Abstract
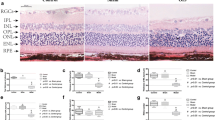
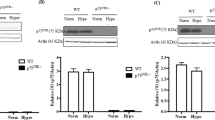
Ischemia occurs in diabetic retinopathy with neuronal loss, edema, glial cell reactivity and oxidative stress. Epacs, consisting of Epac1 and Epac2, are cAMP mediators playing important roles in maintenance of endothelial barrier and neuronal functions. To investigate the roles of Epacs in the pathogenesis of ischemic retinopathy, transient middle cerebral artery occlusion (tMCAO) was performed on Epac1-deficient (Epac1 -/-) mice, Epac2-deficient (Epac2 -/-) mice, and their wild type counterparts (Epac1 +/+ and Epac2 +/+). Two-hour occlusion and 22-hour reperfusion were conducted to induce ischemia/reperfusion injury to the retina. After tMCAO, the contralateral retinae displayed similar morphology between different genotypes. Neuronal loss, retinal edema and increase in immunoreactivity for aquaporin 4 (AQP4), glial fibrillary acidic protein (GFAP), peroxiredoxin 6 (Prx6) were observed in ipsilateral retinae. Epac2 -/- ipsilateral retinae showed more neuronal loss in retinal ganglion cell layer, increased retinal thickness and stronger immunostaining of AQP4, GFAP, and Prx6 than those of Epac2 +/+. However, Epac1 -/- ipsilateral retinae displayed similar pathology as those in Epac1 +/+ mice. Our observations suggest that Epac2-deficiency led to more severe ischemic retinopathy after retinal ischemia/reperfusion injury.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Osborne NN, Casson RJ, Wood JPM, Chidlow G, Graham M, Melena J. Retinal ischemia: mechanisms of damage and potential therapeutic strategies. Prog Retin Eye Res, 2004, 23: 91–147
Buchi ER. Cell death in the rat retina after a pressure-induced ischaemia-reperfusion insult: an electron microscopic study. I. Ganglion cell layer and inner nuclear layer. Exp Eye Res, 1992, 55: 605–613
Lafuente MP, Villegas-Pérez MP, Sellés-Navarro I, Mayor-Torroglosa S, de Imperial JM, Vidal-Sanz M. Retinal ganglion cell death after acute retinal ischemia is an ongoing process whose severity and duration depends on the duration of the insult. Neuroscience, 2002, 109: 157–168
Nucci C, Tartaglione R, Rombolà L, Morrone LA, Fazzi E, Bagetta G. Neurochemical evidence to implicate elevated glutamate in the mechanisms of high intraocular pressure (IOP)-induced retinal ganglion cell death in rat. Neurotoxicology, 2005, 26: 935–941
Yamamoto H, Schmidt-Kastner R, Hamasaki DI, Yamamoto H, Parel JM. Complex neurodegeneration in retina following moderate ischemia induced by bilateral common carotid artery occlusion in Wistar rats. Exp Eye Res, 2006, 82: 767–779
Bringmann A, Francke M, Pannicke T, Biedermann B, Faude F, Enzmann V, Wiedemann P, Reichelt W, Reichenbach A. Human Muller glial cells: altered potassium channel activity in proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci, 1999, 40: 3316–3323
Kim IB, Kim KY, Joo CK, Lee MY, Oh SJ, Chung JW, Chun MH. Reaction of Muller cells after increased intraocular pressure in the rat retina. Exp Brain Res, 1998, 121: 419–424
Da T, Verkman AS. Aquaporin-4 gene disruption in mice protects against impaired retinal function and cell death after ischemia. Invest Ophthalmol Vis Sci, 2004, 45: 4477–4483
Yanoff M, Fine BS, Brucker AJ, Eagle Jr. Ralph C. Pathology of human cystoid macular edema. Surv Ophthalmol, 1984, 28(Suppl): 505–511
