Abstract
With disease-modifying drugs on the horizon for degenerative ataxias, ecologically valid, finely granulated, digital health measures are highly warranted to augment clinical and patient-reported outcome measures. Gait and balance disturbances most often present as the first signs of degenerative cerebellar ataxia and are the most reported disabling features in disease progression. Thus, digital gait and balance measures constitute promising and relevant performance outcomes for clinical trials.
This narrative review with embedded consensus will describe evidence for the sensitivity of digital gait and balance measures for evaluating ataxia severity and progression, propose a consensus protocol for establishing gait and balance metrics in natural history studies and clinical trials, and discuss relevant issues for their use as performance outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Based on the recent success of preclinical studies in genetic ataxias, and with several clinical trials currently active, targeted, disease-modifying therapies are on the horizon for spinocerebellar ataxias (SCAs) such as SCA1, 2, 3, and 6 and Friedreich’s ataxia [1,2,3,4]. To perform such clinical trials, there is a critical need for markers evaluating therapeutic outcomes [1, 5, 6]. As the genetic ataxias are rare disorders and current clinical and patient-reported measures demonstrate limited responsiveness, it is crucial to identify more sensitive markers of early disease and individual disease progression to enable trials with smaller sample sizes [5,6,7,8].
Gait and balance disturbances often represent the earliest signs of degenerative ataxia [9,10,11] and are reported by people with ataxia as one of the most disabling features affecting functional mobility as the disease progresses [12,13,14,15]. Thus, measures of gait and balance impairments qualify as both ecologically valid markers of progression and treatment response markers in future clinical trials.
Variability measures of ataxic gait and postural sway in stance have been shown to be strongly related to ataxia severity in multiple cross-sectional studies (reviews in [16,17,18,19]), including sensitivity in pre-ataxic disease stages [20,21,22,23]. The pre-ataxic stage includes carriers of SCA mutations before the manifestation of clinical ataxia symptoms defined by a SARA score below the threshold of 3 points [8, 24]. Digital gait and balance measures are now considered promising candidate outcomes for clinical trials and have been integrated into observational trials to yield further evidence [5, 25, 26].
In addition to cross-sectional sensitivity to early ataxia, clinical trials need objective measures that are capable of reflecting the slowing of disease progression within a reasonable study period (e.g. within 1–2 years). Hence, longitudinal, rather than cross-sectional, studies of gait and balance are needed to determine trajectories of digital measures, alongside clinical measures (e.g. the SARA score) and underlying biomarkers of disease progression. Given the limited number of people with these rare diseases, pooling of patient populations in multicenter, natural history studies will be most effective. This calls for highly standardized procedures of assessment [10, 27].
Measures characterizing the temporal and spatial variability of gait patterns in ataxia have been examined using a wide range of recording technologies (see for reviews [16, 18, 28]), from marker-based capturing systems as gold standard [20, 29, 30], electronic gait mats [22, 31,32,33], camera-based systems [34, 35] and body-worn inertial measurement units (IMUs) [21, 23, 33, 36,37,38]. Variability of gait can be measured both in the clinic/laboratory via active monitoring of prescribed tasks and passive monitoring during daily life. We will review the advantages and challenges of different motion recording technologies. For suitability in multicenter clinical trials, it is important to consider aspects like cost, feasibility without a dedicated gait laboratory or specialist staff, time required to prepare for the measurements, need of expertise in data processing, limitations in the spatial measurement range as well as the potential to characterize gait in daily life.
Based on this assessment and our current knowledge on sensitive gait and balance measures in ataxia, we present an evidence-informed proposal for: (i) a common protocol of gait and balance tests for natural history studies, (ii) sensitive gait and balance measures to be calculated and (iii) recommended data acquisition technology. With this consensus proposal, we aim to stimulate further research within the ataxia community on digital gait and balance measures to meet the requirements of future clinical trials.
Aims and Objectives
The aim of this manuscript is to:
-
1.
Summarize patient-reported mobility impairments as well as the associated decline in quality of life resulting from ataxia.
-
2.
Review evidence for specific measures of walking function and standing balance for use in ataxias. Digital outcome measures should fulfill the following clinimetrics:
-
a.
Sensitivity/specificity to premanifest and mild-moderate ataxia;
-
b.
Concurrent validity (e.g. significant correlations with clinical rating scales);
-
c.
Sensitivity to change over time (longitudinal) and in response to therapy (interventions);
-
d.
Test-retest reliability and minimal detectable change (MDC) necessary to detect subtle changes;
-
e.
Meaningfulness to people with ataxia.
-
a.
-
3.
Recommend a common protocol for multicenter, natural history studies to support the inclusion of digital gait and stance tasks as useful outcomes for clinical intervention trials on ataxia. Standardizing the motor tasks and harmonizing the protocols, instructions, metrics, and technologies is important for pooling data on rare forms of ataxia for regulatory approval of digital outcomes for future clinical trials.
-
4.
Identify the remaining necessary steps to implement digital gait and balance outcome measures in the context of future intervention trials.
Consensus has been reached in three rounds as follows: (1) Collecting relevant topics and requirements towards trial-readiness of digital motor performance measures; (2) Discussing content issues related to motor tests, measures and recording technologies; (3) Drafting and reviewing the manuscript.
Patient-Reported Impairments in Mobility and Associated Decline of Quality of Life
There is ample evidence from patient-reported outcome measures [12,13,14, 39] and questionnaires on quality of life [40,41,42,43,44] that walking and balance are central factors of functional (im)mobility in the disease-related decline in quality of life of people with ataxia. To measure the impact of ataxia on quality of life, many studies use the EQ-5D [45], a generic, standardized measure of health-related quality of life, which assesses health status across five dimensions (mobility, self-care, usual activities, pain/discomfort and anxiety/depression). In several studies of SCAs [40,41,42], the most frequently patient-reported problems were with mobility (“I have moderate/severe problems walking”), even in mildly to moderately affected SCA participants [42]. In addition, the mobility dimension revealed the largest progression slopes in long-term evolution of ataxia [43].
Two FDA (Food and Drug Administration) “Voice of the Patient” meetings have confirmed that people with ataxia feel that walking difficulty is the biggest challenge in daily living [12, 13]. In addition, most of them identified “Lack of balance” as the ataxia symptom with the greatest impact on daily life [13]. “My balance and coordination are affected so I leave class early to avoid the crowded halls”, “SCA2 affects my balance and coordination making me look drunk while walking”, “I ended up losing my balance and falling” [13]. Similarly, balance problems were the most commonly reported problem affecting daily quality of life in Friedreich's ataxia [12, 14, 15].
The importance of gait and mobility limitations is also reflected in the recent development of the PROM-Ataxia scale, in which 147 people with ataxia described their disease symptoms and the associated limitations in daily life [39]. Impaired balance and gait were the most frequently mentioned symptoms in both the physical domain and the domain of daily activities [39].
Digital Gait and Balance Measures Quantifying Ataxia
Clinical Gait Assessment
Clinically, ataxic gait is typically characterized by unstable, stumbling walking, increased step width and high gait variability [46,47,48,49]. The characteristic high variability of walking patterns in people with ataxia are thought to result from the complex interaction between cerebellar-induced deficits in balance control and multi-joint coordination, the compensatory/safety strategies used, and inaccurate postural adjustments to apparent losses of balance [50].
Accordingly, there is broad consensus [10, 16,17,18, 21, 50, 51] that the most striking and distinctive features of ataxic gait are the high variabilities in spatial and temporal metrics (e.g. stride-to-stride variability in length, stride width, stride duration). Although people with ataxia may also show altered pace with slow gait velocity, small step length, long double-support phase and wide step width, these metrics are less specific and sensitive to ataxia and may reflect compensatory strategies and general slowing of gait, e.g. to avoid losing balance and falling, rather than primary cerebellar deficits in control of gait [18].
Gait variability measures have been shown to be sensitive and specific for ataxia, as well as significantly related to clinical ratings of ataxia severity, such as the ICARS [52], SARA [24], BARS [53] and FARS [54] in multiple cross-sectional studies [21,22,23, 29,30,31, 34, 36, 37, 50, 55,56,57,58] (see Table 1 for an overview).
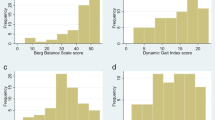
Recently, in one of the largest studies (N=301) using a comprehensive set of gait measures from body-worn, inertial sensors, Shah et al. showed that variability measures were the most discriminative gait characteristics for mild-to-moderate SCA as well as for pre-ataxic SCA compared to healthy controls [21]. This study included measures of gait that depend on measuring foot orientation in space (such as toe-out angle, toe-off angle, the elevation of the feet at mid-swing), as well as trunk measures. The most sensitive and specific measures of gait variability, based on the Receiver-Operating Characteristic (ROC) area under the curve (AUC) to discriminate SCA from control performance, are summarized in Fig. 1A. Figure 1B shows significant correlations between these most sensitive gait variability measures and the SARA ataxia score [21].
(A) Area under the ROC (receiver operating characteristic) Curve (AUC) in descending order for each gait measure discriminating people with spinocerebellar ataxia (SCA) from healthy controls (HC). (B) Pearson correlation of the four most discriminative gait measures with clinical SARA scores related to the ataxia severity of each subtype of SCA 1,2,3 and 6 (adapted from [21])
Besides the relationship with clinical ataxia severity, gait variability is also associated with patient-reported balance impairments. For example, gait variability (step length) is related to the number of reported falls [31, 66, 74] (see Fig. 2) and predicts future falls in cerebellar gait disorders [75]. In addition, gait variability (e.g. lateral step deviation) is correlated with the subjective confidence in daily life activities of balance, measured by the ABC-score (Activities-specific Balance Confidence) [36].
Correlations between the step length coefficient of variation (CV) and the falls/year (A) and SARA scores (B) in 17 ataxic participants. Pearson’s R coefficient (R) and significance (p) are reported (adapted from [74])
Unlike cerebellar ataxia in SCA, Friedreich's ataxia (FRDA) mainly affects afferent connections to the cerebellum, resulting in a combined cerebellar-sensory ataxia. Therefore, the most sensitive/specific gait characteristics may be different in this population. Serrao et al. [50] have conducted the only study comparing the gait characteristics of individuals with FRDA and SCA (SCA1 and SCA2) and found that gait pattern impairments were relatively consistent between groups with the exception of a shorter step length in FRDA. In cross-sectional studies in FRDA, stride length variability was correlated with balance outcomes [58, 71] during self-selected and fast walking speeds, whereas mean spatiotemporal parameters were correlated with falling frequency [76] and lower limb co-ordination [58]. In addition, double support time variability is sensitive to disease duration, balance decline, and ataxia (as measured by the FARS [58] and SARA [70]).
