Abstract
Low plasma levels of High Density Lipoprotein (HDL) cholesterol (HDL-C) are associated with increased risks of atherosclerotic cardiovascular disease (ASCVD). In cell culture and animal models, HDL particles exert multiple potentially anti-atherogenic effects. However, drugs increasing HDL-C have failed to prevent cardiovascular endpoints. Mendelian Randomization studies neither found any genetic causality for the associations of HDL-C levels with differences in cardiovascular risk. Therefore, the causal role and, hence, utility as a therapeutic target of HDL has been questioned. However, the biomarker “HDL-C” as well as the interpretation of previous data has several important limitations: First, the inverse relationship of HDL-C with risk of ASCVD is neither linear nor continuous. Hence, neither the-higher-the-better strategies of previous drug developments nor previous linear cause-effect relationships assuming Mendelian randomization approaches appear appropriate. Second, most of the drugs previously tested do not target HDL metabolism specifically so that the futile trials question the clinical utility of the investigated drugs rather than the causal role of HDL in ASCVD. Third, the cholesterol of HDL measured as HDL-C neither exerts nor reports any HDL function. Comprehensive knowledge of structure-function-disease relationships of HDL particles and associated molecules will be a pre-requisite, to test them for their physiological and pathogenic relevance and exploit them for the diagnostic and therapeutic management of individuals at HDL-associated risk of ASCVD but also other diseases, for example diabetes, chronic kidney disease, infections, autoimmune and neurodegenerative diseases.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
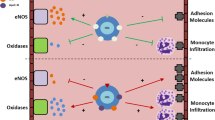
Low plasma levels of high density lipoprotein (HDL) cholesterol (HDL-C) are associated with increased risks of atherosclerotic cardiovascular diseases (ASCVD), notably coronary heart disease (CHD) (Emerging Risk Factors Collaboration 2009; Madsen et al. 2021). HDL particles exert a broad spectrum of biological activities many of which are considered as anti-atherogenic, for example mediation of cholesterol efflux from macrophage foam cells and reverse transport of cholesterol to the liver, promotion of endothelial integrity and function, inhibition of inflammation by suppression of myelopoiesis and transmigration of leukocytes through the endothelium as well as macrophage activation, inhibition of lipid oxidation as well as inactivation of oxidized lipids (Fig. 1) (Von Eckardstein and Kardassis 2015; Robert et al. 2021; Rohatgi et al. 2021). Furthermore, atherosclerosis could be decreased or even reverted in several animal models by transgenic over-expression or exogenous application of apolipoprotein (apoA-I), i.e. the most abundant protein of HDL (Hoekstra and Van Eck 2015; Lee-Rueckert et al. 2016). However, in humans, drugs increasing HDL-C such as fibrates, nicotinic acid (niacin), or inhibitors of cholesteryl ester transfer protein (CETP) have failed to prevent fatal or non-fatal cardiovascular endpoints (Keene et al. 2014; Riaz et al. 2019). Infusions of reconstituted HDL (rHDL) did not lead to regression of atherosclerosis in coronary or carotid arteries (He et al. 2021). Moreover, in several inborn errors of human HDL metabolism and genetic mouse models with altered HDL metabolism, low or high HDL-C levels were not always associated with the differences in cardiovascular risk and atherosclerotic plaque load, respectively, that were expected from epidemiology (Hoekstra and Van Eck 2015; Lee-Rueckert et al. 2016; Zanoni and von Eckardstein 2020). For example, the loss of scavenger receptor B1 (SR-BI) function aggravates the risk of ASCVD events in human carriers of SCARB1 mutations and promotes atherosclerosis in Scarb1 knock-out mice despite increasing HDL-C levels (Hoekstra and Van Eck 2015; Lee-Rueckert et al. 2016; Zanoni et al. 2016). Because of these ambiguous data, the causal role of HDL in the pathogenesis of atherosclerosis as well as the suitability of HDL-C as a therapeutic target is nowadays scrutinized if not doubted (Madsen et al. 2021; März et al. 2017). Both the previous euphoria and the current skepticism in the discussion of HDL’s role in health and disease, specifically in ASCVD but also beyond, have been suffering from several misconceptions, which are described in the first part of this review. As the conclusion, several perspectives for the clinical exploitation of HDL are presented in the second part.
Possible pathophysiological relationships of low HDL cholesterol with its associated diseases (modified from Von Eckardstein and Kardassis 2015)
2 Possible Reasons for HDL-C’s Clinical Futility
2.1 Lack of Causality
Mendelian randomization studies have been a successful tool to support the causality of LDL cholesterol (LDL-C) in atherosclerosis: Single nucleotide polymorphisms (SNPs) and rare genetic variants that are associated with lower or higher LDL-C levels are associated with lower and higher risk, respectively, of ASCVD events. The associations of genetically determined LDL-C with ASCVD risk are even stronger than the associations of measured LDL-C. This is because the genetic information includes both time and dosage of exposure to the harmful LDL-C whereas the measured LDL-C only records the dosage of the harm (Borén et al. 2020). Mendelian randomization studies also support causality of hypertriglyceridemia and elevated apoB levels as well as hypertension in the pathogenesis of ASCVD (Benn and Nordestgaard 2018). Conversely, this genetic strategy rather excluded genetic causality of HDL-C and apoA-I levels in the manifestation of ASCVD, at least after maximal adjustment for confounding lipid traits such as apoB and triglyceride levels (Richardson et al. 2020; Voight et al. 2012). However, it is important to note the limitations of Mendelian Randomization studies. With respect to HDL-C the most important limitation is the assumption of a continuous relationship between the risk factor and the clinical endpoint. This is true for the association of LDL-C or nonHDL-C with major cardiovascular events but not for HDL-C, where no difference in risk is observed among individuals with HDL-C levels above the 60th percentile (Emerging Risk Factors Collaboration 2009; Johannesen et al. 2020; Madsen et al. 2017; see Sect. 2.2).
HDL-C levels below the widely accepted risk thresholds of 1.0 mmol/L or 40 mg/dL are frequently confounded by other risk factors of ASCVD, notably hypertriglyceridemia, manifest diabetes mellitus type 2 (T2DM) or impaired fasting glucose, smoking, chronic inflammatory diseases (chronic obstructive lung disease, rheumatic diseases) or biomarkers of inflammation (e.g., elevated C-reactive protein), overweight or obesity (Fig. 1; Assmann et al. 1996; Damen et al. 2017). Due to the links of HDL metabolism with the metabolism of triglyceride-rich lipoproteins, it has been suggested that low HDL-C is an indirect long-term indicator of postprandial hypertriglyceridemia and hence exposure of atherogenic remnants like elevated glycated hemoglobin A1c is a long-term marker of disturbed glucose metabolism but a non-causal risk factor of glycation-induced organ damage (Langsted et al. 2020).
2.2 Epidemiology and Human Genetics Disprove “the Higher the Better” Concept
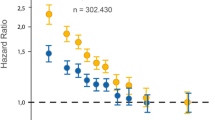
The association of HDL-C with risk of ASCVD events has been described for decades to be inverse. The resulting widespread reception of HDL-C as the “good cholesterol” led to the application of “the higher the better” strategies to both patient counselling and drug development. However, the meta-analysis of 68 population studies with more than 300,000 participants and 2,785 incident myocardial infarctions by the Emerging Risk Factors Collaboration found the unadjusted risk of myocardial infarction gradually decreasing from the first decile to the eighth decile (i.e., until about 1.5 mmol/L or 58 mg/dL) but no significant changes at higher levels of HDL-C. After adjustment for possible confounders, statistically significant dose-dependent risk decreases happen within the lower six deciles until about 50 mg/dL (1.3 mmol/L) but not above this threshold (Emerging Risk Factors Collaboration 2009). Similar observations were made in more than 110,000 and 630,000 participants of the Copenhagen General Population (CGPS) and Copenhagen City Heart Studies (CCHS) and CANHEART studies (Madsen et al. 2017; Wijeysundera et al. 2017). In Denmark, the decreases in risk of cardiovascular events reached plateaus at 1.5 mmol/L in men and 2.0 mmol/L in women (Madsen et al. 2017). In Canada, below the reference interval ranging from 50 to 60 mg/dL, the incidence of ASCVD events increased with every decreasing 10 mg/dL interval of HDL-C. Above the threshold of 60 mg/dL, the ASCVD risks were overall significantly lower compared to the reference interval, but did not differ between the increasing 10 mg/dL strata, neither in men nor in women (Wijeysundera et al. 2017). Of note, the associations of HDL-C with total as well as disease-specific mortalities including cardiovascular mortality are even parabolic (U-shaped): Both in the Danish and Canadian studies, the inverse associations of HDL-C with total mortality reached their nadirs at 1.8–1.9 mmol/L (70–75 mg/dL) and 2.3–2.4 mmol/L (90–95 mg/dL) in men and women, respectively. Beyond these thresholds, the risk of dying became gradually higher with further increasing HDL-C levels (Ko et al. 2016; Madsen et al. 2017).
The discontinuous and even parabolic associations of HDL-C with cardiovascular morbidity and mortality, respectively, have been largely ignored both in the execution of Mendelian randomization studies and in the design of randomized controlled studies that aimed at the lowering of cardiovascular risk by increasing of HDL-C: both have been based on the assumption of continuous the-higher-the-better associations. The majority of the trials on fibrates, niacin, or CETP inhibitors did not define any upper limit of HDL-C for inclusion into the trial (Table 1). No Mendelian Randomization study restricted the analysis to the ranges where changes in HDL-C are associated with changes in risk, e.g. to the lower five or six deciles (Richardson et al. 2020; Voight et al. 2012). Of note, a large register study of a lipid clinics in Boston among individuals with HDL-C below 25 mg/dL (0.8 mmol/L) found an increased prevalence of ASCVD events in carriers of mutations in the genes of APOA1, ABCA1, LCAT, and LPL (Geller et al. 2018). Also studies in Dutch and Canadian families affected by loss of function mutations in APOA1, ABCA1, or LCAT found the prevalence of ASCVD events increased among mutation carriers, but only if HDL-C was below the fifth percentile (Abdel-Razek et al. 2018; Tietjen et al. 2012). Conversely, mutations in the genes of CETP, SCARB1, and LIPG, which cause increases in HDL-C show heterogenous associations with ASCVD. Loss of function mutations in LIPG encoding endothelial lipase do not alter the risk of ASCVD (Voight et al. 2012). The associations of loss of function mutations in CETP and SCARB1 with ASCVD are controversial: Rare SCARB1 mutations were associated with increased CVD risk in one study but not in another (Helgadottir et al. 2018; Zanoni et al. 2016). CETP deficiency was originally associated with reduced risk of ASCVD and increased life expectancy but later studies found diverse associations of loss of function mutations in CETP with ASCVD, namely increased risk in the Honolulu Heart Study but decreased risk in a Japanese population study (Moriyama et al. 1998; Yamashita and Matsuzawa 2016; Zhong et al. 1996). Likewise, the common polymorphisms in CETP which are associated with lower CETP mass and activity, LDL-C, and triglycerides but higher HDL-C were showed diverse associations with ASCVD in different studies: meta-analyses found lower risks of ASCVD associated with loss of function alleles of CETP (Kathiresan 2012; Niu and Qi 2015), but there are several individual studies which found the opposite (Agerholm-Larsen et al. 2000; Borggreve et al. 2006). Loss of function mutations in APOC3 cause higher HDL-C levels and reduce cardiovascular risk (Crosby et al. 2014; Pollin et al. 2008), but this may reflect proatherogenic features of apoC-III beyond its influence on HDL-C and triglyceride levels (Riwanto et al. 2013; Zewinger et al. 2020; Zvintzou et al. 2017) (see also Sect. 3.2).
2.3 Limitations of HDL Modifying Drugs
The futility of the most recent randomized controlled trials (RCTs) on fenofibrate (ACCORD Study Group 2010; Keech et al. 2005), nicotinic acid (AIM-HIGH Investigators 2011; HPS2-THRIVE Collaborative Group 2014), and cholesteryl ester transfer protein (CETP)-inhibitors (ILLUMINATE, Dal-OUTCOME, ACCELERATE, REVEAL) (Barter et al. 2007b; HPS3/TIMI55–REVEAL Collaborative Group 2017; Lincoff et al. 2017; Schwartz et al. 2012) is frequently used as the argument to question the causality of HDL in the pathogenesis of atherosclerosis. However, this conclusion overlooks that – except the CETP inhibitor dalcetrapib (Schwartz et al. 2012) – none of these drugs is specifically altering HDL-C. Especially fibrates and nicotinic acid exert stronger effects on other lipoprotein traits than on HDL-C. Thus, their failure to reduce ASCVD events should primarily prompt to scrutinize the suitability of these pharmacological strategies rather than the causality of HDL in ASCVD. Moreover, one should be oblivious to meta-analyses that demonstrated futility of fibrates or nicotinic acid if combined with statins but efficacy if used as monotherapies (Keene et al. 2014; Riaz et al. 2019). Likewise, genetic studies indicate that potential efficacy of CETP inhibitors in ASCVD prevention may be hampered by the combination with statins (Ference et al. 2017).
2.3.1 Neither Fibrates nor Nicotinic Acid Specifically Target HDL Metabolism
Fibrates are agonists of the peroxisome proliferator agonist receptor alpha (PPARα). As such they regulate the transcription of several genes which are relevant in the metabolism of HDL metabolism (e.g., APOA1, PLTP, SCARB1) but also triglyceride-rich lipoproteins (Montaigne et al. 2021; Zandbergen and Plutzky 2007). As the result, fibrates cause increases in HDL-C of maximally 15% and decreases in triglycerides of 25–50%. The rather moderate effect on HDL-C is partially explained by the induction of APOA1 and SCARB1 genes, which enhances production and catabolism of HDL, respectively. As the result, the flux of HDL and probably reverse cholesterol transport are affected by fibrates more profoundly than reflected by changes in HDL-C. Triglycerides rather than HDL-C were the most profoundly altered lipoprotein traits. The two gemfibrozil utilizing trials – the primary prevention Helsinki Heart Study (Frick et al. 1987) and the secondary prevention study VA-HIT (Rubins et al. 1999) – were the only ones which found significant reductions of ASCVD events by the fibrate intervention vs. placebo. Only three trials (VA-HIT, BIP, and ACCORD) pre-defined plasma levels of HDL-C as inclusion criterion (ACCORD Study Group 2010; Bezafibrate Infarction Prevention Study 2000; Rubins et al. 1999). For ACCORD, the threshold was rather high with 55 mg/dL (1.42 mmol/L) (ACCORD study group 2010). Post-hoc analyses of the fibrate trials demonstrated relative risk reductions for subgroups of patients with HDL-C and triglycerides levels <35 mg/dL (0.9 mmol/L) and >200 mg/dL (2.3 mmol/L) ranging from 27% (FIELD, Keech et al. 2005) to −65% (Helsinki Heart Study) (Sacks et al. 2010). Currently the Pemafibrate to Reduce Cardiovascular OutcoMes by Reducing Triglycerides IN patiENts With diabeTes (PROMINENT) trial tests prospectively the efficacy of the novel combined PPARα/PPARδ agonist pemafibrate (NCT03071692) (Pradhan et al. 2018).
Nicotinic acid (niacin) is an agonist of the G-protein coupled receptor GPR109A (HM74A or PUMA-G) (Offermanns 2014). As such, it primarily inhibits the lipolysis in adipocytes and secondarily, by reducing the free fatty acid flux, the lipogenesis and VLDL production in the liver. Reduced free fatty acid exposure may also promote ABCA1 activity in the liver and hence the production of nascent HDL (Chapman et al. 2010; KAmanna et al. 2013). In addition CETP activity was found decreased upon treatment with nicotinic acid due to direct and indirect inhibitory effects via production as well as activity of the protein and diminished pool of VLDL and hence acceptor particles, respectively (Chapman et al. 2010). In addition, nicotinic acid lowers plasma levels of LDL-C and lipoprotein(a) (Lp(a)). Despite these multiple beneficial effects on lipoproteins, in both the AIM-HIGH and HPS-THRIVE trials, the combination of statins with nicotinic acid was not superior to statin monotherapy in preventing ASCVD events (Table 2) (AIM-HIGH Investigators 2011; HPS2-THRIVE Collaborative Group 2014). Only AIM-HIGH defined inclusion criteria based on HDL-C (<1.05 mmol/L or <40 mg/dL). However, post-hoc analyses did not find any evidence that low HDL-C defines a subgroup of patients who benefit from nicotinic acid (Guyton et al. 2013; HPS2 THRIVE Collaborative Group 2014) However, in a meta-analysis monotherapy of nicotinic acid was found effective in reducing cardiovascular morbidity and mortality (Keene et al. 2014). Because of futility and the occurrence of flushes as very unpleasant and frequent side effects, nicotinic acid is no longer available for treatment in many countries.
2.3.2 CETP Inhibitors Block Rather than Promote Reverse Cholesterol Transport
CETP links the metabolism of HDL and apoB containing lipoproteins by exchanging cholesteryl esters of HDL against triglycerides of VLDL and LDL (Chapman et al. 2010). As the result of inhibiting this exchange, the most effective CETP inhibitors – torcetrapib, evacetrapib, and anacetrapib – cause increases of HDL-C by 75 (Torcetrapib) to 130% (Evacetrapib) and decreases of LDL-C by 25% (torcetrapib) to 40% (Anacetrapib) (Barter et al. 2007b; HPS3/TIMI55–REVEAL Collaborative Group 2017; Lincoff et al. 2017). The weaker CETP inhibitor dalcetrapib increases HDL-C by 30% without causing any drop in LDL-C (Schwartz et al. 2012). CETP inhibitors also decrease Lp(a) by up to 35% through an as yet unknown mechanism (Gencer and Mach 2020). Despite their at first sight beneficial effects on lipoprotein traits, three trials were prematurely stopped; the ILLUMINATE trial because of excess morbidity and mortality in the torcetrapib arm possibly due to off target effects of torcetrapib (Barter et al. 2007b). ACCELERATE and dal-OUTCOME were stopped prematurely because of futility of evacetrapib and dalcetrapib, respectively (Lincott et al. 2017; Schwartz et al. 2012). Only the combination of statin with anacetrapib in the REVEAL trial showed some superiority towards statin only therapy (HPS3/TIMI55–REVEAL Collaborative Group 2017). However, with a 9% relative risk reduction or the primary endpoint, the added value of anacetrapib was small and attributed to the decrease in LDL-C rather than to the increase in HDL-C. Because of the parallel successful development of PCSK9 inhibitors, which are much more effective in lowering LDL-C and event rates, the development of anacetrapib was stopped. Dalcetrapib, however, is further developed towards a personalized indication: post-hoc analyses of the Dal-OUTCOME study revealed that the carrier status for a mutation in the adenylate cyclase subtype 9 encoding ADCY9 gene discriminated individuals who did or did not benefit from dalcetrapib treatment by lower ASCVD event rates (Tardif et al. 2015). However, the same mutation discriminated responders and non-responders neither to anacetrapib nor to evacetrapib in the REVEAL and ACCELERATE trials, respectively (Hopewell et al. 2019; Nissen et al. 2018). Conversely, the interaction of CETP with ADCY9 was recapitulated in genetic mouse models (Rautureau et al. 2018). The Dal-GenE trial currently investigates prospectively, whether patients selected for the ADCY9 genotype benefit from treatment with dalcetrapib (NCT02525939) (Tardif et al. 2020).
