Abstract
The water present within trees when sugars and cellulose are formed is the source of hydrogen and oxygen atoms that are incorporated into tree-ring cellulose (see Chaps. 10 and 11). However, the isotope composition of relevant water pools is often unknown when trying to interpret δ18O and δ2H isotopic records in tree rings. This chapter focuses on the factors that can influence the O and H isotope ratios of source waters for trees. Trees generally use water that originated as precipitation, but this does not mean that the isotope ratios of water used by trees—predominantly taken up by roots from soils—and incorporated in cellulose exactly matches precipitation isotope ratios. Precipitation isotope ratios vary in space and time, and only a fraction of all precipitation infiltrates soils, reaches roots, and is ultimately taken up by trees. Considering species, soils, and climates may allow for predicting which fraction of water resides in the root-zone during the growing seasons, and how its isotope ratios deviate from that of average precipitation. Here we provide an overview of the terrestrial water cycle and the associated transport and fractionation processes that influence the stable isotope ratios of water used by trees. We highlight obstacles and opportunities to be considered, towards more accurately interpreting the records of O and H isotope ratios in tree cellulose.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
1 Introduction
Understanding the isotopic composition of the water that supplies trees (δSource) and is incorporated into sugars and cellulose is central to interpreting tree-ring δ18O and δ2H. When using stable isotope ratios in tree rings, δSource is often subtracted from the isotopic values in cellulose (δcellulose) to isolate the fractionation effects that reflect physiological and climatic controls over leaf gas exchange (Chap. 10, 11, 16); however, especially when using δcellulose as a proxy for past conditions, measurements of δSource are rarely available. Thus, predictions and assumptions are often necessary for interpreting tree-ring δ18O and δ2H. Analytical approaches have relied on predicting δSource by averaging across some compilation of isotopic values of local precipitation (e.g., Anderson et al. 1998), sometimes implicitly assuming static values (e.g., Helliker and Richter 2008). However, δSource varies throughout years, among years, among trees, and among sites, yielding measurement and prediction challenges.
The value of δSource that is recorded in an annual tree ring (after accounting for fractionation) should equal the mean isotopic ratio of xylem water (δxylem), weighted by the photosynthetic rate and integrated over the time when that tree grows radially. However, not only are δxylem measurements generally unavailable for long tree-ring records, using them would involve uncertainties (e.g., regarding the timing of sugars forming cellulose; see Chap. 13). Instead, δSource is commonly assumed to be constant and equal to the isotopic value of long-term-mean precipitation, but such assumptions have known limitations (e.g., as discussed in Chap. 16 and Roden and Siegwolf 2012). To better account for variations in δSource and more precisely interpret tree-ring cellulose isotope ratios, several factors should be considered if δSource estimates are needed (see Fig. 18.1).
To estimate the isotopic composition of δ source waters to trees, we need to not only consider the average isotopic composition of precipitation at a site. We must also consider which water reaches the soil layers from which roots take up water during the time when plants are transpiring, and how the isotope ratio of that water was influenced by mixing and fractionation processes before reaching roots. While these steps can rarely be quantitatively addressed, asking these questions can help in evaluating which factors should be considered to improve the accuracy of δSource estimates when interpreting tree-ring isotope ratios
The objective of this chapter is to provide an overview of how precipitation, soil, and tree water-uptake processes manifest in variations in δSource, and how our understanding of those processes informs the interpretation of tree-ring isotope ratios. The numerous biochemical synthesis and translocation processes that further alter δcellulose in trees, convolving the relationship between δSource and δcellulose are discussed in Chaps. 10, 11, and 13, and not covered here. Understanding the processes that control δSource found in the xylem can lead to practical assumptions and new ways of using tree-ring δ18O and δ2H, despite the uncertainties in δSource.
2 Precipitation Inputs and Their Spatially and Temporally Varying Isotope Ratios
Isotope ratios of precipitation (δPrecip) have been measured and monitored since the 1950s (Dansgaard 1953, 1954), producing a rich body of data and theory describing controls on meteoric-water isotope-ratio variation in space and time. Even these earliest studies recognized that isotope ratios of rainfall were related to meteorology, and that different storm systems and different climatic regimes produced rain and snow with contrasting isotope ratios (Dansgaard 1954; Craig 1961). By the mid-1960s, many of the underlying principles had been established, including equilibrium and kinetic fractionation effects that lead to isotopic differences between liquid (isotopically heavier) and vapor-phase (isotopically lighter) water (Fig. 18.2; Dansgaard 1964; Craig and Gordon 1965). These fractionation factors also lead to the progressive depletion of heavy isotopes from the atmosphere as air masses cool and lose water as liquid- or solid-phase condensate (following a so-called Rayleigh distillation process; Kendall and Caldwell 1998). Together these processes explain most of the isotopic variation in meteoric waters, though progress building on this early work has improved understanding of how dynamics and transport within the atmospheric water cycle are expressed in δPrecip across different systems and timescales. The fundamental theory of isotope fractionation in the water cycle was reviewed by Gat (1996), and Bowen et al. (2019) provide an update focusing on the integrated expression of these processes in large-scale climatic and hydrological systems.
