Abstract
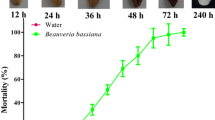
Monochamus alternatus, the main vector beetles of invasive pinewood nematode, has established a symbiotic relationship with a native ectotrophic fungal symbiont, Sporothrix sp. 1, in China. The immune response of M. alternatus to S. sp. 1 in the coexistence of beetles and fungi is, however, unknown. Here, we report that immune responses of M. alternatus pupae to infection caused by ectotrophic symbiotic fungus S. sp. 1 and entomopathogenic fungus Beauveria bassiana differ significantly. The S. sp. 1 did not kill the beetles while B. bassiana killed all upon injection. The transcriptome results showed that the numbers of differentially expressed genes in M. alternatus infected with S. sp. 1 were 2-fold less than those infected with B. bassiana at 48 hours post infection. It was noticed that Toll and IMD pathways played a leading role in the beetle’s immune system when infected by symbiotic fungus, but upon infection by entomopathogenic fungus, only the Toll pathway gets triggered actively. Furthermore, the beetles could tolerate the infection of symbiotic fungi by retracing their Toll and IMD pathways at 48 h. This study provided a comprehensive sequence resource of M. alternatus transcriptome for further study of the immune interactions between host and associated fungi.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Agaisse, H., Petersen, U.M., Boutros, M., Mathey-Prevot, B., and Perrimon, N. (2003). Signaling role of hemocytes in Drosophila JAK/STAT-dependent response to septic injury. Dev Cell 5, 441–450.
Alves, M., Pereira, A., Matos, P., Henriques, J., Vicente, C., Aikawa, T., Hasegawa, K., Nascimento, F., Mota, M., Correia, A., and Henriques, I. (2016). Bacterial community associated to the pine wilt disease insect vectors Monochamus galloprovincialis and Monochamus alternatus. Sci Rep 6, 23908.
Ashburner, M., Ball, C.A., Blake, J.A., Botstein, D., Butler, H., Cherry, J.M., Davis, A.P., Dolinski, K., Dwight, S.S., Eppig, J.T., Harris, M.A., Hill, D.P., Issel-Tarver, L., Kasarskis, A., Lewis, S., Matese, J.C., Richardson, J.E., Ringwald, M., Rubin, G.M., and Sherlock, G. (2000). Gene Ontology: tool for the unification of biology. Nat Genet 25, 25–29.
Bäckhed, F., Ley, R.E., Sonnenburg, J.L., Peterson, D.A., and Gordon, J.I. (2005). Host-bacterial mutualism in the human intestine. Science 307, 1915–1920.
Baeg, G.H., Zhou, R., and Perrimon, N. (2005). Genome-wide RNAi analysis of JAK/STAT signaling components in Drosophila. Genes Dev 19, 1861–1870.
Currie, C.R., Wong, B., Stuart, A.E., Schultz, T.R., Rehner, S.A., Mueller, U.G., Sung, G.H., Spatafora, J.W., and Straus, N.A. (2003). Ancient tripartite coevolution in the attine ant-microbe symbiosis. Science 299, 386–388.
De Gregorio, E., Spellman, P.T., Tzou, P., Rubin, G.M., and Lemaitre, B. (2002). The Toll and Imd pathways are the major regulators of the immune response in Drosophila. EMBO J 21, 2568–2579.
Dori-Bachash, M., Avrahami-Moyal, L., Protasov, A., Mendel, Z., and Freeman, S. (2015). The occurrence and pathogenicity of Geosmithia spp. and common blue-stain fungi associated with pine bark beetles in planted forests in Israel. Eur J Plant Pathol 143, 627–639.
Ferron, P. (1978). Biological control of insect pests by entomogenous fungi. Annu Rev Entomol 23, 409–442.
Garver, L.S., de Almeida Oliveira, G., and Barillas-Mury, C. (2013). The JNK pathway is a key mediator of Anopheles gambiae antiplasmodial immunity. PLoS Pathog 9, e1003622.
Gillespie, J.P., Kanost, M.R., and Trenczek, T. (1997). Biological mediators of insect immunity. Annu Rev Entomol 42, 611–643.
Gottar, M., Gobert, V., Matskevich, A.A., Reichhart, J.M., Wang, C., Butt, T.M., Belvin, M., Hoffmann, J.A., and Ferrandon, D. (2006). Dual detection of fungal infections in Drosophila via recognition of glucans and sensing of virulence factors. Cell 127, 1425–1437.
