Abstract
Triglycerides are critical lipids as they provide an energy source that is both compact and efficient. Due to its hydrophobic nature triglyceride molecules can pack together densely and so be stored in adipose tissue. To be transported in the aqueous medium of plasma, triglycerides have to be incorporated into lipoprotein particles along with other components such as cholesterol, phospholipid and associated structural and regulatory apolipoproteins. Here we discuss the physiology of normal triglyceride metabolism, and how impaired metabolism induces hypertriglyceridemia and its pathogenic consequences including atherosclerosis. We also discuss established and novel therapies to reduce triglyceride-rich lipoproteins.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Apolipoprotein B
- Chylomicrons
- Hypertriglyceridemia
- Metabolism
- Therapies
- Triglyceride-rich lipoproteins (TRL)
- Very low density lipoproteins (VLDL)
1 Introduction
Interest in triglyceride-rich lipoproteins (TRLs) has for long been rather low, but recent results demonstrating that TRLs are causally associated with atherosclerotic cardiovascular disease (ASCVD) have generated major interest in these lipoproteins. Hypertriglyceridemia is quite common today and approximately 25% of US adults are estimated to have hypertriglyceridemia (triglyceride [TG] level ≥ 1.7 mmol/L]). TRLs are synthesized in the liver as very low-density lipoproteins (VLDL) and in the intestine as chylomicrons. During lipolysis TRLs are converted to atherogenic cholesterol-ester enriched lipoprotein remnant particles. Dysregulation of the normal metabolism of TRLs leads to excess formation of these atherogenic lipoprotein remnant particles (Chapman et al. 2011; Nordestgaard and Varbo 2014; Boren et al. 2014; Dallinga-Thie et al. 2016). Since humans are postprandial most of the day, we continuously generate atherogenic remnant particles. Consequently, the continuous generation of remnants after each meal may be an important causal risk factor for the development of atherosclerosis. Genetic studies have also identified key regulators of the metabolism of TRLs and major emphasis is now directed at evaluating their potential as novel candidate targets for dyslipidemia and premature ASCVD risk (Dallinga-Thie et al. 2016). Here we discuss how TRLs are synthesized and metabolized.
2 Hepatic Formation and Secretion of VLDL
The assembly of VLDL is a complex process and involves a stepwise lipidation of apoB100, the principal apolipoprotein on VLDL, in the liver (Olofsson et al. 2000; Olofsson and Boren 2005). ApoB100 is a large protein consisting of one globular N-terminal structure, two domains of amphipathic β-sheets and two domains of amphipathic α-helices (Segrest et al. 2001) ApoB differs from other apolipoproteins in that it is highly hydrophobic. Therefore, it cannot equilibrate between different lipoproteins but remains bound to the particle on which it was secreted into plasma. Thus, every VLDL particle contains one molecule of apoB100. This is generally thought to be explained by the presence of antiparallel β-sheets with a width of approximately 30 Å, which form very strong lipid-binding structures (Segrest et al. 2001).
The lipidation cascade starts with a cotranslational transfer of triglycerides to nascent apoB polypeptides during the assembly of nascent VLDL mediated by the microsomal triglyceride-transfer protein (MTP) in the rough endoplasmic reticulum (ER) (Fig. 1) (Boren et al. 1992; Rustaeus et al. 1998). The critical role of MTP in VLDL assembly is demonstrated by the rare, autosomal-recessive disorder abetalipoproteinemia. The disorder results from mutations in the gene encoding the large subunit of MTP and is characterized by nearly a complete absence of apoB-containing lipoproteins including VLDL. To date, over different 30 mutations in the MTP gene have been described for ABL (Lee and Hegele 2014). Absence of MTP leads to premature proteosomal degradation of nascent apoB and therefore absence of VLDL and chylomicron production. The patients are characterized by hypocholesterolemia and the absence of apoB-containing lipoproteins (Paquette et al. 2016).
Assembly and secretion of apoB100-containing lipoproteins. ApoB is synthesized and translocated into the lumen of the endoplasmic reticulum (ER) (1). The growing nascent apoB polypeptide is cotranslationally lipidated by the lipid transfer protein MTTP to form a partially lipidated pre-VLDL particle (2). If apoB fails to be lipidated and acquire a correct protein folding (3), it is sorted to posttranslational degradation (4). The triglyceride-poor pre-VLDL particle exits the ER by Sar1/CopII vesicles that bud off (6) from specific sites on the ER membrane (Gusarova et al. 2003). The vesicles fuse to form the ER Golgi intermediate compartment (ERGIC) (7), which then fuses with the cis-Golgi (8). The triglyceride-poor particles are either secreted as smaller VLDL2 particles (9) or further lipidated (10) to form mature triglyceride-rich VLDL1 particles, which are then secreted (11). The formation of triglyceride-rich VLDL1 particles is highly dependent on the presence of triglyceride-containing cytosolic lipid droplets. These lipid droplets are formed as small primordial droplets from microsomal membranes (12) and increase in size by fusion (13). The triglycerides within the droplets undergo lipolysis and are re-esterified (14) before they lipidate the triglyceride-poor VLDL to form triglyceride-rich VLDL. Hepatic triglycerides originate from influx of free fatty acids, hepatic de novo lipogenesis (DNL), or hepatic uptake of lipoprotein particles
The cotranslational lipidation stabilizes the nascent apoB polypeptide and results in the formation of a nascent pre-VLDL lipoprotein particle (Bostrom et al. 1988). The immature pre-VLDL undergoes further lipidation in the secretory pathway, forming a triglyceride-poor VLDL particle (Stillemark-Billton et al. 2005). This particle can either be secreted from the liver as a smaller VLDL particle (i.e., VLDL2) or undergo further lipidation to form a larger triglyceride-rich (i.e., VLDL1) (Stillemark-Billton et al. 2005; Stillemark et al. 2000). The lipidation cascade is still not fully understood, but has been shown to involve several proteins including the GTP-binding protein ADP-ribosylation factor 1 (ARF-1) (Asp et al. 2005).
The conversion of smaller triglyceride-poor VLDL particles to large triglyceride-rich VLDL likely involves the fusion of cytoplasmic lipid droplets to the smaller VLDL particle. Thus, this bulk addition of triglycerides differs from the initial stepwise addition of triglycerides. The formation of the large mature VLDL particles is therefore dependent on the presence of cytosolic lipid droplets (Wiggins and Gibbons 1992; Salter et al. 1998; Gibbons et al. 2000). Therefore, it’s not surprising that hepatic accumulation of triglycerides, non-alcoholic fatty liver disease (NAFLD), is linked to oversecretion of large VLDL1 particles (Adiels et al. 2006a, b). However, not all forms of fatty liver disease are linked to increased hepatic secretion of VLDL1, indicating that the hepatic stores of triglycerides in some genetic forms of NAFLD, like PNPLA3, are not accessible for VLDL formation. However, the molecular mechanisms are still unclear. Interestingly, the amounts of triglycerides that are added to triglyceride-rich poor VLDL seem to be constant. Thus, subjects with type 2 diabetes secrete more – not larger – VLDL1 particles than non-diabetic controls (Adiels et al. 2005, 2006a, b). Thus, bulk addition of triglycerides from the cytoplasmic lipid droplets seems to be a highly regulated process.
3 Regulators of Hepatic VLDL Secretion
Hepatic triglyceride accumulation stimulates hepatic VLDL1 secretion, and the sources for liver fat are: (1) plasma fatty acids generated by lipolysis of the peripheral adipose tissue; (2) fatty acids synthesized in the liver from carbohydrates through hepatic de novo lipogenesis (DNL); (3) fatty acids that come from the diet; and (4) hepatic uptake of triglyceride-rich lipoproteins (TRLs) (Parks and Hellerstein 2006; Barrows and Parks 2006). Most of the hepatic triglycerides originate from circulating fatty acids, since the hepatic uptake of fatty acids is not regulated. Thus, increased levels of circulating fatty acids are directly connected to increased hepatic uptake of fatty acids (Tamura and Shimomura 2005). Lipolysis of adipose tissue (in particular the visceral adipose tissue) is the principal contributor (approx. 80%) of the plasma NEFA pool (Tamura and Shimomura 2005). It is therefore not surprising that visceral adiposity is strongly associated with NAFLD and oversecretion of VLDL1 particles (Parks and Hellerstein 2006; Barrows and Parks 2006; Farquhar et al. 1965; Parks et al. 1999; Havel 1961; Donnelly et al. 2005).
Normally the hepatic DNL plays a minor role (<5%) (Barrows and Parks 2006), but in conditions of increased plasma glucose and hyperinsulinemia it has been shown to generate ≈25% of liver triglycerides (Donnelly et al. 2005). The explanation is that glucose is the substrate for hepatic DNL and that hyperinsulinemia is linked to increased expression of factors needed for hepatic DNL such as SREBP1-c (Browning and Horton 2004; Shimomura et al. 1999), the carbohydrate response element–binding protein (ChREBP) (Koo et al. 2001), and PPARγ (Edvardsson et al. 1999; Chao et al. 2000; Westerbacka et al. 2007).
In addition, there is evidence that VLDL1 and VLDL2 are regulated independently. Ethanol overconsumption seems to stimulate VLDL1 production in humans (Fielding et al. 2000), whereas endogenous cholesterol synthesis correlates with VLDL2-apoB but not VLDL1-apoB production (Prinsen et al. 2003). This finding may explain why VLDL2, but not VLDL1, is increased in patients with increased plasma cholesterol such as moderate hypercholesterolemia (Gaw et al. 1995) and familial hypercholesterolemia (James et al. 1989).
4 Synthesis and Secretion of Chylomicrons from the Intestine
Chylomicrons are synthesized in the enterocytes of the small intestine and each chylomicron contains one molecule of apoB48. The apoB48 protein corresponds exactly to the N-terminal 48% of apoB100. The explanation for this is that both proteins are encoded by the same gene. The mRNA for apoB48 is generated from the apoB100 mRNA by a posttranscriptional editing process during which a deamination of a cytidine (at nucleotide 6,666) to a uridine converts a glutamine codon to a stop codon. The mechanism has been extensively reviewed (Davidson and Shelness 2000; Anant and Davidson 2001; Wang et al. 2003). The assembly of chylomicrons is a highly complex multistep process, and less is still known about chylomicron assembly than VLDL assembly (Xiao et al. 2019; Hussain et al. 2005). However, it is known that in addition to MTTP, intestinal assembly of chylomicron requires Sar1 GTPase, which is critical for the intracellular transport of apoB48-containing particles from ER to the Golgi (Julve et al. 2016).
