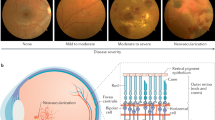
Abstract
Diabetic retinopathy (DR) is one of the most common retinal diseases world-wide. It has a complex pathology that involves the vasculature of the inner retina and breakdown of the blood-retinal barrier. Extensive research has determined that DR is not only a vascular disease but also has a neurodegenerative component and that essentially all types of cells in the retina are affected, leading to chronic loss of visual function. A great deal of work using animal models of DR has established the loss of neurons and pathology of other cell types, including supporting glial cells. There has also been an increased emphasis on measuring retinal function in the models, as well as further validation and extension of the animal studies by clinical and translational research. This article will attempt to summarize the more recent developments in research towards understanding the complexities of retinal neurodegeneration and functional vision loss in DR.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gastinger MJ, Singh RS, Barber AJ. Loss of cholinergic and dopaminergic amacrine cells in streptozotocin-diabetic rat and ins2akita-diabetic mouse retinas. Invest Ophthalmol Vis Sci, 2006, 47: 3143–3150
Gastinger MJ, Kunselman AR, Conboy EE, Bronson SK, Barber AJ. Dendrite remodeling and other abnormalities in the retinal ganglion cells of ins2 akita diabetic mice. Invest Ophthalmol Vis Sci, 2008, 49: 2635–2642
Bloodworth JM Jr., Molitor DL. Ultrastructural aspects of human and canine diabetic retinopathy. Invest Ophthalmol, 1965, 4: 1037–1048
Bloodworth JM Jr. Diabetic microangiopathy. Diabetes, 1963, 12: 99–114
Cunha-Vaz JG. Pathophysiology of diabetic retinopathy. British J Ophthalmol, 1978, 62: 351–355
Cunha-Vaz JG. Studies on the pathophysiology of diabetic retinopathy. The blood-retinal barrier in diabetes. Diabetes, 1983, 32(Suppl 2): 20–27
Bloodworth JM Jr. Diabetic retinopathy. Diabetes, 1962, 11: 1–22
Wolter JR. Diabetic retinopathy. Am J Ophthalmol, 1961, 51: 1123–1139
Mizutani M, Kern TS, Lorenzi M. Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J Clin Invest, 1996, 97: 2883–2890
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest, 1998, 102: 783–791
Kanamori A, Nakamura M, Mukuno H, Maeda H, Negi A. Diabetes has an additive effect on neural apoptosis in rat retina with chronically elevated intraocular pressure. Curr Eye Res, 2004, 28: 47–54
Martin PM, Roon P, Van Ells TK, Ganapathy V, Smith SB. Death of retinal neurons in streptozotocin-induced diabetic mice. Invest Ophthalmol Vis Sci, 2004, 45: 3330–3336
Barber AJ, Antonetti DA, Kern TS, Reiter CE, Soans RS, Krady JK, Levison SW, Gardner TW, Bronson SK. The ins2akita mouse as a model of early retinal complications in diabetes. Invest Ophthalmol Vis Sci, 2005, 46: 2210–2218
Ning X, Baoyu Q, Yuzhen L, Shuli S, Reed E, Li QQ. Neuro-optic cell apoptosis and microangiopathy in kkay mouse retina. Int J Mol Med, 2004, 13: 87–92
Park SH, Park JW, Park SJ, Kim KY, Chung JW, Chun MH, Oh SJ. Apoptotic death of photoreceptors in the streptozotocin-induced diabetic rat retina. Diabetologia, 2003, 46: 1260–1268
Feit-Leichman RA, Kinouchi R, Takeda M, Fan Z, Mohr S, Kern TS, Chen DF. Vascular damage in a mouse model of diabetic retinopathy: relation to neuronal and glial changes. Invest Ophthalmol Vis Sci, 2005, 46: 4281–4287
Barber AJ. A new view of diabetic retinopathy: a neurodegenerative disease of the eye. Prog Neuro-Psychopharmacol Biol Psychiatry, 2003, 27: 283–290
