Abstract
Powdery mildew is one of the serious diseases of garden pea which causes a large number of yield losses. Genetic resistance is quite effective, being cost-effective and environment friendly than fungicide applications. In the present studies an initial attempt has been made to identify resistant genotypes against powdery mildew disease developed from hybridization followed by validation of the disease. The experimental material comprised of 48 genotypes that includes 44 advanced breeding lines was evaluated for powdery mildew incidence in Randomized Complete Block Design with three replications at two locations under field conditions [Palampur (winter 2017–18 and 2018–19) and Kukumseri (summer 2018)] and in vitro at Palampur [detached leaf method and polyhouse conditions]. Ten lines viz., SP7, SN-1, SN-6-1, SN-7-1, SN-2, SN-5-2, SN-6-2, SN-10, SN-21 and SP-281 showed resistant reaction along with check Palam Sumool while 27 lines were identified as moderately resistant in comparison to susceptible check Azad P-1. Besides, six lines namely, SP-2, SP-5, SP-10, SP-24, SA-4 and SP-12-1 gave moderately susceptible reaction along with checks Pb-89 and Palam Priya. Only, SP-19 was categorized as susceptible. The high yielding lines SP-3, SP-6 and SP-22 showed moderately resistant reaction in both natural and artificial conditions. Validation of resistance using molecular markers revealed that neither the parental genotypes nor the progenies possess the er1 gene of JI1559. The er2 linked marker ScOPX-171700 was polymorphic between Palam Sumool and Palam Priya but the marker didn’t show polymorphism between er2 harboring line (JI2480). These results suggested that the lines showing resistance under field conditions may have some other genes or alleles for resistance and further confirmation is needed by developing mapping populations with specific gene or gene combinations.
Similar content being viewed by others
Introduction
Garden pea (Pisum sativum L.) is an important nitrogen-fixing vegetable crop of Fabaceae family and forms a significant component of sustainable cropping systems, helps to preserve soil health and output1. It is the oldest model of plant genetics and one of the most important legumes in the world2. Garden pea is quite palatable and excellent food for human consumption3,4 and provides an exceptionally diverse nutrient profile viz., vitamins, minerals and lysine, a limiting amino acid in cereals1. Also, fresh pea pods are good source of folic acid, ß-sitosterol, vitamin C and K5. Antibacterial, antidiabetic, antifungal, anti-inflammatory, anti-hypercholesterolemia and anti-cancerous properties further support its dietary benefits6. India ranks second both in area and production in the world7.
Various biotic and abiotic stresses make the local landraces less profitable to farmers due to the reduction in yield. To overcome the further economic loss in the context of biotic and abiotic stresses, there is a dire need to breed resistant and high yielding varieties. Powdery mildew in garden pea caused by Erysiphe pisi DC. is a serious disease that can cause 25–50 per cent yield losses8. Genetic resistance is quite effective strategy for disease management as it is more cost-effective and environment friendly. The genetics of powdery mildew resistance (PMR) in pea is relatively well understood with three major reported loci er1, er2 and Er39,10,11. Different modes of inheritance i.e. single recessive12,13, single dominant14,15 and duplicate recessive gene action16,17 have been reported for powdery mildew resistance. The recessive ‘er1’ gene is responsible for resistance to majority of the naturally occurring powdery mildew disease18,19,20,21 and was commonly used in pea breeding for development of PMR cultivars. Later, numerous new alleles have been identified which were derived either from natural or artificial mutagenized population22,23. The chromosomal position of PMR genes viz., ‘er2’ and ‘Er3’ are known but cloning has not been reported yet. The DNA markers linked to resistance genes provide an alternative to powdery mildew screening and provide an accurate measure as they are not affected by epistatic interactions. They can be used to confirm the presence of multiple resistance genes thereby, increasing efficiency of selection and reducing time span for the introgression of resistance genes.
High yield, specific pod characteristics (proper filling, long, dark green, sweet) and resistant to pests and diseases are the main criteria opted by the breeders for garden pea improvement. The focus on improvement of these specific traits has led to narrowing its genetic base. The varieties like ‘Azad P-1’, ‘Lincoln’, ‘Arkel’ etc. are still preferred by the growers due to desirable horticultural traits though the varieties have become vulnerable to a plethora of biotic and abiotic stresses particularly powdery mildew24 and has led to low/stagnant yield and a major impediment in pea improvement. Various studies were conducted for screening and identification of pea genotypes for PMR25,26,27 but these studies have not undertaken validation using molecular markers. In this perspective, four diverse parents selected on the basis of diverse phenological traits were involved in three inter-varietal crosses to isolate transgressive segregants in the recent years that have resulted in 44 progenies with desirable pod characteristics. The present investigation was, therefore, planned with the objective of screening of newly identified genotypes for powdery mildew resistance both in vivo and in-vitro conditions and validation of resistance using molecular markers.