Clinical Balance Assessment in Stance
Standing balance tasks allow the evaluation of ataxia-related, static balance impairments in a “purer” form, without the influence of locomotor dynamics or impairments in multi-joint coordination for goal-directed leg placement. Thus, measures of postural sway during quiet, unsupported stance (static posturography) provide a method to quantify the quality of postural (balance) control [77].
The cerebellum is responsible for integrating somatosensory, vestibular and visual inputs for control of balance and people with cerebellar ataxia become more dependent upon vision to control balance compared to controls [78,79,80,81]. Thus, postural balance tasks with eyes closed are particularly difficult for people with FRDA, likely associated with difficulty using proprioceptive feedback due to spinocerebellar degeneration [62, 82]. Postural stability with eyes closed is highly responsive to disease progression early in FRDA; however, 66% of independently ambulant individuals with FRDA cannot stand with eyes closed [62]. An early study by Diener et al. [83], compared postural sway eyes open (left) and eyes closed (right) for a healthy control and a person with FRDA (Fig. 3). Historically, studies have used a force plate to quantify postural sway as displacement of the body center of pressure, whereas more recent studies have used an inertial sensor placed near the body center of mass (Lumbar 2 level) to quantify anterior-posterior and mediolateral linear accelerations and/or angular velocities [84, 85] (Fig. 4). An overview of postural sway studies in people with ataxia is provided in Table 2.
Recording of sway path (SP) in anteroposterior and lateral direction and the calculated sway direction histogram (SDH). (A) Normal subject. (B) Predominantly lateral sway and very large sway eyes closed, in a patient with Friedreich’s ataxia. Adapted from [80]
(A) Representative examples of representative statokinesiograms (postural sway path) during a 30-s, feet-together, eyes-open stance in a healthy control individual, an individual with pre-ataxic SCA6 and an individual with manifest SCA6. (B) Both sway ellipse area and sway mean velocity are correlated with severity of ataxia, as measured by the SARA in SCA 1,2,3 and 6 [85]
Gait and Stance Tasks with Increased Balance Challenge
Stance Tasks with Increased Balance Challenge
Postural sway characteristics may be more sensitive to detect early and pre-ataxic stages in more complex stance tasks, such as standing with feet together, on a foam surface or with feet in a tandem position or by eliminating visual feedback with eye closure [20, 23, 90, 96]. Pre-ataxic SCA2 participants showed a significantly larger postural sway and jerk both with feet together and in tandem positions compared to healthy controls (HCs) [23]. In fact, the more challenging the standing position, the stronger the relationship between postural sway and years to estimated disease onset in pre-ataxic SCA (types 1, 2, 3 and 6) [20]. Figure 5 shows the increased sensitivity of tandem stance compared to feet together stance in pre-ataxic participants compared to controls. Figure 6 shows the relationship between time to genetically-estimated disease onset [97] and postural sway [98] under various conditions: standing balance task with (A) eyes open, (B) eyes closed and (C) eyes closed on a foam cushion. Thus, studies on pre-ataxic SCA or people with ataxia close to disease onset should include stance tasks in more complex conditions. Adopting a wide stance on a firm surface with eyes open may not be challenging enough to identify impairments in standing balance in such populations.
Postural sway abnormalities in pre-ataxic SCA2 participants (Pre-SCA2) in comparison to healthy participants (HC) for a stance task with feet together and for tandem stance. Shown are stance measures jerks (A) and Path Length (B). ns: P > 0.0013 (after Bonferroni correction); **, P < 0.005; ***, P < 0.0005; Adapted from [23]
Relationship between body sway and estimated time to disease onset for pre-ataxic mutation carriers in different stance tasks. Shown are relationships for genetically-based estimates of onset according to [97]. Each circle represents one participant. Body sway (length of sway path) was determined in three different stance conditions: (A) feet closed (Romberg test, RB) and eyes open; (B) feet closed (Romberg) and eyes closed; (C) feet closed (Romberg test, RB) and eyes closed on a foam cushion (mattress). P-values indicate significant correlations between durations to estimated disease onset and body sway. Reprinted with permission from [20]
Gait Tasks with Increased Balance Challenge
Tandem gait increases the demands on dynamic balance control and also on the accuracy of targeted foot movements and has therefore been shown to be very sensitive to detect mild cerebellar damage [51, 88, 99], including sub-clinical cerebellar deficits [29]. Recent studies revealed increases in body sway and stride time variability when walking in tandem in pre-ataxic SCA mutation carriers [20, 23] with correlations [20] to genetically determined estimations of disease onset [97]. The tandem gait is therefore a very sensitive test in the earliest stages of ataxia, but people with moderate to severe impairments are often unable to perform the test safely.
Turning movements represent a highly relevant component of everyday walking behaviour, since 35–45% of steps occur within turns [100]. Compared to straight walking, turning movements are more challenging in terms of dynamic balance [101,102,103,104], as they involve a stronger demand for anticipatory postural adjustments [105] and trunk-limb coordination strategies [106]. A recent study demonstrated that a measure — lateral velocity change (LVC) — which was used to quantify dynamic balance during turning, is sensitive to pre-ataxic stages and shows strong correlations to self-reported balance confidence in daily life as measured by the ABC-score (r > 0.65) [107]. Thus, dynamic balance measures while turning seem particularly sensitive for detecting subtle changes in ataxia and should be included in studies of pre-ataxic and early disease stages.
Quantifying Walking Behaviour in Daily Life
There is legitimate concern that, despite the heightened potential for reproducibility, the assessment of gait in the clinic may not adequately reflect mobility function during daily life [108, 109]. Under a brief examination in the outpatient clinic, a person with ataxia may appear to walk and display balance better than caregivers report observing during their daily lives. Furthermore, a single, or sparsely spaced, measure of mobility cannot assess day-to-day or other clinically relevant windows of change, such as daily motor fluctuations or effects of fatigue.
Advances in wearable sensor technology enable not only standardized gait and stance assessments in clinical settings, but also allow recordings of gait behavior in everyday life.
Remote monitoring of mobility provides an extended period of observation in the more natural home setting, adding ecological validity to the observed measures.
A first cross-sectional study on daily-life gait in degenerative cerebellar ataxia showed that it was feasible to measure in home environments and that stride-to-stride gait variability measures from inertial sensors — in spite of increased gait variability in real-life walking also in healthy participants [110] — demonstrate high sensitivity to small cross-sectional differences in disease severity, with higher effect sizes in daily-life walking compared to the SARA and clinical gait assessment [36]. Namely, lateral step deviation and a compound measure of spatial step variability (SPcmp) distinguished people with ataxia from healthy controls with a discrimination accuracy of 0.86. Both gait measures were highly correlated with clinical ataxia severity (SARA, effect size ρ=0.76) and patient-reported balance confidence (ABC-score, ρ=0.66). These measures detected group differences even when the difference was only 1 point in the clinical SARAposture&gait subscore, with the highest effect sizes observed for real-life walking (effect size d=0.67, Fig. 7). The compound measure SPcmp — integrating variability in the anterior-posterior as well as in the medio-lateral dimension — hereby seems to benefit from capturing different compensation strategies employed in different disease stages.
Differences between subgroups of participants with cerebellar ataxia (CA) stratified according to gait and posture ataxia severity as determined by the SARAp&g subscore [reprinted by permission from [36]]. Subgroups: CAMild: SARAg&p [0:2], CAMod: SARAg&p= [3–4], CASev: SARAg&p [5–6]. Shown are group differences for constrained lab-based walking and real-life walking). LatStepDev and the compound measure of spatial variability were sensitive in distinguishing these severity subgroups also during real-life walking
A recent study examined ankle movements captured remotely by one IMU over a period of 1 week [73]. Individuals with ataxia revealed smaller, slower, and less powerful ankle submovements during natural behaviour at home. A composite measure based on ankle submovements strongly correlated with ataxia rating scale scores (Pearson’s r = 0.82–0.88), and self-reported function (r = 0.81) (PROM-Ataxia) [39], and had high test-retest reliability (ICC=0.95).
In addition to the analysis of straight walking episodes, analyzing turning movements in daily life distinguished not only ataxic, but also pre-ataxic, participants from healthy controls (effect sizes δ=0.68 and δ=0.53 respectively). Moreover, a measure of dynamic balance during turning detected a significant longitudinal change in a one-year follow-up assessment of people with degenerative cerebellar ataxia, with a large effect size (rprb=0.66) [38].
Challenges to Quantify Walking Behaviour in Daily Life
Despite these promising results, several challenges remain in recording gait in daily life. The total number of days or hours per day required to obtain reliable gait measures are uncertain. Studies in in other neurological diseases suggest that three days of monitoring may be sufficient to capture real-life gait performance; however, longer periods (e.g. 6 to 10 days) may be needed to fully capture day-to-day variability and establish strong correlations with patient-reported clinical measures [111].
In rea-life walking, gait measures are substantially influenced by contextual and environmental factors [112,113,114], both for healthy individuals and clinical populations (Parkinson’s disease, dementia, multiple sclerosis, cerebral palsy) [115,116,117,118,119,120]. Performance measures such as mean gait speed and especially gait variability measures are sensitive to gait bout length and other contextual factors [115, 118, 120] (e.g. stride length variability, stride duration variability). The analysis of shorter walking bouts for indoor walking — compared to longer walks outdoors — inherently delivers increased variability measures for both healthy controls and people with ataxia [110, 118]. Thus, it remains an open question whether the analysis should be restricted to a specific size of gait bouts, averaged over all gait bouts [115] or gait bouts matched according to macroscopic gait parameters [121]. Further work is required to determine the influence of data aggregation on real-world gait data in people with ataxia, particularly concerning the calculation of gait variability outcomes.
