Abstract
Purpose
Intestinal protozoan parasites among Asian schoolchildren are a subject of concern due to their prevalence and potential health impact. Understanding and addressing this issue is crucial for public health in the region.
Methods
We conducted a comprehensive search for articles published up to December 2023 across four databases, including Scopus, PubMed, ProQuest, and Web of Science. To estimate the combined prevalence, a random-effects model with a 95% confidence interval (CI) was applied, and the statistical analysis was performed using meta-analysis packages in R version (3.6.1). This study is registered with PROSPERO (CRD42023481146).
Results
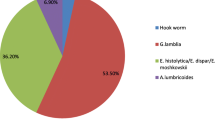
Among 131 eligible articles, the prevalence of intestinal protozoan parasites was 0.208 (95% CI = 0.180–0.238). Lebanon and Tajikistan had the highest country-level prevalence at 0.851 and 0.836, respectively, with Giardia duodenalis being the most prevalent species at 0.082.
Conclusion
In summary, our study highlights the urgent public health issue of protozoan parasites among Asian schoolchildren due to poor sanitation and water quality. Immediate interventions are essential, considering climate and socioeconomic factors, to combat these infections and improve overall health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intestinal parasites pose a significant health challenge in developing nations, where they cause substantial morbidity and mortality rates. Among those most severely impacted are children from economically disadvantaged families. It is noteworthy that more than 267 million preschool-aged children and 568 million school-aged children reside in areas characterized by the widespread and intense transmission of these parasites [1,2,3].
These children frequently find themselves in situations where they need the means to obtain uncontaminated and safe drinking water, proper sanitation facilities, or even basic toilets in their homes. This leaves them exceptionally susceptible to various health risks and challenges [4, 5].
On a global scale, the prevalence of intestinal protozoan parasites (IPPs) affects nearly 3.5 billion individuals, highlighting the extent of this problem. Furthermore, these parasites contribute significantly to the high annual incidence of over one billion cases of diarrheal illnesses [6].
These parasites are ubiquitous in regions characterized by tropical and subtropical climates, which often include countries in the developing world. In such areas, the environmental conditions are conducive to the proliferation of these parasites, making them a significant health concern. The challenges posed by these infections are particularly pronounced in developing nations, where limited access to clean water, sanitation facilities, and healthcare resources can exacerbate the impact of these parasites on the population's health and well-being [4, 7, 8].
Among intestinal protozoan parasites, Cryptosporidium spp., Entamoeba histolytica, and Giardia duodenalis are the predominant sources of infections in the human population [9, 10].
The symptomatic infection manifests as diarrhea, abdominal discomfort, and malabsorption, resulting in malnutrition and weight reduction, especially in children [11].
The most commonly utilized methods for detecting IPPs from stool samples include direct smear microscopy and various concentration techniques [12].
Nowadays, more precise methods such as copro-parasitological exams and DNA-based techniques are employed for more accurate identification of these infections [13].
In recent years, an escalating apprehension has arisen regarding the welfare of children in Asia, with a specific focus on the menace of parasitic infections. The presence of IPPs represents a significant public health challenge, emphasizing the need for a comprehensive understanding of their prevalence and geographical distribution among preschool and school-age children. These insights are essential for crafting precise and effective intervention strategies. The primary aim of this systematic review and meta-analysis is to address the substantial gap in our current knowledge by collecting and analyzing existing data to shed light on the current situation.
Methods
Search strategy
The current study followed the guidelines outlined by the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) [14]. We conducted an extensive search across four databases (including PubMed, Scopus, Web of Science, and ProQuest) to retrieve relevant papers published up to December 2023, without time limitation (Supplementary Table 1). Our search employed terms related to the prevalence, frequency, epidemiology, incidence, parasitic diseases, parasites, parasitic infections, protozoan parasites, protozoan infections, protozoan diseases, protozoan pathogens, intestinal protozoans, preschool and/or school-age children, and Asia, using both AND and/or Boolean operators. The names of the 48 countries in Asia were also included in the search terms list.
Duplicate papers were automatically excluded using EndNote software X9 version. Additionally, we manually scrutinized the reference lists to identify any pertinent studies that may not have been accessible through the database search. Two authors independently conducted the searches, evaluated titles and abstracts, and thoroughly reviewed the full-text articles.