Material and methods
Experimental material
The experimental material comprised of 48 genotypes of garden pea of which 44 were advanced breeding lines isolated from three inter-varietal crosses for high pod yield along with four recommended varieties as standard checks. The details are given in the Table 1.
Site of experiment
The experimental material was evaluated at two diverse locations under field conditions at Palampur for two years during winter season (2017–18 and 2018–19) and Kukumseri during summer season of 2018 at 2nd picking and final harvest stage to identify the resistant breeding lines of garden pea against powdery mildew.
Field screening
Observations on powdery mildew disease severity under field conditions were recorded at peak harvest stage (second/third picking) and also at seed maturity stage macroscopically by following methodology suggested by Banyal and Tyagi28. Five infection types were recorded as 0, 1, 2, 3 and 4 scale of Mains and Deitz (Table 2)29.
In-vitro screening
The genotypes were also evaluated under in-vitro conditions in polyhouse at Palampur by planting 3–5 seeds of each genotype in individual pots. The in-vitro multiplied conidial inoculum of the disease was dusted on the plants with camel hair brush for uniform development of disease infestation to facilitate screening of genotypes for resistance. Simultaneously, all the lines were also screened by using detached leaf method28 under laboratory conditions. Detached leaf assay under in-vitro conditions was conducted using three replications of each variety in the petri dishes under artificial conditions against Palampur isolate of Erysiphe pisi along with susceptible and resistant check varieties in the Department of Vegetable Science and Floriculture Laboratory at CSKHPKV, Palampur. The leaves along with petiole were detached from 15 to 30 days old seedlings of each accession and floated on tap water in petri dishes. Fifty ppm of benzimidazole was added to enhance the longevity of the detached floating leaves which were inoculated with the isolate. The petri dishes were incubated at room temperature in laboratory. Observations on the disease development were made at 24 h interval up to twelve days. Infection types were recorded based on macroscopic and microscopic density of mycelia and sporulation at 9 days interval.
Molecular validation of powdery mildew resistance
Isolation, purification and quantification of plant genomic DNA
The total genomic DNA was extracted from all the 48 genotypes and er1 and er2 harboring lines (JI- 1559 and JI-2480) followed by PCR amplification using er1 and er2 linked markers. The details of molecular markers are presented in Table 3. DNA was isolated from young leaf tissue by using CTAB method given by Murray and Thompson30. The extracted DNA samples of all genotypes were loaded on 0.8% agarose gel (1gm/100 ml 1X TAE buffer) and run at 90 V for 40 min to determine the quality and quantity of DNA.
Genomic DNA amplification in polymerase chain reaction (PCR)
DNA amplification was carried out in a 12.5 µl reaction volume containing 20 ng template DNA, 0.2 mM of each dNTP, 0.2 µM of each primer, 1.5 mM MgCl2, 1X PCR buffer (10 mM Tris–HCl, 50 mM KCl, pH 8.3) and 1U Taq polymerase. PCR amplification for ScOPX171400 and ScOPE161600 carried out in a thermocycler using initial denaturation at 94 °C for 5 min followed by 39 cycles at 94 °C for 30 s, 55 °C for 30 s, 72 °C for 1 min and a final extension at 72 °C for 5 min followed by rapid cooling at 4 °C. The same PCR conditions were used for AD60 except that extension step of the reiterative PCR cycles was carried out for 30 s. The details of markers used for validation is given in Table 331,32,33.
Analysis of PCR product
10 µl of each PCR product was mixed with 3 µl of 6X gel loading dye (0.25% bromophenol blue and 40% sucrose) and electrophoresis was carried out using 4% agarose gel prepared in 1X TAE buffer and ethidium bromide (0.5 µg/ml). The gels were run at a constant voltage of 120 V for 1.5 h. The gel was visualised by using Gel Documentation system (Labnet, ENDURO™ GDS, Aplegen). Presence of appropriate size product of the markers linked to powdery mildew resistant genes was recorded for 48 genotypes.
