Abstract
Background
Pregnant persons are susceptible to significant complications following COVID-19, even death. However, worldwide COVID-19 vaccination coverage during pregnancy remains suboptimal.
Objective
This study assessed the safety and effectiveness of COVID-19 vaccines administered to pregnant persons and shared this evidence via an interactive online website.
Methods
We followed Cochrane methods to conduct this living systematic review. We included studies assessing the effects of COVID-19 vaccines in pregnant persons. We conducted searches every other week for studies until October 2023, without restrictions on language or publication status, in ten databases, guidelines, preprint servers, and COVID-19 websites. The reference lists of eligible studies were hand searched to identify additional relevant studies. Pairs of review authors independently selected eligible studies using the web-based software COVIDENCE. Data extraction and risk of bias assessment were performed independently by pairs of authors. Disagreements were resolved by consensus. We performed random-effects meta-analyses of adjusted relative effects for relevant confounders of comparative studies and proportional meta-analyses to summarize frequencies from one-sample studies using R statistical software. We present the GRADE certainty of evidence from comparative studies. Findings are available on an interactive living systematic review webpage, including an updated evidence map and real-time meta-analyses customizable by subgroups and filters.
Results
We included 177 studies involving 638,791 participants from 41 countries. Among the 11 types of COVID-19 vaccines identified, the most frequently used platforms were mRNA (154 studies), viral vector (51), and inactivated virus vaccines (17). Low to very low-certainty evidence suggests that vaccination may result in minimal to no important differences compared to no vaccination in all assessed maternal and infant safety outcomes from 26 fewer to 17 more events per 1000 pregnant persons, and 13 fewer to 9 more events per 1000 neonates, respectively. We found statistically significant reductions in emergency cesarean deliveries (9%) with mRNA vaccines, and in stillbirth (75–83%) with mRNA/viral vector vaccines. Low to very low-certainty evidence suggests that vaccination during pregnancy with mRNA vaccines may reduce severe cases or hospitalizations in pregnant persons with COVID-19 (72%; 95% confidence interval [CI] 42–86), symptomatic COVID-19 (78%; 95% CI 21–94), and virologically confirmed SARS-CoV-2 infection (82%; 95% CI 39–95). Reductions were lower with other vaccine types and during Omicron variant dominance than Alpha and Delta dominance. Infants also presented with fewer severe cases or hospitalizations due to COVID-19 and laboratory-confirmed SARS-CoV-2 infection (64%; 95% CI 37–80 and 66%; 95% CI 37–81, respectively).
Conclusions
We found a large body of evidence supporting the safety and effectiveness of COVID-19 vaccines during pregnancy. While the certainty of evidence is not high, it stands as the most reliable option available, given the current absence of pregnant individuals in clinical trials. Results are shared in near real time in an accessible and interactive format for scientists, decision makers, clinicians, and the general public. This living systematic review highlights the relevance of continuous vaccine safety and effectiveness monitoring, particularly in at-risk populations for COVID-19 impact such as pregnant persons, during the introduction of new vaccines.
Clinical Trial Registration
PROSPERO: CRD42021281290.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Pregnant persons are susceptible to significant complications following COVID-19, even at the present stage of the pandemic, but vaccination coverage during pregnancy remains suboptimal. |
Our living systematic review found a large body of evidence supporting the safety and effectiveness of COVID-19 vaccines during pregnancy. |
The lower effectiveness during Omicron variant dominance, which decreases over time, and the less robust evidence for non-mRNA vaccines emphasized the need for continuous vaccine safety and effectiveness monitoring among pregnant persons. |
1 Introduction
The COVID-19 pandemic caused a significant global health crisis leading to over 770 million cases and seven million deaths worldwide [1]. The development and distribution of COVID-19 vaccines have been critical steps in controlling the spread of the virus and mitigating its impact on health worldwide [2, 3]. Many COVID-19 vaccines were authorized for use [4, 5]. Although clinical trials demonstrated promising results regarding the efficacy and safety of these vaccines [6], real-world effectiveness and safety data on these vaccines continue to emerge.
Early in the pandemic, pregnant persons with COVID-19 were found to be at a greater risk of experiencing severe illness than non-pregnant adults [7,8,9]. Various factors, including maternal age, high body mass index, non-white race/ethnicity, pre-existing health conditions, and gestational diabetes mellitus, increased the risk of severe COVID-19 during pregnancy further, including a higher risk of admission to intensive care units or needing invasive ventilation than non-pregnant women of reproductive age [10, 11]. Pregnant persons also exhibited an elevated likelihood of experiencing adverse pregnancy and birth outcomes associated with SARS-CoV-2 infection [12]. In particular, pregnant persons with COVID-19 are prone to delivering preterm babies, who may require admission to neonatal intensive care units (NICUs). The rates of SARS-CoV-2 infection in pregnant women varied significantly by region, with the highest rates observed in Latin America and the Caribbean (19%) and in lower- to middle-income countries (13%). Lower-income to middle-income countries (LMICs) also reported significantly higher rates of maternal mortality (0.68%) and stillbirths (1.09%) than high-income countries (HICs). [13]
Unfortunately, completed randomized controlled trials (RCTs) of COVID-19 vaccines did not include pregnant persons in their eligible population [14]. Consequently, the efficacy and safety of multiple vaccine products during pregnancy were not fully assessed at the time of their recommendation for widespread use in response to the public health emergency. Many regulatory bodies advised the use of COVID-19 vaccines for pregnant persons on the premise that the benefits of vaccination would outweigh the potential risks [15]. It was therefore crucial to provide a timely comprehensive evaluation of the available evidence on the safety and benefits of COVID-19 vaccines in this population. Considering the growing body of evidence, we conducted a living systematic review (LSR) and meta-analysis designed to periodically assess the effectiveness, safety, and immunogenicity of COVID-19 vaccines to inform clinical practice, public health policy, and future research efforts.
2 Methods
We followed the Cochrane and World Health Organization (WHO) methods [16,17,18] and the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) statement/extension for reporting this LSR [19, 20]. The protocol was registered in the PROSPERO database (CRD42021281290) and published elsewhere [21], but we summarize the main methodology here.
2.1 Inclusion Criteria
We included RCTs, quasi-experimental studies, and observational studies assessing COVID-19 vaccination during pregnancy, irrespective of publication status (pre-print or standard publication), publication year, and language. Case reports for unexpected adverse events were also included. Study participants were pregnant persons, irrespective of prior exposure to SARS-CoV-2, age, comorbidities, immune status, or baseline risk with or without exposure to virologically confirmed SARS-CoV-2 infection.
We included all COVID-19 vaccines authorized by the WHO or national regulatory authorities, regardless of dosage or administration schedule. Any comparison group was considered, including usual care, no intervention, another COVID-19 vaccine, or other active comparators. We also included non-comparative studies; therefore, a control group was not mandatory. We considered any safety outcomes, i.e., pregnancy-related maternal or obstetric outcomes, adverse events following immunization (AEFIs), infant outcomes, efficacy or effectiveness outcomes, and immunogenicity results available in published studies.
2.2 Search Strategy
We conducted searches every other week from January 2020 to October 2023 of published and unpublished studies without restrictions on language or publication status (see the full search in the Electronic Supplementary Material [ESM]). The searches were conducted in the Cochrane Library databases, MEDLINE, EMBASE, Latin American and Caribbean Health Sciences Literature (LILACS), Science Citation Index Expanded (SCI-EXPANDED), China Network Knowledge Information (CNKI), Chinese Biomedical Literature Database (CBM), Chinese Science Journal Database (VIP), WHO Database of publications on SARS-CoV-2, EPPI-Centre map of the current evidence on COVID-19, guidelines published by national and international professional societies (e.g., American College of Obstetricians and Gynecologists, Royal College of Obstetricians and Gynecologists, International Federation of Gynecology and Obstetrics), pre-print servers (ArXiv, BiorXiv, medRxiv, search.bioPreprint), and COVID-19 research websites, including the WHO Global research on coronavirus disease (COVID-19) website, COVID-19 Vaccine Tracker, the L-OVE Platform, and the COVID-19 Living Evidence. Additional relevant studies were identified by hand searching the reference lists of the identified systematic reviews.