At first sight the negative outcomes of the CETP inhibitor trials were surprising, not only because of the beneficial effects on the lipoprotein profile but also because several large genetic studies demonstrated lower prevalences or incidences of cardiovascular events among carriers of low activity CETP alleles (Kathiresan 2012). However, later population genetic studies showed an interaction between CETP and HMGCR alleles. In the presence of HMGCR alleles that reduce HMG-CoA reductase activity and thereby mimic treatment effects of statins, CETP alleles that cause low CETP activity and mimic the effects of CETP inhibitors did not confer any additional cardiovascular risk reduction (Ference et al. 2017). Likewise, torcetrapib treatment reduced atherosclerosis in apoE3 Leiden*CETP transgenic mice, if provided as monotherapy but not if provided in combination with statins (de Haan et al. 2008) whereas anacetrapib treatment enhanced the anti-atherogenic effect of atorvastatin (Kühnast et al. 2015). Nevertheless, the question is raised if the anti-atherogenicity of CETP inhibition depends on the capacity of the LDL receptor pathway: if this is fully functional, for example, as the result of statin treatment, CETP will promote reverse cholesterol transport and should not be blocked (von Eckardstein 2020). Only in situations, where LDL removal by the LDL receptor pathway is compromised, it may be useful to withheld cholesterol from LDL by CETP inhibition for hepatic removal through LDL receptor independent pathways involving direct HDL/receptor interactions, for example with SR-BI. As an alternative explanation, it was proposed that CETP inhibition renders HDL dysfunctional by prolonging the half-life of HDL particles and thereby making them susceptible to adverse alterations in the lipid and protein composition or oxidative and enzymatic modifications of protein or lipid components. However, the classical function of HDL, mediation of cholesterol efflux from macrophages was rather increased upon treatment of humans or animals with Evacetrapib, Anacetrapib, or Dalcetrapib (Brodeur et al. 2017; Metzinger et al. 2020; Nicholls et al. 2015; Simic et al. 2017; Tardif et al. 2015). Interestingly endothelial functions were not improved or even impaired in apoE3 Leiden*CETP transgenic mice upon treatment with evacetrapib and anacetrapib, respectively, despite increasing CEC and paraoxonase activity (Simic et al. 2017).
2.3.3 Combination with High-Intensity Statins: The Winner Takes it All
The combination of statins with fenofibrate, nicotinic acid, or CETP inhibitors was motivated by post-hoc meta-analyses of statin trials, which found the residual risk of patients treated with statins to be significantly associated with low HDL-C levels (Boekholdt et al. 2013). A closer look to post-hoc analyses of individual trials, however, reveals that these associations became weaker the lower LDLC levels were reached. For example, in the WOSCOP study, where mean levels of LDL-C were lowered from 5.0 mmol/L in the placebo arm to 3.6 mmol/L in the pravastatin arm, low baseline levels of HDL-C were associated with increased risk of ASCVD events in both treatment groups (West of Scotland Coronary Prevention Study Group 1998). However, more than 10 years later in the JUPITER study (LDL-C at baseline <3.37 mmol/L), both baseline and on-treatment levels of HDL-C were significantly associated with residual risk only in the placebo group with a mean on treatment LDL-C of 2.8 mmol/L, but not in the rosuvastatin group with an on treatment mean LDL-C level of 1.42 mmol/L (Ridker et al. 2010). Similar discrepant observations were made in the secondary prevention trials CARE and LIPID vs. TNT: HDL-C levels explained part of the residual risk in both placebo and pravastatin groups of CARE and LIPID (mean baseline LDL-C 3.80 mmol/L, on treatment LDL-C 2.85 mmol/L) (Sacks et al. 2000). However, in the TNT trial (baseline LDL-C 2.55 mmol/L), low HDL-C increased ASCVD risk in the low-dose atorvastatin group (on treatment LDL-C 2.60 mmol/L) but not in the high dose atorvastatin group (on treatment LDL-C 2.0 mmol/L) (Barter et al. 2007a). It thus appears that the optimized control of LDL-C by high intensity statin therapy alleviates the residual risk associated with low HDL-C levels. In this regard it is also noteworthy that contemporary observational studies in general populations as well as in patients with clinically manifest ASCVD find weaker associations of HDL-C with first and recurrent cardiovascular events, respectively, than historical studies which recruited their participants in the pre-statin era (Bolibar et al. 2000; Colantonio et al. 2016; Schwartz et al. 2012). These secular trends are usually explained by the generally improved risk factor control. However, one must also be aware of the change in the methodology of HDL-C measurements that occurred in parallel with the triumphal procession of statins. Since about 1990, non-traceable and biased homogenous assays have replaced the previous cholesterol quantification after manual precipitation of apoB containing lipoproteins. One can hence not exclude that changes in the analytics affected the prognostic value of HDL-C (Miller et al. 2010).
2.4 Wrong Biomarker “the Good Cholesterol”
By contrast to the disease causing cholesterol in LDL (Borén et al. 2020), the cholesterol in HDL (that is HDL-C) neither exerts nor reflects any of the potentially anti-atherogenic activities of HDL. HDL-C is only a non-functional surrogate marker for estimating the HDL pool size without deciphering the heterogeneous composition and, hence, functionality of HDL (Rohatgi et al. 2021; Annema and von Eckardstein 2013, 2016). Differences in the molar content of apoA-I, phosphatidylcholines, cholesterol, and cholesteryl ester cause differences of HDL subclasses in shape, size, and charge. HDL particles carry hundreds of different quantitatively minor proteins and lipid species many of which are not just passive cargo (like cholesterol) but biologically active and susceptible to quantitative and qualitative modifications by diseases or interventions (Rohatgi et al. 2021, Annema and von Eckardstein 2013, 2016). These functionally active components hence have a much bigger chance than HDL-C to serve as a causal biomarker that can be exploited towards the development, targeting, and monitoring of therapies.
The most obvious candidate for a functional HDL biomarker is the plasma concentration of apoA-I which is not only a mandatory structural component of the bulk of HDL but also exerts several biological activities of HDL, for example activation of ABCA1 and LCAT to efflux and esterify cholesterol, respectively, or binding to SR-BI and other HDL receptors. In both epidemiological and clinical studies, apoA-I levels show inverse associations with ASCVD events, which however are not stronger than those of HDL-C (Emerging Risk Factors Collaboration 2009). Neither did Mendelian Randomization studies unravel any causal genetic relationship between apoA-I levels and ASCVD (Karjalainen et al. 2020; Richardson et al. 2020).
Other widely investigated HDL biomarker candidates include numbers and sizes of HDL particles. However, the outcomes of their evaluation in epidemiological and clinical studies are controversial. Some studies found HDL particle number (HDL-P) superior to HDL-C (Chandra et al. 2015; Kuller et al. 2007; Mackey et al. 2012; Otvos et al. 2006; Singh et al. 2020), others vice versa (El Harchaoui et al. 2009; Mora et al. 2009; Parish et al. 2012; Qi et al. 2015). Interestingly, within the JUPITER trial HDL-P was superior to HDL-C in the prediction of events among statin treated probands but inferior among placebo treated probands (Mora et al. 2013). Some studies found small HDL particles more strongly related with outcomes than large HDL particles (Ditah et al. 2016; Kim et al. 2016; McGarrah et al. 2016; Silbernagel et al. 2017), other studies found the opposite (Li et al. 2016; Arsenault et al. 2009). A recent meta-analysis of four studies concluded similar strong associations of small, medium, and large HDL particles with the incidence of ASCVD events (hazard ratio and 95% confidence interval 0.91 and 0.87 to 0.96) (Wu et al. 2018). With a hazard ratio and 95% confidence interval of 0.82 and 0.78 to 0.87, the total number of HDL particles showed stronger associations. Interestingly a recent Mendelian Randomization study found protective associations between the concentration of medium and – less so but also significantly – small HDL particles with coronary artery disease (Zhao et al. 2021). Drug interventions in lipoprotein metabolism result in diverse changes of HDL particle size and numbers. For example, treatment with nicotinic acid and CETP inhibitors increases HDL-C levels more profoundly than HDL-P, reflecting the shift to larger particles. Vice versa, upon treatment with fibrates, HDL-P increases more strongly than HDL-C (Rosenson et al. 2015).
3 Consequences and Perspectives
3.1 The Search for Novel HDL-Biomarkers
The further development of HDL as a therapeutic target is mainly limited by the availability of biomarkers that reflect the functional and causal role of HDL in the pathogenesis of atherosclerosis. To this end, bioassays of HDL function were recently developed and validated in population and clinical studies. Among them, cholesterol efflux capacity (CEC) has been investigated most extensively. In these studies, different macrophage cell lines treated with different drugs to enhance the cellular cholesterol efflux machinery were utilized as donors of radioactively or fluorescently labeled cholesterol. ApoB depleted plasma or serum was used as acceptors and as surrogate of HDL to avoid laborious ultracentrifugation. The heterogeneity of assays together with the heterogeneity of populations investigated has contributed to some discrepant findings (Anastasius et al. 2018). Nevertheless, a recent meta-analysis of eight prospective studies and more than 10,000 participants with more than 3,000 events found a significant inverse association of CEC with ASCVD events (HR 0.86, 95% CI: 0.76–0.98). In a subgroup of five studies also mortality was related to CEC (HR 0.77, 95% CI: 0.80–1.0). Although CEC correlates with HDL-C, the associations of CEC with cardiovascular outcomes were independent of HDL-C (Soria-Florido et al. 2020). However, the concept of CEC as a proxy of HDL functionality has several limitations. First, as a laborious and difficult if not impossible to standardize bioassay it is a research rather than diagnostic tool, primarily for proof of concept studies and secondarily for the identification of functional molecular markers (Anastasius et al. 2018). Second, CEC should not be considered as an overall proxy of HDL functionality because other functions of HDL neither correlate nor share molecular determinants with CEC (Cardner et al. 2020). Third, although most intensively investigated, it is not clear that mediation of cholesterol efflux is the most relevant atheroprotective function of HDL. In fact, changes in CEC upon treatment with CETP inhibitors did not predict correctly the clinical outcomes of these drug interventions. They led to increases in CEC of apoB-free sera or plasmas but not to any reduction in cardiovascular event rates and coronary atherosclerosis, respectively (Brodeur et al. 2017; Metzinger et al. 2020; Nicholls et al. 2015; Simic et al. 2017; Tardif et al. 2015).
Despite these limitations, CEC has been used as the reference to develop molecular biomarkers that can be measured in clinical laboratories. One example is the derivation of an algorithm which integrates the information of differently sized HDL particles as measured by NMR. The estimated NMR-based CEC correlated very well with the in vitro measured CEC (R2 > 0.8) and predicted incident CHD events with a hazards ratio of 0.86; 95% CI, 0.79–0.93, adjusted for traditional risk factors and HDL-C) (Kuusisto et al. 2019). Another example is a proteomic score integrating the information of apolipoproteins A-I, C-I, C-II, C-III, and C-IV showed good correlation with CEC as well as significant association with the presence of coronary artery disease and cardiovascular mortality independently of clinical risk factors including conventionally measured concentrations of apoA-I and apoB (Jin et al. 2019; Natarajan et al. 2019). Replication studies are needed to validate these surrogate scores of CEC.
Several laboratories have used tandem mass spectrometry to search for protein or lipid components of HDL as functional biomarkers. The most recent update of the HDL Proteome Watch data bank (http://homepages.uc.edu/~davidswm/HDLproteome.html; accessed July 15, 2021) documents more than 200 proteins which were identified in HDL by at least three of 40 independent studies and are therefore considered as highly confident components of HDL. Even higher numbers of lipid species were identified by mass spectrometry of HDL (Cardner et al. 2020; Kontush et al. 2013). The concentrations of these molecules vary from less than 1 μmol/L to more than 1 mmol/L (Annema and von Eckardstein 2013; Rohatgi et al. 2021). Already in view of the average HDL particle concentration of about 20 μmol/L it is clear that only some lipids (e.g., unesterified cholesterol, cholesteryl esters, phosphatidylcholines) or proteins (e.g., apoA-I) are present on each particle with several copies. Other low abundant lipids (e.g., sphingosine-1-phosphate, oxysterols) and proteins (apoM or LCAT) are dispersed throughout different particles. Interestingly, these molecules are non-randomly distributed among HDL particles. For example, the presence of sphingosine-1-phosphate is linked to the presence of its chaperone apoM (Christoffersen et al. 2011). By combining two immunoaffinity chromatography procedures, one with anti-apoA-I antibodies and one with an antibody against one of 16 other HDL-associated proteins, 16 HDL subclasses with distinct proteomes and little intraindividual variation over 3–24 months were identified (Furtado et al. 2018). Many proteins of each HDL subspecies exert related functions, for example in lipid transport, hemostasis, oxidation, or inflammation suggesting that specific functions beyond cholesterol efflux are exerted by distinct subspecies of HDL rather than the bulk of HDL. In agreement with this concept, a recent systems biology approach found distinct functions of HDL determined by clusters of distinct proteins and lipids carried by HDL with little overlap between the functions (Cardner et al. 2020). Moreover, in four prospective nested case–control studies, the presence or absence of distinct proteins was found to determine the association of apoA-I levels with incident cardiovascular events (Sacks et al. 2020). For example, apoA-I levels in particles that contain apoE or apoC-I but not their apoE or apoC-I- free counterparts showed the expected inverse association with incident ASCVD events. Vice versa, apoA-I levels in apoC-III-free particles but not particles containing apoC-III showed the expected inverse association with incident ASCVD events (Sacks et al. 2020). Cholesterol levels in apoC-III containing HDL even showed a positive association with incident ASCVD (Jensen et al. 2018). Moreover, apoC-III containing HDL was found to interfere with the capacity of HDL to inhibit the apoptosis of endothelial cells and to promote efflux from macrophages (Riwanto et al. 2013; Zvintzou et al. 2017). This makes apoC-III an interesting target for therapy beyond lowering of triglycerides (Zewinger et al. 2020). Other studies found the enrichment of HDL with either pulmonary surfactant protein B or serum amyloid protein A associated with increased risk of mortality in patients with diabetic end-stage nephropathy, heart failure, or CHD (Emmens et al. 2018; Kopecky et al. 2015; Zewinger et al. 2015).
Several mass spectrometric studies demonstrated gross alterations in the lipidome of HDL in patients with acute or chronic CHD as well as changes in response to statin therapy or body weight reduction (Cardner et al. 2020; Khan et al. 2018; Meikle et al. 2019; Orsoni et al. 2016; Sutter et al. 2015). However, to date, only signatures of lipid species in total plasma but not in HDL have been explored for their prognostic performance in prospective studies (Hilvo et al. 2019; Mundra et al. 2016). NMR-based studies identified some more general lipid traits of HDL to be associated with incident disease. However, they represent classes or subclasses rather than species of lipids and they are strongly intercorrelated with each other as well as measures of particle size or numbers so that they are not pursued as biomarkers beyond the latter indices (Cardner et al. 2020; Hafiane and Genest 2015; Rosenson et al. 2011).
3.2 Ongoing and Novel Drug Developments
After the failure of CETP inhibitors, only few drug developments targeting HDL have been continued or newly started. Some of the latter targets are pleiotropic and HDL is a bystander rather than the focus of these drug developments.
3.2.1 Reconstituted HDL, apoA-I Mimetic Peptides, and Recombinant LCAT
After infusions of artificially reconstituted HDL (rHDL) were found to reduce atherosclerosis in hypercholesterolemic rabbits, several formulations of rHDL were developed for investigation of their atheroprotective effects (He et al. 2021. Because rather large amounts of protein are needed, only short-term applications in acute clinical settings are feasible, for example in patients with acute coronary syndrome (ACS). rHDL containing phosphatidylcholines together with the recombinant apoA-I Milano variant (ETC-216, MDCO-216) or recombinant wild type apoA-I plus sphingomyelin (Cer001) or apoA-I isolated from plasma (CSL111, CSL112) were initially tested in phase II trials for their short-term effects on coronary atherosclerosis which was assessed by intravascular ultrasound of ACS patients. Whereas initial studies showed some regression of coronary atherosclerosis upon treatment with ETC.-216 or CSL111 (Tardif et al. 2007; Nissen et al. 2003), later larger studies with MDCO-216 or Cer001 did not (Nicholls et al. 2018a, b). Neither did Cer001 cause regression or prevent progression of carotid atherosclerosis in patients with genetic HDL deficiency (Zheng et al. 2020). Currently only one formulation – CSL112 – is further pursued by a large randomized and controlled phase III trial (ApoA-I Event Reducing in Ischemic Syndrome II = AEGIS II). Seventeen thousand four hundred patients with myocardial infarction are randomized to 4 weekly infusions of either 6 g CSL112 or placebo within 5 days of the event (Gibson et al. 2021). The primary outcome is the time to first occurrence of the composite of CV death, MI, or stroke through 90 days. Secondary outcomes include the total number of hospitalizations for coronary, cerebral, or peripheral ischemia through 90 days and time to first occurrence of the composite primary outcome through 180 and 365 days. Results are expected to become available in 2023.
In addition to rHDL containing full length apoA-I, also apoA-I mimetic peptides are developed for treatment of atherosclerosis. They showed promising results in vitro and in preclinical animal models. Three of them have been tested for safety and effects on HDL-C and HDL function. For two of them – L4F and D4F – results have been reported. They did not cause any changes in HDL-C or anti-inflammatory HDL functions. No results have been reported for ETC642. The clinical development of FX-5A is planned (Wolska et al. 2021).
Application of recombinant LCAT is an alternative strategy to increase HDL-C or promote HDL metabolism by substitution of components (Freeman et al. 2020). In a randomized controlled study 32 patients were treated with three weekly injections of different dosages of recombinant LCAT (MEDI6012) or placebo (Bonaca et al. 2021). Compared to placebo, MEDI6012 caused dose-dependent increases of HDL-C by 66% to 144% at day 19. Interestingly the initial bolus injection led to a more than 40% increase of HDL-C within 30 min. MEDI6012 caused neither any severe side effects nor the generation of neutralizing antibodies. In an ongoing phase IIb trial (REAL-TIMI 63B), the application of 2 dosages of MEDI6012 to ACS patients is currently investigated for its effect on infarct size in more than 400 ACS patients (https://clinicaltrials.gov/ct2/show/NCT03578809). Half of the patients receive additional 4-weekly injections of MEDI6012 or placebo for 12 weeks for investigation of LCAT’s effects on coronary calcification. Of note, recombinant LCAT is also attractive for the use as enzyme replacement in familial LCAT deficiency, however with the goal to prevent the development and progression of nephropathy in these patients (Freeman et al. 2020; Vaisman et al. 2019).
3.2.2 Apabetalone
Apabetalone (RVX208) is an inhibitor of bromodomain and extraterminal (BET) proteins that regulate the expression of multiple genes by interference with histone acetylation. RVX208 was initially developed because it strongly induced APOA1 gene expression in cultivated hepatocytes and caused a profound increase of HDL-C and apoA-I levels in non-human primates by 90 and 60%, respectively (Ghosh et al. 2017). Also mice responded with substantial increases of HDL cholesterol. Atherosclerosis was suppressed in apoE deficient mice. However, in humans treatment with apabetalone caused very moderate increases in HDL-C and apoA-I levels but also reduced CRP levels. The development of the drug was stopped after a recent phase III trial (BETonMACE) in patients with acute coronary syndrome and type 2 diabetes did not reveal any reduction of clinical events compared with placebo. (Ray et al. 2020).