Water isotope variations in dual-isotope space and the effects of climate variations on environmental water isotope ratios. Incoming precipitation generally falls along a meteoric water line (MWL) which can be described by either the global meteoric water line (GMWL, δ2H = 8δ18O + 10), or a local meteoric water lines (LMWL) if available for a specific site. The range of precipitation isotope ratio in a given location is strongly related to its temperature, elevation, continentality. The slope of 8, reflecting the relative equilibrium fractionation factors between δ2H and δ18O, defines the GMWL but it is also common that LMWLs have similar slopes. Variation in precipitation isotope ratios along those MWLs is strongly affected by season. Phase changes result in fractionation, with lighter isotopologues favoring the vapor phase. Thus, fractionation from evaporation causes the remaining liquid water to become isotopically enriched in the heavy isotopes because the departing vapor contains more light isotopes. Fractionation caused by evaporation also results in water isotopes deviating from the MWL, following an evaporation line with a lower slope than that of the MWL. This evaporation line slope relates to the relative humidity, where evaporation lines associated with higher humidity are more similar to the equilibrium fractionation line; at very low humidity, the slope is controlled by the relative diffusivity ratios of 1H2H16O to 1H216O versus 1H218O to 1H216O. Deviations from the MWL can be measured as d-excess—the deviation in δ2H units from the line passing through the origin (defined by the standard V-SMOW) along the equilibrium fractionation slope (equal to 8); thus, d-excess = δ2H -8δ18O and the GMWL has a d-excess of 10. To account for LMWLs not always matching the GMWL, lc-excess—the deviation in δ2H units from the LMWL, where LMWL lc-excess = 0—better quantifies the effects of evaporative fractionation on pools supplied by local precipitation
An assumption in many tree-ring studies has been that water used by trees reflects the isotopic composition of annually- or seasonally-averaged local precipitation (e.g., Anderson et al. 2002; Danis et al. 2006; Evans 2007; Zeng et al. 2016; Lin et al. 2019), which are well-known for the modern climate across much of the Earth through a combination of monitoring and statistical modeling (Aggarwal et al. 2010; Bowen 2010). This assumption implies that the processes delivering water to trees naturally integrate across precipitation inputs at a given site. However, systematic variation in δPrecip exists at many spatial and temporal scales relevant to ecohydrology, and the processes routing meteoric water to plants are selective so the validity of this assumption is often compromised. Here we introduce relevant sources of δPrecip variability, working from large to small spatiotemporal scales, and we discuss the question of routing and selectivity of water uptake in subsequent sections.
Long-term, annual-average δPrecip exhibits substantial (i.e. >1 ‰ δ18O) variation at spatial scales of tens to thousands of kilometers (Fig. 18.3a). In non-mountainous regions, local isotopic variation (at scales less than 100 km) is more limited (e.g., Price et al. 2008; Liu et al. 2010). In contrast, areas of high topographic relief form precipitation due to orographic lifting and produce strong and systematic decreases in δPrecip with increasing elevation (Dansgaard 1964; Poage and Chamberlain 2001). The average decrease in δ18O with elevation is approximately –0.28 ‰ per 100 m (Poage and Chamberlain 2001) and approximately –2.2 ‰ δ2H per 100 m. Consequently, downslope routing of groundwater, streamwater, or other surface waters that generated in higher elevations can result in water available to trees that is isotopically lighter than local precipitation; in such systems the resulting isotopic contrast among locally-available water sources has been used to identify sources of plant water uptake (Dawson and Ehleringer 1991; Chimner and Cooper 2004).
Maps of global variations in a typical average precipitation δ18O and b seasonal amplitude (i.e., absolute differences between mean values and either typical mid-summer or typical mid-winter values) of precipitation δ18O (adapted from Allen et al. 2019a). Circles reflect the locations of precipitation isotope monitoring sites, and their fitted average values and seasonal anomalies from those average values. In extra-tropical regions, the highest values occur in summer; elsewhere, the timing of seasonal cycles varies with the migration of the Inter Tropical Convergence Zone
Old groundwater (which could be a tree-water source in areas where that groundwater exfiltrates) can be isotopically distinct from modern precipitation. Evidence from paleo-waters, climate proxy records, and models also clearly suggest large shifts in average δPrecip as Earth’s climate system has changed. For example, estimated δPrecip across the northeastern USA during the last glacial period (~20,000 years ago) were likely lighter in 18O by 8 ‰ or more relative to modern precipitation (Jouzel et al. 1994). Paleo-waters are relatively common in the subsurface (Jasechko et al. 2017), and although these old waters mostly exist at depths that prohibit their use by plants, they represent an isotopically distinctive source of water that may, in rare cases, be accessed.