2.3 Selection of Studies and Data Collection
Pairs of review authors independently screened each title and abstract and retrieved all potentially relevant full-text studies. Pairs of review authors independently selected the full texts, documenting the reasons for the exclusion of ineligible studies. We resolved disagreements through discussion with the review team. This process was performed using the web-based software COVIDENCE [22]. Study data were abstracted independently by pairs of review authors and stored using REDCap electronic data capture tools [23] hosted and designed by the Institute for Clinical Effectiveness and Health Policy. If needed, we contacted the study authors for additional data. Data extraction items included study identification elements, methods, participants’ characteristics, countries involved, group allocation, intervention, outcomes, risk of bias, and summary of results.
2.4 Risk of Bias Assessment
For observational cohort, case-control, cross-sectional, and case-series studies, we used the National Institutes of Health Quality Assessment Tools [24]. After answering the different signaling questions, the reviewers (see the signaling questions in Tables S2, S3, S4, and S5 of the ESM) classified the study quality as good, fair, or poor.
2.5 Data Synthesis
We performed meta-analyses for each comparison according to the Cochrane Handbook of Systematic Reviews of Interventions and used a random-effects meta-analysis for the primary analysis [25]. Only studies with adjusted effect measures (e.g., adjustment for at least two confounders as age, smoking status, parity, body mass index) or using matched controls were included for comparative meta-analyses. We also performed proportional meta-analyses to summarize frequencies from one-sample studies and conducted targeted searches for background rates of each maternal-infant event. These background rates encompass global, HIC, and LMIC rates and serve as reference points for contextualizing the findings within the frame of maternal-infant health knowledge. R statistical software was used for this project [26] to analyze the data. The main packages selected for data analyses were Meta [27], Metafor [28], and Tidyverse [29]. For the selection process of the study data for each meta-analysis, we avoided double-counting populations or comparators through an algorithm, which selected the data from each study with the greatest amount of available information (significant quantity of trimesters, vaccines, types of variants) that complied with the selected analysis. When one study had more than one effect estimate for the same outcome, we included in the analysis only the data with the most follow-up and the largest sample size. The researchers performed a validation process to ensure the validity of the endpoint selection algorithm monthly.
We extracted or calculated hazard ratios (HRs), risk ratios (RRs), or odds ratios (ORs) with a 95% confidence interval (CI) for dichotomous outcomes and the mean difference or standardized mean difference for continuous outcomes. We also calculated proportions with a 95% CI for non-comparative studies using arcsine transformation [30].
We estimated the vaccine efficacy/effectiveness (VE) by determining the percentage reduction in disease risk among vaccinated persons compared with unvaccinated persons based on adjusted relative effects [31]. We analyzed maternal safety outcomes, including pregnancy-related outcomes and AEFIs, and infant safety outcomes mainly based on standardized case definitions developed by the Global Alignment of Immunization Safety Assessment in Pregnancy (GAIA) and the Brighton Collaboration [32].
For maternal and infant VE, we included the prevention of laboratory-confirmed SARS-CoV-2 infection and symptomatic COVID-19 by nucleic acid amplification tests and their complications, including hospital and intensive care unit admissions and deaths, among pregnant persons vaccinated during pregnancy and their offspring. Results regarding immunogenicity outcomes will be presented in a separate publication.
We performed the following pre-specified subgroup analyses when analyzing the primary outcomes: pregnancy trimester (first/early pregnancy 0–12 weeks; second trimester 13–27 weeks or third trimester/late pregnancy 28 weeks to full term), vaccine platform (mRNA, viral vector, inactivated virus), dominant variant of SARS-CoV-2 among the study population (Alpha, Beta, Delta, Omicron), and vaccination schedule (primary series and booster).
In the “Summary of Findings” tables, we summarized the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) certainty of evidence from comparative studies [33]. The estimates were downgraded for serious and very serious imprecision if 95% CIs were < 0.90 or > 1.10, and < 0.5 or > 1.50, respectively (or VE reaching 0% for effectiveness), and for serious and very serious inconsistency if not all 95% CIs are at the same side of the minimal important difference and I2 values were > 60% and > 75%, respectively.
2.6 Data Visualization
We developed an online interactive dashboard for data visualization using Microsoft Power BI [34], available at https://www.safeinpregnancy.org/living-systematic-review/. The most relevant variables can be selected among maternal and neonatal safety and effectiveness outcomes. They are presented in figures, tables, and maps. The living meta-analysis section is available for users as an interactive tool developed as a Shiny application through RStudio [35]. The application allows the users to display meta-analyses of interest by outcome selecting by filters such as trimester, vaccine platform, vaccine doses, population, and comparator, among others. The research team designed an algorithm for the endpoint selection of each study included in the living meta-analysis.
3 Results
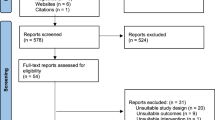
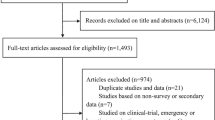
We included in this review 177 studies (see the study flow diagram in Fig. 1) that provided data on the safety, effectiveness, and/or immunogenicity of COVID-19 vaccines in pregnant persons and their infants, published until October 2023 (56% in 2022). The studies included a total of 638,791 vaccinated pregnant persons from 41 countries who were exposed to mRNA (154 studies), viral vector (51), and inactivated virus vaccines (17). Only ten studies (6%) were from LMICs and 23 studies (13%) were from upper-middle-income countries, the rest were from HICs. Among the included studies, 42% reported pregnancy outcomes, 35% maternal AEFI, 37% infant safety outcomes, 16% VE, and 39% immunogenicity results.
For this analysis, we included 137 studies reporting safety or effectiveness outcomes (immunogenicity outcomes will be reported elsewhere). Most were conducted in the USA (43 [31%]), Israel (25 [18%]), Brazil (7 [5%]), and seven (5%) were multi-country. These studies used one or more study designs: cohort studies (95 [69%]), cross-sectional (12 [9%]), case-control (10 [7%]), case series (10 [7%]), case report (9 [7 %]), and controlled clinical trial (1 [1%]). The only included RCT reported only AEFIs. It is noteworthy that 21 of the studies reported data from surveillance systems (15%). Out of 137 publications, 33 reported adjusted measures and were included in the meta-analysis.
Among the ten COVID-19 vaccine products identified, the most frequently assessed were the BNT162b2 (Pfizer/BioNTech) mRNA vaccine in 108 studies (79%), the mRNA-1273 (Moderna) vaccine in 71 studies (52%), and the Janssen/Johnson & Johnson (Ad26,COV2,S) in 24 studies (18%) (Table S1 of the ESM). All descriptive information is available online in real time at https://www.safeinpregnancy.org/living-systematic-review/ (last updated January 2024). It can be filtered by publication date, country/region, study design, population, and vaccine type and product from the sidebar, the map, or the figures. A snapshot of 10/30/2023 is presented in the evidence map in Fig. 2.
3.1 Risk of Bias of Included Studies
The comprehensive quality assessment according to each study design revealed a substantial proportion of studies meeting the criteria for good or fair quality. Among the cross-sectional and cohort studies, 93% (99/107) were rated as good or fair quality, while 70% (7/10) of case-series studies reached the same categories. All the case-control studies were deemed to be of good or fair quality (10/10) and the only controlled clinical trial included met the criteria for fair quality. The risk of bias for the included studies by study design is presented in Tables S2, S3, S4, and S5, of the ESM.
3.2 Effects of COVID-19 Vaccines During Pregnancy
We present the “Summary of Findings” tables with the most important outcomes by subgroup meta-analyses/estimates. Maternal pregnancy-related safety outcomes and infant safety outcomes are presented by trimester and by vaccines, and efficacy by vaccine type and SARS-CoV-2 variant dominance. All remaining results, organized by outcome groups, are also presented in our LSR platform at https://www.safeinpregnancy.org/summary-tables/.