Results
Field screening
The analysis of variance revealed significant differences among 48 genotypes over environments for powdery mildew disease reaction (Table 4). The powdery mildew disease reaction at Palampur during winter season of 2017–18 and 2018–19 revealed that majority of the breeding lines showed resistant to moderately resistant reaction except SP-2 and SA-4 (Table 5; Supplementary Table-1). The SA-4 found to had moderately susceptible reaction during 2nd year. The check variety ‘Palam Sumool’ showed resistant reaction while ‘Pb-89’ was categorized as moderately resistant. Conversely, Palam Priya and Azad-P1 revealed moderately susceptible and susceptible disease reaction, respectively during both the years.
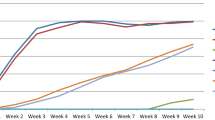
At Kukumseri during summer 2018, the disease was scored at two harvest stages i.e. during second picking and at crop maturity/last picking (Table 5). Azad-P1 witnessed susceptible reaction at early stages (Fig. 1) while 5 newly developed breeding lines were scored as 0 (HR) while 31 and 8 genotypes were rated as 1 (R) and 2 (MR) infection types, respectively at the same growth stage. Of the 36 lines with resistant reaction (Infection types-0 and 1), 17 lines retained infection type 1 till maturity, 19 lines rated as moderately resistant whereas, 5 lines viz., SP-2, SP-10, SP-19, SP-24 and SA-4 recorded moderately susceptible reaction. Checks Pb-89 and Palam Priya were categorized as moderately susceptible with infection type 3 whereas, Palam Sumool showed resistant reaction.
The reaction type of 48 pea lines over the environments (Table 5) exhibited differential response to powdery mildew under field conditions. Among these, 40 lines were resistant (0, 1 and 2 infection types) and 8 were susceptible (3 and 4 infection types). Majority of the newly developed breeding lines showed resistant reaction except SP-2, SP-7, SP-19, SP-24 and SA-4 while Palam Sumool was the only variety with resistant reaction amongst the four checks. The high yielding lines namely, SP-3, SP-6 and SP-22 were moderately resistant as they recorded similar disease reaction (infection type 2) at both the locations Kukumseri and Palampur and therefore, were categorized as moderately resistant.
In-vitro screening
All these genotypes were also screened in-vitro for powdery mildew disease reaction following detached leaf method and under naturally ventilated polyhouse conditions by raising them in pots. It was observed that out of the 48 lines evaluated under polyhouse conditions, 14, 26, 6 and 2 genotypes exhibited infection type 1 (R), 2 (MR), 3 (MS) and 4 (S), respectively (Table 6; Supplementary Table 2). Broadly, 40 genotypes were classified as resistant (infection types 1 and 2) and 8 as susceptible (infection types 3 and 4) under protected environment. Likewise, detached leaf method also revealed similar disease reactions for majority of the lines with 38 genotypes categorized as resistant (infection types 1 and 2) and 10 as susceptible (infection types 3 and 4). The Fig. 2 depicts the susceptible reaction of Azad-P1and Lincoln, moderately susceptible reaction of Pb-89, moderately resistant reaction of SP-3, SP-6, SP-22 and resistant reaction of Palam Sumool and SP-7.
Overall, 10 lines viz., SP7, SN-1, SN-6-1, SN-7-1, SN-2, SN-5-2, SN-6-2, SN-10, SN-21 and SP-28-1 showed resistant reaction (infection type 1) along with check Palam Sumool while 27 lines were identified as moderately resistant (infection type 2). In contrary, standard check Azad-P1 and line SP-19 were categorized as susceptible whereas, checks Pb-89 and Palam Priya along with 6 breeding lines namely, SP-2, SP-5, SP-10, SP-24, SA-4 and SP-12-1 revealed moderately susceptible reaction. The summary of disease reaction of various genotypes under field and in-vitro conditions is presented in Table 7.