Cheung AK, Fung MK, Lo AC, Lam TT, So KF, Chung SS, Chung SK. Aldose reductase deficiency prevents diabetes-induced bloodretinal barrier breakdown, apoptosis, and glial reactivation in the retina of db/db mice. Diabetes, 2005, 54: 3119–3125
Cheung AK, Lo AC, So KF, Chung SS, Chung SK. Gene deletion and pharmacological inhibition of aldose reductase protect against retinal ischemic injury. Exp Eye Res, 2007, 85: 608–616
Gloerich M, Bos JL. Epac: defining a new mechanism for cAMP action. Annu Rev Pharmacol Toxicol, 2010, 50: 355–375
Bos JL. Linking Rap to cell adhesion. Curr Opin Cell Biol, 2005, 17: 123–128
Rampersad SN, Ovens JD, Huston E, Umana MB, Wilson LS, Netherton SJ, Lynch MJ, Baillie GS, Houslay MD, Maurice DH. Cyclic AMP phosphodiesterase 4D (PDE4D) Tethers EPAC1 in a vascular endothelial cadherin (VE-Cad)-based signaling complex and controls cAMP-mediated vascular permeability. J Biol Chem, 2010, 285: 33614–33622
Nijholt IM, Dolga AM, Ostroveanu A, Luiten PG, Schmidt M, Eisel UL. Neuronal AKAP150 coordinates PKA and Epac-mediated PKB/Akt phosphorylation. Cell Signal, 2008, 20: 1715–1724
Penzes P, Woolfrey KM, Srivastava DP. Epac2-mediated dendritic spine remodeling: implications for disease. Mol Cell Neurosci, 2011, 46: 368–380
Woolfrey KM, Srivastava DP, Photowala H, Yamashita M, Barbolina MV, Cahill ME, Xie Z, Jones KA, Quilliam LA, Prakriya M, Penzes P. Epac2 induces synapse remodeling and depression and its disease-associated forms alter spines. Nat Neurosci, 2009, 12: 1275–1284
Kawasaki H, Springett GM, Mochizuki N, Toki S, Nakaya M, Matsuda M, Housman DE, Graybiel AM. A family of cAMP-binding proteins that directly activate Rap1. Science, 1998, 282: 2275–2279
Whitaker CM, Cooper NGF. Differential distribution of exchange proteins directly activated by cyclic AMP within the adult rat retina. Neuroscience, 2010, 165: 955–967
Kai AK, Lam AK, Chen Y, Tai AC, Zhang X, Lai AK, Yeung PK, Tam S, Wang J, Lam KS, Vanhoutte PM, Bos JL, Chung SS, Xu A, Chung SK. Exchange protein activated by cAMP 1 (Epac1)-deficient mice develop beta-cell dysfunction and metabolic syndrome. FASEB J, 2013, 27: 4122–4135
Zhang CL, Katoh M, Shibasaki T, Minami K, Sunaga Y, Takahashi H, Yokoi N, Iwasaki M, Miki T, Seino S. The cAMP sensor Epac2 is a direct target of antidiabetic sulfonylurea drugs. Science, 2009, 325: 607–610
Cheung SS, Leung JW, Lam AK, Lam KS, Chung SS, Lo AC, Chung SK. Selective over-expression of endothelin-1 in endothelial cells exacerbates inner retinal edema and neuronal death in ischemic retina. PLoS One, 2011, 6: e26184
Li SY, Fu ZJ, Ma H, Jang WC, So KF, Wong D, Lo AC. Effect of lutein on retinal neurons and oxidative stress in a model of acute retinal ischemia/reperfusion. Invest Ophthalmol Vis Sci, 2009, 50: 836–843
Fu ZJ, Li SY, Kociok N, Wong D, Chung SK, Lo AC. Aldose reductase deficiency reduced vascular changes in neonatal mouse retina in oxygen-induced retinopathy. Invest Ophthalmol Vis Sci, 2012, 53: 5698–5712
Li SY, Yang D, Yeung CM, Yu WY, Chang RC, So KF, Wong D, Lo AC. Lycium barbarum polysaccharides reduce neuronal damage, blood-retinal barrier disruption and oxidative stress in retinal ischemia/reperfusion injury. PLoS One, 2011, 6: e16380
Shibasaki T, Takahashi H, Miki T, Sunaga Y, Matsumura K, Yamanaka M, Zhang C, Tamamoto A, Satoh T, Miyazaki J, Seino S. Essential role of Epac2/Rap1 signaling in regulation of insulin granule dynamics by cAMP. Proc Natl Acad Sci USA, 2007, 104: 19333–19338