The results of each comparative (see https://www.safeinpregnancy.org/meta-analysis/) and non-comparative (see https://www.safeinpregnancy.org/proportional-meta-analyses/) meta-analysis are shown on the online and interactive LSR platform through forest plot/meta-analyses and summary tables organized by outcome.
3.2.1 Maternal Pregnancy-Related Safety Outcomes
The pooled absolute and relative effects of adjusted comparative studies on maternal pregnancy-related safety outcomes and their certainty of evidence are presented in Table 1 and the pooled proportions of outcomes and background rates, including the supporting references, are in Table S6 of the ESM.
For this particular set of outcomes, the level of evidence ranged from low to very low certainty. Evidence suggests a lower risk of stillbirth among those who received at least one dose of the COVID-19 mRNA vaccine compared with those who did not when the exposure was in the first or second trimester (1,2T) or in the third trimester (3T) [RR 0.17; 95% CI 0.07–0.43 and RR 0.25; 95% CI 0.08–0.80, respectively], but there was no statistically significant difference between arms when the exposure was in the 2T (RR 1.12; 95% CI 0.52–2.40) and at any trimester. Pooled estimation of three studies [36,37,38] of vaccination at any trimester with mRNA/viral vector showed a RR of 0.49 (95% CI 0.20–1.19; I2 87%) (Fig. 3). We found similar findings when we analyzed this outcome by vaccine platform (Fig. S1 of the ESM).
Adjusted risk of stillbirth associated with COVID-19 vaccination during pregnancy versus unvaccinated pregnant population, by trimester of exposure. A Alpha, CI confidence interval, D Delta, DV dominant variant, G good, GQ global quality, NS not specified, O Omicron, VT vaccine type, VV viral vector
Three observational studies reported adjusted effect measures for miscarriage/abortion [39,40,41]. Pooled studies of two of them [39, 41] did not show a higher risk of miscarriage/abortion among vaccinated pregnant women with mRNA in 1T or 1/2T (OR 0.91; 95% CI 0.70–1.20; I2 77%) with at least one dose during pregnancy (Fig. S2 of the ESM). The third study assessing viral vector vaccines found similar results (RR 0.84 95% CI 0.48–1.47) [not shown in the plot because it uses a different effect measure] [40]. Only one study reported adjusted effect measures for a gestational diabetes outcome [42] and did not show a higher risk after vaccination at 2T and 3T (RR 1.21; 95% CI 0.93–1.58 and RR 0.99; 95% CI 0.77–1.27, respectively) between vaccinated pregnant people with the mRNA vaccine and unvaccinated groups.
The pooled estimation of three studies reporting adjusted effect measures [42,43,44] did not show a higher risk of hypertensive disorders of pregnancy between vaccinated pregnant women with the mRNA vaccine administered in 2,3T and unvaccinated pregnant women (RR 1.07; 95% CI 0.81–1.40; p = 0.49; I2 0%) [Fig. S3 of the ESM]. Six studies reported adjusted effect measures for emergency cesarean delivery [37, 42,43,44,45,46]. The single study meeting the trimester filter showed a lower incidence of this outcome in the vaccinated group at 2,3T with the mRNA vaccine (RR 0.97; 95% CI 0.94–1.00).
A postpartum hemorrhage-adjusted outcome was reported by six studies [36, 42, 43, 45,46,47]. Pooled estimates of five of them ([36, 37, 42, 43, 46] showed a trend toward a higher incidence of postpartum hemorrhage in those receiving mRNA vaccines during 2,3T (regardless of the completeness of the scheme) versus unvaccinated pregnant persons (RR 1.44; 95% CI 0.85–2.45; I2 89%). The subgroup analysis by vaccination schedule (partial, completed, and booster) showed different incidences of this outcome (RR 1.09; 95% CI 0.56–2.12 [45]; RR 0.95; 95% CI 0.86–1.05 [36, 37, 43, 46] and RR 3.88; 95% CI 2.41–6.25 [46], respectively) (Figs. S4 and S5 of the ESM).
The background rates of maternal pregnancy-related safety outcomes were within the CIs of the proportional meta-analysis estimations, which means that proportions were not unexpected in vaccinated individuals. The subgroup analysis by trimester of exposure and vaccine type showed similar results to each other or presented a significant imprecision.
As an example, we illustrated the pooled proportions of stillbirth by trimester in Fig. 4 and by vaccine type in Fig. S6 of the ESM. The rest of the outcomes are shown in https://www.safeinpregnancy.org/proportional-meta-analyses/.
3.2.2 Maternal AEFIs
The pooled proportions of AEFIs among vaccinated pregnant persons are presented by vaccine platform in Table S7 of the ESM. The pooled anaphylaxis proportion by vaccine type was 2 per 100,000 vaccinated persons (95% CI 0–92) with mRNA vaccines, and no events were reported with viral vector vaccines [48]. For serious adverse events, proportions were 0.25% (95% CI 0.13–0.41) and 0.41% (95% CI 0.10–2.24), and for lymphadenopathy, 4.41% (95% CI2.48–6.83) and 6.36 % (95% CI 4.70–8.24) with mRNA and viral vector vaccines respectively. The pooled proportion of myocarditis was 0.00% (95% CI 0.00–1.19) and the proportion of seizures was 0.05% (95% CI 0.00–0.26), both with mRNA vaccines. The pooled proportion of fever was 6.88% (95% CI 4.15–10.21) with mRNA vaccines and 30.96% (95% CI 7.47–26.15) with viral vector vaccines. The observed pooled proportion of headache was 19.83% (95% CI 14.20–26.14) with mRNA vaccines, 12.40% (95% CI 0.00–44.20) with viral vector vaccines, and 10.05% (95% CI 4.03–18.00) with inactivated virus vaccines. Fatigue proportions were 40.91% (95% CI 33.16–48.89) with mRNA, 27.76% (95% CI 0.00–94.00) with viral vector vaccines, and 11.46% (95% CI 2.02–25.42) with inactivated virus vaccines, respectively. The rest of the analyzed AEFI outcomes (chills, diarrhea, injection-site reactions, joint pain, myalgia, rash, and vomiting) are shown in Table S7 of the ESM, and in our interactive LSR online platform (https://www.safeinpregnancy.org/proportional-meta-analyses/=).
The pooled proportions by vaccine schedule (partial, complete, and booster) were generally similar with few exceptions. The proportion of headache, fatigue, fever, and lymphadenopathy was more than double with the second dose compared withthe first dose (shown in https://www.safeinpregnancy.org/proportional-meta-analyses/)
Only two cohort studies reported comparative effect measures for AEFIs between pregnant and non-pregnant persons [49, 50]. Kachikis et al. [49] observed that in comparison to non-pregnant non-lactating persons (N = 4726), pregnant persons (N = 2009) experienced more local reactions to a COVID-19 booster or third dose (adjusted OR 1.2; 95% CI 1.0–1.4 but fewer systemic reactions (adjusted OR 0.7; 95% CI 0.6–0.8). The majority of pregnant individuals (97.6%) reported no obstetric concerns following vaccination.
Shapiro et al. [50] reported that pregnant persons (N = 1650 first dose and 1014 second dose) experienced side effects less frequently than non-pregnant persons (N = 6600 first dose and 4052 second dose). Pregnancy was a weak predictor for reporting any side effect in general and in particular fatigue, myalgia, headache, chills, and fever. Estimation of each study and meta-analyses of both studies are reported in Table S8 of the ESM.
3.2.3 Infant Safety Outcomes Following COVID-19 Vaccination During Pregnancy
The pooled absolute and relative effects of adjusted comparative studies on infant safety outcomes following COVID-19 vaccination during pregnancy and their certainty of evidence are presented in Table 2. Pooled proportions and background rates, including the supporting references, are shown in Table S9 of the ESM. For this particular set of outcomes, the level of evidence ranged from low to very low certainty.
Three studies reported adjusted effect measures for any congenital malformations [36, 45, 51]. Regardless of the trimester of exposure, the studies did not show an increased risk of congenital malformations in the vaccinated group (RR 0.69 for 1T, 0.80 for 2,3T, and 0.72 for 1,2,3T). Pooled studies by vaccine type (RR 0.71; 95% CI 0.47–1.06 and RR 0.89; 95% CI 0.56–0.93 for mRNA and viral vector, respectively). Analysis by vaccine schedule showed similar results (Figs. S7, S8, and S9 of the ESM).