The newly synthesized chylomicrons carrying dietary lipids and fat-soluble vitamins are secreted through lacteal endothelial gaps that are present in the postprandial phase into the venous system blood system through the lymphatic system. Thus, unlike other nutrients dietary lipids bypass the hepatic portal system.
Over the last years several surprising findings have been made (Lambert and Parks 2012). First, studies have demonstrated that the intestine stores triglycerides and that lipids secreted after a meal may have been consumed in an earlier meal (Mattes 2002; Robertson et al. 2002; Chavez-Jauregui et al. 2010). This may explain the early rise in postprandial plasma triglycerides since the intestine does not have to absorb dietary lipids and then form chylomicrons, but instead start secreting stored triglycerides in chylomicrons. We have also realized that the release of chylomicrons is linked to a taste–gut–brain axis (Khan and Besnard 2009) Interestingly, chylomicrons can be secreted when fat (Mattes 2009) or glucose (Robertson et al. 2003) is merely tasted but not consumed. Lastly, contrary to what was believed, recent studies have shown that apoB48-containing particles are secreted not only as chylomicrons but also as less triglyceride-rich lipoprotein particles (isolated in the VLDL density range) both in the fasting state and postprandially (Bjornson et al. 2019a, b).
5 Disorders of the Synthesis of TRLs
Abetalipoproteinemia (ABL) (also known as the Bassen–Kornzweig syndrome) is a rare autosomal-recessive disease that is characterized by very low plasma concentrations of TG and cholesterol (under 30 mg/dL) and undetectable levels of LDL and apoB. The rare recessive genetic disease is caused by loss-of-function mutations in the MTTP gene encoding for the microsomal triglyceride-transfer protein (MTP).
Clinic: Mutations in MTP leads to impairment of the formation of triglyceride-rich VLDL and chylomicrons. Patients with ABL (and compound heterozygous and homozygous FHBL) have therefore very low plasma total cholesterol and generally low plasma triglycerides. LDL-C when measured by direct methods, and apoB, will be absent or their concentrations will be very low. Patients may display neurological, hematological (acanthocytosis on peripheral blood smear and anemia), and gastro-intestinal symptoms due to deficiency in lipophilic vitamins and fat malabsorption (Paquette et al. 2016). The deficiency of vitamin E could lead to severe neurological disorders including spinocerebellar degeneration with ataxia and retinitis pigmentosa (Welty 2014). In addition, the impaired secretion of hepatic triglycerides may lead to hepatic steatosis (Welty 2014). The clinical phenotype and severity differs as the type and combination of MTTP mutations influence the clinical phenotype and treatment response (Paquette et al. 2016). Subjects who carry a single MTTP mutation may have normal plasma lipid levels or may have LDL-cholesterol and apoB concentrations similar to those seen in heterozygous familial hypobetalipoproteinemia (Lee and Hegele 2014; Paquette et al. 2016).
Treatment: Early diagnosis and treatment is important to prevent neurologic complications of this disease. Reversal of existing neurologic disease can also be achieved. Treatment involves a low-fat diet, supplementation with essential fatty acids and high oral doses of fat-soluble vitamins, vitamins A and E (Paquette et al. 2016; Welty 2014; Linton et al. 1993). High dose of oral fat-soluble vitamins bypasses the chylomicron pathway, and vitamins are carried via the portal circulation (Lee and Hegele 2014; Paquette et al. 2016).
Chylomicron retention disease (CRD). In addition to MTP, chylomicron formation requires Sar1 GTPase, one of the subunits of the coat protein (COPII) complex, which is critical for the vesicular transport of apoB-48-containing particles from endoplasmic reticulum to the Golgi (Julve et al. 2016). Loss-of-function mutations in SAR1B, the gene encoding Sar1 homolog B GTPase causes CRD (also known as Anderson disease) (Julve et al. 2016), a rare autosomal-recessive disorder characterized by an intestinal defect in lipid transport due to a failure of chylomicron formation in enterocytes (Julve et al. 2016).
Clinic: The failure to synthesize chylomicrons results in severe malabsorption with steatorrhea, fat-soluble vitamin deficiency, low blood cholesterol levels, and failure to thrive in infancy (Julve et al. 2016).
Treatment: Same as ABL.
Familial hypobetalipoproteinemia (FHBL) is an autosomal codominant disorder characterized by apoB <5th percentile and LDL-cholesterol usually between 20 and 50 mg/dL (Welty 2014; Linton et al. 1993). Over 60 different mutations in apoB producing truncated forms of apoB, ranging from apoB to apoB89, have been reported (Welty 2014; Linton et al. 1993). These truncated forms of apoB are named according to the percent length of the native apoB100 molecule. Truncated forms of apoB shorter than apoB30 are seldom detectable in human plasma as lipoproteins since these truncated proteins undergo intracellular degradation. Although one allele if affected only in heterozygous FBHL, the plasma levels are normally closer to one quarter to one third of normal, due to low hepatic secretion of the truncated forms of apoB combined with decreased production and increased clearance of VLDL and LDL produced by the normal allele (Welty et al. 1997; Elias et al. 1999; Aguilar-Salinas et al. 1995; Parhofer et al. 1996).
Clinic: Heterozygous FHBL is often asymptomatic and not diagnosed unless a lipid profile is obtained. In contrast, the clinical presentation of homozygous FHBL is similar to ABL. Early diagnosis of homozygous FHBL is therefore important. As the hepatic secretion of triglyceride-rich lipoproteins is impaired, FHBL has been shown to associate with hepatic steatosis and mild elevation of liver enzymes (Welty 2014). In 32 FHBL subjects, the hepatic fat content was increased to 14.0 ± 12.0% compared to 5.2 ± 5.9%, respectively, for 33 controls matched for age, sex, and indices of adiposity (Tanoli et al. 2004).
Treatment: For homozygous FHBL treatment involves a low-fat diet, supplementation with essential fatty acids and high oral doses of fat-soluble vitamins, vitamins A and E (Welty 2014; Linton et al. 1993).
6 Metabolism of Triglyceride-Rich Lipoproteins
After secretion of chylomicrons and VLDL, the lipoproteins are exposed to lipoprotein lipase (LPL) on the capillary endothelial cells within adipose tissue, skeletal muscle, and the heart, leading to hydrolyzation of the triglycerides, allowing the delivery of non-esterified free fatty acids (NEFA) to adipose tissue, skeletal muscle and the heart.
As the triglycerides are removed from the particles, they shrink and their density increases (Goldberg 1996); chylomicrons become chylomicron remnants, and large triglyceride-rich VLDL1 particles become smaller VLDL2 and subsequently intermediate density lipoproteins (IDL). The IDL particles can be further hydrolyzed to LDL particles by action of the hepatic lipase (HL). Since all human TRLs contain a substantial amount of cholesterol esters, hydrolysis of triglycerides leads to enrichment of cholesterol esters. Consequently, TRL remnants are enriched in cholesteryl esters (Dallinga-Thie et al. 2010).
Although roughly 80% of the increase in postprandial plasma triglycerides consists of chylomicrons and their remnants (Cohn et al. 1993), the majority of particles (around 80%) comprise of liver-derived VLDL and their remnants (Karpe et al. 1995; Schneeman et al. 1993). Also, the area under the curve for apoB100 is 10-fold higher than that of apoB48 (Vakkilainen et al. 2002), and the production rate of apoB100 is 15–20 times higher than that of apoB48 (Lichtenstein et al. 1992; Welty et al. 1999). We have earlier shown that chylomicrons and VLDL particles are not cleared equally by the lipoprotein lipase pathway, and that chylomicrons seem to be the preferred substrate (Adiels et al. 2012). Therefore, the major contribution to an atherogenic lipoprotein profile from chylomicrons is likely its interference with apoB100 catabolism.
ApoB-containing particles with a diameter of about 70 nm or smaller can penetrate the arterial endothelial layer, and subsequently become retained in the artery wall. Thus, cholesterol-rich remnants (i.e., both chylomicron remnants and VLDL remnants) can lead to cholesterol deposition in growing lesions, accelerated atherosclerosis, and enhanced CVD risk in a similar manner as LDL.
Genetic deficiency of LPL leads to the rare autosomal-recessive disorder familial LPL deficiency. These patients usually display milky plasma (accumulation of chylomicrons) and very severe hypertriglyceridemia with episodes of abdominal pain (pancreatitis), eruptive cutaneous xanthomata, and hepatosplenomegaly.
7 Deciphering the Pathogenesis of Hypertriglyceridemia
Insulin resistance and hypertriglyceridemia are associated with an atherogenic dyslipidemia characterized by prolonged postprandial hyperlipidemia, accumulation of small dense LDL (sdLDL) and low HDL cholesterol. The mechanism that leads to the formation of sdLDL is well clarified; the cholesteryl ester transfer protein (CETP) transfers triglycerides from VLDL1 to LDL. This results in formation of triglyceride-rich LDL. These lipoprotein particles are the preferred substrate for hepatic lipase (HL) that depletes triglycerides from the triglyceride-rich LDL. As large triglyceride-rich VLDL1 particles are the substrate for CETP, accumulation of TRLs is a prerequisite for sdLDL formation (Adiels et al. 2006b; Packard 2003; Georgieva et al. 2004). The enzymes also act on HDL, resulting in the formation of sdHDL that are efficiently removed from circulation. The combined action of CETP and HL thus results in the formation of sdLDL and low HDL cholesterol (Verges 2005; Taskinen 2003). Several studies indicate that increased sdLDL is associated with increased CVD risk (Austin et al. 1990; Lamarche et al. 1997; Gardner et al. 1996; Vakkilainen et al. 2003). However, it is still unclear if sdLDL is a marker of an atherogenic dyslipidemia or causatively linked to the increased CVD risk (Sacks and Campos 2003).
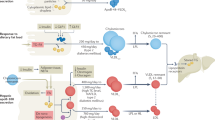
To elucidate the pathophysiology of the hypertriglyceridemia in obese subjects we have performed a series of kinetic studies with stable isotopes. These studies have shown that the impaired lipid metabolism is caused by dual mechanisms: increased secretion of triglyceride-rich VLDL1 from the liver and delayed clearance of TRLs from the circulation (Borén et al. 2015; Taskinen et al. 2011). This is illustrated in Fig. 2 by two pathways: a synthesis pathway and a clearance pathway. Interestingly, the synthesis pathway explained only 20% of the variation of plasma triglycerides (Borén et al. 2015). In contrast, the clearance pathway explained 50% of the variation in the total plasma triglycerides. Thus, the impaired catabolism of VLDL1-triglycerides is the most important determinant of the plasma triglyceride concentration in subjects with abdominal obesity and dyslipidemia.
