Abstract
A low-cost locally available agricultural waste-based Anchote peel adsorbent was studied to remove the methyl orange (MO) dye from wastewater which was created at laboratory conditions. The adsorbent was characterized by Fourier infrared and x-ray powder diffraction spectroscopic techniques. The adsorption capacity of the proposed adsorbent was investigated using the batch adsorption method. The best performance was achieved after careful optimization of experimental parameters such as adsorbent dose (0.4 g), initial dye concentration (70 mg/L), contact time (140 min), pH of the solution (8.0), and temperature (40 °C), respectively. The removal of MO from water using anchote peel adsorbent achieved a removal efficiency of 94.47% following the pseudo-second-order kinetic model, Freundlich isotherm, and chemisorption mechanism, respectively. The adsorbent showed heterogeneous surfaces and the adsorption of MO was thermodynamically spontaneous. Furthermore, the present results reveal that Anchote peel adsorbent is promising in future for the removal of organic dyes and other contaminants like toxic heavy metals from water and wastewater.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Water is the most abundant and the scarcest substance required to sustain life in this World. The reason for this is only a very small portion of water (< 3%) is accessible for human utilization, and also the available part is highly affected by natural and artificial pollution (Padowski et al. 2015; Wei et al. 2021). Water pollutants can be organic and inorganic pollutants, thermal, physical, and biological. Many industries such as textile, rubber, plastics, printing leather, cosmetics, paper, pharmaceutical, agricultural, food technology, hair coloring and photoelectrochemical cells widely use dyes and discharge in the form of waste into water bodies (Tkaczyk et al. 2020). The contamination of water resources with dyes becomes a global concern because of their inertness and possible health effects. Among the different organic dyes anionic, cationic, and nonionic, azo dyes are a large class of synthetic dyes containing azo groups (–N=N–). The presence of aromatic rings and –N=N– groups in azo dyes structure make them highly toxic, carcinogenic, and teratogenic as well as harmful to the environment and biological organisms (Bai et al. 2020; Haque et al. 2021). From the large quantities of the azo dyes used in textile industries, up to 20% are eventually discharged to the environment as effluent thereby causing high toxicity and mutagenicity in aquatic life and humans (Mahmood et al. 2016).
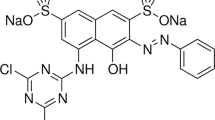
Methyl orange (MO) is a common anionic azo dye that is harmful to the environment and biological systems, so it must be treated mildly before it can be discharged into the environment (Wu et al. 2021). MO is soluble in water, and is resistant to degradation; its removal from aqueous solutions by existing water treatment methods is difficult. Moreover, MO persists in the environment if not treated correctly and presents a hazard to living organisms. The leakage of dyes into wastewater has been identified as a factor affecting human health because of the carcinogenic and mutagenic effects of dyes (Alabbad 2020).
In this study, MO was selected as a target because of its wider applications in textile, printing, paper manufacturing, pharmaceutical, food industries, and chemical laboratories, as well as its negative effects on the environment, animals, and human beings (Alseddig et al. 2017). Thus, wastewater treatment was proposed using different methods to reduce health and environmental problems. There are various methods employed including photo-catalytic and electrochemical combined treatments, photo-catalytic degradation, sonochemical degradation, biodegradation, electrochemical degradation, adsorption process, chemical coagulation or flocculation, ozonation, cloud point extraction, oxidation, nano-filtration, chemical precipitation, ion-exchange, reverse osmosis and ultra-filtration (Bharathi and Ramesh 2013; Mittal and Mittal 2015). Contrary to other methods, adsorption is capable of removing the dyes from concentrated wastewater with low operating costs and high flexibility. Thus, adsorption is the most selective and effective technology for water pollution remediation to remove pollutants from complex matrices (Dai et al. 2018). The removal of different ionic dyes from aqueous media using adsorption techniques are reported by different scholars (Dai et al. 2018; Latif et al. 2019; Kadhom et al. 2020). In particular, the adsorption of dyes from water using agricultural wastes which are loose, porous in structure, and have amine, carbonyl, carboxyl, and reactive hydroxyl groups become attractive due to their dual advantages. These advantages are reducing the environmental burden and using it as a cost-effective and reusable adsorbent based on the principle of treating waste using waste materials (Huang 2017; Kadhom et al. 2020). In line with this, many research reports indicated that the removal of MO from wastewater through the application of an adsorption process using agricultural wastes attracted numerous attention across the World (Sigh et al. 2003; Mittal et al. 2007; Dakhil 2020). Subsequently, pomel peel (Zhang et al. 2020), eggshell (Belay and Hayelom 2014), corn leaf (Fadhil and Eisa 2019), chitosan (Conde et al. 2020), halloysite and chrysotile nanotubes (Wu et al. 2021) adsorbents were used to remove MO from water. These adsorbents were reported as having good performance for the adsorption of MO from water. Hence, the effort for exploiting other locally available and low-cost adsorbents is still demanding.
