Abstract
The progressive reduction of synthetic agrochemical fertilizers is one of the key factors in the shift from conventional agriculture to sustainable farming. Nitrogen (N) is the ruling element in the development of agricultural production, but its use in the mineral form or its excessive use causes several environmental issues. Since the release of N nanocomposites coincides with their uptake by crops, N loss reduces while enhancing plant uptake due to nano fertilizers application. Additionally, an intercropping legume with cereal as an eco-friendly pattern could improve and rationalize the nitrogenous inputs. Therefore, a two-year field trial was conducted to determine the efficacy of nano-chitosan-loaded N (CS-NNPs) for saving mineral N amounts applied in maize-based on maize-soybean intercropping and enhancing land productivity. Methods In a randomized split-plot design in three replicates, three intercropping patterns, in addition to the sole crops, and three N levels were implemented. Intercropping involved three intercrop configurations [planting maize rows (M) alternated with soybean rows (S) in patterns of 4M:2S, 2M:4S, and 3M:3S)], in addition to planting sole maize crop (SMC) and sole soybean crop (SSC). N fertilization treatments included adding 288 kg N ha−1 (MN100%) and two levels of CS-NNPs composite involving 216 kg N ha−1 + 2 foliar sprays of CS-NNPs (MN75% + 2CS-NNPs), and 144 kg N ha−1 + 3 foliar sprays of CS-NNPs composite (MN50% + 3CS-NNPs). Under the tested treatments, the agronomic traits, intercropping indices, and economic benefits were estimated. Results Findings revealed that the application of SMC × MN75% + 2CS-NNPs, followed by 4M:2S × MN75% + 2CS-NNPs showed the highest growth, biological yield, and grain yield of maize. The interaction of SSC × MN75% + 2CS-NNPs, followed by 2M:4S × MN75% + 2CS-NNPs resulted in the highest seed yield components, biological yield, straw yield, and seed yield of soybean. Application of 2M:4S × MN100%, 2M:4S × MN50% + 3NNPs, and 3M:3S × MN100% recorded the maximum total land equivalent ratio. While applications of 2M:4S × MN100%, 2M:4S × MN75% + 2CS-NNPs, and 3M:3S × MN100% achieved the highest land equivalent coefficient, land-use efficiency, area time equivalent ratio, and percent yield difference. Likewise, both interactions of 2M:4S × MN75% + 2CS-NNPs and 3M:3S × MN100% recorded the highest system productivity index. Better yield advantage of maize-soybean intercrop compared with the monocrop since total actual yield loss values were positive and higher than zero in all interactions of intercropping pattern × N fertilization. Fertilizing maize with MN50% + 3CS NNPs grown under the 2M:4S pattern had the highest positive aggressivity values. Conclusion The productivity shortfall accompanying the 25% N reduction was compensated by the application of CS-NNPs. Thus, N applied to the maize intercropped with soybeans can be rationalized. This undoubtedly has a good economic payoff for the maize growers with the conservation of the agricultural environment. In maize production systems, it is advisable to fertilize the plants using 216 kg instead of 288 kg nitrogen ha−1 when nano chitosan-loaded nitrogen composite twice (0.48 kg nitrogen ha−1) applied.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sustainable agriculture is a pattern of agriculture that aims to increase the efficiency of resources utilization (Alghamdi et al., 2023; Zulfiqar et al., 2019), provide the present-day demands as well as those of future generations, restore diversity to agricultural ecosystems, and achieve environmental balance through its effective management (Mekdad et al., 2022; Mousavi & Eskandari, 2011). Intercropping is considered an example of a sustainable agricultural system that contributes to achieving agro-ecological balance, better utilization of available growth resources such as nutrients, water, and light, increasing the productivity per unit of land, and reducing yield damage by pests, diseases, and weeds (Brooker et al., 2015). In arid and semi-arid regions of the world, particularly in Africa, intercropping has anciently been practiced for several decades, and it is still acquiring popularity among smallholder crop growers (Ghosh et al., 2006; Kermah et al., 2017). Intercropping is a combination of two or more crops cultivated in the same field and growing season and aimed to increase diversity in the agricultural ecosystem (Fathi, 2014). The difference in response of intercropping components to edaphic and climatic factors is always served to maximize their total output (Thierfelder et al., 2017). Legumes and non-legumes (Saudy & El-Metwally, 2009; Saudy & El-Bagoury, 2014; Sheha et al., 2023) as well as C3 and C4 crops were successful systems in different intercropping patterns, where the differences in plant architecture helped a lot in this situation (Layek et al., 2018).
Several studies were conducted to evaluate the productivity of maize-soybean in intercropping systems, for example, Sani et al. (2014) found that the maize yield was higher in intercropping than in monoculture crops. In addition, the maize-based intercropping system with legumes (i.e., soybean, cowpea, and pea) helps in improving soil health as well as crop yield (Beedy et al., 2010; Saudy, 2015). Maize plant development is strongly dependent on the abundance of soil nitrogen (N) and N use efficiency for biomass production and yield (Sonnewald, 2013). It is well documented hat nitrogen (N) is regarded as one of the most significant prime nutrients are needed by crop palms. Accordingly, N supply improves and maximizes the growth and yield of various field crops (Noureldin et al., 2013; Saudy, 2014; Saudy & El-Metwally, 2023; Saudy et al., 2020). On the other hand, soybean is regarded as a legume plant, a fixer for atmospheric N when properly nodulated, and thus is less dependent for growth on soil N content (Flynn & Idowu, 2015).
Nanoscience technology is a promising way for achieving a revolution in agriculture (Chen & Yada, 2011). Nanoparticle (nanoscale particles, NSPs) materials, with a size of 1–100 nm, created by nanotechnology (Suguna et al., 2017) can be exploited in several ecological aspects (Khan et al., 2017). Compared to the bulk form, transforming the substance into a nano shape can quite change its physical–chemical traits (Ferreira et al., 2011). Huge attempts have been implemented to minimize nutrient losses while increasing the crop yield quantity and quality (Abd-Elrahman et al. 2022; Yang et al., 2016; Saudy & El-Metwally, 2019). In this regard, approximately 50–70% losses in conventional N fertilizers occurred in various ways resulting in reduced fertilizer efficiency and an increase in production cost (Wang et al., 2015). On the contrary, the release of nano N compounds is coinciding with its uptake by crops (Dwivedi et al., 2016). Hence, the losses of N were reduced by regulating the demand-based release while enhancing the plant uptake process owing to the application of porous nanomaterials such as chitosan (Panpatte et al., 2016). Chemically, chitosan is obtained by deacetylation of chitin (Sorlier et al., 2001). The controlled-release chitosan microspheres loaded with nitrogen could be slowly liberated into the soil meeting the required nutrient of plants at different stages with reducing the nutrient loss and improving fertilizer utilization (Giroto et al., 2017). Due to its potential to increase the uptake of water and nutrients availability, chitosan improves plant growth and development (Bibi et al., 2021; Hidangmayum et al., 2019). Root and vegetative growth, plant pigments and dry matter production were enhanced by chitosan supply (Ali et al., 2020; Marzouk et al., 2022). Furthermore, chitosan contributes to raising the defensive mechanism stimulation against abiotic stresses (Guan et al., 2009a, 2009b). Balanced fertilizer management will increase agricultural production, which amounted to 35–40% (Khodabin et al., 2022; Shaaban et al., 2023a). It has been keenly observed that the application of nano-formulated fertilizers has significant potential to increase crop productivity (Millán et al., 2008). However, very limited information relevant to the integration between intercropping and nanocarrier-loaded N are available. Keeping in view the above-mentioned aspects, the current study hypothesized that the application of nano-chitosan-loaded N could have the potential to improve the productivity of the different intercropped maize and soybean along with saving the applied mineral N in maize. Thus, the study objective was to evaluate the effect of different maize-soybean intercropping patterns on yield traits, intercropping indices, and economic benefits under foliage-sprayed nano-chitosan-loaded N.
Materials and Methods
Experimental Field Site
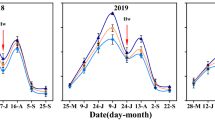
A two-year field experiment was conducted at Gemmieza Agricultural Research Station in El-Gharbia governorate, Egypt (latitude: 30° 48′ 11′′ N and longitude: 31° 08′ 22′′ E) during the 2019 and 2020 cropping summer seasons. The study region has four distinct agro-seasons and is characterized by semi-arid climatic conditions with no precipitation during the experimental period (May–September). The experiment site meteorological (temperatures, relative air humidity, wind speed, and solar radiation), obtained from Central Laboratory of Meteorology, Ministry of Agriculture and Land Reclamation, Egypt, are illustrated in Fig. 1. Following the zigzag sampling method, a representative composite sample was randomly collected from the upper 0.4m soil horizon from the experimental field directly before the sowing date to determine the soil's physico-chemical properties (Table 1) following the methods suggested by Page et al. (1982) and Klute (1986). The hydrometer method suggested by Gee and Or (2002) was used to identify the particle size distribution of soil. The pH was determined in the soil paste extract using a pH meter in a 1:2.5 (w/ν) soil: H2O suspension. Electrical conductivity (ECe in dS m−1) was also measured directly in the saturated soil paste using a Metler conductivity meter. Collin's calcimeter apparatus was used to measure volumetrically the CaCO3 content of the experimental soil. Organic matter content (%) was calculated by multiplying the soil organic carbon (determined following the Walkley–Black dichromate acid procedure) by the conversion factor of 1.724. The N content (mg kg−1 soil) was determined using the H2SO4-HClO4 (3:1, v/v) micro Kjeldahl method (Medical Instruments Co., Ningbo, China). The molybdenum blue colorimetric procedure using a UV-160A spectrophotometer (Shimadzu, Japan) was used for measuring the soil P content (mg kg−1 soil) in the same H2SO4-HClO4 digestion mixture used to determine N content. Potassium content (mg kg−1 soil) was determined using NH4OAC at pH 7 procedure using an ICP-AES inductively coupled plasma atomic-emission spectrometry (Thermo Inc., IRIS-Advan, MA, USA). The soil is clay in texture and typified as Vertic Torrifluvents according to the soil taxonomy, USDA (Soil Survey Staff, 1999).
Experimental Layout, Treatment Details, and Crops Management
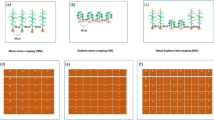
The experiment was laid out in a randomized complete block design with a split-plot arrangement with three replicates. The main plots involved three intercrop patterns (planting four maize rows alternated with two rows of soybean (4M:2S), planting two maize rows alternated with four rows of soybean (2M:4S), and planting three maize rows alternated with two rows of soybean (3M:3S), in addition to planting sole maize crop (SMC) and sole soybean crop (SSC) (Fig. 2). Sub-plots were allocated to three N fertilization treatments including fertilization with 288 kg N ha−1 (100% of mineral N (MN100%), as a recommended dose (RD) as well as nano-chitosan loaded N (CS-NNPs) involving 216 kg N ha−1 as 75% of RD + 2 foliar sprays of CS-NNPs composite (MN75% + 2CS-NNPs) and 144 kg N ha−1 as 50% of RD + 3 foliar sprays of CS-NNPs composite (MN50% + 3CS-NNPs). Mineral N in the form of urea (CO(NH2)2) containing 46% N was applied to maize plants in two equal portions at 20 and 35 days after planting (DAP). According to CS-NNPs treatment, the spray solution was applied to synchronize 35 and 50 DAP (for the treatment involved two sprays) and 35, 50, and 65 DAP (for the treatment involved three sprays), at a rate of 500 mg N L−1 using 480 L ha−1 of water as carrier/solvent. Tween-20, a surfactant agent, was mixed with each applied spray to guarantee an efficacious penetration to CS-NNPs in maize leaves. Each sub-plot area in mono or intercropping patterns was included 6 rows, 3M in length, and 0.7 m apart, forming an area of 12.6 m2. Grains of yellow maize single-cross 168, a local hybrid commonly cultivated by farmers, were manually planted on May 29, 2019, and May 20, 2020, on one side of the ridge at a uniform depth of 0.03–0.05 m with two grains per hill at an inter-hill spacing of 0.25 m. Similarly, the soybean Giza 111 cultivar seeds were planted on one side of the ridge at 0.03–0.05 m depth at an inter-seed spacing of 0.2M.
