Abstract
The delayed titration of guideline-directed drug therapy (GDMT) is a complex event influenced by multiple factors that often result in poor prognosis for patients with heart failure (HF). Individualized adjustments in GDMT titration may be necessary based on patient characteristics, and every clinician is responsible for promptly initiating GDMT and titrating it appropriately within the patient’s tolerance range. This review examines the current challenges in GDMT implementation and scrutinizes titration considerations within distinct subsets of HF patients, with the overarching goal of enhancing the adoption and effectiveness of GDMT. The authors also underscore the significance of establishing a novel management strategy that integrates cardiologists, nurse practitioners, pharmacists, and patients as a unified team that can contribute to the improved promotion and implementation of GDMT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Heart failure (HF) is a global challenge in the cardiovascular field, especially chronic HF, which has high mortality rates and risks of recurrent hospitalizations [1]. The emergence of new types of anti-HF drugs in recent years has provided more ideas for the prevention and treatment of HF [2,3,4]. Early use of guideline-directed medical therapy (GDMT) is crucial for reducing the mortality rate of patients with HF, preventing patient readmissions, and improving their quality of life [5].
Large-scale, multicenter randomized controlled trials (RCTs) have conclusively shown that novel GDMTs, such as angiotensin receptor neprilysin inhibitors (ARNIs), β-blockers, mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter 2 inhibitors (SGLT2is), significantly improve the prognosis of patients suffering from heart failure with reduced ejection fraction (HFrEF) or heart failure with mildly reduced ejection fraction (HFmrEF) [3, 4, 6]. Recent studies have also demonstrated the effectiveness of SGLT2is in patients with heart failure with preserved ejection fraction (HFpEF) [7], breaking the conventional belief that HFpEF is a “drug-resistant” condition. In the specific context of African American patients with advanced HF, classified as New York Heart Association (NYHA) functional class III–IV HFrEF, the therapeutic regimen incorporating hydralazine and isosorbide dinitrate has emerged as a pivotal augmentation to optimal GDMT. This combination leverages the enhanced nitric oxide signaling pathway to induce vasodilation, effectively ameliorating symptoms while concurrently diminishing morbidity and mortality rates [3]. Additionally, for patients exhibiting persistent sinus tachycardia despite comprehensive GDMT, the combination of ivabradine offers a novel therapeutic approach. Ivabradine selectively inhibits sinoatrial node activity, thereby decelerating sinus rhythm without adversely impacting myocardial contractility or intracardiac conduction [8]. This nuanced approach is substantiated by the findings of the SHIFT trial, which revealed the efficacy of ivabradine in reducing both HF hospitalizations and mortality [hazard ratio (HR) 0.74 for both) relative to a placebo control group [9]. These insights underscore the imperative for a meticulously tailored pharmacotherapy and GDMT titration framework, catering to the distinct needs of various patients with HF subsets, thereby optimizing the clinical outcomes achievable through personalized treatment strategies.
The optimal titration sequence for GDMT remains controversial, but existing HF guidelines emphasize the importance of early and full-dose use of GDMT, provided patients can tolerate it [3, 4]. Unfortunately, clinical guidelines are not always fully followed in practice, and many patients receive doses below the minimum effective level or do not receive all the drugs that could improve their prognosis [10]. The delayed titration of GDMT can be attributed to various factors, including therapeutic inertia, complex treatment strategies, and varying interpretations of the guidelines by noncardiologists [11, 12]. The current challenge for clinicians is how to implement high-quality GDMT early and continuously.
2 Current Dilemma with GDMT
The 2021 European Society of Cardiology (ESC) guidelines [4] for HF have replaced the previous medication regimen with a new GDMT that emphasizes early and simultaneous initiation of ARNIs, β-blockers, MRAs, and SGLT2is. Cardiac resynchronization therapy, implantable cardioverter defibrillators or diuretic therapy may be subsequently added based on the patient’s response to treatment and edema [4]. Moreover, findings from the STRONG-HF study revealed that an expedited titration protocol for GDMT in patients experiencing acute HF yielded superior outcomes in diminishing rates of HF readmissions and all-cause mortality when juxtaposed with traditional approaches to GDMT adjustment (15.2% versus 23.3%, P = 0.0021) [13]. The timely and judicious application of GDMT has emerged as a critical determinant in enhancing the long-term prognostic outlook for individuals afflicted with HF.
Although the ESC guidelines [4] provide clear recommendations for HF management, their implementation in clinical practice is limited. We conducted a comprehensive search of the PubMed database covering the period from 1 January 2013 to January 2024. Our aim was to collate and analyze literature on the use of GDMT in the management of HF. The search strategy was meticulously designed to capture the most relevant studies using a combination of the key terms “guideline-directed medical therapy” OR “GDMT” AND “heart failure.” This approach facilitated the identification of pertinent articles that are now summarized in Table 1 of our review. The CHAMP-HF registry data [14] revealed that a significant proportion of outpatients were not prescribed recommended medications, and only a minority of patients received targeted doses of medication. Furthermore, advanced age, hypotension, poor cardiac function, renal insufficiency, and recent hospitalization for HF were frequently associated with lower dosages. Only 1% of eligible patients achieved all recommended target doses of medication. Similarly, the ESC HF long-term registry [15] showed that less than one-third of patients received medications at recommended doses despite high prescription rates of GDMT. In long-term follow-up, discontinuation of medication is very common, with up to 55% of patients discontinuing angiotensin-converting enzyme inhibitors (ACEIs) after 1 year of treatment [16]. This interruption in GDMT integrity may be a significant factor in the poor prognosis of patients with chronic HF.
The reasons for the hindered implementation of GDMT are currently believed to stem from three factors: patient-related factors, treatment-related factors, and healthcare-related factors (Fig. 1) [12]. Patients with good adherence typically receive higher doses of GDMT, which is a complex factor related to the patient’s personality, healthcare knowledge level, and family environment [17, 18]. Negative emotions, such as fatigue and depression resulting from long-term medication use, also play an important role, and regular psychological counseling and comfort may be overlooked blind spots. Furthermore, patients with HF with a high frailty burden are less likely to receive targeted doses of GDMT, which is associated with increased risks of HF hospitalization or death [19].
Factors contributing to the underuse of GDMT in patients with HF. This diagram illustrates the three principal components affecting the widespread adoption and utilization of GDMT for HF treatment: patient-associated factors, medication-related factors, and healthcare system-based factors. HF, heart failure; GDMT, guideline-directed medical therapy
Adverse drug reactions are a prevalent concern among clinicians, with renal impairment and hyperkalemia being the primary reasons for discontinuing ACEIs or angiotensin receptor inhibitors (ARBs) and MRAs, respectively [20, 21]. Moreover, asthma and bradycardia remain the most common causes of β-blocker cessation [12]. Full doses of ARNIs exhibit favorable antiventricular remodeling effects [22]. Nevertheless, some patients may experience a decrease in the estimated glomerular filtration rate (eGFR) during the initial use of ARNIs [23], and its potent hypotensive effect also restricts its application in patients with HF [24]. Despite evidence indicating that an ARNI-induced transient decrease in the eGFR does not result in increased adverse reactions and provides sustained benefits for patients with chronic HF [25], noncardiologists remain hesitant to prescribe sufficient ARNIs due to concerns regarding potential medical complications in these patients. Considering the inherent barriers and differences in development among various specialties, effectively promoting the concept of constantly updated clinical guidelines across disciplines remains a challenging task for future cross-disciplinary development.
Clinical inertia is a significant factor leading to a decline in healthcare service quality, while sequential healthcare services have been linked to improved clinical outcomes [26]. Especially in developing regions, the vast number of patients, complex affairs, and imbalanced medical education will further widen the gap in the quality of medical care services between regions. Therefore, providing specialized training for healthcare professionals and recommending the establishment of multisectoral HF rehabilitation centers can offer more substantial advantages to patients with HF.