3.2.3 PPAR Modulators
The PPARa modulator Pemafibrate is developed primarily for the treatment of hypertriglyceridemia and the related cardiovascular risk. In a phase II dose finding study, pemafibrate dose-dependently decreased triglycerides and increased HDL-C by up to 42% and 21%, respectively, compared to 30% and 14% by fenofibrate (Arai et al. 2017). In hypertriglyceridemic patients, treatment with pemafibrate caused increases of HDL-C by about 16% as well as CEC (Yamashita et al. 2018). In apoE2 knock-in mice, pemafibrate increased HDL-C, CEC as well as macrophage-to-feces reverse cholesterol transport, and reduced the extent of atherosclerotic lesions (Hennuyer et al. 2016). The PROMINENT trial investigates the effect of pemafibrate vs. placebo on cardiovascular outcomes of 10,000 participants with diabetes mellitus type 2, triglycerides 200–499 mg/dL (2.26–5.64 mmol/L), HDL-C level ≤40 mg/dL(1.03 mmol/L) during a maximal follow-up of 5 years (Pradhan et al. 2018).
3.2.4 ANGPTL3 and Endothelial Lipase
Angiopoietin-like protein 3 (ANGPTL3) is mainly produced by the liver and an endogenous inhibitor of both lipoprotein lipase and endothelial lipase (EL). After the discovery of loss of function mutations in the ANGPTL3 gene as a cause of panhypolipoproteinemia and reduced cardiovascular risk (Arca et al. 2020), antisense oligonucleotides as well as monoclonal antibodies were developed for the treatment of hypertriglyceridemia and hypercholesterolemia. In fact, treatment of hypertriglyceridemia with the antisense oligonucleotide Vupanorsen and refractory hypercholesterolemia with the monoclonal antibody Evinacumab caused pronounced decreases of triglyceride levels and LDL-C but also HDL-C (Gaudet et al. 2020; Rosenson et al. 2020) The clinical implication of the 20–30% decrease in HDL-C is not known but very likely reflects the increased activity of EL upon ANGPTL3 inhibition (Wu et al. 2020).
Conversely, also EL inhibitors are developed with the aim to increase HDL-C. Treatment of non-human primates with the monoclonal anti-EL antibody MEDI5884 dose-dependently increased HDL-C and apoA-I levels by up to 100% and 30%, respectively (Le Lay et al. 2021). In a phase I study, human volunteers also experienced increases in HDL-C and apoA-I as well as particle number and size. CEC and anti-inflammatory activities of HDL were also improved. However, endothelial lipase inhibition also caused increases in LDL-C, albeit more profoundly in non-human primates than in humans. In non-human primates this unwanted effect could be blocked by PCSK9 inhibition. Nevertheless, one must wonder whether the risk of increasing LDL-C is well taken, especially since loss-of-function alleles of LIPG encoding EL do not confer any cardiovascular risk reduction despite increasing HDL-C thus questioning the clinical utility of EL inhibition (Voight et al. 2012).
3.2.5 ApoC-III Inhibition
Antisense oligonucleotides against apoC-III (Volanesorsen) exert pronounced triglyceride lowering effects by reducing the production of VLDL as well as by promoting lipolysis and remnant removal by disinhibiting lipoprotein lipase and remnant receptors, respectively. Probably secondarily to the lowering of triglycerides, interference with apoC-III in patients with chylomicronemia also leads to increases of HDL-C levels by 40% (Witztum et al. 2019). Although not tested, one must assume that Volanesorsen also decreases the content of apoC-III in HDL. In view of the positive rather than inverse association of apoC-III containing HDL with cardiovascular outcomes (Jensen et al. 2018; Sacks et al. 2020); as well as the noxious effects of apoC-III on HDL functionality towards cholesterol efflux and endothelial survival and inflammation (Riwanto et al. 2013; Zewinger et al. 2020; Zvintzou et al. 2017), one may hypothesize that apoC-III inhibition also exerts anti-atherogenic effects by improving HDL function. However, this hypothesis needs to be tested.
3.2.6 HDL-C Lowering Therapies: Probucol and Androgens
For a long time, lowering of HDL-C has been considered as a safety issue in drug development. This has changed as the causal role of HDL in ASCVD has been questioned. Even more so, the association of high HDL-C with increased mortality and morbidity for certain diseases (Bowe et al. 2016a; Ko et al. 2016; Madsen et al. 2017) raises the question whether under certain conditions HDL-C lowering may be useful. In view of the genetic association of loss of function mutations in SR-BI with increased cardiovascular risk (Zanoni et al. 2016) and the finding of increased atherosclerosis in Scarb1 knock-out mice (Hoekstra and van Eck 2015), especially therapies that lower HDL-C by upregulation of SR-BI in the liver may be interesting.
Probucol is an old drug which was originally developed to exploit its anti-oxidative effects on LDL. Although it was shown to induce regression of atherosclerosis and xanthomas and has been rather widely used in Japan, the development and clinical application of probucol has not been consequently pursued (Yamashita et al. 2015). The main reason was the about 30% lowering of HDL-C. Activation of CETP and SR-BI has been elucidated as the underlying mechanism. In the most recent PROSPECTIVE trial, 876 Japanese patients with CHD and LDL-C ≥140 mg/dL without medication or those treated with lipid-lowering drugs received optimal lipid-lowering treatment together with placebo or probucol 500 mg/day. After 3 years, LDL-C and HDL-C were 8.5 mg/dL and 16.3 mg/dL, respectively, lower in the probucol than in the placebo group. The event rates did not differ significantly between the groups, although by trend, CHD events happened less frequently in the probucol group (Arai et al. 2021). Interestingly, the combined analysis of the PROMINENT and IMPACT trials showed reductions of cerebrovascular events, however in the absence of any effect on carotid atherosclerosis (Yamashita et al. 2021). Although futile, the data raise the question of whether probucol is a treatment option for patients with high HDL-C.
The stimulatory effects of testosterone on hepatic SR-BI expression are probably the main reasons for the substantial differences in HDL-C levels between males and females (Chiba-Falek et al. 2010; Langer et al. 2002). Even more so, the HDL-lowering effects of testosterone have contributed to the caution on the use of testosterone for the treatment of the aging male syndrome, transgender patients, or female sexual dysfunction as well as for male contraception (Thirumalai et al. 2015; Wu and von Eckardstein 2003). The effects of testosterone replacement on hard cardiovascular endpoints have not been investigated. However, a recent randomized controlled trial in 1007 men with overweight or obesity as well as disturbed or manifest diabetes at baseline showed benefits of 1,000 mg intramuscular testosterone vs. placebo injection on glycemic control and incidence of diabetes during 2 years of follow-up (Wittert et al. 2021).
3.3 Other Disease Targets
As late onset diseases, ASCVDs have not been rate limiting in the evolution of species. Therefore, one must envisage that the broad spectrum of HDL’s protective functions has rather evolved to prevent other diseases or secure survival and healing of their victims. Such diseases or their clinical complications may serve as more appropriate targets than ASCVD for the therapeutic exploitation of HDL (Von Eckardstein and Rohrer 2016). Indeed, recent epidemiological and genetic studies unraveled several associations of HDL-C and genetic loci intimately related to HDL metabolism with non-cardiovascular diseases as well as mortality (Table 3) (Kjeldsen et al. 2021a; Madsen et al. 2021). Please note the diverse directions of these associations which reach from inverse (diabetes, autoimmune diseases) over parabolic (infections, chronic kidney disease, mortality) to positive (Alzheimer’s disease, age related macular degeneration), re-emphasizing that the kinetics of HDL metabolism and HDL function rather than the concentration of HDL particles are relevant.
3.3.1 Diabetes
Low levels of HDL-C are frequent in patients with diabetes mellitus type 2. This finding even precedes the manifestation of hyperglycemia and is hence an indicator of increased risk for incident diabetes (Haase et al. 2015; Schmidt et al. 2005; von Eckardstein et al. 2000; White et al. 2016; Wilson et al. 2007). Because of multiple effects of insulin on HDL metabolism, most of which are indirect via free fatty acids or triglyceride-rich lipoproteins, these associations have been explained for a long time by reverse causality: diabetes and pre-diabetes cause low HDL-C rather than vice versa (Parhofer 2015; Vollenweider et al. 2015; von Eckardstein and Widmann 2014). However, increasing evidence from in vitro as well as in vivo studies indicates that HDL exerts protective functions on the function and survival of pancreatic beta cells as well as on the sensitivity of target cells to insulin (Cochran et al. 2021; Manandhar et al. 2020; Vollenweider et al. 2015; von Eckardstein and Widmann 2014; Yalcinkaya et al. 2020). Also mitochondrial function and thereby cellular energy metabolism is modulated by HDL (Lehti et al. 2013). In humans, the potentially anti-diabetic effects of HDL are best illustrated by the acute glucose lowering effect of rHDL infusion (Drew et al. 2009) as well as by the findings of post-hoc analyses of the CETP inhibitor trials: Participants who received the CETP inhibitors showed better glycemic control and experienced less often new-onset diabetes as compared to the placebo treated controls (Barter et al. 2011; Masson et al. 2018; Menon et al. 2020; Schwartz et al. 2020). Mendelian randomization studies yielded controversial results on the genetic causality of HDL-C in diabetes (Fall et al. 2015; Haase et al. 2015; White et al. 2016).
3.3.2 Chronic Kidney Disease
The Veterans Administration study showed a parabolic association of HDL-C with >30% declining estimated glomerular filtration rate (eGFR) or the incidence of eGFR <60 ml/min and also provided evidence for genetic causality (Bowe et al. 2016b). Mutations in APOA1, APOE; APOL1, and LCAT are causes of genetic nephropathies (Strazzella et al. 2021). However, it is not clear whether their pathogenesis involves HDL: Certain missense mutations in APOA1 cause familial amyloidosis, which also affect other organs (Zanoni and von Eckardstein 2020). Specific mutations in APOE cause lipid glomerulopathy which however has been suggested to develop in response to disturbed interactions of apoB containing lipoproteins with the LDL receptor or due to accumulation of the structural defective apoE (Saito et al. 2020). ApoL1 is the trypanolytic factor which is transported by a minor subfraction of HDL (Friedman and Pollak 2020). Certain apoL1 variants that protect the host from infections with Trypanosoma brucei rhodesiense and gambiense dramatically increase the risk of chronic kidney disease, notably focal segmental glomerulosclerosis, in their African and Afroamerican carriers (Friedman and Pollak 2020). LCAT deficiency is a classical HDL deficiency syndrome causing a nephropathy that eventually progresses to end-stage renal disease (Pavanello and Calabresi 2020). However, the pathogenic mechanism depends on the accumulation of lipoprotein X rather than the absence of HDL (Vaisman et al. 2019). This is best illustrated by the absence of nephropathy in patients with fish-eye disease where partial loss of LCAT causes the same decrease in HDL as classical LCAT deficiency, however no nephropathy (Pavanello and Calabresi 2020).
Treatment with fibrates improves albuminuria but worsens glomerular filtration rate (Speer et al. 2021). Niacin has no effect on renal function. Small studies also indicate protective effects of probucol towards acute kidney injury, for example of patients exposed to contrast agents (Xin et al. 2019). The pleiotropic effects of these drugs on lipoprotein metabolism and the lack of association between changes in renal function and HDL-C under their treatment do not allow any conclusion on the role of HDL modifying therapies for prevention or treatment of CKD. Furthermore the exploitation of HDL towards kidney disease is also hampered by the currently unknown mechanisms how HDL exerts renal protection by HDL. Since small HDL particles are undergoing glomerular filtration and tubular re-uptake by the megalin/cubilin co-receptors, it may be that HDL delivers protective molecules to the kidney, for example sphingosine-1-phosphate (Bisgaard and Christoffersen 2019; Strazzella et al. 2021).
3.3.3 Infections
The Copenhagen General Population study found U-shaped associations between HDL-C and the incidence of infections (Madsen et al. 2018). The associations with bacterial infections were stronger than with viral infections. Upon limited adjustment, gastroenteritis, bacterial pneumonia, skin and urinary tract infections, as well as sepsis were more prevalent among individuals with HDL-C <1.0 mmol/L as compared to individuals with higher HDL-C. Upon full adjustment the associations with gastroenteritis and pneumonia remained significant (Madsen et al. 2018). A preliminary Mendelian randomization analysis with two loci (CETP and LIPC) provided initial evidence of genetic causality (Madsen et al. 2018). Genetic causality also exists for the association of low HDL-C with the incidence of sepsis as well as with the chance of survival in patients with sepsis (Trinder et al. 2020). Evidence from population studies as well as experiments in genetic animal models points to the importance of CETP in this process (Blauw et al. 2020; Trinder et al. 2019, 2021). However also HDL particles per se as well as specific structural components of HDL exert several antibacterial activities such as binding and removal of lipopolysaccharides, protection of epithelial and endothelial barriers, or modulatory effects on leukocyte functions (Catapano et al. 2014; Meilhac et al. 2020; Pirillo et al. 2015; Robert et al. 2021; Rohatgi et al. 2021; Trakaki and Marsche 2021). The special association of HDL-C with gastroenteritis may also mirror an important role of HDL that is locally produced in the intestine (Ko et al. 2020). Similarly, the protection from pneumonia may mirror the high exposure of the lung to newly synthesized HDL due to first pass effects (Gordon et al. 2016). In sepsis models, genetically modified mice overexpressing human apoA-I showed improved survival (Meilhac et al. 2020; Morin et al. 2015). Interestingly, the clinical development of CSL111 was originally aiming at the treatment of sepsis since their infusion into volunteers exerted several beneficial effects on inflammation, coagulation, and fibrinolysis (Pajkrt et al. 1996, 1997). Most recent experiments in preclinical sepsis models demonstrated better survival of mice treated with CSL111 (Tanaka et al. 2020). The better chances of survival from sepsis by patients carrying low CETP activity alleles suggest CETP inhibitors as interesting drugs for the treatment of patients with sepsis (Trinder et al. 2019, 2021). However, an important caveat comes from the ILLUMINATE study where torcetrapib treatment was associated with excess mortality due to infections (Barter et al. 2007b).
Although the association of HDL-C with the incidence of viral infections was not statistically significant in the Copenhagen General Population Study (Madsen et al. 2018), it is noteworthy that at least in vitro HDL or apoA-I interferes with the entry or fusion of viruses with target cells (Meilhac et al. 2020; Pirillo et al. 2015). HDLs also induce viral inactivation by immune cells and protect cells from virus-induced damage (Pirillo et al. 2015). The COVID19 pandemia also raised the question of whether HDL interferes with SARS-CoV2 infections. In the UK Biobank study, a linear inverse and independent association was found between pre-infection HDL-C levels and the risk of hospitalization for severe COVID19 (Hilser et al. 2021; Lassale et al. 2021). Mendelian Randomization rather excluded any causal role of HDL in preventing SARS-CoV2 infection. (Hilser et al. 2021). Nevertheless, since SR-BI is an entry route of several viruses including SARS-CoV2 into cells (Pirillo et al. 2015; Wei et al. 2020), competition of this interaction by HDL is an intriguing hypothesis. In addition, the protective effects of HDL on the survival and function of cells may also help infected cells, for example of the lung epithelium or the endothelium (Robert et al. 2021), to combat and survive the entered viruses. Interestingly, dalcetrapib is currently tested towards its effect on the course of COVID19 infections. (Talasaz et al. 2021; https://clinicaltrials.gov/ct2/show/NCT04676867).
Finally, HDL also exerts protective activities towards infections with protozoa. The best example is the protection of humans from Trypanosoma brucei by apoL1 transported by a subfraction of HDL containing also haptoglobin related protein. This complex kills Trypanosome brucei by causing lysosomal swelling (Friedman and Pollak 2020). A similar HDL-related mechanism appears operative towards Leishmania (Samanovic et al. 2009).
3.3.4 Autoimmune Diseases
In an analysis of more than 110,000 participants of The Copenhagen General Population and the Copenhagen City Heart Study, low HDL-C concentrations were associated with elevated risk of developing the composite end point of 42 different autoimmune diseases (Madsen et al. 2019). Among them, the associations of celiac disease, idiopathic thrombocytopenic purpura, Sjögren’s disease, diabetes type 1, inflammatory bowel diseases, and Graves’ disease showed the strongest and individually significant associations with HDL-C. Currently, no data are available to prove or disprove causality of these associations (Madsen et al. 2021). Neither are the mechanisms understood. They may involve immunomodulatory effects of HDL which are relevant in the development of auto-immunity (Catapano et al. 2014; Pirillo et al. 2015; Rohatgi et al. 2021; Trakaki and Marsche 2021) or protective effects of HDL towards organs attacked by the immune system so that the onset of organ damage or failure and thereby the clinical diagnosis of manifest disease is delayed. For example, the anti-apoptotic effects of HDL on pancreatic beta cells may delay the loss of insulin production in the course of progressing type 1 diabetes (von Eckardstein and Widmann 2014; Yalcinkaya et al. 2020). For inflammatory bowel diseases, preclinical models generated evidence for the therapeutic potential of HDL: Intestinal inflammation was increased in Apoa1 knock-out mice but decreased in mice which overexpressed human APOA1 or were fed with apoA-I mimetic peptides (Gerster et al. 2014; Meriwether et al. 2019; Nowacki et al. 2016).
3.3.5 Cancer
Several epidemiological studies found inverse associations between HDL-C and cancer in general as well as specific cancers such as breast cancer or colorectal cancer (Ganjali et al. 2021; Madsen et al. 2021; Pirro et al. 2018). Currently, there is no evidence of causality. However, several confounders of low HDL-C are associated with increased risk of several cancers, for example smoking, overweight and obesity, type 2 diabetes, or chronic inflammatory diseases. There is hence some likelihood that low HDL-C is a confounder of other causal risk factors rather than reflecting loss of anti-cancer functions. Even if HDL is not causally related to cancer, it will offer opportunities for therapeutic or diagnostic exploitation: probably to satisfy their high need of cholesterol for growth, many cancers show a high expression of lipoprotein receptors including SR-BI (Hoekstra and Sorci-Thomas 2017; Kinslechner et al. 2018; Velagapudi et al. 2018). This can be exploited by using rHDL for the delivery of anti-cancer drugs or tracers for imaging. In fact, according experiments in preclinical models showed promising results (Morin et al. 2018; Rajora and Zheng 2016).