In both seasons, maize plants were fertilized with P and K fertilizers at rates of 54 kg P2O5 ha−1 in the form of monocalcium phosphate (15.5% P2O5) during land preparation and 119 kg K2O ha−1 in the form of potassium sulfate (48% K2O) at 28 DAP. The soybean seeds were inoculated immediately before planting with Bradyrhizobium japonicum as a bacterial symbiont (obtained from the Egyptian Agriculture Ministry). Soybean plants received a starter N dose of 15 kg ha−1 due to the low N-fixing potential of legumes at the initial growth stages, and such a dose is necessary to ensure healthy growth and uniform soybean plant stands (Gai et al., 2017). A surface flood irrigation system was used (common practice in the study area), where plants were irrigated 12–15 days interval. Weeding was done on all experimental plots at 24 DAP concurrently with hilling-up for maize and soybean plants by displacing soil from the unplanted inter-ridge space.
Preparation and Characterization of CS-NNPs Composite
Following the protocol of Corradini et al. (2010), the CS-NNPs composite was prepared by the polymerization of methacrylic acid (PMAA) in a CS solution followed by the loading of the N element in two consecutive steps. Firstly, the nano-chitosan (CS-NPs) suspension was prepared using a waterish solution of 0.2% (w/v) of medium molecular weight CS (190–310 kDa and ≥ 75% deacetylated) and PMAA (0.5%; v/v) under continuous magnetic stirring at 600 rpm for 12 h using an MSH-20D stirrer device (Wise Stir, Korea). Secondly, 0.2Mmol of alkaline K2S2O8, a radical initiator agent, was added to this mixture with continuous stirring until attaining a clear homogeneous solution. These reactant substances were sealed under refluxing conditions and stirred well at 70 °C for 2 h to form chitosan polymerization methacrylic acid nanoparticles (CS-PMAA-NPs) and then quickly cooled in an ice-bath (de Moura et al., 2008; Hasaneen et al., 2014). This CS-PMMAA-NPs suspension is used for N loading. An appropriate amount of CO(NH2)2 equivalent to 500 ppm N is dissolved in a given volume of the above suspension under magnetic stirring for 8 h at 25 °C to obtain the CS-NNPs. The surface of nanosheets (NSs)-like morphology and the elemental distribution of CS-NNPs were characterized by a field-emission scanning electron microscope (FE-SEM) apparatus (Carl ZEISS AG, sigma 500 VP, Germany) attached with energy-dispersive X-ray spectroscopy (EDS) unit. The average size and zeta potential of the obtained CS-NNPs were determined by measuring zeta size using a Zetasizer Nano-ZS system (Malvern Instruments Ltd., Worcestershire, UK).
Agronomic Traits and Maize Photosynthetic Partitioning of Maize-Soybean Intercrop
At the fully ripe (BBCH 89, kernels hard and shiny; Meier, 2001) stage of maize, five plants were randomly selected from each sub-plot to measure plant height, leaves no. plant−1, ear leaf area, leaf area index (LAI), ear length, kernels weight ear−1, ears weight plant−1, biological yield plant−1, and grain yield plant−1. Similarly, ten soybean plants, at full seed maturity (BBCH 89) stage, were randomly selected to measure plant height, branches no. plant−1, pods no. plant−1, seed yield plant−1, and 100-seed weight. Grain and seed yield for maize and soybean, respectively, as well as biological yield, were determined based on sub-plot yield plus the yield of the individual maize and soybean plants sampled before and were converted into t ha−1. For adjusting grain/seed yields of maize and soybean-based on 14% and 12% moisture content, respectively. A 200-g subsample of grain/seed was weighed, dried at 105 ± 2 °C to a constant weight, and then weighed additional for final adjusted grain yield computation. To determine the grain, biological, and vegetative yield contributions per unit leaf area in maize crop, the relative photosynthetic potentials (RPPs) for grain, biological, and vegetative yields, were calculated according to McGraw (1977) and Snyder and Carlson (1984) as follows: RPP for grain yield (g m−2) = grain yield plant−1/LAI, RPP for biological yield (g m−2) = biological yield plant−1(g)/LAI, and RPP for vegetative yield (g m−2) = RPP for biological yield—RPP for grain yield.
Biological Efficiency of Maize-Soybean Intercrop
The biological efficiency of the maize-soybean intercropping system was evaluated using the land equivalent ratio (LER), land equivalent coefficient (LEC), land-use efficiency (LUE), area time equivalent ratio (ATER), system productivity index (SPI), and percent yield difference (PYD). The LER as described by Mead and Willey (1980) indicates the relative area of the monocrop that is required to produce an equivalent yield obtainable under intercropping as follows:
where LERm and LERs indicate the partial LER of maize and soybean under the intercropping patterns, respectively, whereas Ymi and Ysi represent the grain and seed yield of maize and soybean, respectively, under the intercropping patterns. In contrast, Yms and Yss indicate the respective yields under the sole crop. LER values above 1 refer to intercropping yield advantage, whilst those below 1 refer to a disadvantage of intercropping thus, advising for the cultivation of the respective crops as monocrops (Machiani et al., 2018; Mead & Willey, 1980).
The LEC proposed by Adetiloye et al. (1983) is a product of LERm and LERs as follows: LEC = (Ymi × Ysi)/(Yms × Yss). The LUE was calculated using LER and ATER values (Mead & Willey, 1980; Yaseen et al., 2014) according to the following equation. LUE = [LER + (ATER/2)] × 100. The ATER was suggested as a modification for LER (Mead & Willey, 1980). It was calculated to compare the yield advantage of maize-soybean intercropping to the sole cropping, considering the number of hectare-days from planting to maturity by the component crops (Hiebsch & McCollum, 1987; Doubi et al. 2016) under intercropping patterns as follows: ATER = [(LERm × dm) + (LERs × ds)]/D, where dm and ds denote the growth period of maize and soybean, respectively, in days from planting to maturity. D denotes the duration of the component crops in days with the longest growing period.
The SPI was used to evaluate the yield stability of the intercropping patterns by standardizing the soybean yield as a secondary crop in terms of maize as a primary crop (Agegnehu et al., 2006; Odo, 1991) as follows: SPI = Ymi + [(Yms/Yss) × Ysi]. The PYD proposed by Afe and Atanda (2015), by definition, indicates the percentage yield difference between the sole crop and the intercrop. The sole crop yield is assumed to be 100% and a loss in one component crop's yield is commonly compensated by an increase in the companion crop's yield. In contrast to the other indices, the greater the PYD value, the lower the intercropping system's efficiency, and vice-versa (Afe & Atanda, 2015). The PYD was calculated as: PYD = 100—[(Yms – Ymi)/Yms) + (Yss – Ysi)/Yss)] × 100.
Interspecific Competition Indices in Maize-Soybean Intercrop
Relative crowding coefficient (RCC), actual yield loss (AYL), and aggressivity (AG) were utilized to evaluate the interspecific competitive effect between the maize and soybean crops in different intercropping patterns. The RCC proposed by Ghosh (2004) is used to evaluate the relative dominance or aggressiveness of either soybean on maize or vice-versa (Gitari et al., 2020; Lithourgidis et al., 2011) in intercropping patterns. The RCC was calculated as:
where RCCm and RCCs represent the partial RCC of maize and soybean under the intercropping patterns, respectively. Zm denotes the sown proportion (%) of maize to soybean in the mixture whereas Zs denotes that of soybean to maize. RCC value > 1 indicates the species is more competitive, when RCC value = 1 there is no competition between the two intercropped species, and when RCC < 1 the species is less efficient in environmental resource utilization resulting in a relative productivity loss. The AYL is a competition indicator that reflects the equivalent yield loss or gain of component crops compared with their pure stand, providing more detailed information about the inter- and intra-competition of intercrops (Banik, 1996). In contrast to LER, AYL considers the actual sown proportion of land in the field occupied by the intercrops.
where AYLm and AYLs refer to the proportional productivity loss of maize and soybean, respectively, under an intercropping pattern relative to their productivities in pure stand. Positive and negative AYL values denote the accrued advantage and disadvantage, respectively, in intercrops when the primary objective is to compare the yield on a per-plant basis (Dhima et al., 2007; Machiani et al., 2018). The AG is adopted as a competition indicator to measure how much the relative productivity increase of a given crop is higher than that of the other crop (Gitari et al., 2020; McGilchrist, 1965) in an intercropping pattern as follows:
where AGm and AGs represent the aggressivity of maize and soybean, respectively, under the intercropping patterns. If AGm or AGs = 0, both crops in the intercropping pattern are evenly competitive, if AGm is positive then the maize crop is dominant over the soybean, if AGm is negative then the maize is the dominated crop.
Economic Efficiency of Maize-Soybean Intercrop
The economic efficiency indices, e.g., total revenue (TR), net profit (NP), income equivalent ratios (IER), maize equivalent yield (MEY), monetary advantage index (MAI), relative value total (RVT), and replacement value of intercropping (RVI) were calculated to assess the economic feasibility of maize-soybean intercropping patterns integrated with mineral-NNPs fertilization. The TR was computed by considering the economic yields (e.g., grain and seed for maize and soybean, respectively) based on prevailing market prices of agricultural inputs, human labor, and returns during the 2019 and 2020 seasons in U.S. dollars (US$). All monetary values were converted to US$ according to the average local official exchange price, which amounted to 17.87 and 17.88 Egyptian pounds for each US$ 1 in the 2019 and 2020 seasons, respectively. Output prices were US$ 199.86 and 199.74 t−1 for maize grains and US$ 447.68 and 447.43 t−1 for soybean seeds in the 2019 and 2020 seasons, respectively. The TR (US$ ha−1) = monetary value of maize grains + monetary value of soybean seeds. The NP (US$ ha−1) = [(Y × P) – VPC], where Y is the economic yield (grain or seed) in t ha−1, P is the yield price in US$ t−1, and VPC is the variable production costs (e.g., seeds, fertilizers, irrigation, human labor, power, etc.) in US$ ha−1 (Abdul Rahman et al., 2021). The IER concept (Devasenapathy, 2008) is defined as the land area required under monocropping to that under intercropping to obtain similar total income under a comparable agricultural management level. IER implements a comparable concept to LER; however, in opposition to LER, which measures crop productivity, instead, IER utilizes for gross income measurement. The IER was calculated as:
where IERm and IERs represent the partial IER of maize and soybean under the intercropping patterns, respectively. Pm and Ps donate market prices of maize (grain) and soybean (seed), respectively. IER values > 1 indicate the economic profitability advantage for intercropping compared to the respective crops as monocrops. The economic yields (grain and seed) were converted into MEY (Gitari et al., 2019) according to the following equation: MEY = Ymi + [(Ysi × Ps)/Pm]. The MAI was computed (Finney 1990; Ghosh, 2004) as the following equation: MAI = [(Ymi × Pm) + (Ysi × Ps)] × [(LER – 1)/LER]. Despite the LER indicator being the most often used in agronomic research, it does not consider the economic value of the intercropped crops. The RVT described by Alabi and Esobhawan (2006) provides a solution to such a flaw. This is particularly favorable for farmers who are targeting the economic profitability of the intercropping enterprise. The RVT was calculated as: RVT = [(Ymi × Pm) + (Ysi × Ps)]/(Yms × Pm). The RVI (Moseley, 1994; Singh et al., 2015) is an indicator that accounts for the VPC of maize as the main crop in a sole stand (VPCms); hence it is a better index than the RVT and was calculated as follows: RVI = [(Ymi × Pm) + (Ysi × Ps)]/[(Yms × Pm)—VPCms
Statistical Analysis
A combined analysis was performed for the data of the two experimental seasons by Bartlett’s chi-square test after verifying the homogeneity of experimental error variance. All obtained data, except for economic analysis indices, were statistically analyzed according to the technique of ANOVA for split-plot design (Gomez & Gomez, 1984) utilizing GenStat computer software (Release 12.1, VSN International Ltd, Oxford, UK) package. The maize-soybean intercropping treatments, foliar applied-nano chitosan-loaded N levels, and their interaction set as considered fixed factors, while growing years, replications, and their interaction were considered random factors. The differences among treatment means were compared at a significant level of p ≤ 0.05 by Duncan’s multiple range as a post hoc test (Steel & Torrie, 1980). Graphs were constructed using GraphPad Prism 6.1 graphical software.