3 Subgroups of Patients with HF and GDMT Exploration
3.1 Patients with HF and Hypotension
Hypotension and peripheral circulatory hypoperfusion often occur in chronic patients with HF with severe left ventricular insufficiency (e.g., postmyocardial infarction), and systemic hypotension is frequently associated with unfavorable clinical outcomes [27]. Apart from inducing inadequate organ perfusion, hypotension may impede the initiation or titration of GDMT, thereby further elevating the mortality risk in patients with HF. Canesin et al. [28] reported that patients with stable NYHA functional class IV HF and a mean systolic blood pressure below 105 mmHg had a 7.6-fold increase in mortality risk. Lee’s study also revealed a significant increase in the risk of death for patients with HFrEF with systolic blood pressure less than 110 mmHg [29]. The Carvedilol Prospective Randomized Cumulative Survival Trial conducted a substudy in 2004, which showed that patients with HF with systolic blood pressure below 95 mmHg had an 18% increased risk of death compared with those with systolic blood pressure above 125 mmHg (P < 0.001) [30]. Furthermore, a subsequent European study demonstrated a 24% higher mortality rate in patients with HF with systolic blood pressure at or below 120 mmHg [31]. Therefore, maintaining a minimum systolic blood pressure is crucial for reducing mortality risk in patients with HF.
Evidence suggests that standardized implementation of GDMT can significantly improve outcomes for patients with HF [3, 4]. However, the use of certain medications may lead to hypotension, which can interfere with GDMT implementation, resulting in a therapeutic dilemma between optimal GDMT treatment and potential harm from low blood pressure. The current clinical practice is to achieve the recommended target dose or maximum tolerated dose determined by clinical trials in the absence of evidence of organ hypoperfusion [32]. However, studies have shown that GDMT prescription rates are only 62.7% in patients with blood pressure less than 100 mmHg [33]. Therefore, it is necessary to consider when to intervene in patients with low blood pressure to maintain the minimum blood pressure and ensure complete implementation of GDMT.
ARNIs are the most potent antihypertensive drugs in GDMT, and their unique antihypertensive properties limit their use in patients with HF with low blood pressure [34]. Currently, there is a lack of high-quality clinical studies regarding the standard titration of ARNIs in hypotensive populations, but it is generally not recommended to administer ARNIs in patients with systolic blood pressure less than 100 mmHg to ensure adequate perfusion of vital organs [35]. The negative inotropic effect of β-blockers is a significant obstacle to their use in patients with HF. However, the COPERNICUS trial [30] revealed that β-blockers did not further reduce blood pressure in patients with systolic blood pressure between 85 and 95 mmHg compared with placebo. In fact, this group of patients experienced the greatest benefits from β-blocker treatment [30]. Nevertheless, it is obvious that reducing or discontinuing β-blockers is reasonable when patients exhibit significant hypotension or shock-related symptoms. The antihypertensive effects of MRAs and SGLT2is are mild and do not require discontinuation due to hypotension [36, 37], which can be used in patients with HF with low blood pressure for long-term maintenance. Recent research has shown that the significant benefits of GDMT in patients with HFrEF with normal blood pressure extend to those with normal renal function and hypotension, and the standard implementation of GDMT is associated with improved clinical outcomes even in hospitalized patients with lower blood pressure [33]. Moreover, drug-induced orthostatic hypotension emerges as a notable concern during the titration of GDMT, particularly among the elderly patient cohort. Existing clinical guidelines advocate for the preliminary assessment of orthostatic hypotension prior to the commencement or escalation of antihypertensive regimens, in addition to ongoing surveillance of medication safety throughout the therapeutic course [38]. Interestingly, recent evidence from a comprehensive meta-analysis revealed that intensified antihypertensive therapy can also significantly reduce the risk of cardiovascular or all-cause mortality in patients with orthostatic hypotension (HR 0.81) or postural hypotension (HR 0.80) [39]. This finding fuels the ongoing discourse regarding the imperative for routine orthostatic hypotension screening before the initiation of GDMT. While opinions diverge, there is a consensus on the prudence of modifying antihypertensive drug dosages in response to the manifestation of orthostatic hypotension symptoms. This strategy underscores the delicate balance required in optimizing blood pressure control while mitigating the risk of adverse effects, thereby ensuring a patient-centric approach to the management of hypertension within the framework of GDMT.
3.2 Patients with HF and Renal Dysfunction
Renal dysfunction, indicated by elevated blood creatinine or decreased eGFR, is commonly associated with disease progression and poor clinical outcomes [40, 41]. In chronic conditions, such as HF, diabetes, and hypertension, a continuous decline in eGFR indicates progressive loss of renal units, potentially related to intraglomerular hypertension or chronic hypoperfusion damage [42]. In patients with chronic HF, chronic renal dysfunction resulting from cardiorenal syndrome is frequently observed, often impeding the standard titration of GDMT. However, renal function impairment does not always indicate a poor long-term prognosis [43]. For example, in patients with acute HF, renal impairment may only reflect short-term changes in hemodynamic status and not necessarily permanent loss of functional renal units [44].
ACEIs and ARBs have been associated with a potential decrease in the eGFR. In patients with poor renal compensatory ability, the use of ACEIs or ARBs may lead to a rapid decline in renal function [45]. Although it has been established that a reduction in the eGFR following the administration of ACEIs does not impact long-term benefits [46], there is limited evidence on the role of renin–angiotensin–aldosterone system (RAAS) inhibitors in patients with HF, and the safety of these agents should be confirmed through observational studies with extended follow-up periods. According to a subgroup analysis of patients with HF and chronic kidney disease (CKD), compared with enalapril, ARNIs significantly delayed the decrease in the eGFR by 23.5% while also reducing the risk of cardiorenal events and mortality [47]. Similarly, the PARAGON-HF trial revealed that ARNIs could reduce the risk of combined endpoint events, including HF hospitalization and cardiovascular death, by 21% in patients with an eGFR < 60 ml/min/1.732 [48]. Although increasing evidence supports the renal protective effects of ARNIs, the temporary decline in renal function during their initiation may raise concerns among some clinicians. This concern varies among specialists and may stem more from the desire to avoid potential medical disputes rather than a lack of clinical evidence. It is important to address these concerns and encourage further titration of ARNIs based on individual patient factors and close monitoring.
Spironolactone can effectively suppress the harmful effects of aldosterone on mesangial cell proliferation, podocyte injury, sclerotic changes, and arteriolar hyalinosis [49]. When used in combination with RAAS inhibitors, it can further enhance the clinical outcomes of patients with HF. The EMPHASIS-HF study demonstrated that MRA can significantly reduce the risk of hospitalization for HF and cardiovascular death in patients with an eGFR < 60 ml/min/1.73 m2 [50]. Furthermore, the EPHESUS trial showed that MRA may cause a slight decrease in eGFR in the short term, but it does not impact the overall rate of eGFR decrease [51]. MRA therapy is generally safe for patients with HF with normal or mildly impaired renal function. However, due to the lack of sufficient data on safety and efficacy, the use of MRA is not recommended for patients with severely impaired renal function (eGFR ≤ 30 ml/min/1.73 m2) [52].
SGLT2i has been demonstrated to reduce cardiovascular death risk in all types of patients with HF, particularly in patients with HFpEF [53]. Therefore, SGLT2i therapy should be initiated early and at an appropriate dosage for all patients with HF. Although there may be a temporary decrease in the eGFR during the initial stages of SGLT2i therapy, long-term follow-up data indicate that SGLT2i usage can slow the rate of eGFR decrease, ultimately leading to improved renal outcomes in patients with HF [54, 55].