Validation of resistance using molecular markers
For validation of powdery mildew resistance, three markers namely, AD60 and ScOPE-161600 linked to er1 and ScOPX-171400 linked to er2 were used. The er1 donor JI1559 and er2 donor JI 2480 were used for the validation. The amplification products for er1 and er2 linked markers are presented in the Fig. 3. The markers linked to ‘Er3’ were not used in this study because the segregation pattern in F2 generations of respective cross combinations from which advance breeding lines isolated were observed to be in the ratio of 3 (susceptible): 1 (resistant) indicating the presence of recessive gene in the donor parent (Palam Sumool).
er1 linked markers SCOPE-16-1600 and AD-60 and er2 linked marker SCOPX-17. Legend: M: DNA ladder, L: Lincoln, 2480: er1 harboring line, 1559: er2 harboring line, 1: SP-1, 2: SP-2, 3: SP-3, 4: SP-5, 5: SP-6, 6: SP-7, 7: SP-10, 8: SP-12, 9: SP-14, 10: SP-15, 11: SP-17, 12: SP-18, 13: SP-19, 14: SP-22, 15: SP-24, 16: SN-1, 17: SN-4, 18: 19: 20: 21: SN-11-1, 22: SN-15, 23: SN-2, 24: SN-5-2, 25: SN-6-2, 26: SN-7-2, 27: SN-8, 28: SN-10, 29: SN-11-2, 30: SN-13, 31: SN-19, 32: SN-21, 34: SA-1, 35: SA-4, 36: PSPP-8, 37: SP-12-1, 38: SP-12-2, 39: SP-15-1, 40: SP-23-1, 41: SP-28-1, 42: SN-8-2, 43: SN-3-1, 44: SN-9-2, 45: Palam priya, 46: Azad-P1, 47: Palam sumool, 48: Pb-89.
Two markers used for validation of er1, the marker ScOPE-161600 was not polymorphic between the er1 donor JI 1559 and parental genotypes and the amplicons were similar for the progenies as well (Fig. 3) therefore, the marker cannot be used to trace the presence of er1 in the crosses. The marker AD60 exhibited polymorphism between er1 donor JI1559 and parental genotypes of crosses Palam Sumool × Palam Priya (Cross-1) and Palam Sumool × Pb-89 (Cross-2). However, all the progeny plants of crosses inherited the non-JI1559 alleles of AD60. Therefore, neither the parental genotypes nor the progenies comprise the er1 gene of JI1559. This suggests that Palam Sumool and Pb-89 do not contain er1 gene of JI1559.
The er2 linked marker ScOPX-171700 was polymorphic between Palam Sumool and Palam Priya (Fig. 3) but the marker didn’t show polymorphism between er2 harboring line (JI2480). Surprisingly, the marker amplified identical amplicons in er2 harboring line JI2480 and susceptible genotypes Palam Priya and Azad-P1. However, none of the progenies of the cross-1 inherited ScOPX-171400 alleles from either er2 donor or Palam Priya and the amplicons of progenies match Palam Sumool.
Discussion
Powdery mildew disease significantly affects the quality and quantity of pea production and is considered as one of the economically important diseases of pea. The use of fungicide is available as an alternative to control the disease but genetic resistance is more favored being more sustainable and eco-friendlier. In the present investigation, four diverse parents selected on the basis of diverse phenological traits were involved in three inter-varietal crosses to isolate transgressive segregants in the recent years that have resulted in 44 progenies with desirable pod characteristics viz., long and dark green pods having high yield and resistance to powdery mildew disease. The powdery mildew resistant progenies were selected in epiphytotic and artificial conditions besides high yield to meet farmer’s preference.
Earlier, many resistant lines have been identified for resistance against powdery mildew using natural and artificial methods26,34,35,36. The validation of resistance in the cultivars can be achieved through in vitro screening by inoculating disease pathogen. However, the recessive nature of the powdery mildew resistance genes and difficulties associated with the handling of obligate pathogens like Erysiphe pisi, complicates the selection of resistant phenotypes. During field screening the progression of disease development after first harvest was drastic which may be due to warm days and cool nights favoring disease development. Thompson and Kelly reported that the disease incidence is severe when days are warm and dry and night temperatures are low37. Fondevilla and Rubiales also stated that powdery mildew is particularly damaging in late sowings or in late maturing varieties8. North-western Indian Himalayan region is natural hot spot for the manifestation of powdery mildew disease26. Accordingly at Kukumseri, the disease pressure was higher and disease progressed severely which might be attributed to favourable environmental conditions for disease development and more virulent race of pathogens suggesting it as the hot spot for the disease development (Fig. 1). Based on the high discrimination of the genotypes, Kukumseri was identified as the ideal environment for screening of the genotypes. The variable reaction of genotypes at different locations suggests the difference in virulence of pathogen, variable environmental conditions and variations in the genetic makeup of resistance genes in the accessions14,26,38. Due to the variable reaction of the genotypes, there was a need to screen the germplasm under artificial conditions using ample pathogen population.