Inclusion and exclusion criteria
Full-text articles were considered eligible upon satisfying the inclusion criteria outlined below:
-
1-
They included cross-sectional research that reported the presence of IPPs among Asian schoolchildren.
-
2-
They were original articles published in peer-reviewed journals.
-
3-
Both the full-text and abstract of the articles were available in English.
-
4-
The articles provided information on the total sample size and the precise number of individuals who tested positive for intestinal protozoan parasites.
Studies that fell into the category of case series, case reports, letters, editorials, publications without original data, review articles, articles with inconclusive findings, non-English-language publications, and studies reporting IPPs in samples from sources other than human subjects were excluded from the analysis conducted in this study. Microsoft Excel® version 2016 was used to systematically gather the following data from the included articles: author names, publication year, climate, annual precipitation, humidity levels, annual rainfall, average temperature, gender, educational status, the Global Burden of Disease (GBD) regions, district/city/province, age, mean age, income level, source of sample, diagnostic method, and type of IPPs (Tables 1, 2, 3).
Quality assessment
We employed the Newcastle–Ottawa Scale to evaluate the quality of the study, as detailed in Supplementary Table 2 [15]. The scoring system was based on the following components and their respective score ranges:
-
Selection (up to a maximum of 5 stars).
-
Comparability (up to a maximum of 2 stars).
-
Outcome (up to a maximum of 3 stars).
Data synthesis and statistical analysis
Multiple statistical methods were employed to comprehensively analyze data concerning the prevalence of IPPs among schoolchildren in Asia. A 95% confidence interval (95% CI) was used to calculate the overall pooled prevalence. To estimate this pooled prevalence, a random-effects model with a Freeman-Tukey double arcsine transformation was utilized. To assess potential publication bias, Begg's rank test was applied, and publication bias was also evaluated using the Luis Furuya-Kanamori (LFK) index and the Doi plot [16]. An LFK index falling outside the ± 2, ± 2, and ± 1 range was considered significantly asymmetrical, slightly asymmetrical, and symmetrical (indicating the absence of publication bias), respectively.
Additionally, heterogeneity among the included studies was evaluated using Cochrane's Q test and the inconsistency index (I2 statistics), where I2 values of 0–25% were classified as low heterogeneity, 25–50% as moderate heterogeneity, and 50–75% as high heterogeneity. Statistical significance was defined as a p-value less than 0.05. All statistical analyses were conducted using the meta and metasens packages in R (version 3.6.1) [17]. This review was registered in PROSPERO (CRD42023481146).
Results
Characteristics of included studies
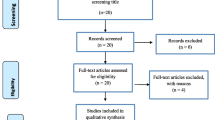
The current study involved a systematic search that resulted in the identification of 10,767 articles, out of which 195 full-text papers were selected for a detailed assessment to determine their eligibility. After our evaluations, we excluded eight studies due to lack of sufficient data, three studies that contained overlapping data, six studies that lacked essential participant details, and 47 studies that did not present original data, such as letters, reviews, workshops, and theses. Finally, 131 papers met the critical appraisal criteria for inclusion in the meta-analysis (Fig. 1).
The prevalence of IPPs among schoolchildren has been reported in thirty-two Asian countries. The largest number of reports were related to Nepal (35 studies), followed by Iran (15 studies) (Table 2).
The estimated pooled prevalence of IPPs among Asian schoolchildren was 0.208 (95% CI = 0.180–0.238) (Fig. 2).
Forest plots for random-effects meta-analysis of intestinal protozoan parasites among Asian schoolchildren (the box indicate the effect size of the studies (prevalance) and the whiskers indicate its confidence interval for corresponding effect size. There is no specific difference between white and black bars, only studies with a very narrow confidence interval are shown in white. In the case of diamonds, their size indicates the size of the effect, and their length indicate confidence intervals)
The studies included in this review employed parasitology techniques comprising microscopic methods (concentration with and without flotation or sedimentation, and other direct smear techniques), culture method, molecular approach (conventional PCR and real-time PCR), and staining methods (Lugol's iodine, trichrome, and Ziehl–Neelsen) (Table 2).