In contrast to ARNIs, MRAs, and SGLT2is, β-blockers do not cause rapid deterioration of the eGFR in the short term [56]. Studies have shown that bisoprolol significantly reduces the combined risk of all-cause mortality and hospitalization for HF exacerbation in patients with CKD without interacting with ACEIs or ARBs, which could worsen renal function [57]. This finding supports the long-term implementation of bisoprolol in patients with HF. Some studies have indicated that the cardiovascular protective effects of bisoprolol may extend to stage 3B CKD, but there is currently a lack of research data on its effectiveness in patients with stage 4 and stage 5 CKD [58].
There is currently a lack of clinical evidence regarding evaluation criteria and treatment methods for renal function deterioration after initiating or implementing GDMT, but treatment methods for RAAS inhibitor-induced renal function deterioration may be used as a reference [59, 60]. The absolute value of serum creatinine is not a key indicator, and attention should be given to its dynamic changes. If the serum creatinine level increases to less than 30% of the baseline value, the ARNI can continue to be used. If the serum creatinine level exceeds 30% of the baseline level, the dose of ARNIs should be promptly reduced or discontinued, and the cause should be identified. If the serum creatinine level exceeds 50% of the baseline level, the ARNI should be discontinued [61]. In patients with HF with nonsevere renal impairment, early and aggressive use of β-blockers, MRAs, and SGLT2is is recommended [4]. Regular monitoring of renal function after initiating or implementing GDMT and adjusting individualized implementation plans based on renal function can provide greater benefits to patients.
3.3 Patients with HF and Electrolyte Imbalance
Patients with HF who self-administer diuretics for long-term volume management are at high risk of electrolyte imbalances, particularly potassium loss or accumulation. These electrolyte disturbances can interfere with the titration of GDMT, potentially leading to a reduction in the long-term use of certain medications, such as MRAs. Additionally, in developing regions, regular monitoring of electrolytes and timely feedback to physicians to adjust treatment plans may pose challenges, potentially hindering the implementation of GDMT.
Both ARNIs and MRAs can cause hyperkalemia, which may be a concern for doctors in patients with HF with renal dysfunction [62]. The EMPHASIS-HF trial showed that MRA still had significant cardiovascular benefits in patients with HF with hyperkalemia [63]. Recent studies by Karola et al. [64] have shown that the combination of ARNI and MRA not only improves cardiovascular outcomes in patients but also does not increase the incidence of renal function deterioration or hyperkalemia. Akshay et al. [65] also reported that the risk of hyperkalemia was significantly lower when ARNI was used in combination with MRA than when it was used in combination with enalapril and MRA. In addition, a secondary analysis of the EMPEROR-Reduced trial revealed that the combination of MRA and SGLT2i improved patients’ symptoms and health status, significantly reduced cardiovascular mortality, and had no significant adverse effects [66]. Starting MRA and SGLT2i simultaneously can reduce the risk of hyperkalemia, slow the progression of kidney disease, and reduce the probability of MRA discontinuation [67]. These studies provide reliable evidence for the safety of early implementation of GDMT.
Although existing studies suggest a lower incidence of hyperkalemia during GDMT implementation [68, 69], clinicians should still be concerned about hyperkalemia as a life-threatening emergency. Regular electrolyte monitoring and medication dose adjustment are cost-effective but require good patient compliance. Active health education and promotion during hospitalization are crucial for patient awareness. Severe hyperkalemia should prompt further endocrine evaluation and a review of concomitant medical therapies and diet [58]. Additionally, preventing the use of hypokalemia drugs in patients with HF appears feasible. Sodium zirconium cyclosilicate (SZC), a novel selective cation exchanger, immediately decreases serum potassium by approximately 1.0 mEq/l in patients with HF with hyperkalemia [70, 71]. Short-term studies have not reported adverse events, including hypokalemia [72], but caution is still necessary when using SZC in patients with HF with mild hyperkalemia to prevent iatrogenic hypokalemia. Therefore, individualized serum potassium monitoring and the use of new potassium-binding agents in patients with HF at increased risk of hyperkalemia can optimize RAAS inhibition therapy and more effectively manage hyperkalemia.
3.4 Patients with HF and Atrial Fibrillation
Atrial fibrillation and HF are twin diseases with complex interactive mechanisms that promote each other’s development [73, 74]. The combination of these two diseases leads to a significant increase in incidence and mortality. In addition to standard GDMT titration, patients with atrial fibrillation and HF also require standardized management of atrial fibrillation through the “ABC pathway,” which includes the use of anticoagulants, control of heart rate and rhythm, and management of complications [75].
The MISOAC-AF database [76] revealed that only 30.7% of patients with atrial fibrillation and HF received standard doses of GDMT at 3 months postdischarge. Lower baseline eGFR values were also associated with reduced prescription of HF disease-modifying drugs, except for bisoprolol. Zak et al. [77] reported that the use of GDMT was associated with lower mortality rates in patients with atrial fibrillation and HF. Better adherence to guidelines was associated with better 60 day and 1 year prognoses in patients with atrial fibrillation and HF [78]. Observational studies have shown that rhythm control of atrial fibrillation is associated with improved quality of life and functional capacity and decreased all-cause mortality in patients with HF [79]. Furthermore, the AATAC-AF study, CASTLE-AF study, and CABANA study have all demonstrated that ablation procedures are superior to drug therapy in patients with atrial fibrillation and HF [80,81,82], providing more robust evidence for early rhythm control strategies for atrial fibrillation. Combining early rhythm control treatment with standard GDMT may result in better clinical outcomes for patients with concomitant atrial fibrillation and HF.
3.5 Patient Medication Adherence: The Unsung Challenge in HF Management
An “ideal” treatment plan not only involves physicians developing reasonable strategies but also necessitates patient cooperation and adherence, reflecting the fundamental trust between doctors and patients [83]. Medication nonadherence is prevalent among patients with HF, particularly elderly patients, and is potentially linked to various comorbidities and complex treatment plans. Poor medication adherence is significantly associated with all-cause mortality and major cardiovascular hospitalization outcomes [for adherence < 80%: HR 2.19, 95% confidence interval (CI) 1.72‒2.79, P < 0.0001] [84]. Approximately 36% of caregivers, 56% of patients, and 63% of healthcare providers regard comorbidities, suboptimal drug treatment, and poor medication adherence as critical factors contributing to patient readmission [85]. Comorbidities can complicate HF management, requiring modifications to GDMT for effective treatment. Furthermore, the illness itself and depression caused by prolonged medication cannot be disregarded. Patients experiencing depressive symptoms and lacking knowledge about their condition encounter significant challenges in adhering to treatment. In another study among elderly patients with HF, the overall adherence rate was 72%, which improved with increased education and disease awareness and was inversely associated with depressive symptoms [86]. Therefore, medication adherence should be prioritized in regular follow-up care for patients with HF, and measures to enhance medication adherence should be a crucial component of the self-care plans of patients with HF [87].
Currently, the majority of patients have access to a wide range of medical information. However, for patients lacking medical literacy, it is challenging to sift through this vast amount of information and identify what is truly effective. Regular health education during hospitalization has proven to be an effective approach for addressing this issue. It not only enhances patients’ fundamental understanding of their condition but also facilitates the promotion of GDMT implementation through the use of printed materials such as booklets, flyers, and posters within the ward [12]. Moreover, leveraging mobile phones, educational websites, and social media for postdischarge education can further enhance patients’ adherence to treatment [88, 89]. An RCT highlighted that telehealth utilization in HF management enhances patient adherence to treatment, reduces rehospitalization, and diminishes treatment costs [90]. The coronavirus disease 2019 (COVID-19) pandemic further emphasized the efficacy of remote healthcare in overcoming numerous care-related obstacles for patients with HF [91], indicating the viability of improving GDMT adherence via remote monitoring. Economic considerations are also crucial. Financial constraints significantly impede the adoption of GDMT among patients with HF [92]. The integration of ARNIs and SGLT2is into GDMT introduces a notable economic challenge due to their elevated costs. Insights from the Medical Expenditure Panel Survey revealed that approximately one-third of patients with HF experience subjective financial distress due to medical expenses, with 13.2% completely unable to bear such costs [93, 94]. Individuals aged 65 years or younger and those with limited educational attainment are particularly vulnerable and their financial strain is often exacerbated by a lack of insurance coverage [94]. This burden is even more acute in developing nations, where inadequate insurance coverage or minimal reimbursement compels patients to either forego essential medications or resort to less expensive alternatives, such as substituting ARNIs with ACEIs or ARBs. These dynamics highlight the critical need for improvements in the quality and coverage of insurance schemes, especially in light of the expanding spectrum of HF treatment options. Therefore, encouraging comprehensive public reimbursement and providing financial support for HF medication can alleviate the financial burden on patients while fostering wider implementation of GDMT [12]. Additionally, if psychological disorders such as anxiety or depression are identified during follow-up, it is important to offer psychological counseling and recommend that patients seek assistance at a dedicated counseling clinic. In some cases, drug or behavioral therapy, under the guidance of a psychiatric specialist, may be necessary.