Five pooled studies [36,37,38, 42, 46] that recorded the incidence of an Apgar score ≤5 minutes revealed no statistically significant difference between vaccinated during 2T-3T or any T and unvaccinated groups (RR 0.74; 95% CI 0.33–1.88 and RR 0.90; 95% CI 0.77–1.04, respectively); pooled results were homogenous (I2 29% and 50%, respectively). Subgroup analyses by vaccine type or vaccine schedule do not show any differences (Figs. S10 and S11 of the ESM).
Nine studies reported adjusted effect measures for prematurity (gestational age at delivery < 37 weeks) [36,37,38, 42, 44, 46, 51, 52]. Pooled estimations did not show a higher incidence of preterm infants in vaccinated persons exposed at any trimester (RR 0.89; 95% CI 0.73–1.07). There were some differences by trimester but never reached statistical significance. When we analyzed pooled studies by vaccine schedule and type of vaccine (mRNA and viral vector), there was no increased risk of prematurity either (Figs. S12 and S13 of the ESM).
Ten studies reported adjusted effect measures for small for gestational age [36, 37, 42,43,44,45,46, 51, 52]. Pooled estimations did not show a higher incidence of small for gestational age cases in vaccinated persons exposed at any trimester (RR 0.98; 95% CI 0.94–1.01). There were some differences by trimester but these never reached statistical significance. When we analyzed pooled studies by vaccine status and type of vaccine (mRNA and viral vector), there was no increased risk for preterm infants (Figs. S14 and S15 of the ESM).
Six studies reported adjusted effect measures for NICU hospitalization [36,37,38, 44, 45, 51]. Pooled estimations did not show a higher incidence of NICU hospitalizations and even showed a lower incidence among vaccinated pregnant persons at 3T and 2,3T (RR 0.92; 95% CI 0.87–0.97 and RR 0.92; 95% CI 0.87–0.97, respectively) [Fig. S16 of the ESM]. A subgroup analysis by vaccine platform and vaccine status did not show any relevant difference between groups.
Only one study reported adjusted effect measures for respiratory distress in the newborn (not due to COVID-19) [43] and one reported for infant death [51] among pregnant persons receiving mRNA vaccination at any trimester. There was no increased risk (RR 0.84; 95% CI 0.42–1.68).
The background rates of infant safety outcomes were within the CIs of the proportional meta-analysis performed (see Table S9 of the ESM). Data were analyzed by trimester of exposure and vaccine type. The pooled proportions showed no important differences by vaccine type, except for low birth weight, which presented half the proportion with viral vector vaccines than with mRNA vaccines (2.25%; 95% CI 1.25–3.50 vs 5.56%; 95% CI 4.23–7.06, respectively) [Fig. S17 of the ESM; see https://www.safeinpregnancy.org/proportional-meta-analyses/ for the rest of the outcomes].
3.2.4 Effectiveness Outcomes
The absolute and relative effects of adjusted comparative studies and their certainty of evidence by vaccine type and SARS-CoV-2 variant dominance are presented in Table 3. Seven studies reported adjusted effect measures of vaccination compared with no vaccination on the VE on severe or hospitalized COVID-19 [53,54,55,56,57,58,59]. The available evidence, characterized by low to very low certainty, indicates a potential reduction in severe or hospitalized COVID-19 cases among mothers with varying vaccine types. The comparison group for all VE analyses was 0 doses (unvaccinated pregnant individuals). Specifically, combining across variants, the VE was estimated at 72% (95% CI 42–86) with mRNA vaccines, 49% (95% CI 0–74) with viral vector vaccines, and 61% (95% CI 0–93) with inactivated vaccine regardless of vaccine schedule (see Fig. 5). Different follow-ups, which can be explored online activating days to outcome, did not change significantly the estimations. The VE during Omicron SARS-CoV-2 dominance was 58% (95% CI 15–79) for the primary series and 65% (95% CI 33–81) after a booster dose regardless of the timing of vaccination. Schrag et al. reported an estimated VE of 86% (95% CI 28–97) against hospitalization in the Omicron period from 7 to 119 days after the receipt of a booster dose. This suggests a high level of protection during this period. However, the booster dose did not provide a statistically significant protective effect beyond 120 days before virologically confirmed SARS-CoV-2 or hospitalization [59] (Figs. S18 and S19 of the ESM).
Effectiveness of COVID-19 vaccination compared to no vaccination during pregnancy, by vaccine type: severe or hospitalized maternal COVID-19. Global quality (GQ): Fair (F), Good (G); Vaccine type (VT): Inactivated virus (IV), Ribonucleic acid (RNA), Viral vector (VV), Not specified (NS); Vaccination status (VS): Complete (C), Booster (B); Trimester (Trim): 1 (1st), 2 (2nd), 3 (3rd), Not specified (NS); Dominant variant (DV): Alpha (A), Delta (D), Omicron (O), Not specified (NS)
The pattern was similar for symptomatic COVID-19 by type of vaccine with VE of 78% (95% CI 21–94) for mRNA, 39% (95% CI 26–49) for inactivated virus, and 25% (95% CI 0–100) for viral vector regardless the SARS-CoV-2 variant dominance. The VE with mRNA or viral vector vaccines during Omicron SARS-CoV-2 dominance was 56% (95% CI 21–75) and was not possible to compare with a single type of vaccine.
The same happens regarding laboratory-confirmed SARS-CoV-2 infection by vaccine type, with VE 82% (95% CI 39–95) with mRNA vaccines, 27% (95% CI 0–47) with inactivated vaccines, and 20% (95% CI 0–36) with viral vector vaccines. However, the reduction was lower during Omicron dominance: VE 30% (95% CI 19–39).
Low-certainty evidence suggests that mothers’ vaccination may reduce severe or hospitalized COVID-19 in their infants, based on three studies that reported adjusted effect measures for this outcome in infants up to 6 months of age [14, 60, 61]. Pooled studies showed a VE of 79% (95% CI 45–92) with the mRNA vaccine and complete scheme (two doses) in mothers evaluating during Delta dominance and 48% (95% CI 32–60) during Omicron dominance. One study [14] showed that a third vaccine dose during pregnancy bolstered the protection of infants raising the VE to 80% (63–88%) during Omicron dominance and maintaining protection for 6 months. These meta-analyses by trimester of exposure are available at https://www.safeinpregnancy.org/meta-analysis/.
4 Discussion
To our knowledge, this is the first LSR that has regularly evaluated, updated, and publicly disseminated the latest findings on the safety and effectiveness of COVID-19 vaccines during pregnancy, including 177 studies involving 631,957 pregnant persons worldwide exposed to ten COVID-19 vaccine products. Out of the 137 publications included in meta-analyses, 33 provided adjusted measures and thus incorporated them into the comparative meta-analysis.
4.1 Main Findings
We found no associations between COVID-19 vaccination during pregnancy with at least one dose and adverse maternal-pregnancy-related and infant outcomes, regardless of the trimester of exposure and type of vaccine. Outcomes included miscarriage, gestational diabetes, hypertensive disorders, congenital anomalies, Apgar score at 5 minutes < 7, prematurity, small for gestational age, NICU admission or hospitalization, and respiratory distress. In fact, we observed statistically significant reductions in stillbirth with mRNA/viral vector vaccines administered in the early stages of pregnancy and in emergency cesarean deliveries with mRNA vaccines at any time. Postpartum hemorrhage was the only outcome where we found a non-statistically significant higher incidence in vaccinated versus non-vaccinated pregnant persons. However, only three studies were included in this meta-analysis. The pooled proportions of maternal and infant safety outcomes were in line with what was anticipated, considering the background rates reported globally in HIC and LMIC settings. We only identified two studies directly comparing the effects of vaccination in pregnant versus non-pregnant persons on AEFIs [49, 50]. For most of the maternal AEFIs reported and analyzed, such as chills, eye irritation, facial swelling, rash, fatigue, fever, gastrointestinal, myalgias, systemic reactions, vomiting, nausea, and diarrhea, the frequency was lower in pregnant than in non-pregnant persons. The only exception was injection-site reactions, with a 20% higher frequency among pregnant persons.