Predictors of VLDL1-triglyceride secretion and catabolism. Liver fat content (P < 0.01) and total fat mass (P < 0.05) are important independent predictors of VLDL1-TG secretion rate (SR). The plasma concentration of apoC-III correlated strongly with plasma TG and the fractional catabolism of VLDL1-TG. VLDL1-TG kinetics explained 76% of the variation in the total plasma triglycerides. Of these, ≈20% was explained by the secretion pathway, whereas ≈50% was explained by the clearance pathway. Thus, indices of catabolism were stronger predictors of plasma triglycerides than parameters of secretion. The associations between liver fat and fat mass vs plasma TG (dotted lines) are likely secondary and mediated via VLDL1 SR. Likewise, the direct effect of apoC-III on plasma TG (dotted line) is likely explained by effect(s) of apoC-III beyond lipoprotein lipase-independent pathways of triglyceride metabolism (Borén et al. 2015)
The synthesis pathway includes liver fat and total fat mass as these remained independent predictors of VLDL1-triglyceride secretion rate in a stepwise multivariable regression analysis (Borén et al. 2015). Increased liver fat is linked to impaired regulation of VLDL production and a continuous oversecretion of VLDL1 (Adiels et al. 2006b; Poulsen et al. 2016) (Fig. 2), and adipose tissue is the major source of fatty acids in the NEFA pool that determines the hepatic uptake of fatty acids. In the clearance pathway, the plasma concentration of apoC-III is shown to correlate strongly with plasma triglycerides and clearance of VLDL1-triglycerides.
8 Regulation of Hydrolysis of TRLs and the LPL Pathway
The clearance of triglycerides is directly linked to the lipolysis of TRLs by LPL (Ginsberg et al. 1986). Full activity of LPL requires the interaction with the transport protein glycosylphosphatidylinositol anchored high density lipoprotein binding protein 1 (GPI-HBP1) and the lipase maturation factor 1 (LMF 1) present at the surface of capillaries (Sandesara et al. 2019). The LPL activity is modulated by several regulators including apoC-I, apoC-II, apoC-III, angiopoietin-like 3 (ANGPTL3), ANGPTL4 and ANGPTL8 (Kersten 2014). Insulin and apoC-III are the key regulators of LPL activity. Insulin stimulates the expression of LPL in endothelial cells whereas apoC-III inhibits LPL activity and thereby reduces the clearance of TRLs (Ginsberg and Brown 2011; Zheng et al. 2010; Yao 2012). The seminal role of LPL for the catabolic rate of TRLs and the conversion of large VLDL particles into smaller particles is demonstrated in studies in subjects with LPL gene mutations (Ooi et al. 2012). In addition, strong evidence supports the critical roles of apoC-III and apoE for suppressing or stimulating, respectively, clearance of apoB-containing lipoproteins from the circulation (Mendivil et al. 2010; Sacks 2015; Zheng et al. 2007).
ApoC-III is displacing apo-CII, an activator of LPL, from lipoprotein surfaces and thus impairing the actual lipolytic process (Sacks 2015; Gordts et al. 2016; Larsson et al. 2013). In addition, apoC-III has a wide range of actions action on triglyceride metabolism beyond its LPL-dependent effects (Taskinen and Boren 2015; Norata et al. 2015). For example, apoC-III interferes with the binding of apoB and apoE to hepatic lipoprotein receptors including heparin sulfate proteoglycan receptor (HSPG), low-density lipoprotein receptors (LDLR), and LDLR related protein 1 receptor (LRPl). This raises the option that high apoC-III would also inhibit the receptor mediated hepatic uptake of TRL remnants (Huff and Hegele 2013). The fact that an apoC-III ASO (antisense oligonucleotides) that inhibits apoC-III synthesis greatly reduced serum triglycerides in subjects with familial LPL deficiency demonstrates that apoC-III inhibits also hepatic clearance of remnants by LPL-independent pathways (Gaudet et al. 2014). However, recent results indicate that apoC-III inhibits turnover of TRLs primarily through a hepatic clearance mechanism mediated by the LDLR/LRPl axis, since apoC-III ASO treatment in LDLR/LRP1 deficient mice did not lower plasma TG levels (Gordts et al. 2016). Interestingly, we have recently reported that apoC-III metabolism is significantly perturbed in subjects with type 2 diabetes and that the apoC-III secretion rate was markedly higher in subjects with diabetes compared with BMI-matched non-diabetic subjects (Adiels et al. 2019). Improved glycemic control with liraglutide therapy reduced significantly apoC-III secretion rate and, thereby, apoC-III levels in type 2 diabetic subjects (Matikainen et al. 2019). These findings suggest that glucose homeostasis is a regulator of apoC-III metabolism and that the secretion rate of apoC-III seems to be an important driver for the elevation of TRLs in type 2 diabetes.
Angiopoietin-like protein (ANGPTL) family includes three members (ANGPTL3, ANGPTL4, and ANGPTL8) that are important modulators of lipoprotein metabolism (Kersten 2017; Christopoulou et al. 2019; Romeo et al. 2009; Li et al. 2020). Both ANGPTL3 and ANGPTL4 are endogenous inhibitors of LPL, and loss-of-function (LOF) mutations in ANGPTL3 and 4 associate with low triglyceride levels (Romeo et al. 2009; Minicocci et al. 2013; Zhang 2012) and reduced CVD risk (Dewey et al. 2017; Myocardial Infarction Genetics Investigators CAEC et al. 2016; Dewey et al. 2016). ANGPTL3 deficiency has been reported to reduce hepatic VLDL secretion and lower LDL-cholesterol (Wang et al. 2015a). Interestingly, ANGPTL3 gene silencing has been shown to associate not only with reduced hepatic secretion of apoB-containing lipoproteins, but also with enhanced uptake of particles via the LDL receptor. This likely explains the reduction of IDL cholesterol levels in subjects with familial hypobetalipoproteinemia (Xu et al. 2018). Consequently, targeting ANGPTL3 and ANGPTL4 genes has emerged as a promising goal for triglyceride lowering therapies (Tsimikas 2018; Gaudet et al. 2017a; Keech and Jenkins 2017; Bauer et al. 2016).
The inhibitory action of ANGPTL8 on LPL function requests the presence of ANGPTL3 (Kersten 2017; Haller et al. 2017; Luo and Peng 2018), as ANGPTL8 seems to enhance the inhibitory action of ANGPTL3 on LPL (Chi et al. 2017). Interestingly, these two proteins seem to work together to orchestrate responses of both glucose and lipid metabolism in fasting and in feeding (Wang et al. 2015b). Interestingly, ANGPTL3 is exclusively expressed in the liver being as a true hepatokine while ANGPTL8 is expressed both in adipose tissue and in the liver. The co-operative action of these two proteins seems to regulate the uptake of triglyceride-derived fatty acids either in the adipose tissue for storage or in cardiomyocytes and skeletal muscle for oxidation (Li et al. 2020; Vatner et al. 2018; Davies 2018).
ApoE plays a pivotal role in both triglyceride and cholesterol metabolism (Marais 2019). It predominantly associates with triglyceride-rich lipoproteins to mediate the clearance of their remnants after enzymatic lipolysis in the circulation (Marais 2019; Mahley and Huang 2007; Nakajima et al. 2019). Plasma levels of apoE and other lipids and lipoproteins are under strong genetic influence by APOE polymorphism – a combination of two genetic variants (rs429358 and rs7412) giving rise to six common APOE genotypes, ɛ22, ɛ32, ɛ33, ɛ42, ɛ43, and ɛ44 (Marais 2019; Mahley and Huang 2007). Both ɛ2 and ɛ4 alleles are associated with unfavorable lipid profiles, and the ɛ4 allele is a strong genetic risk factor for Alzheimer disease and by far the strongest hit in genome-wide association studies of longevity. The apoE variants relate to different amino acids at positions 112 and 158: cysteine in both for apoE2, arginine at both sites for apoE4, and respectively cysteine and arginine for apoE3 that is viewed as the wild type. High levels of plasma apoE have been shown to associate with increased risk of ischemic heart disease (Rasmussen et al. 2019). Hence both a quantitative importance of plasma apoE levels and a qualitative genetically determined effect appear to be important for cardiovascular disease (Rasmussen et al. 2019).
9 Role of Triglyceride-Rich Lipoproteins in Atherogenesis
It is well established that hypercholesterolemia is causatively linked to atherosclerotic cardiovascular disease and that lowering of cholesterol-rich LDL levels reduces cardiovascular events (Boren et al. 2020). However, cholesterol-lowering medication only prevents up to half of these events. Recent advances in human genetics indicate that the remaining “residual risk” of ASCVD is linked to elevated plasma triglyceride levels. Since triglyceride itself is not thought to contribute to atherogenesis, a consensus view has emerged that the remaining risk is linked to increased formation of “remnant” particles. These are derived from TRLs in the blood when the triglycerides are removed by the enzyme LPL (Boren et al. 2020). The remnant particles are not efficiently lowered by the available cholesterol-lowering medications.
To enter the artery wall, lipoproteins must cross the endothelium by transcytosis, a vesicular transport process. While chylomicrons and large VLDLs cannot undergo transcytosis because of their size, smaller chylomicron and VLDL remnants can and do penetrate the arterial wall. Thus, TRL remnants, in addition to LDL, may be retained in the arterial wall (Chapman et al. 2011). Even though remnant particles remain richer in triglycerides than cholesterol, their large size means that they contain up to twofold more cholesterol content per particle than LDL. However, the relative atherogenicity of remnants relative to LDL remains unclear.
10 Therapies to Reduce Triglyceride-Rich Lipoproteins
Lifestyle changes to lower plasma triglycerides – The first approach to lower moderately increased plasma triglycerides is to alter lifestyle (Laufs et al. 2020). Focus on a healthier diet and physical activity are cornerstones of lifestyle recommendations. Patients should reduce net caloric intake and lessen intake of sucrose, fructose, and alcohol. Diets rich in saturated fatty acids should be replaced with food enriched in monounsaturated and polyunsaturated fat (Laufs et al. 2020). It should be remembered though that scientific evidence for dietary recommendations is sparse. In addition, it is genuinely hard to persuade patients to change lifestyle and to make lifestyle changes that last. Thus, pharmaceutical approaches are often required.
Pharmacological therapies to lower plasma triglycerides – All commonly used cholesterol-lowering drugs as statins, ezetimibe, PCSK9 inhibitors only discreetly reduce triglyceride levels (around 5–15%), even though statins are somewhat more efficiently in reducing triglycerides and TRL remnants than PCSK9 inhibitors. Fibrates, omega-3-fatty acids, and niacin are somewhat more efficiently in reducing triglyceride levels (25–45%).