Results
Characterization of CS-NNPs Composite
The FE-SEM images (Fig. 3a, b) revealed that the CS-NNPs composite appeared in excellent 2D nanosheets. Few nanosheets are wrinkled during the freeze-drying process that is applied to get a solid sample (as required for FE-SEM imaging) from CS-NNPs composite suspension. The elemental distribution results (Fig. 3c-d) declare the uniform distribution of constituent elements within the prepared CS-NNPs as a nano-fertilizer and the successful N loading to the nano-chitosan surface. The average particle size and zeta potential (ζ) for the prepared CS-NNPs composite in this study were 45.3 ± 2 nm (Fig. 3e).
Characterization of nano-chitosan-loaded N (CS-NNPs) composite (a, b) FE-SEM images of CS-NNPs composite, c the corresponding Energy Dispersive Spectroscopy (EDS) elemental distribution (e.g., C, O, Na, P, Sm, and N indicate carbon, oxygen, sodium, phosphorus, samarium, and nitrogen, respectively), and d EDS image for N shows its uniform distribution over the nanocomposite surface. e Size distribution of the constituent CS-NNPs as estimated from zeta sizer measurements
Agronomic Traits and Maize Photosynthetic Partitioning of Maize-Soybean Intercrop
Different maize-soybean intercropping treatments (IPs) significantly (p ≤ 0.05) affected the growth, yield, and yield components of maize and soybean (Tables 2, 3, 4 and 5). Monocropping of maize (SMC) or soybean (SSC) resulted in statistically significant higher values of most growth and yield traits than the tested intercropped treatments. Ear leaf area, LAI, ear length, grain yield plant−1, and biological and grain yields of intercropped maize were, higher under the 4M:2S pattern (723.4 cm2, 4.13, 24.2 cm, 167.3 g, and 303.6 g, respectively) than 2M:4S, which recorded the lowest values of these traits. However, for soybean, the plant height, branches no. plant−1, pods no. plant−1, seed yield plant−1, 100-seed weight, yield in terms of biological, straw, and seed were, higher under the 2M:4S pattern (108.2 cm, 2.92, 104.7, 21.8 g, 16.8 g, 8.32 t ha−1, 6.19 t ha−1 and 2.13 t ha−1, respectively) than under 4M:2S pattern, which recorded the lowest values of these traits.
Growth and yield-related parameters of maize and soybean were affected significantly by N fertilization applications (Tables 2, 3, 4 and 5). The application of MN75% + 2CS-NNPs treatment significantly (p ≤ 0.05) increased plant height (by 5.7 and 1.4%), leaves no. plant−1 (by 2.8 and 1.4%), ear leaf area (by 15.1 and 5.7%), LAI (by 14.9 and 6.0%), ear length (by 11.3 and 9.5%), kernels weight ear−1 (by 16.1 and 10.6%), ears weight plant−1 (by 13.6 and 6.8%), grain yield plant−1 (by 9.4 and 8.2%), biological yield (by 20.3 and 8.7%), and grain yield (by 6.0 and 8.4%) of maize compared with MN100% and MN50% + 3CS-NNPs treatments, respectively. Also, the highest increase in soybean plant height, branches no. plant−1, pods no. plant−1, seed yield plant−1, 100-seed weight, yield in terms of biological, straw, and seed due to N fertilization was observed under application of MN75% + 2CS-NNPs treatment, with increases of 5.3–8.3%, 20.9–23.9%, 7.9–3.8%, 11.1–16.2%, 4.2–6.1%, 15.2–9.3%, 17.9–9.9%, and 17.0–12.2% compared with MN100% and MN50% + 3CS-NNPs treatments, respectively. There is a significant IP × CS-NNPs interaction effect on growth and yield-related parameters of maize and soybean. For maize crop, the application of SMC × MN75% + 2CS-NNPs interaction, followed by 4M:2S × MN75% + 2CS-NNPs interaction owned the highest plant height, LAI, biological yield, and grain yield. For soybean, the interaction application of SSC × MN75% + 2CS-NNPs, followed by 2M:4S × MN75% + 2CS-NNPs resulted in the highest branches no. plant−1, seed yield plant−1, 100-seed weight, biological yield, straw yield, and seed yield.
Results presented in Fig. 4a-c, illustrate the effects of IPs and N fertilization on RPPs for biological, vegetative, and grain of maize. The RPPs for biological and vegetative in maize were 33.9 and 36.4%, and 48.0 and 42.0% higher under 2M:4S and 3M:3S, respectively, than under SMC, which recorded the lowest values of these parameters. The highest statistical values of RPPs for grain were given by SMC, 4M:2S, and 3M:3S, while the 2M:4S exhibited the lowest value. The effect of N fertilization was only significant on the RPP for grain. In this regard, the fertilization with MN100% significantly exceeded both MN75% + 2NNPs and MN50% + 3CS-NNPs treatments by 5.2% and 8.1%, respectively. Concerning the IP × CS-NNPs interaction, 4M:2S × MN100% recorded the highest RPP for grain compared with the lowest value of 4M:2S × MN50% + 3NNPs.
Relative photosynthetic potential (RPP) for biological (a), vegetative (b), and grain (c) yields of maize as affected by intercropping pattern (IP), fertilization with mineral nitrogen (N) and nano chitosan-loaded N (CS-NNPs) composite, and their interaction (data pooled over 2019 and 2020 years). SMC sole maize crop, 4M:2S 4Maize:2 soybean, 2M:4S 2Maize:4 soybean, 3M:3S 3Maize:3 soybean, MN100% 288 kg mineral N ha−1, MN75%+2CS-NNPs 216 kg mineral N ha−1 + 2 sprays of CS-NNPs, MN50%+3CS-NNPs 144 kg mineral N ha−1 + 3 sprays of CS-NNPs. Data are mean value ± SE. Bars with the same letter are not significant at p ≤ 0.05
Biological Efficiency of Maize-Soybean Intercrop
The partial LERm significantly (p ≤ 0.05) increased (0.77) as the proportion of soybean decreased (4M:2S), whereas partial LERs increased (0.77) with decreasing maize planting ratio (2M:4S) in the evaluated IPs. The total LER value for each tested intercropping pattern was greater than the unity (Fig. 5a), indicating a yield advantage compared to pure stands. Total LER increased significantly (p ≤ 0.05) when the soybean proportion increased. For example, 2M:4S recorded a higher total LER (1.37) compared to 4M:2S (1.28) and 3M:3S (1.32). Fertilization with MN100% recorded the maximum value of total LER surpassing MN75% + 2CS-NNPs and MN50% + 3CS-NNPs treatments. The interaction application of 2M:4S × MN100%, 2M:4S × MN50% + 3NNPs, and 3M:3S × MN100% resulted in the highest total LER (1.40, 1.38, and 1.43, respectively).
Land equivalent ratio (LER; a) and income equivalent ratio (IER; b) for maize (LERm and IERm) and soybean (LERs and IERs) as affected by intercropping pattern (IP), as affected by intercropping pattern (IP), fertilization with mineral nitrogen (N) and nano chitosan-loaded N (CS-NNPs) composite, and their interaction (data pooled over 2019 and 2020 years). 4M:2S 4Maize:2 soybean, 2M:4S 2Maize:4 soybean, 3M:3S 3Maize:3 soybean, MN100% 288 kg mineral N ha−1, MN75%+2CS-NNPs 216 kg mineral N ha−1 + 2 sprays of CS-NNPs, MN50%+3CS-NNPs 144 kg mineral N ha−1 + 3 sprays of CS-NNPs. Data are mean value ± SE. Bars with the same letter are not significant at p ≤ 0.05. The dashed line represents an LER or IER when equal to 1
Intercropping maize with soybean resulted in a significant (p ≤ 0.05) effect on LEC, LUE, ATER, SPI, and PYD (Table 6). Generally, all these indices increased with increasing the ratio of soybean in the intercropping pattern. Across CS-NNPs treatments, LEC, LUE, ATER, SPI, and PYD were low (0.39, 188.8, 1.22, 13.9, and 27.8) in 4M:2S, intermediate (0.43, 195.1, 1.27, 14.4, and 31.8) in 4M:2S and high (0.46, 203.3, 1.32, 14.9, and 37.1), respectively in 2M:4S. Regarding N fertilization treatments, MN100% treatment resulted in the highest values of LEC, LUE, ATER, SPI, and PYD, followed by MN75% + 2NNPs and MN50% + 3NNPs. Notably, there was no significant difference in SPI between MN100% and MN75% + 2NNPs treatments. Without any statistically significant difference among them, the interaction applications of 2M:4S × MN100%, 2M:4S × MN75% + 2CS-NNPs, and 3M:3S × MN100% resulted in the highest LEC, LUE, ATER, and PYD. Likewise, both interactions of 2M:4S × MN75% + 2CS-NNPs and 3M:3S × MN100% recorded the highest SPI which were statistically parallel.
Interspecific Competition Indices in Maize-Soybean Intercrop
Based on pooled data across the seasons, only maize under the 2M:4S pattern had a higher RCC value (Fig. 6a). Higher (3.07 and 5.76) and lower (1.89 and 4.01) RCCm and total RCC were under 2M:4S and 4M:2S intercropping patterns, respectively. RCCs did not change with changing in IPs. MN75% + 2CS-NNPs and MN50% + 3CS-NNPs resulted in 34.5% and 40.5% for RCCs and 44.5% and 39.9% for total RCC lower compared to MN100% treatment. Regarding IPs × N fertilization interaction, MN75% + 2CS-NNPs or MN50% + 3CS-NNPs, when interacted with any of the evaluated IPs, had lower RCCm and RCCs than MN100%. AYLm was positive and greater than AYLs in 2M:4S (Fig. 6b). Maize was the dominant crop as indicated by positive values of partial AYLm that varied significantly between IPs in decreasing order: 2M:4S (0.81) < 3M:3S (0.35) < 4M:2S (0.15). A higher partial AYLs (0.44) was found when the intercrops were fertilized with MN100%. In addition, the highest partial AYLm values were recorded under 2M:4S × MN50% + 3CS-NNPs (0.97), followed by the 2M:4S × MN75% + 2CS-NNPs (0.74) and 2M:4S × MN100% (0.71). Total AYL values were positive and higher than zero in all interactions of IPs × N fertilization interaction, indicating a better yield advantage of maize-soybean intercropping compared with the monocropping pattern. AG index was affected significantly by the IPs, N fertilization treatments, and their interaction (Fig. 7). Maize under 2M:4S and 3M:3S patterns had the highest positive AGm values (0.65 and 0.06), respectively, indicating the highly competitive ability of this species under maize-soybean intercropping levels. The dominance (greater positive AGm values) of maize over soybean was more noticeable under N fertilization treatments, especially when both intercrops were fertilized with MN75% + 2CS-NNPs (0.11) or MN50% + 3CS-NNPs (0.23) higher than MN100%. About the IPs × N fertilization interaction, the 2M:4S × MN50% + 3CS-NNPs had a higher positive AGm value (0.87), followed by 2M:4S × MN50% + 3CS-NNPs (0.62) and 2M:4S × MN100% (0.46).