3.3.6 Behind the Blood Brain Barrier: Alzheimer’s Disease and Age Related Macular Degeneration
Until recently, only cross-sectional studies and smaller prospective cohort studies described associations of HDL-C with neurodegenerative diseases including Alzheimer’s disease (AD) and age related macular degeneration (AMD). They reported discrepant associations ranging from inverse over none to positive (Kjeldsen et al. 2021a). The situation became clearer but also surprising by recent reports of analyses in the Copenhagen General Population and Copenhagen City Heart Studies. High HDL-C levels >95th percentiles increase the risk of dementia and AD (Kjeldsen et al. 2021b). This association became even more prominent after adjustment for APOE genotypes. Also the risk of AMD was found to increase with HDL-C and even more so with apoA-I levels (Nordestgaard et al. 2021) confirming data on more than 30,000 individuals from the EYE-RISK and European Eye Epidemiology Consortia (Colijn et al. 2019). Of note, in that study higher HDL-C was most strongly associated with increased risk of early AMD. Mendelian Randomization studies found evidence for genetic causality of higher HDL-C for the higher risk of AMD but not AD (Burgess and Smith 2017; Chen et al. 2010; Fan et al. 2017; Neale et al. 2010; Ostergaard et al. 2015; Proitsi et al. 2014), the latter perhaps because of the non-linear relationship. CETP, APOE, and LIPC were important drivers of the genetic association between HDL-C and AMD (Colijn et al. 2019; Chen et al. 2010; Neale et al. 2010). However, candidate gene approaches as well as genome-wide association studies found ABCA1 as a genetic determinant of both AD and AMD risks (Bellenguez et al. 2020; Nordestgaard et al. 2015; Fritsche et al. 2016). Likewise, tissue specific knock-out experiments in mice indicate that loss of ABCA1 function in neurons and retinal pigment epithelial cells compromise neurocognitive and retinal functions, respectively (Behl et al. 2021; Storti et al. 2019). These discrepant associations of AD and AMD with high HDL-C levels in peripheral blood but locally reduced cholesterol efflux in the brain and retina probably reflect the tight separation of these compartments by the blood brain barrier. Some HDL functions in the brain are executed by HDL-like particles that contain apoE instead of apoA-I and that are produced by astrocytes within the central nervous system (Button et al. 2019). Especially the association of APOE genotypes with risk of AD, although mechanistically not resolved, has been traditionally ascribed to apoE endogenously produced by the CNS rather than supplied by the systemic circulation. Of note however, also the concentration of apoE in plasma of peripheral blood has been associated with risk of AD (Rasmussen et al. 2018). Moreover, anti-apoA-I immunoreactivity is found in the brain. These findings indicate a limiting role of the blood brain barrier for any protective role of HDL in CNS diseases such as AD and AMD. Any therapeutic exploitation of HDL for CNS diseases will have to address the interaction of HDL with the blood brain barrier, either as a target of HDL’s protective actions, for example in amyloid beta clearance, or as a barrier that must be surmounted by HDL to exert protective functions within the CNS (Button et al. 2019; Robert et al. 2021). The latter is also important for the use of HDL-like nanoparticles that are currently investigated as vehicles for drug delivery into the brain (D'Arrigo 2020; Kadiyala et al. 2019; Kim et al. 2020).
3.4 Implications for Nowaday’s Clinical Practice
As the result of the futile intervention trials, HDL-C unlike LDL-C has not become any treatment goal (Grundy et al. 2019; Mach et al. 2020). However, HDL-C continues to be part of ASCVD risk assessment, both directly and indirectly by using HDL-C for the calculation of nonHDL cholesterol or even LDL cholesterol (Grundy et al. 2019; Mach et al. 2020; Martin et al. 2013; Sampson et al. 2020). Especially in asymptomatic patients without any lipid modifying treatment, a low HDL-C level is considered as a risk factor of developing ASCVD. As such, HDL-C is a component of most clinical risk prediction rules that are promoted by guidelines for the prevention of ASCVD (Grundy et al. 2019; Mach et al. 2020). Unfortunately, these algorithms do not realize the discontinuous relationship of HDL-C with risk, but still de-escalate risk estimates in individuals with very high HDL-C levels. This may be a reason why, for example, in the Copenhagen City Heart study the inclusion of HDL-C impaired rather than improved the prognostic performance of SCORE promoted by ESC and EAS (Mortensen et al. 2015). With the same reasoning, clinical laboratories as well as clinicians and practitioners should stop the still widely spread clinical practice to calculate total cholesterol/HDL-C- or LDL-C/HDL-C ratios because they may underestimate the risk of individuals with high HDL-C (de Wolf et al. 2020; Nordestgaard et al. 2020). The same concern may relate to the atherogenic index – the logarithmically transformed ratio of the molar concentrations of plasma triglycerides to HDL-C – especially because some studies found the joint presence of high HDL-C and hypertriglyceridemia associated with increased ASCVD risk (Jeppesen et al. 1998; von Eckardstein et al. 1999). Finally, low HDL-C continues to be a component of definitions for the metabolic syndrome, which indicates increased risks not only for ASCVD but also for diabetes and other obesity related diseases (Alberti et al. 2009).
Whether reflecting compromised anti-atherogenic functions or indicating indirectly a proatherogenic situation, the finding of low HDL-C levels should prompt physicians and patients to optimize the control of other risk factors (März et al. 2017). The lost association of low HDL-C with increased risk upon intensive statin therapy indicates the importance of consequent LDL-C lowering in these patients. Additional important measures include cessation of smoking, correction of obesity and overweight, and treatment of hypertension. In view of the inconsistent outcomes of according randomized controlled trials it is a matter of uncertainty and controversy whether or not hypertriglyceridemia which frequently confounds low HDL-C should be targeted by drug treatment (Ginsberg et al. 2021).
The discussion on therapeutic consequences of high HDL-C levels is in its infancy. It is not clear whether the associations of high HDL-C with increased mortality and risks of CKD, infectious diseases, AD, or AMD are causal. An important potential confounder is excess alcohol consumption (Madsen et al. 2021). Potential candidates for HDL-C lowering drugs are probucol, ANGPTL3 inhibitors, or androgens. In the absence of HDL-C lowering treatments with proven efficacy, it is advisable to focus on risk factor control also in patients with high HDL-C as described for patients with low HDL-C.
References
Abdel-Razek O, Sadananda SN, Li X et al (2018) Increased prevalence of clinical and subclinical atherosclerosis in patients with damaging mutations in ABCA1 or APOA1. J Clin Lipidol 12:116–121
ACCORD Study Group, Ginsberg HN, Elam MB, Lovato LC, Crouse JR 3rd, Leiter LA, Linz P, Friedewald WT, Buse JB, Gerstein HC, Probstfield J, Grimm RH, Ismail-Beigi F, Bigger JT, Goff DC Jr, Cushman WC, Simons-Morton DG, Byington RP (2010) Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med 362(17):1563–1574. Erratum in: N Engl J Med. 2010 May 6;362(18):1748. https://doi.org/10.1056/NEJMoa1001282
Agerholm-Larsen B, Nordestgaard BG, Steffensen R, Jensen G, Tybjaerg-Hansen A (2000) Elevated HDL cholesterol is a risk factor for ischemic heart disease in white women when caused by a common mutation in the cholesteryl ester transfer protein gene. Circulation 101(16):1907–1912. https://doi.org/10.1161/01.cir.101.16.1907
AIM-HIGH Investigators, Boden WE, Probstfield JL, Anderson T, Chaitman BR, Desvignes-Nickens P, Koprowicz K, McBride R, Teo K, Weintraub W (2011) Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med 365(24):2255–2267. Erratum in: N Engl J Med. 2012 Jul 12;367(2):189. https://doi.org/10.1056/NEJMoa1107579
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA et al (2009) Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120:1640–1645
Anastasius M, Luquain-Costaz C, Kockx M, Jessup W, Kritharides L (2018) A critical appraisal of the measurement of serum ‘cholesterol efflux capacity’ and its use as surrogate marker of risk of cardiovascular disease. Biochim Biophys Acta Mol Cell Biol Lipids 1863(10):1257–1273. https://doi.org/10.1016/j.bbalip.2018.08.002
Annema W, von Eckardstein A (2013) High-density lipoproteins. Multifunctional but vulnerable protections from atherosclerosis. Circ J 77(10):2432–2448. https://doi.org/10.1253/circj.cj-13-1025
Annema W, von Eckardstein A (2016) Dysfunctional high-density lipoproteins in coronary heart disease: implications for diagnostics and therapy. Transl Res 173:30–57. https://doi.org/10.1016/j.trsl.2016.02.008
Arai H, Yamashita S, Yokote K, Araki E, Suganami H, Ishibashi S, K-877 Study Group (2017) Efficacy and safety of K-877, a novel selective peroxisome proliferator-activated receptor α modulator (SPPARMα), in combination with statin treatment: two randomised, double-blind, placebo-controlled clinical trials in patients with dyslipidaemia. Atherosclerosis 261:144–152. https://doi.org/10.1016/j.atherosclerosis.2017.03.032
Arai H, Bujo H, Masuda D, Ishibashi T, Nakagawa S, Tanabe K, Kagimura T, Kang HJ, Kim MH, Sung J, Kim SH, Kim CH, Park JE, Ge J, Oh BH, Kita T, Saito Y, Fukushima M, Matsuzawa Y, Yamashita S (2021) Integrated analysis of two probucol trials for the secondary prevention of atherosclerotic cardiovascular events: PROSPECTIVE and IMPACT. J Atheroscler Thromb. https://doi.org/10.5551/jat.62821
Arca M, D'Erasmo L, Minicocci I (2020) Familial combined hypolipidemia:angiopoietin-like protein-3 deficiency. Curr Opin Lipidol 31(2):41–48. https://doi.org/10.1097/MOL.0000000000000668
Arsenault BJ, Lemieux I, Després JP, Gagnon P, Wareham NJ, Stroes ES, Kastelein JJ, Khaw KT, Boekholdt SM (2009) HDL particle size and the risk of coronary heart disease in apparently healthy men and women: the EPIC-Norfolk prospective population study. Atherosclerosis 206(1):276–281. https://doi.org/10.1016/j.atherosclerosis.2009.01.044
Assmann G, Schulte H, von Eckardstein A, Huang Y (1996) High-density lipoprotein cholesterol as a predictor of coronary heart disease risk. The PROCAM experience and pathophysiological implications for reverse cholesterol transport. Atherosclerosis 124 Suppl:S11–S20. https://doi.org/10.1016/0021-9150(96)05852-2
Barter P, Gotto AM, LaRosa JC, Maroni J, Szarek M, Grundy SM, Kastelein JJ, Bittner V, Fruchart JC, Treating to New Targets Investigators (2007a) HDL cholesterol, very low levels of LDL cholesterol, and cardiovascular events. N Engl J Med 357(13):1301–1310. https://doi.org/10.1056/NEJMoa064278
Barter PJ, Caulfield M, Eriksson M, Grundy SM, Kastelein JJ, Komajda M, Lopez-Sendon J, Mosca L, Tardif JC, Waters DD, Shear CL, Revkin JH, Buhr KA, Fisher MR, Tall AR, Brewer B, ILLUMINATE Investigators (2007b) Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med 357(21):2109–2122. https://doi.org/10.1056/NEJMoa0706628
Barter PJ, Rye K-A, Tardif J-C, Waters DD, Boekholdt SM, Breazna A, Kastelein JJ (2011) Effect of torcetrapib on glucose, insulin, and hemoglobin a 1c in subjects in the investigation of lipid level management to understand its impact in atherosclerotic events (ILLUMINATE) trial. Circulation 124:555–562
Behl T, Kaur I, Sehgal A, Kumar A, Uddin MS, Bungau S (2021) The interplay of ABC transporters in Aβ translocation and cholesterol metabolism: implicating their roles in Alzheimer’s disease. Mol Neurobiol 58(4):1564–1582. https://doi.org/10.1007/s12035-020-02211-x
Bellenguez C, Küçükali F, Jansen I, Andrade V, Moreno-grau S (2020) New insights on the genetic etiology of Alzheimer’s and related dementia. MedRxiv:1–35. https://doi.org/10.1101/2020.10.01.20200659
Benn M, Nordestgaard BG (2018) From genome-wide association studies to Mendelian randomization: novel opportunities for understanding cardiovascular disease causality, pathogenesis, prevention, and treatment. Cardiovasc Res 114(9):1192–1208. https://doi.org/10.1093/cvr/cvy045
Bezafibrate Infarction Prevention (BIP) Study (2000) Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery disease. Circulation 102(1):21–27. https://doi.org/10.1161/01.cir.102.1.21
Bisgaard LS, Christoffersen C (2019) Apolipoprotein M/sphingosine-1-phosphate:novel effects on lipids, inflammation and kidney biology. Curr Opin Lipidol 30(3):212–217. https://doi.org/10.1097/MOL.0000000000000606
Blauw LL, Wang Y, Willems van Dijk K, Rensen PCN (2020) A novel role for CETP as immunological gatekeeper: raising HDL to cure sepsis? Trends Endocrinol Metab 31(5):334–343. https://doi.org/10.1016/j.tem.2020.01.003
Boekholdt SM, Arsenault BJ, Hovingh GK, Mora S, Pedersen TR, Larosa JC, Welch KM, Amarenco P, Demicco DA, Tonkin AM, Sullivan DR, Kirby A, Colhoun HM, Hitman GA, Betteridge DJ, Durrington PN, Clearfield MB, Downs JR, Gotto AM Jr, Ridker PM, Kastelein JJ (2013) Levels and changes of HDL cholesterol and apolipoprotein A-I in relation to risk of cardiovascular events among statin-treated patients: a meta-analysis. Circulation 128(14):1504–1512. https://doi.org/10.1161/CIRCULATIONAHA.113.002670
Bolibar I, von Eckardstein A, Assmann G, Thompson S, ECAT Angina Pectoris Study Group. European Concerted Action on Thrombosis and Disabilities (2000) Short-term prognostic value of lipid measurements in patients with angina pectoris.The ECAT Angina Pectoris Study Group: European Concerted Action on Thrombosis and Disabilities. Thromb Haemost 84(6):955–960
Bonaca MP, George RT, Morrow DA, Bergmark BA, Park JG, Abuhatzira L, Vavere AL, Karathanasis SK, Jin C, She D, Hirshberg B, Hsia J, Sabatine MS (2021) Recombinant human lecithin-cholesterol acyltransferase in patients with atherosclerosis: phase 2a primary results and phase 2b design. Eur Heart J Cardiovasc Pharmacother:pvab001. https://doi.org/10.1093/ehjcvp/pvab001
Borén J, Chapman MJ, Krauss RM, Packard CJ, Bentzon JF, Binder CJ, Daemen MJ, Demer LL, Hegele RA, Nicholls SJ, Nordestgaard BG, Watts GF, Bruckert E, Fazio S, Ference BA, Graham I, Horton JD, Landmesser U, Laufs U, Masana L, Pasterkamp G, Raal FJ, Ray KK, Schunkert H, Taskinen MR, van de Sluis B, Wiklund O, Tokgozoglu L, Catapano AL, Ginsberg HN (2020) Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 41(24):2313–2330. https://doi.org/10.1093/eurheartj/ehz962
Borggreve SE, Hillege HL, Wolffenbuttel BHR, De Jong PE, Zuurman MW, Van Der Steege G, Van Tol A, Dullaart RPF (2006) An increased coronary risk is paradoxically associated with common cholesteryl ester transfer protein gene variations that relate to higher high-density lipoprotein cholesterol: a population-based study. J Clin Endocrinol Metab 91:3382–3388
Bowe B, Xie Y, Xian H, Balasubramanian S, Zayed MA, Al-Aly Z (2016a) High density lipoprotein cholesterol and the risk of all-cause mortality among U.S. veterans. Clin J Am Soc Nephrol 11(10):1784–1793. https://doi.org/10.2215/CJN.00730116
Bowe B, Xie Y, Xian H, Balasubramanian S, Al-Aly Z (2016b) Low levels of high-density lipoprotein cholesterol increase the risk of incident kidney disease and its progression. Kidney Int 89(4):886–896. https://doi.org/10.1016/j.kint.2015.12.034
Brodeur MR, Rhainds D, Charpentier D, Mihalache-Avram T, Mecteau M, Brand G, Chaput E, Perez A, Niesor EJ, Rhéaume E, Maugeais C, Tardif JC (2017) Dalcetrapib and anacetrapib differently impact HDL structure and function in rabbits and monkeys. J Lipid Res 58(7):1282–1291. https://doi.org/10.1194/jlr.M068940
Burgess S, Smith GD (2017) Mendelian randomization implicates high-density lipoprotein cholesterol–associated mechanisms in etiology of age-related macular degeneration. Ophthalmology 124:1165–1174
Button EB, Robert J, Caffrey TM, Fan J, Zhao W, Wellington CL (2019) HDL from an Alzheimer’s disease perspective. Curr Opin Lipidol 30(3):224–234. https://doi.org/10.1097/MOL.0000000000000604