Fibrates are agonists of peroxisome proliferator-activated receptor-α (PPAR-α), acting via transcription factors regulating on lipid and lipoprotein metabolism. The drug has good efficacy in lowering fasting TG levels, as well as postprandial triglycerides and TRL remnants, albeit with marked interindividual variation. In addition, a small LDL-C increase may be observed in subjects with high triglyceride levels. The cardiovascular benefits have been shown to be heterogeneous and less robust than that of statins; when used as monotherapy, fibrates have been demonstrated to reduce CVD risk. However, when used in combination with statins no further reduction in CVD risk was demonstrated, although subgroup analysis indicates that hypertriglyceridemic patients with low HDL-C may benefit from such combination therapy. Results from ongoing trials using pemafibrate, a selective peroxisome proliferator-activated receptor alpha (PPARα) receptor agonist, will show if this approach will be successful (Pradhan et al. 2018).
For decades, omega-3-fatty acids have been used to lower plasma triglycerides and to prevent CVD (Bays et al. 2008). The results from two recent clinical trials using different omega-3 fatty acids have been mixed and somewhat confusing. The REDUCE-IT trial used icosapent ethyl omega-3 fatty acid (4 g daily). The results were positive and resulted in a 25% reduction in CVD and a 20% reduction in plasma triglyceride levels. Interestingly, the treatment also reduced plasma C-reactive protein by 40% (Bhatt et al. 2019). These results strengthen the link between plasma triglycerides and CVD (Myocardial Infarction Genetics Investigators CAEC et al. 2016; Do et al. 2013, 2015). However, the reduction of cardiovascular event was independent of plasma triglyceride levels both at baseline and on treatment, indicating that the reduction of cardiovascular events was only modest due to changes in TRL levels. One potential explanation for the clinical benefits could be the marked attenuation of the postprandial response by eicosapentaenoic acid, the hydrolytic product of icosapent ethyl, as 58% of the study participants had type 2 diabetes that commonly have prolonged postprandial hypertriglyceridemia (Taskinen and Boren 2015). In contrast to the positive outcome from the REDUCE-IT trial, the STRENGTH trial using another omega-3 fatty acid formulation (a combination of eicosapentaenoic acid and docosahexaenoic acid) failed to demonstrate any clinical benefit. The explanation(s) for the different outcomes is still unclear. Possible reasons include that the two trials studied different type of omega-3 fatty acids, and that the REDUCE-IT trial used mineral oil as placebo which may have adverse effects, whereas STRENGTH used a corn oil placebo. A recent Cochrane review of 86 randomized controlled trials with 162,796 participants concluded “evidence suggests that increasing long-chain omega-3 slightly reduces risk of coronary heart disease mortality and events, and reduces serum triglycerides” (Abdelhamid et al. 2020).
The 2019 ESC/EAS guidelines for the management of dyslipidemias recommend that statin treatment remains the first choice for managing high triglycerides (triglycerides >200 mg/dL or 2.3 mmol/L) (Mach et al. 2020). However, the guidelines have taken account of evidence from REDUCE-IT and recommend n-3 PUFAs (particularly icosapent ethyl 2 × 2 g daily) in high-risk patients with persistently elevated plasma triglycerides (between 135 and 499 mg/dL or 1.5 and 5.6 mmol/L) despite statin treatment. In high-risk patients at LDL-C goal with TG >200 mg/dL or >2.3 mmol/L, fenofibrate or bezafibrate may be considered in combination with statins (Mach et al. 2020).
11 Development of Novel Interventions
Efficient interventions to reduce plasma levels of TRLs and TRL remnants are still missing, and development of the development of strategies to treat the large numbers of hypertriglyceridemic individuals who currently remain at high risk of ASCVD despite optimal treatment according to current guidelines is urgently needed. Genetic studies have demonstrated that apoC-III and angiopoietin-like protein 3 (Angptl3) are critical regulators of triglyceride metabolism, and both have been developed as drug targets.
Statins and omega-3 fatty acids modestly reduce plasma apoC-III levels by less than 20% (Ooi et al. 2008; Maki et al. 2011; Morton et al. 2016; Dunbar et al. 2015). However, development of antisense oligos and siRNA has made it possible to develop high efficiency therapies. For example, antisense therapeutic oligonucleotides conjugated with N-acetyl galactosamine-conjugated (GalNAc) adducts (i.e., the ligand of the hepatic asialoglycoprotein receptor) have been developed. These are very efficient in reducing APOC3 expression (Graham et al. 2013). For example, results from the APPROACH trial, a 52-week randomized, double-blind, phase 3 in 66 patients with familial chylomicronemia syndrome, demonstrated that the drug resulted in an impressive 77% decrease in plasma triglyceride levels (Witztum et al. 2019). The antisense therapeutic oligonucleotides have also been shown to markedly lower plasma apoC-III and triglycerides levels in subjects with severe or uncontrolled hypertriglyceridemia (Gaudet et al. 2015; Gouni-Berthold 2017) and in subjects with diabetic dyslipidemia (Digenio et al. 2016). Intriguingly, the intervention not only improved the diabetic dyslipidemia, but also improved whole-body insulin sensitivity (by 57%).
To target Angptl3, both a monoclonal antibody and therapeutic oligonucleotides have been developed. Recent results have demonstrated that anti-ANGPTL3 therapies reduce both marked hypertriglyceridemia (around 75% reduction) and severely elevated LDL-cholesterol in subjects with familial hypercholesterolemia (around 23% reduction) (Gaudet et al. 2017b). The finding that anti-Angptl3 lowers LDL-C in subjects lacking functional LDL receptors indicates that the underlying mechanism is independent of the LDL receptor pathway. In line, a GalNac-modified antisense-oligonucleotide has recently been shown to reduce both plasma triglycerides and LDL-cholesterol (by 63.1% and 32.9%, respectively) (Graham et al. 2017). Interestingly, results from murine models indicate that the antisense-oligonucleotide seems to reduce hepatic steatosis. These results have prompted ongoing human studies.
Other ongoing projects involve lipoprotein lipase gene therapy, oral inhibitors of intestinal DGAT1 to reduce dietary fat absorption and triglyceride synthesis, and treatments targeting apoC-III and Angptl4 (Laufs et al. 2020).
12 Conclusion
Whether plasma triglycerides constitute an independent risk factor for CVD has been debated for decades, but there is now strong support for a causative role of TRLs in CVD. These studies equally indicate that cholesterol-enriched TRL remnant play a key role in the pathophysiology of atherosclerotic vascular disease. We are now beginning to understand the complex regulation of triglyceride metabolism. Hopefully, this molecular understanding will be translated into targeted treatment for the atherogenic dyslipidemia associated with hypertriglyceridemia.
References
Abdelhamid AS, Brown TJ, Brainard JS, Biswas P, Thorpe GC, Moore HJ, Deane KH, Summerbell CD, Worthington HV, Song F, Hooper L (2020) Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev 3:CD003177
Adiels M, Packard C, Caslake MJ, Stewart P, Soro A, Westerbacka J, Wennberg B, Olofsson SO, Taskinen MR, Boren J (2005) A new combined multicompartmental model for apolipoprotein B-100 and triglyceride metabolism in VLDL subfractions. J Lipid Res 46(1):58–67
Adiels M, Olofsson SO, Taskinen MR, Boren J (2006a) Diabetic dyslipidaemia. Curr Opin Lipidol 17(3):238–246
Adiels M, Taskinen MR, Packard C, Caslake MJ, Soro-Paavonen A, Westerbacka J, Vehkavaara S, Hakkinen A, Olofsson SO, Yki-Jarvinen H, Boren J (2006b) Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia 49(4):755–765
Adiels M, Matikainen N, Westerbacka J, Soderlund S, Larsson T, Olofsson SO, Boren J, Taskinen MR (2012) Postprandial accumulation of chylomicrons and chylomicron remnants is determined by the clearance capacity. Atherosclerosis 222(1):222–228
Adiels M, Taskinen MR, Bjornson E, Andersson L, Matikainen N, Soderlund S, Kahri J, Hakkarainen A, Lundbom N, Sihlbom C, Thorsell A, Zhou H, Pietilainen KH, Packard C, Boren J (2019) Role of apolipoprotein C-III overproduction in diabetic dyslipidaemia. Diabetes Obes Metab 21(8):1861–1870
Aguilar-Salinas CA, Barrett PH, Parhofer KG, Young SG, Tessereau D, Bateman J, Quinn C, Schonfeld G (1995) Apoprotein B-100 production is decreased in subjects heterozygous for truncations of apoprotein B. Arterioscler Thromb Vasc Biol 15(1):71–80