Relative crowding coefficient (RCC; a) and actual yield loss (AYL; b) for maize (RCCm and AYLm) and soybean (RCCs and AYLs) as affected by intercropping pattern (IP), fertilization with mineral nitrogen (N) and nano chitosan-loaded N (CS-NNPs) composite, and their interaction (data pooled over 2019 and 2020 years). 4M:2S 4Maize:2 soybean, 2M:4S 2Maize:4 soybean, 3M:3S 3Maize:3 soybean, MN100% 288 kg mineral N ha−1, MN75%+2CS-NNPs 216 kg mineral N ha−1 + 2 sprays of CS-NNPs, MN50%+3CS-NNPs 144 kg mineral N ha−1 + 3 sprays of CS-NNPs. Data are mean value ± SE. Bars with the same letter are not significant at p ≤ 0.05
Aggressivity (AG) for maize (AGm) and soybean (AGs) as affected by intercropping pattern (IP), fertilization with mineral nitrogen (N), and nano chitosan-loaded N (CS-NNPs) composite, and their interaction (data pooled over 2019 and 2020 years). 4M:2S 4Maize:2 soybean, 2M:4S 2Maize:4 soybean, 3M:3S 3Maize:3 soybean, MN100% 288 kg mineral N ha−1, MN75%+2CS-NNPs 216 kg mineral N ha−1 + 2 sprays of CS-NNPs, MN50%+3CS-NNPs 144 kg mineral N ha−1 + 3 sprays of CS-NNPs. Data are mean value ± SE. Bars with the same letter are not significant at p ≤ 0.05
Economic Efficiency of Maize-Soybean Intercrop
Generally, the total IER values for IPs, N fertilization treatments, and their interaction were greater than unity (Fig. 5b), indicating better economic profitability over pure stands. IP with a higher maize proportion (4M:2S) had significantly greater partial IERm (0.77) compared to those with a higher soybean proportion (2M:4S), which had greater partial IERs (0.77). The overall IER for the IPs indicated that 2M:4S had a significantly higher value of 1.37 compared to 1.28 and 1.32 recorded with 4M:2S and 3M:3S, respectively. The partial IERs and overall IER for the fertilization treatments indicated that MN100% had significantly higher values (0.70 and 1.38), followed by MN75% + 2CS-NNPs (0.62 and 1.29) and MN50% + 3CS-NNPs (0.60 and 1.30), respectively. The interaction application of 3M:3S × MN100%, 2M:4S × MN100%, and 2M:4S × MN50% + 3CS-NNPs had the greatest overall IER (1.43, 1.40, and 1.38, respectively), followed by 2M:4S × MN75% + 2CS-NNPs (1.32). The highest TR (2444.5, 2350.2, and 2381.9 US $ ha−1), NP (1496.4, 1347.1, and 1323.8 US $ ha−1) and MEY (12.2, 11.8, and 11.9) were recorded under 4M:2S, 2M:4S, and 3M:3S when fertilized with MN75% + 2CS-NNPs, respectively (Table 7). For MAI, the highest values (685.4, 630.1, 609.1, and 574.4) were obtained under interaction application of 3M:3S × MN100%, 2M:4S × MN50% + 3CS-NNPs, 2M:4S × MN100%, and 2M:4S × MN75% + 2CS-NNPs, respectively. The highest RVT values (1.10, 1.07, 1.07, and 1.06) were observed under the interactive application of 2M:4S × MN50% + 3CS-NNPs, 3M:3S × MN100%, 4M:2S × MN50% + 3CS-NNPs, and 4M:2S × MN75% + 2CS-NNPs, respectively. The RVI values were at the maximum level (2.28, 2.23, and 2.14) with the application of 2M:4S × MN50% + 3CS-NNPs, 4M:2S × MN50% + 3CS-NNPs, and 3M:3S × MN50% + 3CS-NNPs, respectively.
Discussion
It has been documented that legumes can rapidly cover the soil surface, reducing soil erosion and weed infestation (Berdjour et al., 2020; Mekdad et al., 2021), promoting N fixation (Yong et al., 2018), reducing insect pests and disease attacks (Chang et al., 2020), and improving land-use efficiency (Kermah et al., 2017; Shaaban et al., 2023b). Thus, the findings of this study revealed the superiority of growth and productivity of intercrops (maize/soybean) under the sole cropping over the intercropping levels. This may be due to niche complementarities induced by the spatiotemporal differences in the exploitation of the available environmental resources among intercrops (Dong et al., 2018; Kermah et al., 2017). Moreover, maize roots have been reported to penetrate soil deeper than soybean roots under optimum growth conditions (Gao et al., 2010). This probably enabled maize to access nutrients from various soil layers by lengthening its roots beneath neighboring soybean roots, therefore sparing water and mineral nutrients in the surface soil horizon for soybean uptake (Li et al., 2006). The sunlight and row spacing design have a direct influence on the component crop’s competitiveness in light interception (Yang et al., 2017). Within this context, Lv et al. (2014) reported that although the primary explanation for the advantages of maize–soybean intercrop is competition for mineral nutrients, soil water, and sunshine, their findings suggest that competition for nutrients was more important than for sunlight. Moreover, the lesser interspecific competition between intercrops for preemption of available environmental resources above (sunlight, space, etc.) soil surface may be a reason behind the higher productivity of the maize-soybean intercrop patterns tested than their corresponding pure crops when considering the entire intercropping entity. Yang et al. (2017) and Feng et al. (2020) reported that under intercropping conditions, there is improved light capture and utilization of water and soil nutrients, resulting in a better yield advantage compared with sole cropping.
Compared with pure maize stand, intercropping treatments significantly increased RPPs for biological, vegetative, and grain, which might be attributed to increasing light interception, particularly in post-silking and early grain-filling periods (Ning et al., 2013). Soybean is a highly shade-sensitive plant (Liu et al., 2015; Yang et al., 2014), so the higher soybean yield under pure stand may be due to the acquisition of more light interception and available resources than under intercropping. Moreover, soybean plants under the 4M:2S and 3M:3S patterns have more shade by maize for most of their growth duration than under the 2M:4S pattern, which resulted in a seed yield reduction, particularly for plants in intercropped border rows in agreeing with Liu et al. (2017). The shading resulting from maize under intercropping reduced the photosynthetic capacity of soybean plants, particularly in lower parts, which decreased not only the photosynthetic capacity but final seed yield (Yang et al., 2014; Zhuang & Yu-Bi, 2013). Because of the higher sensitivity to shade, soybean plants under 4M:2S or 3M:3S patterns showed vigorous shade avoidance plastic responses (i.e., small leaves, lengthier and slender internodes, and less steep leaves) to adapt to maize shading (Li et al., 2021), which resulted in lower growth and seed yield, especially for plants closer to maize in accordance with Liu et al. (2015) and Li et al. (2021). In the same context, Pierik and De Wit (2014) and Zhao et al. (2019) suggested that soybean yield, under maize-soybean intercropping conditions, is probably to be reduced due to the shading of maize as a companion crop. Under the narrow spacing, the ideotype root system architecture of maize grown under intercropping is denser than monocropping (Hui et al., 2018; Yang et al., 2016), thus a more extensive root led to more water and nutrient absorption from soil (Zhou et al., 2019). More rooting requires allocating more photo-assimilates to roots, where root growth and development need large quantities of carbohydrates from shoots as a source of root tissue components and a respiration substrate (Thaler & Pagès, 1998), and thus translocation of more carbohydrates preferentially towards roots. This is a possible explanation for the maize yield reduction under intercropping compared with monocropping. Another suggested reason for the reduction of maize yield is the height of soybean plants, which reached 108.2 cm in the 2M:4S pattern, causing shading of the lower and middle leaves of maize, reducing the photosynthesis capacity and yield (Yan et al., 2011). This is also supported by Zhou et al. (2021) who found that shading for the middle leaves of intercropped maize during the silking period led to their early senescence and lower nutrient remobilization, thus decreasing the leaf photosynthetic capacity and dry matter accumulated at maturation.
The results showed that LER values were enhanced for all tested intercropping patterns over the monocropping of maize or soybean under all N fertilization treatments. These findings concur with those reported by Lv et al. (2014), Yang et al. (2016), and Chen et al. (2019), who demonstrated that higher productivity on per-unit farmland basis and lower land-use intensity are achieved under intercropping than the corresponding monocropping and LER of maize-soybean intercropping ranges from 0.93 to 2.20. The results regarding the LER values indicate that interspecific facilitation was stronger than interspecific competition, implying that intercropping increased land-use intensity (Machiani et al., 2018). The higher partial LERm and LERs observed in 4M:2S and 2M:4S patterns, respectively, than those others suggested that the higher plant density of the intercropped maize and soybean, suppressed the other when they are grown in close association (Berdjour et al., 2020; Layek et al., 2014). In addition, one of the key explanations for the higher LER of the maize-soybean intercropping system could be due to the biological N fixation of soybean in maize-based intercropping systems (Yong et al., 2018). Li et al. (2016) reported that root exudates of maize can promote the biological N fixation from the atmosphere by faba bean under intercropping. Under intercropping, cereal plant root exudates supply a carbon source for soil microbiota to build their community structure, thereby enhancing soil N content (Coskun et al., 2017). Assessment of the intercropping treatments based on the LER findings in this study supported our results that the maize crop had higher RCC and AG values relative to soybean. The intercropping’s aggressiveness demonstrated that companion crops did not compete equally. Amanullah et al. (2020) revealed that higher AG values were obtained by cereals (pearl millet and sorghum) than in their companion’s mungbean crop. The N fixation by legumes in addition to the over-shaded occurred by maize on soybean plants under intercropping may be potential reasons for increasing the maize competitiveness indicators (AG and RCC) and maize yield (Barillot et al., 2014; Kaci et al., 2018; Mouradi et al., 2018). Moreover, about 50 to 70% of the required N for soybean can obtain from the air through N-fixing bacteria, which established functioning nodules on their roots, leading to limited competition with maize by intercropping for below-ground resources (Conley & Christmas, 2005; Mueller et al., 2015). The results of AYL showed that maize and soybean crops presented positive AYL values, indicating that both crops were to the advantage of intercropping patterns. The highest AYL values were observed by maize when intercropped with soybean in the 2M:4S pattern in accordance with the LER values. The higher-yielding of 2M:4S over those other patterns as for total LEC, LUE, ATER, SPI, and PYD, indicated a yield advantage over intercrops due to better utilization of above and below-ground growth resources under 2M:4S intercropping system (Gitari et al., 2020; Lithourgidis et al., 2011). Another reason behind higher values under the 2M:4S pattern than in other patterns may be due to more fixed N occurring by 4 rows of soybean than 2 rows of soybean in other patterns as agreed with Hinsinger et al. (2011) when intercropped maize with faba bean.
The higher values of the economic indices were a clear indication of the economic feasibility of maize when growing in close association with soybean. Among all intercropping treatments, the 2M:4S and 3M:3S were the most economic profitability and had higher productivity advantage-based LER and economic benefits than those other patterns. Similarly, Banik et al. (2006), reported a better economic advantage of intercropping due to positive MAI values. Therefore, these intercropping pattern advantages can be ascribed to the optimum utilization of environmental growth resources by the intercrop (maize and soybean) coordinates.
The low amount of available N in the experimental soil (23.5 mg N kg−1 soil) indicates the necessity of plant supply with N for maximizing yield, particularly under the intercropping system. Supplying the intercrops with enough N levels is essential for less competition severity during the co-growth period and high productivity (Nasar et al., 2021). Nitrogen deficiency stress may exacerbate the inter-and intra-specific competitions that often exist under intercropping systems. Thus, practices that help in alleviating such stress should be adopted. The results revealed that soil application of 75% MN with two foliar CS-NNPs sprays significantly improved growth, yield, and yield components of maize and soybean crops compared with MN100% as a bulk-size soil application. It is expected that lowering the N amount than normal will cause reductions in crop growth and productivity. However, despite the N supply being reduced by 25%, the application of two foliar CS-NNPs sprays kept/improved maize yield and yield components. Herein, it should be referred to the important role of chitosan, in general, and particularly nano chitosan. Despite N fertilizer applications being intended to provide the N requirements for maize crop, soybean plants benefited from the draft spray of nano chitosan-loaded nitrogen composite, thus, increasing the advantage of intercropping.