Barber AJ, Gardner TW, Abcouwer SF. The significance of vascular and neural apoptosis to the pathology of diabetic retinopathy. Invest Ophthalmol Vis Sci, 2011, 52: 1156–1163
Barber AJ, Lieth E. Agrin accumulates in the brain microvascular basal lamina during development of the blood-brain barrier. Dev Dyn, 1997, 208: 62–74
Abu-El-Asrar AM, Dralands L, Missotten L, Al-Jadaan IA, Geboes K. Expression of apoptosis markers in the retinas of human subjects with diabetes. Invest Ophthalmol Vis Sci, 2004, 45: 2760–2766
Aung MH, Park HN, Han MK, Obertone TS, Abey J, Aseem F, Thule PM, Iuvone PM, Pardue MT. Dopamine deficiency contributes to early visual dysfunction in a rodent model of type 1 diabetes. J Neurosci, 2014, 34: 726–736
Lopes de Faria JM, Russ H, Costa VP. Retinal nerve fibre layer loss in patients with type 1 diabetes mellitus without retinopathy. British J Ophthalmol, 2002, 86: 725–728
Ozdek S, Lonneville YH, Onol M, Yetkin I, Hasanreisoglu BB. Assessment of nerve fiber layer in diabetic patients with scanning laser polarimetry. Eye (London, England), 2002, 16: 761–765
Takahashi H, Goto T, Shoji T, Tanito M, Park M, Chihara E. Diabetes-associated retinal nerve fiber damage evaluated with scanning laser polarimetry. Am J Ophthalmol, 2006, 142: 88–94
Sugimoto M, Sasoh M, Ido M, Wakitani Y, Takahashi C, Uji Y. Detection of early diabetic change with optical coherence tomography in type 2 diabetes mellitus patients without retinopathy. Ophthalmologica, 2005, 219: 379–385
Biallosterski C, van Velthoven ME, Michels RP, Schlingemann RO, DeVries JH, Verbraak FD. Decreased optical coherence tomographymeasured pericentral retinal thickness in patients with diabetes mellitus type 1 with minimal diabetic retinopathy. British J Ophthalmol, 2007, 91: 1135–1138
Peng PH, Lin HS, Lin S. Nerve fibre layer thinning in patients with preclinical retinopathy. Can J Ophthalmol, 2009, 44: 417–422
van Dijk HW, Kok PHB, Garvin M, Sonka M, Devries JH, Michels RPJ, van Velthoven MEJ, Schlingemann RO, Verbraak FD, Abramoff MD. Selective loss of inner retinal layer thickness in type 1 diabetic patients with minimal diabetic retinopathy. Invest Ophthalmol Vis Sci, 2009, 50: 3404–3409
van Dijk HW, Verbraak FD, Kok PH, Garvin MK, Sonka M, Lee K, Devries JH, Michels RP, van Velthoven ME, Schlingemann RO, Abramoff MD. Decreased retinal ganglion cell layer thickness in patients with type 1 diabetes. Invest Ophthalmol Vis Sci, 2010, 51: 3660–3665
van Dijk HW, Verbraak FD, Kok PH, Stehouwer M, Garvin MK, Sonka M, DeVries JH, Schlingemann RO, Abramoff MD. Early neurodegeneration in the retina of type 2 diabetic patients. Invest Ophthalmol Vis Sci, 2012, 53: 2715–2719
Vujosevic S, Midena E. Retinal layers changes in human preclinical and early clinical diabetic retinopathy support early retinal neuronal and muller cells alterations. J Diabetes Res, 2013, 2013: 905058
Oshitari T, Hanawa K, Adachi-Usami E. Changes of macular and rnfl thicknesses measured by stratus oct in patients with early stage diabetes. Eye (London, England), 2009, 23: 884–889
Browning DJ, Fraser CM, Clark S. The relationship of macular thickness to clinically graded diabetic retinopathy severity in eyes without clinically detected diabetic macular edema. Ophthalmology, 2008, 115: 533–539.e532
Asefzadeh B, Fisch BM, Parenteau CE, Cavallerano AA. Macular thickness and systemic markers for diabetes in individuals with no or mild diabetic retinopathy. Clin Exp Ophthalmol, 2008, 36: 455–463
Araszkiewicz A, Zozulinska-Ziolkiewicz D, Meller M, Bernardczyk-Meller J, Pilacinski S, Rogowicz-Frontczak A, Naskret D, Wierusz-Wysocka B. Neurodegeneration of the retina in type 1 diabetic patients. Pol Arch Med Wewn, 2012, 122: 464–470