Based on our included studies, we designed a map using QGIS3 software (https://qgis.org/en/site/) to display the prevalence of IPPs among schoolchildren in different regions of Asia (Fig. 3).
Pooled prevalence based on the type of intestinal protozoan parasites, source of samples, gender, and diagnostic techniques
The pooled prevalence of IPPs among Asian schoolchildren was estimated as follows; 0.082 (95% CI = 0.070–0.095) for G. duodenalis with heterogeneity (I2 = 97; τ2 = 0.015; p < 0.001), 0.079 (95% CI = 0.051–0.112) for Blastocystis hominis with heterogeneity (I2 = 98; τ2 = 0.035; p < 0.001), 0.067 (95% CI = 0.025–0.128) for Cryptosporidium spp. with heterogeneity (I2 = 98; τ2 = 0.034; p < 0.001), 0.064 (95% CI = 0.048–0.082) for E. histolytica/dispar with heterogeneity (I2 = 98; τ2 = 0.026; p < 0.001), 0.051 (95% CI = 0.034–0.071) for E. coli with heterogeneity (I2 = 98; τ2 = 0.023; p < 0.001), 0.028 (95% CI = 0.016–0.043) for Endolimax nana with heterogeneity (I2 = 94; τ2 = 0.008; p < 0.001), 0.025 (95% CI = 0.006–0.058) for E. hartmanni with heterogeneity (I2 = 95; τ2 = 0.009; p < 0.001), 0.016 (95% CI = 0.007–0.027) for Cyclospora cayetanensis with heterogeneity (I2 = 62; τ2 = 0.001; p < 0.001), 0.013 (95% CI = 0.005–0.026) for Iodamoeba buetschlii with heterogeneity (I2 = 87; τ2 = 0.007; p < 0.001), 0.010 (95% CI = 0.001–0.025) for Chilomastix mesnili with heterogeneity (I2 = 98; τ2 = 0.007; p < 0.001), 0.005 (95% CI = 0–0.023) for Balantidium coli, 0.004 (95% CI = 0.0008–0.010) for Cystoisospora belli with heterogeneity (I2 = 25; τ2 = 0.0002; p < 0.001), and 0.002 (95% CI = 0–0.010) for Sarcocystis spp. (Table 3).
The highest pooled prevalence estimated based on the source of samples was 0.208 (95% CI = 0.180–0.237) with heterogeneity (I2 = 98; τ2 = 0.039; p < 0.001) for stool (Table 2).
The results of this study show that the male/female ratio is approximately equal for both sexes (OR: 1.019, 95% CI, 0.842–1.234) (Supplementary Fig. 1).
Based on the diagnostic techniques, the highest pooled prevalence (0.851, 95% CI = 0.801–0.890) was related to studies that employed a combined direct smear, staining, and PCR methods (Table 2).
Pooled prevalence based on GBD regions, country, socio-economic status, and educational level
According to different GBD regions, the pooled prevalence ranged from 0.836% to 0.164%, including 0.836 (95% CI = 0.804–0.864) for Central Asia, 0.256 (95% CI = 0.203–0.314) for North Africa and Middle East with heterogeneity (I2 = 99; τ2 = 0.045; p < 0.001), 0.206 (95% CI = 0.147–0.273) for Southeast Asia with heterogeneity (I2 = 98; τ2 = 0.046; p < 0.001), and 0.164 (95% CI = 0.133–0.198) for South Asia with heterogeneity (I2 = 96; τ2 = 0.026; p < 0.001) (Table 2).
At the country level, the highest pooled prevalence was observed for Lebanon (0.851, 95% CI = 0.801–0.890) and Tajikistan (0.836, 95% CI = 0.804–0.864), both with one study (Table 2).
The pooled prevalence estimated based on income level ranged from 0.122 to 0.409, with the highest rate related to the low-income group (0.409, 95% CI = 0.282–0.543) with heterogeneity (I2 = 98; τ2 = 0.046; p < 0.001) (Table 2). Furthermore, based on educational level, the IPPs were most prevalent in the elementary school group (0.252, 95% CI = 0.113–0.424) with heterogeneity (I2 = 99; τ2 = 0.050; p < 0.001) (Table 2).