Concerning the effect of N fertilization treatments (nano-chitosan loaded N, CS-NNPs), chitosan stimulates plant growth and development by boosting water and essential minerals uptake (). Since chitosan adjusted cell osmotic potential by stimulating the enzymatic systems, water and nutrients uptake improved, hence growth and yield parameters enhanced (Guan et al., 2009a, 2009b; Makhlouf et al., 2022; Martins et al., 2018; Yang et al., 2009). Moreover, owing to its potentiality to hydroxylate amino group which offers an effective scavenger of reactive oxygen species (Sun et al., 2008), antioxidant enzymes and hydrogen peroxide (H2O2) signaling pathways were enhanced by chitosan application (Guan et al., 2009a, 2009b). Being chitosan increases the activity of catalase and glutathione peroxidase as well as the stress-protective enzymes (Hidangmayum et al., 2019; Makhlouf et al., 2022) and motivates chlorophyll formation and growth under environmental stress (Kumaraswamy et al., 2021), leaf dry matter and total yield were improved (Marzouk et al., 2022). Due to chitosan application, root growth and water absorption (Zeng & Luo, 2012), and nutrient uptake (Dzung, 2007) were enhanced, thus the yield attributes of intercropped maize soybean improved. Furthermore, the role of nitrogen itself should not be neglected, as N supply can enhance crop yield and quality as well (El-khouly et al., 2018; Saudy & Mubarak, 2014; Saudy & Mubarak, 2015; Saudy et al., 2018).
It is believed that such a beneficial effect of chitosan could be more evident when it is transformed into nano form (Abdel-Aziz et al., 2019; Ashraf et al., 2022). Since chitosan in the form of nanoparticles has a tiny size, high sorption capacity, and diffusible nature with rapid and perfect absorption/uptake by the plants were increased (Rameshaiah et al., 2015). Particles in nano form have a small size (1–100 nm) is clearly associated with the efficiency of their physicochemical features (Torabian et al., 2017). Accordingly, using nano chitosan as a carrier of nutrients could increase nutrient utilization, hence, crop growth and yields. This effect is consistent with this study findings, where CS-NNPs composite helped to gain maize/soybean yields equivalent to the recommended rate (288 kg N ha−1) by the addition of 75% of the required level of N fertilizer.
It has been reported that a safe dose of nano-fertilizers can help plants for growth and development improvements (Gao, 2006; Kale & Gawade, 2016; Zheng et al., 2005). The foliar application provides a better probability of correcting nutritional deficiencies in plants caused by an insufficient supply of nutrients to the roots, and it is usually more efficient and cost-effective (Hong et al., 2021; Saudy et al., 2022). Foliar application boosted nutrient effectiveness and was the most efficient approach for farmers to apply nutrients (Abdel-Aziz et al., 2016; Abou Tahoun et al., 2022). The bulk size fertilizers are released into the surroundings and cannot be consumed by plants, resulting in significant resource and economic waste as well as massive environmental contamination (Rai, 2016). On the other hand, nano-fertilizers exhibited excellent effects (Abou Tahoun et al., 2022; Saudy et al., 2022), because their size ranges from 1 to 100 nm, allowing them to penetrate plant leaves, which are the basic units for photosynthesis and physiological processes (Abdel-Aziz et al., 2016; Wang et al., 2013). Consequently, nano-fertilizers can decrease the nutrient amount required for raising plant productivity. Nano-fertilizers have a high surface area and supply at an active site, which boosts their rate of absorption by plants and, as a result, helps to reduce pollution (El-Ghamry et al., 2018; Rahman et al., 2021). Also, nano-fertilization provides plants with slow-release nutrients during the life cycle thereby decreasing the hazards of leaching, adsorption, surface runoff, and decomposition (Seleiman et al., 2020). The productivity and biomass yield of Glycine max has been increased following the synthesized nano-fertilizer application (Liu & Lal, 2014). The direct application of wheat plants to a specific type of nanoparticle-induced considerable improvements in growth and yield traits evaluated (Abdel-Aziz et al., 2016).
There were significant increases in ear traits of maize owing to nano-N foliar application as compared with the mineral ground application. This might be attributed to the promotion of the N absorption and utilization of nano-fertilizers, leading to an enhancement in the photosynthetic and other metabolic processes leading to an increase in cell division and elongation (Hatwar et al., 2003). Accordingly, under possible generated biotic stress (due to the impact of intercropping) and abiotic stress (due to reduced N supply than normal), using nano-chitosan loaded N is so essential for maize production-based intercropping systems.
Conclusions
Based on the gross productivity of maize (grains) and soybean (seeds), total revenue, and net profit obtained, the treatment of fertilizing maize plants with 216 kg nitrogen ha−1 plus nano chitosan-loaded nitrogen composite twice (0.48 kg nitrogen ha−1) was the most beneficial and promising practice whether under sole or intercropping patterns. Certainly, utilization of land, exploiting with a better rationalization of the available environmental resources by integrating maize-soybean intercrop and nano-chitosan-loaded nitrogen composite will enhance the farm income and sustain its potentiality. Additionally, nano-chitosan loaded nitrogen composite saves the applied nitrogen by approximately 25% of the recommended rate, hence lowering the possible environmental pollution. However, further future investigations regarding nano-carriers of other nutrients in addition to nitrogen should be adopted for an integrated nutrient enrichment program for maize grown with soybeans. Frankly, the nano compounds are still not sufficiently available in the agricultural market. Thus, further researches should be designed to find out more easy and applicable ways to manufacture the nano-nitrogen compounds to be available for maize growers.
Data Availability
Data will be made available on request.
References
Abdel-Aziz, H. M., Hasaneen, M. N., & Omer, A. M. (2016). Nano chitosan-NPK fertilizer enhances the growth and productivity of wheat plants grown in sandy soil. Span Journal of Agricultural Research, 14, e0902. https://doi.org/10.5424/sjar/2016141-8205
Abdel-Aziz, H. M. M., Hasaneen, M. N. A., & Omer, A. M. (2019). Impact of engineered nanomaterials either alone or loaded with NPK on growth and productivity of French bean plants: Seed priming vs foliar application. South+A1443 African Journal of Botany, 125, 102–108. https://doi.org/10.1016/j.sajb.2019.07.005
Abd-Elrahman, Sh. H., Saudy, H. S., & Abd El–FattahHashem, D. A. F. A. (2022). Effect of irrigation water and organic fertilizer on reducing nitrate accumulation and boosting lettuce productivity. Journal of Soil Science and Plant Nutrition, 22, 2144–2155. https://doi.org/10.1007/s42729-022-00799-8
Abdul Rahman, N., Larbi, A., Kotu, B., Asante, M. O., Akakpo, D. B., Bedi, S. M., & Hoeschle-Zeledon, I. (2021). Maize–legume strip cropping effect on productivity, income, and income risk of farmers in northern Ghana. Agronomy Journal, 113, 1574–1585. https://doi.org/10.1002/agj2.20536
Abou Tahoun, A. M., El-Enin, M. M. A., Mancy, A. G., Sheta, M. H., & Shaaban, A. (2022). Integrative soil application of humic acid and foliar plant growth stimulants improves soil properties and wheat yield and quality in nutrient-poor sandy soil of a semiarid region. Journal of Soil Science and Plant Nutrition. https://doi.org/10.1007/s42729-022-00851-7
Adetiloye, P. O., Ezedinma, F. O., & Okigbo, B. N. (1983). A land equivalent coefficient (LEC) concept for the evaluation of competitive and productive interactions in simple to complex crop mixtures. Ecological Modelling, 19, 27–39. https://doi.org/10.1016/0304-3800(83)90068-6
Afe, A. I., & Atanda, S. (2015). Percentage yield difference, an index for evaluating intercropping efficiency. American Journal of Experimental Agriculture, 5, 278–271. https://doi.org/10.9734/AJEA/2015/12405
Agegnehu, G., Ghizaw, A., & Sinebo, W. (2006). Yield performance and land-use efficiency of barley and faba bean mixed cropping in Ethiopian highlands. European Journal of Agronomy, 25, 202–207. https://doi.org/10.1016/j.eja.2006.05.002
Alabi, A., Esobhawan, O. (2006). Relative economic value of maize-okra intercrops in rainforest zone, Nigeria. Journal of Central European Agriculture 7, 433–438. https://www.researchgate.net/publication/27201836
Alghamdi, S. A., Alharby, H. F., Abdelfattah, M. A., Mohamed, I. A., Hakeem, K. R., Rady, M. M., & Shaaban, A. (2023). Spirulina platensis-inoculated humified compost boosts rhizosphere soil hydro-physico-chemical properties and Atriplex nummularia forage yield and quality in an arid saline calcareous soil. Journal of Soil Science and Plant Nutrition. https://doi.org/10.1007/s42729-023-01174-x
Ali, M., Ayyub, C. M., Hussain, Z., Hussain, R., & Rashid, S. (2020). Optimization of chitosan level to alleviate the drastic effects of heat stress in cucumber (Cucumis sativus L.). Journal of Pure and Applied Agriculture, 5, 30–38.
Amanullah, K. S., Khalil, F., & Imranuddin,. (2020). Influence of irrigation regimes on competition indexes of winter and summer intercropping system under semi-arid regions of Pakistan. Science and Reports, 10, 8129. https://doi.org/10.1038/s41598-020-65195-7
Ashraf, U., Zafar, S., Ghaffar, R., Sher, A., Mahmood, S., Noreen, Z., Maqbool, M. M., Saddiqu, S., Ashraf, A. (2022). Impact of nano chitosan-NPK fertilizer on field crops. In: Role of Chitosan and Chitosan-Based Nanomaterials in Plant Sciences, Academic Press, pp 165–183. https://doi.org/10.1016/B978-0-323-85391-0.00008-3
Banik, P. (1996). Evaluation of wheat (Triticum aestivum) and legume intercropping under 1:1 and 2:1 row-replacement series system. Journal of Agronomy and Crop Science, 176, 289–294. https://doi.org/10.1111/j.1439-037X.1996.tb00473.x
Banik, P., Midya, A., Sarkar, B. K., & Ghose, S. S. (2006). Wheat and chickpea intercropping systems in an additive series experiment advantages and weed smothering. European Journal of Agronomy, 24, 325–332. https://doi.org/10.1016/j.eja.2005.10.010
Barillot, R., Escobar-Gutiérrez, A. J., Fournier, C., Huynh, P., & Combes, D. (2014). Assessing the effects of architectural variations on light partitioning within virtual wheat–pea mixtures. Annals of Botany, 114, 725–737. https://doi.org/10.1093/aob/mcu099
Beedy, T. L., Snapp, S. S., Akinnifesi, F. K., & Sileshi, G. W. (2010). Impact of Gliricidia sepium intercropping on soil organic matter fractions in a maize-based cropping system. Agriculture, Ecosystems & Environment, 138, 139–146. https://doi.org/10.1016/j.agee.2010.04.008
Berdjour, A., Dugje, I. Y., Dzomeku, I. K., & Rahman, N. A. (2020). Maize–soybean intercropping effect on yield productivity, weed control and diversity in northern Ghana. Weed Biology and Management, 20, 69–81. https://doi.org/10.1111/wbm.12198
Bibi, A., Ibrar, M., Shalmani, A., Rehan, T., & Quratulain. (2021). A review on recent advances in chitosan applications. Pure and Applied Biology, 10, 1217–1229. https://doi.org/10.19045/bspab.2021.100128
Brooker, R. W., Bennett, A. E., Cong, W., Daniell, T. J., George, T. S., Hallett, P. D., Hawes, C., Iannetta, P. P. M., Jones, H. G., Karley, A. J., Li, L., McKenzie, B. M., Pakeman, R. J., Paterson, E., Schob, C., Shen, J., Squire, G., Watson, C. A., Zhang, C., … White, P. J. (2015). Improving intercropping: A synthesis of research in agronomy, plant physiology and ecology. New Phytologist, 206, 107–117. https://doi.org/10.1111/nph.13132
Chang, X., Yan, L., Naeem, M., Khaskheli, M. I., Zhang, H., Gong, G., Zhang, M., Song, C., Yang, W., Liu, T., & Chen, W. (2020). Maize/soybean relay strip intercropping reduces the occurrence of Fusarium root rot and changes the diversity of the pathogenic Fusarium species. Pathogens, 9, 211. https://doi.org/10.3390/pathogens9030211
Chen, H., & Yada, R. (2011). Nanotechnologies in agriculture: New tools for sustainable development. Trends in Food Science & Technology, 22, 585–594. https://doi.org/10.1016/j.tifs.2011.09.004
Chen, P., Song, C., Liu, X. M., Zhou, L., Yang, H., Zhang, X., Zhou, Y., Du, Q., Pang, T., & Fu, Z. D. (2019). Yield advantage and nitrogen fate in an additive maize-soybean relay intercropping system. Science of the Total Environment, 657, 987–999. https://doi.org/10.1016/j.scitotenv.2018.11.376
Conley, S. P., & Christmas, E. P. (2005). Utilizing inoculants in a corn-soybean rotation. Purdue Extension Purdue University. https://doi.org/10.1094/CM-2007-0104-01-RS
Corradini, E., de Moura, M. R., & Mattoso, L. H. C. (2010). A preliminary study of the incorporation of NPK fertilizer into chitosan nanoparticles. Express Polymer Letters, 4, 509–515. https://doi.org/10.3144/expresspolymlett.2010.64
Coskun, D., Britto, D. T., Shi, W., & Kronzucker, H. J. (2017). How plant root exudates shape the nitrogen cycle. Trends in Plant Science, 22, 661–673.
