Abstract
Chronic kidney disease has been linked to cognitive impairment and morphological brain change. However, less is known about the impact of kidney functions on cerebral cortical thickness. This study investigated the relationship between kidney functions and global or lobar cerebral cortical thickness (CTh) in 259 non-demented elderly persons. Forty-three participants (16.7%) had kidney dysfunction, which was defined as either a glomerular filtration rate (GFR) of <60 ml/min/1.73 m2 or presence of proteinuria. Kidney dysfunction was associated with lower global (β = −0.05, 95% CI = −0.08 to −0.01) as well as frontal, parietal, temporal, occipital, and insular lobar CTh. In the stratified analysis, the associations were more pronounced in women, APOEε4 non-carriers, and participants with a lower cognitive score. Besides, kidney dysfunction significantly increased the risk of cortical thinning, defined as being the lowest CTh tertile, in the insular lobe (adjusted odds ratio = 2.74, 95% CI = 1.31−5.74). Our results suggested that kidney dysfunction should be closely monitored and managed in elderly population to prevent neurodegeneration.
Similar content being viewed by others
Introduction
The worldwide incidence and prevalence of dementia is increasing rapidly in persons of advanced age, which results in a huge global disease burden1. Currently, it is estimated that 46.8 million people worldwide are living with dementia, although in some developed countries the prevalence is gradually lowering possibly due to higher education levels and better control of cardiovascular risk factors2, 3. Long before clinically evident dementia-related functional impairment, generalized brain atrophy progresses gradually and asymptomatically with age, beginning at middle age4. Therefore, it is important to identify potentially preventable factors related to brain atrophy during the subclinical phase. Consequently, the structural neuroimaging, among various potential biomarkers for dementia, serves most closely temporal relationship with the onset of clinically detectable cognitive impairment5. The most commonly applied structural neuroimaging technique is magnetic resonance imaging (MRI). In various MRI imaging sequences, physicians can use three-dimensional T1-weighted images that clearly delineate the volume, shape, and thickness of the respective cerebral cortex. Specifically, the volume of cortical regions is a composite measure related to both thickness and surface area. In these two morphometric parameters, cortical thickness more closely reflects pathological changes related to dementia, such as laminar thinning and neuronal loss, while volume loss is largely the result of a reduction in surface area during normal aging6.
Several factors have been linked to dementia, such as age, sex, education, apolipoprotein E (APOE) ε4 status, and various cardiovascular risk factors such as hypertension, diabetes mellitus, metabolic syndrome, and stroke7. In recent years, brain-kidney interaction is increasingly emphasized. Chronic kidney disease (CKD) and dementia are both common diseases in the elderly, and CKD has been proposed as an independent risk factor for cognitive impairment8. A population-based study had demonstrated that cognitive impairment is common not only in hemodialysis patients, but also in the less severe stages of CKD9. CKD was also linked to subclinical brain MRI abnormalities, such as cerebral small vessel disease, white matter hyperintensities, or global brain atrophy10,11,12,13. However, less is known about the relationship between kidney functions and cortical thickness of the brain, especially in a nondisabled elderly population. Therefore, the present study aimed to investigate the potential impact of kidney dysfunctions on cerebral cortical thickness in a group of independently living older persons. This study further explored whether age, gender, APOEε4 status and cognitive function affect the association between kidney function and cerebral cortical thickness.
Methods
Study Population
This is a cross-sectional study from a prospective cohort study (2011-present). Participants aged 65 years or older were recruited from the annual Elderly Health Checkup (EHC) at National Taiwan University Hospital (NTUH) from 2011 to 2013, and all participants underwent a brain MRI examination, except those with contraindications (e.g., implantation of a cardiac pacemaker, defibrillators or other electronic devices, received intracranial metal coils, or claustrophobia). A total of 397 participants who underwent a brain MRI examination were recruited. To clarify the cross-sectional relationship between kidney function and structural brain change, the MRI examination had to be performed within 6 months of specimen collection, along with questionnaire administration, and other clinical examinations (n = 263). As the study aimed to investigate the brain structural change among elderly people without known destructive or degenerative neurological diseases, we excluded those with a history of stroke (n = 3) or using medication for Alzheimer’s disease (AD, n = 1). After exclusion, 259 participants were included for statistical analysis. Informed consent was obtained from each participant before administration of questionnaires, performing clinical assessments, and collection of biological specimen at NTUH. The research plan, informed consent, questionnaires, and application forms were approved by the Institutional Review Board at NTUH. All methods were carried out in accordance with the approved guidelines and regulations.
