Abstract
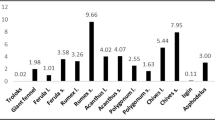
Traditional medicines are composed of herbal formulations and their active ingredients and constituents which play a crucial role in the treatment of various human ailments. Astragalus eremophilus and Melilotus indicus (L.) All. (syn. Melilotusparviflora Desf.) are used traditionally as antiperspirant, tonic, diuretic, laxative and narcotic agents. The current study was designed to investigate the Astragalus eremophilus and Melilotus indicus (L.) All. (syn. Melilotus parviflora Desf.) methanol extracts for their anti-oxidant, antibacterial and antifungal activities. Fine powder of A. eremophilus and M. parviflora was extracted with 70% methanol to get crude methanol extract. Extract was characterized for antioxidant, antibacterial and antifungal activities. Antioxidant activity of various concentrations (3 mg/ml, 1.5 mg/ ml, 0.75 mg/ml, and 0.38 mg/ml) of both plant extracts was analyzed using 2,2-Diphenyl-1-picrylhydrazyl (DPPH) free radical. Salmonella typhemorium, Klebsiellapneumoniae (gram-negatie)andStaphylococcus aureus, Enterococcus faecalis (gram-positive) bacterial strains were used for assessment of antibacterial activities. Antifungal activities of 7.5 mg/ml, 5.0 mg/ml, 2.5 mg/ml (A. eremophilus and M. parviflora) were conducted using Aspergillus niger, Aspergillus flavus, Aspergillus fumigatus and Candida albicons. At high concentration (3 mg/ml), all the tested fractions of A. eremophilus and M. parviflora methanol extracts showed potent antioxidant activities, ranging between 83.8 and 63.33%. Antibacterial activities revealed that A. eremophilus showed a maximum zone of inhibition (8.1 ±0.1) on Salmonella typheno-rium foliowed by Enterococcus faecalis (7.2 ± 0.1), Klebsellesa pneumonia (6.1 ± 0.6), and Staphylococcus aureus (5.1 ±0.4), and at highest concentration (7.5 mg/ml), however, maximum zone of inhibition of Melilotus parviflora was at 7.5 mg/ml foliowed by 5.0 mg/ml and 2.5 mg/ml against Klebsiella pneumonia, Staphylococcus aureus, Salmonella typhemorium and Enterococcus faecalis. Antifungal assessment of both plant extracts showed that the higher concentration (7.5 mg/ml) has significant inhibitory effect as compared to control. The results can lead to the conclusion that A. eremophilus and M. parviflora methanol extracts are indeed sources of potential therapeutic compounds against antibacterial, antifungal and free radical associated disorders.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Adeniji, I. T., Adio, A. F., Iroko, O. A., Kareem, A. A., Jegede, O. C., Kazeem-Ibrahim, F., Adewole, T. O., Adeosu, A. O. (2014) Pre-treatment of seeds of Annona squamosa (sugar apple) a non-timber forest product. Res. Plant Sci. 2, 50–52.
Akinyemi, K. O., Oladapo, O., Okwara, C. E., Ibe, C. C., Fasure, K. A. (2005) Screening of crude extracts of six medicinal plants used in South-West Nigerian unorthodox medicine for anti-methicillin resistant Staphylococcus aureus activity. BMC Compl. Alter. Med. 5, 6–13.
Bruneton, J. (1995) Pharmacognosy, phytochemistry, medicinal plants. Paris, Lavoisier Publishing.
Bocco, A., Marie, E. C., Hubert, R., Claudette, B. (1998) Antioxidant Activity and Phenolic Composition of Citrus Peel and Seed Extracts. J. Agric. FoodChem. 46, 2123–2129.
Brand-Williams, W., Cuvelier, M. E., Berset, C. (1995) Use of free radical method to evaluate antioxidant activity. Lebensm Wiss Technol. 28, 25–30.
Baba, A., Malik, S. A., Baba, A. S. (2015) Determination of total phenolic and flavonoid content, anti-microbial and antioxidant activity of a root extract of Arisaema jacquemonti Blume. J. Taib. Univ. Sci. 9, 449–154.