de Moura, M. R., Aouada, F. A., & Mattoso, L. H. C. (2008). Preparation of chitosan nanoparticles using methacrylic acid. Journal of Colloid and Interface Science, 321, 477–483. https://doi.org/10.1016/j.jcis.2008.02.006
Devasenapathy, P. (2008). Efficiency Indices for Agriculture Management Research. New India Publishing, p. 162. https://books.google.com.eg
Dhima, K. V., Lithourgidis, A. S., Vasilakoglou, I. B., & Dordas, C. A. (2007). Competition indices of common vetch and cereal intercrops in two seeding ratios. Field Crops Research, 100, 249–256. https://doi.org/10.1016/j.fcr.2006.07.008
Dong, N., Tang, M. M., Zhang, W. P., Bao, X. G., Wang, Y., Christie, P., & Li, L. (2018). Temporal differentiation of crop growth as one of the drivers of intercropping yield advantage. Science and Reports, 8, 1–11. https://doi.org/10.1038/s41598-018-21414-w
Doubi, B. S., Kouassi, K. I., Kouakou, K. L., Koffi, K. K., Baudoin, J., & Zoro, B. A. (2016). (Existing competitive indices in the intercropping system of Manihot esculenta Crantz and Lagenaria siceraria (Molina) standley. Journal of Plant Interactions, 11, 178–185. https://doi.org/10.1080/17429145.2016.1266042
Dwivedi, S., Saquib, Q., Al-Khedhairy, A. A., & Musarrat, J. (2016). Understanding the role of nanomaterials in agriculture. In D. Singh, H. Singh, & R. Prabha (Eds.), Microbial inoculants in sustainable agricultural productivity. Springer. https://doi.org/10.1007/978-81-322-2644-4_17
Dzung, N. A. (2007). Chitosan and their derivatives as prospective biosubstances for developing sustainable eco–agriculture. In: Senel, S., Varum, K. M., Sumnu, M. M., Hincal, A. A. (eds). Advances in chitin science X, pp 453–459.
El-Ghamry, A., Mosa, A. A., Alshaal, T., & El-Ramady, H. (2018). Nanofertilizers vs. biofertilizers: new insights. Environment, Biodiversity and Soil Security, 2, 51–72. https://doi.org/10.21608/jenvbs.2018.3880.1029
El-khouly, N. S., Saudy, H. S., & Abd El-Momen, W. T. (2018). Varietal variations of sesame in nitrogen utilization efficacy. Arab Universities Journal of Agricultural Sciences, 26, 1819–1826. https://doi.org/10.21608/ajs.2018.31652
Fathi, N. O. (2014). Crop yield and nitrogen bioavailability mediated by nitrogen fertilization in maize/soybean intercropping system with and without Rhizobium Inoculation. Alexandria Science Exchange Journal, 35, 115–123. https://doi.org/10.21608/ASEJAIQJSAE.2014.2586
Feng, L., Raza, M. A., Shi, J., Ansar, M., Titriku, J. K., Meraj, T. A., Shah, G. A., Ahmed, Z., Saleem, A., Liu, W., Wang, X., Yong, T., Yuan, S., Feng, Y., & Yang, W. (2020). Delayed maize leaf senescence increases the land equivalent ratio of maize soybean relay intercropping system. European Journal of Agronomy, 118, 126092. https://doi.org/10.1016/j.eja.2020.126092
Ferreira, S. A., Pereira, P., Sampaio, P., Coutinho, P. J., & Gama, F. M. (2011). Supramolecular assembled nanogel made of mannan. Journal of Colloid and Interface Science, 361, 97–108. https://doi.org/10.1016/j.jcis.2011.05.020
Flynn, R., & Idowu, J. (2015). Nitrogen fixation by legumes. GUID A-129:1–5. https://aces.nmsu.edu/pubs/a/A129.pdf
Gai, Z., Zhang, J., & Li, C. (2017). Effects of starter nitrogen fertilizer on soybean root activity, leaf photosynthesis and grain yield. PLoS ONE, 12, 1–15. https://doi.org/10.1371/journal.pone.0174841
Gao, S. (2006). Nano biological pesticide appears. In: Market information of pesticides. Supplement: 22–23. https://doi.org/10.1016/j.molstruc.2021.130517 (In Chinese)
Gao, Y., Duan, A., Qiu, X., Liu, Z., Sun, J., Zhang, J., & Wang, H. (2010). Distribution of roots and root length density in a maize/soybean strip intercropping system. Agricultural Water Management, 98, 199–212. https://doi.org/10.1016/j.agwat.2010.08.021
Gee, G. W., & Or, D. (2002). Particle-size analysis. In J. H. Dane & G. C. Topp (Eds.), Methods of soil analysis. Part 4. Physical methods (pp. 255–293). Madison: SSSA.
Ghosh, P. K. (2004). Growth, yield, competition, and economics of groundnut/cereal fodder intercropping systems in the semi-arid tropics of India. Field Crops Research, 88, 227–237. https://doi.org/10.1016/j.fcr.2004.01.015
Ghosh, P. K., Manna, M. C., Bandyopadhyay, K. K., Tripathi, A. K., Wanjari, R. H., Hati, K. M., Misra, A. K., Acharya, C. L., & Subba Rao, A. (2006). Interspecific interaction and nutrient use in soybean/sorghum intercropping system. Agronomy Journal, 98, 1097–1108. https://doi.org/10.2134/agronj2005.0328
Giroto, A. S., Guimarães, G. G., Foschini, M., & Ribeiro, C. (2017). Role of slow-release nanocomposite fertilizers on nitrogen and phosphate availability in soil. Science and Reports, 7, 1–11.
Gitari, H. I., Nyawade, S. O., Kamau, S., Gachene, C. K., Karanja, N. N., & Schulte-Geldermann, E. (2019). Increasing potato equivalent yield increases returns to investment under potato-legume intercropping systems. Open Agriculture, 4, 623–629. https://doi.org/10.1515/opag-2019-0062
Gitari, H. I., Nyawade, S. O., Kamau, S., Karanja, N. N., Gachene, C. K., Raza, M. A., Maitra, S., & Schulte-Geldermann, E. (2020). Revisiting intercropping indices with respect to potato-legume intercropping systems. Field Crops Research, 258, 107957. https://doi.org/10.1016/j.fcr.2020.107957
Gomez, K. A., Gomez, A. A. (1984). Statistical Procedures for Agricultural Research. 2nd Edition, John Wiley and Sons, Inc., London, 13–175. https://books.google.com.eg
Guan, Y. J., Hu, J., Wang, X., & Shao, C. (2009a). Seed priming with chitosan improves maize germination and seedling growth in relation to physiological changes under low temperature stress. Journal of Zhejiang University. Science B, 10, 427–433. https://doi.org/10.1631/jzus.b0820373
Guan, Y. J., Hu, J., Wang, X. J., & Shao, C. X. (2009b). Seed priming with chitosan improves maize germination and seedling growth in relation to physiological changes under low temperature stress. Journal of Zhejiang University. Science. B, 10, 427–433. https://doi.org/10.1631/jzus.B0820373
Hasaneen, M. N. A., Abdel-Aziz, H. M. M., El-Bialy, D. M. A., & Omer, A. M. (2014). Preparation of chitosan nanoparticles for loading with NPK fertilizer. African Journal of Biotechnology, 13, 3158–3164. https://doi.org/10.5897/AJB2014.13699
Hatwar, G. P., Gondane, S. M., & Urkade, S. M. (2003). Effect of micronutrients on growth and yield of chilli. Journal of Soils and Crops, 13, 123–1254. https://doi.org/10.5923/j.re.20201003.01
Hidangmayum, A., Dwivedi, P., Katiyar, D., & Hemantaranjan, A. (2019). Application of chitosan on plant responses with special reference to abiotic stress. Physiology and Molecular Biology of Plants, 25, 313–326. https://doi.org/10.1007/s12298-018-0633-1
Hiebsch, C. K., & McCollum, R. E. (1987). Area-×-time equivalency ratio: A method for evaluating the productivity of intercrops. Agronomy Journal, 79, 15–22. https://doi.org/10.2134/agronj1987.00021962007900010004x
Hinsinger, P., Betencourt, E., Bernard, L., Brauman, A., Plassard, C., Shen, J., Tang, X., & Zhang, F. (2011). P for two, sharing a scarce resource: Soil phosphorus acquisition in the rhizosphere of intercropped species. Plant Physiology, 156, 1078–1086. https://doi.org/10.1104/pp.111.175331
Hong, J., Wang, C., Wagner, D. C., Gardea-Torresdey, J. L., He, F., & Rico, C. M. (2021). Foliar application of nanoparticles: Mechanisms of absorption, transfer, and multiple impacts. Environmental Science: Nano, 8, 1196–1210. https://doi.org/10.1039/d0en01129k
Hui, F., Guo, Y., Li, B., Lv, C., Ma, Y. (2018). Quantification of differences in root system architecture under maize/soybean interspecific interactions. In 2018 6th International Symposium on Plant Growth Modeling, Simulation, Visualization and Applications (PMA) (pp. 39–42). IEEE. https://doi.org/10.1093/aob/mcr073
Kaci, G., Blavet, D., Benlahrech, S., Kouakoua, E., Couderc, P., Deleporte, P., & Ounane, S. M. (2018). The effect of intercropping on the efficiency of faba bean–rhizobial symbiosis and durum wheat soil-nitrogen acquisition in a Mediterranean agroecosystem. Plant, Soil and Environment, 64, 136–146. https://doi.org/10.17221/9/2018-PSE
Kale, A. P., & Gawade, S. N. (2016). Studies on nanoparticle induced nutrient use efficiency of fertilizer and crop productivity. Green Chemistry & Technology Letters, 2, 88–92. https://doi.org/10.18510/gctl.2016.226
Kermah, M., Franke, A. C., Adjei-Nsiah, S., Ahiabor, B. D., Abaidoo, R. C., & Giller, K. E. (2017). Maize-grain legume intercropping for enhanced resource use efficiency and crop productivity in the Guinea savanna of northern Ghana. Field Crops Research, 213, 38–50. https://doi.org/10.1016/j.fcr.2017.07.008
Khan, M. N., Mobin, M., Abbas, Z. K., AlMutairi, K. A., & Siddiqui, Z. H. (2017). Role of nanomaterials in plants under challenging environments. Plant Physiology and Biochemistry, 110, 194–209. https://doi.org/10.1016/j.plaphy.2016.05.038
Khodabin, G., Lightburn, K., Hashemi, S. M., Moghada, M. S., & Jalilian, A. (2022). Evaluation of nitrate leaching, fatty acids, physiological traits, and yield of rapeseed (Brassica napus) in response to tillage, irrigation and fertilizer management. Plant and Soil, 19, 1–8. https://doi.org/10.1007/s11104-021-05294-0
Klute, A. (1986). Water retention: laboratory methods. In: Methods of soil analysis: Part 1 Physical and mineralogical methods 5:635–62. https://doi.org/10.2136/sssabookser5.1.2ed
Kumaraswamy, R. V., Saharan, V., Kumari, S., Choudhary, R. C., Pal, A., Sharma, S. S., Rakshit, S., Raliya, R., & Biswas, P. (2021). Chitosan-silicon nanofertilizer to enhance plant growth and yield in maize (Zea mays L.). Plant Physiology and Biochemistry, 159, 53–66. https://doi.org/10.1016/j.plaphy.2020.11.054
Layek, J., Das, A., Mitran, T., Nath, C., Meena, R. S., Yadav, G. S., Shivakumar, B. G., Kumar, S., & Lal, R. (2018). Cereal+legume intercropping: An option for improving productivity and sustaining soil health. In R. S. Meena, A. Das, G. S. Yadav, & R. Lal (Eds.), Legumes for soil health and sustainable management (pp. 347–386). springer Sci Rev. https://doi.org/10.1007/978-981-13-0253-4_11