Cardner M, Yalcinkaya M, Goetze S, Luca E, Balaz M, Hunjadi M, Hartung J, Shemet A, Kraenkel N, Radosavljevic S, Keel M, Othman A, Karsai G, Hornemann T, Claassen M, Liebisch G, Carreira E, Ritsch A, Landmesser U, Krützfeldt J, Wolfrum C, Wollscheid B, Beerenwinkel N, Rohrer L, von Eckardstein A (2020) Structure-function relationships of HDL in diabetes and coronary heart disease. JCI Insight 5(1):e131491. https://doi.org/10.1172/jci.insight.131491
Catapano AL, Pirillo A, Bonacina F, Norata GD (2014) HDL in innate and adaptive immunity. Cardiovasc Res 103:372–383
Chandra A, Neeland IJ, Das SR, Khera A, Turer AT, Ayers CR, McGuire DK, Rohatgi A (2015) Relation of black race between high density lipoprotein cholesterol content, high density lipoprotein particles and coronary events (from the Dallas Heart Study). Am J Cardiol 115(7):890–894. https://doi.org/10.1016/j.amjcard.2015.01.015
Chapman MJ, Le Goff W, Guerin M, Kontush A (2010) Cholesteryl ester transfer protein: at the heart of the action of lipid-modulating therapy with statins, fibrates, niacin, and cholesteryl ester transfer protein inhibitors. Eur Heart J 31(2):149–164. https://doi.org/10.1093/eurheartj/ehp399
Chen W, Stambolian D, Edwards AO, Branham KE, Othman M, Jakobsdottir J, Tosakulwong N, Pericak-Vance MA, Campochiaro PA, Klein ML, Tan PL, Conley YP, Kanda A, Kopplin L, Li Y, Augustaitis KJ, Karoukis AJ, Scott WK, Agarwal A, Kovach JL, Schwartz SG, Postel EA, Brooks M, Baratz KH, Brown WL, Complications of Age-Related Macular Degeneration Prevention Trial Research Group, Brucker AJ, Orlin A, Brown G, Ho A, Regillo C, Donoso L, Tian L, Kaderli B, Hadley D, Hagstrom SA, Peachey NS, Klein R, Klein BE, Gotoh N, Yamashiro K, Ferris Iii F, Fagerness JA, Reynolds R, Farrer LA, Kim IK, Miller JW, Cortón M, Carracedo A, Sanchez-Salorio M, Pugh EW, Doheny KF, Brion M, Deangelis MM, Weeks DE, Zack DJ, Chew EY, Heckenlively JR, Yoshimura N, Iyengar SK, Francis PJ, Katsanis N, Seddon JM, Haines JL, Gorin MB, Abecasis GR, Swaroop A (2010) Genetic variants near TIMP3 and high-density lipoprotein-associated loci influence susceptibility to age-related macular degeneration. Proc Natl Acad Sci U S A 107(16):7401–7406. https://doi.org/10.1073/pnas.0912702107
Chiba-Falek O, Nichols M, Suchindran S, Guyton J, Ginsburg GS, Barrett-Connor E, McCarthy JJ (2010) Impact of gene variants on sex-specific regulation of human Scavenger receptor class B type 1 (SR-BI) expression in liver and association with lipid levels in a population-based study. BMC Med Genet 11:9. https://doi.org/10.1186/1471-2350-11-9
Christoffersen C, Obinata H, Kumaraswamy SB, Galvani S, Ahnström J, Sevvana M, Egerer-Sieber C, Muller YA, Hla T, Nielsen LB, Dahlbäck B (2011) Endothelium-protective sphingosine-1-phosphate provided by HDL-associated apolipoprotein. MProc Natl Acad Sci U S A 108(23):9613–9618. https://doi.org/10.1073/pnas.1103187108
Cochran BJ, Ong KL, Manandhar B, Rye KA (2021) High density lipoproteins and diabetes. Cell 10(4):850. https://doi.org/10.3390/cells10040850
Colantonio LD, Bittner V, Reynolds K, Levitan EB, Rosenson RS, Banach M, Kent ST, Derose SF, Zhou H, Safford MM, Muntner P (2016) Association of serum lipids and coronary heart disease in contemporary observational studies. Circulation 133(3):256–264. https://doi.org/10.1161/CIRCULATIONAHA.115.011646
Colijn JM, den Hollander AI, Demirkan A, Cougnard-Grégoire A, Verzijden T, Kersten E, Meester-Smoor MA, Merle BMJ, Papageorgiou G, Ahmad S, Mulder MT, Costa MA, Benlian P, Bertelsen G, Bron AM, Claes B, Creuzot-Garcher C, Erke MG, Fauser S, Foster PJ, Hammond CJ, Hense HW, Hoyng CB, Khawaja AP, Korobelnik JF, Piermarocchi S, Segato T, Silva R, Souied EH, Williams KM, van Duijn CM, Delcourt C, Klaver CCW, European Eye Epidemiology Consortium, EYE-RISK Consortium (2019) Increased high-density lipoprotein levels associated with age-related macular degeneration: evidence from the EYE-RISK and European eye epidemiology consortia. Ophthalmology 126(3):393–406. https://doi.org/10.1016/j.ophtha.2018.09.045
Crosby JH, Peloso GM, Auer PL, Crosslin DR, Stitziel N, Lange LA, Lu Y, Tang ZZ, Zhang H, Hindy G et al (2014) Loss-of-function mutations in APOC3, triglycerides, and coronary disease. N Engl J Med 371:22–31
Damen MSMA, Popa CD, Netea MG, Dinarello CA, Joosten LAB (2017) Interleukin-32 in chronic inflammatory conditions is associated with a higher risk of cardiovascular diseases. Atherosclerosis 264:83–91. https://doi.org/10.1016/j.atherosclerosis.2017.07.005
D'Arrigo JS (2020) Biomimetic nanocarrier targeting drug(s) to upstream-receptor mechanisms in dementia: focusing on linking pathogenic cascades. Biomimetics (Basel) 5(1):11. https://doi.org/10.3390/biomimetics5010011
de Haan W, de Vries-van der Weij J, van der Hoorn JW, Gautier T, van der Hoogt CC, Westerterp M, Romijn JA, Jukema JW, Havekes LM, Princen HM, Rensen PC (2008) Torcetrapib does not reduce atherosclerosis beyond atorvastatin and induces more proinflammatory lesions than atorvastatin. Circulation 117(19):2515–2522. https://doi.org/10.1161/CIRCULATIONAHA.107.761965
De Wolf HA, Langlois MR, Suvisaari J, Aakre KM, Baum H, Collinson P, Duff CJ, Gruson D, Hammerer-Lercher A, Pulkki K, Stankovic S, Stavljenic-Rukavina A, Laitinen P, EFLM Task Group on Cardiac Markers (2020) How well do laboratories adhere to recommended guidelines for dyslipidaemia management in Europe? The cardiac MARker guideline uptake in Europe (CAMARGUE) study. Clin Chim Acta 508:267–272. https://doi.org/10.1016/j.cca.2020.05.038
Ditah C, Otvos J, Nassar H, Shaham D, Sinnreich R, Kark JD (2016) Small and medium sized HDL particles are protectively associated with coronary calcification in a cross-sectional population-based sample. Atherosclerosis 251:124–131. https://doi.org/10.1016/j.atherosclerosis.2016.06.010
Drew BG, Duffy SJ, Formosa MF, Natoli AK, Henstridge DC, Penfold SA, Thomas WG, Mukhamedova N, de Courten B, Forbes JM et al (2009) High-density lipoprotein modulates glucose metabolism in patients with type 2 diabetes mellitus. Circulation 119:2103–2111
El Harchaoui K, Arsenault BJ, Franssen R, Després JP, Hovingh GK, Stroes ES, Otvos JD, Wareham NJ, Kastelein JJ, Khaw KT, Boekholdt SM (2009) High-density lipoprotein particle size and concentration and coronary risk. Ann Intern Med 150(2):84–93. https://doi.org/10.7326/0003-4819-150-2-200901200-00006
Emerging Risk Factors Collaboration, Di Angelantonio E, Sarwar N, Perry P, Kaptoge S, Ray KK, Thompson A, Wood AM, Lewington S, Sattar N, Packard CJ, Collins R, Thompson SG, Danesh J (2009) Major lipids, apolipoproteins, and risk of vascular disease. JAMA 302(18):1993–2000. https://doi.org/10.1001/jama.2009.1619
Emmens JE, Jones DJL, Cao TH, Chan DCS, Romaine SPR, Quinn PA, Anker SD, Cleland JG, Dickstein K, Filippatos G, Hillege HL, Lang CC, Ponikowski P, Samani NJ, van Veldhuisen DJ, Zannad F, Zwinderman AH, Metra M, de Boer RA, Voors AA, Ng LL (2018) Proteomic diversity of high-density lipoprotein explains its association with clinical outcome in patients with heart failure. Eur J Heart Fail 20(2):260–267. https://doi.org/10.1002/ejhf.1101
Fall T, Xie W, Poon W, Yaghootkar H, Mägi R, Knowles JW, Lyssenko V, Weedon M, Frayling TM, Ingelsson E (2015) Using genetic variants to assess the relationship between circulating lipids and type 2 diabetes. Diabetes 64:2676–2684
Fan Q, Maranville JC, Fritsche L, Sim X, Cheung CMG, Chen LJ, Gorski M, Yamashiro K, Ahn J, Laude A et al (2017) HDL-cholesterol levels and risk of age-related macular degeneration: a multiethnic genetic study using Mendelian randomization. Int J Epidemiol 46:1891–1902
Ference BA, Kastelein JJP, Ginsberg HN, Chapman MJ, Nicholls SJ, Ray KK, Packard CJ, Laufs U, Brook RD, Oliver-Williams C, Butterworth AS, Danesh J, Smith GD, Catapano AL, Sabatine MS (2017) Association of genetic variants related to CETP inhibitors and statins with lipoprotein levels and cardiovascular risk. JAMA 318(10):947–956. https://doi.org/10.1001/jama.2017.11467
Freeman LA, Karathanasis SK, Remaley AT (2020) Novel lecithin: cholesterol acyltransferase-based therapeutic approaches. Curr Opin Lipidol 31(2):71–79. https://doi.org/10.1097/MOL.0000000000000673
Frick MH, Elo O, Haapa K, Heinonen OP, Heinsalmi P, Helo P, Huttunen JK, Kaitaniemi P, Koskinen P, Manninen V et al (1987) Helsinki heart study: primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N Engl J Med 317(20):1237–1245. https://doi.org/10.1056/NEJM198711123172001
Friedman DJ, Pollak MR (2020) APOL1 and kidney disease: from genetics to biology. Annu Rev Physiol 82:323–342. https://doi.org/10.1146/annurev-physiol-021119-034345
Fritsche LG, Igl W, Bailey JNC, Grassmann F, Sengupta S, Bragg-Gresham JL, Burdon KP, Hebbring SJ, Wen C, Gorski M et al (2016) A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nat Genet 48:134–143
Furtado JD, Yamamoto R, Melchior JT, Andraski AB, Gamez-Guerrero M, Mulcahy P, He Z, Cai T, Davidson WS, Sacks FM (2018) Distinct proteomic signatures in 16 HDL (high-density lipoprotein) subspecies. Arterioscler Thromb Vasc Biol 38(12):2827–2842. https://doi.org/10.1161/ATVBAHA.118.311607
Ganjali S, Banach M, Pirro M, Fras Z, Sahebkar A (2021) HDL and cancer – causality still needs to be confirmed? Update 2020. Semin Cancer Biol 73:169–177. https://doi.org/10.1016/j.semcancer.2020.10.007
Gaudet D, Karwatowska-Prokopczuk E, Baum SJ, Hurh E, Kingsbury J, Bartlett VJ, Figueroa AL, Piscitelli P, Singleton W, Witztum JL, Geary RS, Tsimikas S, O'Dea LSL, Vupanorsen Study Investigators (2020) Vupanorsen, an N-acetyl galactosamine-conjugated antisense drug to ANGPTL3 mRNA, lowers triglycerides and atherogenic lipoproteins in patients with diabetes, hepatic steatosis, and hypertriglyceridaemia. Eur Heart J 41(40):3936–3945. https://doi.org/10.1093/eurheartj/ehaa689
Geller AS, Polisecki EY, Diffenderfer MR et al (2018) Genetic and secondary causes of severe HDL deficiency and cardiovascular disease. J Lipid Res 59:2421–2435
Gencer B, Mach F (2020) Potential of lipoprotein(a)-lowering strategies in treating coronary artery disease. Drugs 80(3):229–239. https://doi.org/10.1007/s40265-019-01243-5
Gerster R, Eloranta JJ, Hausmann M, Ruiz PA, Cosin-Roger J, Terhalle A, Ziegler U, Kullak-Ublick GA, von Eckardstein A, Rogler G (2014) Anti-inflammatory function of high-density lipoproteins via autophagy of IκB kinase. Cell Mol Gastroenterol Hepatol 1(2):171–187.e1. https://doi.org/10.1016/j.jcmgh.2014.12.006
Ghosh GC, Bhadra R, Ghosh RK, Banerjee K, Gupta A (2017) RVX 208: a novel BET protein inhibitor, role as an inducer of apo A-I/HDL and beyond. Cardiovasc Ther 35(4). https://doi.org/10.1111/1755-5922.12265
Gibson CM, Kastelein JJP, Phillips AT, Aylward PE, Yee MK, Tendera M, Nicholls SJ, Pocock S, Goodman SG, Alexander JH, Lincoff AM, Bode C, Duffy D, Heise M, Berman G, Mears SJ, Tricoci P, Deckelbaum LI, Steg PG et al (2021) Rationale and design of ApoA-I event reducing in ischemic syndromes II (AEGIS-II): a phase 3, multicenter, double-blind, randomized, placebo-controlled, parallel-group study to investigate the efficacy and safety of CSL112 in subjects after acute myocardial infarction. Am Heart J 231:121–127
Ginsberg HN, Packard CJ, Chapman JM, Borén J, Aguilar-Salinas CA, Averna M, Ference BA, Gaudet D, Hegele RA, Kersten S, Lewis LF, Lichtenstein AH, Moulin P, Nordestgaard BG, Remaley AT, Staels, ESG S, Taskinen MR, Tokgözoğlu LS, Tybjaerg-Hansen A, Stock JK, Catapano AL (2021) Triglyceride-rich lipoproteins and their remnants: metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies. A consensus statement from the European Atherosclerosis Society. Eur Heart J. (in press)
Gordon EM, Figueroa DM, Barochia AV, Yao X, Levine SJ (2016) High-density lipoproteins and apolipoprotein A-I: potential new players in the prevention and treatment of lung disease. Front Pharmacol 7:323. https://doi.org/10.3389/fphar.2016.00323
Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, Braun LT, de Ferranti S, Faiella-Tommasino J, Forman DE, Goldberg R, Heidenreich PA, Hlatky MA, Jones DW, Lloyd-Jones D, Lopez-Pajares N, Ndumele CE, Orringer CE, Peralta CA, Saseen JJ, Smith SC Jr, Sperling L, Virani SS, Yeboah J (2019) 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice guidelines. Circulation 139(25):e1082–e1143. Erratum in: Circulation. 2019 Jun 18;139(25):e1182–e1186. https://doi.org/10.1161/CIR.0000000000000625
Guyton JR, Slee AE, Anderson T, Fleg JL, Goldberg RB, Kashyap ML, Marcovina SM, Nash SD, O'Brien KD, Weintraub WS, Xu P, Zhao XQ, Boden WE (2013) Relationship of lipoproteins to cardiovascular events: the AIM-HIGH trial (atherothrombosis intervention in metabolic syndrome with low HDL/high triglycerides and impact on global health outcomes). J Am Coll Cardiol 62(17):1580–1584. https://doi.org/10.1016/j.jacc.2013.07.023
Haase CL, Tybjærg-Hansen A, Nordestgaard BG, Frikke-Schmidt R (2015) HDL cholesterol and risk of type 2 diabetes: a Mendelian randomization study. Diabetes 64:3328–3333
Hafiane A, Genest J (2015) High density lipoproteins: measurement techniques and potential biomarkers of cardiovascular risk. BBA Clin 3:175–188. https://doi.org/10.1016/j.bbacli.2015.01.005
He H, Hong K, Liu L, Schwendeman A (2021) Artificial high-density lipoprotein mimicking nanotherapeutics for the treatment of cardiovascular diseases. Wiley Interdiscip Rev Nanomed Nanobiotechnol 14:e1737. https://doi.org/10.1002/wnan.1737
Helgadottir A, Sulem P, Thorgeirsson G, Gretarsdottir S, Thorleifsson G, Jensson BÖ, Arnadottir GA, Olafsson I, Eyjolfsson GI, Sigurdardottir O, Thorsteinsdottir U, Gudbjartsson DF, Holm H, Stefansson K (2018) Rare SCARB1 mutations associate with high-density lipoprotein cholesterol but not with coronary artery disease. Eur Heart J 39(23):2172–2178. https://doi.org/10.1093/eurheartj/ehy169
Hennuyer N, Duplan I, Paquet C, Vanhoutte J, Woitrain E, Touche V, Colin S, Vallez E, Lestavel S, Lefebvre P, Staels B (2016) The novel selective PPARα modulator (SPPARMα) pemafibrate improves dyslipidemia, enhances reverse cholesterol transport and decreases inflammation and atherosclerosis. Atherosclerosis 249:200–208. https://doi.org/10.1016/j.atherosclerosis.2016.03.003
Hilser JR, Han Y, Biswas S, Gukasyan J, Cai Z, Zhu R, Tang WHW, Deb A, Lusis AJ, Hartiala JA, Allayee H (2021) Association of serum HDL-cholesterol and apolipoprotein A1 levels with risk of severe SARS-CoV-2 infection. J Lipid Res 62:100061. https://doi.org/10.1016/j.jlr.2021.100061
Hilvo M, Meikle PJ, Pedersen ER, Tell GS, Dhar I, Brenner H, Schöttker B, Lääperi M, Kauhanen D, Koistinen KM, Jylhä A, Huynh K, Mellett NA, Tonkin AM, Sullivan DR, Simes J, Nestel P, Koenig W, Rothenbacher D, Nygård O, Laaksonen R (2019) Development and validation of a ceramide- and phospholipid-based cardiovascular risk estimation score for coronary artery disease patients. Eur Heart J. https://doi.org/10.1093/eurheartj/ehz387
Hoekstra M, Sorci-Thomas M (2017) Rediscovering scavenger receptor type BI: surprising new roles for the HDL receptor. Curr Opin Lipidol 28(3):255–260. https://doi.org/10.1097/MOL.0000000000000413
Hoekstra M, Van Eck M (2015) Mouse models of disturbed HDL metabolism. Handb Exp Pharmacol 224:301–336. https://doi.org/10.1007/978-3-319-09665-0_9
Hopewell JC, Ibrahim M, Hill M, Shaw PM, Braunwald E, Blaustein RO, Bowman L, Landray MJ, Sabatine MS, Collins R, HPS3/TIMI55 - REVEAL Collaborative Group (2019) Impact of ADCY9 genotype on response to anacetrapib. Circulation 140(11):891–898. https://doi.org/10.1161/CIRCULATIONAHA.119.041546
HPS2-THRIVE Collaborative Group, Landray MJ, Haynes R, Hopewell JC, Parish S, Aung T, Tomson J, Wallendszus K, Craig M, Jiang L, Collins R, Armitage J (2014) Effects of extended-release niacin with laropiprant in high-risk patients. N Engl J Med 371(3):203–212. https://doi.org/10.1056/NEJMoa1300955
HPS3/TIMI55–REVEAL Collaborative Group, Bowman L, Hopewell JC, Chen F, Wallendszus K, Stevens W, Collins R, Wiviott SD, Cannon CP, Braunwald E, Sammons E, Landray MJ (2017) Effects of anacetrapib in patients with atherosclerotic vascular disease. N Engl J Med 377(13):1217–1227. https://doi.org/10.1056/NEJMoa1706444
Jensen MK, Aroner SA, Mukamal KJ, Furtado JD, Post WS, Tsai MY, Tjønneland A, Polak JF, Rimm EB, Overvad K, McClelland RL, Sacks FM (2018) High-density lipoprotein subspecies defined by presence of apolipoprotein C-III and incident coronary heart disease in four cohorts. Circulation 137(13):1364–1373. https://doi.org/10.1161/CIRCULATIONAHA.117.031276