Pooled prevalence based on climate variables
Our analyses indicated that IPPs were most prevalent in schoolchildren in regions with annual rainfall of < 400 mm (0.260, 95% CI = 0.203–0.322) and humidity of < 30 (0.324 (95% CI = 0.254–0.398) with heterogeneities (I2 = 99; τ2 = 0.039; p < 0.001) and (I2 = 99; τ2 = 0.036; p < 0.001), respectively (Table 2).
Moreover, regions with an average temperature of > 20 °C represent the highest rate of prevalence (0.206, 95% CI = 0.166–0.250) with heterogeneity (I2 = 98; τ2 = 0.042; p < 0.001) (Table 2).
Based on climate, we found that regions with tropical rainforest climate had the highest pooled prevalence (0.291, 95% CI = 0.192–0.401) with heterogeneity (I2 = 98; τ2 = 0.046; p < 0.001) (Table 2).
Meta-regression
Heterogeneity was observed for humidity and year of publication. Accordingly, the test showed a statistically significance result for humidity (slop = 0.0009, p < 0.0061) and year of publication (slop = 0.0044, p < 0.0065) for all studies included in the current review (Fig. 4A, B).
A meta-regression graph for the prevalnce of intestinal protozoan parasites among Asian schoolchildren based on humidity (A), and year of publication (B). The pink line is the regression line, which was plotted based on the intercept and the slope of the regession model. The different colour bubbles represent the countries under study and their sizes indicates the effect size of each study
Publication bias and sensitivity analysis
There was no significant publication bias according to Egger’s test (t = 1.10, p = 0.271). Based on the Doi plot test, there was a minor asymmetry (LFK index: 1.27) (Fig. 5A, B). The sensitivity analysis results indicated that the impact of each study on the overall estimates of the current meta-analysis was not statistically significant (Supplementary Fig. 2).
A Sankey plot is designed using R software (version 3.6.1) to represent the taxa of intestinal protozoan parasites studies in Asian schoolchildren per taxonomic order, family, and genus (Supplementary Fig. 3).
Quality assessment
Following the quality assessment, it was revealed that among the 131 studies, 95 were classified as high quality (score of 7–9 points), and 36 were categorized as moderate quality (score of 4–6 points) (Supplementary Table 2).
Discussion
Protozoan parasites play a substantial role in the global public health challenge, particularly affecting developing regions where these infections are prevalent and pose a significant threat to community well-being, with children at heightened risk [1, 18].
To the best of our knowledge, this is the first systematic review and meta-analysis assessing the IPP prevalence among schoolchildren in Asia across the continent. In this study, the prevalence of IPPs among schoolchildren was determined to be relatively high (0.208%), which may be attributable to inadequate hygiene practices among these populations.
Although public health measures are generally stricter in developed countries compared to developing ones, minority groups, institutionalized individuals, and the immunocompromised remain at very high risk. This risk can extend to the broader population, making these groups a public health priority. Giardia duodenalis, Cryptosporidium spp., and Entamoeba spp. are the most frequently reported protozoa linked to enteric infections, and are primarily associated with outbreaks originating from food and water sources. Other protozoa, such as C. cayetanensis, B. coli, C. belli, and Blastocystis spp., are becoming significant causes of illness, particularly affecting travelers to developing regions, immunocompromised individuals, and young children. [1]. The public health sectors recommendations can be affected by the severity and pathogenesis of infections caused by protozoan parasites. The serious clinical symptoms, such as dehydration, malnutrition, or organ damage may lead to public health authorities prioritizing approaches for prevention, detection, and treatment. For instance, in regions with a high prevalence of water- and foodborne protozoan parasites like Giardia and Cryptosporidium, and outbreaks are possible, recommendations may prioritize measures such as enhancing food safety, improving water treatment and sanitation, and educating the public on hygiene practices [19,20,21].
Our results follow a prior study conducted among Asian children, which documented that G. duodenalis (15.1%) was the predominant intestinal parasite [22].
G. duodenalis is known to infect around 200 million people globally, with a higher prevalence rate among schoolchildren and in daycare centers [23]. The primary mode of transmission for G. duodenalis is through the fecal–oral route [24], and the primary sources of transmission include drinking water, food, and vegetables contaminated with cysts of the parasite [25, 26].
