Abstract
How forest management practice impacts the temperature response of soil carbon decomposition remains unclear in Tibetan boreal forests. Here, an experiment was conducted to compare soil carbon decomposition of two layers (organic and mineral) in three Tibetan forests (natural forest, NF; secondary forest, SF; spruce plantation, PF). Soils were incubated at two temperatures (10 °C and 20 °C) for 219 days. Increased temperature often stimulated carbon decomposition rates of organic layer but did not affect them in the mineral soils. Soil carbon decomposition rates in the organic layer followed a pattern of NF > SF > PF over the incubation period. Regardless of forest type, soil carbon decomposition rates and temperature coefficient (Q 10) were higher in the organic layers compared to mineral soils. Moreover, forest type conversion increased Q 10 values in each soil layer. Taken together, our results suggest that forest management practice has much stronger impacts on biochemical properties in the organic layers relative to mineral soils. Moreover, the temperature responses of soil carbon decomposition depend largely on forest management practice and soil layer in this specific area.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Boreal forests cover one third of the world’s forested area and store about 30% of the global terrestrial carbon pool1. It is believed that boreal forest soil, acts as a key carbon pool, plays an increasingly important role in carbon (C) cycling of terrestrial ecosystems. Climate warming is predicted to affect almost all terrestrial ecosystems and will be particularly pronounced in cold biomes2. Temperature is considered to be a key factor that regulates the decomposition of soil organic matter (SOM), which is a large component of the terrestrial carbon budget3, 4. Thus, warming-associated increases in SOM decomposition could profoundly affect the carbon balance in boreal forest soils, with consequent feedbacks to global warming.
The temperature response of soil C decomposition may depend largely on the initial conditions of substrates, such as stocks of SOM, the chemical composition and microbial community5,6,7. In addition, soil responses to climate change could also be complicated by land-use change8. Forest management practice (e.g., artificial reforestation and natural regeneration) often produces significant changes in soil biochemical conditions, which in turn could directly and/or indirectly affect the response of soil C decomposition to climate change9, 10. As a consequence, it is very crucial to synchronously compare the temperature effects on soil C decomposition under different forest managements.
As well known, boreal forests accumulate a large amount of organic material in the surface forest floor (a thick organic layer) as a result of slow decomposition process. An organic layer often includes various stages of decomposed organic matter, such as highly decomposed, septic; moderately decomposed, hemic, and minimally decomposed11. Compared to mineral horizons in the soil profile, they are rich in organic matter, with typically black or dark brown in color. In boreal forest ecosystems, organic layer is considered to be the most active interface where many biochemical cycles between trees and soils occur12. The organic layer and the mineral soil often have different substrate quality and availability for the decomposition of SOM due to different rates of C input, accumulation, and turnover in both layers13, 14. Therefore, the temperature sensitivity of soil C decomposition could be differential between the two soil layers.
The Tibetan forests are typical alpine boreal forests at low latitude, with important consequences for regional and global carbon balance. The magnitude of climate change on the Tibetan Plateau is projected to be large relative to many other regions15. Additionally, a large amount of SOM is stored in the organic layer besides the mineral soil in Tibetan forests16. Therefore, soil C decomposition of Tibetan forests is likely to be more pronounced relative to other forest ecosystems in a warmer world. Over the last decades, the natural coniferous forests have been harvested in large-scale industrial logging, and replaced by secondary forests and dragon spruce plantation under national restoration programs17. Forest management practice (e.g., artificial reforestation or natural regeneration) often induces significant changes in soil physical and biochemical properties12, especially in the organic layer, which in turn might largely regulate the temperature responses of soil C decomposition. Here, an experiment was conducted to explore temperature effects on soil C mineralization of two layers (organic and mineral) in three contrasting forest ecosystems (natural forest, secondary forest and spruce plantation) on the eastern Tibetan Plateau. Specifically, we hypothesized that (1) forest land-use change would lower soil substrate quality and C decomposition; (2) temperature response of soil C decomposition would vary with forest types and soil layers.