Sensitivity to Longitudinal Change
Gait variability and body sway measures have shown their sensitivity to ataxia severity predominantly via cross-sectional correlations with clinical ataxia scores [21,22,23, 29,30,31, 34, 36, 37, 50, 55,56,57]. However, these correlations with scores like SARA are strongly influenced by the range of disease severity (range of observations [122]) in the examined population. For cohorts that encompass a wide range of disease stages, many gait measures, including unspecific ones like gait speed, show significant correlation with disease severity, often predominantly driven by participants at the ends of the examined disease severity spectrum [122] (Fig. 8). Conversely, interventional trial requires the quantification of individual change in relatively short time-frames (e.g. over 1 year) in subjects with mostly mild-to-moderate disease (see 1-year follow-up in Fig. 8).
Illustration of different ways to show sensitivity to changes in ataxia severity. In most studies, cross-sectional analysis (blue) has been performed to show sensitivity to ataxia severity by correlations of balance and gait digital measures with clinical ataxia scores like the SARA, the FARS or the number of falls. These correlations with clinical ataxia scores are strongly influenced by the range of disease severity (range of observations [122]). Longitudinal (red): To serve as valid performance measure in ataxia intervention trials, these gait measures need to prove their sensitivity to individual longitudinal change over short time-spans (e.g. 1 year). In addition, the target population in clinical trials will most likely not encompass the full range of disease severity, but will be limited to, for example, mild-to-moderate disease
Few studies have examined the longitudinal course of gait impairment in ataxia, observing limited sensitivity to changes over time [61, 62, 123]. Changes in medial-lateral sway amplitude during gait using a triaxial accelerometer attached to the upper back were detected after 1.5 years in 25 people with spinocerebellar degeneration [65]. In a recent multicenter study based on 6 depth-imaging cameras (multi-Kinect system [35]), longitudinal analysis of 17 participants with SCA3 revealed significant change in gait measures between baseline and 1-year follow-up in slow gait with large effect sizes (stride length variability: rprb=0.66; lateral sway: rprb=0.73) (Fig. 9A) [25]. In this study, sample size estimation for lateral sway reveals a required cohort size of n=43 for detecting a 50% reduction of natural progression in this measure, compared to n=240 for the clinical ataxia score SARA (Fig. 9B).
(A) Longitudinal analyses of 1-year follow-up assessments: Within-subject changes between baseline and 1-year follow-up for a SCA 3 group. Upper panel: Within-subject changes in the SARA score and the gait measures of lateral sway and Stride length CV in the slow walking condition from baseline (BL) at the 1-year follow-up (FU). Lower panel: Within-subject changes between baseline and 1-year follow-up represented as delta (∆). In all panels, SARA scores of individual participants with cerebellar ataxia are colour coded. Black dotted line = mean change across all participants. The stars indicate significant differences between timepoints (*≡ p<0.05, **≡ p<0.0083 Bonferroni-corrected, ***≡ p<0.001). Effect sizes rprb were determined by matched-pairs rank biserial correlation. (B) Sample size estimations were performed for future intervention trials showing different levels of reduction in progression levels for the different outcome measures: SARA, lateral sway and stride length variability in the walking conditions with preferred and slow speed. The estimated number of participants per study arm is plotted over the assumed therapeutic effect for lowering the 1-year progression in SCA3 (reprinted from [25] with permission)
A change over a 1-year follow-up was also identified in a mixed population of degenerative cerebellar disease for turning stability in daily life [38]. A measure that quantifies dynamic balance during turning — lateral velocity change (LVC) — detected of longitudinal change at 1-year follow-up (effect size: rprb=0.66). Larger multicenter longitudinal studies are needed to confirm and extend these findings and to specify gait, balance and turning metrics for more homogeneous ataxia populations [26, 121].
In FRDA, three studies have examined change due to natural disease progression over 12 months [62, 72, 124]. In the largest cohort of 52 participants, mean cadence (effect size SRM = −0.624) and velocity (SRM = −0.641) at fast speed were the most responsive spatiotemporal gait parameters and had larger effect sizes than the FARS, SARA [24] and mFARS [125]. Step width variability appears to be sensitive to FRDA disease progression, including children and those ambulating with and without an aid [62]. Knee extension range during stance also appears sensitive to disease progression in children [70], but it is yet to be seen if these changes continue in adulthood. In contrast to SCA3 [25], stride length variability at a self-selected speed seemed insensitive to disease progression in participants with FRDA [62, 70, 72].
Recommended Consensus Protocols for the Assessment of Digital Gait and Balance Measures in Ataxia
In the following, we propose a digital gait and balance protocol for natural history studies and interventional trials in degenerative ataxias, on the basis of the (i) accumulated evidence on the sensitivity of gait and stance measures for quantifying ataxia (Tables 1 and 2) and (ii) established requirements for performance measures [126,127,128,129,130,131].
In developing such a protocol, it is important to consider that application is essentially confined to those able to stand and walk independently and that: (i) elementary gait and balance tasks may not be sensitive in early stages of ataxia; and, on the other hand, (ii) more complex gait and balance tasks may not be feasible for people with advanced ataxia. Therefore, we have divided the clinical gait and stance assessment protocol into a basic and a more complex part, with the latter which constituting tasks that are only suited for those participants in the earliest stages of the disease.
Basic Protocol
Table 3 summarizes our recommendations for gait and balance assessments applicable to studies in individuals who are still able to stand and walk independently. Tasks with asterisks* indicate a minimum set. Participants should perform gait tasks (normal, comfortable pace and a self-determined ‘slow’ pace for 2 min over a 10-m pathway with 180° turns over a marker on the ground). Turns should be removed from the walking bouts for the gait analysis and analysed separately. Gait instructions and recommended details about the gait protocol can be found in the supplementary information.
Since gait variability is a sensitive and specific marker of ataxic gait, protocols should include at least 40 steps (20 strides) that have been shown to be required to ensure reliable measures of gait variability [132,133,134] and number of steps analysed should be reported along with results. As shorter distances intrinsically increase variability, the length of the walkway should be standardized in multicenter trials [16] [135] and is recommended to be 10 m.
We included slow-paced walking since slow walking has recently been shown to result in larger effect sizes of variability measures compared to natural paces walking for people with cerebellar ataxia [25, 56, 64]. These observations correspond with theoretical work on modelling human gait control, which postulates higher balance challenges with slow walking [136, 137].
However, 1 min of fast walking is recommended for sensory ataxias like Friedreich’s ataxia, as larger long-term effects have been observed compared to normal walking [62].
In general, recommendations of motor task and gait measures differ for individuals with FRDA compared with other hereditary (cerebellar) ataxias due to a number of factors. First, with the early onset of symptoms, on average 10–15 years of age [138], typical neural maturation, such as decreasing gait variability [139], changes in cognitive-gait interference [140] and sensory reweighting for postural strategies [141] impact gait and balance, concurrent with progression of ataxia. Second, time from symptom onset to wheel-chair dependency is only 10–15 years [142] with gait aid use as a common transition within this time. This provides a small window to include only independently-ambulant individuals in clinical studies, thus potentially necessitating the inclusion of participants who are dependent on walking aids (e.g. a cane or a 4-wheeled frame walker). Although the use of any assistive device or touching for stability makes it difficult to accurately interpret postural control in standing or gait variability in walking, the analysis of functional mobility (e.g. mean gait speed, mean length of walking bouts, see Table 4) in the course of disease may be useful for longitudinal studies.
In addition to gait, standing balance should be assessed with 3 stance tasks (30 s each, eyes open and eyes closed with normal stance standardized with a template, as well as eyes open with feet together). Since people with ataxia tend to compensate for their large postural sway by widening their stance, protocols for measuring postural sway should be carefully controlled, settings standardised and stance width determined using a foot template [143]. The template we recommend is based on average, normal stance width in healthy young and elderly adults with 10 cm between the heels and 10° external rotation of the feet (15 cm wide at the toes) [144]. Since postural sway includes frequencies below 1 Hz, a minimum of 30 s of sway needs to be recorded [145].
In addition, we included a standardized turning task for the performance of 90° and 180° turns.
Protocol for Early Disease Stages
The pre-ataxic phase of SCAs before the clinical manifestation of ataxia symptoms [8, 24] provides a promising window for early therapeutic intervention — both pharmaceutical and rehabilitative - before substantial, irreversible neurodegeneration has occurred [8, 146, 147]. Although some studies have identified gait and balance changes in pre-ataxic mutation carriers [22, 23], changes with larger effect sizes are found for more complex tasks such as tandem stance, tandem walking [20, 23, 51] and turning [38].
Thus, we have included more challenging gait and balance tasks for pre-ataxic or very mildly affected participants (Table 4). Participants are asked to perform a tandem walk and two additional stance tasks (eyes closed with feet together and tandem stance with heel touching opposite toe with eyes open).
In addition to the clinical assessments of prescribed gait and balance tasks we also propose continuous monitoring of gait and turning during daily life for 7 days (minimum 5 hours per day) for both pre-ataxic and manifest ataxia (Table 5).
Test-Retest Reliability and Minimal Detectable Change
Studies of gait and balance ataxia should repeat tests at baseline. Useful gait and balance outcomes need to demonstrate stability of measures over time when no change is expected, such as in re-test within a short timeframe. Test-retest reliability can then be calculated using intraclass correlation coefficients (ICCs) [148] and Bland-Altmann Plot with limits of agreement [149, 150].
One way to investigate the reliability (in terms of technical repeatability) of the gait measures is to divide a 2 minute-walk test into two, 1-minute segments, and calculate the split-half reliability of gait measures ICC (2, 1) [148]. As ataxia can show considerable day-to-day fluctuations [151], a more rigorous way to calculate test-retest reliability is to have the participants repeat the test twice, after a period of rest or on another day.
In a study examining gait measures from the 2-minute walk test, the within session test–retest reliability of the most sensitive/specific gait variability measures discriminating participants with SCA from healthy controls was good-to-excellent (0.83–0.9) [21]. Moreover, high test-retest reliability was reached for ankle movement measures in real life by comparing data recorded over days 1–3 and days 4–6 [73].
Based on the test-retest reliability measured by the ICC, the minimal detectable change (MDC) can be determined. MDC indicates the minimum change that falls outside the measurement error and can be statistically detected with some degree of confidence (e.g. 95 or 90%) from a test-retest reliability design [152,153,154].