Anant S, Davidson NO (2001) Molecular mechanisms of apolipoprotein B mRNA editing. Curr Opin Lipidol 12(2):159–165
Asp L, Magnusson B, Rutberg M, Li L, Boren J, Olofsson SO (2005) Role of ADP ribosylation factor 1 in the assembly and secretion of ApoB-100-containing lipoproteins. Arterioscler Thromb Vasc Biol 25(3):566–570
Austin MA, King M-C, Vranizan KM, Krauss RM (1990) Atherogenic lipoprotein phenotype. A proposed genetic marker for coronary heart disease risk. Circulation 82:495–506
Barrows BR, Parks EJ (2006) Contributions of different fatty acid sources to very low-density lipoprotein-triacylglycerol in the fasted and fed states. J Clin Endocrinol Metab 91(4):1446–1452
Bauer RC, Khetarpal SA, Hand NJ, Rader DJ (2016) Therapeutic targets of triglyceride metabolism as informed by human genetics. Trends Mol Med 22(4):328–340
Bays HE, Tighe AP, Sadovsky R, Davidson MH (2008) Prescription omega-3 fatty acids and their lipid effects: physiologic mechanisms of action and clinical implications. Expert Rev Cardiovasc Ther 6(3):391–409
Bhatt DL, Steg PG, Miller M, Brinton EA, Jacobson TA, Ketchum SB, Doyle RT Jr, Juliano RA, Jiao L, Granowitz C, Tardif JC, Ballantyne CM, Investigators R-I (2019) Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 380(1):11–22
Bjornson E, Packard CJ, Adiels M, Andersson L, Matikainen N, Soderlund S, Kahri J, Hakkarainen A, Lundbom N, Lundbom J, Sihlbom C, Thorsell A, Zhou H, Taskinen MR, Boren J (2019a) Apolipoprotein B48 metabolism in chylomicrons and very low-density lipoproteins and its role in triglyceride transport in normo- and hypertriglyceridemic human subjects. J Intern Med 288(4):422–438
Bjornson E, Packard CJ, Adiels M, Andersson L, Matikainen N, Soderlund S, Kahri J, Sihlbom C, Thorsell A, Zhou H, Taskinen MR, Boren J (2019b) Investigation of human apoB48 metabolism using a new, integrated non-steady-state model of apoB48 and apoB100 kinetics. J Intern Med 285(5):562–577
Boren J, Graham L, Wettesten M, Scott J, White A, Olofsson SO (1992) The assembly and secretion of ApoB 100-containing lipoproteins in Hep G2 cells. ApoB 100 is cotranslationally integrated into lipoproteins. J Biol Chem 267(14):9858–9867
Boren J, Matikainen N, Adiels M, Taskinen MR (2014) Postprandial hypertriglyceridemia as a coronary risk factor. Clin Chim Acta 431:131–142
Borén J, Watts GF, Adiels M, Söderlund S, Chan DC, Hakkarainen A, Lundbom N, Matikainen N, Kahri J, Vergès B, Barrett PHR, Taskinen M (2015) Kinetic and related determinants of plasma triglyceride concentration in abdominal obesity. Multicenter Tracer Kinetic Study. Arterioscler Thromb Vasc Biol 35(10):2218–2224
Boren J, Chapman MJ, Krauss RM, Packard CJ, Bentzon JF, Binder CJ, Daemen MJ, Demer LL, Hegele RA, Nicholls SJ, Nordestgaard BG, Watts GF, Bruckert E, Fazio S, Ference BA, Graham I, Horton JD, Landmesser U, Laufs U, Masana L, Pasterkamp G, Raal FJ, Ray KK, Schunkert H, Taskinen MR, van de Sluis B, Wiklund O, Tokgozoglu L, Catapano AL, Ginsberg HN (2020) Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 41(24):2313–2330
Bostrom K, Boren J, Wettesten M, Sjoberg A, Bondjers G, Wiklund O, Carlsson P, Olofsson SO (1988) Studies on the assembly of apo B-100-containing lipoproteins in HepG2 cells. J Biol Chem 263(9):4434–4442
Browning JD, Horton JD (2004) Molecular mediators of hepatic steatosis and liver injury. J Clin Invest 114(2):147–152
Chao L, Marcus-Samuels B, Mason MM, Moitra J, Vinson C, Arioglu E, Gavrilova O, Reitman ML (2000) Adipose tissue is required for the antidiabetic, but not for the hypolipidemic, effect of thiazolidinediones. J Clin Invest 106(10):1221–1228
Chapman MJ, Ginsberg HN, Amarenco P, Andreotti F, Boren J, Catapano AL, Descamps OS, Fisher E, Kovanen PT, Kuivenhoven JA, Lesnik P, Masana L, Nordestgaard BG, Ray KK, Reiner Z, Taskinen MR, Tokgozoglu L, Tybjaerg-Hansen A, Watts GF, European Atherosclerosis Society Consensus Panel (2011) Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur Heart J 32(11):1345–1361
Chavez-Jauregui RN, Mattes RD, Parks EJ (2010) Dynamics of fat absorption and effect of sham feeding on postprandial lipema. Gastroenterology 139(5):1538–1548
Chi X, Britt EC, Shows HW, Hjelmaas AJ, Shetty SK, Cushing EM, Li W, Dou A, Zhang R, Davies BSJ (2017) ANGPTL8 promotes the ability of ANGPTL3 to bind and inhibit lipoprotein lipase. Mol Metab 6(10):1137–1149
Christopoulou E, Elisaf M, Filippatos T (2019) Effects of angiopoietin-like 3 on triglyceride regulation, glucose homeostasis, and diabetes. Dis Markers 2019:6578327
Cohn JS, Johnson EJ, Millar JS, Cohn SD, Milne RW, Marcel YL, Russell RM, Schaefer EJ (1993) Contribution of apoB-48 and apoB-100 triglyceride-rich lipoproteins (TRL) to postprandial increases in the plasma concentration of TRL triglycerides and retinyl esters. J Lipid Res 34(12):2033–2040
Dallinga-Thie GM, Franssen R, Mooij HL, Visser ME, Hassing HC, Peelman F, Kastelein JJ, Peterfy M, Nieuwdorp M (2010) The metabolism of triglyceride-rich lipoproteins revisited: new players, new insight. Atherosclerosis 211(1):1–8
Dallinga-Thie GM, Kroon J, Boren J, Chapman MJ (2016) Triglyceride-rich lipoproteins and remnants: targets for therapy? Curr Cardiol Rep 18(7):67
Davidson NO, Shelness GS (2000) APOLIPOPROTEIN B: mRNA editing, lipoprotein assembly, and presecretory degradation. Annu Rev Nutr 20:169–193
Davies BSJ (2018) Can targeting ANGPTL proteins improve glucose tolerance? Diabetologia 61(6):1277–1281
Dewey FE, Gusarova V, O’Dushlaine C, Gottesman O, Trejos J, Hunt C, Van Hout CV, Habegger L, Buckler D, Lai KM, Leader JB, Murray MF, Ritchie MD, Kirchner HL, Ledbetter DH, Penn J, Lopez A, Borecki IB, Overton JD, Reid JG, Carey DJ, Murphy AJ, Yancopoulos GD, Baras A, Gromada J, Shuldiner AR (2016) Inactivating variants in ANGPTL4 and risk of coronary artery disease. N Engl J Med 374(12):1123–1133
Dewey FE, Gusarova V, Dunbar RL, O’Dushlaine C, Schurmann C, Gottesman O, McCarthy S, Van Hout CV, Bruse S, Dansky HM, Leader JB, Murray MF, Ritchie MD, Kirchner HL, Habegger L, Lopez A, Penn J, Zhao A, Shao W, Stahl N, Murphy AJ, Hamon S, Bouzelmat A, Zhang R, Shumel B, Pordy R, Gipe D, Herman GA, Sheu WHH, Lee IT, Liang KW, Guo X, Rotter JI, Chen YI, Kraus WE, Shah SH, Damrauer S, Small A, Rader DJ, Wulff AB, Nordestgaard BG, Tybjaerg-Hansen A, van den Hoek AM, Princen HMG, Ledbetter DH, Carey DJ, Overton JD, Reid JG, Sasiela WJ, Banerjee P, Shuldiner AR, Borecki IB, Teslovich TM, Yancopoulos GD, Mellis SJ, Gromada J, Baras A (2017) Genetic and pharmacologic inactivation of ANGPTL3 and cardiovascular disease. N Engl J Med 377(3):211–221
Digenio A, Dunbar RL, Alexander VJ, Hompesch M, Morrow L, Lee RG, Graham MJ, Hughes SG, Yu R, Singleton W, Baker BF, Bhanot S, Crooke RM (2016) Antisense-mediated lowering of plasma apolipoprotein C-III by volanesorsen improves dyslipidemia and insulin sensitivity in type 2 diabetes. Diabetes Care 39(8):1408–1415
Do R, Willer CJ, Schmidt EM, Sengupta S, Gao C, Peloso GM, Gustafsson S, Kanoni S, Ganna A, Chen J, Buchkovich ML, Mora S, Beckmann JS, Bragg-Gresham JL, Chang HY, Demirkan A, Den Hertog HM, Donnelly LA, Ehret GB, Esko T, Feitosa MF, Ferreira T, Fischer K, Fontanillas P, Fraser RM, Freitag DF, Gurdasani D, Heikkila K, Hypponen E, Isaacs A, Jackson AU, Johansson A, Johnson T, Kaakinen M, Kettunen J, Kleber ME, Li X, Luan J, Lyytikainen LP, Magnusson PK, Mangino M, Mihailov E, Montasser ME, Muller-Nurasyid M, Nolte IM, O’Connell JR, Palmer CD, Perola M, Petersen AK, Sanna S, Saxena R, Service SK, Shah S, Shungin D, Sidore C, Song C, Strawbridge RJ, Surakka I, Tanaka T, Teslovich TM, Thorleifsson G, Van den Herik EG, Voight BF, Volcik KA, Waite LL, Wong A, Wu Y, Zhang W, Absher D, Asiki G, Barroso I, Been LF, Bolton JL, Bonnycastle LL, Brambilla P, Burnett MS, Cesana G, Dimitriou M, Doney AS, Doring A, Elliott P, Epstein SE, Eyjolfsson GI, Gigante B, Goodarzi MO, Grallert H, Gravito ML, Groves CJ, Hallmans G, Hartikainen AL, Hayward C, Hernandez D, Hicks AA, Holm H, Hung YJ, Illig T, Jones MR, Kaleebu P, Kastelein JJ, Khaw KT, Kim E, Klopp N, Komulainen P, Kumari M, Langenberg C, Lehtimaki T, Lin SY, Lindstrom J, Loos RJ, Mach F, WL MA, Meisinger C, Mitchell BD, Muller G, Nagaraja R, Narisu N, Nieminen TV, Nsubuga RN, Olafsson I, Ong KK, Palotie A, Papamarkou T, Pomilla C, Pouta A, Rader DJ, Reilly MP, Ridker PM, Rivadeneira F, Rudan I, Ruokonen A, Samani N, Scharnagl H, Seeley J, Silander K, Stancakova A, Stirrups K, Swift AJ, Tiret L, Uitterlinden AG, van Pelt LJ, Vedantam S, Wainwright N, Wijmenga C, Wild SH, Willemsen G, Wilsgaard T, Wilson JF, Young EH, Zhao JH, Adair LS, Arveiler D, Assimes TL, Bandinelli S, Bennett F, Bochud M, Boehm BO, Boomsma DI, Borecki IB, Bornstein SR, Bovet P, Burnier M, Campbell H, Chakravarti A, Chambers JC, Chen YD, Collins FS, Cooper RS, Danesh J, Dedoussis G, de Faire U, Feranil AB, Ferrieres J, Ferrucci L, Freimer NB, Gieger C, Groop LC, Gudnason V, Gyllensten U, Hamsten A, Harris TB, Hingorani A, Hirschhorn JN, Hofman A, Hovingh GK, Hsiung CA, Humphries SE, Hunt SC, Hveem K, Iribarren C, Jarvelin MR, Jula A, Kahonen M, Kaprio J, Kesaniemi A, Kivimaki M, Kooner JS, Koudstaal PJ, Krauss RM, Kuh D, Kuusisto J, Kyvik KO, Laakso M, Lakka TA, Lind L, Lindgren CM, Martin NG, Marz W, MI MC, CA MK, Meneton P, Metspalu A, Moilanen L, Morris AD, Munroe PB, Njolstad I, Pedersen NL, Power C, Pramstaller PP, Price JF, Psaty BM, Quertermous T, Rauramaa R, Saleheen D, Salomaa V, Sanghera DK, Saramies J, Schwarz PE, Sheu WH, Shuldiner AR, Siegbahn A, Spector TD, Stefansson K, Strachan DP, Tayo BO, Tremoli E, Tuomilehto J, Uusitupa M, van Duijn CM, Vollenweider P, Wallentin L, Wareham NJ, Whitfield JB, Wolffenbuttel BH, Altshuler D, Ordovas JM, Boerwinkle E, Palmer CN, Thorsteinsdottir U, Chasman DI, Rotter JI, Franks PW, Ripatti S, Cupples LA, Sandhu MS, Rich SS, Boehnke M, Deloukas P, Mohlke KL, Ingelsson E, Abecasis GR, Daly MJ, Neale BM, Kathiresan S (2013) Common variants associated with plasma triglycerides and risk for coronary artery disease. Nat Genet 45(11):1345–1352