Layek, J., Shivakumar, B. G., Rana, D. S., Munda, S., Lakshman, K., Das, A., & Ramkrushna, G. I. (2014). Soybean–cereal intercropping systems as influenced by nitrogen nutrition. Agronomy Journal, 106, 1933–1946. https://doi.org/10.2134/agronj13.0521
Li, B., Li, Y., Wu, H., Zhang, F., Li, C., Li, X., Lambers, H., & Li, L. (2016). Root exudates drive interspecific facilitation by enhancing nodulation and N2 fixation. Proceedings of the National Academy of Sciences of the United States of America, 113, 6496–6501. https://doi.org/10.1073/pnas.1523580113
Li, L., Sun, J., Zhang, F., Guo, T., Bao, X., Smith, F. A., & Smith, S. E. (2006). Root distribution and interactions between intercropped species. Oecologia, 147, 280–290. https://doi.org/10.1073/pnas.1523580113
Li, S., Van Der, W., Zhu, J., Guo, Y., Li, B., Ma, Y., & Evers, J. B. (2021). Estimating the contribution of plant traits to light partitioning in simultaneous maize/soybean intercropping. Journal of Experimental Botany, 72, 3630–3646. https://doi.org/10.1093/jxb/erab077
Lithourgidis, A. S., Vlachostergios, D. N., Dordas, C. A., & Damalas, C. A. (2011). Dry matter yield, nitrogen content, and competition in pea–cereal intercropping systems. European Journal of Agronomy, 34, 287–294. https://doi.org/10.1016/j.eja.2011.02.007
Liu, R., & Lal, R. (2014). Synthetic apatite Nanoparticles as a phosphorus fertilizer for soybean (Glycine max). Scientific Reports, 4, 6. https://doi.org/10.1038/srep05686
Liu, W., Zou, J., Zhang, J., Yang, F., Wan, Y., & Yang, W. (2015). Evaluation of soybean (Glycine max) stem vining in maize-soybean relay strip intercropping system. Plant Production Science, 18, 69–75. https://doi.org/10.1626/pps.18.69
Liu, X., Rahman, T., Song, C., Su, B., Yang, F., Yong, T., Wu, Y., Zhang, C., & Yang, W. (2017). Changes in light environment, morphology, growth and yield of soybean in maize–soybean intercropping systems. Field Crops Research, 200, 38–46. https://doi.org/10.1016/j.fcr.2016.10.003
Lv, Y., Francis, C., Wu, P., Chen, X., & Zhao, X. (2014). Mize–soybean intercropping interactions above and below ground. Crop Science, 54, 914–922. https://doi.org/10.2135/cropsci2013.06.0403
Machiani, M. A., Javanmard, A., Morshedloo, M. R., & Maggi, F. (2018). Evaluation of competition, essential oil quality and quantity of peppermint intercropped with soybean. Industrial Crops and Products, 111, 743–754. https://doi.org/10.1016/j.indcrop.2017.11.052
Makhlouf, B. S. I., Khalil, S. R. A. E., & Saudy, H. S. (2022). Efficacy of humic acids and chitosan for enhancing yield and sugar quality of sugar beet under moderate and severe drought. Journal of Soil Science and Plant Nutrition. https://doi.org/10.1007/s42729-022-00762-7
Martins, M., Carvalho, M., Carvalho, D. T., Barbosa, S., Doriguetto, A. C., Magalhaes, P. C., & Ribeiro, C. (2018). Physicochemical characterization of chitosan and its effects on early growth, cell cycle and root anatomy of transgenic and non-transgenic maize hybrids. Australian Journal of Crop Science, 12, 56–66. https://doi.org/10.21475/ajcs.18.12.01.pne649
Marzouk, N. M., Abd-Alrahman, H. A., & El-Sawy, S. M. M. (2022). Amino acids sources and chitosan enhance cauliflower yield and quality under heat stress. Asian Jounal of Planetary Sciences, 21, 9–23. https://doi.org/10.3923/ajps.2022.9.23
McGilchrist, C. A. (1965). Analysis of competition experiments. Biomet, 21, 975–985. https://doi.org/10.2307/2528258
McGraw, R. L. (1977). Yield dynamics of Florunner peanuts. Master of Science Thesis. Univ of Fla. https://doi.org/10.3146/pnut.12.2.0005
Mead, R., & Willey, R. (1980). The concept of a ‘land equivalent ratio and advantages in yields from intercropping. Experimental Agriculture, 16, 217–228. https://doi.org/10.1017/S0014479700010978
Meier, U. (2001). Growth stages of mono- and dicotyledonous plants. BBCH Monograph. In: Meier, U. (Ed.), Federal Biological Research Centre for Agriculture and Forestry, 2nd edition. https://www.researchgate.net/profile/Carmen-Feller/publication/281574833
Mekdad, A. A., El-Sherif, A., Rady, M. M., & Shaaban, A. (2022). Culture management and application of humic acid in favor of Helianthus annuus L. oil yield and nutritional homeostasis in a dry environment. Journal of Soil Science and Plant Nutrition, 22, 71–86. https://doi.org/10.1007/s42729-021-00636-4
Mekdad, A. A., El-Enin, M. M. A., Rady, M. M., Hassan, F. A., Ali, E. F., & Shaaban, A. (2021). Impact of level of nitrogen fertilization and critical period for weed control in peanut (Arachis hypogaea L.). Agronomy, 11, 909. https://doi.org/10.3390/agronomy11050909
Millán, G., Agosto, F., Vázquez, M., Botto, L., Lombardi, L., & Juan, L. (2008). Use of clinoptilolite as a carrier for nitrogen fertilizers in soils of the Pampean regions of Argentina. Ciencia e Investigación Agraria, 35, 293–302.
Moseley, W. G. (1994). An equation for the replacement value of agroforestry. Agroforestry Systems, 26, 47–52. https://doi.org/10.1007/BF00705151
Mouradi, M., Farissi, M., Makoudi, B., Bouizgaren, A., & Ghoulam, C. (2018). Effect of faba bean (Vicia faba L.)–rhizobia symbiosis on barley’s growth, phosphorus uptake and acid phosphatase activity in the intercropping system. Annals of Agrarian Science, 3, 1–18. https://doi.org/10.1016/j.aasci.2018.05.003
Mousavi, S. R., & Eskandari, H. (2011). A general overview on intercropping and its advantages in sustainable agriculture. Journal of Applied Environmental and Biological Sciences, 1, 4482–482. https://doi.org/10.3389/fsufs.2021.634361
Mueller, N., Elmore, R., & Shapiro, C. (2015). Soybean inoculation: When, where, and why. Crop Watch No. https://doi.org/10.1017/wsc.2019.8
Nasar, J., Khan, W., Khan, M. Z., Gitari, H. I., Gbolayori, J. F., Moussa, A. A., Mandozai, A., Rizwan, N., Anwari, G., & Maroof, S. M. (2021). Photosynthetic activities and photosynthetic nitrogen use efficiency of maize crop under different planting patterns and nitrogen fertilization. Journal of Soil Science and Plant Nutrition, 21, 2274–2284. https://doi.org/10.1007/s42729-021-00520-1
Ning, P., Li, S., Yu, P., Zhang, Y., & Li, C. (2013). Post-silking accumulation and partitioning of dry matter, nitrogen, phosphorus, and potassium in maize varieties differing in leaf longevity. Field Crops Research, 144, 19–27. https://doi.org/10.1016/j.fcr.2013.01.020
Noureldin, N. A., Saudy, H. S., Ashmawy, F., & Saed, H. M. (2013). Grain yield response index of bread wheat cultivars as influenced by nitrogen levels. Annals of Agricultural Sciences, 58, 147–152. https://doi.org/10.1016/j.aoas.2013.07.012
Odo, P. E. (1991). Evaluation of short and tall sorghum varieties in mixtures with cowpea in the Sudan savanna of Nigeria: Land equivalent ratio, grain yield and system productivity index. Experimental Agriculture, 27, 435–441. https://doi.org/10.9734/jeai/2019/v37i330269
Page, A. L., Keeney, D. R., Baker, D. E., Miller, R. H., Ellis, R., Rhoades, J. D. (1982). Methods of Soil Analysis, Part 2, Chemical and microbiological properties, 2nd Eds. No. 9, ASA-SSSA, Madison, Wisconsin, USA. https://doi.org/10.2136/sssabookser5.1.2ed
Panpatte, D. G., Jhala, Y. K., Shelat, H. N., & Vyas, R. V. (2016). Nanoparticles: The next generation technology for sustainable agriculture. In D. Singh, H. Singh, & R. Prabha (Eds.), Microbial inoculants in sustainable agricultural productivity. Springer. https://doi.org/10.1007/978-81-322-2644-4_18
Pierik, R., & de Wit, M. (2014). Shade avoidance: Phytochrome signalling and other aboveground neighbour detection cues. Journal of Experimental Botany, 65, 2815–2824. https://doi.org/10.1093/jxb/ert389
Rahman, M. H., Haque, K. S., & Khan, M. Z. H. (2021). A review on application of controlled released fertilizers influencing the sustainable agricultural production: A Cleaner production process. Environmental Technology & Innovation, 23, 101697. https://doi.org/10.1016/j.eti.2021.101697
Rai, P. K. (2016). Impacts of particulate matter pollution on plants: Implications for environmental biomonitoring. Ecotoxicology and Environmental Safety, 129, 120–136. https://doi.org/10.1016/j.ecoenv.2016.03.012
Rameshaiah, G. N., Pallavi, J., Shabnam, S. (2015). Nano fertilizers and nano sensors an attempt for developing smart agriculture. International Journal of Engineering Research and General Science 3, 314‒320. http://www.ijergs.org/
Sani, Y. G., Jamshidi, K., & Moghadam, M. R. (2014). Evaluation of Quality and Quantity of Corn and Soybean Grain Yield in Intercropping under Deficit Irrigation. Journal of Biology, Agriculture and Healthcare, 4, 133–139. https://doi.org/10.7176/JBAH
Saudy, H.S., El-Metwally, I. M. (2009). Weed management under different patterns of sunflower–soybean intercropping. Journal of Central European Agriculture 10, 41–52. https://hrcak.srce.hr/41577
Saudy, H. S., El-Bagoury, K. F. (2014). Quixotic coupling between irrigation system and maize-cowpea intercropping for weed suppression and water preserving. African Crop Science Journal 22, 97–108. https://www.ajol.info/index.php/acsj/article/view/104937
Saudy, H. S. (2014). Chlorophyll meter as a tool for forecasting wheat nitrogen requirements after application of herbicides. Archives of Agronomy and Soil Science, 60, 1077–1090. https://doi.org/10.1080/03650340.2013.866226
Saudy, H. S. (2015). Maize–cowpea intercropping as an ecological approach for nitrogen–use rationalization and weed suppression. Archives of Agronomy and Soil Science, 61, 1–14. https://doi.org/10.1080/03650340.2014.920499
Saudy, H. S., & Abd El-MomenEl-khouly, W. R. N. S. (2018). Diversified nitrogen rates influence nitrogen agronomic efficiency and seed yield response index of sesame (Sesamum indicum, L.) cultivars. Communications in Soil Science and Plant Analysis, 49, 2387–2395. https://doi.org/10.1080/00103624.2018.1510949
Saudy, H. S., & El-Metwally, I. M. (2019). Nutrient utilization indices of NPK and drought management in groundnut under sandy soil conditions. Communications in Soil Science and Plant Analysis, 50, 1821–1828. https://doi.org/10.1080/00103624.2019.1635147