The genotypes viz., SN-11-2, SP-18, PSPP-8, SP-15-1, SP-23-1, SN-22, SN-7-2 showed resistant reaction in field screening (Table 5) but they were moderately resistant during in-vitro screening (Table 6). Similarly, the disease reaction of SP-12-1 changed from MR to MS and SP-19 from MS to S. This change in the disease reaction of genotypes expressed the actual response of genotypes which showed that the artificial screening methods provide more reliable results as compared to the field screening. This is because in artificial screening favourable conditions are provided to the pathogen for its better perpetuation. The utility of detached leaf assay for screening garden pea for powdery mildew resistance was also mentioned39. Similarly, Rana et al.26 found 57 accessions out of 701 as resistant to powdery mildew under field conditions but only 14 showed resistance under in vitro conditions. From the results of natural and artificial screening, it can be concluded that high yielding lines viz., SP-3, SP-6 and SP-22 which showed stability for majority of the desirable traits across the environments, also showed moderate resistance both under field and in-vitro conditions (Table 7). Therefore, these lines can be exploited for commercial cultivation even in the hot spot areas.
The DNA markers linked to resistance genes provide an alternative to disease screening of powdery mildew resistance genes and provide an accurate measure as they are not affected by epistatic interactions. They can be used to confirm the presence of multiple resistance genes thereby, increasing efficiency of selection and reducing time span for the introgression of resistance genes. Three monogenic sources of powdery mildew resistance have been identified in pea germplasm, two recessive (er1 and er2) and one dominant (Er3)9.
For validation of powdery mildew resistance, three markers namely, AD60 and ScOPE-161600 linked to er1 and ScOPX-171400 linked to er2 were used (Fig. 3). The results suggested that the ScOPX-171400 is not a universally valid marker and cannot be used to confirm the presence of er2 gene in our parental genotypes and their progenies. The linked markers like ScOPX-171400 and ScOPE-161600 often suffer from the drawback that they show marker haplotypes identical to the resistant parents in many susceptible genotypes thereby limiting their utility in MAS. The main reason attributable to their limitation is that such markers are derived from the genomic regions which have no causative role in the manifestation of resistance and therefore, may be identical in resistant and susceptible genotypes. It is therefore, required that the gene-based markers derived from the functional polymorphisms within the resistance genes should be used to survey the germplasm for the target genes. However, the presence of resistance in many progenies of these crosses suggested that they have inherited some unknown powdery mildew resistance gene different than er1 from the powdery mildew resistant genotype Palam Sumool. The results also suggest that the lines showing resistance under field conditions may have some other genes or alleles for resistance and further confirmation by developing mapping populations with specific gene or gene combinations is needed26 and validation by involving a greater number of markers or developing new markers specific to the gene(s) for powdery mildew resistance.
Conclusion
Ten lines viz., SP7, SN-1, SN-6-1, SN-7-1, SN-2, SN-5-2, SN-6-2, SN-10, SN-21 and SP-28-1 along with Palam Sumool were identified as resistant along with 27 lines as moderately resistant. High yielding genotypes SP-3, SP-6 and SP-22 were moderately resistant. Molecular markers, used to survey the germplasm revealed that resistance in many of the breeding lines might be inherited from resistant parent Palam Sumool. The molecular markers used in the studies revealed the absence of er1 in the progenies while er2 could not be confirmed. Therefore, there is need to involve more number of markers to validate the specific gene providing resistance.
Data availability
The datasets generated during and/or analyzed during the current study are presented in the main manuscript and as additional supporting files. Further, any additional information can be obtained from the corresponding author on reasonable request. The advance breeding lines were isolated from diverse intervarietal crosses by the corresponding author and are in accordance with local legislations and comply with institutional/national/international guidelines.
References
Sharma, A., Sekhon, B. S., Sharma, S. & Kumar, R. Newly isolated inter-varietal garden pea (Pisum sativum L.) progenies (F7) under north western Himalayan conditions of India. Exp. Agri. 56, 76–87 (2020).
Kulaeva, O. A. et al. Pea marker database (PMD)—A new online database combining known pea (Pisum sativum L) gene-based markers. PLoS ONE 12, e018713. https://doi.org/10.1371/journal.pone.0186713 (2017).