Do R, Stitziel NO, Won HH, Jorgensen AB, Duga S, Angelica Merlini P, Kiezun A, Farrall M, Goel A, Zuk O, Guella I, Asselta R, Lange LA, Peloso GM, Auer PL, NES P, Girelli D, Martinelli N, Farlow DN, MA DP, Roberts R, Stewart AF, Saleheen D, Danesh J, Epstein SE, Sivapalaratnam S, Hovingh GK, Kastelein JJ, Samani NJ, Schunkert H, Erdmann J, Shah SH, Kraus WE, Davies R, Nikpay M, Johansen CT, Wang J, Hegele RA, Hechter E, Marz W, Kleber ME, Huang J, Johnson AD, Li M, Burke GL, Gross M, Liu Y, Assimes TL, Heiss G, Lange EM, Folsom AR, Taylor HA, Olivieri O, Hamsten A, Clarke R, Reilly DF, Yin W, Rivas MA, Donnelly P, Rossouw JE, Psaty BM, Herrington DM, Wilson JG, Rich SS, Bamshad MJ, Tracy RP, Cupples LA, Rader DJ, Reilly MP, Spertus JA, Cresci S, Hartiala J, Tang WH, Hazen SL, Allayee H, Reiner AP, Carlson CS, Kooperberg C, Jackson RD, Boerwinkle E, Lander ES, Schwartz SM, Siscovick DS, McPherson R, Tybjaerg-Hansen A, Abecasis GR, Watkins H, Nickerson DA, Ardissino D, Sunyaev SR, O’Donnell CJ, Altshuler D, Gabriel S, Kathiresan S (2015) Exome sequencing identifies rare LDLR and APOA5 alleles conferring risk for myocardial infarction. Nature 518(7537):102–106
Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ (2005) Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 115(5):1343–1351
Dunbar RL, Nicholls SJ, Maki KC, Roth EM, Orloff DG, Curcio D, Johnson J, Kling D, Davidson MH (2015) Effects of omega-3 carboxylic acids on lipoprotein particles and other cardiovascular risk markers in high-risk statin-treated patients with residual hypertriglyceridemia: a randomized, controlled, double-blind trial. Lipids Health Dis 14:98
Edvardsson U, Bergstrom M, Alexandersson M, Bamberg K, Ljung B, Dahllof B (1999) Rosiglitazone (BRL49653), a PPARgamma-selective agonist, causes peroxisome proliferator-like liver effects in obese mice. J Lipid Res 40(7):1177–1184
Elias N, Patterson BW, Schonfeld G (1999) Decreased production rates of VLDL triglycerides and ApoB-100 in subjects heterozygous for familial hypobetalipoproteinemia. Arterioscler Thromb Vasc Biol 19(11):2714–2721
Farquhar JW, Gross RC, Wagner RM, Reaven GM (1965) Validation of an incompletely coupled two-compartment nonrecycling catenary model for turnover of liver and plasma triglyceride in man. J Lipid Res 6:119–134
Fielding BA, Reid G, Grady M, Humphreys SM, Evans K, Frayn KN (2000) Ethanol with a mixed meal increases postprandial triacylglycerol but decreases postprandial non-esterified fatty acid concentrations. Br J Nutr 83(6):597–604
Gardner CD, Fortmann SP, Krauss RM (1996) Association of small low-density lipoprotein particles with the incidence of coronary artery disease in men and women. JAMA 276:875–881
Gaudet D, Brisson D, Tremblay K, Alexander VJ, Singleton W, Hughes SG, Geary RS, Baker BF, Graham MJ, Crooke RM, Witztum JL (2014) Targeting APOC3 in the familial chylomicronemia syndrome. N Engl J Med 371(23):2200–2206
Gaudet D, Alexander VJ, Baker BF, Brisson D, Tremblay K, Singleton W, Geary RS, Hughes SG, Viney NJ, Graham MJ, Crooke RM, Witztum JL, Brunzell JD, Kastelein JJ (2015) Antisense inhibition of apolipoprotein C-III in patients with hypertriglyceridemia. N Engl J Med 373(5):438–447
Gaudet D, Drouin-Chartier JP, Couture P (2017a) Lipid metabolism and emerging targets for lipid-lowering therapy. Can J Cardiol 33(7):872–882
Gaudet D, Gipe DA, Pordy R, Ahmad Z, Cuchel M, Shah PK, Chyu KY, Sasiela WJ, Chan KC, Brisson D, Khoury E, Banerjee P, Gusarova V, Gromada J, Stahl N, Yancopoulos GD, Hovingh GK (2017b) ANGPTL3 inhibition in homozygous familial hypercholesterolemia. N Engl J Med 377(3):296–297
Gaw A, Packard CJ, Lindsay GM, Griffin BA, Caslake MJ, Lorimer AR, Shepherd J (1995) Overproduction of small very low density lipoproteins (Sf 20-60) in moderate hypercholesterolemia: relationships between apolipoprotein B kinetics and plasma lipoproteins. J Lipid Res 36(1):158–171
Georgieva AM, van Greevenbroek MM, Krauss RM, Brouwers MC, Vermeulen VM, Robertus-Teunissen MG, van der Kallen CJ, de Bruin TW (2004) Subclasses of low-density lipoprotein and very low-density lipoprotein in familial combined hyperlipidemia: relationship to multiple lipoprotein phenotype. Arterioscler Thromb Vasc Biol 24(4):744–749
Gibbons GF, Islam K, Pease RJ (2000) Mobilisation of triacylglycerol stores. Biochim Biophys Acta 1483(1):37–57
Ginsberg HN, Brown WV (2011) Apolipoprotein CIII: 42 years old and even more interesting. Arterioscler Thromb Vasc Biol 31(3):471–473
Ginsberg HN, Le NA, Goldberg IJ, Gibson JC, Rubinstein A, Wang-Iverson P, Norum R, Brown WV, Apolipoprotein B (1986) Metabolism in subjects with deficiency of apolipoproteins CIII and AI. Evidence that apolipoprotein CIII inhibits catabolism of triglyceride-rich lipoproteins by lipoprotein lipase in vivo. J Clin Invest 78(5):1287–1295
Goldberg IJ (1996) Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis. J Lipid Res 37(4):693–707
Gordts PL, Nock R, Son NH, Ramms B, Lew I, Gonzales JC, Thacker BE, Basu D, Lee RG, Mullick AE, Graham MJ, Goldberg IJ, Crooke RM, Witztum JL, Esko JD (2016) ApoC-III inhibits clearance of triglyceride-rich lipoproteins through LDL family receptors. J Clin Invest 126(8):2855–2866
Gouni-Berthold I (2017) The role of antisense oligonucleotide therapy against apolipoprotein-CIII in hypertriglyceridemia. Atheroscler Suppl 30:19–27
Graham MJ, Lee RG, Bell TA 3rd, Fu W, Mullick AE, Alexander VJ, Singleton W, Viney N, Geary R, Su J, Baker BF, Burkey J, Crooke ST, Crooke RM (2013) Antisense oligonucleotide inhibition of apolipoprotein C-III reduces plasma triglycerides in rodents, nonhuman primates, and humans. Circ Res 112(11):1479–1490
Graham MJ, Lee RG, Brandt TA, Tai LJ, Fu W, Peralta R, Yu R, Hurh E, Paz E, McEvoy BW, Baker BF, Pham NC, Digenio A, Hughes SG, Geary RS, Witztum JL, Crooke RM, Tsimikas S (2017) Cardiovascular and metabolic effects of ANGPTL3 antisense oligonucleotides. N Engl J Med 377(3):222–232
Gusarova V, Brodsky JL, Fisher EA (2003) Apolipoprotein B100 exit from the endoplasmic reticulum (ER) is COPII-dependent, and its lipidation to very low density lipoprotein occurs post-ER. J Biol Chem 278(48):48051–48058
Haller JF, Mintah IJ, Shihanian LM, Stevis P, Buckler D, Alexa-Braun CA, Kleiner S, Banfi S, Cohen JC, Hobbs HH, Yancopoulos GD, Murphy AJ, Gusarova V, Gromada J (2017) ANGPTL8 requires ANGPTL3 to inhibit lipoprotein lipase and plasma triglyceride clearance. J Lipid Res 58(6):1166–1173
Havel RJ (1961) Conversion of plasma free fatty acids into triglycerides of plasma lipoprotein fractions in man. Metabolism 10:1031–1034
Huff MW, Hegele RA (2013) Apolipoprotein C-III: going back to the future for a lipid drug target. Circ Res 112(11):1405–1408
Hussain MM, Fatma S, Pan X, Iqbal J (2005) Intestinal lipoprotein assembly. Curr Opin Lipidol 16(3):281–285
James RW, Martin B, Pometta D, Fruchart JC, Duriez P, Puchois P, Farriaux JP, Tacquet A, Demant T, Clegg RJ et al (1989) Apolipoprotein B metabolism in homozygous familial hypercholesterolemia. J Lipid Res 30(2):159–169
Julve J, Martin-Campos JM, Escola-Gil JC, Blanco-Vaca F (2016) Chylomicrons: advances in biology, pathology, laboratory testing, and therapeutics. Clin Chim Acta 455:134–148
Karpe F, Bell M, Bjorkegren J, Hamsten A (1995) Quantification of postprandial triglyceride-rich lipoproteins in healthy men by retinyl ester labeling and simultaneous measurement of apolipoproteins B-48 and B-100. Arterioscler Thromb Vasc Biol 15(2):199–207
Keech AC, Jenkins AJ (2017) Triglyceride-lowering trials. Curr Opin Lipidol 28(6):477–487
Kersten S (2014) Physiological regulation of lipoprotein lipase. Biochim Biophys Acta 1841(7):919–933