Acquisition of MRI
Brain MRI was performed using a single 1.5-T scanner with high-resolution T1-weighted volumetric MRI scans. All MRI images were processed using FreeSurfer (http://surfer.nmr.mgh.harvard.edu). The FreeSurfer image analysis suite can perform surface extraction, cortical parcellations and thickness computation14. The cortical thickness (CTh) of each participant was derived from a surface-based pipeline consisting of several stages, including but not limited to the following: image registration via affine transformation to Montreal Neurological Institute atlas, correction of intensity inhomogeneities, removal of skull using a deformable template model, classification of voxels into white matter and non-white matter based on intensity, tessellation of gray-white matter boundaries, automated correction of topological defects, and surface reconstruction to delineate the gray matter-cerebrospinal fluid and gray matter-white matter borders. Ultimately, cortical thickness was obtained at each vertex by computing the shortest distance between the above two surfaces (gray matter-cerebrospinal fluid and gray-white matter). In the FreeSurfer analysis suite, there were 34 predefined regions of interest in each cerebral hemisphere15. Cortical thickness (CTh) was expressed in micrometers as global (whole brain) average and lobar (left and right thickness averaged for frontal, parietal, temporal and occipital lobes) average. The method of cortical thickness measurements has been validated with histological data16 and manual measurements4, and has been used in several previous studies6, 17, 18. Further, total intracranial volume was quantified by automatic segmentation with the FreeSurfer analysis suite. White matter lesions, defined as areas in cerebral white matter that appear abnormally hypointense on T1-weighted or hyperintense on T2-weighted images, were semi-quantitatively graded according to an age-related white matter change rating scale (score 0–3, with a higher score representing more severe lesions) by an experienced neuroradiologist who was unaware of the study population19, 20. Lacunes were defined as small lesions (≤15 mm in diameter) with a low signal on T1-weighted and high signal on T2-weighted images. The presence or absence of lacunes was recorded. In addition to the use of continuous CTh variable for analysis, CTh was further organized into tertiles to identify potential susceptible population from a public health perspective. Cerebral cortical thinning was defined as the lowest tertile (T1), whilenormal CTh was defined as the remaining tertiles (T2 + T3).
Kidney function
To estimate kidney function parameters, the serum creatinine level was obtained from a blood test, and glomerular filtration rate (GFR) was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation21:
In this equation, Scr is serum creatinine (mg/dL), κ is 0.7 for females and 0.9 for males, α is −0.329 for females and −0.411 for males, min indicates the minimum of Scr/κ or 1, and max indicates the maximum of Scr/κ or 1. This equation was more accurate, less biased, and better for precision than the traditionally used Modification of Diet in Renal Disease Study (MDRD) equation21. Urine was also collected from each participant for urinalysis. The level of urinary protein was examined using a urine dipstick and was classified into absence (dipstick reading of − or ± ) or presence (1+ to 4+) of proteinuria, with 1 + approximately corresponding to a proteinuria level of 30 mg/dL,which is the cut-off value for microalbuminuria22, 23. In this study, ‘kidney dysfunction’ was defined if a participant had either a GFR < 60 ml/min/1.73 m2 or presence (≥1+) of proteinuria.
Covariates
A set of relevant covariates was obtained during the study period (2011–2013). Demographic data included age, gender, body mass index (BMI), years of education, and history of smoking. Medical comorbidities such as hypertension, diabetes mellitus, dyslipidemia, metabolic syndrome, or depressive symptoms were documented based on self-report by the participants, available medical records or laboratory data. Laboratory data from blood specimens included total cholesterol, triglycerides, glucose, and APOEε4 status. Cognitive function was evaluated by a trained assistant using the Taiwan version of Montreal Cognitive Assessment (MoCA-T)24, which is a brief test that evaluates several cognitive domains, including visuospatial, executive function, language, verbal memory, attention, and orientation. The MoCA-T scores range from 0 to 30. Participants with MoCA-T score of less than 24 would be classified as having cognitive impairment24. In addition, participants were surveyed using the self-report Center for Epidemiologic Studies Depression (CES-D) scale to assess symptoms of depression, with 20-item score that ranged from 0 to 60. Participants with scores 16 or higher on CES-D scale would be assigned as presence of depressive symptoms25.