Results
Soil properties
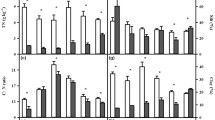
Compared to the mineral soils, SOC, N and P in the organic layer were 2.9–4.7, 2.0–6.3 and 1.2–2.4 times higher among three forests (Fig. 1a,b and c, all p < 0.05). In the organic layer, SOC, N and P in both NF and SF were significantly higher than those in the PF (p < 0.05). However, SOC, N and P was greatest in the SF in the mineral soil (p < 0.05). Compared to the mineral soils, lower C:N ratio of the organic layer was only observed in the NF (Fig. 1d). Similarly, there were no significant differences in C:P ratio among the forests (Fig. 1e). However, C:P ratio was higher in the organic layer in each forest type as compared to mineral soil (p < 0.05). Soil pH increased from NF to SF or PF in both soil layers (Fig. 1f). The statistical analysis showed that the effect of forest conversion on SOC, N, P and pH were dependent on soil layer (Table 1).
Effects of forest type conversion and soil layer on soil properties. Values indicate means ± SE, n = 4. Different letters within the same soil layer denote significant differences among forest types by one-way ANOVA. Asterisk indicates significant differences between the two soil layers by student t-test. NF: natural forest, SF: secondary forest and PF: spruce plantation.
Soil microbial community
Bacteria, fungi and their ratio were significantly affected by forest conversion and soil layer (Fig. 2, Table 1). In the organic layer, bacteria and fungi PLFAs were markedly larger in both NF and SF compared to PF (Fig. 2a). In the mineral layer, bacteria PLFAs were 3.9 and 2.8 times greater, respectively, in the SF and PF compared to NF (Fig. 2a). Bacteria PLFAs in the organic layer were significantly higher as compared to mineral soil in both NF and SF. However, the opposite pattern was true in the PF (Fig. 2a). In addition, fungi PLFAs were higher in the organic layer than in the mineral soil except for the PF (Fig. 2b). However, forest conversion did not affect bacteria:fungi ratio in the organic layer (Fig. 2c). Additionally, obvious difference in bacteria:fungi ratio between soil layers was only observed in the PF (Fig. 2c). The ANOVA results showed that there were interactive effects of forest type and soil layer on bacteria, fungi and their ratio (Table 1).
Effects of forest type conversion and soil layer on soil microbial properties. Values indicate means ± SE, n = 4. Different letters within the same soil layer denote significant differences among forest types by one-way ANOVA. Asterisk indicates significant differences between the two soil layers by student t-test. NF: natural forest, SF: secondary forest and PF: spruce plantation.
Soil C mineralization
Forest type, incubation temperature, soil layer and time all had significant effects on soil C mineralization rate and cumulative C production (Fig. 3, Table 2). In the organic layer, both soil C mineralization rate and cumulative amount of C mineralization at 20 °C were higher than those at 10 °C on most of measurements (Fig. 3a,c). However, temperature often did not affect mineral soil C mineralization (Fig. 3b,d). Irrespective of incubation temperature, soil C mineralization rate was remarkably higher in the organic layer than in the mineral soil in each forest type (Fig. 3a–d, Table 2). In the organic layer, both soil C mineralization rate and cumulative C release followed a pattern of NF > SF > PF during the incubation period (Fig. 3a–d). The ANOVA results indicated that temperature effect on soil C mineralization was dependent on forest type and soil layer (Table 2).
Temperature sensitivity (Q 10)
Forest type and soil layer significantly affected Q 10 values (Fig. 4, Tables 1 and 2). The Q 10 varied from 1.35 to 2.82 across three forest types (Fig. 4). Irrespective of soil layer, Q 10 was higher in the SF as compared to NF and PF. Meanwhile, Q 10 values of organic layers were higher than those of mineral soils (Fig. 4). The statistical analysis showed that the interaction of forest type and soil layer was not significant on Q 10 value (Table 1, p < 0.05).
Effects of forest type conversion and soil layer on temperature coefficient Q 10. Values indicate means ± SE, n = 4. Different letters within the same soil layer denote significant differences among forest types by one-way ANOVA. Asterisk indicates significant differences between the two soil layers by student t-test. NF: natural forest, SF: secondary forest and PF: spruce plantation.
The correlations between soil substrates and C decomposition
SOC, N, P, bacteria and fungi were positively correlated with soil C mineralization rate (Table 3, all p < 0.001). However, pH, C:N and bacteria:fungi had a negative relationship with soil C mineralization rate (Table 3). Similarly, there were a marginal relationship between Q 10 value and SOC, N, P and fungi (Table 3, all p < 0.1). Conversely, Q 10 value decreased with increasing C:N and bacteria:fungi (Table 3, p < 0.1).