COVID-19 VE in pregnant persons against severe complications (severe symptoms, hospitalization, and intensive care unit admission), during the Omicron period, was 58% (95% CI 15–79) after the primary series, and the VE increased to 65% (33–81) with a booster dose. Effectiveness was higher if the booster dose was administered within the past 120 days. Variability of VE by type of vaccine ranged across variants, from 49% with viral vector vaccines to 72% with mRNA vaccines. Even after a booster dose, protection against symptomatic COVID-19 and laboratory-confirmed SARS-CoV-2 infection during Omicron dominance was just 56% (95% CI 21–75) and 30% (95% CI 19-39), respectively. Maternal COVID-19 vaccination protects against severe illness both among pregnant women and their infants up to 6 months of age who are too young to be vaccinated.
The certainty of the evidence in our LSR was determined to be low to very low for all outcomes using the GRADE process primarily owing to the derivation of data from observational studies [33]. Starting for this reason with low certainty evidence, each of the outcomes can be downgraded, resulting in a classification of very low certainty of evidence. This downgrading was attributed to reasons such as imprecision, heterogeneity, or risk of bias, and they were detailed for each outcome and comparison as footnotes in the summary of findings tables. Consequently, the evidence for many considered outcomes is characterized as very uncertain.
In the contemporary landscape, there is an increasing body of information concerning the safety and effectiveness of vaccines administered to pregnant women. These accumulating data indicate that the advantages of vaccination for pregnant women surpass the acknowledged or potential risks. The present LSR builds upon and extends the findings of two prior reviews from our group that foreshadowed the safety outcomes reported in this study [7, 62] and with other published systematic reviews that also supported the safety and effectiveness of COVID-19 vaccines during pregnancy [63,64,65,66,67,68,69,70,71,72,73,74]. We show some similarities and differences. We did not find a statistically significantly lower risk of prematurity as other systematic reviews did [63, 66, 71]. However, our analyses showed that COVID-19 vaccination during pregnancy significantly decreases the incidence of stillbirth and emergency cesarean section compared with the unvaccinated group, which was also demonstrated by other reviews [63, 65, 72]. The AEFIs observed in pregnant persons were the same as found in two systematic reviews assessing the effects of immunization in general populations [75, 76]. Local AEFIs such as pain, swelling, erythema, and redness, as well as systemic AEFIs such as fatigue, headache, and myalgia, were consistently the most frequently reported, irrespective of the population under study. Potentially life-threatening adverse events related to vaccination were rarely reported. The proportion of headache, fatigue, fever, and lymphadenopathy was more than double with the second dose compared with the first dose. This finding was consistent with one review [75] but not with the other [76].
Some of the studies in previous systematic reviews included women vaccinated before pregnancy or non‐pregnant individuals, but we focused on vaccinated versus unvaccinated pregnant persons. Previous systematic reviews occasionally amalgamated unadjusted measures with adjusted measures. In contrast, our approach concentrated on observational studies adjusted for pivotal confounders, conducting distinct analyses for crucial maternal and infant safety as well as VE endpoints. These differences in methodology could explain some of the differences in findings. However, irrespective of the methodological precision exhibited in these antecedent systematic reviews, they uniformly affirmed the safety of COVID-19 vaccines during pregnancy.
In light of the inherent biases present in observational data used for assessing VE, these evaluations typically necessitate specific design parameters and definitions including methodologies such as the test-negative design and the adjustment of VE estimates for key confounders in cohort studies. The reference group for all VE analyses conducted in our study comprises pregnant individuals who received zero vaccine doses. Our findings reveal positive albeit varying levels of effectiveness across all vaccine types, including mRNA and non-mRNA vaccines. Vaccine efficacy/effectiveness appears to decline more rapidly in the Omicron era; however, we observed an improvement in VE after booster doses. Additionally, decreased protection of the primary series after 150 or more days since the second dose against the hospitalization endpoint was more evident during the Omicron period than during the earlier Delta period, highlighting the importance of additional doses among pregnant persons, in line with current recommendations [4]. The current findings contribute to bolstering confidence among both the general populace and clinicians, affirming that COVID-19 vaccination serves as a protective measure against severe maternal SARS-CoV-2 infection.
Our study also provides insight into whether vaccination in pregnancy prevents COVID in the infant (through antibody transfer or indirect protection). The available evidence suggests that vaccination during pregnancy protects against COVID-19 hospitalization (severe disease) in infants during their first 6 months of life, although protection is lower in the Omicron period [14, 60, 61]. Similar studies support these findings and thus an addition potential benefit for vaccination during pregnancy [77, 78].
4.2 Strengths
Our LSR has relevant strengths. Our study contributed to the knowledge on the topic by including new studies and providing additional data related to the certainty of evidence using the GRADE method [33]. The utilization of adjusted measures by key confounders, such as age, gestational age, residential area, smoking status, parity, body mass index, or having a seasonal influenza vaccine, or using matched controls for comparing the safety and effectiveness of COVID-19 vaccines in pregnant individuals with those not vaccinated is a noteworthy facet of this study. This approach is critical to ensure that observed effects are not influenced by other variables, thereby enhancing the reliability and validity of our findings. Considering that the number of studies using this approach is limited, we also performed proportional meta-analyses to include many more studies and to provide an alternative perspective on the effects of COVID-19 vaccination during pregnancy. In this way, we estimated the frequency/incidence of maternal and infant safety outcomes after the pregnant persons were exposed to the vaccines, which is crucial to understanding maternal and infant health across trimesters. While the absence of a control group presents a challenge, background rates of maternal-infant events in global, HIC, and LMIC settings served as reference points for contextualizing the findings within the framework of existing maternal-infant health data.
This comprehensive and methodologically rigorous LSR presents separate results by trimester of vaccination, in addition to vaccine type, as the primary analysis of safety maternal and neonatal outcomes since the first trimester is generally considered the most vulnerable period for the fetus in the context of medication or infectious agents [79]. In the same way, in addition to vaccine type and number of doses, we analyzed VE stratified by dominant variant dominance with a focus on the Omicron SARS-CoV-2 variant, the most concerning at the moment of reporting our study. We conducted an exhaustive and regular search across multiple databases and found more studies than any other published systematic review. Another strength is the visualization of up-to-date information. We developed an interactive and public online platform that presented data with less than a month of delay from its publication. Furthermore, we presented effect estimates through customizable real-time meta-analyses of the effectiveness, maternal pregnancy-related outcomes, maternal AEFIs, and infant safety outcomes following COVID-19 vaccination during pregnancy, representing an innovation for policy making and research. No less important, this project had the support of a Strategic and Technical Advisory Group, including immunization experts and WHO representatives, that provided real-time guidance on collecting and analyzing data and reporting it in a way that was most useful in evidence synthesis and decision making.
4.3 Limitations
Our study has also limitations. We did not find RCTs evaluating the safety and efficacy of COVID-19 vaccination during pregnancy because RCTs considered pregnancy an exclusion criterion during the first years of the pandemic. To minimize the inherent risk of bias of observational studies, we only conducted comparative meta-analyses of adjusted estimations, but that limited the number of studies available for a meta-analysis. Nevertheless, several important confounding factors were not considered and may be missed across included studies. We could not pool many studies by outcomes because effect measures were reported by subgroups exposed to vaccinations and compared with the same control group, so the effect measures’ adjusted nature precluded splitting the control group. Additionally, HRs could not be combined with other effect measures such as RRs or ORs, and further limited the number of studies to meta-analyze. Proportional meta-analyses included many more studies, but as they come from non-comparative studies or unadjusted estimates, they are exposed to a high risk of bias.