Statistical analysis
To examine differences in characteristics between participants with or without kidney dysfunction, a Chi-square test was used for categorical variables, and Student’s t test or Mann-Whitney U-test were used for continuous variables. Correlation between GFR and CTh (global and lobar) was evaluated by Spearman’s rank sum test, and a partial correlation model (represented as ρ) was applied after adjustment of age, gender, years of education, MoCA-T score, and APOEε4 status.
To explore whether the level of GFR or presence of kidney dysfunction were related to global and lobar CTh, multivariable linear regression models were applied. Because one single GFR unit was relatively small, it was expressed as every 10 ml/min/1.73 m2 increase in the regression models. In model 1, important covariates including age, gender, years of education, MoCA-T score, APOEε4 status, and intracranial volume were adjusted. In model 2, degree of white matter lesion, presence of lacunes, BMI, hypertension, diabetes mellitus, and smoking were added to test whether kidney dysfunction related to cortical thinning was independent of cerebral small vessel disease and vascular risk factors. A logistic regression model was used to estimate adjusted odds ratio (aOR) and 95% confidence intervals (CI) with cortical thinning versus normal CTh for GFR values or kidney dysfunction, respectively. The adjusted covariates were the same as the linear regression models described above. False discovery rate correction was used to correct for inflated type I error because of multiple comparisons. An additional stratified analysis was performed based on age groups (65–74 vs ≥75 years old), gender, cognitive status (MoCA-T < 24 vs MoCA-T \(\ge \) 24), and APOEε4 status (carriers vs non-carriers). All analyses were performed using SAS version 9.3 (SAS Institute Inc, Cary, NC, USA). All statistical tests were two-sided, and P values of <0.05 were considered significant.
Results
Characteristics of study population
A total of 259 participants (mean age 73.0 ± 5.2 years old, male 45.6%) from an elderly health checkup program who underwent a comprehensive baseline data collection and brain MRI study were included for analysis. The mean global thickness of the participants was 2.24 ± 0.12 mm. Thirty-five participants had a GFR of <60 ml/min/1.73 m2, thirteen had presence of proteinuria, and a total of 43 participants were classified as having kidney dysfunction. For those with kidney dysfunction, they were generally older, were less educated, had a lower MoCA-T score, had more severe white matter lesions, had thinner global and most of the lobar CTh except the limbic lobe (Table 1). The GFR values were positively correlated with the global and lobar CTh in an unadjusted model (Spearman’s r, 0.16–0.31; all P < 0.05), but partial correlation existed in only the frontal (ρ = 0.16, P = 0.01) and insular (ρ = 0.14, P = 0.03) lobe after a partial adjustment of age, gender, years of educational, MoCA-T score, and APOEε4 status.
Linear association between CTh and kidney function (GFR or kidney dysfunction)
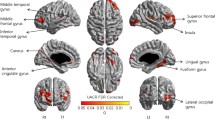
In multiple linear regression models (Table 2), every 10–ml/min/1.73 m2 increase in GFR was significantly associated with a greater CTh for the global (β = 0.01, 95% CI = 0.0005–0.02), frontal (β = 0.01, 95% CI = 0.002–0.02), and insular lobes (β = 0.02, 95% CI = 0.003–0.03) if model 1 was applied; however, the association between GFR and global CTh did not exist if model 2 was applied. Nevertheless, the presence of kidney dysfunction was independently associated with a lower global CTh (β = −0.05, 95% CI = −0.08–−0.01, P = 0.01), as well as frontal, parietal, temporal, occipital and insular lobe CTh (β = −0.04 ~ −0.11; Table 2). The results were similar when model 2 was applied except temporal and limbic lobe. The significant findings remained after correcting multiple tests. In terms of a lower global CTh, kidney dysfunction had similar impacts with 6.5 years of aging (estimated by dividing the coefficient for kidney dysfunction [β = −0.0468] by the coefficient for age per year [β = −0.0072]).