Jeppesen J, Hein HO, Suadicani P, Gyntelberg F (1998) Triglyceride concentration and ischemic heart disease: an eight-year follow-up in the Copenhagen male study. Circulation 97(11):1029–1036. Erratum in: Circulation 1998 May 19;97(19):1995. https://doi.org/10.1161/01.cir.97.11.1029
Jin Z, Collier TS, Dai DLY, Chen V, Hollander Z, Ng RT, McManus BM, Balshaw R, Apostolidou S, Penn MS, Bystrom C (2019) Development and validation of apolipoprotein AI-associated lipoprotein proteome panel for the prediction of cholesterol efflux capacity and coronary artery disease. Clin Chem 65(2):282–290. https://doi.org/10.1373/clinchem.2018.291922
Johannesen CDL, Langsted A, Mortensen MB, Nordestgaard BG (2020) Association between low density lipoprotein and all cause and cause specific mortality in Denmark: prospective cohort study. BMJ 371:m4266. Erratum in: BMJ. 2021 Feb 12;372:n422. https://doi.org/10.1136/bmj.m4266
Kadiyala P, Li D, Nuñez FM, Altshuler D, Doherty R, Kuai R, Yu M, Kamran N, Edwards M, Moon JJ, Lowenstein PR, Castro MG, Schwendeman A (2019) High-density lipoprotein-mimicking nanodiscs for chemo-immunotherapy against glioblastoma multiforme. ACS Nano 13(2):1365–1384. https://doi.org/10.1021/acsnano.8b06842
Kamanna VS, Ganji SH, Kashyap ML (2013) Recent advances in niacin and lipid metabolism. Curr Opin Lipidol 24(3):239–245. https://doi.org/10.1097/MOL.0b013e3283613a68
Karjalainen MK, Holmes MV, Wang Q, Anufrieva O, Kähönen M, Lehtimäki T, Havulinna AS, Kristiansson K, Salomaa V, Perola M, Viikari JS, Raitakari OT, Järvelin MR, Ala-Korpela M, Kettunen J (2020) Apolipoprotein A-I concentrations and risk of coronary artery disease: a Mendelian randomization study. Atherosclerosis 299:56–63. https://doi.org/10.1016/j.atherosclerosis.2020.02.002
Kathiresan S (2012) Will cholesteryl ester transfer protein inhibition succeed primarily by lowering low-density lipoprotein cholesterol? Insights from human genetics and clinical trials. J Am Coll Cardiol 60(20):2049–2052. https://doi.org/10.1016/j.jacc.2012.08.967
Keech A, Simes RJ, Barter P, Best J, Scott R, Taskinen MR, Forder P, Pillai A, Davis T, Glasziou P, Drury P, Kesäniemi YA, Sullivan D, Hunt D, Colman P, d'Emden M, Whiting M, Ehnholm C, Laakso M, FIELD study investigators (2005) Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366(9500):1849–1861. Erratum in: Lancet. 2006 Oct 21;368(9545):1420. Erratum in: Lancet. 2006 Oct 21;368(9545):1415. https://doi.org/10.1016/S0140-6736(05)67667-2
Keene D, Price C, Shun-Shin MJ, Francis DP (2014) Effect on cardiovascular risk of high density lipoprotein targeted drug treatments niacin, fibrates, and CETP inhibitors: meta-analysis of randomised controlled trials including 117,411 patients. BMJ 349:g4379. https://doi.org/10.1136/bmj.g4379
Khan AA, Mundra PA, Straznicky NE, Nestel PJ, Wong G, Tan R, Huynh K, Ng TW, Mellett NA, Weir JM, Barlow CK, Alshehry ZH, Lambert GW, Kingwell BA, Meikle PJ (2018) Weight loss and exercise alter the high-density lipoprotein lipidome and improve high-density lipoprotein functionality in metabolic syndrome. Arterioscler Thromb Vasc Biol 38(2):438–447. https://doi.org/10.1161/ATVBAHA.117.310212
Kim DS, Li YK, Bell GA, Burt AA, Vaisar T, Hutchins PM, Furlong CE, Otvos JD, Polak JF, Arnan MK, Kaufman JD, McClelland RL, Longstreth WT Jr, Jarvik GP (2016) Concentration of smaller high-density lipoprotein particle (HDL-P) is inversely correlated with carotid intima media thickening after confounder adjustment: the multi ethnic study of atherosclerosis (MESA). J Am Heart Assoc 5(5):e002977. https://doi.org/10.1161/JAHA.115.002977
Kim J, Dey A, Malhotra A, Liu J, Ahn SI, Sei YJ, Kenney AM, MacDonald TJ, Kim Y (2020) Engineered biomimetic nanoparticle for dual targeting of the cancer stem-like cell population in sonic hedgehog medulloblastoma. Proc Natl Acad Sci U S A 117(39):24205–24212. https://doi.org/10.1073/pnas.1911229117
Kinslechner K, Schörghofer D, Schütz B, Vallianou M, Wingelhofer B, Mikulits W, Röhrl C, Hengstschläger M, Moriggl R, Stangl H, Mikula M (2018) Malignant phenotypes in metastatic melanoma are governed by SR-BI and its association with glycosylation and STAT5 activation. Mol Cancer Res 16(1):135–146. https://doi.org/10.1158/1541-7786.MCR-17-0292
Kjeldsen EW, Nordestgaard LT, Frikke-Schmidt R (2021a) HDL cholesterol and non-cardiovascular disease: a narrative review. Int J Mol Sci 22(9):4547. https://doi.org/10.3390/ijms22094547
Kjeldsen EW, Thomassen JQ, Juul Rasmussen I, Nordestgaard BG, Tybjærg-Hansen A, Frikke-Schmidt R (2021b) Plasma HDL cholesterol and risk of dementia – observational and genetic studies. Cardiovasc Res:cvab164. https://doi.org/10.1093/cvr/cvab164
Ko DT, Alter DA, Guo H, Koh M, Lau G, Austin PC, Booth GL, Hogg W, Jackevicius CA, Lee DS, Wijeysundera HC, Wilkins JT, Tu JV (2016) High-density lipoprotein cholesterol and cause-specific mortality in individuals without previous cardiovascular conditions: the CANHEART study. J Am Coll Cardiol 68(19):2073–2083. https://doi.org/10.1016/j.jacc.2016.08.038
Ko CW, Qu J, Black DD, Tso P (2020) Regulation of intestinal lipid metabolism: current concepts and relevance to disease. Nat Rev Gastroenterol Hepatol 17(3):169–183. https://doi.org/10.1038/s41575-019-0250-7
Kontush A, Lhomme M, Chapman MJ (2013) Unraveling the complexities of the HDL lipidome. J Lipid Res 54(11):2950–2963. https://doi.org/10.1194/jlr.R036095
Kopecky C, Genser B, Drechsler C, Krane V, Kaltenecker CC, Hengstschläger M, März W, Wanner C, Säemann MD, Weichhart T (2015) Quantification of HDL proteins, cardiac events, and mortality in patients with type 2 diabetes on hemodialysis. Clin J Am Soc Nephrol 10(2):224–231. https://doi.org/10.2215/CJN.06560714
Kühnast S, van der Tuin SJ, van der Hoorn JW, van Klinken JB, Simic B, Pieterman E, Havekes LM, Landmesser U, Lüscher TF, Willems van Dijk K, Rensen PC, Jukema JW, Princen HM (2015) Anacetrapib reduces progression of atherosclerosis, mainly by reducing non-HDL-cholesterol, improves lesion stability and adds to the beneficial effects of atorvastatin. Eur Heart J 36(1):39–48. https://doi.org/10.1093/eurheartj/ehu319
Kuller LH, Grandits G, Cohen JD, Neaton JD, Prineas R, Multiple Risk Factor Intervention Trial Research Group (2007) Lipoprotein particles, insulin, adiponectin, C-reactive protein and risk of coronary heart disease among men with metabolic syndrome. Atherosclerosis 195(1):122–128. https://doi.org/10.1016/j.atherosclerosis.2006.09.001
Kuusisto S, Holmes MV, Ohukainen P, Kangas AJ, Karsikas M, Tiainen M, Perola M, Salomaa V, Kettunen J, Ala-Korpela M (2019) Direct estimation of HDL-mediated cholesterol efflux capacity from serum. Clin Chem 65(8):1042–1050. https://doi.org/10.1373/clinchem.2018.299222
Langer C, Gansz B, Goepfert C, Engel T, Uehara Y, von Dehn G, Jansen H, Assmann G, von Eckardstein A (2002) Testosterone up-regulates scavenger receptor BI and stimulates cholesterol efflux from macrophages. Biochem Biophys Res Commun 296(5):1051–1057. https://doi.org/10.1016/s0006-291x(02)02038-7
Langsted A, Jensen AMR, Varbo A, Nordestgaard BG (2020) Low high-density lipoprotein cholesterol to monitor long-term average increased triglycerides. J Clin Endocrinol Metab 105(4):dgz265. https://doi.org/10.1210/clinem/dgz265
Lassale C, Hamer M, Hernáez Á, Gale CR, Batty GD (2021) Association of pre-pandemic high-density lipoprotein cholesterol with risk of COVID-19 hospitalisation and death: the UK Biobank cohort study. Prev Med Rep 23:101461. https://doi.org/10.1016/j.pmedr.2021.101461
Le Lay JE, Du Q, Mehta MB, Bhagroo N, Hummer BT, Falloon J, Carlson G, Rosenbaum AI, Jin C, Kimko H, Tsai LF, Novick S, Cook B, Han D, Han CY, Vaisar T, Chait A, Karathanasis SK, Rhodes CJ, Hirshberg B, Damschroder MM, Hsia J, Grimsby JS (2021) Blocking endothelial lipase with monoclonal antibody MEDI5884 durably increases high density lipoprotein in nonhuman primates and in a phase 1 trial. Sci Transl Med 13(590):eabb0602. https://doi.org/10.1126/scitranslmed.abb0602
Lee-Rueckert M, Escola-Gil JC, Kovanen PT (2016) HDL functionality in reverse cholesterol transport--challenges in translating data emerging from mouse models to human disease. Biochim Biophys Acta 1861(7):566–583. https://doi.org/10.1016/j.bbalip.2016.03.004
Lehti M, Donelan E, Abplanalp W, Al-Massadi O, Habegger KM, Weber J, Ress C, Mansfeld J, Somvanshi S, Trivedi C, Keuper M, Ograjsek T, Striese C, Cucuruz S, Pfluger PT, Krishna R, Gordon SM, Silva RA, Luquet S, Castel J, Martinez S, D'Alessio D, Davidson WS, Hofmann SM (2013) High-density lipoprotein maintains skeletal muscle function by modulating cellular respiration in mice. Circulation 128(22):2364–2371. https://doi.org/10.1161/CIRCULATIONAHA.113.001551
Li JJ, Zhang Y, Li S, Cui CJ, Zhu CG, Guo YL, Wu NQ, Xu RX, Liu G, Dong Q, Sun J (2016) Large HDL subfraction but not HDL-C is closely linked with risk factors, coronary severity and outcomes in a cohort of nontreated patients with stable coronary artery disease: a prospective observational study. Medicine (Baltimore) 95(4):e2600. https://doi.org/10.1097/MD.0000000000002600
Lincoff AM, Nicholls SJ, Riesmeyer JS, Barter PJ, Brewer HB, Fox KAA, Gibson CM, Granger C, Menon V, Montalescot G, Rader D, Tall AR, McErlean E, Wolski K, Ruotolo G, Vangerow B, Weerakkody G, Goodman SG, Conde D, McGuire DK, Nicolau JC, Leiva-Pons JL, Pesant Y, Li W, Kandath D, Kouz S, Tahirkheli N, Mason D, Nissen SE, ACCELERATE Investigators (2017) Evacetrapib and cardiovascular outcomes in high-risk vascular disease. N Engl J Med 376(20):1933–1942. https://doi.org/10.1056/NEJMoa1609581
Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, Chapman MJ, De Backer GG, Delgado V, Ference BA, Graham IM, Halliday A, Landmesser U, Mihaylova B, Pedersen TR, Riccardi G, Richter DJ, Sabatine MS, Taskinen MR, Tokgozoglu L, Wiklund O, ESC Scientific Document Group (2020) 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 41(1):111–188. Erratum in: Eur Heart J. 2020 Nov 21;41(44):4255. https://doi.org/10.1093/eurheartj/ehz455
Mackey RH, Greenland P, Goff DC Jr, Lloyd-Jones D, Sibley CT, Mora S (2012) High-density lipoprotein cholesterol and particle concentrations, carotid atherosclerosis, and coronary events: MESA (multi-ethnic study of atherosclerosis). J Am Coll Cardiol 60(6):508–516. https://doi.org/10.1016/j.jacc.2012.03.060
Madsen CM, Varbo A, Nordestgaard BG (2017) Extreme high high-density lipoprotein cholesterol is paradoxically associated with high mortality in men and women: two prospective cohort studies. Eur Heart J 38(32):2478–2486. https://doi.org/10.1093/eurheartj/ehx163
Madsen CM, Varbo A, Tybjærg-Hansen A, Frikke-Schmidt R, Nordestgaard BG (2018) U-shaped relationship of HDL and risk of infectious disease: two prospective population-based cohort studies. Eur Heart J 39(14):1181–1190. https://doi.org/10.1093/eurheartj/ehx665
Madsen CM, Varbo A, Nordestgaard BG (2019) Low HDL cholesterol and high risk of autoimmune disease: two population-based cohort studies including 117341 individuals. Clin Chem 65(5):644–652. https://doi.org/10.1373/clinchem.2018.299636
Madsen CM, Varbo A, Nordestgaard BG (2021) Novel insights from human studies on the role of high-density lipoprotein in mortality and noncardiovascular disease. Arterioscler Thromb Vasc Biol 41(1):128–140. https://doi.org/10.1161/ATVBAHA.120.314050
Manandhar B, Cochran BJ, Rye KA (2020) Role of high-density lipoproteins in cholesterol homeostasis and glycemic control. J Am Heart Assoc 9(1):e013531. https://doi.org/10.1161/JAHA.119.013531
Martin SS, Blaha MJ, Elshazly MB, Toth PP, Kwiterovich PO, Blumenthal RS, Jones SR (2013) Comparison of a novel method vs the Friedewald equation for estimating low-density lipoprotein cholesterol levels from the standard lipid profile. JAMA 310(19):2061–2068. https://doi.org/10.1001/jama.2013.280532
März W, Kleber ME, Scharnagl H, Speer T, Zewinger S, Ritsch A, Parhofer KG, von Eckardstein A, Landmesser U, Laufs U (2017) HDL cholesterol: reappraisal of its clinical relevance. Clin Res Cardiol 106(9):663–675. https://doi.org/10.1007/s00392-017-1106-1
Masson W, Lobo M, Siniawski D, Huerín M, Molinero G, Valéro R, Nogueira JP (2018) Therapy with cholesteryl ester transfer protein (CETP) inhibitors and diabetes risk. Diabetes Metab 44(6):508–513. https://doi.org/10.1016/j.diabet.2018.02.005
McGarrah RW, Craig DM, Haynes C, Dowdy ZE, Shah SH, Kraus WE (2016) High-density lipoprotein subclass measurements improve mortality risk prediction, discrimination and reclassification in a cardiac catheterization cohort. Atherosclerosis 246:229–235. https://doi.org/10.1016/j.atherosclerosis.2016.01.012
Meikle PJ, Formosa MF, Mellett NA, Jayawardana KS, Giles C, Bertovic DA, Jennings GL, Childs W, Reddy M, Carey AL, Baradi A, Nanayakkara S, Wilson AM, Duffy SJ, Kingwell BA (2019) HDL phospholipids, but not cholesterol distinguish acute coronary syndrome from stable coronary artery disease. J Am Heart Assoc 8(11):e011792. https://doi.org/10.1161/JAHA.118.011792
Meilhac O, Tanaka S, Couret D (2020) High-density lipoproteins are bug scavengers. Biomol Ther 10(4):598. https://doi.org/10.3390/biom10040598
Menon V, Kumar A, Patel DR, John JS, Riesmeyer J, Weerakkody G, Ruotolo G, Wolski KE, McErlean E, Cremer PC et al (2020) Effect of CETP inhibition with evacetrapib in patients with diabetes mellitus enrolled in the ACCELERATE trial. BMJ Open Diabetes Res Care 8:e000943
Meriwether D, Sulaiman D, Volpe C, Dorfman A, Grijalva V, Dorreh N, Solorzano-Vargas RS, Wang J, O'Connor E, Papesh J, Larauche M, Trost H, Palgunachari MN, Anantharamaiah GM, Herschman HR, Martin MG, Fogelman AM, Reddy ST (2019) Apolipoprotein A-I mimetics mitigate intestinal inflammation in COX2-dependent inflammatory bowel disease model. J Clin Invest 129(9):3670–3685. https://doi.org/10.1172/JCI123700
Metzinger MP, Saldanha S, Gulati J, Patel KV, El-Ghazali A, Deodhar S, Joshi PH, Ayers C, Rohatgi A (2020) Effect of anacetrapib on cholesterol efflux capacity: a substudy of the DEFINE trial. J Am Heart Assoc 9(24):e018136. https://doi.org/10.1161/JAHA.120.018136
Miller WG, Myers GL, Sakurabayashi I, Bachmann LM, Caudill SP, Dziekonski A, Edwards S, Kimberly MM, Korzun WJ, Leary ET, Nakajima K, Nakamura M, Nilsson G, Shamburek RD, Vetrovec GW, Warnick GR, Remaley AT (2010) Seven direct methods for measuring HDL and LDL cholesterol compared with ultracentrifugation reference measurement procedures. Clin Chem 56(6):977–986. https://doi.org/10.1373/clinchem.2009.142810
Montaigne D, Butruille L, Staels B (2021) PPAR control of metabolism and cardiovascular functions. Nat Rev Cardiol. https://doi.org/10.1038/s41569-021-00569-6
Mora S, Otvos JD, Rifai N, Rosenson RS, Buring JE, Ridker PM (2009) Lipoprotein particle profiles by nuclear magnetic resonance compared with standard lipids and apolipoproteins in predicting incident cardiovascular disease in women. Circulation 119(7):931–939. https://doi.org/10.1161/CIRCULATIONAHA.108.816181
Mora S, Glynn RJ, Ridker PM (2013) High-density lipoprotein cholesterol, size, particle number, and residual vascular risk after potent statin therapy. Circulation 128(11):1189–1197. https://doi.org/10.1161/CIRCULATIONAHA.113.002671
Morin EE, Guo L, Schwendeman A, Li XA (2015) HDL in sepsis - risk factor and therapeutic approach. Front Pharmacol 6:244. https://doi.org/10.3389/fphar.2015.00244
Morin EE, Li XA, Schwendeman A (2018) HDL in endocrine carcinomas: biomarker, drug carrier, and potential therapeutic. Front Endocrinol (Lausanne) 9:715. https://doi.org/10.3389/fendo.2018.00715
Moriyama Y, Okamura T, Inazu A, Doi M, Iso H, Mouri Y, Ishikawa Y, Suzuki H, Iida M, Koizumi J et al (1998) A low prevalence of coronary heart disease among subjects with increased high-density lipoprotein cholesterol levels, including those with plasma cholesteryl ester transfer protein deficiency. Prev Med 27:659–667
Mortensen MB, Afzal S, Nordestgaard BG, Falk E (2015) The high-density lipoprotein-adjusted SCORE model worsens SCORE-based risk classification in a contemporary population of 30,824 Europeans: the Copenhagen general population study. Eur Heart J 36(36):2446–2453. https://doi.org/10.1093/eurheartj/ehv251
Mundra PA, Shaw JE, Meikle PJ (2016) Lipidomic analyses in epidemiology. Int J Epidemiol 45(5):1329–1338
Natarajan P, Collier TS, Jin Z, Lyass A, Li Y, Ibrahim NE, Mukai R, McCarthy CP, Massaro JM, D'Agostino RB Sr, Gaggin HK, Bystrom C, Penn MS, Januzzi JL Jr (2019) Association of an HDL apolipoproteomic score with coronary atherosclerosis and cardiovascular death. J Am Coll Cardiol 73(17):2135–2145. https://doi.org/10.1016/j.jacc.2019.01.073
