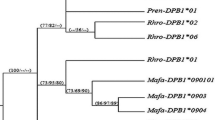
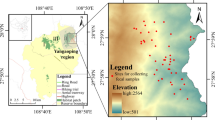
Abstract
Genetic variation is generally believed to be important in studying endangered species’ adaptive potential. Early studies assessed genetic diversity using nearly neutral markers, such as microsatellite loci and mitochondrial DNA (mtDNA), which are very informative for phylogenetic and phylogeographic reconstructions. However, the variation at these loci cannot provide direct information on selective processes involving the interaction of individuals with their environment, or on the capability to resist continuously evolving pathogens and parasites. The importance of genetic diversity at informative adaptive markers, such as major histocompatibility complex (MHC) genes, is increasingly being realized, especially in endangered, isolated species. Small population size and isolation make the golden snub-nosed monkey (Rhinopithecus roxellana) particularly susceptible to genetic variation losses through inbreeding and restricted gene flow. In this study, we compared the genetic variation and population structure of microsatellites, mtDNA, and the most relevant adaptive region of the MHC II-DRB genes in the golden snub-nosed monkey. We examined three Chinese R. roxellana populations and found the same variation patterns in all gene regions, with the population from Shennongjia population, Hubei Province, showing the lowest polymorphism among three populations. Genetic drift that outweighed balancing selection and the founder effect in these populations may explain the similar genetic variation pattern found in these neutral and adaptive genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altizer S, Harvell D, Friedle E. Rapid evolutionary dynamics and disease threats to biodiversity. Trends Ecol Evol, 2003, 18: 589–596
Wahlberg N, Moilanen A, Hanski I. Predicting the occurrence of endangered species in fragmented landscapes. Science, 1996, 273: 1536–1538
Meffe G K, Carroll C G, contributors: Principles of Conservation Biology. 2nd ed. Massachusetts: Sinauer Associates, 1997
Peacock M M, Smith A T. The effects of habitat fragmentation on dispersal patterns, mating behavior and genetic variation in a pica (Ochotona princeps) metapopulation. Oecol, 1997, 112: 524–533
Sommer S. The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front Zool, 2005, 2: 16
Allendorf F W, Luikart G. Conservation and the Genetics of Populations. Malden: Blackwell Publishing, 2007
Brumfield R T, Beerli P, Nickerson D A, et al. The utility of single nucleotide polymorphisms in inferences of population history. Trends Ecol Evol, 2003, 18: 249–256
Morin P A, Luikart G, Wayne R K. The SNP workshop group: SNPs in ecology, evolution and conservation. Trends Ecol Evol, 2004, 19: 208–216
Meyers L A, Bull J J. Fighting change with change: Adaptive variation in an uncertain world. Trends Ecol Evol, 2002, 17: 551–557
Van Tienderen P H, de Haan A A, van der Linden G, et al. Biodiversity assessment using markers for ecologically important traits. Trends Ecol Evol, 2002, 17: 577–582
Kohn M H, Murphy W J, Ostrander E A, et al. Genomics and conservation genetics. Trends Ecol Evol, 2006, 21: 629–637
Bonin A, Nicole F, Pompanon F, et al. Population adaptive index: A new method to help measure intraspecific genetic diversity and prioritize populations for conservation. Conserv Biol, 2007, 21: 697–708
Crandall K A, Bininda-Emonds O R P, Mace G M, et al. Considering evolutionary processes in evolutionary biology. Trends Ecol Evol, 2000, 15: 290–295
Stockwell C A, Hendry A P, Kinnison M T. Contemporary evolution meets conservation biology. Trends Ecol Evol, 2003, 18: 94–101
Siddle H V, Kreiss A, Eldridge M D B, et al. Transmission of a fatal clonal tumor by biting occurs due to depleted MHC diversity in a threatened carnivorous marsupial. Proc Natl Acad Sci USA, 2007, 104: 16221–16226
Miller H C, Allendorf F, Daugherty C H. Genetic diversity and differentiation at MHC genes in island populations of tuatara (Sphenodon spp.). Mol Ecol, 2010, 19: 3894–3908