Discussion
Forest land-use change could affect soil C mineralization directly and/or indirectly thought altering soil substrate conditions, including soil C quantity and quality, substrate availability and microbial properties18,19,20. A number of studies have reported that the conversion from natural forests to secondary forests and/or plantations decreased soil C pool, leading to lower soil C mineralization rates18, 21, 22. For instance, the secondary forest had higher C pools, microbial biomass and C mineralization rate as compared to larch plantations in Northeast China18. In this study, irrespective of incubation temperature, soil C mineralization rates in the organic layer generally followed a tendency of NF > SF > PF over the incubation period. This could be mainly attributed to the changes in soil substrate and microbial properties following the forest type conversion. SOC and N pools were decreased following the conversion from NF to PF. Additionally, both fungi and bacteria are two dominant microbial decomposer groups controlling soil C mineralization23, 24. Our results found that both soil fungi and bacteria PLFAs were markedly higher in the NF and SF than in the PF. This was also supported by statistical analysis because there were significant positive correlations between soil C mineralization rate and SOC, N and microbial PLFAs. Besides, it has been demonstrated that high-quality SOC is of great benefit to microbial carbon use efficiency7. Similar to previous studies25, 26, our results have also shown that soil C mineralization rate is negatively linked to C:N.
In boreal forests, there is an obvious organic layer accumulated in the upper forest floor due to slow decomposition. There are significant differences in soil biochemical properties between organic layer and mineral soil due to different rates of C input, accumulation, and turnover13, 14. Therefore, soil C decomposition rate could differ largely between two soil layers27. In this case, soil C mineralization rate was markedly greater in the organic layer compared to mineral soils in each forest type. This result was consistent with the observations from other boreal ecosystems13, 27. This is mainly because soil C pool and microbial PLFAs are extremely higher in the organic layer relative to mineral soil. It is widely accepted that SOC and microbial biomass directly regulate soil C mineralization in terrestrial ecosystems. Forest management practice profoundly and directly alters litter inputs which control substrate availability and quality for soil C decomposition7, 20. Previous studies have reported that fine roots mainly distributed in the organic layer in Tibetan forests28. Apparently, organic layer is much more vulnerable to forest land-use change as compared to mineral soils. Our results found that forest land-use change caused significant effects on soil biochemical properties, especially in the organic layer.
In recent years, the temperature sensitivity of soil C decomposition has gained much more attention29, 30. The temperature sensitivity of soil C decomposition, the factor by which soil C decomposition rate increases by 10 °C increases, is a key parameter to evaluate the feedback intensity between soil C efflux and climate change. Temperature coefficient Q 10 is the most common measure to assess the temperature sensitivity of SOM decomposition in empirical studies. In the present case, forest land-use change may affect Q 10 value via altering soil substrate availability and lability20, 27. In this study, Q 10 values varied from 1.35 to 2.82 among three forest soils. A current synthesis has demonstrated that the Q 10 values ranged from 1.10 to 5.18 across China’s forests30. Forest type conversion completely change dominant tree species and litter type, consequently affecting soil substrate and microbial properties, which are closely associated with Q 10 value7. For example, the conversion from a primary forest dominated by Quercus liaotungensis to artificial plantations (Larix principis-rupprechtii and Pinus tabulaeformis) or secondary shrub forest significantly lowered Q 10 value in northern China31. However, our results found that forest type conversion increased Q 10 value. However, soil microbial biomass and C release rate were increased after conversion from native broadleaf forest to plantations in subtropical region32. Such differences imply that the effects of forest land-use change on soil C decomposition may vary with climatic zones.
On the other hand, some studies have reported that Q 10 values increased with soil profile, reflecting that a decrease in substrate lability with soil depth27, 33. However, our results observed that Q 10 value was greater in the organic layer as compared to mineral soil. This result was consistent with the findings observed in other boreal soils at high latitudes13, 27. Compared to mineral soils, there was a sharp reduction in soil C decomposition rate in the organic layers during the initial period of the incubation, implying that organic layer contained a small pool of very labile C pools, which was rapidly depleted over the early period of the incubation. Similar patterns have been observed in other boreal soils13, 27. Moreover, it was believed that the larger Q 10 in the organic layer relative to mineral soil may be attributed to the extremely higher C availability, which may cause a decrease in the “cancelling effect”13. Finally, our study also showed that Q 10 was positively associated with fungi PLFAs but negatively with bacteria:fungi ratios. Greater activation energy could be required for soil C mineralization when microbial activity is low, which may partially contribute to a higher Q 10.