Enhancing patients’ self-care awareness and offering psychological counseling play pivotal roles. These interventions enable patients to develop a deeper understanding of their condition, diligently follow treatment plans, and implement vital lifestyle modifications to alleviate the burden of their illness.
3.6 Physicians’ Understanding and Implementation of the GDMT
Doctors play a crucial role as intermediaries between GDMT and patients, and the extent to which physicians understand and implement GDMT significantly impacts its titration strategies. Doctors must recognize that they are responsible for implementing and adjusting all mandatory classes of HF medications [12]. The implementation of the GDMT lacks consistency across various healthcare systems. Physicians working in advanced therapeutic centers or specialized HF clinics have faster access to the latest guideline information and are more receptive to GDMT principles. However, doctors in economically disadvantaged regions face challenges in promptly accessing up-to-date guidelines, leading to delays in GDMT implementation [95]. Moreover, the concept of GDMT implementation appears to be less emphasized among noncardiology specialists, who may exhibit greater concerns regarding potential complications. Nevertheless, further data are required to validate these observations.
At present, the dissemination of guideline principles primarily relies on periodic guideline lectures organized by the National HF Association. Providing appropriate educational courses, workshops, and materials (such as translated versions of ESC guidelines, national guidelines, and treatment updates) tailored to healthcare professionals of varying expertise levels can enhance the implementation of GDMT [12]. However, this approach has inherent delays in information delivery and does not ensure that all doctors accurately grasp the implementation principles of GDMT outlined in the guidelines. HF guideline lectures tend to attract more cardiologists and are not extensively promoted among noncardiology specialists, potentially resulting in insufficient understanding of GDMT implementation among nonspecialist doctors. Additionally, the evidence supporting guideline development often stems from large-scale, high-quality clinical trials. However, in reality, high-risk patients with HF are unlikely to receive the recommended treatments, making them ineligible for evidence-based therapies [96]. One contributing factor to the greater success of implementation strategies in clinical trials is the inclusion of younger, more proactive patients with fewer comorbidities, which may overestimate the potential for optimizing GDMT [97]. Therefore, regular monitoring of patients’ improvement in cardiac function and complications during the actual implementation of GDMT is advantageous for its implementation and adjustment.
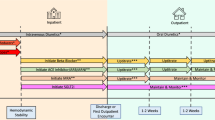
Historically, the crucial roles of nurses and pharmacists in the long-term management of patients with HF have often been disregarded. However, a study conducted by Andrea et al. [98] revealed that when led by nurses, the implementation of GDMT can significantly enhance the optimization of β-blockers and reduce hospitalization rates. Despite evidence supporting the effectiveness of telehealth, its integration into routine clinical practice remains limited, possibly due to the absence of regular professional theoretical training for nurses [12] and their engagement in time-consuming nonclinical tasks, which significantly drains nursing resources. This issue is particularly pronounced in developing countries. Nurse practitioner (NP)-led interventions and care programs have shown promise in managing patients with HF. NPs, as licensed healthcare professionals, provide comprehensive, patient-centered care that improves patient self-care awareness and quality. Their roles include diagnosing and treating patients, prescribing medications, making referrals, managing acute and chronic conditions, and focusing on health promotion, disease prevention, and patient satisfaction [99]. A cohort study during the COVID-19 pandemic demonstrated that an NP-led pilot telehealth program enabled 75% of patients with HF to achieve optimal GDMT within 2 months, with a significant improvement in the left ventricular ejection fraction observed in 77% of patients during follow-up [100]. Additionally, the pivotal roles of nurses and pharmacists in the long-term management of patients with HF have sparked discussions about the need for innovative management strategies. A meta-analysis demonstrated that interventions led by pharmacists and nurses were effective in improving guideline adherence in initiating or implementing GDMT [101]. To optimize the initiation or implementation of GDMT in future patients with HF, a novel management strategy that involving cardiologists, NPs, pharmacists, and patients as a cohesive unit may prove to be the most effective choice (Fig. 2).
Comparative usage and remedial measures for GDMT in different scenarios. This graphic representation utilizes color-coded circles to symbolize four distinct types of GDMT medications. The dimension of each circle directly correlates with the degree of clinical utilization priority: the larger the circle is, the less likely the medication is to be discontinued, and conversely, smaller circles indicate a greater likelihood of discontinuation. ARNI, angiotensin receptor neprilysin inhibitor; GDMT, guideline-directed medical therapy; MRA, mineralocorticoid receptor antagonist; NPs, nurse practitioners; SGLT2i, sodium-glucose cotransporter 2 inhibitors
3.7 Implementation of the GDMT in other Scenarios
The practical application of GDMT across diverse clinical scenarios reveals the intricacies inherent in adapting standard titration protocols to meet the unique needs of specific patient groups. Among these, special consideration is warranted for populations such as pregnant women, elderly individuals with HF, and individuals suffering from concurrent diabetes.
Particularly challenging is the management of peripartum cardiomyopathy (PPCM), a condition characterized by the emergence of maternal HF with systolic dysfunction—evidenced by a left ventricular ejection fraction below 45%—occurring in the final month of pregnancy or within the initial 5 months postdelivery, in the absence of any preexisting cardiac dysfunction [102]. PPCM accounts for approximately 60% of cardiogenic shock incidents during pregnancy or the early postpartum period [103]. The clinical approach to PPCM necessitates a nuanced, multidisciplinary strategy due to the absence of large-scale RCTs that specifically address GDMT titration in this cohort. Consequently, the therapeutic regimen for PPCM often mirrors that for HFrEF, albeit with significant modifications to ensure fetal safety [104]. Medications such as ACEIs, ARBs, and MRAs are contraindicated prior to childbirth. Instead, highly selective β1-adrenergic receptor blockers are recommended. The use of ARNIs and SGLT2is in the postpartum phase is growing, although definitive data on their safety and efficacy remain forthcoming [102].
Most conventional HF medications are deemed safe for breastfeeding, yet the safety profiles of newer pharmacological agents have yet to be fully established. GDMT should be continued for a duration of 12–24 months following the complete recovery of cardiac function. Typically, a significant proportion of women experience an improvement in the left ventricular ejection fraction to over 50% within 6 months of diagnosis. However, the timeline for returning to normative cardiac function varies widely, with some individuals experiencing protracted recovery periods and others never fully regaining prior cardiac health [105]. The mortality rate associated with PPCM can reach 20% [102], underscoring the critical need for women with symptomatic HF to avoid pregnancy. These findings highlight the imperative for tailored GDMT approaches that accommodate the complex medical and ethical considerations unique to specific patient demographics.
