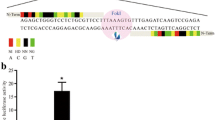
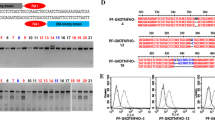
Abstract
Genetically modified pigs are valuable models of human disease and donors of xenotransplanted organs. Conventional gene targeting in pig somatic cells is extremely inefficient. Zinc-finger nuclease (ZFN) technology has been shown to be a powerful tool for efficiently inducing mutations in the genome. However, ZFN-mediated targeting in pigs has rarely been achieved. Here, we used ZFNs to knock out the porcine α-1, 3-galactosyl-transferase (GGTA1) gene, which generates Gal epitopes that trigger hyperacute immune rejection in pig-to-human transplantation. Primary pig fibroblasts were transfected with ZFNs targeting the coding region of GGTA1. Eighteen mono-allelic and four biallelic knockout cell clones were obtained after drug selection with efficiencies of 23.4% and 5.2%, respectively. The biallelic cells were used to produce cloned pigs via somatic cell nuclear transfer (SCNT). Three GGTA1 null piglets were born, and one knockout primary fibroblast cell line was established from a cloned fetus. Gal epitopes on GGTA1 null pig cells were completely eliminated from the cell membrane. Functionally, GGTA1 knockout cells were protected from complement-mediated immune attacks when incubated with human serum. This study demonstrated that ZFN is an efficient tool in creating gene-modified pigs. GGTA1 null pigs and GGTA1 null fetal fibroblasts would benefit research and pig-to-human transplantation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Niemann H, Kues WA. Application of transgenesis in livestock for agriculture and biomedicine. Anim Reprod Sci, 2003, 79: 291–317
Kuwaki K, Tseng YL, Dor FJ, Shimizu A, Houser SL, Sanderson TM, Lancos CJ, Prabharasuth DD, Cheng J, Moran K, Hisashi Y, Mueller N, Yamada K, Greenstein JL, Hawley RJ, Patience C, Awwad M, Fishman JA, Robson SC, Schuurman HJ, Sachs DH, Cooper DK. Heart transplantation in baboons using α1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med, 2005, 11: 29–31
Rogers CS, Stoltz DA, Meyerholz DK, Ostedgaard LS, Rokhlina T, Taft PJ, Rogan MP, Pezzulo AA, Karp PH, Itani OA, Kabel AC, Wohlford-Lenane CL, Davis GJ, Hanfland RA, Smith TL, Samuel M, Wax D, Murphy CN, Rieke A, Whitworth K, Uc A, Starner TD, Brogden KA, Shilyansky J, McCray PB Jr, Zabner J, Prather RS, Welsh MJ. Disruption of the CFTR gene produces a model of cystic fibrosis in newborn pigs. Science, 2008, 321: 1837–1841
Yamada K, Yazawa K, Shimizu A, Iwanaga T, Hisashi Y, Nuhn M, O’Malley P, Nobori S, Vagefi PA, Patience C, Fishman J, Cooper DK, Hawley RJ, Greenstein J, Schuurman HJ, Awwad M, Sykes M, Sachs DH. Marked prolongation of porcine renal xenograft survival in baboons through the use of α1,3-galactosyltransferase gene-knockout donors and the cotransplantation of vascularized thymic tissue. Nat Med, 2005, 11: 32–34
Capecchi MR. Altering the genome by homologous recombination. Science, 1989, 244: 1288–1292
Thomas KR, Capecchi MR. Site-directed mutagenesis by gene targeting in mouse embryo-derived stem cells. Cell, 1987, 51: 503–512
Jin DI, Lee SH, Choi JH, Lee JS, Lee JE, Park KW, Seo JS. Targeting efficiency of α-1,3-galactosyl transferase gene in pig fetal fibroblast cells. Exp Mol Med, 2003, 35: 572–577
Lai L, Kolber-Simonds D, Park KW, Cheong HT, Greenstein JL, Im GS, Samuel M, Bonk A, Rieke A, Day BN, Murphy CN, Carter DB, Hawley RJ, Prather RS. Production of α-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science, 2002, 295: 1089–1092
Hendricks SP, He P, Stults CL, Macher BA. Regulation of the expression of Gal alpha 1-3Gal beta 1-4GlcNAc glycosphingolipids in kidney. J Biol Chem, 1990, 265: 17621–17626
Cooper DK, Koren E, Oriol R. Genetically engineered pigs. Lancet, 1993, 342: 682–683
