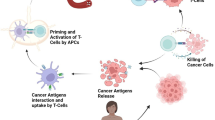
Abstract
Cancer treatment has evolved rapidly due to major advances in tumor immunity research. However, due to the complexity, heterogeneity, and immunosuppressive microenvironment of tumors, the overall efficacy of immunotherapy is only 20%. In recent years, nanoparticles have attracted more attention in the field of cancer immunotherapy because of their remarkable advantages in biocompatibility, precise targeting, and controlled drug delivery. However, the clinical application of nanomedicine also faces many problems concerning biological safety, and the synergistic mechanism of nano-drugs with immunity remains to be elucidated. Our study summarizes the functional characteristics and regulatory mechanisms of nanoparticles in the cancer immune microenvironment and how nanoparticles activate and long-term stimulate innate immunity and adaptive immunity. Finally, the current problems and future development trends regarding the application of nanoparticles are fully discussed and prospected to promote the transformation and application of nanomedicine used in cancer treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Xia C, Dong X, Li H, et al. Cancer statistics in China and United States, 2022: profiles, trends, and determinants. Chin Med J (Engl), 2022,135(5):584–590
Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin, 2022,72(1):7–33
DeVita VT, Chu E. A history of cancer chemotherapy. Cancer Res, 2008,68(21):8643–8653
Chandra RA, Keane FK, Voncken FEM, et al. Contemporary radiotherapy: present and future. Lancet, 2021,398(10295):171–184
Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol, 2020,20(11):651–668
Wang Z, Cao YJ. Adoptive Cell Therapy Targeting Neoantigens: A Frontier for Cancer Research. Front Immunol, 2020,11:176
Saxena M, van der Burg SH, Melief CJM, et al. Therapeutic cancer vaccines. Nat Rev Cancer, 2021,21(6):360–378
Singh AK, McGuirk JP. CAR T cells: continuation in a revolution of immunotherapy. Lancet Oncol, 2020,21(3):e168–e178
Postow MA, Callahan MK, Wolchok JD. Immune Checkpoint Blockade in Cancer Therapy. J Clin Oncol, 2015,33(17):1974–1982
Bagchi S, Yuan R, Engleman EG. Immune Checkpoint Inhibitors for the Treatment of Cancer: Clinical Impact and Mechanisms of Response and Resistance. Annu Rev Pathol, 2021,16:223–249
Petrosky E, Bocchini JA Jr, Hariri S, et al. Use of 9-valent human papillomavirus (HPV) vaccine: updated HPV vaccination recommendations of the advisory committee on immunization practices. MMWR Morb Mortal Wkly Rep, 2015,64(11):300–304
Paz-Ares L, Ciuleanu TE, Cobo M, et al. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomised, open-label, phase 3 trial. Lancet Oncol, 2021,22(2):198–211
Hellmann MD, Paz-Ares L, Bernabe Caro R, et al. Nivolumab plus Ipilimumab in Advanced Non-Small-Cell Lung Cancer. N Engl J Med, 2019,381(21):2020–2031
Hodi FS, Chiarion-Sileni V, Gonzalez R, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol, 2018,19(11):1480–1492
Hou AJ, Chen LC, Chen YY. Navigating CAR-T cells through the solid-tumour microenvironment. Nat Rev Drug Discov, 2021,20(7):531–550
Goldberg MS. Improving cancer immunotherapy through nanotechnology. Nat Rev Cancer, 2019,19(10):587–602
Irvine DJ, Dane EL. Enhancing cancer immunotherapy with nanomedicine. Nat Rev Immunol, 2020,20(5):321–334
Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res, 1986,46(12 Pt 1):6387–6392
Iyer AK, Khaled G, Fang J, et al. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov Today, 2006,11(17–18):812–818
Wang J, Zhang B, Sun J, et al. Nanomedicine-Enabled Modulation of Tumor Hypoxic Microenvironment for Enhanced Cancer Therapy. Adv Ther (Weinh), 2020,3(1):1900083
Jin MZ, Jin WL. The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct Target Ther, 2020,5(1):166
Shields CWt 4th, Wang LL, Evans MA, et al. Materials for Immunotherapy. Adv Mater, 2020,32(13):e1901633
Thomas PD, Kahn M. Kat3 coactivators in somatic stem cells and cancer stem cells: biological roles, evolution, and pharmacologic manipulation. Cell Biol Toxicol, 2016,32(1):61–81
Zhou J, Wang G, Chen Y, et al. Immunogenic cell death in cancer therapy: Present and emerging inducers. J Cell Mol Med, 2019,23(8):4854–4865
Kroemer G, Galluzzi L, Kepp O, et al. Immunogenic cell death in cancer therapy. Annu Rev Immunol, 2013,31:51–72
Banstola A, Jeong JH, Yook S. Immunoadjuvants for cancer immunotherapy: A review of recent developments. Acta Biomater, 2020,114:16–30
Mohamed SIA, Jantan I, Haque MA. Naturally occurring immunomodulators with antitumor activity: An insight on their mechanisms of action. Int Immunopharmacol, 2017,50:291–304
Mitchell MJ, Billingsley MM, Haley RM, et al. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov, 2021,20(2):101–124
Kuai R, Yuan W, Son S, et al. Elimination of established tumors with nanodisc-based combination chemoimmunotherapy. Sci Adv, 2018,4(4):eaao1736
Ding Y, Sun Z, Gao Y, et al. Plasmon-Driven Catalytic Chemotherapy Augments Cancer Immunotherapy through Induction of Immunogenic Cell Death and Blockage of IDO Pathway. Adv Mater, 2021,33(34):e2102188
Feng B, Zhou F, Hou B, et al. Binary Cooperative Prodrug Nanoparticles Improve Immunotherapy by Synergistically Modulating Immune Tumor Microenvironment. Adv Mater, 2018,30(38):e1803001
Mu QG, Lin G, Jeon M, et al. Iron oxide nanoparticle targeted chemo-immunotherapy for triple negative breast cancer. Mater Today (Kidlington), 2021,50:149–169
de Souza ASC, Goncalves LB, Lepique AP, et al. The Role of Autophagy in Tumor Immunology-Complex Mechanisms That May Be Explored Therapeutically. Front Oncol, 2020,10:603661
Wang X, Li M, Ren K, et al. On-Demand Autophagy Cascade Amplification Nanoparticles Precisely Enhanced Oxaliplatin-Induced Cancer Immunotherapy. Adv Mater, 2020,32(32):e2002160
Li W, Yang J, Luo L, et al. Targeting photodynamic and photothermal therapy to the endoplasmic reticulum enhances immunogenic cancer cell death. Nat Commun, 2019,10(1):3349
Yan S, Zeng X, Tang Y, et al. Activating Antitumor Immunity and Antimetastatic Effect Through Polydopamine-Encapsulated Core-Shell Upconversion Nanoparticles. Adv Mater, 2019,31(46):e1905825
Chen C, Ni X, Jia S, et al. Massively Evoking Immunogenic Cell Death by Focused Mitochondrial Oxidative Stress using an AIE Luminogen with a Twisted Molecular Structure. Adv Mater, 2019,31(52):e1904914
He C, Duan X, Guo N, et al. Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy. Nat Commun, 2016,7:12499
Xu C, Jiang Y, Huang J, et al. Second Near-Infrared Light-Activatable Polymeric Nanoantagonist for Photothermal Immunometabolic Cancer Therapy. Adv Mater, 2021,33(36):e2101410