This value is a fundamental technical metric that indicates the “noise” above which a change can be considered beyond technical error and potentially daily fluctuations. The ability of a measure to detect change over time in this sense has been referred to as internal responsiveness of an outcome [155].
Recommended Recording Technology
While sensitive digital gait measures have been identified using a variety of recording technologies (Reviews in [16, 18, 28, 109]), not all are equally suitable for multicentre clinical trials that need to include centers without a dedicated motion laboratory or specialised technical staff (see Table 6 for comparison of recording technologies in clinical trials). While laboratory-based, optical motion analysis systems remain the gold standard for gait analysis, they are expensive, resource intensive, and largely immobile, which limits their accessibility in clinical settings [135, 156]. Other cost-efficient camera-based systems (e.g. Kinect) often have limitations in the recording space, which reduce the length (≤5 m) of the captured walkway important for gait variability.
Given the described influences of walkway length on step variability measures, it is important to consider the influence of technical restrictions of the equipment on study design, gait protocol and parameter definitions [16, 109].
Wearable IMU sensor technology for quantifying gait and balance has recently become feasible for large, multicenter clinical trials without sophisticated gait laboratories or expert researchers.
These body-worn sensors enable the recording of longer gait distances, are portable and instantly provide gait metrics without post-trial human analysis, making IMUs are easy to use in clinical settings. Moreover, they wearable IMUs allow movement monitoring in daily life [36, 157, 158] (Reviews in [109, 159]). Therefore, wearable sensor technology has been identified as the most appropriate technology at this time to conduct such multicenter studies of digital gait and balance measures in ataxia [127, 160].
However, IMUs require specific algorithms [161,162,163] and technical verification of the accuracy of the hardware, raw signals and software (with the algorithms producing the gait and balance metrics) [161163]. Sensors employed for clinical trials must be validated by comparison with laboratory gold-standards [160]. For example, a study of SCA14 showed good-to-excellent between-methods consistency with APDM’s Opal sensors (using Mobility Lab) and measures of mean gait metrics derived from GAITRite, with ICCs >0.9, except for stride length (ICC=0.84) [33].
The optimal number of sensors in the trade-off between gait data quality and participants burden currently appears to be three: one sensor at the lower lumbar spine, and one sensor on the top of each foot. This configuration has substantial advantages for the quantification of ataxic foot placement characteristics, compared to only one sensor on the lumbar spine, as is widely used for activity-monitoring [117, 164, 165]. First, several measures showing the best sensitivity to ataxia require sensors on the feet (e.g. lateral step deviation and pitch angle at heel strike) [21, 36]. Second, in order to accurately measure step variability (e.g. variability in stride length and stride duration), accurate determination of step events (initial and final foot contacts) is crucial. At least current recording techniques and algorithms using only one IMU or mobile phone at the pelvis show limited accuracy and reliability of gait variability measurements, which is probably low due to inaccurate identification of heel strike and toe-off events [57, 166, 167] (see review in [135]). In addition to the IMUs on the feet, the lumbar sensor provides measures of trunk instability (range of motion and jerkiness of the trunk), as well as measures of turning (turn velocity, duration, etc.). Postural sway in standing can also be measured with an IMU on the lumbar spine, but recent studies suggest that even more sensitive measures of ataxic sway can be obtained from an IMU on the sternum [85, 168].
Future promising developments in recording technology could include the combination of IMUs with pressure sensitive insoles [169] as well as video-based technologies using machine learning methods to generate specific movement features (see for review [170]).
Recommended Inclusion/Exclusion Criteria
The following inclusion criteria for establishing digital balance and gait metrics are recommended as follows: (1) genetically confirmed hereditary cerebellar ataxia; (2) degenerative cerebellar ataxia in the absence of any unrelated signs of other CNS disease; (3) age between 18 and 80 years for SCAs. For Friedreich’s ataxia, younger participants are required due to the earlier onset: age between 8 and 65 years (see [62]); (4) able to walk for 2 min and stand for 30 s with eyes open, without walking aids. The exclusion criteria should include the following: (1) severe visual or hearing disturbances, (2) cognitive impairment limiting ability to follow protocol instructions, (3) orthopaedic or unrelated neurological constraints affecting standing and walking, and (4) drug or alcohol history which related to ataxia.
If participants who require walking aids for the 2-minute walk are recruited, they must be analysed separately, and the use of an assistive device needs to be documented. Natural history studies should also include age-and sex-matched healthy control subjects without known neurological or musculoskeletal impairments that affect balance or walking.
Recommended Clinical Assessments and Patient-Reported Outcomes
Every natural history study of ataxic gait and balance should include clinical measures of ataxia symptoms, such as the SARA [24] for SCA, mFARs [125] for Friedreich’s Ataxia; the INAS for other non-ataxia symptoms, and patient-reported outcomes including the new PROM-Ataxia [39], EQ-5 [45], and the Activities-specific Balance Confidence (ABC) Scale [107] (See Table 7). The most sensitive measures of gait and balance should then be correlated with clinical assessment of severity of ataxia [24] for Concurrent Validity (see Figs. 1, 2, 4) as well as with patient-reported scales for meaningfulness to people with ataxia [39, 107] (see also the AGI Consensus Recommendations for Clinical Outcome Assessments [171]).
Avoiding falls is also meaningful to people with ataxia [31, 172] so longitudinal studies should include prospective monitoring of falls over time using recommended guidelines [173, 174]. Falls can be defined as a “sudden, unintended contact with the ground” and should be queried 1–2 times per month via email or texts for accurate recall, with phone follow-ups if a fall occurred or if the queries are not answered. Gait and balance measures that predict falls or separate fallers from non-fallers can help determine which measures are meaningful to people with ataxia.
To quantify progression and treatment responses, gait and balance measures should capture longitudinal changes that correspond to minimal clinically important differences (MCID) [175] and functional changes in patient-centred outcome measures [1, 160, 176]. MCID for how much a balance or gait measure needs to change for a person with ataxia to perceive a small difference can be captured by asking them to rate their change in balance or gait (after longitudinal progression or treatment) on a 7-point Likert scale [177, 178] (Patient global impression of change (PGI-C), see Table 7) [179]. The Likert scale has 3 points indicating worse balance/gait and 3 points indicating better balance/gait with 0 indicating no change. A MCID would correspond to a patient reporting a +1 or −1 on the Likert Scale [177]. Of course, difficulties of recollection of one’s own balance and gait impairments in the past 6–12 months is a limitation of this approach. By concept, the choice of MCID anchor may differ between contexts of use [179].
Regulatory Considerations for Digital Gait and Balance Outcomes for Clinical Trials
The regulatory pathway for including endpoints derived from body-worn sensors varies across regulatory agencies.
The FDA has a program for Clinical Outcome Assessments (COAs), that are the most suitable for gait and balance quantification for clinical trials and that are not specific to a single, individual drug development program [131, 179]. COAs are defined as measures that reflect how a person feels, functions, or survives. The FDA has specified five different types of COAs. Measures from body-worn sensors during prescribed tasks, such as walking and standing, would be considered as performance outcomes (PerfO). Many criteria are considered as part of the qualification process including those described here, such as reliability, validity, and meaningfulness [160] .
In particular, trial endpoints must first be based on a meaningful aspect of health, such as ability to perform ambulatory activities, from which various concepts of interests, such as walking and balance cascade. Figure 10 illustrates examples of these concepts [182].
Examples of how a variety of concepts of interest cascade from a single meaningful aspect of health across select conditions and clinical populations (adapted from [182])
As described above, it is well known that impairments in gait and balance due to ataxia are some of the most important symptoms, as they have a direct impact upon the quality of life of people with ataxia [40,41,42] and their ability to perform normal daily activities independently [12, 13]. While there are now patient-oriented measures that address gait and balance impairments in daily life (PROM-Ataxia, ABC-score [39, 107]), there is yet no study showing their longitudinal sensitivity. When such studies have been completed, changes in gait and balance measures should be interpreted against changes in patient-reported outcomes. Moreover, gait and balance measures, and especially how they change over time, need to be linked to meaningfulness in different stages of the disease.
One possible approach for such a link is to establish Meaningful Score Regions (MSR) or Meaningful Score Changes (MSC) for gait and stance metrics by relating them to different levels of gait-related Patient Global Impression of Severity or Patient Global Impression of Change (see Table 7) [179]. However, it is not yet exactly clear, which specific aspects of functional mobility should be rated relative to specific gait and balance metrics: e.g.: mobility, walking quality, postural stability, unsteadiness, fear of falling, gait variability.
Another issue that has not yet been fully resolved arises from the problem that while a discrete increase in gait variability does not noticeably affect a person with ataxia in the early stages of the disease, a gradual increase in gait variability leads to a much higher risk of falls later in the course of the disease [31, 183]. Thus, a therapy-induced reduction in gait variability in the earlier stages of the disease may have a significant impact on later disease progression and thereby prolonging independent ambulation.
Therefore, evidence is needed on what changes in gait and balance performance measures are meaningful for people with ataxia not only in their current condition, but also as the disease progresses over time.
Summary
The tasks recommended in this consensus protocol are strongly associated with typical daily activities that people perform regularly. Standing, walking and turning are performed throughout the day and are critical functional tasks in daily life. These are also activities that are impaired early in ataxia and are frequently identified as the most disabling and thereby meaningful and relevant to daily functioning.
Digital gait and standing balance measures represent promising endpoints for upcoming interventional trials. Our consensus recommendations to allow multisite pooling of natural history data on gait and balance impairments for ataxia include:
a) Protocols: Include a minimum of a 2-minute walk (use a walkway with the standardized length of 10 meters) and a 30-second standing task with additional conditions or greater challenge for pre-ataxic ataxia;
b) Recording Technology: 3 body-worn inertial (IMU) sensors (one at the pelvis or sternum and two on the feet);
c) Sensitivity/Specificity: Calculate AUC of ROC curves to identify gait and balance measures that best separate individuals with ataxia from age-matched controls;
d) Test-retest reliability: Show test-retest reliability and calculate a Minimal Detectable Change (MDC);
e) Meaningfulness: Calculate Minimal Clinically Important Change MCID for sensitive digital measures by including a patient-reported scale of perceived change;
f) Concurrent Validity: Include standard neurological scales of severity (e.g. SARA or mFARS);
g) Longitudinal assessment of natural course: Test participants every 6 months for 2 years. Demonstrate longitudinal changes over a reasonable study period (e.g. within 1–2 years) to enable sample size estimation for future clinical trials.
h) Daily life: monitoring of walking behavior over 7 days of daily life (for a minimum of 5 h daily), with the same IMU system.