Anchote (Coccinia abyssinica) is an indigenous vegetable tuber crop cultivated for human consumption by Western and South-eastern Ethiopian communities of the Oromo people (Regasa et al. 2018). The skin or peel removed from Anchote tuber during cooking is discarded as waste without use for a long time. However, these agricultural waste biomaterials have different organic compounds with various functional groups to interact with other molecules. Therefore, in the present study, Anchote peel waste biomaterials were applied as cost-effective, cheap, and effective adsorbents for the removal of MO from synthetic wastewater using the batch adsorption method. The adsorption efficiency, adsorption isotherm, adsorption kinetics, and adsorption thermodynamics of the Anchote peel adsorbent have been investigated for better performances for the removal of MO in the current study.
Materials and methods
Chemicals and apparatus
All chemicals and reagents used were analytical reagent grades. Analytical standard of methyl orange dye (MO) was obtained from (Loba Chemie and Qualigens, India). Analytical grade reagent hydrochloric acid (37%) (Merck, Germany) and sodium hydroxide (97%) (CDH, India) were used in the present investigation.
MO stock solution (1000 mg/L) was prepared with distilled water, and working solutions (10–100 mg/L) were prepared by diluting the stock solution before the batch adsorption experiment.
Ultraviolet–visible (UV–vis) spectrophotometer (Shimadzu DU-8800D, Japan) was used for measuring the concentration of MO dye before and after each batch of adsorption experiments.
Fourier Transform Infrared (FTIR) spectrophotometry (Shimadzu FTIR 8400S, Japan) and X-ray powder diffraction (XRD) (model Xpert MPD, Philips, Netherlands) spectrometry were used for the characterization of Anchote peel adsorbent materials. The chemical and crystal nature of the Anchote peel powder was performed using FTIR and XRD. FTIR spectra were recorded and used to study the available functional units and possible molecular interactions. XRD data were taken with CuKα radiation (λ = 1.5418 Å), on the powder diffractometer operated in the 2θ = 0.5–80° range and step scan of Δ2θ = 0.5°.
Biosorbent preparation and characterization
Anchote peel (C. abyssinica) was collected from the local fruit stalls in Nekemte City, Ethiopia washed thoroughly with distilled water to remove impurities, chopped into small pieces, sun-dried for four days, and finally oven-dried at 105 °C until it became crispy. The dried Anchote peels were pulverized into the fine powder by a mechanical grinder and sieved through a 1 mm sieve. The obtained Anchote peel powder was used for experimental purposes. The fine powder of the adsorbent was kept in a plastic bag for the subsequent experiment and adsorbent characterization.
Adsorption studies
Adsorption of MO dye onto Anchote peel adsorbent
The batch adsorption experiments were used to determine the efficiency of the adsorbents for the removal of MO dye from an aqueous solution. The adsorption experiments were carried out by taking 25 mL of adsorbate solutions with varying initial concentrations (10–100 mg/L) in different conical flasks (100 mL). The fixed quantity of adsorbent was added to each flask and covered in an isothermal water bath shaker and shaken with a speed of 120 rpm at 298 K to achieve equilibrium condition. After filtration using Whatman filter paper grade 42, the residual concentration of MO was measured three times (n = 3) by a UV–vis spectrophotometer at a pre-optimized wavelength of 462.10 nm. The experimental parameters affecting the batch adsorption of MO were optimized based on the design of the experiment using the Minitab method. The extent of dye adsorption can be expressed by three adsorption parameters, namely, adsorption capacity (Qm), adsorption capacity at any time (t) (Qt), and removal efficiency (also known as percent removal, %R) given by the following Eq. (Douglas and Vermeulen 1981):
where Co and Ce are the initial and the equilibrium MO dye concentrations (mg/L), V is the volume of solution (mL) and W is the amount of adsorbent used (g).
Adsorption isotherm study
The equilibrium relationship between the adsorbate dosage and the adsorbent uptake with time was investigated by using Freundlich and Langmuir isotherms.
Langmuir assumes that a monolayer is formed at the maximum adsorption that occurs on localized sites on the homogeneous surface to attain the Langmuir isotherm linear form given by the following Eq. (Cheung et al. 2001):
where Ce is the equilibrium dye concentration (mg/L), qe (mg/gm) is the amount of dye adsorbed at equilibrium, qm (mg/gm) is the amount of dye adsorbed at saturation and Kl (g/L) is Langmuir constant.