Bronson-Castain KW, Bearse MA, Jr., Neuville J, Jonasdottir S, King-Hooper B, Barez S, Schneck ME, Adams AJ. Adolescents with type 2 diabetes: Early indications of focal retinal neuropathy, retinal thinning, and venular dilation. Retina, 2009, 29: 618–626
van Dijk HW, Verbraak FD, Stehouwer M, Kok PH, Garvin MK, Sonka M, DeVries JH, Schlingemann RO, Abramoff MD. Association of visual function and ganglion cell layer thickness in patients with diabetes mellitus type 1 and no or minimal diabetic retinopathy. Vis Res, 2011, 51: 224–228
Frisen L. New, sensitive window on abnormal spatial vision: rarebit probing. Vis Res, 2002, 42: 1931–1939
Zhi Z, Chao JR, Wietecha T, Hudkins KL, Alpers CE, Wang RK. Noninvasive imaging of retinal morphology and microvasculature in obese mice using optical coherence tomography and optical microangiography. Invest Ophthalmol Vis Sci, 2014, 55: 1024–1030
Yang JH, Kwak HW, Kim TG, Han J, Moon SW, Yu SY. Retinal neurodegeneration in type ii diabetic otsuka long-evans tokushima fatty rats. Invest Ophthalmol Vis Sci, 2013, 54: 3844–3851
McLenachan S, Chen X, McMenamin PG, Rakoczy EP. Absence of clinical correlates of diabetic retinopathy in the ins2akita retina. Clin Exp Ophthalmol, 2013, 41: 582–592
Hombrebueno JR, Chen M, Penalva RG, Xu H. Loss of synaptic connectivity, particularly in second order neurons is a key feature of diabetic retinal neuropathy in the ins2akita mouse. PLoS One, 2014, 9: e97970
VanGuilder HD, Brucklacher RM, Patel K, Ellis RW, Freeman WM, Barber AJ. Diabetes downregulates presynaptic proteins and reduces basal synapsin i phosphorylation in rat retina. Eur J Neurosci, 2008, 28: 1–11
Gaspar JM, Baptista FI, Galvao J, Castilho AF, Cunha RA, Ambrosio AF. Diabetes differentially affects the content of exocytotic proteins in hippocampal and retinal nerve terminals. Neuroscience, 2010, 169: 1589–1600
Masser DR, VanGuilder Starkey HD, Bixler GV, Dunton W, Bronson SK, Freeman WM. Insulin treatment normalizes retinal neuroinflammation but not markers of synapse loss in diabetic rats. Exp Eye Res, 2014, 125: 95–106
Kurihara T, Ozawa Y, Nagai N, Shinoda K, Noda K, Imamura Y, Tsubota K, Okano H, Oike Y, Ishida S. Angiotensin II type 1 receptor signaling contributes to synaptophysin degradation and neuronal dysfunction in the diabetic retina. Diabetes, 2008, 57: 2191–2198
Sasaki M, Ozawa Y, Kurihara T, Kubota S, Yuki K, Noda K, Kobayashi S, Ishida S, Tsubota K. Neurodegenerative influence of oxidative stress in the retina of a murine model of diabetes. Diabetologia, 2010, 53: 971–979
D’Cruz TS, Weibley BN, Kimball SR, Barber AJ. Post-translational processing of synaptophysin in the rat retina is disrupted by diabetes. PLoS One, 2012, 7: e44711
Qin Y, Xu G, Wang W. Dendritic abnormalities in retinal ganglion cells of three-month diabetic rats. Curr Eye Res, 2006, 31: 967–974
Meyer-Rusenberg B, Pavlidis M, Stupp T, Thanos S, Meyer-Rusenberg B, Pavlidis M, Stupp T, Thanos S. Pathological changes in human retinal ganglion cells associated with diabetic and hypertensive retinopathy. Graefes Arch Clin Exp Ophthalmol, 2007, 245: 1009–1018
Simonsen SE. Prognostic value of erg (oscillatory potential) in juvenile diabetics. Acta Ophthalmol Suppl, 1974, 123: 223–224
Frost-Larsen K, Larsen HW, Simonsen SE. Oscillatory potential and nyctometry in insulin-dependent diabetics. Acta Ophthalmol (Copenh), 1980, 58: 879–888