Association between kidney function (GFR or kidney dysfunction) and cortical thinning
No significant association was observed between GFR and global or lobar cortical thinning in the logistic regression models (Table 3), except borderline finding in the frontal lobe (aOR = 0.74, 95% CI = 0.66–0.99, P = 0.04) which was attenuated after correction for multiple comparisons. On the other hand, the presence of kidney dysfunction was associated with an increased risk of insular lobar cortical thinning (aOR = 2.74, 95% CI = 1.31–5.74, P = 0.01; Table 3). However, the effect did not exist for global or other lobar cortical thinning.
Stratified analyses by important potential confounders
No significant interaction existed between kidney dysfunction and age, gender, cognitive or APOEε4 status for global CTh (P interaction = 0.98, 0.08, 0.54, and 0.06, respectively). If using the linear regression model 1, GFR had a significant impact on global CTh in those participants with cognitive impairment (MoCA-T < 24) (β = 0.02, 95% CI = 0.002–0.04; Table 4). In contrast, kidney dysfunction was independently associated with lower global CTh in subgroups of younger age (aged 65–74 years-old, β = −0.05, 95% CI = −0.10 to −0.01), female (β = −0.09, 95% CI = −0.15 to −0.04), and APOEε4 non-carriers (β = −0.06, 95% CI = −0.10 to −0.02; Table 4). The impact of kidney dysfunction on women was equivalent to 20.5 years of aging, while the impact on APOE ε4 non-carriers was equivalent to 6.7 years of aging.
Discussion
The present study demonstrated that decreased kidney function was associated with cerebral cortical thinning in an independently living elderly population. The association was more salient and consistent when combining a lower GFR and proteinuria within the kidney dysfunction population, rather than focusing on GFR alone. Traditionally, a lower GFR was viewed as a hallmark of CKD. Nevertheless, proteinuria was increasingly being viewed as another important indicator and a screening tool for CKD26. Proteinuria revealed dysfunction of the glomerular barrier and often preceded any detectable decline in renal filtration function26. In fact, current guideline of diagnosing CKD required the presence of markers of kidney damage (such as proteinuria) or decreased GFR for more than 3 months23. As our study had a cross-sectional design, we were unable to diagnose participants with CKD but could only label them as having “kidney dysfunction”. Nonetheless, our findings implied that both glomerular filtration rate and barrier dysfunction may contribute to the morphological changes in the cerebral cortex.
Previous studies usually investigated the impact of reduced kidney function on the whole brain volume. For example, in a study including 1253 community-dwelling elders without dementia, a low GFR or the presence of microalbuminuria was significantly associated with generalized brain atrophy12. A Japanese study on 610 adults recruited from health examination confirmed that a lower GFR and the presence of CKD were both associated with generalized cerebral atrophy13. One study in the Netherlands, however, did not demonstrate a significant association between GFR and total gray matter volume in 434 non-disabled elderly persons10.
There were only a few studies that explored the relationship between kidney function and cortical thickness. A Korean study investigated the regional cortical thickness among 162 patients clinically diagnosed with AD, and the results indicated that patients in the lowest GFR quartile had a significantly thinner cortex than those in the highest quartile, especially in the temporal and parietal lobes27. A recently published study from the United States examined the associations between various systemic markers of disease and cortical thickness in 138 healthy middle-aged and elder adults28. They found that kidney function was related to cortical thickness in widespread association and sensory-motor areas. Our findings paralleled their results, however, there were several differences between the two studies that should be emphasized. First, we included not only the glomerular filtration rate but also proteinuria, a potent marker for early kidney damage, compared to their use of blood markers such as GFR, creatinine, and BUN to represent kidney function. Besides, their study employed the surface-based general linear model analysis which incorporated covariate once a time, while we applied a multivariable linear regression to embrace all potential confounders. In doing so, common confounding factors, such as age, sex, educational level, APOEε4 carrier status, and cognitive function could be incorporated into the model, in order to examine these associations in a more comprehensive method. In summary, both studies addressed an important viewpoint that kidney function was closely related to brain morphology even among healthy individuals.
The reasons that kidney dysfunction had a negative effect on the cortical thickness were probably multifactorial (Fig. 1). It had been observed that systemic catabolism of amyloid beta was largely performed by kidney or liver in mice29. In both probable AD patients and normal controls, the serum creatinine level was significantly correlated with plasma amyloid beta (either A β 40 or A β 42)30. Consequently, the accumulation of A β in the body and its downstream neurodegeneration was still the main hypothetical model for the pathophysiological process in AD5. Therefore, reduced kidney function could lead to impaired peripheral excretion and an abnormal accumulation of circulating A β, ultimately resulting in cortical thinning.