In a recent meta-analysis, the study revealed that the factors associated with an increased risk of giardiasis include being exposed to sewage or wastewater, consuming untreated drinking water, and engaging in recreational activities in water. Remarkably, the study also found that having contact with pets was a significant risk factor for giardiasis, particularly in children. Moreover, traveling to foreign countries was identified as another risk factor, especially in industrialized nations [22]. In children under the age of five, giardiasis can result in the development of severe, acute diarrhea, and several research studies have put evidence suggesting that chronic giardiasis, if left untreated or recurrent, may have lasting consequences in terms of growth retardation, impacting the long-term physical development of affected individuals [27]. This highlights the significance of addressing and managing giardiasis, especially in young children, to mitigate potential health and growth-related issues.
Our country-based analysis revealed that Lebanon and Tajikistan represent the highest prevalence rates of IPPs. However, we need to interpret it cautiously due to the low number of studies related to these two countries, which may cause bias in the results towards a higher prevalence rate. In Lebanon, many households lack adequate sanitation systems, leading to fecal contamination through ground seepage.
Lebanon faced an issue regarding wastewater management, which resulted in large-scale water pollution. According to research, nearly 74% of samples collected from rivers, which are the primary source of irrigation water in the country, surpassed the microbiological acceptability standards for this purpose. Moreover, in this country, the water sources are poorly managed, and freshwater is scarce, leading farmers to rely highly on untreated water sources [28].
In Tajikistan, it is estimated that approximately half of the rural households rely on untreated water sources for drinking, and this failure to meet the drinking water and poor sanitation standards in the country can partly be attributed to the contaminated water supply, which is associated with transmission of both waterborne and foodborne parasitic protozoa [29, 30].
Our findings based on different GBD regions found that Central Asia accounted for the highest prevalence rate of IPPs, which was in parallel with our country-based results. In this part of Asia, in Kyrgyzstan, a survey on children between the ages of 6 and 15 revealed that the overall prevalence of IPPs was found to be relatively high (41%) [31].
In Central Asian countries, the limited availability of high-quality drinking water is one of several issues related to public health. The aging and poor condition of water pipeline networks lead people to resort to alternative, often untreated, water sources. Additional factors also contribute significantly to the problem, including contamination of the water supply sources discharges from industrial and agricultural activities [32].
The infection rate is directly linked to factors such as sanitation, proper disposal of feces with good hygiene practices, access to safe drinking water, and other related factors [33, 34]. In regions with poor sanitary settings, public tap or standpipes as a source of drinking water supply emerged as a protective factor against IPPs, particularly concerning giardiasis [35].
Multiple reports have consistently highlighted the increased occurrence of IPPs in poor communities residing in countries with low to lower-middle income status, with a particular focus on various Asian nations. As anticipated, our review showed that the most significant incidence of IPPs in schoolchildren occurred in countries characterized by low and lower-middle income levels [36, 37].
Our review highlighted that regions with a tropical rainforest climate had the highest incidence of IPPs among schoolchildren in Asia, underscoring the significance of climate conditions as essential factors that affect the prevalence of IPPs in this context.
Parasitic intestinal infections are prevalent in tropical and subtropical regions, particularly in areas like Sub-Saharan Africa, Latin America, China, and East Asia. The warm and humid climate in these regions creates favorable conditions for the transmission and distribution of parasites, contributing to the high prevalence of these infections [6]. Furthermore, these regions experience significant population growth coupled with elevated poverty rates, which further escalates the risk of parasite transmission [38,39,40].
In the current study, the highest prevalence of IPPs was related to primary and elementary school-aged children, which might be attributed to their weaker immune systems, increased contact with soil and other contaminated materials, and their limited adherence to health standards [41].
Based on our analysis of various methods, the highest prevalence was related to the studies that employed a combination of direct smear, staining, and PCR techniques that are regarded as an approach for the qualitative diagnosis of intestinal protozoan parasites. PCR offers a higher detection sensitivity than light microscopy, making it particularly valuable for identifying a low number of parasites in stool samples [42]. Nevertheless, using a combination of microscopy with immunoassay or molecular methods has resulted in a notable improvement in both sensitivity and specificity over the last two decades [43].