Kersten S (2017) Angiopoietin-like 3 in lipoprotein metabolism. Nat Rev Endocrinol 13(12):731–739
Khan NA, Besnard P (2009) Oro-sensory perception of dietary lipids: new insights into the fat taste transduction. Biochim Biophys Acta 1791(3):149–155
Koo SH, Dutcher AK, Towle HC (2001) Glucose and insulin function through two distinct transcription factors to stimulate expression of lipogenic enzyme genes in liver. J Biol Chem 276(12):9437–9445
Lamarche B, Tchernof A, Moorjani S, Cantin B, Dagenais GR, Lupien PJ, Despres JP (1997) Small, dense low-density lipoprotein particles as a predictor of the risk of ischemic heart disease in men. Prospective results from the Quebec Cardiovascular Study. Circulation 95(1):69–75
Lambert JE, Parks EJ (2012) Postprandial metabolism of meal triglyceride in humans. Biochim Biophys Acta 1821(5):721–726
Larsson M, Vorrsjo E, Talmud P, Lookene A, Olivecrona G (2013) Apolipoproteins C-I and C-III inhibit lipoprotein lipase activity by displacement of the enzyme from lipid droplets. J Biol Chem 288(47):33997–34008
Laufs U, Parhofer KG, Ginsberg HN, Hegele RA (2020) Clinical review on triglycerides. Eur Heart J 41(1):99–109c
Lee J, Hegele RA (2014) Abetalipoproteinemia and homozygous hypobetalipoproteinemia: a framework for diagnosis and management. J Inherit Metab Dis 37(3):333–339
Li J, Li L, Guo D, Li S, Zeng Y, Liu C, Fu R, Huang M, Xie W (2020) Triglyceride metabolism and angiopoietin-like proteins in lipoprotein lipase regulation. Clin Chim Acta 503:19–34
Lichtenstein AH, Hachey DL, Millar JS, Jenner JL, Booth L, Ordovas J, Schaefer EJ (1992) Measurement of human apolipoprotein B-48 and B-100 kinetics in triglyceride-rich lipoproteins using [5,5,5-2H3]leucine. J Lipid Res 33(6):907–914
Linton MF, Farese RV Jr, Young SG (1993) Familial hypobetalipoproteinemia. J Lipid Res 34(4):521–541
Luo M, Peng D (2018) ANGPTL8: an important regulator in metabolic disorders. Front Endocrinol (Lausanne) 9:169
Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, Chapman MJ, De Backer GG, Delgado V, Ference BA, Graham IM, Halliday A, Landmesser U, Mihaylova B, Pedersen TR, Riccardi G, Richter DJ, Sabatine MS, Taskinen MR, Tokgozoglu L, Wiklund O, ESC Scientific Document Group (2020) 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 41(1):111–188
Mahley RW, Huang Y (2007) Atherogenic remnant lipoproteins: role for proteoglycans in trapping, transferring, and internalizing. J Clin Invest 117(1):94–98
Maki KC, Bays HE, Dicklin MR, Johnson SL, Shabbout M (2011) Effects of prescription omega-3-acid ethyl esters, coadministered with atorvastatin, on circulating levels of lipoprotein particles, apolipoprotein CIII, and lipoprotein-associated phospholipase A2 mass in men and women with mixed dyslipidemia. J Clin Lipidol 5(6):483–492
Marais AD (2019) Apolipoprotein E in lipoprotein metabolism, health and cardiovascular disease. Pathology 51(2):165–176
Matikainen N, Soderlund S, Bjornson E, Pietilainen K, Hakkarainen A, Lundbom N, Taskinen MR, Boren J (2019) Liraglutide treatment improves postprandial lipid metabolism and cardiometabolic risk factors in humans with adequately controlled type 2 diabetes: a single-centre randomized controlled study. Diabetes Obes Metab 21(1):84–94
Mattes RD (2002) Oral fat exposure increases the first phase triacylglycerol concentration due to release of stored lipid in humans. J Nutr 132(12):3656–3662
Mattes RD (2009) Brief oral stimulation, but especially oral fat exposure, elevates serum triglycerides in humans. Am J Physiol Gastrointest Liver Physiol 296(2):G365–G371
Mendivil CO, Zheng C, Furtado J, Lel J, Sacks FM (2010) Metabolism of very-low-density lipoprotein and low-density lipoprotein containing apolipoprotein C-III and not other small apolipoproteins. Arterioscler Thromb Vasc Biol 30(2):239–245
Minicocci I, Santini S, Cantisani V, Stitziel N, Kathiresan S, Arroyo JA, Marti G, Pisciotta L, Noto D, Cefalu AB, Maranghi M, Labbadia G, Pigna G, Pannozzo F, Ceci F, Ciociola E, Bertolini S, Calandra S, Tarugi P, Averna M, Arca M (2013) Clinical characteristics and plasma lipids in subjects with familial combined hypolipidemia: a pooled analysis. J Lipid Res 54(12):3481–3490
Morton AM, Furtado JD, Lee J, Amerine W, Davidson MH, Sacks FM (2016) The effect of omega-3 carboxylic acids on apolipoprotein CIII-containing lipoproteins in severe hypertriglyceridemia. J Clin Lipidol 10(6):1442–1451.e4
Myocardial Infarction Genetics Investigators CAEC, Stitziel NO, Stirrups KE, Masca NG, Erdmann J, Ferrario PG, Konig IR, Weeke PE, Webb TR, Auer PL, Schick UM, Lu Y, Zhang H, Dube MP, Goel A, Farrall M, Peloso GM, Won HH, Do R, van Iperen E, Kanoni S, Kruppa J, Mahajan A, Scott RA, Willenberg C, Braund PS, van Capelleveen JC, Doney AS, Donnelly LA, Asselta R, Merlini PA, Duga S, Marziliano N, Denny JC, Shaffer CM, El-Mokhtari NE, Franke A, Gottesman O, Heilmann S, Hengstenberg C, Hoffman P, Holmen OL, Hveem K, Jansson JH, Jockel KH, Kessler T, Kriebel J, Laugwitz KL, Marouli E, Martinelli N, MI MC, Van Zuydam NR, Meisinger C, Esko T, Mihailov E, Escher SA, Alver M, Moebus S, Morris AD, Muller-Nurasyid M, Nikpay M, Olivieri O, Lemieux Perreault LP, AlQarawi A, Robertson NR, Akinsanya KO, Reilly DF, Vogt TF, Yin W, Asselbergs FW, Kooperberg C, Jackson RD, Stahl E, Strauch K, Varga TV, Waldenberger M, Zeng L, Kraja AT, Liu C, Ehret GB, Newton-Cheh C, Chasman DI, Chowdhury R, Ferrario M, Ford I, Jukema JW, Kee F, Kuulasmaa K, Nordestgaard BG, Perola M, Saleheen D, Sattar N, Surendran P, Tregouet D, Young R, Howson JM, Butterworth AS, Danesh J, Ardissino D, Bottinger EP, Erbel R, Franks PW, Girelli D, Hall AS, Hovingh GK, Kastrati A, Lieb W, Meitinger T, Kraus WE, Shah SH, McPherson R, Orho-Melander M, Melander O, Metspalu A, Palmer CN, Peters A, Rader D, Reilly MP, Loos RJ, Reiner AP, Roden DM, Tardif JC, Thompson JR, Wareham NJ, Watkins H, Willer CJ, Kathiresan S, Deloukas P, Samani NJ, Schunkert H (2016) Coding variation in ANGPTL4, LPL, and SVEP1 and the risk of coronary disease. N Engl J Med 374(12):1134–1144
Nakajima K, Tokita Y, Tanaka A, Takahashi S (2019) The VLDL receptor plays a key role in the metabolism of postprandial remnant lipoproteins. Clin Chim Acta 495:382–393
Norata GD, Tsimikas S, Pirillo A, Catapano AL (2015) Apolipoprotein C-III: from pathophysiology to pharmacology. Trends Pharmacol Sci 36(10):675–687
Nordestgaard BG, Varbo A (2014) Triglycerides and cardiovascular disease. Lancet 384(9943):626–635
Olofsson SO, Boren J (2005) Apolipoprotein B: a clinically important apolipoprotein which assembles atherogenic lipoproteins and promotes the development of atherosclerosis. J Intern Med 258(5):395–410
Olofsson SO, Stillemark-Billton P, Asp L (2000) Intracellular assembly of VLDL: two major steps in separate cell compartments. Trends Cardiovasc Med 10(8):338–345
Ooi EM, Watts GF, Chan DC, Chen MM, Nestel PJ, Sviridov D, Barrett PH (2008) Dose-dependent effect of rosuvastatin on VLDL-apolipoprotein C-III kinetics in the metabolic syndrome. Diabetes Care 31(8):1656–1661
Ooi EM, Russell BS, Olson E, Sun SZ, Diffenderfer MR, Lichtenstein AH, Keilson L, Barrett PH, Schaefer EJ, Sprecher DL (2012) Apolipoprotein B-100-containing lipoprotein metabolism in subjects with lipoprotein lipase gene mutations. Arterioscler Thromb Vasc Biol 32(2):459–466
Packard CJ (2003) Triacylglycerol-rich lipoproteins and the generation of small, dense low-density lipoprotein. Biochem Soc Trans 31(Pt 5):1066–1069
Paquette M, Dufour R, Hegele RA, Baass A (2016) A tale of 2 cousins: an atypical and a typical case of abetalipoproteinemia. J Clin Lipidol 10(4):1030–1034
Parhofer KG, Barrett PH, Aguilar-Salinas CA, Schonfeld G (1996) Positive linear correlation between the length of truncated apolipoprotein B and its secretion rate: in vivo studies in human apoB-89, apoB-75, apoB-54.8, and apoB-31 heterozygotes. J Lipid Res 37(4):844–852
Parks EJ, Hellerstein MK (2006) Thematic review series: patient-oriented research. Recent advances in liver triacylglycerol and fatty acid metabolism using stable isotope labeling techniques. J Lipid Res 47(8):1651–1660
Parks EJ, Krauss RM, Christiansen MP, Neese RA, Hellerstein MK (1999) Effects of a low-fat, high-carbohydrate diet on VLDL-triglyceride assembly, production, and clearance. J Clin Invest 104(8):1087–1096
Poulsen MK, Nellemann B, Stodkilde-Jorgensen H, Pedersen SB, Gronbaek H, Nielsen S (2016) Impaired insulin suppression of VLDL-triglyceride kinetics in nonalcoholic fatty liver disease. J Clin Endocrinol Metab 101(4):1637–1646
Pradhan AD, Paynter NP, Everett BM, Glynn RJ, Amarenco P, Elam M, Ginsberg H, Hiatt WR, Ishibashi S, Koenig W, Nordestgaard BG, Fruchart JC, Libby P, Ridker PM (2018) Rationale and design of the Pemafibrate to reduce cardiovascular outcomes by reducing triglycerides in patients with diabetes (PROMINENT) study. Am Heart J 206:80–93
