Abstract
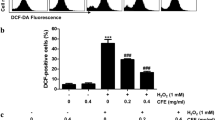
The fruit of the Prunus mume (Siebold) Siebold & Zucc, Rosaceae (Korean name: Maesil) has long been used as a health food or valuable medicinal material in traditional herb medicine in Southeast Asian countries. In this study, we determined the potential therapeutic efficacy of the ethanol extract of P. mume fruits (EEPM) against H2O2-induced oxidative stress and apoptosis in the murine skeletal muscle myoblast cell line C2C12, and sought to understand the associated molecular mechanisms. The results indicated that exposure of C2C12 cells to H2O2 caused a reduction in cell viability by increasing the generation of intracellular reactive oxygen species and by disrupting mitochondrial membrane permeability, leading to DNA damage and apoptosis. However, pretreatment of the cells with EEPM before H2O2 exposure effectively attenuated these changes, suggesting that EEPM prevented H2O2-induced mitochondria-dependent apoptosis. Furthermore, the increased ex-pression and phosphorylation of nuclear factor erythroid 2-related factor 2 (Nrf2) and up-regulation of heme oxygenase-1 (HO-1), a phase II antioxidant enzyme, were detected in EEPM-treated C2C12 cells. We also found that zinc protoporphyrin IX, an HO-1 inhibitor, attenuated the protective effects of EEPM against H2O2-induced reactive oxygen species accumulation and cytotoxicity. Therefore, these results indicate that the activation of the Nrf2/HO-l pathway might be involved in the protection of EEPM against H2O2-induced cellular oxidative damage. In conclusion, these results show that EEPM contributes to the prevention of oxidative damage and could be used as a nutritional agent for oxidative stress-related diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ayala-Pena, S., 2013. Role of oxidative DNA damage in mitochondrial dysfunction and Huntington’s disease pathogenesis. Free Radic. Biol. Med. 62, 102–110.
Chen, Y.H., Yet, S.F., Perrella, M.A., 2003. Role of heme oxygenase-1 in the regulation of blood pressure and cardiac function. Exp. Biol. Med. (Maywood) 228, 447–453.
Chistiakov, D.A., Sobenin, I.A., Revin, V.V., Orekhov, A.N., Bobryshev, Y.V., 2014. Mitochondrial aging and age-related dysfunction of mitochondria. Biomed. Res. Int., https://doi.org/10.1155/2014/238463.
Deavall, D.G., Martin, E.A., Horner, J.M., Roberts, R., 2012. Drug-induced oxidative stress and toxicity. J. Toxicol. 2012, 645460.
Enomoto, S., Yanaoka, K., Utsunomiya, H., Niwa, T., Inada, K., Deguchi, H., Ueda, K., Mukoubayashi, C., Inoue, I., Maekita, T., Nakazawa, K., Iguchi, M., Arii, K., Tamai, H., Yoshimura, N., Fujishiro, M., Oka, M., Ichinose, M., 2010. Inhibitory effects of Japanese apricot (Prunus mume Siebold et Zucc; Ume) on Helicobacter pylori-related chronic gastritis. Eur. J. Clin. Nutr. 64, 714–719.
Fleury, C., Mignotte, B., Vayssiére, J.L., 2002. Mitochondrial reactive oxygen species in cell death signaling. Biochimie 84, 131–141.
Guerra-Araiza, C., Álvarez-Mejía, A.L., Sńchez-Torres, S., Farfan-Garcia, E., Mondragón-Lozano, R., Pinto-Almazán, R., Salgado-Ceballos, H., 2013. Effect of natural exogenous antioxidants on aging and on neurodegenerative diseases. Free Radic. Res. 47, 451–462.
Hybertson, B.M., Gao, B., Bose, S.K., McCord, J.M., 2011. Oxidative stress in health and disease: the therapeutic potential of Nrf2 activation. Mol. Aspects Med. 32, 234–246.
Jeong, J.T., Moon, J.H., Park, K.H., Shin, C.S., 2006. Isolation and characterization of a new compound from Prunus mume fruit that inhibits cancer cells. J. Agric. Food Chem. 54, 2123–2128.
Jung, B.C., Ko, J.H., Cho, S.J., Koh, H.B., Yoon, S.R., Han, D.U., Lee, B.J., 2010. Immune-enhancing effect of fermented Maesil (Prunus mume Siebold & Zucc.) with probiotics against Bordetella bronchiseptica in mice. J. Vet. Med. Sci. 72, 1195–1202.