Freundlich isotherm uses Freundlich empirical to describe the distribution of a solute between solid and aqueous phases at equilibrium expressed as (Baup et al. 2000):
where Kf (g/L)1/n and n are Freundlich constants, which give a measure of both intensity and capacity of adsorption, respectively.
Adsorption kinetic studies
Adsorption kinetics was used for determining the mechanism of dye adsorption onto the adsorbent material, the dye adsorption rate onto the adsorbent’s particle surface, and show the influence of different conditions on the speed of the adsorption process (Al-Harby et al. 2021). Therefore, pseudo-first-order kinetic model and pseudo-second-order kinetic model were studied to understand the kinetics of the adsorption of the MO dye onto Anchote peel adsorbent at 25 °C.
Pseudo-first-order kinetic model
This model also known as Lagergren Model determines the relationship between the change in time and the adsorption capacity with an order of one as shown here (Lagergren 1898).
where Qe and Qt are the adsorption capacity at equilibrium and time t (mg g−1), respectively, k1 is the pseudo-first-order rate constant (min−1) and t is the time (min). The values of Qm and k1 can be determined from the intercept and the slope of the linear plot of log (Qm − Qt) versus t.
Pseudo-second-order kinetic model
The pseudo-second-order kinetic model is the model that shows the relationship between the adsorption capacity and concentration with second order. Pseudo-second-order Kinetic is expressed by (Ho and McKay 1998):
where K2 (gm/mg.min) is pseudo-second-order constant.
Intraparticle diffusion model
Kinetic data obtained from the adsorption study were further analyzed using the intraparticle diffusion model (Weber and Morris 1963). This model is used to determine the rate-controlling for the adsorption process and is expressed by:
where qt is the number of dye ions adsorbed at time t in mg/g, kint is the intraparticle diffusion constant (mg g−1 min−1/2), and C is a constant (mg g−1) that is directly proportional to the boundary layer thickness. Both values of kint and C can be obtained by calculations from the slope and intercept resulting from the linear curve of qt versus t1/2.
Adsorption thermodynamic studies
The characteristic energy change associated with the adsorption of dyes on the surface of the adsorbent can be obtained from thermodynamic parameters. Thus, Gibbs free energy change, ∆G° (kJ mol−1), enthalpy change, ∆H° (kJ mol−1), and entropy change, ∆S° (J mol−1 K−1) were calculated for the adsorption of MO dye onto the Anchote peel adsorbent using four different temperatures (293, 303, 313, and 323 K) by using the following equations to identify the value at which optimum performance is observed.
From ΔG = − RT lnKc and ΔG = ΔH° − TΔS°, then by rearrangement,
where R is the universal gas constant (8.314 J mol−1 K−1), T is the absolute temperature (K) and Kc is the distribution coefficient (QE/Ce). By plotting ln Kc versus 1/T, the thermodynamic parameters can be acquired.
Statistical analysis
In this study, all experiments were performed in triplicate, and the results are expressed as the mean ± standard deviation. The computation of the isotherm, kinetic models, and thermodynamic results was performed using the Solver add-in (Microsoft Excel) to compute the parameters of the isotherm and kinetic models (Tran et al. 2017).
Results and discussion
Characterization of Anchote peel adsorbent
The characterization of adsorbent materials is very important to better understand the nature of the materials. Information obtained from the characteristic of adsorbents is necessary as their physical and chemical properties can influence the adsorption behavior. Thus, the characterization of Anchote peel was carried out by using FTIR and XRD (Fig. 1A and B). FTIR spectra were employed to ascertain the bond types, structures, and functional groups in the Anchote peel agricultural waste and determine the functional groups responsible for the adsorption of the dye (Fig. 1C). The bands observed between 3800 and 2700 cm−1 are due to –C–H,= C–H, and hydroxyl groups (O–H groups) stretching (Samiey and Ashoori 2012). The presence of double peaks with equivalent halves near 3629–3315 cm−1 belongs to the primary amines (NH2) symmetric stretching that confirms the presence of amides. The absence of a single broad peak at about 3400–3300 cm−1 and C–N stretching near 1300–1000 cm−1 show the compound is amide (Benerjee and Chattopadhyaya 2017). The peak at 2295 cm−1 is due to the C≡N stretching vibration while the two strong peaks at 1678 and 1441 cm−1 are owing to the presence of C=O and N–H groups. The C=O from carboxylic acids, ketones, or esters can be associated with the band at 1678 cm−1 and the presence of C=C that can be related to the lipid fraction of the Anchote peel was identified at 1510 cm−1 (Pang et al. 2019). Furthermore, the peak at 708 cm−1 corresponds to NH out-of-plane bending (Ahmad et al. 2009). Thus, the biomaterial mainly consists of long-chain amine compounds containing carbonyl functional units. The presence of both amine and carbonyl groups in the bionanomaterial makes it the ideal adsorbent for the adsorption of many compounds of interest.