Hartley, S.E., and Gange, A.C. (2009). Impacts of plant symbiotic fungi on insect herbivores: mutualism in a multitrophic context. Annu Rev Entomol 54, 323–342.
Hou, Y., Wang, X.L., Saha, T.T., Roy, S., Zhao, B., Raikhel, A.S., and Zou, Z. (2015). Temporal coordination of carbohydrate metabolism during mosquito reproduction. PLoS Genet 11, e1005309.
Lemaitre, B. (2004). The road to Toll. In: Nature Reviews Immunology. (New York: Nature Publishing), pp. 521–527.
Lemaitre, B., Nicolas, E., Michaut, L., Reichhart, J.M., and Hoffmann, J.A. (1996). The dorsoventral regulatory gene cassette spätzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86, 973–983.
Lemaitre, B., Reichhart, J.M., and Hoffmann, J.A. (1997). Drosophila host defense: differential induction of antimicrobial peptide genes after infection by various classes of microorganisms. Proc Natl Acad Sci USA 94, 14614–14619.
Lhocine, N., Ribeiro, P.S., Buchon, N., Wepf, A., Wilson, R., Tenev, T., Lemaitre, B., Gstaiger, M., Meier, P., and Leulier, F. (2008). PIMS modulates immune tolerance by negatively regulating Drosophila innate immune signaling. Cell Host Microbe 4, 147–158.
Lowenberger, C. (2001). Innate immune response of Aedes aegypti. Insect Biochem Mol Biol 31, 219–229.
Lu, M., Hulcr, J., and Sun, J. (2016). The role of symbiotic microbes in insect invasions. Annu Rev Ecol Evol Syst 47, 487–505.
Müller, C.A., Autenrieth, I.B., and Peschel, A. (2005). Intestinal epithelial barrier and mucosal immunity. CMLS Cell Mol Life Sci 62, 1297–1307.
Macdonald, T.T., and Monteleone, G. (2005). Immunity, inflammation, and allergy in the gut. Science 307, 1920–1925.
Maehara, N., and Futai, K. (2000). Population changes of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae), on fungi growing in pine-branch segments. Appl Entomol Zool 35, 413–417.
Mamiya, Y., and Enda, N. (1972). Transmission of Bursaphelenchus Lignicolus (Nematoda: Aphelenchoididae) by Monochamus Alternatus (Coleoptera: Cerambycidae). Nematologica 18, 159–162.
Meng, Q., Yu, H.Y., Zhang, H., Zhu, W., Wang, M.L., Zhang, J.H., Zhou, G.L., Li, X., Qin, Q.L., Hu, S.N., and Zou, Z. (2015). Transcriptomic insight into the immune defenses in the ghost moth, Hepialus xiaojinensis, during an Ophiocordyceps sinensis fungal infection. Insect Biochem Mol Biol 64, 1–15.
Muta, T., and Iwanaga, S. (1996). The role of hemolymph coagulation in innate immunity. Curr Opin Immunol 8, 41–47.
Park, J.M., Brady, H., Ruocco, M.G., Sun, H., Williams, D.A., Lee, S.J., Kato, T., Richards, N., Chan, K., Mercurio, F., Karin, M., and Wasserman, S.A. (2004). Targeting of TAK1 by the NF- B protein Relish regulates the JNK-mediated immune response in Drosophila. Genes Dev 18, 584–594.
Rämet, M., Lanot, R., Zachary, D., and Manfruelli, P. (2002). JNK signaling pathway is required for efficient wound healing in Drosophila. Dev Biol 241, 145–156.
Repe, A., Kirisits, T., Piškur, B., de Groot, M., Kump, B., and Jurc, M. (2013). Ophiostomatoid fungi associated with three spruce-infesting bark beetles in Slovenia. Ann For Sci 70, 717–727.
Schmidt, O., Theopold, U., and Strand, M. (2001). Innate immunity and its evasion and suppression by hymenopteran endoparasitoids. Bioessays 23, 344–351.
Silverman, N., and Maniatis, T. (2001). NF-kappaB signaling pathways in mammalian and insect innate immunity. Genes Dev 15, 2321–2342.
Strand, M.R., and Pech, L.L. (1995). Immunological basis for compatibility in parasitoid-host relationships. Annu Rev Entomol 40, 31–56.