Based on our current knowledge, the same gait and balance digital outcomes can be used for a range of cerebellar ataxias, with the most evidence currently available on the most common SCA including 1, 2, 3 and 6 (see Fig. 1B and [21]). In Friedreich’s ataxia, the gait measures with the greatest sensitivity to longitudinal changes may vary somewhat due to the predominance of sensory ataxia, the younger age range during adolescence, and the more rapid dependence on walking aids.
In general, the most sensitive tasks and measures will likely depend primarily on the disease stage of the targeted trial population. More complex movement tasks, such as tandem walking, tandem stance or eyes-closed stance are proposed for pre-ataxic and early ataxic participants. Gait variability over a natural-pace 2-minute walk and postural sway area during a feet-together, eyes-open stance for 30 s are currently the most promising outcomes for prescribed tasks in the clinic/laboratory. Although active monitoring of prescribed gait and standing tasks currently provide the most reliable data, daily life monitoring holds great promise for providing even more meaningful measures of actual functional mobility. The goal is to identify the most sensitive gait and balance measures, or a composite measure, that has a larger effect size than current clinical scales to detect disease progression so that clinical trials for these rare diseases could be conducted with smaller cohorts.
Recommendations for Further Studies
Existing studies have provided significant evidence that digital gait and balance measures can be sensitive performance markers for ataxia with excellent reliability and validity characteristics. In addition, ataxia-related gait changes are related to meaningful aspects of health, e.g. a high risk of falls is associated with increased gait variability and increased postural sway. However, we need to establish the meaningful score difference for digital gait and balance outcomes by relating them to patient global impression of severity (PGI-S) or change (PGI-C) [178, 179]. Furthermore, future studies should compare active versus passive monitoring of gait as useful outcomes for ataxia, as well as explore whether reliable measures of gait can be obtained from individuals using walking aids.
There is a consensus that a large, international effort to collect digital balance and gait measures longitudinally is necessary (such as performed in other neurodegenerative diseases like Parkinson’s disease [184]). This data, and in general as much data as possible collected through the proposed consensus protocol, should be openly available to support the further development of digital gait and balance outcomes for ataxia clinical trials.
References
Ashizawa T, Oz G, Paulson HL. Spinocerebellar ataxias: prospects and challenges for therapy development. Nat Rev Neurol. 2018;14(10):590–605.
Klockgether T, Mariotti C, Paulson HL. Spinocerebellar ataxia. Nat Rev Dis Primers. 2019;5(1):24.
Scoles DR, Pulst SM. Antisense therapies for movement disorders. Mov Disord. 2019;34(8):1112–9.
Coarelli G, Wirth T, Tranchant C, Koenig M, Durr A, Anheim M. The inherited cerebellar ataxias: an update. J Neurol. 2023;270(1):208–22.
Klockgether T, Ashizawa T, Brais B, et al. Paving the way toward meaningful trials in ataxias: an ataxia global initiative perspective. Mov Disord. 2022;37(6):1125–30.
Saute JAM, Jardim LB. Planning future clinical trials for Machado-Joseph disease. In: Nóbrega C, Pereira de Almeida L, editors. Polyglutamine disorders. Cham: Springer International Publishing; 2018. p. 321–48.
Jacobi H, du Montcel ST, Bauer P, et al. Long-term disease progression in spinocerebellar ataxia types 1, 2, 3, and 6: a longitudinal cohort study. Lancet Neurol. 2015;14(11):1101–8.
Maas RP, van Gaalen J, Klockgether T, van de Warrenburg BP. The preclinical stage of spinocerebellar ataxias. Neurology. 2015;85(1):96–103.
Globas C, du Montcel ST, Baliko L, et al. Early symptoms in spinocerebellar ataxia type 1, 2, 3, and 6. Mov Disord. 2008;23(15):2232–8.
Ilg W, Branscheidt M, Butala A, et al. Consensus paper: neurophysiological assessments of ataxias in daily practice. Cerebellum. 2018;17(5):628–53.
Luo L, Wang J, Lo RY, et al. The initial symptom and motor progression in Spinocerebellar ataxias. Cerebellum. 2017;16(3):615–22.
National Ataxia Foundation (NAF). The voice of the patient: Friedreich’s ataxia. Summary report resulting from an externally led patient-focused drug development meeting. Availlable from: https://www.ataxia.org/ataxiapfdd/2017.
Rosen A, Hagen S, Trace K, Compton A. The voice of the patient: living with polglutamine spinocerebellar ataxias (SCA) and dentatorubal-pallidoluysion atrophy (DRPLA). https://www.ataxia.org/ataxiapfdd/: National Ataxia Foundation 2021.
Seabury J, Alexandrou D, Dilek N, et al. Patient-reported impact of symptoms in Friedreich ataxia. Neurology. 2023;100(8):e808–21. https://doi.org/10.1212/WNL.0000000000201598.
Lowit A, Greenfield J, Cutting E, Wallis R, Hadjivassiliou M. Symptom burden of people with progressive ataxia, and its wider impact on their friends and relatives: a cross-sectional study AMRC. Health Open Res. 2023;3:28. https://doi.org/10.12688/amrcopenres.13036.2.
Buckley E, Mazza C, McNeill A. A systematic review of the gait characteristics associated with cerebellar ataxia. Gait & posture. 2018;60:154–63.
Milne SC, Murphy A, Georgiou-Karistianis N, Yiu EM, Delatycki MB, Corben LA. Psychometric properties of outcome measures evaluating decline in gait in cerebellar ataxia: a systematic review. Gait & posture. 2018;61:149–62.
Ilg W, Timmann D. Gait ataxia--specific cerebellar influences and their rehabilitation. Mov Disord. 2013;28(11):1566–75.
Marquer A, Barbieri G, Perennou D. The assessment and treatment of postural disorders in cerebellar ataxia: a systematic review. Ann Phys Rehabil Med. 2014;57(2):67–78.
Ilg W, Fleszar Z, Schatton C, et al. Individual changes in preclinical spinocerebellar ataxia identified via increased motor complexity. Mov Disord. 2016;31(12):1891–900.
Shah VV, Rodriguez-Labrada R, Horak FB, et al. Gait variability in spinocerebellar ataxia assessed using wearable inertial sensors. Mov Disord. 2021;36(12):2922–31.
Rochester L, Galna B, Lord S, Mhiripiri D, Eglon G, Chinnery PF. Gait impairment precedes clinical symptoms in spinocerebellar ataxia type 6. Mov Disord. 2014;29(2):252–5.
Velazquez-Perez L, Rodriguez-Labrada R, Gonzalez-Garces Y, et al. Prodromal spinocerebellar ataxia type 2 subjects have quantifiable gait and postural sway deficits. Mov Disord. 2021;36(2):471–80.
Schmitz-Hubsch T, du Montcel ST, Baliko L, et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology. 2006;66(11):1717–20.
Ilg W, Muller B, Faber J, et al. Digital gait biomarkers allow to capture 1-year longitudinal change in spinocerebellar ataxia type 3. Mov Disord. 2022;37(11):2295–301.
Johnson M, Gomez C, Horak F. IDEA study 2019.
Paap BK, Roeske S, Durr A, et al. Standardized assessment of hereditary ataxia patients in clinical studies. Mov Disord Clin Pract. 2016;3(3):230–40.
Ngo T, Pathirana PN, Horne MK, Corben LA, Harding IH, Szmulewicz DJ. Technological evolution in the instrumentation of ataxia severity measurement. IEEE Access. 2023;11:14006–27. https://doi.org/10.1109/ACCESS.2023.3243178.
Ilg W, Christensen A, Mueller OM, Goericke SL, Giese MA, Timmann D. Effects of cerebellar lesions on working memory interacting with motor tasks of different complexities. J Neurophysiol. 2013;110(10):2337–49.
Ilg W, Schatton C, Schicks J, Giese MA, Schols L, Synofzik M. Video game-based coordinative training improves ataxia in children with degenerative ataxia. Neurology. 2012;79(20):2056–60.
Schniepp R, Wuehr M, Schlick C, et al. Increased gait variability is associated with the history of falls in patients with cerebellar ataxia. J Neurol. 2014;261(1):213–23.
Schniepp R, Wuehr M, Neuhaeusser M, et al. Locomotion speed determines gait variability in cerebellar ataxia and vestibular failure. Mov Disord. 2012;27(1):125–31.
Schmitz-Hubsch T, Brandt AU, Pfueller C, et al. Accuracy and repeatability of two methods of gait analysis - GaitRite und Mobility Lab - in subjects with cerebellar ataxia. Gait & posture. 2016;48:194–201.
Summa S, Tartarisco G, Favetta M, et al. Validation of low-cost system for gait assessment in children with ataxia. Comput Methods Programs Biomed. 2020;196:105705.
Muller B, Ilg W, Giese MA, Ludolph N. Validation of enhanced kinect sensor based motion capturing for gait assessment. PLoS One. 2017;12(4):e0175813.
Ilg W, Seemann J, Giese M, et al. Real-life gait assessment in degenerative cerebellar ataxia: toward ecologically valid biomarkers. Neurology. 2020;95(9):e1199–210.
Zhou H, Nguyen H, Enriquez A, et al. Assessment of gait and balance impairment in people with spinocerebellar ataxia using wearable sensors. Neurol Sci. 2022;43(4):2589–99.
Thierfelder A, Seemann J, John N, et al. Real-life turning movements capture subtle longitudinal and preataxic changes in cerebellar ataxia. Mov Disord. 2022;37(5):1047–58.
Schmahmann JD, Pierce S, MacMore J, L'Italien GJ. Development and validation of a patient-reported outcome measure of ataxia. Mov Disord. 2021;36(10):2367–77.