Another explanation might be related to a background atherosclerotic state and cerebrovascular dysregulation. Many studies have found that CKD independently contributed to subclinical brain lesions on MRI, such as silent brain infarcts, lacunes, white matter hyperintensities, or cerebral microbleeds10, 11, 31,32,33,34,35. These were mainly lesions of cerebral small vessel disease and could lead to brain atrophy and cortical thinning36. In our study, the independent association between GFR and cortical thickness was attenuated after adjustment of white matter lesions, lacunes, and other vascular risk factors (model 2), but the association persisted between kidney dysfunction and cortical thickness. These results implied that both an excretion abnormality (GFR per se) and endovascular dysfunction (proteinuria) were associated with cortical thinning, and the underlying pathophysiological process may be independent of cerebral small vessel disease.
Yet another mechanism may contribute to the chronic inflammatory state that exists in patients with CKD37. It has been shown that inflammation could mediate the relationship between metabolic syndrome and cortical thinning38. Additional work to explore the contribution of inflammatory markers in the kidney dysfunction related cortical thinning is worthy of investigation.
One interesting finding was that kidney dysfunction was associated with profound cortical thinning in the insular lobe. Although the function of the insula was not fully understood, it was usually linked to emotional process, autonomic function and interoception39, 40. Abnormal changes in insular cortical thickness have been found in patients with chronic musculoskeletal pain, abdominal pain, migraine or smoking addiction39, 41,42,43. In patients with mild AD, depression and anxiety symptoms were found to be associated with thinning of the insular cortex44. We did not find a significant correlation between insular cortical thickness and depressive symptoms scores (based on Center for Epidemiological Studies Depression scale) in our study population. Possible factors contributing to the kidney-dysfunction related insular thinning included circulating toxin, accumulation of A β, or oxidative stress brought by endothelial dysfunction.
Several demographic factors have been found to be associated with changes in cortical thickness. Increasing age was generally associated with less cortical thickness4, 45, 46. In our subgroup analysis, we found that in the young-old group (65–74 years-old), kidney dysfunction was significantly related to a lower global CTh, while the old-old group (\(\ge \)75 years-old) also demonstrated a non-significant relationship. Hence, increasing age did not seem to have an important effect on kidney-dysfunction related cortical thinning. However, close monitoring of kidney function during earlier lifespan was important since the accumulation of Aβ could start from middle age5. Secondly, women tended to have a greater cortical thickness than their age-matched counterpart45, 46, and our results were in line with these findings (women vs men: + 47.7 μm, 95% CI = 21.4–74.1 μm in age-adjusted global cortical thickness). Interestingly, we found that kidney dysfunction had more prominent effects on global CTh in women (equivalent to an aging effect of 20.5 years). Whether this was because men had more competing risk factors of neurodegeneration, or women were more susceptible to kidney dysfunction, is worthy of further exploration.
Generally, APOEε4 carriers, even those with normal cognition, had lower cortical thickness17, 47. We found that APOEε4 status demonstrated a borderline modification effect (P interaction = 0.06) on the association between kidney dysfunction and global CTh, and APOEε4 non-carriers were more likely to be affected by kidney dysfunction. A recent study that demonstrated the association between kidney function factor and CTh did not find APOEε4 status being an effect modifier28. However, their finding was in fact concordant with ours that the negative impact of kidney function on the CTh was more prominent among APOEε4 non-carriers. The results suggested that kidney function should be carefully assessed and monitored in APOEε4 non-carriers who have a risk of neurodegeneration. Further, the association between higher GFR and greater global CTh was found only in participants with cognitive impairment (MoCA-T < 24). Interestingly, participants with cognitive impairment did not have lower CTh than those without (2.23 ± 0.12 mm vs 2.24 ± 0.12 mm, respectively; P = 0.61). It could be speculated that in those who already have cognitive impairment, worsening systemic factors such as a reduction in GFR would have a greater effect on brain morphology.