Neale BM, Fagerness J, Reynolds R, Sobrin L, Parker M, Raychaudhuri S, Tan PL, Oh EC, Merriam JE, Souied E, Bernstein PS, Li B, Frederick JM, Zhang K, Brantley MA Jr, Lee AY, Zack DJ, Campochiaro B, Campochiaro P, Ripke S, Smith RT, Barile GR, Katsanis N, Allikmets R, Daly MJ, Seddon JM (2010) Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC). Proc Natl Acad Sci U S A 107(16):7395–7400. https://doi.org/10.1073/pnas.0912019107
Nicholls SJ, Ruotolo G, Brewer HB, Kane JP, Wang MD, Krueger KA, Adelman SJ, Nissen SE, Rader DJ (2015) Cholesterol efflux capacity and pre-beta-1 HDL concentrations are increased in dyslipidemic patients treated with evacetrapib. J Am Coll Cardiol 66(20):2201–2210. https://doi.org/10.1016/j.jacc.2015.09.013
Nicholls SJ, Puri R, Ballantyne CM, Jukema JW, Kastelein JJP, Koenig W, Wright RS, Kallend D, Wijngaard P, Borgman M, Wolski K, Nissen SE (2018a) Effect of infusion of high-density lipoprotein mimetic containing recombinant apolipoprotein A-I milano on coronary disease in patients with an acute coronary syndrome in the MILANO-PILOT trial: a randomized clinical trial. JAMA Cardiol 3:806–814
Nicholls SJ, Andrews J, Kastelein JJP, Merkely B, Nissen SE, Ray KK, Schwartz GG, Worthley SG, Keyserling C, Dasseux JL, Griffith L, Kim SW, Janssan A, Di Giovanni G, Pisaniello AD, Scherer DJ, Psaltis PJ, Butters J (2018b) Effect of serial infusions of CER-001, a pre-β high-density lipoprotein mimetic, on coronary atherosclerosis in patients following acute coronary syndromes in the CER-001 atherosclerosis regression acute coronary syndrome trial: a randomized clinical trial. JAMA Cardiol 3:815–822
Nissen SE, Tsunoda T, Tuzcu EM, Schoenhagen P, Cooper CJ, Yasin M, Eaton GM, Lauer MA, Sheldon WS, Grines CL, Halpern S, Crowe T, Blankenship JC, Kerensky R (2003) Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes: a randomized controlled trial. JAMA 290:2292–2300
Nissen SE, Pillai SG, Nicholls SJ, Wolski K, Riesmeyer JS, Weerakkody GJ, Foster WM, McErlean E, Li L, Bhatnagar P, Ruotolo G, Lincoff AM (2018) ADCY9 genetic variants and cardiovascular outcomes with evacetrapib in patients with high-risk vascular disease: a nested case-control study. JAMA Cardiol 3(5):401–408. https://doi.org/10.1001/jamacardio.2018.0569
Niu W, Qi Y (2015) Circulating cholesteryl ester transfer protein and coronary heart disease: mendelian randomization meta-analysis. Circ Cardiovasc Genet 8(1):114–121. https://doi.org/10.1161/CIRCGENETICS.114.000748
Nordestgaard LT, Tybjærg-Hansen A, Nordestgaard BG, Frikke-Schmidt R (2015) Loss-of-function mutation in ABCA1 and risk of Alzheimer’s disease and cerebrovascular disease. Alzheimers Dement 11(12):1430–1438. https://doi.org/10.1016/j.jalz.2015.04.006
Nordestgaard BG, Langlois MR, Langsted A, Chapman MJ, Aakre KM, Baum H, Borén J, Bruckert E, Catapano A, Cobbaert C, Collinson P, Descamps OS, Duff CJ, von Eckardstein A, Hammerer-Lercher A, Kamstrup PR, Kolovou G, Kronenberg F, Mora S, Pulkki K, Remaley AT, Rifai N, Ros E, Stankovic S, Stavljenic-Rukavina A, Sypniewska G, Watts GF, Wiklund O, Laitinen P, European Atherosclerosis Society (EAS) and the European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) Joint Consensus Initiative (2020) Quantifying atherogenic lipoproteins for lipid-lowering strategies: consensus-based recommendations from EAS and EFLM. Atherosclerosis 294:46–61. https://doi.org/10.1016/j.atherosclerosis.2019.12.005
Nordestgaard LT, Tybjærg-Hansen A, Frikke-Schmidt R, Nordestgaard BG (2021) Elevated apolipoprotein A1 and HDL cholesterol associated with age-related macular degeneration: 2 population cohorts. J Clin Endocrinol Metab 106(7):e2749–e2758. https://doi.org/10.1210/clinem/dgab095
Nowacki TM, Remaley AT, Bettenworth D, Eisenblätter M, Vowinkel T, Becker F, Vogl T, Roth J, Tietge UJ, Lügering A, Heidemann J, Nofer JR (2016) The 5A apolipoprotein A-I (apoA-I) mimetic peptide ameliorates experimental colitis by regulating monocyte infiltration. Br J Pharmacol 173(18):2780–2792. https://doi.org/10.1111/bph.13556
Offermanns S (2014) Free fatty acid (FFA) and hydroxy carboxylic acid (HCA) receptors. Annu Rev Pharmacol Toxicol 54:407–434. https://doi.org/10.1146/annurev-pharmtox-011613-135945
Orsoni A, Thérond P, Tan R, Giral P, Robillard P, Kontush A, Meikle PJ, Chapman MJ (2016) Statin action enriches HDL3 in polyunsaturated phospholipids and plasmalogens and reduces LDL-derived phospholipid hydroperoxides in atherogenic mixed dyslipidemia. J Lipid Res 57(11):2073–2087
Ostergaard SD, Mukherjee S, Sharp SJ, Proitsi P, Lotta LA, Day FR, Perry JR, Boehme KL, Walter S, Kauwe JS et al (2015) Associations between potentially modifiable risk factors and Alzheimer disease: a Mendelian randomization study. PLoS Med 12:e1001841
Otvos JD, Collins D, Freedman DS, Shalaurova I, Schaefer EJ, McNamara JR, Bloomfield HE, Robins SJ (2006) Low-density lipoprotein and high-density lipoprotein particle subclasses predict coronary events and are favorably changed by gemfibrozil therapy in the veterans affairs high-density lipoprotein intervention trial. Circulation 113(12):1556–1563. https://doi.org/10.1161/CIRCULATIONAHA.105.565135
Pajkrt D, Doran JE, Koster F, Lerch PG, Arnet B, van der Poll T, ten Cate JW, van Deventer SJ (1996) Antiinflammatory effects of reconstituted high-density lipoprotein during human endotoxemia. J Exp Med 184(5):1601–1608. https://doi.org/10.1084/jem.184.5.1601
Pajkrt D, Lerch PG, van der Poll T, Levi M, Illi M, Doran JE, Arnet B, van den Ende A, ten Cate JW, van Deventer SJ (1997) Differential effects of reconstituted high-density lipoprotein on coagulation, fibrinolysis and platelet activation during human endotoxemia. Thromb Haemost 77(2):303–307
Parhofer KG (2015) Interaction between glucose and lipid metabolism: more than diabetic dyslipidemia. Diabetes Metab J 39(5):353–362
Parish S, Offer A, Clarke R, Hopewell JC, Hill MR, Otvos JD, Armitage J, Collins R, Heart Protection Study Collaborative Group (2012) Lipids and lipoproteins and risk of different vascular events in the MRC/BHF heart protection study. Circulation 125(20):2469–2478. https://doi.org/10.1161/CIRCULATIONAHA.111.073684
Pavanello C, Calabresi L (2020) Genetic, biochemical, and clinical features of LCAT deficiency: update for 2020. Curr Opin Lipidol 31(4):232–237. https://doi.org/10.1097/MOL.0000000000000697
Pirillo A, Catapano AL, Norata GD (2015) HDL in infectious diseases and sepsis. Handb Exp Pharmacol 224:483–508. https://doi.org/10.1007/978-3-319-09665-0_15
Pirro M, Ricciuti B, Rader DJ, Catapano AL, Sahebkar A, Banach M (2018) High density lipoprotein cholesterol and cancer: marker or causative? Prog Lipid Res 71:54–69. https://doi.org/10.1016/j.plipres.2018.06.001
Pollin TI, Damcott CM, Shen H, Ott SH, Shelton J, Horenstein RB, Post W, McLenithan JC, Bielak LF, Peyser PA et al (2008) A null mutation in human APOC3 confers a favorable plasma lipid profile and apparent cardioprotection. Science 322:1702–1705
Pradhan AD, Paynter NP, Everett BM, Glynn RJ, Amarenco P, Elam M, Ginsberg H, Hiatt WR, Ishibashi S, Koenig W, Nordestgaard BG, Fruchart JC, Libby P, Ridker PM (2018) Rationale and design of the pemafibrate to reduce cardiovascular outcomes by reducing triglycerides in patients with diabetes (PROMINENT) study. Am Heart J 206:80–93. https://doi.org/10.1016/j.ahj.2018.09.011
Proitsi P, Lupton MK, Velayudhan L, Newhouse S, Fogh I, Tsolaki M, Daniilidou M, Pritchard M, Kloszewska I, Soininen H et al (2014) Genetic predisposition to increased blood cholesterol and triglyceride lipid levels and risk of Alzheimer disease: a Mendelian randomization analysis. PLoS Med 11:e1001713
Qi Y, Fan J, Liu J, Wang W, Wang M, Sun J, Liu J, Xie W, Zhao F, Li Y, Zhao D (2015) Cholesterol-overloaded HDL particles are independently associated with progression of carotid atherosclerosis in a cardiovascular disease-free population: a community-based cohort study. J Am Coll Cardiol 65(4):355–363. https://doi.org/10.1016/j.jacc.2014.11.019
Rajora MA, Zheng G (2016) Targeting SR-BI for cancer diagnostics, imaging and therapy. Front Pharmacol 7:326. https://doi.org/10.3389/fphar.2016.00326
Rasmussen KL, Tybjærg-Hansen A, Nordestgaard BG, Frikke-Schmidt R (2018) Plasma apolipoprotein E levels and risk of dementia: a Mendelian randomization study of 106,562 individuals. Alzheimers Dement 14(1):71–80. https://doi.org/10.1016/j.jalz.2017.05.006
Rautureau Y, Deschambault V, Higgins MÈ, Rivas D, Mecteau M, Geoffroy P, Miquel G, Uy K, Sanchez R, Lavoie V, Brand G, Nault A, Williams PM, Suarez ML, Merlet N, Lapointe L, Duquette N, Gillis MA, Samami S, Mayer G, Pouliot P, Raignault A, Maafi F, Brodeur MR, Levesque S, Guertin MC, Dubé MP, Thorin É, Rhainds D, Rhéaume É, Tardif JC (2018) ADCY9 (adenylate cyclase type 9) inactivation protects from atherosclerosis only in the absence of CETP (cholesteryl Ester transfer protein). Circulation 138(16):1677–1692. https://doi.org/10.1161/CIRCULATIONAHA.117.031134
Ray KK, Nicholls SJ, Buhr KA, Ginsberg HN, Johansson JO, Kalantar-Zadeh K, Kulikowski E, Toth PP, Wong N, Sweeney M, Schwartz GG, BETonMACE Investigators and Committees (2020) Effect of apabetalone added to standard therapy on major adverse cardiovascular events in patients with recent acute coronary syndrome and type 2 diabetes: a randomized clinical trial. JAMA 323(16):1565–1573. https://doi.org/10.1001/jama.2020.3308
Riaz H, Khan SU, Rahman H, Shah NP, Kaluski E, Lincoff AM, Nissen SE (2019) Effects of high-density lipoprotein targeting treatments on cardiovascular outcomes: a systematic review and meta-analysis. Eur J Prev Cardiol 26(5):533–543. https://doi.org/10.1177/2047487318816495
Richardson TG, Sanderson E, Palmer TM, Ala-Korpela M, Ference BA, Davey Smith G, Holmes MV (2020) Evaluating the relationship between circulating lipoprotein lipids and apolipoproteins with risk of coronary heart disease: a multivariable Mendelian randomisation analysis. PLoS Med 17(3):e1003062. https://doi.org/10.1371/journal.pmed.1003062
Ridker PM, Genest J, Boekholdt SM, Libby P, Gotto AM, Nordestgaard BG, Mora S, MacFadyen JG, Glynn RJ, Kastelein JJ, JUPITER Trial Study Group (2010) HDL cholesterol and residual risk of first cardiovascular events after treatment with potent statin therapy: an analysis from the JUPITER trial. Lancet 376(9738):333–339. https://doi.org/10.1016/S0140-6736(10)60713-1
Riwanto M, Rohrer L, Roschitzki B, Besler C, Mocharla P, Mueller M, Perisa D, Heinrich K, Altwegg L, von Eckardstein A, Lüscher TF, Landmesser U (2013) Altered activation of endothelial anti- and proapoptotic pathways by high-density lipoprotein from patients with coronary artery disease: role of high-density lipoprotein-proteome remodeling. Circulation 127(8):891–904. https://doi.org/10.1161/CIRCULATIONAHA.112.108753
Robert J, Osto E, von Eckardstein A (2021) The endothelium is both a target and a barrier of HDL’s protective functions. Cell 10(5):1041. https://doi.org/10.3390/cells10051041
Rohatgi A, Westerterp M, von Eckardstein A, Remaley A, Rye KA (2021) HDL in the 21st century: a multifunctional roadmap for future HDL research. Circulation 143(23):2293–2309. https://doi.org/10.1161/CIRCULATIONAHA.120.044221
Rosenson RS, Brewer HB Jr, Chapman MJ, Fazio S, Hussain MM, Kontush A, Krauss RM, Otvos JD, Remaley AT, Schaefer EJ (2011) HDL measures, particle heterogeneity, proposed nomenclature, and relation to atherosclerotic cardiovascular events. Clin Chem 57(3):392–410. https://doi.org/10.1373/clinchem.2010.155333
Rosenson RS, Davidson MH, Le NA, Burkle J, Pourfarzib R (2015) Underappreciated opportunities for high-density lipoprotein particles in risk stratification and potential targets of therapy. Cardiovasc Drugs Ther 29(1):41–50. https://doi.org/10.1007/s10557-014-6567-0
Rosenson RS, Burgess LJ, Ebenbichler CF, Baum SJ, Stroes ESG, Ali S, Khilla N, Hamlin R, Pordy R, Dong Y, Son V, Gaudet D (2020) Evinacumab in patients with refractory hypercholesterolemia. N Engl J Med 383(24):2307–2319. https://doi.org/10.1056/NEJMoa2031049
Rubins HB, Robins SJ, Collins D, Fye CL, Anderson JW, Elam MB, Faas FH, Linares E, Schaefer EJ, Schectman G, Wilt TJ, Wittes J (1999) Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med 341(6):410–418. https://doi.org/10.1056/NEJM199908053410604
Sacks FM, Tonkin AM, Shepherd J, Braunwald E, Cobbe S, Hawkins CM, Keech A, Packard C, Simes J, Byington R, Furberg CD (2000) Effect of pravastatin on coronary disease events in subgroups defined by coronary risk factors: the prospective pravastatin pooling project. Circulation 102(16):1893–1900. https://doi.org/10.1161/01.cir.102.16.1893
Sacks FM, Carey VJ, Fruchart JC (2010) Combination lipid therapy in type 2 diabetes. N Engl J Med 363(7):692–694; author reply 694-5. https://doi.org/10.1056/NEJMc1006407
Sacks FM, Liang L, Furtado JD, Cai T, Davidson WS, He Z, McClelland RL, Rimm EB, Jensen MK (2020) Protein-defined subspecies of HDLs (high-density lipoproteins) and differential risk of coronary heart disease in 4 prospective studies. Arterioscler Thromb Vasc Biol 40(11):2714–2727. https://doi.org/10.1161/ATVBAHA.120.314609
Saito T, Matsunaga A, Fukunaga M, Nagahama K, Hara S, Muso E (2020) Apolipoprotein E-related glomerular disorders. Kidney Int 97(2):279–288. https://doi.org/10.1016/j.kint.2019.10.031
Samanovic M, Molina-Portela MP, Chessler AD, Burleigh BA, Raper J (2009) Trypanosome lytic factor, an antimicrobial high-density lipoprotein, ameliorates Leishmania infection. PLoS Pathog 5(1):e1000276. https://doi.org/10.1371/journal.ppat.1000276
Sampson M, Ling C, Sun Q, Harb R, Ashmaig M, Warnick R, Sethi A, Fleming JK, Otvos JD, Meeusen JW, Delaney SR, Jaffe AS, Shamburek R, Amar M, Remaley AT (2020) A new equation for calculation of low-density lipoprotein cholesterol in patients with normolipidemia and/or hypertriglyceridemia. JAMA Cardiol 5(5):540–548. Erratum in: JAMA Cardiol.2020 May 1;5(5):613. https://doi.org/10.1001/jamacardio.2020.0013
Schmidt MI, Duncan BB, Bang H, Pankow JS, Ballantyne CM, Golden SH, Folsom AR, Chambless LE (2005) Identifying individuals at high risk for diabetes: the atherosclerosis risk in communities study. Diabetes Care 28:2013–2018
Schwartz GG, Olsson AG, Abt M, Ballantyne CM, Barter PJ, Brumm J, Chaitman BR, Holme IM, Kallend D, Leiter LA, Leitersdorf E, McMurray JJ, Mundl H, Nicholls SJ, Shah PK, Tardif JC, Wright RS, dal-OUTCOMES Investigators (2012) Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med 367(22):2089–2099. https://doi.org/10.1056/NEJMoa1206797
Schwartz GG, Leiter LA, Ballantyne CM, Barter PJ, Black DM, Kallend D, Laghrissi-Thode F, Leitersdorf E, McMurray JJ, Nicholls SJ et al (2020) Dalcetrapib reduces risk of new-onset diabetes in patients with coronary heart disease. Diabetes Care 43:1077–1084
Silbernagel G, Pagel P, Pfahlert V, Genser B, Scharnagl H, Kleber ME, Delgado G, Ohrui H, Ritsch A, Grammer TB, Koenig W, März W (2017) High-density lipoprotein subclasses, coronary artery disease, and cardiovascular mortality. Clin Chem 63(12):1886–1896. https://doi.org/10.1373/clinchem.2017.275636
Simic B, Mocharla P, Crucet M, Osto E, Kratzer A, Stivala S, Kühnast S, Speer T, Doycheva P, Princen HM, van der Hoorn JW, Jukema JW, Giral H, Tailleux A, Landmesser U, Staels B, Lüscher TF (2017) Anacetrapib, but not evacetrapib, impairs endothelial function in CETP-transgenic mice in spite of marked HDL-C increase. Atherosclerosis 257:186–194. https://doi.org/10.1016/j.atherosclerosis.2017.01.011
Singh K, Chandra A, Sperry T, Joshi PH, Khera A, Virani SS, Ballantyne CM, Otvos JD, Dullaart RPF, Gruppen EG, Connelly MA, Ayers CR, Rohatgi A (2020) Associations between high-density lipoprotein particles and ischemic events by vascular domain, sex, and ethnicity: a pooled cohort analysis. Circulation 142(7):657–669. https://doi.org/10.1161/CIRCULATIONAHA.120.045713
Soria-Florido MT, Schröder H, Grau M, Fitó M, Lassale C (2020) High density lipoprotein functionality and cardiovascular events and mortality: a systematic review and meta-analysis. Atherosclerosis 302:36–42. https://doi.org/10.1016/j.atherosclerosis.2020.04.015
Speer T, Ridker PM, von Eckardstein A, Schunk SJ, Fliser D (2021) Lipoproteins in chronic kidney disease: from bench to bedside. Eur Heart J 42(22):2170–2185. https://doi.org/10.1093/eurheartj/ehaa1050
