Abstract
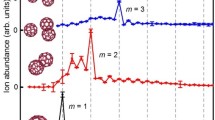
Copper nanoparticles are promising, low-cost candidates for the catalytic splitting of water and production of hydrogen gas. The present gas-phase study, based on the synthesis of copper-water complexes in ultracold helium nanodroplets followed by electron ionization, attempts to find evidence for dissociative water adsorption and H2 formation. Mass spectra show that H2O–Cu complexes containing dozens of copper and water molecules can be formed in the helium droplets. However, ions that would signal the production and escape of H2, such as (H2O)n−2(OH)2Cum+ or the isobaric (H2O)n−1OCum+, could not be detected. We do observe an interesting anomaly though: While the abundance of stoichiometric (H2O)nCum+ ions generally exceeds that of protonated or dehydrogenated ions, the trend is reversed for (H2O)OHCu2+ and (H2O)2OHCu2+; these ions are more abundant than (H2O)2Cu2+ and (H2O)3Cu2+, respectively. Moreover, (H2O)2OHCu2+ is much more abundant than other ions in the (H2O)n−1OHCu2+ series. A byproduct of our experiment is the observation of enhanced stability of He6Cu+, He12Cu+, He24Cu+, and He2Cu2+.
Graphical abstract

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Z.X. Yang, L.G. Xie, D.W. Ma, G.T. Wang, J. Phys. Chem. C 115, 6730 (2011)
J. Xiong, X.D. Wu, Q.J. Xue, J. Colloid Interface Sci. 390, 41 (2013)
N.K. Das, S. Ghosh, A. Priya, S. Datta, S. Mukherjee, J. Phys. Chem. C 119, 24657 (2015)
T. Kruk, K. Szczepanowicz, J. Stefanska, R.P. Socha, P. Warszynski, Colloids Surf. B-Biointerfaces 128, 17 (2015)
Z.K. He, J.W. Fu, B. Cheng, J.G. Yu, S.W. Cao, Appl. Catal. B-Environ. 205, 104 (2017)
Y.P. Liu et al., Adv. Mater. 29 (2017)
G. Hultquist, M.J. Graham, O. Kodra, S. Moisa, R. Liu, U. Bexell, J.L. Smialek, Corros. Sci. 95, 162 (2015)
A.J. Johansson, C. Lilja, T. Brinck, J. Chem. Phys. 135, 084709 (2011)
C.M. Lousada, A.J. Johansson, P.A. Korzhavyi, J. Phys. Chem. C 119, 14102 (2015)
K. Andersson, G. Ketteler, H. Bluhm, S. Yamamoto, H. Ogasawara, L.G.M. Pettersson, M. Salmeron, A. Nilsson, J. Am. Chem. Soc. 130, 2793 (2008)
P. Kappen, J.D. Grunwaldt, B.S. Hammershoi, L. Troger, B.S. Clausen, J. Catal. 198, 56 (2001)
M. Estrella et al., J. Phys. Chem. C 113, 14411 (2009)
S. Huseyinova, J. Blanco, F.G. Requejo, J.M. Ramallo-Lopez, M.C. Blanco, D. Buceta, M.A. Lopez-Quintela, J. Phys. Chem. C 120, 15902 (2016)
L. Chen et al., Phys. Chem. Chem. Phys. 12, 9845 (2010)
J.H. Stenlid, A.J. Johansson, T. Brinck, Phys. Chem. Chem. Phys. 16, 2452 (2014)
J.H. Stenlid, A.J. Johansson, L. Kloo, T. Brinck, J. Phys. Chem. C 120, 1977 (2016)
M.L. Jiang, Q. Zeng, T.T. Zhang, M.L. Yang, K.A. Jackson, J. Chem. Phys. 136, 104501 (2012)
P.M. Holland, A.W. Castleman, J. Chem. Phys. 76, 4195 (1982)
T.F. Magnera, D.E. David, D. Stulik, R.G. Orth, H.T. Jonkman, J. Michl, J. Am. Chem. Soc. 111, 5036 (1989)
N.F. Dalleska, K. Honma, L.S. Sunderlin, P.B. Armentrout, J. Am. Chem. Soc. 116, 3519 (1994)
P.J.E. Boussard, P.E.M. Siegbahn, M. Svensson, Chem. Phys. Lett. 231, 337 (1994)
A.M. El-Nahas, N. Tajima, K. Hirao, J. Mol. Struct. THEOCHEM 469, 201 (1999)