The XRD spectrum of Anchote peel powder is depicted in Fig. 1D. It was perceived that the concentration values increased with increasing an angle up to a certain transition angle and the anomaly decreased further. The intensity of this adsorbent started from 10 degrees and increased up to 25 degrees, and then decreased gradually. A broad peak was observed at 2θ = 20°, corresponding to an interlayer spacing (d-spacing) of 1.75 nm indicating the noncrystalline nature of the adsorbent powder. From the broad spectrum, one can deduce that the Anchote peel material is highly amorphous and porous which is a characteristic of plant-based natural polymers (Lata 2017). Such kind of feature is important for adsorption since it increases the surface area to volume ratio of the material. The highest peak observed between 18o and 25o corresponds to the carbohydrate, particularly cellulose (Jiang et al. 2013; Ferrer et al. 2016) indicating the low crystallinity of the adsorbent.
Adsorption studies
Effect of adsorbent dose
Adsorbent dye removal efficiency in treating polluted wastewater mainly depends on the number of active adsorption sites available on the adsorbent. The amount of adsorbent used in adsorption is particularly important because it determines the adsorbate-adsorbent equilibrium in the system and predicts the treatment cost of adsorbent per unit of dye solution (Wanyonyi et al. 2014). The effect of the adsorbent dose for Anchote peel adsorbent was investigated using the adsorbent dose of different quantities in the range of 0.1–0.5 g/L (Fig. 2). As can be seen from the results, the removal efficiency increased rapidly from 92.6 to 97.8% and then decreased due to saturation. The increase in removal efficiency from 0.1 to 0.4 g of adsorbent might be due to the increase in total surface area and the availability of more binding sites for adsorption.
Thereafter, no significant change has been observed, indicating that the adsorption saturation is reached at the adsorbent dose greater or equal to 0.4 g/L. Thus, this concentration was chosen as the optimum dose for further studies. The effect of increasing the initial MO dye concentration on its adsorption showed positive effects, and the dye adsorption followed the Freundlich isotherm model.
Effect of contact time and initial MO concentration
The effects of contact time and initial dye concentration on the adsorption capacity were determined based on different times of agitation and different initial concentrations of MO as shown in Fig. 4. The obtained result shows the adsorption increases with contact time and finally reaches the equilibrium condition at 140 min (Fig. 3A). However, the increase is relatively higher during the initial 10 min.
The rapid increase in adsorption during the initial stage may presumably be due to the availability of vacant active sites on the surface of the adsorbent. The slow increase at the later stage is due to the slow diffusion of dye into the adsorbent pores because the external sites are completely occupied. Similarly, the adsorption capacity increases with the increase in the initial dye concentration (Fig. 3B). It is also inferred that the adsorption capacity increases with increasing initial dye concentration until 70 mg/L and then continued with slight changes showing saturation of the adsorption.
Effect of pH and temperature
The effect of pH on the removal efficiency of MO dye by Anchote peel adsorbent was evaluated as depicted in Fig. 4A. It has been observed that the removal efficiency increased from 50.0% to 96.23% on increasing the pH from 2.0 to 8.0 for MO-Anchote peel adsorbent. Afterward, there is no major change in the removal efficiency with a further increase in pH values. MO is an anionic dye (carries the negative charge); in an acidic medium, the hydronium ions are predominant in the solution; therefore, the vacant sites of the adsorbent will be occupied by these cations, and the positive sites of the support will adsorb the MO molecules. The number of positively charged sites on the adsorbent decreased as the pH increased. Hence the adsorption of the dye molecules to the surface of the adsorbent increased as the pH value increased (Ahmad and Alrozi 2011; Akar et al. 2013). As the pH of the system increases, the number of negatively charged sites increases, decreasing the biomaterial's adsorption capacity (Kaur et al. 2013). At high pH, the negative charge of the adsorbent sites, therefore, does not promote the adsorption of the MO dye anions due to electrostatic repulsion. In addition, the low adsorption rate of MO at high pH is due to the presence of excess hydroxyl ions, which will compete with the anionic molecules of dye for the adsorption sites (Conde et al. 2020).