The present study had some limitations. First, this study was designed to be cross-sectional, and hence, the causality of kidney function and cortical thinning could not be directly inferred. Second, we used urinary dipstick as a semi-quantitative analysis for proteinuria. A more precise method would have been the quantitative measurement of the urinary albumin-to-creatinine ratio to determine the degree of albuminuria. That said, a dipstick test result of less than 1+ usually had a high negative predictive value with a minimal risk of missed diagnosis of microalbuminuria48. Additional study to explore a possible linear relationship between the level of albuminuria and CTh is desirable. Third, we did not use a commonly used case-control method to compare differences in regional CTh49,50,51, as we studied a relatively homogeneous group of participants without any known neurodegenerative diseases or significant medical illness. Moreover, we focused on a potential preclinical association between kidney function changes and CTh. We did not perform the surface-based analysis to generate maps of regional differences by the Freesurfer suite like some researchers did28, since we applied a multivariable linear regression approach to embrace all potential confounders into one model. Lastly, this study was conducted among Taiwanese elderly adults, who were mainly Han Chinese, so it was limited to generalize our study results to other racial or ethnic groups.
In summary, our findings suggest that kidney dysfunction is associated with cerebral cortical thinning in community-dwelling, non-demented elderly population. Further prospective study is warranted to evaluate whether interventions to protect kidney function or to reduce proteinuria could effectively slow the progression of cerebral cortical thinning. Thus, the critical role of kidney function can be better elucidated in the process of neurodegeneration.
References
Prince, M. et al. The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement 9, 63–75 e62, doi:10.1016/j.jalz.2012.11.007 (2013).
Langa, K. M. Is the risk of Alzheimer’s disease and dementia declining? Alzheimer’s Research & Therapy 7, doi:10.1186/s13195-015-0118-1 (2015).
Langa, K. M. et al. A Comparison of the Prevalence of Dementia in the United States in 2000 and 2012. JAMA Intern Med 177, 51–58, doi:10.1001/jamainternmed.2016.6807 (2016).
Salat, D. H. et al. Thinning of the cerebral cortex in aging. Cereb Cortex 14, 721–730, doi:10.1093/cercor/bhh032 (2004).
Jack, C. R. et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. The Lancet Neurology 12, 207–216, doi:10.1016/s1474-4422(12)70291-0 (2013).
Dickerson, B. C. et al. Differential effects of aging and Alzheimer’s disease on medial temporal lobe cortical thickness and surface area. Neurobiol Aging 30, 432–440, doi:10.1016/j.neurobiolaging.2007.07.022 (2009).
Chen, J. H., Lin, K. P. & Chen, Y. C. Risk factors for dementia. J Formos Med Assoc 108, 754–764, doi:10.1016/s0929-6646(09)60402-2 (2009).
Etgen, T., Chonchol, M., Forstl, H. & Sander, D. Chronic kidney disease and cognitive impairment: a systematic review and meta-analysis. Am J Nephrol 35, 474–482, doi:10.1159/000338135 (2012).
Elias, M. F., Dore, G. A. & Davey, A. Kidney disease and cognitive function. Contrib Nephrol1 79, 42–57, doi:10.1159/000346722 (2013).
Ikram, M. A. et al. Kidney function is related to cerebral small vessel disease. Stroke 39, 55–61, doi:10.1161/STROKEAHA.107.493494 (2008).
Khatri, M. et al. Chronic kidney disease is associated with white matter hyperintensity volume: the Northern Manhattan Study (NOMAS). Stroke 38, 3121–3126, doi:10.1161/strokeaha.107.493593 (2007).
Knopman, D. S. et al. Associations of microalbuminuria with brain atrophy and white matter hyperintensities in hypertensive sibships. J Neurol Sci 271, 53–60, doi:10.1016/j.jns.2008.03.009 (2008).
Yakushiji, Y. et al. Marked cerebral atrophy is correlated with kidney dysfunction in nondisabled adults. Hypertens Res 33, 1232–1237, doi:10.1038/hr.2010.171 (2010).
Fischl, B. & Dale, A. M. Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc Natl Acad Sci USA 97, 11050–11055, doi:10.1073/pnas.200033797 (2000).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980, doi:10.1016/j.neuroimage.2006.01.021 (2006).
Rosas, H. D. et al. Regional and progressive thinning of the cortical ribbon in Huntington’s disease. Neurology 58, 695–701, doi:10.1212/WNL.58.5.695 (2002).
Burggren, A. C. et al. Reduced cortical thickness in hippocampal subregions among cognitively normal apolipoprotein E e4 carriers. Neuroimage 41, 1177–1183, doi:10.1016/j.neuroimage.2008.03.039 (2008).