Shao D, Zhang F, Chen F, et al. Biomimetic Diselenide-Bridged Mesoporous Organosilica Nanoparticles as an X-ray-Responsive Biodegradable Carrier for ChemoImmunotherapy. Adv Mater, 2020,32(50):e2004385
Song R, Li T, Ye J, et al. Acidity-Activatable Dynamic Nanoparticles Boosting Ferroptotic Cell Death for Immunotherapy of Cancer. Adv Mater, 2021,33(31):e2101155
Su Z, Xiao Z, Wang Y, et al. Codelivery of Anti-PD-1 Antibody and Paclitaxel with Matrix Metalloproteinase and pH Dual-Sensitive Micelles for Enhanced Tumor Chemoimmunotherapy. Small, 2020,16(7):e1906832
Zhou P, Qin J, Zhou C, et al. Multifunctional nanoparticles based on a polymeric copper chelator for combination treatment of metastatic breast cancer. Biomaterials, 2019,195:86–99
Hu CM, Zhang L, Aryal S, et al. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc Natl Acad Sci USA, 2011,108(27):10980–10985
Kroll AV, Fang RH, Zhang L. Biointerfacing and Applications of Cell Membrane-Coated Nanoparticles. Bioconjug Chem, 2017,28(1):23–32
Bahmani B, Gong H, Luk BT, et al. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat Commun, 2021,12(1):1999
Kroll AV, Fang RH, Jiang Y, et al. Nanoparticulate Delivery of Cancer Cell Membrane Elicits Multiantigenic Antitumor Immunity. Adv Mater, 2017,29(47)
Liu WL, Zou MZ, Liu T, et al. Expandable Immunotherapeutic Nanoplatforms Engineered from Cytomembranes of Hybrid Cells Derived from Cancer and Dendritic Cells. Adv Mater, 2019,31(18):e1900499
Deng G, Sun Z, Li S, et al. Cell-Membrane Immunotherapy Based on Natural Killer Cell Membrane Coated Nanoparticles for the Effective Inhibition of Primary and Abscopal Tumor Growth. ACS Nano, 2018,12(12):12096–12108
Cao H, Dan Z, He X, et al. Liposomes Coated with Isolated Macrophage Membrane Can Target Lung Metastasis of Breast Cancer. ACS Nano, 2016,10(8):7738–7748
Chen Q, Bai H, Wu W, et al. Bioengineering Bacterial Vesicle-Coated Polymeric Nanomedicine for Enhanced Cancer Immunotherapy and Metastasis Prevention. Nano Lett, 2020,20(1):11–21
Kim OY, Park HT, Dinh NTH, et al. Bacterial outer membrane vesicles suppress tumor by interferon-gamma-mediated antitumor response. Nat Commun, 2017,8(1):626
Min Y, Roche KC, Tian S, et al. Antigen-capturing nanoparticles improve the abscopal effect and cancer immunotherapy. Nat Nanotechnol, 2017,12(9):877–882
Wang B, An J, Zhang H, et al. Personalized Cancer Immunotherapy via Transporting Endogenous Tumor Antigens to Lymph Nodes Mediated by Nano Fe3O4. Small, 2018,14(38):e1801372
Lei K, Kurum A, Tang L. Mechanical Immunoengineering of T cells for Therapeutic Applications. Acc Chem Res, 2020,53(12):2777–2790
Zhou Z, Zhang B, Zai W, et al. Perfluorocarbon nanoparticle-mediated platelet inhibition promotes intratumoral infiltration of T cells and boosts immunotherapy. Proc Natl Acad Sci USA, 2019,116(24):11972–11977
Yin Q, Yu W, Grzeskowiak CL, et al. Nanoparticle-enabled innate immune stimulation activates endogenous tumor-infiltrating T cells with broad antigen specificities. Proc Natl Acad Sci USA, 2021,118(21):e2016168118
Xu C, Jiang Y, Han Y, et al. A Polymer Multicellular Nanoengager for Synergistic NIR-II Photothermal Immunotherapy. Adv Mater, 2021,33(14):e2008061