Storti F, Klee K, Todorova V, Steiner R, Othman A, van der Velde-Visser S, Samardzija M, Meneau I, Barben M, Karademir D, Pauzuolyte V, Boye SL, Blaser F, Ullmer C, Dunaief JL, Hornemann T, Rohrer L, den Hollander A, von Eckardstein A, Fingerle J, Maugeais C, Grimm C (2019) Impaired ABCA1/ABCG1-mediated lipid efflux in the mouse retinal pigment epithelium (RPE) leads to retinal degeneration. elife 8:e45100. https://doi.org/10.7554/eLife.45100
Strazzella A, Ossoli A, Calabresi L (2021) High-density lipoproteins and the kidney. Cell 10(4):764. https://doi.org/10.3390/cells10040764
Sutter I, Velagapudi S, Othman A, Riwanto M, Manz J, Rohrer L, Rentsch K, Hornemann T, Landmesser U, von Eckardstein A (2015) Plasmalogens of high-density lipoproteins (HDL) are associated with coronary artery disease and anti-apoptotic activity of HDL. Atherosclerosis 241(2):539–546. https://doi.org/10.1016/j.atherosclerosis.2015.05.037
Talasaz AH, Sadeghipour P, Aghakouchakzadeh M, Dreyfus I, Kakavand H, Ariannejad H, Gupta A, Madhavan MV, Van Tassell BW, Jimenez D, Monreal M, Vaduganathan M, Fanikos J, Dixon DL, Piazza G, Parikh SA, Bhatt DL, Lip GY, Stone GW, Krumholz HM, Libby P, Goldhaber SZ, Bikdeli B (2021) Lipid-modulating agents for prevention or treatment of COVID-19 in randomized trials. medRxiv. https://doi.org/10.1101/2021.05.03.21256468
Tanaka S, Genève C, Zappella N, Yong-Sang J, Planesse C, Louedec L, Viranaïcken W, Bringart M, Montravers P, Denamur E, Duranteau J, Couret D, Meilhac O (2020) Reconstituted high-density lipoprotein therapy improves survival in mouse models of sepsis. Anesthesiology 132(4):825–838. https://doi.org/10.1097/ALN.0000000000003155
Tardif JC, Grégoire J, L'Allier PL, Ibrahim R, Lespérance J, Heinonen TM, Kouz S, Berry C, Basser R, Lavoie MA, Guertin MC, Rodés-Cabau J, Effect of rHDL on Atherosclerosis-Safety and Efficacy (ERASE) Investigators (2007) Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: a randomized controlled trial. JAMA 297:1675–1682
Tardif JC, Rhéaume E, Lemieux Perreault LP, Grégoire JC, Feroz Zada Y, Asselin G, Provost S, Barhdadi A, Rhainds D, L'Allier PL, Ibrahim R, Upmanyu R, Niesor EJ, Benghozi R, Suchankova G, Laghrissi-Thode F, Guertin MC, Olsson AG, Mongrain I, Schwartz GG, Dubé MP (2015) Pharmacogenomic determinants of the cardiovascular effects of dalcetrapib. Circ Cardiovasc Genet 8(2):372–382. https://doi.org/10.1161/CIRCGENETICS.114.000663
Tardif JC, Dubé MP, Pfeffer MA, Waters DD, Koenig W, Maggioni AP, McMurray JJV, Mooser V, White HD, Heinonen T, Black DM, Guertin MC, dal-GenE Investigators (2020) Study design of dal-GenE, a pharmacogenetic trial targeting reduction of cardiovascular events with dalcetrapib. Am Heart J 222:157–165. https://doi.org/10.1016/j.ahj.2020.01.007
Thirumalai A, Rubinow KB, Page ST (2015) An update on testosterone, HDL and cardiovascular risk in men. Clin Lipidol 10(3):251–258. https://doi.org/10.2217/clp.15.10
Tietjen I, Hovingh GK, Singaraja R et al (2012) Increased risk of coronary artery disease in Caucasians with extremely low HDL cholesterol due to mutations in ABCA1, APOA1, and LCAT. Biochim Biophys Acta 1821:416–424
Trakaki A, Marsche G (2021) Current understanding of the immunomodulatory activities of high-density lipoproteins. Biomedicine 9(6):587. https://doi.org/10.3390/biomedicines9060587
Trinder M, Genga KR, Kong HJ, Blauw LL, Lo C, Li X, Cirstea M, Wang Y, Rensen PCN, Russell JA, Walley KR, Boyd JH, Brunham LR (2019) Cholesteryl ester transfer protein influences high-density lipoprotein levels and survival in sepsis. Am J Respir Crit Care Med 199(7):854–862. https://doi.org/10.1164/rccm.201806-1157OC
Trinder M, Walley KR, Boyd JH, Brunham LR (2020) Causal inference for genetically determined levels of high-density lipoprotein cholesterol and risk of infectious disease. Arterioscler Thromb Vasc Biol 40(1):267–278. https://doi.org/10.1161/ATVBAHA.119.313381
Trinder M, Wang Y, Madsen CM, Ponomarev T, Bohunek L, Daisely BA, Julia Kong H, Blauw LL, Nordestgaard BG, Tybjærg-Hansen A, Wurfel MM, Russell JA, Walley KR, Rensen PCN, Boyd JH, Brunham LR (2021) Inhibition of cholesteryl ester transfer protein preserves high-density lipoprotein cholesterol and improves survival in sepsis. Circulation 143(9):921–934. https://doi.org/10.1161/CIRCULATIONAHA.120.048568
Vaisman BL, Neufeld EB, Freeman LA, Gordon SM, Sampson ML, Pryor M, Hillman E, Axley MJ, Karathanasis SK, Remaley AT (2019) LCAT enzyme replacement therapy reduces LpX and improves kidney function in a mouse model of familial LCAT deficiency. J Pharmacol Exp Ther 368(3):423–434. https://doi.org/10.1124/jpet.118.251876
Velagapudi S, Schraml P, Yalcinkaya M, Bolck HA, Rohrer L, Moch H, von Eckardstein A (2018) Scavenger receptor BI promotes cytoplasmic accumulation of lipoproteins in clear-cell renal cell carcinoma. J Lipid Res 59(11):2188–2201. https://doi.org/10.1194/jlr.M083311
Voight BF, Peloso GM, Orho-Melander M, Frikke-Schmidt R, Barbalic M, Jensen MK, Hindy G, Hólm H, Ding EL, Johnson T, Schunkert H, Samani NJ, Clarke R, Hopewell JC, Thompson JF, Li M, Thorleifsson G, Newton-Cheh C, Musunuru K, Pirruccello JP, Saleheen D, Chen L, Stewart A, Schillert A, Thorsteinsdottir U, Thorgeirsson G, Anand S, Engert JC, Morgan T, Spertus J, Stoll M, Berger K, Martinelli N, Girelli D, McKeown PP, Patterson CC, Epstein SE, Devaney J, Burnett MS, Mooser V, Ripatti S, Surakka I, Nieminen MS, Sinisalo J, Lokki ML, Perola M, Havulinna A, de Faire U, Gigante B, Ingelsson E, Zeller T, Wild P, de Bakker PI, Klungel OH, Maitland-van der Zee AH, Peters BJ, de Boer A, Grobbee DE, Kamphuisen PW, Deneer VH, Elbers CC, Onland-Moret NC, Hofker MH, Wijmenga C, Verschuren WM, Boer JM, van der Schouw YT, Rasheed A, Frossard P, Demissie S, Willer C, Do R, Ordovas JM, Abecasis GR, Boehnke M, Mohlke KL, Daly MJ, Guiducci C, Burtt NP, Surti A, Gonzalez E, Purcell S, Gabriel S, Marrugat J, Peden J, Erdmann J, Diemert P, Willenborg C, König IR, Fischer M, Hengstenberg C, Ziegler A, Buysschaert I, Lambrechts D, Van de Werf F, Fox KA, El Mokhtari NE, Rubin D, Schrezenmeir J, Schreiber S, Schäfer A, Danesh J, Blankenberg S, Roberts R, McPherson R, Watkins H, Hall AS, Overvad K, Rimm E, Boerwinkle E, Tybjaerg-Hansen A, Cupples LA, Reilly MP, Melander O, Mannucci PM, Ardissino D, Siscovick D, Elosua R, Stefansson K, O'Donnell CJ, Salomaa V, Rader DJ, Peltonen L, Schwartz SM, Altshuler D, Kathiresan S (2012) Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet 380(9841):572–580. Erratum in: Lancet. 2012 Aug 11;380(9841):564. https://doi.org/10.1016/S0140-6736(12)60312-2
Vollenweider P, Von Eckardstein A, Widmann C (2015) HDLs, diabetes and metabolic syndrome. In: High density lipoproteins: handbook of experimental pharmacology. Springer, Cham, pp 405–421
von Eckardstein A (2020) LDL contributes to reverse cholesterol transport. Circ Res 127(6):793–795. https://doi.org/10.1161/CIRCRESAHA.120.317721
Von Eckardstein A, Kardassis DM (2015) High density lipoproteins: from biological understanding to clinical exploitation. Handbook of experimental pharmacology. Springer, Cham
Von Eckardstein A, Rohrer L (2016) HDLs in crises. Curr Opin Lipidol 27:264–273
von Eckardstein A, Widmann C (2014) High-density lipoprotein, beta cells, and diabetes. Cardiovasc Res 103(3):384–394
von Eckardstein A, Schulte H, Assmann G (1999) Increased risk of myocardial infarction in men with both hypertriglyceridemia and elevated HDL cholesterol. Circulation 99(14):1925
von Eckardstein A, Schulte H, Assmann G (2000) Risk for diabetes mellitus in middle-aged Caucasian male participants of the PROCAM study: implications for the definition of impaired fasting glucose by the American Diabetes Association. Prospective Cardiovascular Münster. J Clin Endocrinol Metab 85(9):3101–3108. https://doi.org/10.1210/jcem.85.9.6773
Wei C, Wan L, Yan Q, Wang X, Zhang J, Yang X, Zhang Y, Fan C, Li D, Deng Y, Sun J, Gong J, Yang X, Wang Y, Wang X, Li J, Yang H, Li H, Zhang Z, Wang R, Du P, Zong Y, Yin F, Zhang W, Wang N, Peng Y, Lin H, Feng J, Qin C, Chen W, Gao Q, Zhang R, Cao Y, Zhong H (2020) HDL-scavenger receptor B type 1 facilitates SARS-CoV-2 entry. Nat Metab 2(12):1391–1400. https://doi.org/10.1038/s42255-020-00324-0
West of Scotland Coronary Prevention Study Group (1998) Influence of pravastatin and plasma lipids on clinical events in the West of Scotland Coronary Prevention Study (WOSCOPS). Circulation 97(15):1440–1445. https://doi.org/10.1161/01.cir.97.15.1440
White J, Swerdlow DI, Preiss D, Fairhurst-Hunter Z, Keating BJ, Asselbergs FW, Sattar N, Humphries SE, Hingorani AD, Holmes MV (2016) Association of lipid fractions with risks for coronary artery disease and diabetes. JAMA Cardiol 1:692–699
Wijeysundera HC, Koh M, Alter DA, Austin PC, Jackevicius CA, Tu JV, Ko DT (2017) Association of high-density lipoprotein cholesterol with non-fatal cardiac and non-cardiac events: a CANHEART substudy. Open Heart 4(2):e000731. https://doi.org/10.1136/openhrt-2017-000731
Wilson PWF, Meigs JB, Sullivan L, Fox CS, Nathan DM, D’Agostino RB (2007) Prediction of incident diabetes mellitus in middle-aged adults: the Framingham offspring study. Arch Intern Med 167:1068–1074
Wittert G, Bracken K, Robledo KP, Grossmann M, Yeap BB, Handelsman DJ, Stuckey B, Conway A, Inder W, McLachlan R, Allan C, Jesudason D, Fui MNT, Hague W, Jenkins A, Daniel M, Gebski V, Keech A (2021) Testosterone treatment to prevent or revert type 2 diabetes in men enrolled in a lifestyle programme (T4DM): a randomised, double-blind, placebo-controlled, 2-year, phase 3b trial. Lancet Diabetes Endocrinol 9(1):32–45. https://doi.org/10.1016/S2213-8587(20)30367-3
Witztum JL, Gaudet D, Freedman SD, Alexander VJ, Digenio A, Williams KR, Yang Q, Hughes SG, Geary RS, Arca M, Stroes ESG, Bergeron J, Soran H, Civeira F, Hemphill L, Tsimikas S, Blom DJ, O'Dea L, Bruckert E (2019) Volanesorsen and triglyceride levels in familial chylomicronemia syndrome. N Engl J Med 381(6):531–542. https://doi.org/10.1056/NEJMoa1715944
Wolska A, Reimund M, Sviridov DO, Amar MJ, Remaley AT (2021) Apolipoprotein mimetic peptides: potential new therapies for cardiovascular diseases. Cell 10(3):597. https://doi.org/10.3390/cells10030597
Wu FC, von Eckardstein A (2003) Androgens and coronary artery disease. Endocr Rev 24(2):183–217. https://doi.org/10.1210/er.2001-0025
Wu Y, Fan Z, Tian Y, Liu S, Liu S (2018) Relation between high density lipoprotein particles concentration and cardiovascular events: a meta-analysis. Lipids Health Dis 17(1):142. https://doi.org/10.1186/s12944-018-0732-6
Wu L, Soundarapandian MM, Castoreno AB, Millar JS, Rader DJ (2020) LDL-cholesterol reduction by ANGPTL3 inhibition in mice is dependent on endothelial lipase. Circ Res 127(8):1112–1114. https://doi.org/10.1161/CIRCRESAHA.120.317128
Xin W, Lin Z, Zhang T, Jia S (2019) Probucol for the prevention of contrast-induced acute kidney injury in patients undergoing coronary angiography or percutaneous coronary intervention: a meta-analysis of randomized controlled trials. Clin Nephrol 92(1):36–43. https://doi.org/10.5414/CN109701
Yalcinkaya M, Kerksiek A, Gebert K, Annema W, Sibler R, Radosavljevic S, Lütjohann D, Rohrer L, von Eckardstein A (2020) HDL inhibits endoplasmic reticulum stress-induced apoptosis of pancreatic β-cells in vitro by activation of smoothened. J Lipid Res 61(4):492–504. https://doi.org/10.1194/jlr.RA119000509
Yamashita S, Matsuzawa Y (2016) Re-evaluation of cholesteryl ester transfer protein function in atherosclerosis based upon genetics and pharmacological manipulation. Curr Opin Lipidol 27(5):459–472. https://doi.org/10.1097/MOL.0000000000000332
Yamashita S, Masuda D, Matsuzawa Y (2015) Did we abandon probucol too soon? Curr Opin Lipidol 26(4):304–316. https://doi.org/10.1097/MOL.0000000000000199
Yamashita S, Arai H, Yokote K, Araki E, Suganami H, Ishibashi S, K-877 Study Group (2018) Effects of pemafibrate (K-877) on cholesterol efflux capacity and postprandial hyperlipidemia in patients with atherogenic dyslipidemia. J Clin Lipidol 12(5):1267–1279.e4. https://doi.org/10.1016/j.jacl.2018.06.010
Yamashita S, Arai H, Bujo H, Masuda D, Ohama T, Ishibashi T, Yanagi K, Doi Y, Nakagawa S, Yamashiro K, Tanabe K, Kita T, Matsuzaki M, Saito Y, Fukushima M, Matsuzawa Y, PROSPECTIVE Study Group (2021) Probucol trial for secondary prevention of atherosclerotic events in patients with coronary heart disease (PROSPECTIVE). J Atheroscler Thromb 28(2):103–123. https://doi.org/10.5551/jat.55327
Zandbergen F, Plutzky J (2007) PPARalpha in atherosclerosis and inflammation. Biochim Biophys Acta 1771(8):972–982. https://doi.org/10.1016/j.bbalip.2007.04.021
Zanoni P, von Eckardstein A (2020) Inborn errors of apolipoprotein A-I metabolism: implications for disease, research and development. Curr Opin Lipidol 31(2):62–70. https://doi.org/10.1097/MOL.0000000000000667
Zanoni P, Khetarpal SA, Larach DB, Hancock-Cerutti WF, Millar JS, Cuchel M, DerOhannessian S, Kontush A, Surendran P, Saleheen D, Trompet S, Jukema JW, De Craen A, Deloukas P, Sattar N, Ford I, Packard C, Aa M, Alam DS, Di Angelantonio E, Abecasis G, Chowdhury R, Erdmann J, Nordestgaard BG, Nielsen SF, Tybjærg-Hansen A, Schmidt RF, Kuulasmaa K, Liu DJ, Perola M, Blankenberg S, Salomaa V, Männistö S, Amouyel P, Arveiler D, Ferrieres J, Müller-Nurasyid M, Ferrario M, Kee F, Willer CJ, Samani N, Schunkert H, Butterworth AS, Howson JM, Peloso GM, Stitziel NO, Danesh J, Kathiresan S, Rader DJ, CHD Exome+ Consortium, CARDIoGRAM Exome Consortium, Global Lipids Genetics Consortium (2016) Rare variant in scavenger receptor BI raises HDL cholesterol and increases risk of coronary heart disease. Science 351(6278):1166–1171. https://doi.org/10.1126/science.aad3517
Zewinger S, Drechsler C, Kleber ME, Dressel A, Riffel J, Triem S, Lehmann M, Kopecky C, Säemann MD, Lepper PM, Silbernagel G, Scharnagl H, Ritsch A, Thorand B, de las Heras Gala T, Wagenpfeil S, Koenig W, Peters A, Laufs U, Wanner C, Fliser D, Speer T, März W (2015) Serum amyloid A: high-density lipoproteins interaction and cardiovascular risk. Eur Heart J 36(43):3007–3016. https://doi.org/10.1093/eurheartj/ehv352
Zewinger S, Reiser J, Jankowski V, Alansary D, Hahm E, Triem S, Klug M, Schunk SJ, Schmit D, Kramann R, Körbel C, Ampofo E, Laschke MW, Selejan SR, Paschen A, Herter T, Schuster S, Silbernagel G, Sester M, Sester U, Aßmann G, Bals R, Kostner G, Jahnen-Dechent W, Menger MD, Rohrer L, März W, Böhm M, Jankowski J, Kopf M, Latz E, Niemeyer BA, Fliser D, Laufs U, Speer T (2020) Apolipoprotein C3 induces inflammation and organ damage by alternative inflammasome activation. Nat Immunol 21(1):30–41. https://doi.org/10.1038/s41590-019-0548-1
Zhao Q, Wang J, Miao Z, Zhang NR, Hennessy S, Small DS, Rader DJ (2021) A Mendelian randomization study of the role of lipoprotein subfractions in coronary artery disease. elife 10:e58361. https://doi.org/10.7554/eLife.58361
Zheng KH, Kaiser Y, van Olden CC, Santos RD, Dasseux JL, Genest J, Gaudet D, Westerink J, Keyserling C, Verberne HJ, Leitersdorf E, Hegele RA, Descamps OS, Hopkins P, Nederveen AJ, Stroes ESG (2020) No benefit of HDL mimetic CER-001 on carotid atherosclerosis in patients with genetically determined very low HDL levels. Atherosclerosis 311:13–19
Zhong S, Sharp DS, Grove JS, Bruce C, Yano K, Curb JD, Tall AR (1996) Increased coronary heart disease in Japanese-American men with mutation in the cholesteryl ester transfer protein gene despite increased HDL levels. J Clin Invest 97(12):2917–2923. https://doi.org/10.1172/JCI118751
Zvintzou E, Lhomme M, Chasapi S, Filou S, Theodoropoulos V, Xapapadaki E, Kontush A, Spyroulias G, Tellis CC, Tselepis AD, Constantinou C, Kypreos KE (2017) Pleiotropic effects of apolipoprotein C3 on HDL functionality and adipose tissue metabolic activity. J Lipid Res 58(9):1869–1883. https://doi.org/10.1194/jlr.M077925
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this chapter
Cite this chapter
von Eckardstein, A. (2021). High Density Lipoproteins: Is There a Comeback as a Therapeutic Target?. In: von Eckardstein, A., Binder, C.J. (eds) Prevention and Treatment of Atherosclerosis . Handbook of Experimental Pharmacology, vol 270. Springer, Cham. https://doi.org/10.1007/164_2021_536
Download citation
DOI: https://doi.org/10.1007/164_2021_536
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-86075-2
Online ISBN: 978-3-030-86076-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)