Limitations
This study has faced particular limitations that should be noted. Firstly, there were limitations in the number of studies available for specific subgroups of Asian schoolchildren. In some cases, only one article addressed the prevalence of protozoan parasites for certain types of parasites. Secondly, our analyses might have been influenced by publication bias, stemming from the absence of or a limited number of studies available from specific geographical regions. Lastly, some of the studies in our analyses exhibited small-study effects, which can be attributed to factors such as limited sample size and the absence of a susceptible highly sensitive diagnostic technique. Despite these limitations, it's essential to acknowledge that this study offers the most comprehensive insights into the prevalence of intestinal protozoan parasites among Asian schoolchildren.
Conclusion
The final remarks summarize the findings of the examination and meta-analysis, affirming the prevailing risk factors like climate and socioeconomic aspects in certain Asian countries. This underscores the ongoing risk of protozoan infections to children, along with the necessity to monitor the protozoan infection trends/patterns. It is imperative to update public health recommendations for surveillance of protozoan infections particularly given the emerging links between certain protozoan pathogenesis and other chronic diseases, and infections.
Author contributors
MB, AVE, and PK contributed to the study design. ZM, ON, AKS, and MP searched for primary publications, screened, and appraised primary studies. ZM and AA extracted the data. MB, ON, and AVE contributed to the methodology. MO made a contribution to data analysis and interpretation. AVE, MB, and PK wrote the study manuscript. MB, AVE, and PK reviewed and edited the manuscript. All authors read the manuscript and participated in preparing the final version.
Availability of data and materials
The datasets used and/or analyzed during the current study are included in the manuscript.
References
Fletcher SM, Stark D, Harkness J, Ellis J. Enteric protozoa in the developed world: a public health perspective. Clin Microbiol Rev. 2012;25:420–49.
Eslahi AV, Olfatifar M, Zaki L, Pirestani M, Sotoodeh S, Farahvash MA, et al. The worldwide prevalence of intestinal helminthic parasites among food handlers: a systematic review and meta-analysis. Food Control. 2023;2: 109658.
WHO. Soil-transmitted helminth infections. WHO Fact Sheets. World Health Organization Geneva, Switzerland; 2020.
Siddig HS, Mohammed IA, Mohammed MN, Bashir AM. Prevalence of intestinal parasites among selected group of primary school children in Alhag Yousif Area, Khartoum, Sudan. Int J Med Res Heal Sci. 2017;6:125–31.
Gupta R, Rayamajhee B, Sherchan SP, Rai G, Mukhiya RK, Khanal B, et al. Prevalence of intestinal parasitosis and associated risk factors among school children of Saptari district, Nepal: a cross-sectional study. Trop Med Health. 2020;48:1–9.
Eslahi AV, Olfatifar M, Zaki L, Saryazdi AK, Barikbin F, Maleki A, et al. Global prevalence of intestinal protozoan parasites among food handlers: A systematic review and meta-analysis. Food Control. 2022;2: 109466.
Huh S, Yu J-R, Kim J-I, Gotov C, Janchiv R, Seo J-S. Intestinal protozoan infections and echinococcosis in the inhabitants of Dornod and Selenge, Mongolia (2003). Korean J Parasitol. 2006;44:171.
Sutrave S, Richter MH. The Truman Show for protozoan parasites: a review of in vitro cultivation platforms. PLoS Negl Trop Dis. 2021;15: e0009668.
Hemphill A, Müller N, Müller J. Comparative pathobiology of the intestinal protozoan parasites Giardia lamblia, Entamoeba histolytica, and Cryptosporidium parvum. Pathogens. 2019;8:116.
Ahmed T, Khanum H, Uddin MS, Barua P, Arju T, Kabir M, et al. Entamoeba histolytica, Giardia lamblia and Cryptosporidium spp. infection in children in an urban slum area of Bangladesh. Bioresearch Commun. 2016;2:175–81.
Eckmann L, Gillin FD. Microbes and microbial toxins: paradigms for microbial-mucosal interactions I. Pathophysiological aspects of enteric infections with the lumen-dwelling protozoan pathogen Giardia lamblia. Am J Physiol Liver Physiol. 2001;280:1–6.