Spurgin L G, Richardson D S. How pathogens drive genetic diversity: MHC, mechanisms and misunderstandings. Proc Roy Soc B-Biol Sci, 2010, 277: 979–988
Schwensow N, Fietz J, Dausmann K H. Neutral versus adaptive genetic variation in parasite resistance: Importance of major histocompatibility complex supertypes in a free-ranging primate. Heredity, 2007, 99: 265–277
Bernatchez L, Landry C. MHC studies in nonmodel vertebrates: What have we learned about natural selection in 15 years? J Evol Biol, 2003, 16: 363–377
Spurgin L G, Richardson D S. How pathogens drive genetic diversity: MHC, mechanisms and misunderstandings. Proc Roy Soc B, 2010, 277: 979–988
Dengjel J, Schoor O, Fischer R, et al. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc Natl Acad Sci USA, 2005, 102: 7922–7927
Ohta T. On the pattern of polymorphisms at major histocompatibility complex loci. J Mol Evol, 1998, 46: 633–638
Radwan J, Biedrzycka A, Babik W. Does reduced MHC diversity decrease viability of vertebrate populations? Biol Conserv, 2010, 143: 537–544
Stenzel A, Lu T, Koch W A, et al. Patterns of linkage disequilibrium in the MHC region on human chromosome 6p. Hum Genet, 2004, 114: 377–385
Kelley J, Walter L, Trowsdale J. Comparative genomics of major histocompatibility complexes. Immunogenetics, 2005, 56: 683–695
Li M, Liu Z J, Gou J X, et al. Phylogeography and population struc ture of the golden monkeys (Rhinopithecus roxellana): Inferred from mitochondrial DNA sequences. Am J Primatol, 2007, 69: 1195–1209
Pan D, Hu H X, Meng S J, et al. A high polymorphism level in Rhinopithecus roxellana. Int J Primatol, 2009, 30: 337–351
Thompson J D, Gibson T J, Plewniak F, et al. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res, 1997, 25: 4876–4882
Tamura K, Dudley J, Nei M, et al. MEGA 4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol, 2007, 24: 1596–1599
Bryja J, Charbonnel N, Berthier K, et al. Density-related changes in selection pattern for major histocompatibility complex genes in fluctuating populations of voles. Mol Evol, 2007, 16: 5084–5097
Kennedy L J, Ryvar R, Gaskell R M, et al. Sequence analysis of MHC DRB alleles in domestic cats from the United Kingdom. Immunogenetics, 2002, 54: 348–352
Reche P A, Reinherz E L. Sequence variability analysis of human class I and class II MHC molecules: Functional and structural correlates of amino acid polymorphisms. J Mol Biol, 2003, 331: 623–641
Guindon S, Gascuel O. A simple, fast and accurate algorithm to estimate large phylogenies by max-imum likelihood. Sys Biol, 2003, 52: 696–704
Posada D, Crandall K A. Modeltest: Testing the model of DNA substitution. Bioinformatics, 1998, 14: 817–818
Huson D H, Bryant D. Application of phylogenetic networks in evolutionary studies. Mol Biol Evol, 2006, 23: 254–267
Hudson R R. Two-locus sampling distributions and their application. Genetics, 2001, 159: 1805–1817
McVean G, Awadalla P, Fearnhead P. A coalescennt-based method for detecting and estimating recombination from gene sequences. Genetics, 2002, 160: 1231–1241
Richman A D, Herrera L G, Nash D, et al. Relative roles of mutation and recombination in generating allelic polymorphism at an MHC class II locus in Peromyscus maniculatus. Genet Res Camb, 2003, 82: 89–99
Delport W, Poon A F, Frost S D, et al. Datamonkey 2010: A suite of phylogenetic analysis tools for evolutionary biology. Bioinformatics, 2010, 26: 2455–2457
Librado P, Rozas J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics, 2009, 25: 1451–1452
Yang Z. PAML 4: Phylogenetic analysis by maximum likelihood. Mol Biol Evol, 2007, 24: 1586–1591
Anisimova M, Nielsen R, Yang Z. Effect of recombination on the accuracy of the likelihood method for detecting positive selection at amino acid sites. Genetics, 2003, 164: 1229–1236
Goldman N, Yang Z. A codon-based model of nucleotide substitution for protein-coding DNA sequences. Mol Biol Evol, 1994, 11: 725–736