Galili U. The α-gal epitope (Galα1-3Galβ1-4GlcNAc-R) in xenotran-splantation. Biochimie, 2001, 83: 557–563
Good AH, Cooper DK, Malcolm AJ, Ippolito RM, Koren E, Neethling FA, Ye Y, Zuhdi N, Lamontagne LR. Identification of carbohydrate structures that bind human antiporcine antibodies: implications for discordant xenografting in humans. Transplant Proc, 1992, 24: 559–562
Phelps CJ, Koike C, Vaught TD, Boone J, Wells KD, Chen SH, Ball S, Specht SM, Polejaeva IA, Monahan JA, Jobst PM, Sharma SB, Lamborn AE, Garst AS, Moore M, Demetris AJ, Rudert WA, Bottino R, Bertera S, Trucco M, Starzl TE, Dai Y, Ayares DL. Production of α1,3-galactosyltransferase-deficient pigs. Science, 2003, 299: 411–414
Wu Z, Chen J, Ren J, Bao L, Liao J, Cui C, Rao L, Li H, Gu Y, Dai H, Zhu H, Teng X, Cheng L, Xiao L. Generation of pig induced pluripotent stem cells with a drug-inducible system. J Mol Cell Biol, 2009, 1: 46–54
Ross JW, Whyte JJ, Zhao J, Samuel M, Wells KD, Prather RS. Optimization of square-wave electroporation for transfection of porcine fetal fibroblasts. Transgenic research, 2010, 19: 611–620
Petersen B, Lucas-Hahn A, Oropeza M, Hornen N, Lemme E, Hassel P, Queisser AL, Niemann H. Development and validation of a highly efficient protocol of porcine somatic cloning using preovulatory embryo transfer in peripubertal gilts. Cloning Stem Cells, 2008, 10: 355–362
Onishi A, Iwamoto M, Akita T, Mikawa S, Takeda K, Awata T, Hanada H, Perry AC. Pig cloning by microinjection of fetal fibroblast nuclei. Science, 2000, 289: 1188–1190
Chen G, Sun H, Yang H, Kubelik D, Garcia B, Luo Y, Xiang Y, Qian A, Copeman L, Liu W, Cardella CJ, Wang W, Xiong Y, Wall W, White DJ, Zhong R. The role of anti-non-gal antibodies in the development of acute humoral xenograft rejection of hdaf transgenic porcine kidneys in baboons receiving anti-gal antibody neutralization therapy. Transplantation, 2006, 81: 273–283
Diamond LE, Quinn CM, Martin MJ, Lawson J, Platt JL, Logan JS. A human CD46 transgenic pig model system for the study of discordant xenotransplantation. Transplantation, 2001, 71: 132–142
McCurry KR, Kooyman DL, Alvarado CG, Cotterell AH, Martin MJ, Logan JS, Platt JL. Human complement regulatory proteins protect swine-to-primate cardiac xenografts from humoral injury. Nat Med, 1995, 1: 423–427
McGregor CG, Teotia SS, Byrne GW, Michaels MG, Risdahl JM, Schirmer JM, Tazelaar HD, Walker RC, Logan JS. Cardiac xenotransplantation: progress toward the clinic. Transplantation, 2004, 78: 1569–1575
Fujimura T, Takahagi Y, Shigehisa T, Nagashima H, Miyagawa S, Shirakura R, Murakami H. Production of α1,3-galactosyltransferase gene-deficient pigs by somatic cell nuclear transfer: a novel selection method for gal α1,3-Gal antigen-deficient cells. Mol Reprod Dev, 2008, 75: 1372–1378
Kirkeby S, Moe D. Binding of Griffonia simplicifolia 1 isolectin B4 (GS1 B4) to α-galactose antigens. Immunol Cell Biol, 2001, 79: 121–127
Hauschild J, Petersen B, Santiago Y, Queisser AL, Carnwath JW, Lucas-Hahn A, Zhang L, Meng X, Gregory PD, Schwinzer R, Cost GJ, Niemann H. Efficient generation of a biallelic knockout in pigs using zinc-finger nucleases. Proc Natl Acad Sci USA, 2011, 108: 12013–12017
Li P, Estrada JL, Burlak C, Tector AJ. Biallelic knockout of the α-1,3 galactosyltransferase gene in porcine liver-derived cells using zinc finger nucleases. J Surg Res, 2013, 181: e39–45
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at springerlink.fh-diploma.de
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Bao, L., Chen, H., Jong, U. et al. Generation of GGTA1 biallelic knockout pigs via zinc-finger nucleases and somatic cell nuclear transfer. Sci. China Life Sci. 57, 263–268 (2014). https://doi.org/10.1007/s11427-013-4601-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4601-2