The temperature has a pronounced effect on the adsorption capacity of the adsorbents. The high temperature will destroy the covalent bond thus it reduces the holding capacity of the adsorbents to trap the dye molecules. The adsorption capacity largely relies on the chemical interaction between the functional groups of the adsorbent surface and adsorbate (Jayarajan et al. 2011). The effect of temperature on the dye uptake of the adsorbent was studied by using different temperatures such as 15 °C, 20 °C, 25 °C, 30 °C, 35 °C, 40 °C, 45 °C, and 50 °C as indicated in Fig. 4B. The dye removal efficiency of 94.16% was achieved at the temperature of 40 °C and then continued with a slight increase with a further increase in temperature. The dye uptake increases with temperature because the adsorption of the MO dye increases with the increase in temperature the reason that the dye molecules can obtain sufficient energy to interact with the active site on the adsorbent.
Isotherm study
Adsorption isotherms provide information on the capacity of the adsorbent and the equilibrium relationships between adsorbent and adsorbate at fixed temperatures (Oyelude et al. 2018). Consequently, to postulate the suitable isotherm, we evaluated the equilibrium relations of the batch adsorption of MO using Anchote peel as an adsorbent biomaterial.
The MO adsorption parameters were determined to investigate the adsorption isotherm model that best fits the observed data of the adsorption process (Table 1). As shown in Fig. 5, the result confirmed that the Freundlich isotherm is the best reliable model with the studied process than the Langmuir isotherm model. Furthermore, the result reveals that the change in the behavior of the adsorption process occurred upon the addition of a specified MO amount to the heterogeneous surface of the proposed adsorbent which can be inferred from the higher R2 value for the Freundlich model. In the Freundlich isotherm, the factor 1/n, was found to be greater than unity, suggesting that the adsorption of MO onto Anchote peel adsorbent is cooperative. Anchote peel adsorbent showed its surface heterogeneity and the exponential distribution of the active sites and their energies to interact with the MO for better performances (Al-Ghouti and Da'ana 2020). Thus, the presence of a heterogeneous surface with a multilayer adsorption mechanism enhanced the increase in adsorption with the increase in the MO concentration (Sewu et al. 2019).
Kinetic studies
The adsorption kinetics of MO adsorption onto the Anchote peel adsorbent was studied using the pseudo-first-order and pseudo-second-order models. The kinetic parameters for both models were determined using Eqs. (6) and (7) as well as their respective plots (Table 2). The adsorption kinetics and the correlation coefficient (R2) determined to confirm that the adsorption data fit best with pseudo-second-order when compared to the pseudo-first-order kinetic model, as shown in Fig. 6 and Table 2. Furthermore, the R2 (0.996) indicated in Table 2 leads to infer that the pseudo-second-order kinetic model is the best one to represent the adsorption results of MO. This implies that the adsorbent has heterogeneous surfaces and thus the adsorption rate is limited by the chemisorption mechanism (chemical and valence forces by exchanging electrons between the adsorbent and the adsorbate) (Sterenzon et al. 2022). The result from this study is in agreement with the previous literature reports for the adsorption of MO on different adsorbents (Chen et al. 2011; Fadhil and Eisa 2019; Conde et al. 2020; Wu et al. 2021).
Thermodynamic studies
To understand the energy associated with the adsorption of MO onto the Anchote peel adsorbent, the thermodynamic plot is shown in Fig. 7, and the values obtained for ΔG°, ΔS°, and ΔH° at 25 °C are − 8.53 kJ/mol, − 14.14 kJ/mol, and − 27.90 kJ/mol, respectively, as shown in Table 3. The increase in ΔG° from − 9.05 to − 8.21 kJ mol−1 values with an increase in temperature indicated more efficient adsorption at higher temperatures due to the high mobility of more MO ions to the adsorbent surfaces (Adebayoa et al. 2019). The trend of free energy with temperature further indicates the spontaneous nature and feasibility of the adsorption process with an increase in temperature. Thus, the proposed adsorbent is thermodynamically suitable for removing MO from aqueous media with increased affinity at higher temperatures (Lawal et al. 2019).
The negative value of ∆H° (− 14.14 kJ/mol) and ∆S° (− 27.90 kJ/1 mol) confirmed the exothermic nature of the process and the increased predictability at the solid-solution interface during the adsorption process and a good affinity of the adsorbent for MO.
Intraparticle diffusion model
This model is suggested to describe the adsorption of MO onto anchote peel better than pseudo-first-order and pseudo-second-order kinetics equations. It is an empirical model that describes the dye uptake mechanism and varies almost proportionally with t1/2 rather than with the contact time, t. When adsorbate in solution is mixed with the adsorbent, there occurs transport of the MO dye into the pores of particles from the solution through the interface between the solution and the adsorbent. The plot of qt against t1/2 shown in Fig. 8 is linear passing through the origin for the known initial concentration of MO implying that the adsorption process followed the intraparticle diffusion model (Benerjee and Chattopadhyaya 2017). This implies that the system is characterized by the high concentration of MO, vigorous mixing, and large particle size of Anchote peel powder. The plot shows that the quantity of MO adsorbed is proportional to the initial concentration of MO in an aqueous solution and the square root of time taken for adsorption (Weber and Morris 1963).