Schmitz-Hubsch T, Coudert M, Giunti P, et al. Self-rated health status in spinocerebellar ataxia--results from a European multicenter study. Mov Disord. 2010;25(5):587–95.
Joyce MR, Nadkarni PA, Kronemer SI, et al. Quality of life changes following the onset of cerebellar ataxia: symptoms and concerns self-reported by ataxia patients and informants. Cerebellum. 2022;21(4):592–605.
Bolzan G, Leotti VB, de Oliveira CM, et al. Quality of life since pre-ataxic phases of spinocerebellar ataxia type 3/Machado-Joseph disease. Cerebellum. 2022;21(2):297–305.
Jacobi H, du Montcel ST, Bauer P, et al. Long-term evolution of patient-reported outcome measures in spinocerebellar ataxias. J neurol. 2018;265(9):2040–51.
Maas R, Helmich RCG, van de Warrenburg BPC. The role of the cerebellum in degenerative ataxias and essential tremor: insights from noninvasive modulation of cerebellar activity. Mov Disord. 2020;35(2):215–27.
EuroQol G. EuroQol--a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208.
Holmes G. The cerebellum of man. Brain. 1939;62:1–30.
Hallett M, Massaquoi SG. Physiologic studies of dysmetria in patients with cerebellar deficits. Can J Neurol Sci. 1993;20(Suppl 3):S83–92.
Diener HC, Dichgans J. Cerebellar and spinocerebellar gait disorders. In: Bronstein AM, Brandt T, Woollacott, eds. Clinical disorders of posture and gait. first ed. London: Arnold, 1996:147–155.
Morton SM, Bastian AJ. Cerebellar control of balance and locomotion. Neuroscientist. 2004;10(3):247–59.
Serrao M, Pierelli F, Ranavolo A, et al. Gait pattern in inherited cerebellar ataxias. Cerebellum. 2012;11(1):194–211.
Cabaraux P, Agrawal SK, Cai H, et al. Consensus paper: ataxic gait. Cerebellum. 2023;22(3):394–430. https://doi.org/10.1007/s12311-022-01373-9.
Trouillas P, Takayanagi T, Hallett M, et al. International Cooperative Ataxia Rating Scale for pharmacological assessment of the cerebellar syndrome. The Ataxia Neuropharmacology Committee of the World Federation of Neurology. J Neurol Sci. 1997;145(2):205–11.
Schmahmann JD, Gardner R, MacMore J, Vangel MG. Development of a brief ataxia rating scale (BARS) based on a modified form of the ICARS. Mov Disord. 2009;24(12):1820–8.
Lynch DR, Farmer JM, Tsou AY, et al. Measuring Friedreich ataxia: complementary features of examination and performance measures. Neurology. 2006;66(11):1711–6.
Ilg W, Golla H, Thier P, Giese MA. Specific influences of cerebellar dysfunctions on gait. Brain. 2007;130(Pt 3):786–98.
Wuehr M, Schniepp R, Ilmberger J, Brandt T, Jahn K. Speed-dependent temporospatial gait variability and long-range correlations in cerebellar ataxia. Gait & posture. 2013;37(2):214–8.
Hickey A, Gunn E, Alcock L, et al. Validity of a wearable accelerometer to quantify gait in spinocerebellar ataxia type 6. Physiol Meas. 2016;37(11):N105–17.
Milne SC, Hocking DR, Georgiou-Karistianis N, Murphy A, Delatycki MB, Corben LA. Sensitivity of spatiotemporal gait parameters in measuring disease severity in Friedreich ataxia. Cerebellum. 2014;13(6):677–88.
Palliyath S, Hallett M, Thomas SL, Lebiedowska MK. Gait in patients with cerebellar ataxia. Mov Disord. 1998;13(6):958–64.
Morton SM, Bastian AJ. Relative contributions of balance and voluntary leg-coordination deficits to cerebellar gait ataxia. J Neurophysiol. 2003;89(4):1844–56.
Serrao M, Chini G, Casali C, et al. Progression of gait ataxia in patients with degenerative cerebellar disorders: a 4-year follow-up study. Cerebellum. 2017;16(3):629–37.
Milne SC, Kim SH, Murphy A, et al. The responsiveness of gait and balance outcomes to disease progression in Friedreich ataxia. Cerebellum. 2022;21(6):963–75. https://doi.org/10.1007/s12311-021-01348-2.
Terayama K, Sakakibara R, Ogawa A. Wearable gait sensors to measure ataxia due to spinocerebellar degeneration. Neurol Clin Neurosci. 2018;6(1):9–12.
Phan D, Nguyen N, Pathirana PN, Horne M, Power L, Szmulewicz D. Quantitative assessment of ataxic gait using inertial sensing at different walking speeds. Annu Int Conf IEEE Eng Med Biol Soc. 2019;2019:4600–3.
Shirai S, Yabe I, Takahashi-Iwata I, et al. The responsiveness of triaxial accelerometer measurement of gait ataxia is higher than that of the scale for the assessment and rating of ataxia in the early stages of spinocerebellar degeneration. Cerebellum. 2019;18(4):721–30.
Castiglia SF, Trabassi D, Tatarelli A, et al. Identification of gait unbalance and fallers among subjects with cerebellar ataxia by a set of trunk acceleration-derived indices of gait. Cerebellum. 2023;22(1):46–58.
Lee J, Oubre B, Daneault JF, et al. Analysis of gait sub-movements to estimate ataxia severity using ankle inertial data. IEEE Trans Biomed Eng. 2022;69(7):2314–23.
Gouelle A, Norman S, Sharot B, Salabarria S, Subramony S, Corti M. Gauging gait disorders with a method inspired by motor control theories: a pilot study in Friedreich’s ataxia. Sensors. 2021;21(4):1144. https://doi.org/10.3390/s21041144.
Kadirvelu B, Gavriel C, Nageshwaran S, et al. A wearable motion capture suit and machine learning predict disease progression in Friedreich’s ataxia. Nat Med. 2023;29(1):86–94.
Vasco G, Gazzellini S, Petrarca M, et al. Functional and gait assessment in children and adolescents affected by Friedreich’s ataxia: a one-year longitudinal study. PLoS One. 2016;11(9):e0162463.
Stephenson J, Zesiewicz T, Gooch C, et al. Gait and balance in adults with Friedreich’s ataxia. Gait & posture. 2015;41(2):603–7.
Zesiewicz TA, Stephenson JB, Kim SH, et al. Longitudinal gait and balance decline in Friedreich’s ataxia: a pilot study. Gait & posture. 2017;55:25–30.
Eklund NM, Ouillon J, Pandey V, et al. Real-life ankle submovements and computer mouse use reflect patient-reported function in adult ataxias. Brain. Communications. 2023;5(2):fcad064. https://doi.org/10.1093/braincomms/fcad064.
Caliandro P, Conte C, Iacovelli C, et al. Exploring risk of falls and dynamic unbalance in cerebellar ataxia by inertial sensor assessment. Sensors. 2019;19(24):5571. https://doi.org/10.3390/s19245571.
Schlick C, Rasoul A, Wuehr M, et al. Gait variability predicts a subset of falls in cerebellar gait disorders. J neurol. 2017;264(11):2322–4.
Mueller A, Paterson E, McIntosh A, et al. Digital endpoints for self-administered home-based functional assessment in pediatric Friedreich’s ataxia. Ann Clin Transl Neurol. 2021;8(9):1845–56.
Peterka RJ. Sensorimotor integration in human postural control. J Neurophysiol. 2002;88(3):1097–118.
Bunn LM, Marsden JF, Giunti P, Day BL. Stance instability in spinocerebellar ataxia type 6. Mov Disord. 2013;28(4):510–6.
Dichgans J, Diener HC, Mauritz KH. What distinguishes the different kinds of postural ataxia in patients with cerebellar diseases. Adv Otorhinolaryngol. 1983;30:285–7.
Diener HC, Dichgans J. Pathophysiology of cerebellar ataxia. Mov Disord. 1992;7(2):95–109.
Diener HC, Dichgans J, Bacher M, Gompf B. Quantification of postural sway in normals and patients with cerebellar diseases. Electroencephalogr Clin Neurophysiol. 1984;57(2):134–42.
Schwabova J, Zahalka F, Komarek V, et al. Uses of the postural stability test for differential diagnosis of hereditary ataxias. J Neurol Sci. 2012;316(1):79–85.
Dichgans J, Diener HC. Clinical evidence for functional compartmentalization of the cerebellum. In: Bloedel JR, Dichgans J, Precht W, editors. Cerebellar Functions. Berlin: Springer Verlag; 1984. p. 126–47.
Mancini M, Salarian A, Carlson-Kuhta P, et al. ISway: a sensitive, valid and reliable measure of postural control. J Neuroeng Rehabil. 2012;9:59.
Shah VV, McNames J, Casey H, et al. Digital sway measures for spinocerebellar ataxia. Mov Disord. 2022;37(S2):S227.
Asahina M, Nakajima M, Kojima S, Hirayama K. Postural sway in patients with hereditary ataxia. Rinsho Shinkeigaku. 1994;34(11):1105–10.
Gatev P, Thomas S, Lou JS, Lim M, Hallett M. Effects of diminished and conflicting sensory information on balance in patients with cerebellar deficits. Mov Disord. 1996;11(6):654–64.
Van de Warrenburg BP, Bakker M, Kremer BP, Bloem BR, Allum JH. Trunk sway in patients with spinocerebellar ataxia. Mov Disord. 2005;20(8):1006–13.
Matsushima A, Yoshida K, Genno H, et al. Clinical assessment of standing and gait in ataxic patients using a triaxial accelerometer. Cerebellum Ataxias. 2015;2:9.
Nanetti L, Alpini D, Mattei V, et al. Stance instability in preclinical SCA1 mutation carriers: a 4-year prospective posturography study. Gait & posture. 2017;57:11–4.
Fleszar Z, Mellone S, Giese M, et al. Real-time use of audio-biofeedback can improve postural sway in patients with degenerative ataxia. Ann Clin Transl Neurol. 2019;6(2):285–94.
Nguyen N, Phan D, Pathirana PN, Horne M, Power L, Szmulewicz D. Quantification of axial abnormality due to cerebellar ataxia with inertial measurements. Sensors. 2018;18(9):2791. https://doi.org/10.3390/s18092791.