Liu, Y. et al. Education increases reserve against Alzheimer’s disease–evidence from structural MRI analysis. Neuroradiology 54, 929–938, doi:10.1007/s00234-012-1005-0 (2012).
Wahlund, L. O. et al. A new rating scale for age-related white matter changes applicable to MRI and CT. Stroke 32, 1318–1322, doi:10.1161/01.STR.32.6.1318 (2001).
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I. & Zimmerman, R. A. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol 149, 351–356, doi:10.2214/ajr.149.2.351 (1987).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 150, 604–612, doi:10.7326/0003-4819-150-9-200905050-00006 (2009).
Kumai, Y. et al. Proteinuria and clinical outcomes after ischemic stroke. Neurology 78, 1909–1915, doi:10.1212/WNL.0b013e318259e110 (2012).
Chapter 1: Definition and classification of CKD. Kidney Int Suppl (2011) 3, 19–62, doi:10.1038/kisup.2012.64 (2013).
Tsai, C. F. et al. Psychometrics of the Montreal Cognitive Assessment (MoCA) and its subscales: validation of the Taiwanese version of the MoCA and an item response theory analysis. Int Psychogeriatr 24, 651–658, doi:10.1017/s1041610211002298 (2012).
Thomas, J. L., Jones, G. N., Scarinci, I. C., Mehan, D. J. & Brantley, P. J. The utility of the CES-D as a depression screening measure among low-income women attending primary care clinics. The Center for Epidemiologic Studies-Depression. International journal of psychiatry in medicine 31, 25–40, doi:10.2190/FUFR-PK9F-6U10-JXRK (2001).
Agrawal, V., Marinescu, V., Agarwal, M. & McCullough, P. A. Cardiovascular implications of proteinuria: an indicator of chronic kidney disease. Nature reviews. Cardiology 6, 301–311, doi:10.1038/nrcardio.2009.11 (2009).
Cho, E. B. et al. Effect of kidney dysfunction on cortical thinning in patients with probable Alzheimer’s disease dementia. J Alzheimers Dis 33, 961–968, doi:10.3233/JAD-2012-121180 (2013).
Coutinho, A. M. et al. Differential associations between systemic markers of disease and cortical thickness in healthy middle-aged and older adults. Neuroimage 146, 19–27, doi:10.1016/j.neuroimage.2016.11.021 (2016).
Ghiso, J. et al. Systemic catabolism of Alzheimer’s Abeta40 and Abeta42. J Biol Chem2 79, 45897–45908, doi:10.1074/jbc.M407668200 (2004).
Arvanitakis, Z., Lucas, J. A., Younkin, L. H., Younkin, S. G. & Graff-Radford, N. R. Serum creatinine levels correlate with plasma amyloid Beta protein. Alzheimer disease and associated disorders 16, 187–190, doi:10.1097/00002093-200207000-00009 (2002).
Kobayashi, M. et al. Relationship between silent brain infarction and chronic kidney disease. Nephrol Dial Transplant 24, 201–207, doi:10.1093/ndt/gfn419 (2009).
Kobayashi, S., Ikeda, T., Moriya, H., Ohtake, T. & Kumagai, H. Asymptomatic cerebral lacunae in patients with chronic kidney disease. American Journal of Kidney Diseases 44, 35–41, doi:10.1053/j.ajkd.2004.03.026 (2004).
Moodalbail, D. G. et al. Systematic review of structural and functional neuroimaging findings in children and adults with CKD. Clin J Am Soc Nephrol 8, 1429–1448, doi:10.2215/CJN.11601112 (2013).
Shima, H. et al. Decreased kidney function is a significant factor associated with silent cerebral infarction and periventricular hyperintensities. Kidney Blood Press Res 34, 430–438, doi:10.1159/000328722 (2011).
Wada, M. et al. Cerebral small vessel disease and chronic kidney disease (CKD): results of a cross-sectional study in community-based Japanese elderly. J Neurol Sci 272, 36–42, doi:10.1016/j.jns.2008.04.029 (2008).
Pantoni, L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol 9, 689–701, doi:10.1016/s1474-4422(10)70104-6 (2010).