Moges F, Belyhun Y, Tiruneh M, Kebede Y, Mulu A, Kassu A, et al. Comparison of formol-acetone concentration method with that of the direct iodine preparation and formol-ether concentration methods for examination of stool parasites. Ethiop J Heal Dev. 2010;24:2.
Venturini E, Scarso S, Prelazzi GA, Niccolai C, Bianchi L, Montagnani C, et al. Epidemiology and clinical features of intestinal protozoan infections detected by Real-time PCR in non-native children within an Italian tertiary care children’s hospital: a cross-sectional study. Travel Med Infect Dis. 2021;43: 102107.
Selçuk AA. A guide for systematic reviews: PRISMA. Turkish Arch Otorhinolaryngol. 2019;57:57.
Abdoli A, Olfatifar M, Eslahi AV, Moghadamizad Z, Samimi R, Habibi MA, et al. A systematic review and meta-analysis of protozoan parasite infections among patients with mental health disorders: an overlooked phenomenon. Gut Pathog. 2024;16:7.
Barendregt JJ, Doi SA. MetaXL user guide Version. 2016;4:2011–6.
Team RC. R Core Team R. R: A Language and Environment for Statistical Computing R Foundation for Statistical Computing, Vienna, Austria; 2016. ISBN 3–900051–07–0
Elmonir W, Elaadli H, Amer A, El-Sharkawy H, Bessat M, Mahmoud SF, et al. Prevalence of intestinal parasitic infections and their associated risk factors among preschool and school children in Egypt. PLoS ONE. 2021;16: e0258037.
Bourli P, Eslahi AV, Tzoraki O, Karanis P. Waterborne transmission of protozoan parasites: a review of worldwide outbreaks–an update 2017–2022. J Water Health. 2023;21:1421–47.
EFSA Panel on Biological Hazards (BIOHAZ), Koutsoumanis K, Allende A, Alvarez-Ordóñez A, Bolton D, Bover-Cid S, et al. Public health risks associated with food-borne parasites. EFSA J. 2018;16:e05495.
Omarova A, Tussupova K, Berndtsson R, Kalishev M, Sharapatova K. Protozoan parasites in drinking water: A system approach for improved water, sanitation and hygiene in developing countries. Int J Environ Res Public Health. 2018;15:495.
Kalavani S, Matin S, Rahmanian V, Meshkin A, Taghipour A, Abdoli A. Prevalence of Giardia duodenalis among Asian children: a systematic review and meta-analysis. Int Health. 2023;2:37.
Heresi GP, Murphy JR, Cleary TG. Giardiasis. Semin Pediatr Infect Dis. 2000. p. 189–95.
Dixon BR. Giardia duodenalis in humans and animals-Transmission and disease. Res Vet Sci. 2021;135:283–9.
Hunter PR, Thompson RCA. The zoonotic transmission of Giardia and Cryptosporidium. Int J Parasitol. 2005;35:1181–90.
Siwila J. The Triple Food-borne Protozoan Parasites: Cryptosporidium spp, Giardia duodenalis, Cyclospora cayetanensis Hope in Transmission Reduction. Curr Clin Microbiol Rep. 2023;2:1–9.
Maikai BV, Umoh JU, Lawal IA, Kudi AC, Ejembi CL, Xiao LH. Molecular characterizations of Cryptosporidium, Giardia, and Enterocytozoon in humans in Kaduna State. Nigeria Exp Parasitol. 2012;131:452–6.
El Safadi D, Osman M, Hanna A, Hajar I, Kassem II, Khalife S, et al. Parasitic Contamination of Fresh Leafy Green Vegetables Sold in Northern Lebanon. Pathogens. MDPI; 2023. p. 1014.
Matthys B, Bobieva M, Karimova G, Mengliboeva Z, Jean-Richard V, Hoimnazarova M, et al. Prevalence and risk factors of helminths and intestinal protozoa infections among children from primary schools in western Tajikistan. Parasit Vectors. 2011;4:1–13.
United Nations Development Programme Tajikistan R of T. Progress toward the Millennium Development Goals (Tajikistan 2003). United Nations Development Programme Dushanbe, Tajikistan; 2003.