Burt, S. (2004) Essential oils, their antibacterial properties and potential applications in foods a review. Int. J. Food. Micro. 94, 223–253.
Bylka, W., Goslinska, O. (2001) Determination of isocytisoside andantimicrobial activity of etha-nolic extract from Aquilegia vulgaris. J. Acta. Polo. Pharm. 58, 241–243.
Chhetri, H. R., Yogol, N. S., Sherchan, J., Mansoor, K. C. A., Thapa, P. S. (2008) Phytochemical and antimicrobial evaluations of some medicinal plants of Nepal. Kathmandu University J. Sci. Eng. Tech. 1, 49–54.
Chaudhary, L. B., Rana, T. S., Anand, K. K. (2008) Current status of the systematics of Astragalus L. (Fabaceae) with special reference to the Himalayan species in India. Taiwania 53, 338–355.
Choudhary, M. I., Dur-e-Shahwar, Z., Perveen, A., Atta-ur-Rahman, Ali, I. (1995) Antifungal steroidal lactones from Withania coagulance. Phytochemistry 40, 1243–1246.
Doughari, J. H. (2006) Antimicrobial activity of Tamarindus indica Linn. Trop. J. Pharm. Res. 5, 597–603.
De, M., De, A. K., Banerjee, A. B. (1999) Antimicrobial screening of some Indian spices. Phytother. Res. 13, 616–618.
De Quan, Y. U. (1999) Recent works on anti-tumor constituent from Annonaceae plants in China. Pure Appl. Chem. 71, 1119–1122.
De Luca, V., (2011) Monoterpenoid indole alkaloid biosynthesis. In: Ashihara, H., Crozier, A., Komamine, A. (eds), Plant Metabolism and Biotechnology John Wiley and Sons Ltd., Chichester, pp. 263–291.
Duenas, M., Hernandez, T. Estrella, I. (2006) Assessment of in vitro antioxidant capacity of the seed coat and the cotyledon of legumes in relation to their phenolic contents. FoodChem. 98, 95–103.
Fellenberg, M. A., Espinoza, A., Pena, I., Alarcón, J. (2011) Antioxidant and Bacteriostatic Effects of the Addition of Extract of Quillay Polyphenols (Quillaja saponaria) in the Marinade of Broiler Chicken. Brazilian J. Poultry Sci. 1, 9–11.
Ganapaty, S., Steve, T. R., Venkata, R. K., Neeharika, V. (2001) Areview of phytochemical studies of Rauwolfia species. Indian Drugs 38, 601–612.
Holtz, C. (2007) Global health care: Issues and policies. Jones & Bartlett Publishers, Sudbury, MA.
Hyat, M. Q., Khan, M. A., Ashraf M., Jabeen, S. (2009) Ethnobotany of the Genus Artemisia L. (Asteraceae) in Pakistan. Ethnobot. Res. Appl. 7, 147–162.
Imperato, R. J. (1981) Modern and Traditional Medicine: The Case of Mali. Annals Intl. Med 95, 650–651.
Izzotti, A., Sacca, S. C., Longobardi, M., Cartiglia, C. (2010) Mitochondrial damage in the trabecular meshwork of patients with glaucoma. Arch. Ophthalmol. 128, 724–730.
Kalimuthu, K., Vrjayakumar, S., Senthilkumar, R. (2010) Antimicrobial Activity of the Biodiesel Plant, Jatropha Curcas L. Intl. J. Pharma. Biosci. 1, 1–5.
Khan, A. M., Qureshi, A. M., Gillani, S. A., Ullah, R. (2011) Antimicrobial activity of selected medicinal plants of Margalla Hills, Islamabad Pakistan. J. Med. Plants Res. 5, 4665–1670.
Kavanagh, R. (1963) Analytical microbiology. Academie Press London.
Kilani, S., Sghaier, M. E-., Limem, I., Bouhlel, I., Boubaker, J., Bhouri, W., Skandrani, I., Neffatti, A., Ammarb, R. A., Dijoux-Franca, R. A., Ghediram, K., Chekir-Ghedira, L. (2008) In vitro evaluation of antibacterial, antioxidant, cytotoxic and apoptotic activities of the tubers in fusion and extracts of Cyperus rotundus. Bioresource Technol. 99, 9004–9008.