The mechanism of adsorption of MO on Anchote peel adsorbent is due to intraparticle diffusion. Briefly, the active functional groups in anchote biosorbent such as –OH and –C=O groups can form covalent or non-covalent (hydrogen bonding, ionic interaction, π–π stacking, etc.) while the –NH2 groups can interact with covalent (imine bonds), non-covalent interaction and semi-covalent (amide bonds) with the MO dye molecules (Fig. 9).
Various agricultural waste materials-based adsorbents have been investigated for the removal of MO dye from water. Therefore, the adsorptive capacity of the adsorbent investigated here was compared with those reported in the literature (Table 4). Anchote peel used in this study is a naturally available material with cost low adsorbent which exists in a natural environment as waste and can be used with small treatment and modification. The values of the adsorption capacity given in Table 4 show the proposed adsorbent is promising in removing the MO dye from an aqueous solution when compared with pomelo peel, eggshell, corn leaves, chitosan, halloysite, and chrysotile nanotubes.
Conclusion
Sustainable wastewater treatment technology can be achieved by establishing innovative, cost-effective, and environmentally materials. Toxic organic dye removal from wastewater using adsorption technology and agricultural wastes is interesting in this regard. In the present work, Anchote peel adsorbents were found to be an effective low-cost adsorbent for the removal of MO dye from an aqueous solution. The maximum adsorption capacity was achieved at the optimized experimental conditions of adsorbent dose, initial dye concentration, contact time, pH, and temperature of the solution of 0.4 g, 70 mg L−1, 140 min, 8.0, and 40 °C, respectively. Anchote peel is indeed a viable and affordable adsorbent for the adsorptive removal of MO dye from water with an adsorption capacity of 103.03 mg/g, percentage removal of 94.47% with a relative standard deviation of 7.35% (n = 3) according to the present result. The adsorption data best fits with the pseudo-second-order kinetic model, Freundlich isotherm, chemisorption mechanism, and thermodynamically spontaneous. The proposed biomaterials can be further applied for the water pollutants removal with a further physical and chemical treatment to enhance the performance of the adsorbent. Finally, the use of agricultural wastes such as Anchote peel as adsorbent is economical, practical, and more viable, sound in physicochemical nature, and thus its application to actual wastewater treatment will be the focus of our future work.
References
Adebayoa GB, Jamiua W, Okoroa HK, Okeolaa FO, Adesinaa AK, Feyisetan OA (2019) Kinetics, thermodynamics and isothermal modelling of liquid phase adsorption of methylene blue onto moringa pod husk activated carbon. S Afr J Chem 72:263–273
Ahmad MA, Alrozi R (2011) Removal of malachite green dye from aqueous solution using rambutan peel-based activated carbon: equilibrium, kinetic and thermodynamic studies. Chem Eng J 171:510–516
Ahmad A, Rafatullah M, Ibrahim MH, Hashim R (2009) Scavenging behavior of meranti sawdust in removal of methylene blue from aqueous solution. J Hazard Mater 170:357
Akar E, Altinişik A, Seki Y (2013) Using activated carbon produced from spent tea leaves for the removal of malachite green from aqueous solution. Ecol Eng 52:19–27
Alabbad EA (2020) Efficient removal of methyl orange from wastewater by polymeric chitosaniso-vanillin. Open Chem J 7:16–25
Al-Ghouti MA, Da’ana DA (2020) Guidelines for the use and interpretation of adsorption isotherm models: a review. J Hazard Mater 393:122383
Al-Harby NF, Albahly EF, Mohamed NA (2021) Kinetics, isotherm and thermodynamic studies for efficient adsorption of congo red dye from aqueous solution onto novel cyanoguanidine-modified chitosan adsorbent. Polymers 13:4446
Alseddig A, Eljiedi A, Kamari A (2017) Removal of methyl orange and methylene blue dyes from aqueous solution using lala clam (Orbicularia orbiculata) shell. AIP Conf Proc 2017:1847
Bai YN, Wang XN, Zhang F, Wu J, Zhang W, Lu YZ, Fu L, Lau TC, Zeng RJ (2020) High-rate anaerobic decolorization of methyl orange from synthetic azo dye wastewater in a methane-based hollow fiber membrane bioreactor. J Hazard Mater 388:121753