Kim, B.H., Oh, I., Kim, J.H., Jeon, J.E., Jeon, B., Shin, J., Kim, T.Y., 2014. Anti-inflammatory activity of compounds isolated from Astragalus sinicus L. in cytokine-induced keratinocytes and skin. Exp. Mol. Med. 21, e87.
Kim, R., Emi, M., Tanabe, K., Murakami, S., Uchida, Y., Arihiro, K., 2006. Regulation and interplay of apoptotic and non-apoptotic cell death. J. Pathol. 208, 319–326.
Kita, M., Kato, M., Ban, Y., Honda, C., Yaegaki, H., Ikoma, Y., Moriguchi, T., 2007. Carotenoid accumulation in Japanese apricot (Prunus mume Siebold & Zucc): molecular analysis of carotenogenic gene expression and ethylene regulation. J. Agric. Food Chem. 55, 3414–3420.
Lee, H.S., Lee, G.S., Kim, S.H., Kim, H.K., Suk, D.H., Lee, D.S., 2014. Anti-oxidizing effect of the dichloromethane and hexane fractions from Orostachys japonicus in LPS-stimulated RAW 264.7 cells via upregulation of Nrf2 expression and activation of MAPK signaling pathway. BMB Rep. 47, 98–103.
Lee, J.A., Ko, J.H., Jung, B.C., Kim, T.H., Hong, J.I., Park, Y.S., Lee, B.J., 2013. Fermented Prunus mume with probiotics inhibits 7, 12-dimethylbenz[a]anthracene and 12-o-tetradecanoyl phorbol-13-acetate induced skin carcinogenesis through alleviation of oxidative stress. Asian Pac. J. Cancer Prev. 14, 2973–2978.
Martin, D., Rojo, A.I., Salinas, M., Diaz, R., Gallardo, G., Alam, J., de Galarreta, C.M., Cuadrado, A., 2004. Regulation of heme oxygenase-1 expression through the phosphatidylinositol 3-kinase/Akt pathway and the Nrf2 transcription factor in response to the antioxidant phytochemical carnosol. J. Biol. Chem. 279, 8919–8929.
Matsuda, H., Morikawa, T., Ishiwada, T., Managi, H., Kagawa, M., Higashi, Y., Yoshikawa, M., 2003. Medicinal flowers. VIII. Radical scavenging constituents from the flowers of Prunus mume: structure of prunose III. Chem. Pharm. Bull. (Tokyo) 51, 440–443.
Maynard, S., Schurman, S.H., Harboe, C., de Souza-Pinto, N.C., Bohr, V.A., 2009. Base excision repair of oxidative DNA damage and association with cancer and aging. Carcinogenesis 30, 2–10.
Mitani, T., Horinishi, A., Kishida, K., Kawabata, T., Yano, F., Mimura, H., Inaba, N., Yamanishi, H., Oe, T., Negoro, K., Mori, H., Miyake, Y., Hosoda, A., Tanaka, Y., Mori, M., Ozaki, Y., 2013. Phenolics profile of mume, Japanese apricot (Prunus mume Sieb, et Zucc.) fruit. Biosci. Biotechnol. Biochem. 77, 1623–1627.
Niture, S.K., Khatri, R., Jaiswal, A.K., 2014. Regulation of Nrf2-an update. Free Radic. Biol. Med. 66, 36–44.
Park, C., Jin, C.Y., Kim, G.Y., Jeong, Y.K, Kim, W.J., Choi, Y.H., 2011. Induction of apoptosis by ethanol extract of Prunus mume in U937 human leukemia cells through activation of caspases. Oncol. Rep. 26, 987–993.
Rogakou, E.P., Pilch, D.R., Orr, A.H., Ivanova, V.S., Bonner, W.M., 1998. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 273, 5858–5868.
Sang, S., Lapsley, K., Jeong, W.S., Lachance, P.A., Ho, CT., Rosen, R.T., 2002. Antioxidative phenolic compounds isolated from almond skins (Prunus amygdalus Batsch). J. Agric. Food Chem. 50, 2459–2463.
Seo, K., Ki, S.H., Shin, S.M., 2014. Methylglyoxal induces mitochondrial dysfunction and cell death in liver. Toxicol. Res. 30, 193–198.
Son, T.G., Kawamoto, E.M., Yu, Q.S., Greig, N.H., Mattson, M.P., Camandola, S., 2013. Naphthazarin protects against glutamate-induced neuronal death via activation of the Nrf2/ARE pathway. Biochem. Biophys. Res. Commun. 433, 602–606.