Simonsen SE. The value of the oscillatory potential in selecting juvenile diabetics at risk of developing proliferative retinopathy. Acta Ophthalmol (Copenh), 1980, 58: 865–878
Li Q, Zemel E, Miller B, Perlman I. Early retinal damage in experimental diabetes: electroretinographical and morphological observations. Exp Eye Res, 2002, 74: 615–625
Shinoda K, Rejdak R, Schuettauf F, Blatsios G, Volker M, Tanimoto N, Olcay T, Gekeler F, Lehaci C, Naskar R, Zagorski Z, Zrenner E. Early electroretinographic features of streptozotocin-induced diabetic retinopathy. Clin Exp Ophthalmol, 2007, 35: 847–854
Kohzaki K, Vingrys AJ, Bui BV, Kohzaki K, Vingrys AJ, Bui BV. Early inner retinal dysfunction in streptozotocin-induced diabetic rats. Invest Ophthalmol Vis Sci, 2008, 49: 3595–3604
Hancock HA, Kraft TW. Oscillatory potential analysis and ergs of normal and diabetic rats. Invest Ophthalmol Vis Sci, 2004, 45: 1002–1008
Aung MH, Kim MK, Olson DE, Thule PM, Pardue MT. Early visual deficits in streptozotocin-induced diabetic long evans rats. Invest Ophthalmol Vis Sci, 2013, 54: 1370–1377
Bush RA, Hawks KW, Sieving PA. Preservation of inner retinal responses in the aged royal college of surgeons rat. Evidence against glutamate excitotoxicity in photoreceptor degeneration. Invest Ophthalmol Vis Sci, 1995, 36: 2054–2062
Sugawara T, Sieving PA, Iuvone PM, Bush RA. The melatonin antagonist luzindole protects retinal photoreceptors from light damage in the rat. Invest Ophthalmol Vis Sci, 1998, 39: 2458–2465
Bui BV, Fortune B. Ganglion cell contributions to the rat full-field electroretinogram. J Physiol, 2004, 555: 153–173
Abraham FA, Haimovitz J, Berezin M. The photopic and scotopic visual thresholds in diabetics without diabetic retinopathy. Metab Pediatr Syst Ophthalmol, 1988, 11: 76–77
Aylward GW, Billson FA. The scotopic threshold response in diabetic retinopathy—a preliminary report. Aus New Zealand J Ophthalmol, 1989, 17: 369–372
Nasralah Z, Robinson W, Jackson G, Barber A. Measuring visual function in diabetic retinopathy: progress in basic and clinical research. J Clin Exp Ophthalmol, 2013, 4: 1000306
Ha Y, Saul A, Tawfik A, Zorrilla EP, Ganapathy V, Smith SB. Diabetes accelerates retinal ganglion cell dysfunction in mice lacking sigma receptor 1. Mol Vis, 2012, 18: 2860–2870
Bui BV, Loeliger M, Thomas M, Vingrys AJ, Rees SM, Nguyen CTO, He Z, Tolcos M. Investigating structural and biochemical correlates of ganglion cell dysfunction in streptozotocin-induced diabetic rats. Exp Eye Res, 2009, 88: 1076–1083
Prusky GT, Alam NM, Beekman S, Douglas RM. Rapid quantification of adult and developing mouse spatial vision using a virtual optomotor system. Invest Ophthalmol Vis Sci, 2004, 45: 4611–4616
Kirwin SJ, Kanaly ST, Hansen CR, Cairns BJ, Ren M, Edelman JL. Retinal gene expression and visually evoked behavior in diabetic long evans rats. Invest Ophthalmol Vis Sci, 2011, 52: 7654–7663
Akimov NP, Renteria RC. Spatial frequency threshold and contrast sensitivity of an optomotor behavior are impaired in the ins2akita mouse model of diabetes. Behav Brain Res, 2012, 226: 601–605
Umino Y, Solessio E. Loss of scotopic contrast sensitivity in the optomotor response of diabetic mice. Invest Ophthalmol Vis Sci, 2013, 54: 1536–1543
Muir ER, Renteria RC, Duong TQ. Reduced ocular blood flow as an early indicator of diabetic retinopathy in a mouse model of diabetes. Invest Ophthalmol Vis Sci, 2012, 53: 6488–6494
Lee CA, Li G, Patel MD, Petrash JM, Benetz BA, Veenstra A, Amengual J, von Lintig J, Burant CJ, Tang J, Kern TS. Diabetesinduced impairment in visual function in mice: Contributions of p38 mapk, rage, leukocytes, and aldose reductase. Invest Ophthalmol Vis Sci, 2014, 55: 2904–2910