Prinsen BH, Romijn JA, Bisschop PH, de Barse MM, Barrett PH, Ackermans M, Berger R, Rabelink TJ, de Sain-van der Velden MG (2003) Endogenous cholesterol synthesis is associated with VLDL-2 apoB-100 production in healthy humans. J Lipid Res 44(7):1341–1348
Rasmussen KL, Tybjaerg-Hansen A, Nordestgaard BG, Frikke-Schmidt R (2019) Plasma levels of apolipoprotein E, APOE genotype, and all-cause and cause-specific mortality in 105 949 individuals from a white general population cohort. Eur Heart J 40(33):2813–2824
Robertson MD, Henderson RA, Vist GE, Rumsey RD (2002) Extended effects of evening meal carbohydrate-to-fat ratio on fasting and postprandial substrate metabolism. Am J Clin Nutr 75(3):505–510
Robertson MD, Parkes M, Warren BF, Ferguson DJ, Jackson KG, Jewell DP, Frayn KN (2003) Mobilisation of enterocyte fat stores by oral glucose in humans. Gut 52(6):834–839
Romeo S, Yin W, Kozlitina J, Pennacchio LA, Boerwinkle E, Hobbs HH, Cohen JC (2009) Rare loss-of-function mutations in ANGPTL family members contribute to plasma triglyceride levels in humans. J Clin Invest 119(1):70–79
Rustaeus S, Stillemark P, Lindberg K, Gordon D, Olofsson SO (1998) The microsomal triglyceride transfer protein catalyzes the post-translational assembly of apolipoprotein B-100 very low density lipoprotein in McA-RH7777 cells. J Biol Chem 273(9):5196–5203
Sacks FM (2015) The crucial roles of apolipoproteins E and C-III in apoB lipoprotein metabolism in normolipidemia and hypertriglyceridemia. Curr Opin Lipidol 26(1):56–63
Sacks FM, Campos H (2003) Clinical review 163: cardiovascular endocrinology: low-density lipoprotein size and cardiovascular disease: a reappraisal. J Clin Endocrinol Metab 88(10):4525–4532
Salter AM, Wiggins D, Sessions VA, Gibbons GF (1998) The intracellular triacylglycerol/fatty acid cycle: a comparison of its activity in hepatocytes which secrete exclusively apolipoprotein (apo) B100 very-low-density lipoprotein (VLDL) and in those which secrete predominantly apoB48 VLDL. Biochem J 332(Pt 3):667–672
Sandesara PB, Virani SS, Fazio S, Shapiro MD (2019) The forgotten lipids: triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk. Endocr Rev 40(2):537–557
Schneeman BO, Kotite L, Todd KM, Havel RJ (1993) Relationships between the responses of triglyceride-rich lipoproteins in blood plasma containing apolipoproteins B-48 and B-100 to a fat-containing meal in normolipidemic humans. Proc Natl Acad Sci U S A 90(5):2069–2073
Segrest JP, Jones MK, De Loof H, Dashti N (2001) Structure of apolipoprotein B-100 in low density lipoproteins. J Lipid Res 42(9):1346–1367
Shimomura I, Bashmakov Y, Horton JD (1999) Increased levels of nuclear SREBP-1c associated with fatty livers in two mouse models of diabetes mellitus. J Biol Chem 274(42):30028–30032
Stillemark P, Boren J, Andersson M, Larsson T, Rustaeus S, Karlsson KA, Olofsson SO (2000) The assembly and secretion of apolipoprotein B-48-containing very low density lipoproteins in McA-RH7777 cells. J Biol Chem 275(14):10506–10513
Stillemark-Billton P, Beck C, Boren J, Olofsson SO (2005) Relation of the size and intracellular sorting of apoB to the formation of VLDL 1 and VLDL 2. J Lipid Res 46(1):104–114
Tamura S, Shimomura I (2005) Contribution of adipose tissue and de novo lipogenesis to nonalcoholic fatty liver disease. J Clin Invest 115(5):1139–1142
Tanoli T, Yue P, Yablonskiy D, Schonfeld G (2004) Fatty liver in familial hypobetalipoproteinemia: roles of the APOB defects, intra-abdominal adipose tissue, and insulin sensitivity. J Lipid Res 45(5):941–947
Taskinen MR (2003) Diabetic dyslipidaemia: from basic research to clinical practice. Diabetologia 46(6):733–749
Taskinen MR, Boren J (2015) New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis 239(2):483–495
Taskinen MR, Adiels M, Westerbacka J, Soderlund S, Kahri J, Lundbom N, Lundbom J, Hakkarainen A, Olofsson SO, Orho-Melander M, Boren J (2011) Dual metabolic defects are required to produce hypertriglyceridemia in obese subjects. Arterioscler Thromb Vasc Biol 31(9):2144–2150
Tsimikas S (2018) RNA-targeted therapeutics for lipid disorders. Curr Opin Lipidol 29(6):459–466
Vakkilainen J, Mero N, Schweizer A, Foley JE, Taskinen MR (2002) Effects of nateglinide and glibenclamide on postprandial lipid and glucose metabolism in type 2 diabetes. Diabetes Metab Res Rev 18(6):484–490
Vakkilainen J, Steiner G, Ansquer JC, Aubin F, Rattier S, Foucher C, Hamsten A, Taskinen MR (2003) Relationships between low-density lipoprotein particle size, plasma lipoproteins, and progression of coronary artery disease: the Diabetes Atherosclerosis Intervention Study (DAIS). Circulation 107(13):1733–1737
Vatner DF, Goedeke L, Camporez JG, Lyu K, Nasiri AR, Zhang D, Bhanot S, Murray SF, Still CD, Gerhard GS, Shulman GI, Samuel VT (2018) Angptl8 antisense oligonucleotide improves adipose lipid metabolism and prevents diet-induced NAFLD and hepatic insulin resistance in rodents. Diabetologia 61(6):1435–1446
Verges B (2005) New insight into the pathophysiology of lipid abnormalities in type 2 diabetes. Diabetes Metab 31(5):429–439
Wang AB, Liu DP, Liang CC (2003) Regulation of human apolipoprotein B gene expression at multiple levels. Exp Cell Res 290(1):1–12
Wang Y, Gusarova V, Banfi S, Gromada J, Cohen JC, Hobbs HH (2015a) Inactivation of ANGPTL3 reduces hepatic VLDL-triglyceride secretion. J Lipid Res 56(7):1296–1307
Wang Y, McNutt MC, Banfi S, Levin MG, Holland WL, Gusarova V, Gromada J, Cohen JC, Hobbs HH (2015b) Hepatic ANGPTL3 regulates adipose tissue energy homeostasis. Proc Natl Acad Sci U S A 112(37):11630–11635
Welty FK (2014) Hypobetalipoproteinemia and abetalipoproteinemia. Curr Opin Lipidol 25(3):161–168
Welty FK, Lichtenstein AH, Barrett PH, Dolnikowski GG, Ordovas JM, Schaefer EJ (1997) Decreased production and increased catabolism of apolipoprotein B-100 in apolipoprotein B-67/B-100 heterozygotes. Arterioscler Thromb Vasc Biol 17(5):881–888
Welty FK, Lichtenstein AH, Barrett PHR, Dolnikowski GG, Schaefer EJ (1999) Human apolipoprotein (Apo) B-48 and ApoB-100 kinetics with stable isotopes. Arterioscler Thromb Vasc Biol 19(12):2966–2974
Westerbacka J, Kolak M, Kiviluoto T, Arkkila P, Siren J, Hamsten A, Fisher RM, Yki-Jarvinen H (2007) Genes involved in fatty acid partitioning and binding, lipolysis, monocyte/macrophage recruitment, and inflammation are overexpressed in the human fatty liver of insulin-resistant subjects. Diabetes 56(11):2759–2765
Wiggins D, Gibbons GF (1992) The lipolysis/esterification cycle of hepatic triacylglycerol. Its role in the secretion of very-low-density lipoprotein and its response to hormones and sulphonylureas. Biochem J 284:457–462
Witztum JL, Gaudet D, Freedman SD, Alexander VJ, Digenio A, Williams KR, Yang Q, Hughes SG, Geary RS, Arca M, Stroes ESG, Bergeron J, Soran H, Civeira F, Hemphill L, Tsimikas S, Blom DJ, O’Dea L, Bruckert E (2019) Volanesorsen and triglyceride levels in familial chylomicronemia syndrome. N Engl J Med 381(6):531–542
Xiao C, Stahel P, Lewis GF (2019) Regulation of chylomicron secretion: focus on post-assembly mechanisms. Cell Mol Gastroenterol Hepatol 7(3):487–501
Xu YX, Redon V, Yu H, Querbes W, Pirruccello J, Liebow A, Deik A, Trindade K, Wang X, Musunuru K, Clish CB, Cowan C, Fizgerald K, Rader D, Kathiresan S (2018) Role of angiopoietin-like 3 (ANGPTL3) in regulating plasma level of low-density lipoprotein cholesterol. Atherosclerosis 268:196–206
Yao Z (2012) Human apolipoprotein C-III – a new intrahepatic protein factor promoting assembly and secretion of very low density lipoproteins. Cardiovasc Hematol Disord Drug Targets 12(2):133–140
Zhang R (2012) Lipasin, a novel nutritionally-regulated liver-enriched factor that regulates serum triglyceride levels. Biochem Biophys Res Commun 424(4):786–792
Zheng C, Khoo C, Ikewaki K, Sacks FM (2007) Rapid turnover of apolipoprotein C-III-containing triglyceride-rich lipoproteins contributing to the formation of LDL subfractions. J Lipid Res 48(5):1190–1203
Zheng C, Khoo C, Furtado J, Sacks FM (2010) Apolipoprotein C-III and the metabolic basis for hypertriglyceridemia and the dense low-density lipoprotein phenotype. Circulation 121(15):1722–1734
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this chapter
Cite this chapter
Borén, J., Taskinen, MR. (2021). Metabolism of Triglyceride-Rich Lipoproteins. In: von Eckardstein, A., Binder, C.J. (eds) Prevention and Treatment of Atherosclerosis . Handbook of Experimental Pharmacology, vol 270. Springer, Cham. https://doi.org/10.1007/164_2021_520
Download citation
DOI: https://doi.org/10.1007/164_2021_520
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-86075-2
Online ISBN: 978-3-030-86076-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)