4.4 Implications for Practice and Research
The implications of this LSR for public health policy, clinical practice, and research are extensive. As the global vaccination campaign against COVID-19 continues, it is imperative to have evidence-based guidelines for pregnant individuals. Although the most urgent phase of the pandemic is over, pregnant women continue to be at risk of adverse outcomes, and they are still recommended to receive a dose of vaccine in every pregnancy, which is why data on the safety and effectiveness of pregnancy must continue to be explored, particularly for non-mRNA vaccines for which there are fewer data. The real-time nature of this LSR permitted timely updates, ensuring access to the most current and pertinent information for making informed decisions regarding vaccination during pregnancy.
5 Conclusions
This LSR established a robust foundation for evaluating the safety and effectiveness of COVID-19 vaccines during pregnancy. By presenting the evidence in an accessible and interactive format that included GRADE summary of findings tables, we also provided a valuable resource to the scientific community, the decision makers, the clinicians, and the general public to protect pregnant persons and their neonates even at the current stage in the pandemic. Further research, including studies with larger sample sizes from different countries and sociodemographic diversity, is required to confirm the external validity of our findings. This work underscores the significance of ongoing research and continuous vaccine safety and effectiveness monitoring, in populations at particular risk for adverse outcomes such as pregnant persons.
References
WHO coronavirus (COVID-19) dashboard. Available from: https://covid19.who.int/. Accessed 20 Apr 2024.
World Health Organization, Others. WHO SAGE roadmap for prioritizing uses of COVID-19 vaccines in the context of limited supply: an approach to inform planning and subsequent recommendations based on epidemiological setting and vaccine supply scenarios, first issued 20 October 2020, latest update 16 July 2021. World Health Organization; 2021. Available from: https://apps.who.int/iris/handle/10665/342917. Accessed 20 Apr 2024.
COVID-19 vaccines. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines. Accessed 20 Apr 2024.
World Health Organization. WHO SAGE roadmap for prioritizing uses of COVID-19 vaccines: an approach to optimize the global impact of COVID-19 vaccines, based on public health goals, global and national equity, and vaccine access and coverage scenarios. Updated 10 November 2023. World Health Organization; 2023. Report No.: WHO/2019-nCoV/Vaccines/SAGE/Prioritization/2022.1. Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-Vaccines-SAGE-Prioritization-2023.1. Accessed 20 Apr 2024.
WHO. COVID-19 vaccine tracker and landscape. Available from: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. Accessed 20 Apr 2024.
Korang SK, von Rohden E, Veroniki AA, Ong G, Ngalamika O, Siddiqui F, et al. Vaccines to prevent COVID-19: a living systematic review with Trial Sequential Analysis and network meta-analysis of randomized clinical trials. PLoS ONE. 2022;17: e0260733. https://doi.org/10.1371/journal.pone.0260733.
Ciapponi A, Bardach A, Mazzoni A, Alconada T, Anderson SA, Argento FJ, et al. Safety of components and platforms of COVID-19 vaccines considered for use in pregnancy: a rapid review. Vaccine. 2021;39:5891–908. https://doi.org/10.1016/j.vaccine.2021.08.034.
Zambrano LD, Ellington S, Strid P, Galang RR, Oduyebo T, Tong VT, et al. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status: United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641–7. https://doi.org/10.15585/mmwr.mm6944e3.
Ciapponi A, Bardach A, Comandé D, Berrueta M, Argento FJ, Rodriguez Cairoli F, et al. COVID-19 and pregnancy: an umbrella review of clinical presentation, vertical transmission, and maternal and perinatal outcomes. PLoS ONE. 2021;16: e0253974. https://doi.org/10.1371/journal.pone.0253974.
Vouga M, Favre G, Martinez-Perez O, Pomar L, Acebal LF, Abascal-Saiz A, et al. Maternal outcomes and risk factors for COVID-19 severity among pregnant women. Sci Rep. 2021;11:13898. https://doi.org/10.1038/s41598-021-92357-y.
Allotey J, Fernandez S, Bonet M, Stallings E, Yap M, Kew T, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ. 2019. https://doi.org/10.1136/bmj.m3320.
Smith ER, Oakley E, Grandner GW, Ferguson K, Farooq F, Afshar Y, et al. Adverse maternal, fetal, and newborn outcomes among pregnant women with SARS-CoV-2 infection: an individual participant data meta-analysis. BMJ Glob Health. 2023;8: e009495. https://doi.org/10.1136/bmjgh-2022-009495.
Sheikh J, Lawson H, Allotey J, Yap M, Balaji R, Kew T, et al. Global variations in the burden of SARS-CoV-2 infection and its outcomes in pregnant women by geographical region and country’s income status: a meta-analysis. BMJ Glob Health. 2022;7: e010060. https://doi.org/10.1136/bmjgh-2022-010060.
Jorgensen SCJ, Hernandez A, Fell DB, Austin PC, D’Souza R, Guttmann A, et al. Maternal mRNA COVID-19 vaccination during pregnancy and delta or omicron infection or hospital admission in infants: test negative design study. BMJ. 2023;380: e074035. https://doi.org/10.1136/bmj-2022-074035.
Badell ML, Dude CM, Rasmussen SA, Jamieson DJ. Covid-19 vaccination in pregnancy. BMJ. 2022;378: e069741. https://doi.org/10.1136/bmj-2021-069741.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions. John Wiley & Sons; 2019. Available from: https://play.google.com/store/books/details?id=cTqyDwAAQBAJ. Accessed 20 Apr 2024.
Elliott JH, Synnot A, Turner T, Simmonds M, Akl EA, McDonald S, et al. Living systematic review: 1 Introduction: the why, what, when, and how. J Clin Epidemiol. 2017;91:23–30.
World Health Organization. Regional Office for Europe. Guidance on an adapted evidence to recommendation process for National Immunization Technical Advisory Groups. World Health Organization. Regional Office for Europe; 2022. Report No.: WHO/EURO:2022-5497-45262-64756. Available from: https://apps.who.int/iris/handle/10665/356896. Accessed 20 Apr 2024.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 2021;18: e1003583. https://doi.org/10.1371/journal.pmed.1003583.
Kahale LA, Elkhoury R, El Mikati I, Pardo-Hernandez H, Khamis AM, Schünemann HJ, et al. Tailored PRISMA 2020 flow diagrams for living systematic reviews: a methodological survey and a proposal. F1000Res. 2021;10:192.
Ciapponi A, Berrueta M, Ballivian J, Bardach A, Mazzoni A, Anderson S, et al. Safety, immunogenicity, and effectiveness of COVID-19 vaccines for pregnant persons: a protocol for systematic review and meta analysis. Medicine. 2023;102: e32954. https://doi.org/10.1097/MD.0000000000032954.
Covidence. Covidence systematic review software, veritas health innovation. Melbourne (VIC); 2021.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap): a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81. https://doi.org/10.1016/j.jbi.2008.08.010.
NIH. Study quality assessment tools. 2020. Available from: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools. Accessed 20 Apr 2024.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88. https://doi.org/10.1016/0197-2456(86)90046-2.
The R project for statistical computing. Available from: https://www.R-project.org/. Accessed 17 Jun 2024.
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22:153–60. https://doi.org/10.1136/ebmental-2019-300117.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48. Available from: https://www.jstatsoft.org/article/view/v036i03. Accessed 20 Apr 2024.
Wickham H, Averick M, Bryan J, Chang W, McGowan L, François R, et al. Welcome to the tidyverse. J Open Source Softw. 2019;4:1686.
Freeman MF, Tukey JW. Transformations related to the angular and the square root. Ann Math Stat. 1950;21:607–11.
Taylor I, Knowelden J. Principles of epidemiology. Little, Brown; 1964. Available from: https://play.google.com/store/books/details?id=STRrAAAAMAAJ. Accessed 20 Apr 2024.
Bonhoeffer J, Kochhar S, Hirschfeld S, Heath PT, Jones CE, Bauwens J, et al. Global alignment of immunization safety assessment in pregnancy: the GAIA project. Vaccine. 2016;34:5993–7. https://doi.org/10.1016/j.vaccine.2016.07.006.