D. Feller, E.D. Glendening, W.A. de Jong, J. Chem. Phys. 110, 1475 (1999)
H.M. Lee, S.K. Min, E.C. Lee, J.H. Min, S. Odde, K.S. Kim, J. Chem. Phys. 122, 064314 (2005)
J.D. Herr, R.P. Steele, J. Phys. Chem. A 120, 10252 (2016)
T. Iino, K. Ohashi, Y. Mune, Y. Inokuchi, K. Judai, N. Nishi, H. Sekiya, Chem. Phys. Lett. 427, 24 (2006)
T. Iino, K. Ohashi, K. Inoue, K. Judai, N. Nishi, H. Sekiya, J. Chem. Phys. 126, 194302 (2007)
P.D. Carnegie, A.B. McCoy, M.A. Duncan, J. Phys. Chem. A 113, 4849 (2009)
B.M. Marsh, J. Zhou, E. Garand, J. Phys. Chem. A 118, 2063 (2014)
B.M. Marsh, J. Zhou, E. Garand, Phys. Chem. Chem. Phys. 17, 25786 (2015)
A.F. Sweeney, J.T. O’Brien, E.R. Williams, P.B. Armentrout, Int. J. Mass Spectrom. 378, 270 (2015)
A.F. Sweeney, P.B. Armentrout, J. Phys. Chem. A 118, 10210 (2014)
V.A. Mikhailov, P.E. Barran, A.J. Stace, Phys. Chem. Chem. Phys. 1, 3461 (1999)
G.E. Froudakis, M. Muhlhauser, S.C. Farantos, A. Sfounis, M. Velegrakis, Chem. Phys. 280, 43 (2002)
L.F. Gomez, E. Loginov, R. Sliter, A.F. Vilesov, J. Chem. Phys. 135, 154201 (2011)
H. Schöbel, P. Bartl, C. Leidlmair, S. Denifl, O. Echt, T.D. Märk, P. Scheier, Eur. Phys. J. D 63, 209 (2011)
S. Ralser, J. Postler, M. Harnisch, A.M. Ellis, P. Scheier, Int. J. Mass Spectrom. 379, 194 (2015)
A. Mauracher, M. Daxner, J. Postler, S.E. Huber, S. Denifl, P. Scheier, J.P. Toennies, J. Phys. Chem. Lett. 5, 2444 (2014)
O. Echt, D. Kreisle, M. Knapp, E. Recknagel, Chem. Phys. Lett. 108, 401 (1984)
S. Denifl et al., J. Chem. Phys. 132, 234307 (2010)
S. Krückeberg, L. Schweikhard, J. Ziegler, G. Dietrich, K. Lutzenkirchen, C. Walther, J. Chem. Phys. 114, 2955 (2001)
C.E. Klots et al., Z. Phys. D 21, 335 (1991)
K. Hansen, U. Näher, Phys. Rev. A 60, 1240 (1999)
L. An der Lan, P. Bartl, C. Leidlmair, R. Jochum, S. Denifl, O. Echt, P. Scheier, Chem. Eur. J. 18, 4411 (2012)
P. Bartl, C. Leidlmair, S. Denifl, P. Scheier, O. Echt, J. Phys. Chem. A 118, 8050 (2014)
T. Döppner, T. Diederich, S. Gode, A. Przystawik, J. Tiggesbäumker, K.H. Meiwes-Broer, J. Chem. Phys. 126, 244513 (2007)
M. Goulart et al., Phys. Chem. Chem. Phys. 20, 9554 (2018)
F. Tramonto, P. Salvestrini, M. Nava, D.E. Galli, J. Low Temp. Phys. 180, 29 (2015)
D. Prekas, C. Lüder, M. Velegrakis, J. Chem. Phys. 108, 4450 (1998)
A. Yousef, S. Shrestha, L.A. Viehland, E.P.F. Lee, B.R. Gray, V.L. Ayles, T.G. Wright, W.H. Breckenridge, J. Chem. Phys. 127, 154309 (2007)
X.F. Tong, C.L. Yang, M.S. Wang, X.G. Ma, D.H. Wang, J. Chem. Phys. 134, 024306 (2011)
X.Y. Li, X.Y. Cheng, X. Cao, Struct. Chem. 23, 1831 (2012)
Acknowledgments
Open Access funding provided by Austrian Science Fund (FWF).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contribution to the Topical Issue “Atomic Cluster Collisions”, edited by Alexey Verkhovtsev, Andrey V. Solov’yov, Germán Rojas-Lorenzo, and Jesús Rubayo Soneira.
Rights and permissions
Open Access This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Raggl, S., Gitzl, N., Martini, P. et al. Helium nanodroplets doped with copper and water. Eur. Phys. J. D 72, 130 (2018). https://doi.org/10.1140/epjd/e2018-90150-7
Received:
Revised:
Published:
DOI: https://doi.org/10.1140/epjd/e2018-90150-7