Conclusions and implications
In summary, this study explored variations of soil C mineralization rate and its temperature sensitivity in the organic layer and mineral soil among three contrasting forests. Our results revealed that forest land-use change caused significant changes in substrate properties (e.g., C pools and PLFAs), especially in the organic layer. Both forest type and soil layer significantly influenced soil C mineralization rate and Q 10 value. Taken together, the results demonstrated that soil C mineralization and its temperature sensitivity was a complex process that was susceptible to both direct and/or indirect controls derived from forest type conversion.
The findings in this study have the following important implications. On the one hand, forest management practice dramatically reduced soil C pools in both organic and mineral soils but significantly increase the Q 10 of soil C decomposition. Thus, effective measures should be taken to manage the current primary forests to mitigate warming in this specific area. On the other hand, because soil C pool stored in the organic layer is very large in boreal forests and global warming is relatively pronounced in the surface layer. The higher Q 10 value of the organic layer highlights its importance in boreal forests under a warming scenario. The differences in temperature response between the two layers should be considered when predicting soil C dynamic in boreal forests under a warmer world.
Materials and Methods
Study area
This study was conducted at the Long-term Research Station of Alpine Forest Ecosystems, which is located in the eastern Tibetan Plateau, China (102°53′–102°57′E, 31°14′–31°19′N). Mean temperature ranges from 2 to 4 °C and annual mean precipitation equals 850 mm. The soil was classified as dark brown forest soil with a 10–15 cm deep organic matter layer. Natural coniferous forest (NF), secondary birch forest (SF) and dragon spruce plantation (PF) are three dominant forest types due to local forest management practice. In July 2015, four 20 × 20 m plots were randomly established in each forest type. The basic conditions of three forests were recorded (Supplementary Table S1).
Soil sampling
Soil samples of the organic layer and the upper mineral soil (10 cm) were collected in each plot. The organic layer was identified from the mineral soil via its color, texture and consistency13. Nine cores (5 cm diameter) were taken randomly from each plot and soil samples from same layer were mixed to get one composite sample. Each composite sample was passed through a sieve, and any visible living plant material was removed manually from the sieved soil. The sieved soil was kept in the refrigerator at 4 °C prior to the analysis of microbial properties. A sub-sample of each soil was air-dried and ground prior to chemical analysis.
Sample analyses
Soil organic carbon (SOC) was measured using the dichromate oxidation sulfate ferrous titration method. Soil nitrogen (N) was analyzed following the Kjeldahl digestion procedure. Soil phosphorus (P) was determined using the phosphomolybdenum yellow colorimetry method. Soil pH was measured with a Calomel electrode at 1:5 soil-to-water ratio. The phospholipid fatty acids (PLFAs) were extracted and quantified using a modified method previously described by White6. Bacteria were identified by the following PLFAs: i15:0, a15:0, 16:0, 17:0, a17:0, 16:1w7c, 15:0, 16:1w5t, i17:0, 16:1w9c, 18:1w7c, 18:00, cy19:0, cy17:0, i16:0 and 20:5. Fungi was determined by the PLFAs 18:3, 18:2w6,9c, 18:1w9c and 20:1w9c.
Soil C mineralization
Fresh soil samples (100 g) of the two layers were adjusted to 60% water holding capacity, which was considered optimal for microbial activity34. The soil samples were incubated in 1 L jars at two temperatures (10 °C and 20 °C). Empty jars without soils were used as controls. CO2 production was measured on 2, 5, 8, 15, 22, 29, 36, 43, 50, 57, 71, 85, 99, 113, 134, 155, 187 and 219 days after the incubation by using alkali absorption method. Soil samples were remoistened to keep moisture at each measuring time. The rate of soil C mineralization was calculated per unit mass in the unit time for average rate, and accumulative C production was the CO2 in the sum of unit time.
Temperature sensitivity
The temperature sensitivity of soil C decomposition, the factor by which soil C decomposition rate increases by 10 °C increases, is a key ecological parameter in ecosystem carbon cycle models. Temperature coefficient Q 10 is the most common measure to assess the temperature sensitivity of C decomposition in empirical studies. Therefore, Q 10 was also applied in this study to compare temperature sensitivity of soil C decomposition among forest types using the method stated in Leifeld and Fuhrer35.
Where R 20 and R 10 are the average C mineralization rates at 20 °C and 10 °C, respectively. W is the difference of incubation temperature.