Saudy, H. S., & El-Metwally, I. M. (2023). Effect of irrigation, nitrogen sources and metribuzin on performance of maize and its weeds. Communications in Soil Science and Plant Analysis, 54, 22–31. https://doi.org/10.1080/00103624.2022.2109659
Saudy, H. S., El-Samad, G. A. A., El-Temsah, M. E., & El-Gabry, Y. A. E. G. (2022). Effect of iron, zinc, and manganese nano-form mixture on the micronutrient recovery efficiency and seed yield response index of sesame genotypes. Journal of Soil Science and Plant Nutrition, 22, 732–742. https://doi.org/10.1007/s42729-021-00681-z
Saudy, H. S., Hamed, M. F., & Abd El–MomenHussein, W. R. H. (2020). Nitrogen use rationalization and boosting wheat productivity by applying packages of humic, amino acids and microorganisms. Communications in Soil Science and Plant Analysis, 51, 1036–1047. https://doi.org/10.1080/00103624.2020.1744631
Saudy, H. S., & Mubarak, M. (2014). Does silicon alleviate the injuries of nitrogen deficiency and fenoxaprop-p-ethyl herbicide in wheat (Triticum aestivum, L)? Arab Universities Journal of Agricultural Sciences, 22, 347–360. https://doi.org/10.21608/ajs.2014.14740
Saudy, H. S., & Mubarak, M. (2015). Mitigating the detrimental impacts of nitrogen deficit and fenoxaprop-p-ethyl herbicide on wheat using silicon. Communications in Soil Science and Plant Analysis, 46, 913–923. https://doi.org/10.1080/00103624.2015.1011753
Seleiman, M. F., Almutairi, K. F., Alotaibi, M., Shami, A., Alhammad, B. A., & Battaglia, M. L. (2020). Nano-fertilization as an emerging fertilization technique: Why can modern agriculture benefit from its use? Plants, 10, 2. https://doi.org/10.3390/plants10010002
Shaaban, A., El-Mageed, T. A. A., El-Momen, W. R. A., Saudy, H. S., & Al-Elwany, O. A. (2023a). The integrated application of phosphorous and zinc affects the physiological status, yield and quality of canola grown in phosphorus-suffered deficiency saline soil. Gesunde Pflanzen. https://doi.org/10.1007/s10343-023-00843-2
Shaaban, A., Mahfouz, H., Megawer, E. A., & Saudy, H. S. (2023b) Physiological changes and nutritional value of forage clitoria grown in arid agro-ecosystem as influenced by plant density and water deficit. Journal of Soil Science and Plant Nutrition. https://doi.org/10.1007/s42729-023-01294-4
Sheha, A. M., Abou El-Enin, M. M., El-Hashash, E. F., Rady, M. M., El-Serafy, R. S., & Shaaban, A. (2023). The productivity and overall benefits of faba bean-sugar beet intercropping systems interacted with foliar-applied nutrients. Journal of Plant Nutrition, 46, 1683–1700. https://doi.org/10.1080/01904167.2022.2093747
Singh, R. J., Ahlawat, I. P. S., & Sharma, N. K. (2015). Resource use efficiency of transgenic cotton and peanut intercropping system using modified fertilization technique. International Journal of Plant Production, 9, 523–540. https://doi.org/10.1016/S2095-3119(20)63318-4
Snyder, F. W., & Carlson, G. E. (1984). Selecting for partitioning of photosynthetic products in crops. Agron, 37, 47–72. https://doi.org/10.1111/j.1469-8137.2008.02705.x
Soil Survey Staff. (1999). Soil taxonomy: A basic system of soil classification for making and interpreting soil surveys (2nd ed., p. 864). United States Department of Agriculture.
Sonnewald U (2013) Physiology of development. Strasburger’s Plant Sciences, Springer, Berlin, Heidelberg, Germany, p. 411–530. https://springerlink.fh-diploma.de/content/pdf/10.1007%252F978-3-642-15518-5_6.
Sorlier, P., Denuzière, A., Viton, C., & Domard, A. (2001). Relation between the degree of acetylation and the electrostatic properties of chitin and chitosan. Biomacromole, 2, 765–772.
Steel, R. G., & Torrie, H. H. (1980). Principles and procedures of statistics: A biometrical approach (2nd ed.). McGraw Hill Inc.
Suguna, S., Shankar, S., Jaganathan, S. K., & Manikandan, A. (2017). Novel synthesis of spinel MnxCo1−xAl2O4 (x= 00 to 10) nanocatalysts: Effect of Mn2+ doping on structural, morphological, and opto–magnetic properties. Journal of Superconductivity and Novel Magnetism, 30, 691–699. https://doi.org/10.1007/s10948-016-3866-7
Sun, T., Yao, Q., Zhou, D., & Mao, F. (2008). Antioxidant activity of N-carboxymethyl chitosan oligosaccharides. Bioorganic & Medicinal Chemistry Letters, 18, 5774–5776. https://doi.org/10.1016/j.bmcl.2008.09.072
Thaler, P., & Pagès, L. (1998). Modelling the influence of assimilate availability on root growth and architecture. Plant and Soil, 201, 307–320. https://doi.org/10.1023/A:1004380021699
Thierfelder, C., Chivenge, P., Mupangwa, W., Rosenstock, T. S., Lamanna, C., & Eyre, J. X. (2017). How climate-smart is conservation agriculture (CA)? its potential to deliver on adaptation, mitigation, and productivity on smallholder farms in southern Africa. Food Security, 9, 537–560. https://doi.org/10.1007/s12571-017-0665-3
Torabian, S., Zahedi, M., & Khoshgoftar, A. H. (2017). Effects of foliar spray of nano–particles of FeSO4 on the growth and ion content of sunflower under saline condition. Journal of Plant Nutrition, 40, 615–623. https://doi.org/10.1080/01904167.2016.1240187
Wang, W. N., Tarafdar, J. C., & Biswas, P. (2013). Nanoparticle synthesis and delivery by an aerosol route for watermelon plant foliar uptake. Journal of Nanoparticle Research, 15, 1417. https://doi.org/10.1007/s11051-013-1417-8
Wang, Z. H., Miao, Y. F., & Li, S. X. (2015). Effect of ammonium and nitrate nitrogen fertilizers on wheat yield in relation to accumulated nitrate at different depths of soil in drylands of China. Field Crops Research, 183, 211–224. https://doi.org/10.1016/j.fcr.2015.07.019
Yan, H., Shang, A., Peng, Y., Yu, P., & Li, C. (2011). Covering middle leaves and ears reveals differential regulatory roles of vegetative and reproductive organs in root growth and nitrogen uptake in maize. Crop Science, 51, 265–272. https://doi.org/10.2135/cropsci2010.03.0180
Yang, F., Hu, J., Li, J., Wu, X., & Qian, Y. (2009). Chitosan enhances leaf membrane stability and antioxidant enzyme activities in apple seedlings under drought stress. Plant Growth Regulation, 58, 131–136. https://doi.org/10.1007/s10725-009-9361-4
Yang, F., Huang, S., Gao, R., Liu, W., Yong, T., Wang, X., Wu, X., & Yang, W. (2014). Growth of soybean seedlings in relay strip intercropping systems in relation to light quantity and red: Far-red ratio. Field Crops Research, 155, 245–253. https://doi.org/10.1016/j.fcr.2013.08.011
Yang, F., Liao, D., Fan, Y., Gao, R., Wu, X., Rahman, T., Yong, T., Liu, W., Liu, J., Du, J., Shu, K., Wang, X., & Yang, W. (2017). Effect of narrow-row planting patterns on crop competitive and economic advantage in maize soybean relay strip intercropping system. Plant Production Science, 20, 1–11. https://doi.org/10.1080/1343943X.2016.1224553
Yang, H., Xu, M., Koide, R. T., Liu, Q., Dai, Y., Liu, L., & Bian, X. (2016). Effects of ditch-buried straw return on water percolation, nitrogen leaching and crop yields in a rice–wheat rotation system. Journal of the Science of Food and Agriculture, 96, 1141–1149. https://doi.org/10.1002/jsfa.7196
Yaseen, M., Singh, M., & Ram, D. (2014). Growth, yield, and economics of vetiver (Vetiveria zizanioides L. Nash) under intercropping system. Industrial Crops and Products, 61, 417–21. https://doi.org/10.1016/j.indcrop.2014.07.033
Yong, T., Chen, P., Dong, Q., Du, Q., Yang, F., Wang, X., Liu, W., & Yang, W. (2018). Optimized nitrogen application methods to improve nitrogen use efficiency and nodule nitrogen fixation in a maize-soybean relay intercropping system. Journal of Integrative Agriculture, 17, 60345–60347. https://doi.org/10.1016/S2095-3119(17)61836-7
Zeng, D., & Luo, X. (2012). Physiological effects of chitosan coating on wheat growth and activities of protective enzyme with drought tolerance. Open Journal of Soil Sciences, 2, 282–288. https://doi.org/10.4236/ojss.2012.23034
Zhao, Y., Zhan, H., Dai, X., Shan, D., & Wang, T. (2019). Comprehensive evaluation and screening identification index of shade tolerance of intercropping soybean. Chinese Journal of Oil Crop Sciences, 41, 81–91. https://doi.org/10.7505/j.issn.1007-9084.2019.01.011
Zheng, L., Hong, F., Lu, S., & Liu, C. (2005). Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biological Trace Element Research, 104, 83–91. https://doi.org/10.1385/BTER:104:1:083
Zhou, T., Wang, L., Sun, X., Wang, X., Pu, T., Yang, H., Rengel, Z., Liu, W., & Yang, W. (2021). Improved post-silking light interception increases yield and P-use efficiency of maize in maize/soybean relay strip intercropping. Field Crops Research, 262, 108054. https://doi.org/10.1016/j.fcr.2020.108054
Zhou, T., Wang, L., Yang, H., Gao, Y., Liu, W., & Yang, W. (2019). Ameliorated light conditions increase the P uptake capability of soybean in a relay-strip intercropping system by altering root morphology and physiology in the areas with low solar radiation. Science of the Total Environment, 688, 1069–1080. https://doi.org/10.1016/j.scitotenv.2019.06.344
Zhuang, L., & Yu-Bi, H. U. (2013). Function analysis of phytochrome b gene in maize. Pakistan Journal of Botany, 45, 1215–1220. https://doi.org/10.1104/pp.19.00239
Zulfiqar, F., Navarro, M., Ashraf, M., Akram, N. A., & Munné-Bosch, S. (2019). Nano fertilizers use for sustainable agriculture: Advantages and limitations. Plant Science, 289, 1–11. https://doi.org/10.1016/j.plantsci.2019.110270
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Conceptualization: MMAElE, OAMA, AMS, and AS; investigation, methodology, and data curation: MMAEl-E and AMS; preparing original draft: MMAE-E, AS, HSS, and RS E-S; review and final editing: AS and HSS. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abou El-Enin, M.M., Sheha, A.M., El-Serafy, R.S. et al. Foliage-Sprayed Nano-Chitosan-Loaded Nitrogen Boosts Yield Potentials, Competitive Ability, and Profitability of Intercropped Maize-Soybean. Int. J. Plant Prod. 17, 517–542 (2023). https://doi.org/10.1007/s42106-023-00253-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42106-023-00253-4