Schünemann G, Vist J, Higgins N, Santesso J, Deeks P. Chapter 14: completing ‘summary of findings’ tables and grading the certainty of the evidence. In: Higgins J, Thomas J, editors. Cochrane handbook for systematic reviews of interventions version. London: Cochrane; 2019. p. 6.
Safe in pregnancy: living systematic review. Available from: https://safeinpregnancy.org/lsr/. [Accessed 20 Apr 2024.]
Chang W, Cheng J, Allaire J, Xie Y, McPherson J. Shiny: web application framework for R. R package version.
Hui L, Marzan MB, Rolnik DL, Potenza S, Pritchard N, Said JM, et al. Reductions in stillbirths and preterm birth in COVID-19-vaccinated women: a multicenter cohort study of vaccination uptake and perinatal outcomes. Am J Obstet Gynecol. 2023;228:585.e1-585.e16. https://doi.org/10.1016/j.ajog.2022.10.040.
Fell DB, Dimanlig-Cruz S, Regan AK, Håberg SE, Gravel CA, Oakley L, et al. Risk of preterm birth, small for gestational age at birth, and stillbirth after covid-19 vaccination during pregnancy: population based retrospective cohort study. BMJ. 2022;378: e071416. https://doi.org/10.1136/bmj-2022-071416.
Magnus MC, Örtqvist AK, Dahlqwist E, Ljung R, Skår F, Oakley L, et al. Association of SARS-CoV-2 vaccination during pregnancy with pregnancy outcomes. JAMA. 2022;327:1469–77. https://doi.org/10.1001/jama.2022.3271.
Kharbanda EO, Haapala J, DeSilva M, Vazquez-Benitez G, Vesco KK, Naleway AL, et al. Spontaneous abortion following COVID-19 vaccination during pregnancy. JAMA. 2021;326:1629–31. https://doi.org/10.1001/jama.2021.15494.
Magnus MC, Gjessing HK, Eide HN, Wilcox AJ, Fell DB, Håberg SE. COVID-19 vaccination during pregnancy and first-trimester miscarriage. N Engl J Med. 2021;385:2008–10. https://doi.org/10.1056/NEJMc2114466.
Citu IM, Citu C, Gorun F, Sas I, Bratosin F, Motoc A, et al. The risk of spontaneous abortion does not increase following first trimester mRNA COVID-19 vaccination. J Clin Med Res. 2022. https://doi.org/10.3390/jcm11061698.
Dick A, Rosenbloom JI, Gutman-Ido E, Lessans N, Cahen-Peretz A, Chill HH. Safety of SARS-CoV-2 vaccination during pregnancy- obstetric outcomes from a large cohort study. BMC Pregnancy Childbirth. 2022;22:166. https://doi.org/10.1186/s12884-022-04505-5.
Wainstock T, Yoles I, Sergienko R, Sheiner E. Prenatal maternal COVID-19 vaccination and pregnancy outcomes. Vaccine. 2021;39:6037–40. https://doi.org/10.1016/j.vaccine.2021.09.012.
Peretz-Machluf R, Hirsh-Yechezkel G, Zaslavsky-Paltiel I, Farhi A, Avisar N, Lerner-Geva L, et al. Obstetric and neonatal outcomes following COVID-19 vaccination in pregnancy. J Clin Med Res. 2022. https://doi.org/10.3390/jcm11092540.
Blakeway H, Prasad S, Kalafat E, Heath PT, Ladhani SN, Le Doare K, et al. COVID-19 vaccination during pregnancy: coverage and safety. Am J Obstet Gynecol. 2022;226(236):e1-14. https://doi.org/10.1016/j.ajog.2021.08.007.
Dick A, Rosenbloom JI, Karavani G, Gutman-Ido E, Lessans N, Chill HH. Safety of third SARS-CoV-2 vaccine (booster dose) during pregnancy. Am J Obstet Gynecol MFM. 2022;4: 100637. https://doi.org/10.1016/j.ajogmf.2022.100637.
Fell DB, Dhinsa T, Alton GD, Török E, Dimanlig-Cruz S, Regan AK, et al. Association of COVID-19 vaccination in pregnancy with adverse peripartum outcomes. JAMA. 2022;327:1478–87. https://doi.org/10.1001/jama.2022.4255.
Brinkley E, Mack CD, Albert L, Knuth K, Reynolds MW, Toovey S, et al. COVID-19 vaccinations in pregnancy: comparative evaluation of acute side effects and self-reported impact on quality of life between pregnant and nonpregnant women in the United States. Am J Perinatol. 2022;39:1750–3. https://doi.org/10.1055/s-0042-1748158.
Kachikis A, Englund JA, Covelli I, Frank Y, Haghighi C, Singleton M, et al. Analysis of vaccine reactions after COVID-19 vaccine booster doses among pregnant and lactating individuals. JAMA Netw Open. 2022;5: e2230495. https://doi.org/10.1001/jamanetworkopen.2022.30495.
Shapiro Ben David S, Baruch Gez S, Rahamim-Cohen D, Shamir-Stein N, Lerner U, Ekka Zohar A. Immediate side effects of Comirnaty COVID-19 vaccine: a nationwide survey of vaccinated people in Israel, December 2020 to March 2021. Euro Surveill. 2022. https://doi.org/10.2807/1560-7917.ES.2022.27.13.2100540.
Goldshtein I, Steinberg DM, Kuint J, Chodick G, Segal Y, Shapiro ben David S, et al. Association of BNT162b2 COVID-19 vaccination during pregnancy with neonatal and early infant outcomes. JAMA Pediatr. 2022;176:470–7. https://doi.org/10.1001/jamapediatrics.2022.0001.
Lipkind HS, Vazquez-Benitez G, DeSilva M, Vesco KK, Ackerman-Banks C, Zhu J, et al. Receipt of COVID-19 vaccine during pregnancy and preterm or small-for-gestational-age at birth: eight integrated health care organizations, United States, December 15, 2020-July 22, 2021. MMWR Morb Mortal Wkly Rep. 2022;71:26–30. https://doi.org/10.15585/mmwr.mm7101e1.
Paixao ES, Wong KLM, Alves FJO, de Araújo OV, Cerqueira-Silva T, Júnior JB, et al. CoronaVac vaccine is effective in preventing symptomatic and severe COVID-19 in pregnant women in Brazil: a test-negative case-control study. BMC Med. 2022;20:146. https://doi.org/10.1186/s12916-022-02353-w.
Dagan N, Barda N, Biron-Shental T, Makov-Assif M, Key C, Kohane IS, et al. Effectiveness of the BNT162b2 mRNA COVID-19 vaccine in pregnancy. Nat Med. 2021;27:1693–5. https://doi.org/10.1038/s41591-021-01490-8.
Florentino PTV, Alves FJO, Cerqueira-Silva T, de Araújo OV, Júnior JBS, Penna GO, et al. Effectiveness of BNT162b2 booster after CoronaVac primary regimen in pregnant people during omicron period in Brazil. Lancet Infect Dis. 2022;22:1669–70. https://doi.org/10.1016/S1473-3099(22)00728-9.
Morgan JA, Biggio JR Jr, Martin JK, Mussarat N, Chawla HK, Puri P, et al. Maternal outcomes after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107–9. https://doi.org/10.1097/AOG.0000000000004621.
Villar J, Soto Conti CP, Gunier RB, Ariff S, Craik R, Cavoretto PI, et al. Pregnancy outcomes and vaccine effectiveness during the period of omicron as the variant of concern, INTERCOVID-2022: a multinational, observational study. Lancet. 2023;401:447–57. https://doi.org/10.1016/S0140-6736(22)02467-9.
Bosworth ML, Schofield R, Ayoubkhani D, Charlton L, Nafilyan V, Khunti K, et al. Vaccine effectiveness for prevention of covid-19 related hospital admission during pregnancy in England during the alpha and delta variant dominant periods of the SARS-CoV-2 pandemic: population based cohort study. BMJ Med. 2023;2: e000403. https://doi.org/10.1136/bmjmed-2022-000403.