Statistical analysis
Four-way ANOVAs were employed to analyze the effects of forest type, soil layer, incubation temperature and time on soil C mineralization rates and accumulative C production. Two-way ANOVAs were used to test the effects of forest type and soil layer on measured soil variables and Q 10 values. For same layer, one-way ANOVAs were used to identify significant differences in soil properties among forest types. For same forest type, student t-tests were used to compare the effect of the soil layer. The correlations between soil respiration rate, cumulative C production and Q 10 and soil biochemical properties were analyzed by Pearson coefficient. The statistical tests were considered significant at the p < 0.05 level. All statistical tests were performed using IBM SPSS Statistics 20.0.
References
Bradshaw, C. J., Warkentin, I. G. & Sodhi, N. S. Urgent preservation of boreal carbon stocks and biodiversity. Trends. Ecol. Evol. 24, 541–548 (2009).
Solomon, S. Climate change 2007-the physical science basis: Working group I contribution to the fourth assessment report of the IPCC. Vol. 4 (Cambridge University Press, 2007).
Ågren, G. I. & Wetterstedt, J. M. What determines the temperature response of soil organic matter decomposition? Soil Biol. Biochem. 39, 1794–1798 (2007).
Hamdi, S., Moyano, F., Sall, S., Bernoux, M. & Chevallier, T. Synthesis analysis of the temperature sensitivity of soil respiration from laboratory studies in relation to incubation methods and soil conditions. Soil Biol. Biochem. 58, 115–126 (2013).
Riffaldi, R., Saviozzi, A. & Levi-Minzi, R. Carbon mineralization kinetics as influenced by soil properties. Biol. Fertil. Soils 22, 293–298 (1996).
Thiessen, S., Gleixner, G., Wutzler, T. & Reichstein, M. Both priming and temperature sensitivity of soil organic matter decomposition depend on microbial biomass–an incubation study. Soil Biol. Biochem. 57, 739–748 (2013).
Fierer, N., Craine, J. M., Mclauchlan, K. & Schimel, J. P. Litter quality and the temperature sensitivity of decomposition. Ecology 86, 320–326 (2005).
Luo, Y., Wan, S., Hui, D. & Wallace, L. L. Acclimatization of soil respiration to warming in a tall grass prairie. Nature 413, 622–625 (2001).
Chen, G. et al. Accelerated soil carbon turnover under tree plantations limits soil carbon storage. Sci. Rep 6, 19693 (2016).
Fang, X. et al. Land Use Effects on Soil Organic Carbon, Microbial Biomass and Microbial Activity in Changbai Mountains of Northeast China. Chin. Geograp. Sci 24, 297–306 (2014).
Chapin, III, F. S., Matson, P. A. & Vitousek, P. Principles of terrestrial ecosystem ecology. 78–79 (Springer Science and Business Media, 2011).
Yang, W., Feng, R., Zhang, J. & Wang, K. Carbon stock and biochemical properties in the organic layer and mineral soil under three subalpine forests in Western China. Acta Ecol. Sin 27, 4157–4165 (2007).
Xu, W., Li, W., Jiang, P., Wang, H. & Bai, E. Distinct temperature sensitivity of soil carbon decomposition in forest organic layer and mineral soil. Sci. Rep 4, 6512 (2014).
Trumbore, S. E. & Harden, J. Accumulation and turnover of carbon in organic and mineral soils of the BOREAS northern study area. J. Geophys. Res.: Atmospheres 102, 28817–28830 (1997).
Chen, H. et al. The impacts of climate change and human activities on biogeochemical cycles on the Qinghai‐Tibetan Plateau. Global Change Biol 19, 2940–2955 (2013).
Xu, Z. et al. Initial responses of soil CO2 efflux and C, N pools to experimental warming in two contrasting forest ecosystems, Eastern Tibetan Plateau, China. Plant Soil 336, 183–195 (2010).
Liu, Q., Wu, Y. & He, H. Ecological problems of subalpine coniferous forest in the southwest of China. World Sci-Tech R. D. 23, 63–69 (2001).
Yang, K., Shi, W. & Zhu, J. J. The impact of secondary forests conversion into larch plantations on soil chemical and microbiological properties. Plant Soil 368, 535–546 (2013).
Dube, F., Zagal, E., Stolpe, N. & Espinosa, M. The influence of land-use change on the organic carbon distribution and microbial respiration in a volcanic soil of the Chilean Patagonia. Forest Ecol. Manage. 257, 1695–1704 (2009).