The increasing incidence of HF in the aging population underscores the need for refined GDMT titration strategies tailored to elderly individuals. A subanalysis of the STRONG-HF trial underscores the potential of accelerated GDMT titration to significantly curtail the rates of all-cause mortality or HF rehospitalization among elderly patients with HF, despite yielding only modest enhancements in quality of life [106]. Nonetheless, the corpus of evidence-based medicine on the nuances of GDMT titration within this demographic remains scant. The intricacies of managing GDMT in elderly individuals with HF are markedly more complex than those in their middle-aged counterparts. Elderly patients frequently present with a spectrum of comorbidities, compromised nutritional status, and frailty, predisposing them to a heightened risk of adverse events such as hypotension, electrolyte disturbances, or renal dysfunction amid GDMT titration [107]. Furthermore, the preference for quality of life improvements often takes precedence over the extension of survival among many in this cohort. Consequently, clinicians are generally cautious in modulating pharmacotherapy for elderly patients with HF in the clinical milieu [108]. This delineates the criticality of embracing shared decision-making paradigms, engaging both healthcare practitioners and elderly patients with HF in a dialog that respects individual preferences and therapeutic objectives. To conclude, the optimization of GDMT titration for elderly patients with HF necessitates a comprehensive approach that transcends evidence-based guidelines to incorporate the distinct clinical attributes and value systems of this patient group.
Type 2 diabetes mellitus (T2DM) is recognized as an independent risk factor for HF, with coexistent HF and T2DM associated with a poorer prognosis than HF alone [109]. Optimal initiation and adjustment of the GDMT are paramount for improving outcomes in this patient group. Metformin, the first-line therapy for T2DM, is advised with caution in acute HF settings but can be effectively integrated into GDMT for those with stable conditions [110]. ARNIs have been shown to reduce cardiovascular death or HF hospitalization by approximately 20% compared with ACEIs, with consistent benefits in the diabetic population [111]. Clinical evidence indicates that SGLT2is significantly decrease the risk of cardiovascular death across HF spectra, irrespective of T2DM status [54, 112, 113]. The complexity of managing T2DM in patients with HF necessitates vigilant monitoring due to the increased risk of renal and electrolyte imbalances. Glucagon-like peptide-1 receptor agonists (GLP-1RAs), a newer class of antidiabetic medication, have gained attention for their potential in HF management. The STEP-HFpEF trial demonstrated that semaglutide significantly ameliorated weight and HF symptoms and reduced HF events in obese patients with HFpEF [114]. However, it is worth noting that the direct impact of GLP-1RAs on HF risk reduction in patients with T2DM remains debated. Outcome trials have not shown a significant change in HF hospitalization rates with liraglutide, exenatide, semaglutide, lixisenatide, or dulaglutide [110]. Additionally, a recent study revealed that the effects of liraglutide versus placebo on major adverse cardiovascular events were consistent in patients with (HR 0.81, 95% CI 0.65‒1.02) and without (HR 0.88, 95% CI 0.78‒1.00) a history of HF (P interaction = 0.53) [115]. Despite these findings, the indirect benefits of GLP-1RAs, such as weight and blood pressure reduction, lowering hypoglycemic risk, and mitigating atherosclerotic cardiovascular disease, are crucial in devising an effective treatment plan for patients with HF with T2DM.
4 Conclusions
HF poses a significant challenge in the field of cardiovascular diseases. Although the novel GDMT strategy has brought hope for patients with HF, its practical application has been suboptimal, leading to unfavorable long-term outcomes. The delayed implementation of GDMT primarily arises from patient-related factors, treatment-related factors, and healthcare-related factors. Tailored titration strategies for GDMT exist for patients with HF with diverse characteristics. However, initiating GDMT early and ensuring adequate implementation, while considering individual tolerance, significantly reduces cardiovascular mortality in patients with HF. Furthermore, enhancing patient education and providing psychological support, improving the understanding of GDMT principles among noncardiologists, and establishing a novel management strategy that integrates cardiologists, NPs, pharmacists, and patients as a unified team can contribute to the improved promotion and implementation of GDMT.
References
Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GMC, Coats AJS. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2023;118(17):3272–87.
Bauersachs J. Heart failure drug treatment: the fantastic four. Eur Heart J. 2021;42(6):681–3.
Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: executive summary: A report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation. 2022;145(18):e876–94.
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–726.
G. Savarese, T. Kishi, O. Vardeny, S. Adamsson Eryd, J. Bodegård, et al. Heart failure drug treatment—inertia, titration, and discontinuation: a multinational observational study (EVOLUTION HF). JACC Heart Fail. 11(1) (2023).
Straw S, Cole CA, McGinlay M, Drozd M, Slater TA, et al. Guideline-directed medical therapy is similarly effective in heart failure with mildly reduced ejection fraction. Clin Res Cardiol. 2023;112(1):111–22.
Spertus JA, Birmingham MC, Nassif M, Damaraju CV, Abbate A, et al. The SGLT2 inhibitor canagliflozin in heart failure: the CHIEF-HF remote, patient-centered randomized trial. Nat Med. 2022;28(4):809–13.
Riccioni G. Ivabradine: from molecular basis to clinical effectiveness. Adv Ther. 2010;27(3):160–7.
Swedberg K, Komajda M, Böhm M, Borer JS, Ford I, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet. 2010;376(9744):875–85.
Abdin A, Anker SD, Butler J, Coats AJS, Kindermann I, et al. ‘Time is prognosis’ in heart failure: time-to-treatment initiation as a modifiable risk factor. ESC Heart Fail. 2021;8(6):4444–53.
Abdin A, Bauersachs J, Soltani S, Eden M, Frey N, Böhm M. A practical approach to the guideline-directed pharmacological treatment of heart failure with reduced ejection fraction. ESC Heart Fail. 2023;10(1):24–31.
Seferović PM, Polovina M, Adlbrecht C, Bělohlávek J, Chioncel O, et al. Navigating between Scylla and Charybdis: challenges and strategies for implementing guideline-directed medical therapy in heart failure with reduced ejection fraction. Eur J Heart Fail. 2021;23(12):1999–2007.
Mebazaa A, Davison B, Chioncel O, Cohen-Solal A, Diaz R, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. 2022;400(10367):1938–52.
Greene SJ, Butler J, Albert NM, DeVore AD, Sharma PP, et al. Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF registry. J Am Coll Cardiol. 2018;72(4):351–66.
Maggioni AP, Anker SD, Dahlström U, Filippatos G, Ponikowski P, et al. Are hospitalized or ambulatory patients with heart failure treated in accordance with European Society of Cardiology guidelines? Evidence from 12,440 patients of the ESC Heart Failure Long-Term Registry. Eur J Heart Fail. 2013;15(10):1173–84.
Savarese G, Bodegard J, Norhammar A, Sartipy P, Thuresson M, et al. Heart failure drug titration, discontinuation, mortality and heart failure hospitalization risk: a multinational observational study (US, UK and Sweden). Eur J Heart Fail. 2021;23(9):1499–511.
Samsky MD, Lin L, Greene SJ, Lippmann SJ, Peterson PN, et al. Patient perceptions and familiarity with medical therapy for heart failure. JAMA Cardiol. 2020;5(3):292–9.
Trinkley KE, Kahn MG, Allen LA, Haugen H, Kroehl ME, et al. Patient treatment preferences for heart failure medications: a mixed methods study. Patient Prefer Adherence. 2020;14:2225–30.
Khan MS, Segar MW, Usman MS, Singh S, Greene SJ, et al. Frailty, guideline-directed medical therapy, and outcomes in HFrEF: from the GUIDE-IT Trial. JACC Heart Fail. 2022;10(4):266–75.
Rossignol P, Lainscak M, Crespo-Leiro MG, Laroche C, Piepoli MF, et al. Unravelling the interplay between hyperkalaemia, renin-angiotensin-aldosterone inhibitor use and clinical outcomes. Data from 9222 chronic heart failure patients of the ESC-HFA-EORP Heart Failure Long-Term Registry. Eur J Heart Fail. 2020;22(8):1378–89.