Voskoboinik I, Whisstock JC, Trapani JA. Perforin and granzymes: function, dysfunction and human pathology. Nat Rev Immunol, 2015,15(6):388–400
Zhao Q, Gong Z, Li Z, et al. Target Reprogramming Lysosomes of CD8+ T Cells by a Mineralized Metal-Organic Framework for Cancer Immunotherapy. Adv Mater, 2021,33(17):e2100616
Bae J, Parayath N, Ma W, et al. BCMA peptide-engineered nanoparticles enhance induction and function of antigen-specific CD8(+) cytotoxic T lymphocytes against multiple myeloma: clinical applications. Leukemia, 2020,34(1):210–223
Jiang Y, Krishnan N, Zhou J, et al. Engineered Cell-Membrane-Coated Nanoparticles Directly Present Tumor Antigens to Promote Anticancer Immunity. Adv Mater, 2020,32(30):e2001808
Ai L, Xu A, Xu J. Roles of PD-1/PD-L1 Pathway: Signaling, Cancer, and Beyond. Adv Exp Med Biol, 2020,1248:33–59
Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med, 2012,366(26):2455–2465
Zhong Y, Ma Z, Wang F, et al. In vivo molecular imaging for immunotherapy using ultra-bright near-infrared-IIb rare-earth nanoparticles. Nat Biotechnol, 2019,37(11):1322–1331
Wang D, Wang T, Yu H, et al. Engineering nanoparticles to locally activate T cells in the tumor microenvironment. Sci Immunol, 2019,4(37):eaau6584
Lang T, Liu Y, Zheng Z, et al. Cocktail Strategy Based on Spatio-Temporally Controlled Nano Device Improves Therapy of Breast Cancer. Adv Mater, 2019,31(5):e1806202
Chiang CS, Lin YJ, Lee R, et al. Combination offucoidan-based magnetic nanoparticles and immunomodulators enhances tumour-localized immunotherapy. Nat Nanotechnol, 2018,13(8):746–754
Hong J, Kang M, Jung M, et al. T-Cell-Derived Nanovesicles for Cancer Immunotherapy. Adv Mater, 2021,33(33):e2101110
Alhallak K, Sun J, Wasden K, et al. Nanoparticle T-cell engagers as a modular platform for cancer immunotherapy. Leukemia, 2021,35(8):2346–2357
Parayath NN, Stephan SB, Koehne AL, et al. In vitro-transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo. Nat Commun, 2020,11(1):6080
Tang L, Zheng Y, Melo MB, et al. Enhancing T cell therapy through TCR-signaling-responsive nanoparticle drug delivery. Nat Biotechnol, 2018,36(8):707–716
Gaudino SJ, Kumar P. Cross-Talk Between Antigen Presenting Cells and T Cells Impacts Intestinal Homeostasis, Bacterial Infections, and Tumorigenesis. Front Immunol, 2019,10:360
Schuijs MJ, Hammad H, Lambrecht BN. Professional and ‘Amateur’ Antigen-Presenting Cells In Type 2 Immunity. Trends Immunol, 2019,40(1):22–34
Lindenbergh MFS, Stoorvogel W. Antigen Presentation by Extracellular Vesicles from Professional Antigen-Presenting Cells. Annu Rev Immunol, 2018,36:435–459
Cho NH, Cheong TC, Min JH, et al. A multifunctional core-shell nanoparticle for dendritic cell-based cancer immunotherapy. Nat Nanotechnol, 2011,6(10):675–682
Kranz LM, Diken M, Haas H, et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature, 2016,534(7607):396–401
Nuhn L, De Koker S, Van Lint S, et al. Nanoparticle-Conjugate TLR7/8 Agonist Localized Immunotherapy Provokes Safe Antitumoral Responses. Adv Mater, 2018,30(45):e1803397
Liu Y, Crowe WN, Wang L, et al. An inhalable nanoparticulate STING agonist synergizes with radiotherapy to confer long-term control of lung metastases. Nat Commun, 2019,10(1):5108