Steinmann P, Usubalieva J, Imanalieva C, Minbaeva G, Stefiuk K, Jeandron A, et al. Rapid appraisal of human intestinal helminth infections among schoolchildren in Osh oblast. Kyrgyzstan Acta Trop. 2010;116:178–84.
Bekturganov Z, Tussupova K, Berndtsson R, Sharapatova N, Aryngazin K, Zhanasova M. Water related health problems in Central Asia A review. Water. 2016;8:219.
Kuete T, Yemeli FLS, Mvoa EE, Nkoa T, Somo RM, Ekobo AS. Prevalence and risk factors of intestinal helminth and protozoa infections in an urban setting of Cameroon: the case of Douala. Am J Epidemiol Infect Dis. 2015;3:36–44.
Hajare ST, Chekol Y, Chauhan NM. Assessment of prevalence of Giardia lamblia infection and its associated factors among government elementary school children from Sidama zone, SNNPR. Ethiopia PLoS One. 2022;17: e0264812.
Ndeezi G, Mor SM, Ascolillo LR, Tasimwa HB, Nakato R, Kayondo LN, et al. Giardia duodenalis in Ugandan Children Aged 9–36 Months in Kampala, Uganda: Prevalence and Associated Factors. Am J Trop Med Hyg. 2023;109:147.
Hotez PJ, Bottazzi ME, Strych U, Chang L-Y, Lim YAL, Goodenow MM, et al. Neglected tropical diseases among the Association of Southeast Asian Nations (ASEAN): overview and update. PLoS Negl Trop Dis. 2015;9: e0003575.
Fuhrimann S. Health risk assessment along wastewater recovery and reuse systems in Kampala, Uganda and Hanoi. Vietnam: University of Basel; 2015.
Antoszczak Michałand Steverding D, Huczyński A. Anti-parasitic activity of polyether ionophores. Eur J Med Chem. 2019;166:32–47.
Tidman R, Abela-Ridder B, de Castañeda RR. The impact of climate change on neglected tropical diseases: a systematic review. Trans R Soc Trop Med Hyg. 2021;115:147–68.
Ahmed A, Al-Mekhlafi HM, Choy SH, Ithoi I, Al-Adhroey AH, Abdulsalam AM, et al. The burden of moderate-to-heavy soil-transmitted helminth infections among rural malaysian aborigines: an urgent need for an integrated control programme. Parasit Vectors. 2011;4:1–7.
Eyamo T, Girma M, Alemayehu T, Bedewi Z. Soil-transmitted helminths and other intestinal parasites among schoolchildren in Southern Ethiopia. Res Rep Trop Med. 2019;137–43.
Guy RA, Xiao C, Horgen PA. Real-time PCR assay for detection and genotype differentiation of Giardia lamblia in stool specimens. J Clin Microbiol. 2004;42:3317–20.
Hamad I, Raoult D, Bittar F. Repertory of eukaryotes (eukaryome) in the human gastrointestinal tract: taxonomy and detection methods. Parasite Immunol. 2016;38:12–36.
Acknowledgements
We sincerely thank personnel from the Medical Microbiology Research Center, Qazvin University of Medical Sciences, Qazvin, Iran.
Funding
Open access funding provided by the Cyprus Libraries Consortium (CLC). This work was supported by the Medical Microbiology Research Center, Qazvin University of Medical Sciences, Qazvin, Iran and Jahrom University of Medical Sciences, Jahrom, Iran (contract no. IR.QUMS.REC.1402.150). The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Author information
Authors and Affiliations
Contributions
MB, AVE, and PK contributed to the study design. ZM, ON, AKS, and MP searched for primary publications, screened, and appraised primary studies. ZM and AA extracted the data. MB, ON, and AVE contributed to the methodology. MO made a contribution to data analysis and interpretation. AVE, MB, and PK wrote the study manuscript. MB, AVE, and PK reviewed and edited the manuscript. All authors read the manuscript and participated in preparing the final version.
Corresponding authors
Ethics declarations
Conflict of interest
We declare no competing interests.
Ethical approval
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdoli, A., Olfatifar, M., Eslahi, A.V. et al. Prevalence of intestinal protozoan parasites among Asian schoolchildren: a systematic review and meta-analysis. Infection (2024). https://doi.org/10.1007/s15010-024-02339-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s15010-024-02339-1