Komajda M, Schöpe J, Wagenpfeil S, Tavazzi L, Böhm M, et al. Physicians’ guideline adherence is associated with long-term heart failure mortality in outpatients with heart failure with reduced ejection fraction: the QUALIFY international registry. Eur J Heart Fail. 2019;21(7):921–9.
Tsutsui H. Recent advances in the pharmacological therapy of chronic heart failure: Evidence and guidelines. Pharmacol Ther. 2022;238: 108185.
Shi Y, Wang Y, Chen J, Lu C, Xuan H, et al. Effects of angiotensin receptor neprilysin inhibitor on renal function in patients with heart failure: a systematic review and meta-analysis. Postgrad Med J. 2021;postgradmedj-2021-140132. https://doi.org/10.1136/postgradmedj-2021-140132.
Yamamoto K, Rakugi H. Angiotensin receptor-neprilysin inhibitors: comprehensive review and implications in hypertension treatment. Hypertens Res. 2021;44(10):1239–50.
Chatur S, Claggett BL, McCausland FR, Rouleau J, Zile MR, et al. Variation in renal function following transition to sacubitril/valsartan in patients with heart failure. J Am Coll Cardiol. 2023;81(15):1443–55.
Pereira Gray DJ, Sidaway-Lee K, White E, Thorne A, Evans PH. Continuity of care with doctors-a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8(6): e021161.
Levy WC, Mozaffarian D, Linker DT, Sutradhar SC, Anker SD, et al. The Seattle Heart Failure model: prediction of survival in heart failure. Circulation. 2006;113(11):1424–33.
Canesin MF, Giorgi D, Oliveira MTd, Wajngarten M, Mansur AJ, et al. Ambulatory blood pressure monitoring of patients with heart failure. A new prognosis marker. Arq Bras Cardiol. 2002;78(1):83–9.
Lee TT, Chen J, Cohen DJ, Tsao L. The association between blood pressure and mortality in patients with heart failure. Am Heart J. 2006;151(1):76–83.
Rouleau JL, Roecker EB, Tendera M, Mohacsi P, Krum H, et al. Influence of pretreatment systolic blood pressure on the effect of carvedilol in patients with severe chronic heart failure: the Carvedilol Prospective Randomized Cumulative Survival (COPERNICUS) study. J Am Coll Cardiol. 2004;43(8):1423–9.
Metra M, Torp-Pedersen C, Swedberg K, Cleland JGF, Di Lenarda A, et al. Influence of heart rate, blood pressure, and beta-blocker dose on outcome and the differences in outcome between carvedilol and metoprolol tartrate in patients with chronic heart failure: results from the COMET trial. Eur Heart J. 2005;26(21):2259–68.
Miller WL, Skouri HN. Chronic systolic heart failure, guideline-directed medical therapy, and systemic hypotension—less pressure but maybe more risk (does this clinical scenario need more discussion?). J Card Fail. 2009;15(2):101–7.
Izumi K, Kohno T, Goda A, Takeuchi S, Shiraishi Y, et al. Low blood pressure and guideline-directed medical therapy in patients with heart failure with reduced ejection fraction. Int J Cardiol. 2023;370:255–62.
Campbell DJ. Long-term neprilysin inhibition—implications for ARNIs. Nat Rev Cardiol. 2017;14(3):171–86.
Rosano GMC, Moura B, Metra M, Böhm M, Bauersachs J, et al. Patient profiling in heart failure for tailoring medical therapy. A consensus document of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2021;23(6):872–81.
Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413–24.
Khan J, Graham FJ, Masini G, Iaconelli A, Friday JM, et al. Congestion and use of diuretics in heart failure and cardiomyopathies: a practical guide. Curr Cardiol Rep. 2023;25(5):411–20. https://doi.org/10.1007/s11886-023-01865-y.
Lahrmann H, Cortelli P, Hilz M, Mathias CJ, Struhal W, Tassinari M. EFNS guidelines on the diagnosis and management of orthostatic hypotension. Eur J Neurol. 2006;13(9):930–6.
Juraschek SP, Hu J-R, Cluett JL, Ishak AM, Mita C, et al. Orthostatic hypotension, hypertension treatment, and cardiovascular disease: an individual participant meta-analysis. JAMA. 2023;330(15):1459–71.
Giamouzis G, Kalogeropoulos AP, Butler J, Karayannis G, Georgiopoulou VV, et al. Epidemiology and importance of renal dysfunction in heart failure patients. Curr Heart Fail Rep. 2013;10(4):411–20.
Jingi AM, Nkoke C, Noubiap JJ, Teuwafeu D, Mambap AT, et al. Prevalence, correlates and in-hospital outcomes of kidney dysfunction in hospitalized patients with heart failure in Buea-Cameroon. BMC Nephrol. 2022;23(1):8.
Denic A, Mathew J, Lerman LO, Lieske JC, Larson JJ, et al. Single-nephron glomerular filtration rate in healthy adults. N Engl J Med. 2017;376(24):2349–57.
Harjola V-P, Mullens W, Banaszewski M, Bauersachs J, Brunner-La Rocca H-P, et al. Organ dysfunction, injury and failure in acute heart failure: from pathophysiology to diagnosis and management. A review on behalf of the Acute Heart Failure Committee of the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur J Heart Fail. 2017;19(7):821–36.
Malhotra R, Craven T, Ambrosius WT, Killeen AA, Haley WE, et al. Effects of intensive blood pressure lowering on kidney tubule injury in CKD: a longitudinal subgroup analysis in SPRINT. Am J Kidney Dis. 2019;73(1):21–30.
Zhou S-H, Huang Q, Zhou Y, Cai X-X, Cui Y, et al. Captopril related kidney damage: renal afferent arteriolar responses to angiotensin II and inflammatory signaling. Sheng Li Xue Bao. 2022;74(1):125–33.
McAlister FA, Ezekowitz J, Tonelli M, Armstrong PW. Renal insufficiency and heart failure: prognostic and therapeutic implications from a prospective cohort study. Circulation. 2004;109(8):1004–9.
Damman K, Gori M, Claggett B, Jhund PS, Senni M, et al. Renal effects and associated outcomes during angiotensin-neprilysin inhibition in heart failure. JACC Heart Fail. 2018;6(6):489–98.
Solomon SD, McMurray JJV, Anand IS, Ge J, Lam CSP, et al. Angiotensin-neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609–20.
Briet M, Schiffrin EL. Aldosterone: effects on the kidney and cardiovascular system. Nat Rev Nephrol. 2010;6(5):261–73.
Rossignol P, Dobre D, McMurray JJV, Swedberg K, Krum H, et al. Incidence, determinants, and prognostic significance of hyperkalemia and worsening renal function in patients with heart failure receiving the mineralocorticoid receptor antagonist eplerenone or placebo in addition to optimal medical therapy: results from the Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure (EMPHASIS-HF). Circ Heart Fail. 2014;7(1):51–8.
Rossignol P, Cleland JGF, Bhandari S, Tala S, Gustafsson F, et al. Determinants and consequences of renal function variations with aldosterone blocker therapy in heart failure patients after myocardial infarction: insights from the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study. Circulation. 2012;125(2):271–9.
Mullens W, Martens P, Testani JM, Tang WHW, et al. Renal effects of guideline-directed medical therapies in heart failure: a consensus document from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2022;24(4):603–19.
Pandey AK, Dhingra NK, Hibino M, Gupta V, Verma S. Sodium-glucose cotransporter 2 inhibitors in heart failure with reduced or preserved ejection fraction: a meta-analysis. ESC Heart Fail. 2022;9(2):942–6.
McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008.
Zannad F, Ferreira JP, Pocock SJ, Zeller C, et al. Cardiac and kidney benefits of empagliflozin in heart failure across the spectrum of kidney function: insights from EMPEROR-reduced. Circulation. 2021;143(4):310–21.