Sepehya, S., Bhardwaj, S. K. & Dhiman, S. Quality attributes of garden pea (Pisum sativum L.) as influenced by Integrated Nutrient Management under mid hill conditions. J. Krishi Vigyan 3, 78–83 (2015).
Gari, A. T. Pea weevil (Bruchus pisorum L.) resistance and genetic diversity in field pea (Pisum sativum L.). Doctoral Thesis, Swedish University of Agricultural Sciences Alnarp (2015).
Rana, C. et al. Stability analysis of garden pea (Pisum sativum L.) genotypes under North Western Himalayas using joint regression analysis and GGE biplots. Genet. Resour. Crop Evol. 8, 999–1010 (2021).
Rungruangmaitree, R. & Jiraungkoorskul, W. Pea, Pisum sativum, and its anticancer activity. Pharmacogn. Rev. 11, 39–42 (2017).
Singh, N., Sharma, R. & Kayastha, R. Economic analysis of pea (Pisum sativum) in Himachal Pradesh. Econ. Aff. 65, 191–195. https://doi.org/10.46852/0424-2513.2.2020.9 (2020).
Fondevilla, S. & Rubiales, D. Powdery mildew control in pea: A review. Agron. Sustain. Dev. 32, 401–409 (2012).
Fondevilla, S., Rubiales, D., Moreno, M. T. & Torres, A. M. Identification and validation of RAPD and SCAR markers linked to the gene Er3 conferring resistance to Erysiphe pisi DC. in pea. Mol. Breed. 22, 193–200 (2008).
Srivastava, R. K., Mishra, S. K., Singh, A. K. & Mohapatra, T. Development of coupling phase SCAR marker linked to the powdery mildew resistance gene er1 in pea (Pisum sativum L.). Euphytica 186, 855–866 (2012).
Pavan, S. et al. Identification of a complete set of functional markers for the selection of er1 powdery mildew resistance in Pisum sativum L. Mol. Breed. 31, 247–253 (2013).
Harland, S. C. Inheritance of immunity to mildew in peruvian forms of Pisum sativum. Heredity 2, 263–269 (1948).
Leon, D. P., Checa, O. E. & Obando, P. A. Inheritance of resistance of two pea lines to powdery mildew. Agron. J. 112, 2466–2471 (2020).
Fondevilla, S., Torres, A. M., Moreno, M. T. & Rubiales, D. Identification of a new gene for resistance to powdery mildew in Pisum fulvum, a wild relative of pea. Breed. Sci. 57, 181–184 (2007).
Bobkov, S. V. & Selikhova, T. N. Introgession of powdery mildew resistance into cultural pea from wild accession of P. fulvum. IOP Conf. Ser. Earth Environ. Sci. 650, 012091 (2021).
Sokhi, S. S., Jhooty, J. S. & Bains, S. S. Resistance in pea against powdery mildew. Indian Phytopathol. 32, 571–574 (1979).
Kumar, H. & Singh, R. B. Genetic analysis of adult plant resistance to powdery mildew in pea (Pisum sativum L.). Euphytica 30, 147–151 (1981).
Vaid, A. & Tyagi, P. D. Genetics of powdery mildew resistance in pea. Euphytica 96, 203–206 (1997).
Sharma, B. The Pisum genus has only one recessive gene for powdery mildew resistance. Pisum Genet. 35, 22–27 (2003).
Janila, P. & Sharma, B. RAPD and SCAR markers for powdery mildew resistance gene er in pea. Plant Breed. 123, 271–274 (2004).
Ek, M. et al. Microsatellite markers for powdery mildew resistance in pea (Pisum sativum L.). Hereditas 142, 86–91 (2005).
Humphry, M., Reinstädler, A., Ivanov, S., Bisseling, T. & Panstruga, R. Durable broad-spectrum powdery mildew resistance in pea er1 plants is conferred by natural loss-of-function mutations in PsMLO1. Mol. Plant Pathol. 12, 866–878 (2011).
Pavan, S. et al. Pea powdery mildew er1 resistance is associated to loss-of-function mutations at a MLO homologous locus. Theor. Appl. Genet. 123, 1425–1431 (2011).