Atukorale PU, Raghunathan SP, Raguveer V, et al. Nanoparticle Encapsulation of Synergistic Immune Agonists Enables Systemic Codelivery to Tumor Sites and IFNbeta-Driven Antitumor Immunity. Cancer Res, 2019,79(20):5394–5406
Pan Y, Yu Y, Wang X, et al. Tumor-Associated Macrophages in Tumor Immunity. Front Immunol, 2020,11:583084
Mehla K, Singh PK. Metabolic Regulation of Macrophage Polarization in Cancer. Trends Cancer, 2019,5(12):822–834
Locati M, Curtale G, Mantovani A. Diversity, Mechanisms, and Significance of Macrophage Plasticity. Annu Rev Pathol, 2020,15:123–147
Yunna C, Mengru H, Lei W, et al. Macrophage M1/M2 polarization. Eur J Pharmacol, 2020,877:173090
Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity, 2014,41(1):49–61
Cassetta L, Pollard JW. Tumor-associated macrophages. Curr Biol, 2020,30(6):R246–R248
Zhang M, He Y, Sun X, et al. A high M1/M2 ratio of tumor-associated macrophages is associated with extended survival in ovarian cancer patients. J Ovarian Res, 2014,7:19
Mantovani A, Allavena P. The interaction of anticancer therapies with tumor-associated macrophages. J Exp Med, 2015,212(4):435–445
Zhou J, Tang Z, Gao S, et al. Tumor-Associated Macrophages: Recent Insights and Therapies. Front Oncol, 2020,10:188
Li C, Xu X, Wei S, et al. Tumor-associated macrophages: potential therapeutic strategies and future prospects in cancer. J Immunother Cancer, 2021,9(1):e001341
Zanganeh S, Hutter G, Spitler R, et al. Iron oxide nanoparticles inhibit tumour growth by inducing pro-inflammatory macrophage polarization in tumour tissues. Nat Nanotechnol, 2016,11(11):986–994
Rong L, Zhang Y, Li WS, et al. Iron chelated melaninlike nanoparticles for tumor-associated macrophage repolarization and cancer therapy. Biomaterials, 2019,225:119515
Zhang F, Parayath NN, Ene CI, et al. Genetic programming of macrophages to perform anti-tumor functions using targeted mRNA nanocarriers. Nat Commun, 2019,10(1):3974
Gao X, Li S, Ding F, et al. A Virus-Mimicking Nucleic Acid Nanogel Reprograms Microglia and Macrophages for Glioblastoma Therapy. Adv Mater, 2021,33(9):e2006116
Rodell CB, Arlauckas SP, Cuccarese MF, et al. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat Biomed Eng, 2018,2(8):578–588
Majeti R, Chao MP, Alizadeh AA, et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell, 2009,138(2):286–299
Chao MP, Alizadeh AA, Tang C, et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell, 2010,142(5):699–713
Willingham SB, Volkmer JP, Gentles AJ, et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci U S A, 2012,109(17):6662–6667
Feng M, Jiang W, Kim BYS, et al. Phagocytosis checkpoints as new targets for cancer immunotherapy. Nat Rev Cancer, 2019,19(10):568–586
Advani R, Flinn I, Popplewell L, et al. CD47 Blockade by Hu5F9-G4 and Rituximab in Non-Hodgkin’s Lymphoma. N Engl J Med, 2018,379(18):1711–1721
Rao L, Wu L, Liu Z, et al. Hybrid cellular membrane nanovesicles amplify macrophage immune responses against cancer recurrence and metastasis. Nat Commun, 2020,11(1):4909
Rao L, Zhao SK, Wen C, et al. Activating Macrophage-Mediated Cancer Immunotherapy by Genetically Edited Nanoparticles. Adv Mater, 2020,32(47):e2004853
Apostolopoulos V, McKenzie IF. Role of the mannose receptor in the immune response. Curr Mol Med, 2001,1(4):469–474
Azad AK, Rajaram MV, Schlesinger LS. Exploitation of the Macrophage Mannose Receptor (CD206) in Infectious Disease Diagnostics and Therapeutics. J Cytol Mol Biol, 2014,1(1):1000003