Badve SV, Roberts MA, Hawley CM, Cass A, Garg AX, et al. Effects of beta-adrenergic antagonists in patients with chronic kidney disease: a systematic review and meta-analysis. J Am Coll Cardiol. 2011;58(11):1152–61.
Ghali JK, Wikstrand J, Van Veldhuisen DJ, Fagerberg B, Goldstein S, et al. The influence of renal function on clinical outcome and response to beta-blockade in systolic heart failure: insights from Metoprolol CR/XL Randomized Intervention Trial in Chronic HF (MERIT-HF). J Card Fail. 2009;15(4):310–8.
Beldhuis IE, Lam CSP, Testani JM, Voors AA, Van Spall HGC, et al. Evidence-based medical therapy in patients with heart failure with reduced ejection fraction and chronic kidney disease. Circulation. 2022;145(9):693–712.
Dzau VJ. Renal effects of angiotensin-converting enzyme inhibition in cardiac failure. Am J Kidney Dis. 1987;10(1 Suppl 1):74–80.
Packer M, Lee WH, Medina N, Yushak M, Kessler PD. Functional renal insufficiency during long-term therapy with captopril and enalapril in severe chronic heart failure. Ann Intern Med. 1987;106(3):346–54.
Gan L, Lyu X, Yang X, Zhao Z, Tang Y, et al. Application of angiotensin receptor-neprilysin inhibitor in chronic kidney disease patients: chinese expert consensus. Front Med (Lausanne). 2022;9: 877237.
Henrysson J, Thunström E, Chen X, Fu M, Basic C. Hyperkalaemia as a cause of undertreatment with mineralocorticoid receptor antagonists in heart failure. ESC Heart Fail. 2023;10(1):66–79.
Eschalier R, McMurray JJV, Swedberg K, van Veldhuisen DJ, Krum H, et al. Safety and efficacy of eplerenone in patients at high risk for hyperkalemia and/or worsening renal function: analyses of the EMPHASIS-HF study subgroups (Eplerenone in Mild Patients Hospitalization And SurvIval Study in Heart Failure). J Am Coll Cardiol. 2013;62(17):1585–93.
Jering KS, Zannad F, Claggett B, Mc Causland FR, Ferreira JP, et al. Cardiovascular and renal outcomes of mineralocorticoid receptor antagonist use in PARAGON-HF. JACC Heart Fail. 2021;9(1):13–24.
Desai AS, Vardeny O, Claggett B, McMurray JJV, Packer M, et al. Reduced risk of hyperkalemia during treatment of heart failure with mineralocorticoid receptor antagonists by use of sacubitril/valsartan compared with enalapril: a secondary analysis of the PARADIGM-HF Trial. JAMA Cardiol. 2017;2(1):79–85.
Ferreira JP, Zannad F, Pocock SJ, Anker SD, Butler J, et al. Interplay of mineralocorticoid receptor antagonists and empagliflozin in heart failure: EMPEROR-Reduced. J Am Coll Cardiol. 2021;77(11):1397–407.
Harrington J, Fonarow GC, Khan MS, Hernandez A, Anker S, et al. Medication-attributable adverse events in heart failure trials. JACC Heart Fail. 2023;11(4):425–36.
Cutshall BT, Duhart BT, Saikumar J, Samarin M, Hutchison L, et al. Assessing guideline-directed medication therapy for heart failure in end-stage renal disease. Am J Med Sci. 2018;355(3):247–51.
Butzner M, Riello RJ, Sarocco P, Desai N. Adverse drug effects across patients with heart failure: a systematic review. Am J Manag Care. 2022;28(3):e113–20.
Packham DK, Rasmussen HS, Lavin PT, El-Shahawy MA, Roger SD, et al. Sodium zirconium cyclosilicate in hyperkalemia. N Engl J Med. 2015;372(3):222–31.
Oshima A, Imamura T, Narang N, Kinugawa K. Management of hyperkalemia in chronic heart failure using sodium zirconium cyclosilicate. Clin Cardiol. 2021;44(9):1272–5.
Imamura T, Oshima A, Narang N, Kinugawa K. Clinical implications of sodium zirconium cyclosilicate therapy in patients with systolic heart failure and hyperkalemia. J Clin Med. 2021;10(23).
Carlisle MA, Fudim M, DeVore AD, Piccini JP. Heart failure and atrial fibrillation, like fire and fury. JACC Heart Fail. 2019;7(6):447–56.
Zhang Z, Xiao Y, Dai Y, Lin Q, Liu Q. Device therapy for patients with atrial fibrillation and heart failure with preserved ejection fraction. Heart Fail Rev. 2024;29(2):417–30. https://doi.org/10.1007/s10741-023-10366-7.
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021;42(5):373–498.
Moysidis DV, Kartas A, Samaras A, Papazoglou AS, Patsiou V, et al. Prescription rates and prognostic implications of optimally targeted guideline-directed medical treatment in heart failure and atrial fibrillation: insights from the MISOAC-AF Trial. J Cardiovasc Pharmacol. 2023;81(3):203–11.
Loring Z, Shrader P, Allen LA, Blanco R, Chan PS, et al. Guideline-directed therapies for comorbidities and clinical outcomes among individuals with atrial fibrillation. Am Heart J. 2020;219:21–30.
Ahn M-S, Yoo BS, Yoon J, Lee S-H, Kim JY, et al. Guideline-directed therapy at discharge in patients with heart failure and atrial fibrillation. Heart. 2020;106(4):292–8.
Kelly JP, DeVore AD, Wu J, Hammill BG, Sharma A, et al. Rhythm control versus rate control in patients with atrial fibrillation and heart failure with preserved ejection fraction: insights from get with the guidelines-heart failure. J Am Heart Assoc. 2019;8(24): e011560.
Di Biase L, Mohanty P, Mohanty S, Santangeli P, Trivedi C, et al. Ablation versus amiodarone for treatment of persistent atrial fibrillation in patients with congestive heart failure and an implanted device: results from the AATAC multicenter randomized trial. Circulation. 2016;133(17):1637–44.
Packer DL, Mark DB, Robb RA, Monahan KH, Bahnson TD, et al. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321(13):1261–74.
Marrouche NF, Brachmann J, Andresen D, Siebels J, Boersma L, et al. Catheter ablation for atrial fibrillation with heart failure. N Engl J Med. 2018;378(5):417–27.
Giunta H. Treatment incentives and the nature of the doctor-patient relationship. Am J Bioeth. 2016;16(10):77–8.
Fitzgerald AA, Powers JD, Ho PM, Maddox TM, Peterson PN, et al. Impact of medication nonadherence on hospitalizations and mortality in heart failure. J Card Fail. 2011;17(8):664–9.
Annema C, Luttik M-L, Jaarsma T. Reasons for readmission in heart failure: Perspectives of patients, caregivers, cardiologists, and heart failure nurses. Heart Lung. 2009;38(5):427–34.
van der Wal MHL, Jaarsma T, Moser DK, Veeger NJGM, van Gilst WH, et al. Compliance in heart failure patients: the importance of knowledge and beliefs. Eur Heart J. 2006;27(4):434–40.
Ruppar TM, Cooper PS, Mehr DR, Delgado JM, Dunbar-Jacob JM. Medication adherence interventions improve heart failure mortality and readmission rates: systematic review and meta-analysis of controlled trials. J Am Heart Assoc. 2016;5(6):e002606. https://doi.org/10.1161/JAHA.115.002606.
Athilingam P, Jenkins B. Mobile phone apps to support heart failure self-care management: integrative review. JMIR Cardio. 2018;2(1): e10057.
Wakefield BJ, Boren SA, Groves PS, Conn VS. Heart failure care management programs: a review of study interventions and meta-analysis of outcomes. J Cardiovasc Nurs. 2013;28(1):8–19. https://doi.org/10.1097/JCN.0b013e318239f9e1.