Suh, D.Y., Hyun, M.W., Kim, J.J., Son, S.Y., and Kim, S.H. (2013). Ophiostoma ips from pinewood nematode vector, Japanese pine sawyer beetle (Monochamus alternatus), in Korea. Mycobiology 41, 59–62.
Tu, Q., Cameron, R.A., Worley, K.C., Gibbs, R.A., and Davidson, E.H. (2012). Gene structure in the sea urchin Strongylocentrotus purpuratus based on transcriptome analysis. Genome Res 22, 2079–2087.
Valanne, S., Wang, J.H., and Rämet, M. (2011). The Drosophila toll signaling pathway. J Immunol 186, 649–656.
Vandepitte, K., de Meyer, T., Helsen, K., van Acker, K., Roldán-Ruiz, I., Mergeay, J., and Honnay, O. (2014). Rapid genetic adaptation precedes the spread of an exotic plant species. Mol Ecol 23, 2157–2164.
Vicente, C., Espada, M., Vieira, P., and Mota, M. (2012). Pine Wilt Disease: a threat to European forestry. Eur J Plant Pathol in press doi: 10.1007/s10658-012-9979-3.
Wang, L., Feng, Z., Wang, X., Wang, X., and Zhang, X. (2010). DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26, 136–138.
Wang, Y.H., Hu, Y., Xing, L.S., Jiang, H., Hu, S.N., Raikhel, A.S., and Zou, Z. (2015). A critical role for CLSP2 in the modulation of antifungal immune response in mosquitoes. PLoS Pathog 11, e1004931.
Wraight, S.P., Carruthers, R.I., Jaronski, S.T., Bradley, C.A., Garza, C.J., and Galaini-Wraight, S. (2000). Evaluation of the entomopathogenic fungi Beauveria bassiana and Paecilomyces fumosoroseus for microbial control of the silverleaf whitefly, Bemisia argentifolii. Biol Control 17, 203–217.
Xiong, G.H., Xing, L.S., Lin, Z., Saha, T.T., Wang, C., Jiang, H., and Zou, Z. (2015). High throughput profiling of the cotton bollworm Helicoverpa armigera immunotranscriptome during the fungal and bacterial infections. BMC Genomics 16, 321.
Xu, L., Lou, Q., Cheng, C., Lu, M., and Sun, J. (2015). Gut-associated bacteria of Dendroctonus valens and their involvement in verbenone production. Microb Ecol 70, 1012–1023.
Yassine, H., Kamareddine, L., and Osta, M.A. (2012). The mosquito melanization response is implicated in defense against the entomopathogenic fungus Beauveria bassiana. PLoS Pathog 8, e1003029.
Zhao, L., Mota, M., Vieira, P., Butcher, R.A., and Sun, J. (2014). Interspecific communication between pinewood nematode, its insect vector, and associated microbes. Trends Parasitol 30, 299–308.
Zhao, L., Zhang, S., Wei, W., Hao, H., Zhang, B., Butcher, R.A., and Sun, J. (2013). Chemical signals synchronize the life cycles of a plant-parasitic nematode and its vector beetle. Curr Biol 23, 2038–2043.
Zou, Z., Evans, J.D., Lu, Z., Zhao, P., Williams, M., Sumathipala, N., Hetru, C., Hultmark, D., and Jiang, H. (2007). Comparative genomic analysis of the Tribolium immune system. Genome Biol 8, R177.
Acknowledgments
This work was supported by the Forestry Industry Research Special Funds for Public Welfare Project (201204501), National Key Plan for Scientific Research and Development of China (2016YFC1200604, 2016YFD0500300), the High Technology Research and Development Program (HTRDP) of China (2014AA020529), National Natural Science Foundation of China (31572272, 31370650, 31402013, 31221091, 31672291, L1524009), the CAS Key Research Projects of the Frontier Science (QYZDBSSW- SMC014), and CAS (2015-SM-C-02).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting Information
Table S1 FPKM values of DETs in intracellular signaling pathways of fungal infected M. alternatus
Table S2 Primers used in quantitative real-time PCR analysis
The supporting information is available online at http://life.scichina.com and https://springerlink.fh-diploma.de. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, W., Meng, J., Ning, J. et al. Differential immune responses of Monochamus alternatus against symbiotic and entomopathogenic fungi. Sci. China Life Sci. 60, 902–910 (2017). https://doi.org/10.1007/s11427-017-9102-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-017-9102-y