Akchurin, O. M. & Kaskel, F. Update on inflammation in chronic kidney disease. Blood Purif 39, 84–92, doi:10.1159/000368940 (2015).
Kaur, S. S. et al. Inflammation as a mediator of the relationship between cortical thickness and metabolic syndrome. Brain Imaging Behav, doi:10.1007/s11682-014-9330-z (2014).
Maleki, N. et al. Female migraineurs show lack of insular thinning with age. Pain 156, 1232–1239, doi:10.1097/j.pain.0000000000000159 (2015).
Gu, X., Hof, P. R., Friston, K. J. & Fan, J. Anterior insular cortex and emotional awareness. The Journal of comparative neurology 521, 3371–3388, doi:10.1002/cne.23368 (2013).
Baliki, M. N., Schnitzer, T. J., Bauer, W. R. & Apkarian, A. V. Brain morphological signatures for chronic pain. PLoS One 6, e26010, doi:10.1371/journal.pone.0026010 (2011).
Jorgensen, K. N. et al. Cigarette smoking is associated with thinner cingulate and insular cortices in patients with severe mental illness. Journal of psychiatry & neuroscience: JPN 40, 241–249 (2015).
Hubbard, C. S. et al. Abdominal Pain, the Adolescent and Altered Brain Structure and Function. PLoS One 11, e0156545, doi:10.1371/journal.pone.0156545 (2016).
Hayata, T. T. et al. Cortical correlates of affective syndrome in dementia due to Alzheimer’s disease. Arq Neuropsiquiatr 73, 553–560, doi:10.1590/0004-282x20150068 (2015).
van Velsen, E. F. et al. Brain cortical thickness in the general elderly population: the Rotterdam Scan Study. Neurosci Lett 550, 189–194, doi:10.1016/j.neulet.2013.06.063 (2013).
Hilal, S. et al. Risk Factors and Consequences of Cortical Thickness in an Asian Population. Medicine (Baltimore) 94, e852, doi:10.1097/MD.0000000000000852 (2015).
Fennema-Notestine, C. et al. Presence of ApoE epsilon4 allele associated with thinner frontal cortex in middle age. J Alzheimers Dis 26(Suppl 3), 49–60, doi:10.3233/jad-2011-0002 (2011).
White, S. L. et al. Diagnostic accuracy of urine dipsticks for detection of albuminuria in the general community. Am J Kidney Dis 58, 19–28, doi:10.1053/j.ajkd.2010.12.026 (2011).
Seo, S. W. et al. Effects of demographic factors on cortical thickness in Alzheimer’s disease. Neurobiol Aging 32, 200–209, doi:10.1016/j.neurobiolaging.2009.02.004 (2011).
Jubault, T. et al. Patterns of cortical thickness and surface area in early Parkinson’s disease. Neuroimage 55, 462–467, doi:10.1016/j.neuroimage.2010.12.043 (2011).
Chen, J. et al. Reduced cortical thickness, surface area in patients with chronic obstructive pulmonary disease: a surface-based morphometry and neuropsychological study. Brain Imaging Behav, doi:10.1007/s11682-015-9403-7 (2015).
Acknowledgements
Funding for this study was provided by grants from the Ministry of Science and Technology in Taiwan (104-2314-B-002-038-MY3, 103-2314-B-002-033-MY3, 101-2314-B-002-126-MY3, and 100-2314-B-002-103). These funding sources had no role in the design, methods, subject recruitment, data collection, analysis and preparation of paper. We thank Dr. Michael Pao Chen Yang for English editing and proofreading.
Author information
Authors and Affiliations
Contributions
C.H.C. conceived this work, analyzed the data, wrote the manuscript, and had primary responsibility for final content. P.H.T. and Y.C.C. analyzed the data and wrote the manuscript. J.H.C., Y.C.C. and M.J.C. designed the study and recruited the study participants. Y.F.C. assessed the neuro-imaging and served as the consultant of neuro-radiology. C.J.Y. served as the consultant of nephrology. T.F.C. and M.J.C. served as the consultant of neurology. S.C.T. and S.J.Y. conducted the medical examinations. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, CH., Chen, YF., Chiu, MJ. et al. Effect of Kidney Dysfunction on Cerebral Cortical Thinning in Elderly Population. Sci Rep 7, 2337 (2017). https://doi.org/10.1038/s41598-017-02537-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02537-y
- Springer Nature Limited