Surh, Y.J., Kundu, J.K., Na, H.K., 2008. Nrf2 as a master redox switch in turning on the cellular signaling involved in the induction of cytoprotective genes by some chemopreventive phytochemicals. Planta Med. 74, 1526–1539.
Tada, K., Kawahara, K., Matsushita, S., Hashiguchi, T., Maruyama, I., Kanekura, T., 2012. MK615, a Prunus mume Steb. Et Zucc (‘Urne’) extract, attenuates the growth of A375 melanoma cells by inhibiting the ERKl/2-Id-l pathway. Phytother. Res. 26, 833–838.
Terazawa, R., Akimoto, N., Kato, T., Itoh, T., Fujita, Y., Hamada, N., Deguchi, T., Iinuma, M., Noda, M., Nozawa, Y., Ito, M., 2013. A kavalactone derivative inhibits lipopolysaccharide-stimulated iNOS induction and NO production through activation of Nrf2 signaling in BV2 microglial cells. Pharmacol. Res. 71, 34–43.
Tsuji, R., Koizumi, H., Fujiwara, D., 2011. Effects of a plum (Prunus mume Siebold and Zucc.) ethanol extract on the immune system in vivo and in vitro. Biosci. Biotechnol. Biochem. 75, 2011–2013.
Wang, C.H., Wu, S.B., Wu, Y.T., Wei, Y.H., 2013. Oxidative stress response elicited by mitochondrial dysfunction: implication in the pathophysiology of aging. Exp. Biol. Med. (Maywood) 238, 450–460.
Wen, J., Shi, W., 2012. Revisionofthe Maddenia clade of Prunus (Rosaceae). PhytoKeys 2012, 39–59.
Wojcik, M., Burzynska-Pedziwiatr, I., Wozniak, L.A., 2010. A review of natural and synthetic antioxidants important for health and longevity. Curr. Med. Chem. 17, 3262–3288.
Wu, Y.T., Wu, S.B., Lee, W.Y., Wei, Y.H., 2010. Mitochondrial respiratory dysfunction-elicited oxidative stress and posttranslational protein modification in mitochondrial diseases. Ann. N. Y. Acad. Sci. 1201, 147–156.
Yan, X.T., Lee, S.H., Li, W., Sun, Y.N., Yang, S.Y., Jang, H.D., Kim, Y.H., 2014. Evaluation of the antioxidant and anti-osteoporosis activities of chemical constituents of the fruits of Prunus mume. Food Chem. 156, 408–415.
Yingsakmongkon, S., Miyamoto, D., Sriwilaijaroen, N., Fujita, K., Matsumoto, K., Jam-pangern, W., Hiramatsu, H., Guo, CT., Sawada, T., Takahashi, T., Hidari, K., Suzuki, T., Ito, M., Ito, Y., Suzuki, Y., 2008. In vitro inhibition of human influenza A virus infection by fruit-juice concentrate of Japanese plum (Prunus mume SIEB, et ZUCC). Biol. Pharm. Bull. 31, 511–515.
Zhang, J., Gu, H.D., Zhang, L., Tian, Z.J., Zhang, Z.Q., Shi, X.C., Ma, W.H., 2011. Protective effects of apricot kernel oil on myocardium against ischemia-reperfusion injury in rats. Food Chem. Toxicol. 49, 3136–3141.
Acknowledgments
This work was supported by the High Value-added Food Technology Development Program (314043-3), Ministry of Agriculture, Food and Rural Affairs, and the R&D program of MOTIE/KEIT (10040391, Development of Functional Food Materials and Device for Prevention of Aging-associated Muscle Function Decrease).
Author information
Authors and Affiliations
Contributions
JSK, DJK, GYK and CP contributed in running the laboratory work, analysis of the data and drafted the paper. HJC, SK and HSK contributed to critical reading of the manuscript. HJK, BWK, CMK and YHC designed the study, supervised the laboratory work and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kang, J.S., Kim, D.J., Kim, GY. et al. Ethanol extract of Prunus mume fruit attenuates hydrogen peroxide-induced oxidative stress and apoptosis involving Nrf2/HO-l activation in C2C12 myoblasts. Revista Brasileira de Farmacognosia 26, 184–190 (2016). https://doi.org/10.1016/j.bjp.2015.06.012
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.06.012