Liu XH, Li Y, Xu HL, et al. Quantitative assessment of postural instability in spinocerebellar ataxia type 3 patients. Ann Clin Transl Neurol. 2020;7(8):1360–70.
Galvao AF, Lemos T, Martins CP, Horsczaruk CHR, Oliveira LAS, Ferreira AS. Body sway and movement strategies for control of postural stability in people with spinocerebellar ataxia type 3: a cross-sectional study. Clin Biomech (Bristol, Avon). 2022;97:105711.
Ngo T, Pathirana PN, Horne MK, et al. Balance deficits due to cerebellar ataxia: a machine learning and cloud-based approach. IEEE Trans Biomed Eng. 2021;68(5):1507–17.
Velazquez-Perez L, Sanchez-Cruz G, Rodriguez-Labrada R, Velazquez-Manresa M, Hechavarria-Pupo R, Almaguer-Mederos LE. Postural instability in prodromal spinocerebellar ataxia type 2: insights into cerebellar involvement before onset of permanent ataxia. Cerebellum. 2017;16(1):279–81.
Tezenas du Montcel S, Durr A, Rakowicz M, et al. Prediction of the age at onset in spinocerebellar ataxia type 1, 2, 3 and 6. J Med Genet. 2014;51(7):479–86.
Khasnis A, Gokula RM. Romberg’s test. J Postgrad Med. 2003;49(2):169–72.
Stolze H, Klebe S, Petersen G, et al. Typical features of cerebellar ataxic gait. J Neurol Neurosurg Psychiatry. 2002;73(3):310–2.
Glaister BC, Bernatz GC, Klute GK, Orendurff MS. Video task analysis of turning during activities of daily living. Gait & posture. 2007;25(2):289–94.
Stack E, Ashburn A. Fall events described by people with Parkinson’s disease: implications for clinical interviewing and the research agenda. Physiother Res Int. 1999;4(3):190–200.
Patla AE, Adkin A, Ballard T. Online steering: coordination and control of body center of mass, head and body reorientation. Exp Brain Res. 1999;129(4):629–34.
Weerdesteyn V, Hollands KL, Hollands MA. Gait adaptability. Handb Clin Neurol. 2018;159:135–46.
Fino PC, Horak FB, Curtze C. Inertial sensor-based centripetal acceleration as a correlate for lateral margin of stability during walking and turning. IEEE Trans Neural Syst Rehabil Eng. 2020;28(3):629–36.
Xu D, Carlton LG, Rosengren KS. Anticipatory postural adjustments for altering direction during walking. J Mot Behav. 2004;36(3):316–26.
Hase K, Stein RB. Turning strategies during human walking. J Neurophysiol. 1999;81(6):2914–22.
Powell LE, Myers AM. The activities-specific balance confidence (ABC) scale. J Gerontol A Biol Sci Med Sci. 1995;50A(1):M28–34.
Geh CL, Beauchamp MR, Crocker PR, Carpenter MG. Assessed and distressed: white-coat effects on clinical balance performance. J Psychosom Res. 2011;70(1):45–51.
Buckley C, Alcock L, McArdle R, et al. The role of movement analysis in diagnosing and monitoring neurodegenerative conditions: insights from gait and postural control. Brain Sci. 2019;9(2):34. https://doi.org/10.3390/brainsci9020034.
Tamburini P, Storm F, Buckley C, Bisi MC, Stagni R, Mazza C. Moving from laboratory to real life conditions: influence on the assessment of variability and stability of gait. Gait & posture. 2018;59:248–52.
Meyer BM, Depetrillo P, Franco J, et al. How much data is enough? A reliable methodology to examine long-term wearable data acquisition in gait and postural sway. Sensors. 2022;22(18):6982. https://doi.org/10.3390/s22186982.
Del Din S, Galna B, Godfrey A, et al. Analysis of free-living gait in older adults with and without Parkinson’s disease and with and without a history of falls: identifying generic and disease-specific characteristics. J Gerontol A Biol Sci Med Sci. 2019;74(4):500–6.
Mc Ardle R, Morris R, Hickey A, et al. Gait in mild Alzheimer’s disease: feasibility of multi-center measurement in the clinic and home with body-worn sensors: a pilot study. J Alzheimers Dis. 2018;63(1):331–41.
Roth N, Wieland GP, Küderle A, et al. Do we walk differently at home? A context-aware gait analysis system in continuous real-world environments. 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC); 2021. 1932-1935.
Shah VV, McNames J, Harker G, et al. Effect of bout length on gait measures in people with and without Parkinson’s disease during daily life. Sensors. 2020;20(20).
Shah VV, McNames J, Mancini M, et al. Laboratory versus daily life gait characteristics in patients with multiple sclerosis, Parkinson’s disease, and matched controls. J Neuroeng Rehabil. 2020;17(1):159.
Del Din S, Godfrey A, Galna B, Lord S, Rochester L. Free-living gait characteristics in ageing and Parkinson's disease: impact of environment and ambulatory bout length. J Neuroeng Rehabil. 2016;13(1):46.
Storm FA, Nair KPS, Clarke AJ, Van der Meulen JM, Mazza C. Free-living and laboratory gait characteristics in patients with multiple sclerosis. PLoS One. 2018;13(5):e0196463.
Celik Y, Stuart S, Woo WL, Godfrey A. Wearable inertial gait algorithms: impact of wear location and environment in healthy and Parkinson’s populations. Sensors. 2021;21(19):6476. https://doi.org/10.3390/s21196476.
Carcreff L, Gerber CN, Paraschiv-Ionescu A, et al. Comparison of gait characteristics between clinical and daily life settings in children with cerebral palsy. Sci Rep. 2020;10(1):2091.
Seemann J, Giese M, Schoels L, Synofzik M, Ilg W. Context-matched gait measures capture longitudinal change in real life in Cerebellar Ataxia. International Symposium on Posture and Gait Research 2022; Montreal, CA. Available at https://ispgr.org/wp-content/uploads/2022/06/ISPGR_Abstracts_June21.pdf.
Janse RJ, Hoekstra T, Jager KJ, et al. Conducting correlation analysis: important limitations and pitfalls. Clin Kidney J. 2021;14(11):2332–7.
Morton SM, Tseng YW, Zackowski KM, Daline JR, Bastian AJ. Longitudinal tracking of gait and balance impairments in cerebellar disease. Mov Disord. 2010;25(12):1944–52.
Summa S, Schirinzi T, Bernava GM, et al. Development of SaraHome: a novel, well-accepted, technology-based assessment tool for patients with ataxia. Comput Methods Programs Biomed. 2020;188:105257.
Rummey C, Corben LA, Delatycki MB, et al. Psychometric properties of the Friedreich Ataxia Rating Scale. Neurol Genet. 2019;5(6):371.
Goldsack JC, Coravos A, Bakker JP, et al. Verification, analytical validation, and clinical validation (V3): the foundation of determining fit-for-purpose for Biometric Monitoring Technologies (BioMeTs). NPJ Digit Med. 2020;3:55.
Walton MK, Cappelleri JC, Byrom B, et al. Considerations for development of an evidence dossier to support the use of mobile sensor technology for clinical outcome assessments in clinical trials. Contemp Clin Trials. 2020;91:105962.
Badawy R, Hameed F, Bataille L, et al. Metadata concepts for advancing the use of digital health technologies in clinical research. Digit Biomark. 2019;3(3):116–32.
Stephenson D, Alexander R, Aggarwal V, et al. Precompetitive consensus building to facilitate the use of digital health technologies to support Parkinson disease drug development through regulatory science. Digit Biomark. 2020;4(1):28–49.
Manta C, Mahadevan N, Bakker J, et al. EVIDENCE publication checklist for studies evaluating connected sensor technologies: explanation and elaboration. Digit Biomark. 2021;5(2):127–47.
US Food and Drug Administration (FDA). Digital Health Technologies for remote Data Acquisition in Clinical Investigations: Guidance for Industry, Investigators, and Other Stakeholders. Available from https://www.fda.gov/media/155022/download: Food and Drug Administration, U.S. Department of Health and Human Services; 2021. FDA-2021-D-1128.
Riva F, Bisi MC, Stagni R. Gait variability and stability measures: minimum number of strides and within-session reliability. Comput Biol Med. 2014;50:9–13.
Rennie L, Lofgren N, Moe-Nilssen R, Opheim A, Dietrichs E, Franzen E. The reliability of gait variability measures for individuals with Parkinson's disease and healthy older adults - the effect of gait speed. Gait & posture. 2018;62:505–9.
Kroneberg D, Elshehabi M, Meyer AC, et al. Less Is More - Estimation of the number of strides required to assess gait variability in spatially confined settings. Front Aging Neurosci. 2018;10:435.
Kobsar D, Charlton JM, Tse CTF, et al. Validity and reliability of wearable inertial sensors in healthy adult walking: a systematic review and meta-analysis. J Neuroeng Rehabil. 2020;17(1):62.
Firmani F, Park EJ. Theoretical analysis of the state of balance in bipedal walking. J Biomech Eng. 2013;135(4):041003.
Jansen K, De Groote F, Duysens J, Jonkers I. How gravity and muscle action control mediolateral center of mass excursion during slow walking: a simulation study. Gait & posture. 2014;39(1):91–7.
Bidichandani SI, Delatycki MB. Friedreich Ataxia. In: Adam MP, Pagon RA, Bird TD, Dolan CR, Stephens K, editors. GeneReviewsTM. Seattle: University of Washington; 2017.
Hausdorff JM, Zemany L, Peng C, Goldberger AL. Maturation of gait dynamics: stride-to-stride variability and its temporal organization in children. J Appl Physiol. 1999;86(3):1040–7.
Kraan CM, Tan AHJ, Cornish KM. The developmental dynamics of gait maturation with a focus on spatiotemporal measures. Gait & posture. 2017;51:208–17.
Mallau S, Vaugoyeau M, Assaiante C. Postural strategies and sensory integration: no turning point between childhood and adolescence. PLoS One. 2010;5(9). https://doi.org/10.1371/journal.pone.0013078.
Pandolfo M. Friedreich ataxia: the clinical picture. J neurol. 2009;256(Suppl 1):3–8.