Sharma, A., Bhardwaj, A., Katoch, V. & Sharma, J. Assessment of genetic diversity of garden pea (Pisum sativum) as perspective to isolate horticulturally desirable transgressive segregants. Indian J. Agric. Sci. 83, 1334–1339 (2013).
Davidson, J. A., Krysinska-Kaczmarek, M., Kimber, R. B. E. & Ramsey, M. D. Screening field pea germplasm for resistance to downy mildew (Peronospora viciae) and powdery mildew (Erysiphe pisi). Australas. Plant Pathol. 33, 413–417 (2004).
Rana, J. C. et al. Screening of pea germplasm for resistance to powdery mildew. Euphytica 189, 271–282. https://doi.org/10.1007/s10681-012-0798-6 (2013).
Ikram, A. et al. Screening of resistant germplasm against powdery mildew of pea and its management through nutrients and plant activators. Asian J. Agric. Biol. 8(1), 85–91. https://doi.org/10.35495/ajab.2019.10.441 (2020).
Banyal, D. K. & Tyagi, P. D. Development of powdery mildew of pea in relation to different climatic condition in Himachal Pradesh. Plant Dis. Res. 13, 154–156 (1998).
Mains, E. B. & Dietz, S. M. Physiologic form of barley, Erysiphe graminis hordei Marchal. Phytopathology 20, 229–239 (1930).
Murray, M. G. & Thompson, W. F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 8(19), 4321–4326 (1980).
Katoch, V. et al. Molecular mapping of pea powdery mildew resistance gene er2 to pea linkage group III. Mol. Breed. 25, 229–237 (2010).
Tiwari, K. R., Penner, G. A. & Warkentin, T. D. Identification of coupling and repulsion phase RAPD markers for powdery mildew resistance gene er-1 in pea. Genome 41, 440–444 (1998).
Loridon, K. et al. Microsatellite marker polymorphism and mapping in pea (Pisum sativum L.). Theor. Appl. Genet. 111, 1022–1031 (2005).
Azmat, M. A. et al. Single recessive gene controls powdery mildew resistance in pea. Int. J. Veg. Sci. 16, 278–286 (2010).
Rehman, A. et al. Estimation of genetic diversity of pea germplasm against powdery mildew (Erysiphe pisi) disease and its chemosynthetic management. Pakistan J. Phytopathol. 26, 309–313 (2014).
Banyal, D. K., Chaudhary, J. & Singh, A. Evaluation of pea (Pisum sativum) germplasm for inheritance of resistance to powdery mildew (Erysiphe pisi). Indian Phytopathol. 68, 166–171 (2015).
Thompson, H. C. & Kelly, W. C. in Vegetable Crops. 5th, New Delhi, Tata McGraw-Hill. (1982).
Banyal, D. K., Singh, A. & Tyagi, P. D. Pathogenic variability in Erysiphe pisi causing pea powdery mildew. Himachal J. Agric. Res. 32, 87–92 (2005).
Singh, J., Dhall, R. K. & Aujla, I. S. Characterization of resistance response of garden pea (Pisum sativum L.) against powdery mildew (Erysiphe pisi DC.) in sub-tropical plains of India. SABRAO J. Breed. Genet. 47, 384–393 (2015).
Acknowledgements
The authors would like to thank Prof. R.C. Agrawal, National Director and Dr. Anuradha Agrawal, National Coordinator (CAAST), NAHEP, ICAR, New Delhi for constant encouragement to write and publish articles in journals with high impact factor. Also, thanks to Head, Department of Vegetable Science & Floriculture, Chaudhary Sarwan Kumar Himachal Pradesh Krishi Vishvavidyalaya, Palampur- 176062, Himachal Pradesh for providing all the facilities to conduct the research. Authors also want to extend their gratitude to Department of Agricultural Biotechnology, CSKHPKV Palampur for providing the lab facilities for the molecular work.
Author information
Authors and Affiliations
Contributions
C.R. conducted the experiments and analyzed data. A.S. conceptualize the research programme, conducted experiments, analyzed data and wrote the main manuscript text; B., P.S. and R.S.R. helped to frame the tables and review the manuscript; D.K.B. helped to conduct the in vitro screening and R.R. helped in validation using molecular markers.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rana, C., Sharma, A., Rathour, R. et al. In vivo and in vitro validation of powdery mildew resistance in garden pea genotypes. Sci Rep 13, 2243 (2023). https://doi.org/10.1038/s41598-023-28184-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28184-0
- Springer Nature Limited