Baup S, Jaffre C, Wolbert D, Laplanche A (2000) Adsorption of pesticides onto granular activated carbon: determination of surface diffusivities using simple batch experiments. Adsorption 6:219–228
Belay K, Hayelom A (2014) Removal of methyl orange from aqueous solutions using thermally treated egg shell (locally available and low-cost biosorbent). Int J Innov Sci Res 8:43–49
Benerjee S, Chattopadhyaya MC (2017) Adsorption characteristics for the removal of a toxic dye, tartrazine from aqueous solutions by a low-cost agricultural by-product. Arab J Chem 10:S1629–S1638
Bharathi KS, Ramesh ST (2013) Removal of dyes using agricultural waste as low-cost adsorbents: a review. Appl Wate Sci 3:773–790
Chen H, Zhao J, Wu J, Dai G (2011) Isotherm, thermodynamic, kinetics and adsorption mechanism studies of methyl orange by surfactant modified silkworm exuviae. J Hazard Mater 192(1):246–254
Cheung CW, Porter JF, Mckay G (2001) Sorption kinetic analysis for the removal of cadmium ions from effluents using bone char. Water Res 35(3):605–612
Conde MA, Liwaire CLS, Tchakounte AN, Ntinkam CAS, Nzugue DLE, Kede CM (2020) Removal of methyl orange (MO) by chitosan modified by zero valent iron. Int J Eng Res Technol 9(7):1542–1549
Dai Y, Sun Q, Wang W, Lu L, Liu M, Li J, Yang S, Sun Y, Zhang K, Xu J, Zheng W, Hu Z, Yang Y, Gao Y, Chen Y, Zhang X, Gao F, Zhang Y (2018) Utilizations of agricultural waste as adsorbent for the removal of contaminants: a review. Chemosphere 211:235–253
Dakhil IH (2020) Recycling of agriculture wastes for efficient removal of methyl orange dye using batch adsorption unit. IOP Conf Ser Mater Sci Eng 881:012186
Douglas LM, Vermeulen T (1981) Binary Langmuir and Freundlich isotherms for ideal adsorbed solutions. J Phys Chem 85(22):3247–3250
Fadhil HO, Eisa M (2019) Removal of methyl orange from aqueous solutions by adsorption using corn leaves as adsorbent material. J Eng 25(4):55–69
Ferrer A, Alciaturi C, Faneite A, Rios J (2016) Analyses of biomass fibers by XRD, FT-IR, and NIR. In: Vaz S Jr (ed) Analytical techniques and methods for biomass. Springer, Cham. https://doi.org/10.1007/978-3-319-41414-0_3
Haque MM, Haque MA, Mosharaf MK, Marcus PK (2021) Decolorization, degradation and detoxification of carcinogenic sulfonated azo dye methyl orange by newly developed biofilm consortia. Saudi J Biol Sci 28:793–804
Ho YS, McKay G (1998) Sorption of dye from aqueous solution by peat. Chem Eng J 70:115
Huang YY (2017) Research progress of wastewater treatment by agricultural wastes as biological adsorbent. Appl Chem Ind 2:368–372
Jayarajan M, Arunachalam R, Annadurai G (2011) Use of low-cost nanoporous materials of pomelo fruit peel wastes in the removal of textile dye. Res J Environ Sci 5(5):434–443
Jiang L, Fang Z, Li XK, Luo J (2013) Production of 2,3-butanediol from cellulose and Jatropha hulls after ionic liquid pretreatment and dilute-acid hydrolysis. AMB Express 3(48):1–8
Kadhom M, Albayati N, Alalwan H, Al-Furaiji M (2020) Removal of dyes by agricultural waste. Sust Chem Pharmacy 16:100259
Kaur S, Rani S, Mahajan RK (2013) Adsorption kinetics for the removal of hazardous dye congo red by biowaste materials as adsorbents. J Chem 628582. https://doi.org/10.1155/2013/628582
Lagergren S (1898) About the theory of so-called adsorption of soluble substances. K Sven Vetensk Handingarl 24:1
Lata S (2017) Studies on removal of malachite green dye from aqueous solution using plant-based biosorbents. M.Sc. Thesis, National Institute of Technology, Rourkela
Latif S, Rehman R, Mitu L, Imran M, Iqbal S, Kanwal A (2019) Removal of acidic dyes from aqueous media using Citrullus lanatus peels: an agrowaste-based adsorbent for environmental safety. J Chem 2019:1–9. https://doi.org/10.1155/2019/6704953
Lawal IA, Klink M, Ndungu P (2019) Deep eutectic solvent as an efficient modifier of low-cost adsorbent for the removal of pharmaceuticals and dye. Environ Res 179:108837
Mahmood S, Khalid A, Arshad M, Mahmood T, Crowley DE (2016) Detoxification of azo dyes by bacterial oxidoreductase enzymes. Crit Rev Biotechnol 36:639–651