Chiari L, Rocchi L, Cappello A. Stabilometric parameters are affected by anthropometry and foot placement. Clin Biomech (Bristol, Avon). 2002;17(9-10):666–77.
McIlroy WE, Maki BE. Preferred placement of the feet during quiet stance: development of a standardized foot placement for balance testing. Clin Biomech (Bristol, Avon). 1997;12(1):66–70.
Peterka RJ, Loughlin PJ. Dynamic regulation of sensorimotor integration in human postural control. J Neurophysiol. 2004;91(1):410–23.
Jacobi H, Reetz K, du Montcel ST, et al. Biological and clinical characteristics of individuals at risk for spinocerebellar ataxia types 1, 2, 3, and 6 in the longitudinal RISCA study: analysis of baseline data. Lancet Neurol. 2013;12(7):650–8.
Ghanekar SD, Kuo SH, Staffetti JS, Zesiewicz TA. Current and emerging treatment modalities for spinocerebellar ataxias. Expert Rev Neurother. 2022;22(2):101–14.
Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15(2):155–63.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1(8476):307–10.
Berchtold A. Test–retest: agreement or reliability? Methodol Innov. 2016;9:2059799116672875.
Grobe-Einsler M, Taheri Amin A, Faber J, et al. Development of SARA(home), a new video-based tool for the assessment of ataxia at home. Mov Disord. 2021;36(5):1242–6.
de Vet HC, Terwee CB, Knol DL, Bouter LM. When to use agreement versus reliability measures. J Clin Epidemiol. 2006;59(10):1033–9.
Mohandas Nair P, George Hornby T, Louis Behrman A. Minimal detectable change for spatial and temporal measurements of gait after incomplete spinal cord injury. Top Spinal Cord Inj Rehabil. 2012;18(3):273–81.
Beckerman H, Roebroeck ME, Lankhorst GJ, Becher JG, Bezemer PD, Verbeek AL. Smallest real difference, a link between reproducibility and responsiveness. Qual Life Res. 2001;10(7):571–8.
Husted JA, Cook RJ, Farewell VT, Gladman DD. Methods for assessing responsiveness: a critical review and recommendations. J Clin Epidemiol. 2000;53(5):459–68.
Simon SR. Quantification of human motion: gait analysis-benefits and limitations to its application to clinical problems. J Biomech. 2004;37(12):1869–80.
Shah VV, McNames J, Mancini M, et al. Digital biomarkers of mobility in Parkinson’s disease during daily living. J Parkinsons Dis. 2020;10(3):1099–111.
Shah VV, McNames J, Mancini M, et al. Quantity and quality of gait and turning in people with multiple sclerosis, Parkinson’s disease and matched controls during daily living. J neurol. 2020;267(4):1188–96.
Vienne A, Barrois RP, Buffat S, Ricard D, Vidal PP. Inertial sensors to assess gait quality in patients with neurological disorders: a systematic review of technical and analytical challenges. Front Psychol. 2017;8:817.
Byrom B, Watson C, Doll H, et al. Selection of and evidentiary considerations for wearable devices and their measurements for use in regulatory decision making: recommendations from the ePRO consortium. Value Health. 2018;21(6):631–9.
Aminian K, Najafi B, Büla C, Leyvraz PF, Robert P. Spatio-temporal parameters of gait measured by an ambulatory system using miniature gyroscopes. J Biomech. 2002;35(5):689–99.
Mancini M, King L, Salarian A, Holmstrom L, McNames J, Horak FB. Mobility lab to assess balance and gait with synchronized body-worn sensors. J Bioeng Biomed Sci. 2011;Suppl 1:007.
Vajs IA, Bobić VN, Ðurić-Jovičić MD, Janković MM. Open-source application for real-time gait analysis using inertial sensors. 2020 28th Telecommunications Forum (TELFOR); 2020;1–4. https://doi.org/10.1109/TELFOR51502.2020.9306636.
Mc Ardle R, Del Din S, Donaghy P, Galna B, Thomas AJ, Rochester L. The impact of environment on gait assessment: considerations from real-world gait analysis in dementia subtypes. Sensors. 2021;21(3):813. https://doi.org/10.3390/s21030813.
Pham MH, Elshehabi M, Haertner L, et al. Validation of a step detection algorithm during straight walking and Turning in patients with Parkinson’s disease and older adults using an inertial measurement unit at the lower back. Front Neurol. 2017;8:457.
Rentz C, Far MS, Boltes M, et al. System comparison for gait and balance monitoring used for the evaluation of a home-based training. Sensors. 2022;22(13):4975. https://doi.org/10.3390/s22134975.
Hansen C, Ortlieb C, Romijnders R, et al. Reliability of IMU-derived temporal gait parameters in neurological diseases. Sensors. 2022;22(6):2304. https://doi.org/10.3390/s22062304.
Ngo T, Abeysekara LL, Pathirana PN, Horne M, Power L, Szmulewicz DJ. A comparative severity assessment of impaired balance due to cerebellar ataxia using regression models. 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC); 2020 20-24 July 2020. p. 4571-4574.
Parati M, Gallotta M, Muletti M, et al. Validation of pressure-sensing insoles in patients with Parkinson’s disease during overground walking in single and cognitive dual-task conditions. Sensors. 2022;22(17):6392. https://doi.org/10.3390/s22176392.
Hulleck AA, Menoth Mohan D, Abdallah N, El Rich M, Khalaf K. Present and future of gait assessment in clinical practice: towards the application of novel trends and technologies. Front Med Technol. 2022;4:901331.
Klockgether T, Synofzik M; AGI working group on COAs and Registries . Consensus recommendations for clinical outcome assessments and registry development in ataxias: Ataxia Global Initiative (AGI) Working Group Expert Guidance. Cerebellum. 2023. https://doi.org/10.1007/s12311-023-01547-z.
Prosperini L, Fortuna D, Gianni C, Leonardi L, Pozzilli C. The diagnostic accuracy of static posturography in predicting accidental falls in people with multiple sclerosis. Neurorehabil Neural Repair. 2013;27(1):45–52.
Montero-Odasso M, van der Velde N, Martin FC, et al. World guidelines for falls prevention and management for older adults: a global initiative. Age Ageing. 2022;51(9):afac205. https://doi.org/10.1093/ageing/afac205.
Skelton DA, Becker C, Lamb SE, et al. Prevention of Falls Network Europe: a thematic network aimed at introducing good practice in effective falls prevention across Europe. Eur J Ageing. 2004;1(1):89–94.
Crosby RD, Kolotkin RL, Williams GR. Defining clinically meaningful change in health-related quality of life. J Clin Epidemiol. 2003;56(5):395–407.
Viceconti M, Hernandez Penna S, Dartee W, et al. Toward a regulatory qualification of real-world mobility performance biomarkers in Parkinson’s patients using digital mobility outcomes. Sensors. 2020;20(20):5920. https://doi.org/10.3390/s20205920.
Jaeschke R, Singer J, Guyatt GH. Measurement of health status. Ascertaining the minimal clinically important difference. Control Clin Trials. 1989;10(4):407–15.
Byrom B, Breedon P, Tulkki-Wilke R, Platko JV. Meaningful change: defining the interpretability of changes in endpoints derived from interactive and mHealth technologies in healthcare and clinical research. J Rehabil Assist Technol Eng. 2020;7:2055668319892778.
US Food and Drug Administration (FDA). Patient-focused drug development: incorporating clinical outcome assessments into endpoints for regulatory decision-making. Guidance for Industry, Food and Drug Administration Staff, and Other Stakeholders. Available from https://www.regulations.gov/docket/FDA-2023-D-0026: U.S. Food & Drug Administration (FDA); 2023.
Schmitz-Hubsch T, Coudert M, Bauer P, et al. Spinocerebellar ataxia types 1, 2, 3, and 6: disease severity and nonataxia symptoms. Neurology. 2008;71(13):982–9.
Subramony SH, May W, Lynch D, et al. Measuring Friedreich ataxia: interrater reliability of a neurologic rating scale. Neurology. 2005;64(7):1261–2.
Manta C, Patrick-Lake B, Goldsack JC. Digital measures that matter to patients: a framework to guide the selection and development of digital measures of health. Digit Biomark. 2020;4(3):69–77.
Schniepp R, Schlick C, Pradhan C, et al. The interrelationship between disease severity, dynamic stability, and falls in cerebellar ataxia. J neurol. 2016;263(7):1409–17.
Mikolaizak AS, Rochester L, Maetzler W, et al. Connecting real-world digital mobility assessment to clinical outcomes for regulatory and clinical endorsement-the Mobilise-D study protocol. PLoS One. 2022;17(10):e0269615.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Consortia
Contributions
All authors as members of the AGI working group “Gait&Balance” have contributed substantial work for conception, design and drafting or revising the paper.
Corresponding author
Ethics declarations
Competing Interests
-
Fay Horak and Vrutangkumar Shah and James McNames are employees of APDM Precision Motion, Clario, a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by OHSU.
-
Winfried Ilg receives consultancy honoraria from Ionis Pharmaceuticals.
-
Enrico Bertini receives honoraria for Advisory Board from PTC Therapeutics, Biogen, Roche.
-
Christopher Gomez discloses research collaboration with APDM Precision Motion, Clario.
-
Tanja Schmitz-Hübsch discloses research collaboration with a provider of a motion capture system based on Kinect technology (Motognosis GmbH, Berlin, Germany).
-
Helen Dawes is Director of International Affairs Physiobiometrics inc and scientific advisor for Elaros, UK.
-
Kirsi M Kinnunen ist employed by IXICO, London, UK
-
Hasmet Hanagasi, Jane Newman, Sarah Milne, Clara Rentz, Lukas Beichert, Yi Ng, Lisa Alcock, Norlinah Mohamed Ibrahim, Martina Minneropt, Andrea H Nemeth, Bedia Samanci, Vrutangkumar Shah, Susanna Summa, Gessica Vasco report no disclosures.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ilg, W., Milne, S., Schmitz-Hübsch, T. et al. Quantitative Gait and Balance Outcomes for Ataxia Trials: Consensus Recommendations by the Ataxia Global Initiative Working Group on Digital-Motor Biomarkers. Cerebellum 23, 1566–1592 (2024). https://doi.org/10.1007/s12311-023-01625-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-023-01625-2