Schrag SJ, Verani JR, Dixon BE, Page JM, Butterfield KA, Gaglani M, et al. Estimation of COVID-19 mRNA vaccine effectiveness against medically attended COVID-19 in pregnancy during periods of delta and omicron variant predominance in the United States. JAMA Netw Open. 2022;5: e2233273. https://doi.org/10.1001/jamanetworkopen.2022.33273.
Danino D, Ashkenazi-Hoffnung L, Diaz A, Erps AD, Eliakim-Raz N, Avni YS, et al. Effectiveness of BNT162b2 vaccination during pregnancy in preventing hospitalization for severe acute respiratory syndrome coronavirus 2 in infants. J Pediatr. 2023;254:48-53.e1. https://doi.org/10.1016/j.jpeds.2022.09.059.
Halasa NB, Olson SM, Staat MA, Newhams MM, Price AM, Pannaraj PS, et al. Maternal vaccination and risk of hospitalization for covid-19 among infants. N Engl J Med. 2022;387:109–19. https://doi.org/10.1056/nejmoa2204399.
Ciapponi A, Berrueta M, Bardach A, Mazzoni A, Anderson SA, Argento FJ, et al. Safety of COVID-19 vaccines during pregnancy: a systematic review and meta-analysis. 2022. Available from: https://papers.ssrn.com/abstract=4072487. Accessed 25 May 2022.
Rahmati M, Yon DK, Lee SW, Butler L, Koyanagi A, Jacob L, et al. Effects of COVID-19 vaccination during pregnancy on SARS-CoV-2 infection and maternal and neonatal outcomes: a systematic review and meta-analysis. Rev Med Virol. 2023;33: e2434. https://doi.org/10.1002/rmv.2434.
Rimmer MP, Teh JJ, Mackenzie SC, Al Wattar BH. The risk of miscarriage following COVID-19 vaccination: a systematic review and meta-analysis. Hum Reprod. 2023;38:840–52. https://doi.org/10.1093/humrep/dead036.
Ding C, Liu Y, Pang W, Zhang D, Wang K, Chen Y. Associations of COVID-19 vaccination during pregnancy with adverse neonatal and maternal outcomes: a systematic review and meta-analysis. Front Public Health. 2023;11:1044031. https://doi.org/10.3389/fpubh.2023.1044031.
Shafiee A, Kohandel Gargari O, Teymouri Athar MM, Fathi H, Ghaemi M, Mozhgani S-H. COVID-19 vaccination during pregnancy: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2023;23:45. https://doi.org/10.1186/s12884-023-05374-2.
Zhang D, Huang T, Chen Z, Zhang L, Gao Q, Liu G, et al. Systematic review and meta-analysis of neonatal outcomes of COVID-19 vaccination in pregnancy. Pediatr Res. 2023;94:34–42. https://doi.org/10.1038/s41390-022-02421-0.
Tormen M, Taliento C, Salvioli S, Piccolotti I, Scutiero G, Cappadona R, et al. Effectiveness and safety of COVID-19 vaccine in pregnant women: a systematic review with meta-analysis. BJOG. 2023;130:348–57. https://doi.org/10.1111/1471-0528.17354.
Hameed I, Khan MO, Nusrat K, Mahmood S, Nashit M, Malik S, et al. Is it safe and effective to administer COVID-19 vaccines during pregnancy? A systematic review and meta-analysis. Am J Infect Control. 2023;51:582–93. https://doi.org/10.1016/j.ajic.2022.08.014.
Hagrass AI, Almadhoon HW, Al-Kafarna M, Almaghary BK, Nourelden AZ, Fathallah AH, et al. Maternal and neonatal safety outcomes after SAR-CoV-2 vaccination during pregnancy: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2022;22:581. https://doi.org/10.1186/s12884-022-04884-9.
Carbone L, Trinchillo MG, Di Girolamo R, Raffone A, Saccone G, Iorio GG, et al. COVID-19 vaccine and pregnancy outcomes: a systematic review and meta-analysis. Int J Gynaecol Obstet. 2022;159:651–61. https://doi.org/10.1002/ijgo.14336.
Prasad S, Kalafat E, Blakeway H, Townsend R, O’Brien P, Morris E, et al. Systematic review and meta-analysis of the effectiveness and perinatal outcomes of COVID-19 vaccination in pregnancy. Nat Commun. 2022;13:2414. https://doi.org/10.1038/s41467-022-30052-w.
Wu S, Wang L, Dong J, Bao Y, Liu X, Li Y, et al. The dose- and time-dependent effectiveness and safety associated with COVID-19 vaccination during pregnancy: a systematic review and meta-analysis. Int J Infect Dis. 2023;128:335–46. https://doi.org/10.1016/j.ijid.2023.01.018.
Wei SQ, Bilodeau-Bertrand M, Liu S, Auger N. The impact of COVID-19 on pregnancy outcomes: a systematic review and meta-analysis. CMAJ. 2021;193:E540–8. https://doi.org/10.1503/cmaj.202604.
Katoto PDMC, Brand AS, Byamungu LN, Tamuzi JL, Mahwire TC, Kitenge MK, et al. Safety of COVID-19 Pfizer-BioNtech (BNT162b2) mRNA vaccination in adolescents aged 12–17 years: a systematic review and meta-analysis. Hum Vaccin Immunother. 2022;18:2144039. https://doi.org/10.1080/21645515.2022.2144039.
Chen M, Yuan Y, Zhou Y, Deng Z, Zhao J, Feng F, et al. Safety of SARS-CoV-2 vaccines: a systematic review and meta-analysis of randomized controlled trials. Infect Dis Poverty. 2021;10:94. https://doi.org/10.1186/s40249-021-00878-5.
Zerbo O, Ray GT, Fireman B, Layefsky E, Goddard K, Lewis E, et al. Maternal SARS-CoV-2 vaccination and infant protection against SARS-CoV-2 during the first 6 months of life. Res Sq. 2022. https://doi.org/10.21203/rs.3.rs-2143552/v1.
Munoz FM, Posavad CM, Richardson BA, Badell ML, Bunge KE, Mulligan MJ, et al. COVID-19 booster vaccination during pregnancy enhances maternal binding and neutralizing antibody responses and transplacental antibody transfer to the newborn. Vaccine. 2023;41:5296–303. https://doi.org/10.1016/j.vaccine.2023.06.032.
DeSilva M, Munoz FM, Mcmillan M, Kawai AT, Marshall H, Macartney KK, et al. Congenital anomalies: case definition and guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2016;34:6015–26. https://doi.org/10.1016/j.vaccine.2016.03.047.
Acknowledgments
We thank Flor M. Muñoz for her supervision and general support and Federico Rodriguez Cairoli, Victoria Santa María, Natalia Zamora, and Sabra Zaraa for their participation as researchers in the initial stage of the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported, as a whole, by the Bill & Melinda Gates Foundation (INV008443). Under the grant conditions of the Foundation, a Creative Commons Attribution 4.0 Generic License has already been assigned to the author accepted manuscript version that might arise from this submission. The sponsors had no role in conducting the present study.
Conflicts of Interest/Competing Interests
Agustín Ciapponi, Mabel Berrueta, Fernando J. Argento, Jamile Ballivian, Ariel Bardach, Martin E. Brizuela, Noelia Castellana, Daniel Comandé, Sami Gottlieb, Beate Kampmann, Agustina Mazzoni, Edward P.K. Parker, Juan M. Sambade, Katharina Stegelmann, Xu Xiong, Andy Stergachis, and Pierre Buekens have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
Systematic reviews do not require any original research and are not subject to ethics approval.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Material
Data and materials are available in the supplemental material and at https://www.safeinpregnancy.org/living-systematic-review/.
Code Availability
Not applicable.
Authors’ Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by AC, MB, FA, JB, AB, NC, DC, MEB, JMS, KS, and AM. The first draft of the manuscript was written by AC and MB, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ciapponi, A., Berrueta, M., Argento, F.J. et al. Safety and Effectiveness of COVID-19 Vaccines During Pregnancy: A Living Systematic Review and Meta-analysis. Drug Saf 47, 991–1010 (2024). https://doi.org/10.1007/s40264-024-01458-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-024-01458-w