Mittal A, Malviya A, Kaur D, Mittal J, Kurup L (2007) Studies on the adsorption kinetics and isotherms for the removal and recovery of methyl orange from wastewaters using waste materials. J Hazard Mater 148(1–2):229–240
Mittal A, Feather MJH (2015) A remarkable adsorbent for dye removal. In: Sharma SK (ed) Green chemistry for dyes removal from waste water: research trends and applications, Wiley, pp 409–457
Oyelude EO, Awudza JAM, Twumasi SK (2018) Removal of malachite green from aqueous solution using pulverized teak leaf litter: equilibrium, kinetic and thermodynamic studies. Chem Cent J 12:81
Padowski JC, Gorelick SM, Thompson BH, Rozelle S, Fendorf S (2015) Assessment of human–natural system characteristics influencing global freshwater supply vulnerability. Environ Res Lett 10:104014
Pang X, Sellaoui L, Franco D, Dottoc GL, Georgin J, Bajahzar A, Belmabrouk H, Lamin AB, Petriciolet AB, Li Z (2019) Adsorption of crystal violet on biomasses from pecan nutshell, para chestnut husk, araucaria bark, and palm cactus: experimental study and theoretical modeling via monolayer and double layer statistical physics models. Chem Eng J 378:122101
Regasa MB, Fayisa KG, Woldegebriel HH (2018) Bioactive phytochemical screening and antioxidant potential of different solvent extracts of anchote: the underutilized delicious cultural food of oromo people. Ethiop Food Sci Technol 6(4):81–90
Samiey B, Ashoori F (2012) Adsorptive removal of methylene blue by agar: effects of NaCl and ethanol. Chem Cent J 6:1
Sewu DD, Jung H, Kim SS, Lee DS, Woo SH (2019) Decolorization of cationic and anionic dye-laden wastewater by steam-activated biochar produced at an industrial-scale from spent mushroom substrate. Bioresour Technol 277:77–86
Singh KP, Mohan D, Sinha S, Tondon GS, Gosh D (2003) Color Removal from wastewater using low-cost activated carbon derived from agricultural waste material. Ind Eng Chem Res 42(9):1965–1976
Sterenzon E, Vadivel VK, Gerchman Y, Luxbacher T, Narayanan R, Mamane H (2022) Effective removal of acid dye in synthetic and silk dyeing effluent: isotherm and kinetic studies. ACS Omega 7:118–128
Tkaczyk A, Mitrowska K, Posyniak A (2020) Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: a review. Sci Total Environ 717:137222
Tran HN, You SJ, Chao HP (2017) Insight into adsorption mechanism of cationic dye onto agricultural residues-derived hyrochars: Negligible role of p–p interaction. Korean J Chem Eng 34(6):1708–1720
Wanyonyi WC, Onyari JM, Shiundu PM (2014) Adsorption of Congo red dye from aqueous solutions using roots of Eichhornia crassipes: kinetic and equilibrium studies. Energy 10:105
Weber WJ, Morris JC (1963) Intraparticle diffusion during the sorption of surfactants onto activated carbon. J Sanit Eng Div Am Soc Civ Eng 89:53–61
Wei H, Zhao S, Zhang X, Wen B, Su Z (2021) The future of freshwater access: functional material-based nano-membranes for desalination. Mater Today Energy 22:100856
Wu L, Liu X, Lv G, Zhu R, Tian L, Liu M, Li Y, Rao W, Liu T, Liao L (2021) Study on the adsorption properties of methyl orange by natural one-dimensional nano-mineral materials with different structures. Sci Rep 11:10640
Zhang B, Wu Y, Chan L (2020) Removal of methyl orange dye using activated biochar derived from pomelo peel wastes: performance, isotherm, and kinetic studies. J Dispers Sci Technol 41(1):125–136
Acknowledgments
This work was supported by the School of Graduate Studies and the Department of Chemistry of Wollega University, Ethiopia.
Funding
The authors are grateful for the MAPRONANO research fund received from the African Center of Excellence (ACE) Makerere University, Uganda.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to publish the work.
Consent for publication
The authors declare that they have agreed to publish the work without any objection.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hambisa, A.A., Regasa, M.B., Ejigu, H.G. et al. Adsorption studies of methyl orange dye removal from aqueous solution using Anchote peel-based agricultural waste adsorbent. Appl Water Sci 13, 24 (2023). https://doi.org/10.1007/s13201-022-01832-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-022-01832-y