Kang M, Lee SH, Kwon M, et al. Nanocomplex-Mediated In Vivo Programming to Chimeric Antigen Receptor-M1 Macrophages for Cancer Therapy. Adv Mater, 2021,33(43):e2103258
Zheng C, Wang Q, Wang Y, et al. In Situ Modification of the Tumor Cell Surface with Immunomodulating Nanoparticles for Effective Suppression of Tumor Growth in Mice. Adv Mater, 2019,31(32):e1902542
Ji T, Lang J, Ning B, et al. Enhanced Natural Killer Cell Immunotherapy by Rationally Assembling Fc Fragments of Antibodies onto Tumor Membranes. Adv Mater, 2019,31(6):e1804395
Kim KS, Han JH, Park JH, et al. Multifunctional nanoparticles for genetic engineering and bioimaging of natural killer (NK) cell therapeutics. Biomaterials, 2019,221:119418
Biber G, Sabag B, Raiff A, et al. Modulation of intrinsic inhibitory checkpoints using nano-carriers to unleash NK cell activity. EMBO Mol Med, 2022,14(1):e14073
Gao S, Li T, Guo Y, et al. Selenium-Containing Nanoparticles Combine the NK Cells Mediated Immunotherapy with Radiotherapy and Chemotherapy. Adv Mater, 2020,32(12):e1907568
Wei Z, Yi Y, Luo Z, et al. Selenopeptide Nanomedicine Activates Natural Killer Cells for Enhanced Tumor Chemoimmunotherapy. Adv Mater, 2022:e2108167
Nakamura T, Sato T, Endo R, et al. STING agonist loaded lipid nanoparticles overcome anti-PD-1 resistance in melanoma lung metastasis via NK cell activation. J Immunother Cancer, 2021,9(7):e002852
Veglia F, Sanseviero E, Gabrilovich DI. Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity. Nat Rev Immunol, 2021,21(8):485–498
Gabrilovich DI. Myeloid-Derived Suppressor Cells. Cancer Immunol Res, 2017,5(1):3–8
Ledo AM, Sasso MS, Bronte V, et al. Co-delivery of RNAi and chemokine by polyarginine nanocapsules enables the modulation of myeloid-derived suppressor cells. J Control Release, 2019,295:60–73
Ding D, Zhong H, Liang R, et al. Multifunctional Nanodrug Mediates Synergistic Photodynamic Therapy and MDSCs-Targeting Immunotherapy of Colon Cancer. Adv Sci (Weinh), 2021,8(14):e2100712
Zhang P, Miska J, Lee-Chang C, et al. Therapeutic targeting of tumor-associated myeloid cells synergizes with radiation therapy for glioblastoma. Proc Natl Acad Sci U S A, 2019,116(47):23714–23723
Zhang N, Liu S, Shi S, et al. Solubilization and delivery of Ursolic-acid for modulating tumor microenvironment and regulatory T cell activities in cancer immunotherapy. J Control Release, 2020,320:168–178
Ou W, Thapa RK, Jiang L, et al. Regulatory T cell-targeted hybrid nanoparticles combined with immunocheckpoint blockage for cancer immunotherapy. J Control Release, 2018,281:84–96
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that there are no conflicts of interest relevant to this article.
Additional information
This work was supported by grants from the Fundamental Research Funds for the Central Universities (No. 2042021kf0129) and the Natural Science Foundation of Hubei Province of China (No. 2021CFB086).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
He, Jj., Li, Qq., Zhao, C. et al. Advancement and Applications of Nanotherapy for Cancer Immune Microenvironment. CURR MED SCI 43, 631–646 (2023). https://doi.org/10.1007/s11596-023-2763-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2763-0