Xu Z, Wei Y, Gong J, Cho H, Park JK, Sung ER, Huang H, Wu L, Eberhart C, Handa JT, Du Y, Kern TS, Thimmulappa R, Barber AJ, Biswal S, Duh EJ. Nrf2 plays a protective role in diabetic retinopathy in mice. Diabetologia, 2014, 57: 204–213
Gardner TW, Antonetti DA, Barber AJ, LaNoue KF, Levison SW. Diabetic retinopathy: more than meets the eye. Survey Ophthalmol, 2002, 47: S253–262
Antonetti DA, Barber AJ, Khin S, Lieth E, Tarbell JM, Gardner TW. Vascular permeability in experimental diabetes is associated with reduced endothelial occludin content: vascular endothelial growth factor decreases occludin in retinal endothelial cells. Diabetes, 1998, 47: 1953–1959
Murakami T, Felinski EA, Antonetti DA. Occludin phosphorylation and ubiquitination regulate tight junction trafficking and vascular endothelial growth factor-induced permeability. J Biol Chem, 2009, 284: 21036–21046
Abcouwer SF, Gardner TW. Diabetic retinopathy: loss of neuroretinal adaptation to the diabetic metabolic environment. Ann New York Acad Sci, 2014, 1311: 174–190
Lott ME, Slocomb JE, Shivkumar V, Smith B, Gabbay RA, Quillen D, Gardner TW, Bettermann K. Comparison of retinal vasodilator and constrictor responses in type 2 diabetes. Acta Ophthalmol (Oxf), 2012, 90: e434–441
Lott ME, Slocomb JE, Shivkumar V, Smith B, Quillen D, Gabbay RA, Gardner TW, Bettermann K. Impaired retinal vasodilator responses in prediabetes and type 2 diabetes. Acta Ophthalmol (Oxf), 2013, 91: e462–469
Ott C, Raff U, Schmidt S, Kistner I, Friedrich S, Bramlage P, Harazny JM, Schmieder RE. Effects of saxagliptin on early microvascular changes in patients with type 2 diabetes. Cardiovasc Diabetol, 2014, 13: 19
Lasta M, Pemp B, Schmidl D, Boltz A, Kaya S, Palkovits S, Werkmeister R, Howorka K, Popa-Cherecheanu A, Garhofer G, Schmetterer L. Neurovascular dysfunction precedes neural dysfunction in the retina of patients with type 1 diabetes. Invest Ophthalmol Vis Sci, 2013, 54: 842–847
Bearse MA, Jr., Adams AJ, Han Y, Schneck ME, Ng J, Bronson-Castain K, Barez S. A multifocal electroretinogram model predicting the development of diabetic retinopathy. Prog Retinal Eye Res, 2006, 25: 425–448
Ng JS, Bearse MA, Jr., Schneck ME, Barez S, Adams AJ. Local diabetic retinopathy prediction by multifocal erg delays over 3 years. Invest Ophthalmol Vis Sci, 2008, 49: 1622–1628
Cunha-Vaz J, Ribeiro L, Lobo C. Phenotypes and biomarkers of diabetic retinopathy. Prog Retinal Eye Res, 2014, 41: 90–111
Jackson GR, Scott IU, Quillen DA, Walter LE, Gardner TW. Inner retinal visual dysfunction is a sensitive marker of non-proliferative diabetic retinopathy. British J Ophthalmol, 2012, 96: 699–703
Holfort SK, Jackson GR, Larsen M. Dark adaptation during transient hyperglycemia in type 2 diabetes. Exp Eye Res, 2010, 91: 710–714
Jackson GR, Barber AJ. Visual dysfunction associated with diabetic retinopathy. Curr Diabetes Rep, 2010, 10: 380–384
Somfai GM, Tatrai E, Laurik L, Varga B, Olvedy V, Jiang H, Wang J, Smiddy WE, Somogyi A, DeBuc DC. Automated classifiers for early detection and diagnosis of retinopathy in diabetic eyes. BMC Bioinformatics, 2014, 15: 106
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at springerlink.fh-diploma.de
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Barber, A.J. Diabetic retinopathy: recent advances towards understanding neurodegeneration and vision loss. Sci. China Life Sci. 58, 541–549 (2015). https://doi.org/10.1007/s11427-015-4856-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4856-x