Comín-Colet J, Enjuanes C, Verdú-Rotellar JM, Linas A, Ruiz-Rodriguez P, et al. Impact on clinical events and healthcare costs of adding telemedicine to multidisciplinary disease management programmes for heart failure: results of a randomized controlled trial. J Telemed Telecare. 2016;22(5):282–95.
Hernandez LS. Role of telemedicine in improving guideline-directed medical treatment for patients with heart failure during a pandemic. Crit Care Nurs Clin N Am. 2022;34(2):151–6.
Clark KAA, Defilippis EM, Morris AA. Breaking the bank: the financial burden of living with heart failure. J Card Fail. 2022;28(9):1434–6.
Sen S. Heart failure with reduced ejection fraction: clinical and economic burden and insights into current and emerging treatments. Am J Manag Care. 2023;29(10 Suppl):S180–6.
Ali H-JR, Valero-Elizondo J, Wang SY, Cainzos-Achirica M, Bhimaraj A, et al. Subjective financial hardship due to medical bills among patients with heart failure in the United States: the 2014–2018 Medical Expenditure Panel Survey. J Card Fail. 2022;28(9):1424–33.
Pierce JB, Ikeaba U, Peters AE, DeVore AD, Chiswell K, et al. Quality of care and outcomes among patients hospitalized for heart failure in rural vs urban us hospitals: the get with the guidelines-heart failure registry. JAMA Cardiol. 2023;8(4):376–85.
Peterson PN, Rumsfeld JS, Liang L, Hernandez AF, Peterson ED, et al. Treatment and risk in heart failure: gaps in evidence or quality? Circ Cardiovasc Qual Outcomes. 2010;3(3):309–15.
Greene SJ, Fonarow GC, DeVore AD, Sharma PP, Vaduganathan M, et al. Titration of medical therapy for heart failure with reduced ejection fraction. J Am Coll Cardiol. 2019;73(19):2365–83.
Driscoll A, Currey J, Tonkin A, Krum H. Nurse-led titration of angiotensin converting enzyme inhibitors, beta-adrenergic blocking agents, and angiotensin receptor blockers for people with heart failure with reduced ejection fraction. Cochrane Database Syst Rev. 2015;12:CD009889.
King-Dailey K, Frazier S, Bressler S, King-Wilson J. The role of nurse practitioners in the management of heart failure patients and programs. Curr Cardiol Rep. 2022;24(12):1945–56.
McLachlan A, Aldridge C, Morgan M, Lund M, Gabriel R, Malez V. An NP-led pilot telehealth programme to facilitate guideline-directed medical therapy for heart failure with reduced ejection fraction during the COVID-19 pandemic. N Z Med J. 2021;134(1538):77–88.
Zheng J, Mednick T, Heidenreich PA, Sandhu AT. Pharmacist- and nurse-led medical optimization in heart failure: a systematic review and meta-analysis. J Card Fail. 2023;29:1000–13.
Arany Z. Peripartum cardiomyopathy. N Engl J Med. 2024;390(2):154–64.
Banayan J, Rana S, Mueller A, Tung A, Ramadan H, et al. Cardiogenic shock in pregnancy: analysis from the National Inpatient Sample. Hypertens Pregnancy. 2017;36(2):117–23.
Davis MB, Arany Z, McNamara DM, Goland S, Elkayam U. Peripartum cardiomyopathy: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(2):207–21.
McNamara DM, Elkayam U, Alharethi R, Damp J, Hsich E, et al. Clinical outcomes for peripartum cardiomyopathy in north america: results of the IPAC study (investigations of pregnancy-associated cardiomyopathy). J Am Coll Cardiol. 2015;66(8):905–14.
Arrigo M, Biegus J, Asakage A, Mebazaa A, Davison B, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure in elderly patients: a sub-analysis of the STRONG-HF randomized clinical trial. Eur J Heart Fail. 2023;25(7):1145–55.
Cheng JWM, Nayar M. A review of heart failure management in the elderly population. Am J Geriatr Pharmacother. 2009;7(5):233–49.
Kitzman DW, Upadhya B. Age-related divergence of risk-benefit relationship of spironolactone treatment for heart failure with preserved ejection fraction. JACC Heart Fail. 2019;7(12):1029–31.
Kim YS, Ahn Y. Benefits of SGLT2 inhibitor: preventing heart failure and beyond. Korean Circ J. 2019;49(12):1196–8.
Pop-Busui R, Januzzi JL, Bruemmer D, Butalia S, Green JB, et al. Heart failure: an underappreciated complication of diabetes A consensus report of the American Diabetes Association. Diabetes Care. 2022;45(7):1670–90.
Kristensen SL, Preiss D, Jhund PS, Squire I, Cardoso JS et al. Risk related to pre-diabetes mellitus and diabetes mellitus in heart failure with reduced ejection fraction: insights from prospective comparison of ARNI With ACEI to determine impact on global mortality and morbidity in heart failure trial. Circ Heart Fail. 2016;9(1):e002560. https://doi.org/10.1161/CIRCHEARTFAILURE.115.002560.
Packer M, Anker SD, Butler J, Filippatos G, Ferreira JP, et al. Effect of empagliflozin on the clinical stability of patients with heart failure and a reduced ejection fraction: the EMPEROR-reduced trial. Circulation. 2021;143(4):326–36.
Packer M, Butler J, Zannad F, Filippatos G, Ferreira JP, et al. Effect of empagliflozin on worsening heart failure events in patients with heart failure and preserved ejection fraction: EMPEROR-preserved trial. Circulation. 2021;144(16):1284–94.
Kosiborod MN, Abildstrøm SZ, Borlaug BA, Butler J, Rasmussen S, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med. 2023;389(12):1069–84.
Marso SP, Baeres FMM, Bain SC, Goldman B, Husain M, et al. Effects of liraglutide on cardiovascular outcomes in patients with diabetes with or without heart failure. J Am Coll Cardiol. 2020;75(10):1128–41.
Acknowledgments
Figures 1 and 2 were created using the image bank of Servier Medical Art (http://smart.servier.com/), licensed under the Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/licenses/by/3.0/).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This work was supported by the National Natural Science Foundation of China (no. 82070356 and 81770337), the Key Project of Hunan Provincial Science and Technology Innovation (no. 2020SK1013), the Hunan Provincial Natural Science Foundation of China (no. 2021JJ30033 and 2023JJ30791), the Fundamental Research Funds for the Central Universities of Central South University (no. 2024ZZTS0878), the Clinical Medical Technology Innovation Guidance Project of Hunan Science and Technology Agency (no. 2021SK53519), the Changsha Municipal Natural Science Foundation (no. kq2208306), the Hunan Provincial Health Commission Scientific Research Project (no. 20201302), and the National Natural Science Foundation of China (no. 82200068).
Conflicts of Interest/Competing Interests
There are no conflicts of interest or competing interests.
Availability of Data and Material
No new data were generated or analyzed in support of this research.
Authors’ Contributions
Under the guidance of Prof. Q.L. and Y.X. C.W., J.Z., Y.H., and Z.Z. collected relevant information. Z.Z. wrote the manuscript, which was revised by T.T., Q.L., K.W., W.Z., and N.L. All the authors contributed to the article and approved the final manuscript.
Ethics Approval
Not applicable.
Consent to Participate
All the authors participated in the study and made significant intellectual contributions to the manuscript.
Consent for Publication
The manuscript is not currently under consideration elsewhere, and the work reported will not be submitted for publication elsewhere until a final decision has been made as to its acceptability by the Journal.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Zhang, Z., Wang, C., Tu, T. et al. Advancing Guideline-Directed Medical Therapy in Heart Failure: Overcoming Challenges and Maximizing Benefits. Am J Cardiovasc Drugs 24, 329–342 (2024). https://doi.org/10.1007/s40256-024-00646-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40256-024-00646-4