Kabuki, T., Nakajima, H., Arai, M. (2000) Characterization of novel antimicrobial compounds from mango (Mangifera indica L.) Kernel Seeds. Food Chem. 71, 61–66.
Marjorie, M. C. (1999) Plant products as antimicrobial agents. Clinical Microbiol. Rev. 12, 564–582.
Maisuthisakul, R. (2008) Antiradical scavenging activity and polyphenolic compounds extracted from Thai mango seed kernels. Asian J. Food Agric. Ind. 1, 87–96.
Moerman, D. (1998) Native American ethnobotany Timber Press. Oregon.
Magaldi, S., Mata-Essayag, S., Hartung, C. (2004) Well diffusion for antifungal susceptibility testing. IntlJ. Infec. Dis. 8, 39–15.
Mohanta, T. K., Patra, J. K., Rath, S. K., Pal, D. K., Thatoi, H. N. (2007) Evaluation of antimicrobial activity and phytochemical screening of oils and nuts of Semicarpus anacardium L.f. Sci. Res. Essay 2, 486–190.
Nadkarni’s, K. M. (1976) Indian Materia Medica, Volume 1, Popular Prakashan, Mumbai.
Patwardhan, B., Hooper, M. (1992) Ayurveda and future drug development. IntlJ. Alter. Compl. Med 10, 9–11.
Robbers, J., Speedie, M., Tyler, V. (1996) Pharmacognosy andpharmacobiotechnology. Williams and Wilkins, Baltimore.
Sucher, N. J., Carles, M. C. (2008) Genome-Based Approaches to the Authentication of Medicinal Plants. Planta Medica 74, 603–623.
Slik, I. W. F., Poulsen, A. D., Ashton, R. S., Cannon, C. H., Eichhorn, K. A. O., Kartawinata, K., Lanniari, I., Nagamasu, H., Nakagawa, M., Van Nieuwstadt, M. G. L., Payne, J. (2003) A. floristic analysis of the lowland dipterocarp forests of Borneo. Pak. J. Biol. Sci. 9, 2600–2605.
Stepanovic, S., Cirkovic, I., Mijac, V., Svabic-Vlahovic, M. (2003) Influence of the incubation tem-perature, atmosphere and dynamic conditions on biofilm formation by Salmonella spp. Food Microbiol. 20, 339–343.
Tewari, D. N. (2000) Report of the taskforce on conservation and sustainable use of medicinal plants. Planning commission. 1–18.
Valgas, C., De Souza, S. M., Smania, E. F. A. (2007) Screening methods to determine antibacterial activity of natural products. Brazilian J. Microbiol. 38, 369–380.
Villa F., Pitts B., Stewart R. S., Giussani B., Roncoroni S., Albanese D. (2011) Efficacy of zosteric acid sodium salt on the yeast biofilm model Candida albicans. Microbiol. Ecol. 62, 584–598.
WHO. Traditional medicine strategy 2003-2005. Geneva, World Health Organization, 2003. (WHO/ EDM/TRM/2003.1).
World Health Organization (WHO). (1978) The promotion and development of traditional medicine. Technical report series pp. 622.
Xu, L., Sun, N., Kong, J. (1992) Alkaloids of Annona reticulata L. Zhonqquo Zhong Yao Zazhi 17, 295–296.
Yang, X., Yang, L., Zheng, H. (2010) Hypolipidemic and antioxidant effects of mulberry (Morus alba L.) fruit in hyperlipidemia rats. Food Chem. Toxicol. 48, 2374–2379.
Yigit, D., Yigit, N., Mavi, A. (2009) Antioxidant and antimicrobial activities of bitter and sweet apri-cot (Prunus armeniaca L.) kernels. Brazilian J. Med. Biol. Res. 42, 346–352.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Khan, M.N., Ahmed, M., Khan, M.W. et al. In vitro Pharmacological Effects of Astragalus eremophilus and Melilotus parviflora. BIOLOGIA FUTURA 69, 411–422 (2018). https://doi.org/10.1556/018.69.2018.4.4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.69.2018.4.4