Hao, S. et al. The dynamic response of soil respiration to land-use changes in subtropical China. Global Change Biol 16, 1107–1121 (2010).
Ahn, M.-Y., Zimmerman, A. R., Comerford, N. B., Sickman, J. O. & Grunwald, S. Carbon Mineralization and Labile Organic Carbon Pools in the Sandy Soils of a North Florida Watershed. Ecosystems 12, 672–685 (2009).
Ding, Y., Mao, Z., Zhang, L. & Ding, L. Comparison of Soil Organic Carbon Stock of Betula costata Secondary Forest and Mixed Broadleaved Korean Pine Original Forest in Xiaoxing’an Mountains. Bull. Botan. Res 35, 604–611 (2015).
Rousk, J., Brookes, P. C. & Bååth, E. Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. AppL. Environ. Microbiol. 75, 1589–1596 (2009).
Li, J., Wang, G., Allison, S. D., Mayes, M. A. & Luo, Y. Soil carbon sensitivity to temperature and carbon use efficiency compared across microbial-ecosystem models of varying complexity. Biogeochemistry 119, 67–84 (2014).
Xu, X. et al. Soil properties control decomposition of soil organic carbon: Results from data-assimilation analysis. Geoderma 262, 235–242 (2016).
Spohn, M. & Chodak, M. Microbial respiration per unit biomass increases with carbon-to-nutrient ratios in forest soils. Soil Biol. Biochem. 81, 128–133 (2015).
Karhu, K. et al. Temperature sensitivity of organic matter decomposition in two boreal forest soil profiles. Soil Biol. Biochem. 42, 72–82 (2010).
Liu, L. et al. Biomass and carbon stock of fine roots in the representative subalpine and alpine forests in Western Sichuan. J. Sichuan Sci. Tech 1, 7–10 (2008).
Davidson, E. A. & Janssens, I. A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440, 165–173 (2006).
Xu, Z. et al. Temperature sensitivity of soil respiration in China’s forest ecosystems: Patterns and controls. Appl. Soil Ecol. 93, 105–110 (2015).
Quan, Q. et al. Forest type affects the coupled relationships of soil C and N mineralization in the temperate forests of northern China. Sci. Rep 6, 6584 (2014).
Wang, Q., Xiao, F., He, T. & Wang, S. Responses of labile soil organic carbon and enzyme activity in mineral soils to forest conversion in the subtropics. Ann. Forest Sci. 70, 579–587 (2013).
Fontaine, S. et al. Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 450, 277–280 (2007).
Bowden, R. D., Newkirk, K. M. & Rullo, G. M. Carbon dioxide and methane fluxes by a forest soil under laboratory-controlled moisture and temperature conditions. Soil Biol. Biochem. 30, 1591–1597 (1998).
Leifeld, J. & Fuhrer, J. The temperature response of CO2 production from bulk soils and soil fractions is related to soil organic matter quality. Biogeochemistry 75, 433–453 (2005).
Acknowledgements
This research was financially supported by the National Key Research and Development Program of China (2016YFC0502505) and the Natural Science Foundation of China (31570601, 31570445).
Author information
Authors and Affiliations
Contributions
Z.X. and W.Y. designed the study, B.T., K.Y., Y.L. and L.Y.Z. (Liyan Zhuang) sampled the soils, K.Y., Z.L, and R.H. analyzed the samples, Z.X., K.Y, L.Z., L.T., and F.W. calculated and evaluated the data statistically. Finally, Z.X. and K.Y. interpreted the results, designed and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, K., He, R., Yang, W. et al. Temperature response of soil carbon decomposition depends strongly on forest management practice and soil layer on the eastern Tibetan Plateau. Sci Rep 7, 4777 (2017). https://doi.org/10.1038/s41598-017-05141-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-05141-2
- Springer Nature Limited
This article is cited by
-
Carbon and nitrogen cycling on the Qinghai–Tibetan Plateau
Nature Reviews Earth & Environment (2022)
-
Stable isotopic signatures of carbon and nitrogen in soil aggregates following the conversion of natural forests to managed plantations in eastern China
Plant and Soil (2021)
-
Converting natural evergreen broadleaf forests to intensively managed moso bamboo plantations affects the pool size and stability of soil organic carbon and enzyme activities
Biology and Fertility of Soils (2018)