Yang Z, Nielsen R, Goldman N, et al. Codon substitution models for heterogeneous selection pressure at amino acid sites. Genetics, 2000, 155: 431–449
Yang Z, Wong W S W, Nielsen R. Bayes empirical bayes inference of amino acid sites under positive selection. Mol Biol Evol, 2005, 22: 1107–1118
Klein J, Bontrop R E, Dawkins R L, et al. Nomenclature for the major histocompatibility complexes of different species: A proposal. Immunogenetics, 1990, 31: 217–219
Lafont B A P, McGraw C M, Stukes S A, et al. The locus encoding an oligomorphic family of MHC-A alleles (Mane-A*06/Mamu-A*05) is present at high frequency in several macaque species. Immunogenetics, 2007, 59: 211–223
Schwensow N, Eberle M, Sommer S. Compatibility counts: MHC-associated mate choice in a wild promiscuous primate. Proc Roy Soc B, 2008, 275: 555–564
Doxiadis G G M, Rouweler A J M, Groot N G, et al. Extensive sharing of MHC class II alleles between rhesus and cynomolgus macaques. Immunogenetics, 2006, 58: 259–268
Nei M, Gu X, Sitnikova T. Evolution by the birth-and-death process in multigene families of the vertebrate immune system. Proc Natl Acad Sci USA, 1997, 94: 7799–7806
Pan D, Hu H X, Meng S J, et al. Population analysis of golden monkey using mitochondrial control region: High level of polymorphism and its implications. Chin Sci Bull, 2005, 50: 2489–2494
Bollmer J L, Hull J M, Ernest H B, et al. Reduced MHC and neutralvariation in the Galapagos hawk, an island endemic. BMC Evol Biol, 2011, 11: 43
Weber D S, Stewart B S, Schienman J, et al. Major histocompatibility complex variation at three class II loci in the northern elephant seal. Mol Evol, 2004, 13: 711–718
Babik W, Pabijan M, Arntzen J W, et al. Long-term survival of a urodele amphibian despite depleted major histocompatibility complex variation. Mol Evol, 2009, 18: 769–781
Mason R A B, Browning T L, Eldridge D B. Reduced MHC class II diversity in island compared to mainland populations of the black-footed rock-wallaby (Petrogale lateralis lateralis). Conserv Gene, 2011, 12: 91–103
Biedrzycka A, Radwan J. Population fragmentation and major histocompatibility complex variation in the spotted suslik, Spermophilus suslicus. Mol Evol, 2008, 17: 4801–4811
Luo M F, Liu Z J, Pan H J, et al. Historical geographic dispersal of the golden snub-nosed monkey (Rhinopithecus roxellana) and the influence of climatic oscillations. Am J Primatol, 2012, 74: 91–101
Garrigan D, Hedrick P W. Perspective: Detecting adaptive molecular polymorphism: Lessons from the MHC. Evolution, 2003, 57: 1707–1722
Thomas J C, Godfrey P A, Feldgarden M, et al. Candidate targets of balancing selection in the genome of Staphylococcus aureus. Mol Biol Evol, 2012, 29: 1175–1186
Klein J, Sato A, Nikolaidis N. MHC, TSP, and the origin of species: From immunogenetics to evolutionary genetics. Annu Rev Genet, 2007, 41: 281–304
Doxiadis G G M, Rouweler A J M, Groot N G de, et al. Extensive sharing of MHC class II alleles between rhesus and cynomolgus macaques. Immunogenetics, 2006, 58: 259–268
Xu S X, Ren W H, Li S Z, et al. Sequence polymorphism and evolution of three cetacean MHC genes. J Mol Evol, 2009, 69: 260–275
Kauppi L, Sajantila A, Jeffreys A J. Recombination hotspots rather than population history dominate linkage disequilibrium in the MHC class II region. Hum Mol Genet, 2003, 12: 33–40
Carnngton M. Recombination within the human MHC. Immunol Rev, 1999, 167: 245–256
Pan H J, Shi F L, Chang Z F, et al. Mitochondrial DNA variation analysis suggests extreme low genetic diversity in Guizhou snub-nosed monkeys (Rhinopithecus brelichi). Chin Sci Bull, 2011, 56: 2541–2544
Fang S G. Does population increase alone ensure the long-term survival of endangered species? Chin Sci Bull, 2011, 56: 2521–2522
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Luo, M., Pan, H. MHC II DRB variation and trans-species polymorphism in the golden snub-nosed monkey (Rhinopithecus roxellana). Chin. Sci. Bull. 58, 2119–2127 (2013). https://doi.org/10.1007/s11434-013-5713-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5713-6