Pannicke T, Uckermann O, Iandiev I, Biedermann B, Wiedemann P, Perlman I, Reichenbach A, Bringmann A. Altered membrane physiology in Muller glial cells after transient ischemia of the rat retina. Glia, 2005, 50: 1–11
Tso MO. Pathology of cystoid macular edema. Ophthalmology, 1982, 89: 902–915
Lo AC, Chen AY, Hung VK, Yaw LP, Fung MK, Ho MC, Tsang MC, Chung SS, Chung SK. Endothelin-1 overexpression leads to further water accumulation and brain edema after middle cerebral artery occlusion via aquaporin 4 expression in astrocytic end-feet. J Cereb Blood Flow Metab, 2005, 25: 998–1011
Avellini C, Baccarani U, Trevisan G, Cesaratto L, Vascotto C, D’Aurizio F, Pandolfi M, Adani GL, Tell G. Redox proteomics and immunohistology to study molecular events during ischemiareperfusion in human liver. Transplant Proc, 2007, 39: 1755–1760
Eismann T, Huber N, Shin T, Kuboki S, Galloway E, Wyder M, Edwards MJ, Greis KD, Shertzer HG, Fisher AB, Lentsch AB. Peroxiredoxin-6 protects against mitochondrial dysfunction and liver injury during ischemia-reperfusion in mice. Am J Physiol Gastrointest Liver Physiol, 2009, 296: G266–274
Nagy N, Malik G, Fisher AB, Das DK. Targeted disruption of peroxiredoxin 6 gene renders the heart vulnerable to ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol, 2006, 291: H2636–2640
Zhang X, Yeung PK, McAlonan GM, Chung SS, Chung SK. Transgenic mice over-expressing endothelial endothelin-1 show cognitive deficit with blood-brain barrier breakdown after transient ischemia with long-term reperfusion. Neurobiol Learn Mem, 2013, 101: 46–54
Ouyang M, Zhang L, Zhu JJ, Schwede F, Thomas SA. Epac signaling is required for hippocampus-dependent memory retrieval. Proc Natl Acad Sci USA, 2008, 105: 11993–11997
Dwivedi Y, Mondal AC, Rizavi HS, Faludi G, Palkovits M, Sarosi A, Conley RR, Pandey GN. Differential and brain region-specific regulation of Rap-1 and Epac in depressed suicide victims. Arch Gen Psychiatry, 2006, 63: 639–648
Gekel I, Neher E. Application of an Epac activator enhances neurotransmitter release at excitatory central synapses. J Neurosci, 2008, 28: 7991–8002
Roscioni SS, Elzinga CR, Schmidt M. Epac: effectors and biological functions. Naunyn Schmiedebergs Arch Pharmacol, 2008, 377: 345–357
Suzuki S, Yokoyama U, Abe T, Kiyonari H, Yamashita N, Kato Y, Kurotani R, Sato M, Okumura S, Ishikawa Y. Differential roles of Epac in regulating cell death in neuronal and myocardial cells. J Biol Chem, 2010, 285: 24248–24259
Wittchen ES, Hartnett ME. The small GTPase Rap1 is a novel regulator of RPE cell barrier function. Invest Ophthalmol Vis Sci, 2011, 52: 7455–7463
Kolb H. The architecture of functional neural circuits in the vertebrate retina. The Proctor Lecture. Invest Ophthalmol Vis Sci, 1994, 35: 2385–2404
Yang Y, Shu X, Liu D, Shang Y, Wu Y, Pei L, Xu X, Tian Q, Zhang J, Qian K, Wang YX, Petralia RS, Tu W, Zhu LQ, Wang JZ, Lu Y. EPAC null mutation impairs learning and social interactions via aberrant regulation of miR-124 and Zif268 translation. Neuron, 2012, 73: 774–788
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at springerlink.fh-diploma.de
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Liu, J., Yeung, P.K.K., Cheng, L. et al. Epac2-deficiency leads to more severe retinal swelling, glial reactivity and oxidative stress in transient middle cerebral artery occlusion induced ischemic retinopathy. Sci. China Life Sci. 58, 521–530 (2015). https://doi.org/10.1007/s11427-015-4860-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4860-1