Potentially more common are ecosystems in which plants sample unevenly from the seasonal precipitation cycle, and thus the seasonality of δPrecip is an important determinant of source water composition (Fig. 18.3b). Isotopic seasonality is a common feature in the atmospheric water cycle, and is typical of most regions that exhibit climatic seasonality (Bowen 2008). In the extratropics, the lowest isotope ratios are nearly ubiquitously associated with the cold season, whereas isotopic seasonality at lower latitudes is largely associated with seasonal migration of the intertropical convergence zone and associated wet/dry climate cycles (Feng et al. 2009). The largest range of seasonal variation is found in the mid- to high-latitude continental interiors of the Northern Hemisphere, where inter-seasonal δ18O ranges can exceed 20 ‰ (Bowen 2008). Allen et al. (2019a, b) illustrated how seasonality of δPrecip influences δxylem, and that co-occurring species do not necessarily use the same precipitation (more below), so seasonal bias needs to be considered in δSource estimation.
Understanding inter-annual variations in δPrecip is especially important for tree-ring isotope studies. Inter-annual variability in annual-mean δPrecip can result from not only annual or seasonal δPrecip deviating from its normal pattern, but also from anomalous amounts of summer (higher δPrecip) or winter (lower δPrecip) precipitation in given years. Inter-annual variations in annual mean δPrecip often exceed 1.0 ‰ δ18O (Fig. 18.4). Among precipitation monitoring stations with at least five full years of data, standard deviations describing inter-annual variability in amount-weighted annual mean precipitation δ18O ranged from 0.2 to 1.6 ‰ (with a mean of 0.7 ‰ across 27 sites; Fig. 18.4). In general, inter-annual variability is highest—with maximum ranges among annual means exceeding 4 ‰ δ18O—in regions with larger seasonal δ18O amplitudes (r = 0.62), which also tended to have the lowest average δPrecip values.
Amount weighted annual precipitation δ18O means for individual years at GNIP sites. Dots represents individual years and the bars represent ± 2 standard deviations, describing inter-annual variations. These data are from GNIP sites (IAEA/WMO 2020) that have at least five full years of both monthly δ18O and monthly precipitation amounts; note that there is a several-fold larger set of sites if these criteria are loosened, allowing for gaps in records. Nonetheless, these sites (city and country code indicated on the horizontal axis) span a range of climates and latitudes (e.g., ranging from tropical sites in Uganda and Sri Lanka to northern latitudes in Canada). The monitoring sites are ranked by mean δ18O, demonstrating that the lower values are also associated with higher inter-annual variability in δ18O
The literature also includes a growing number of examples of δPrecip variation among events or over the course of individual storm events (e.g., Munksgaard et al. 2012; Coplen et al. 2015; Fischer et al. 2017). This evidence extends back to the work of Dansgaard (1953), who measured sub-daily changes in rainfall isotope ratios in Denmark associated with the passage of weather systems. In particular, this work showed a substantial increase in 18O/16O as condensation altitude lowered throughout a warm frontal storm, and this general pattern has been subsequently documented and modeled for several other well-organized synoptic-scale events (Gedzelman and Lawrence 1990; Coplen et al. 2008; Pfahl et al. 2012; Aemisegger et al. 2015). While inter-event variations may be mostly damped out in soils and thus not detectable in tree rings, such patterns can be strongly accentuated in large-scale systems such as tropical and extratropical cyclones; in those storms, δPrecip changes of 8–10 ‰ (δ18O; 60 to 80 ‰ for δ2H) have been observed over timescales of hours and/or spatial scales of ~100 km (Gedzelman et al. 2003; Coplen et al. 2008; Good et al. 2014a, b). These storms can produce heavy rains with unusually low isotope ratios, in particular (Lawrence and Gedzelman 1996), and evidence suggests that these isotopic anomalies can be passed along to trees and tree rings if the events are especially large or anomalous events (Weiguo et al. 2004; Miller et al. 2006; Berkelhammer and Stott 2008). The evolution of precipitating storms can be complex and spatially heterogeneous, particularly for less organized and/or convective systems. Routine prediction of isotopic variations throughout individual events has not been demonstrated, and many examples of seemingly chaotic fluctuations (Kennedy et al. 2012) suggest that within-storm variation is the norm. However, predicting these patterns remains a research frontier, and thus we do not really know their potential to bias soil water isotope ratios, relative to average δPrecip, if different-sized events infiltrate the soil differently. With that stated, small-scale variations are often less relevant to tree rings because they integrate water signals across longer timescales.
In forests, canopy interception and interception loss can alter the isotopic composition of precipitation reaching soils. During events, slight evaporation occurs from waters stored in canopies that may evaporatively enrich the water that is eventually transmitted downward (i.e., throughfall). Perhaps more importantly (Gat and Tzur 1968), interception also involves the selective transmission of throughfall during events and the omission of the water stored at the end of events that evaporates completely. Given that δPrecip can trend upward or downward throughout events, this selective transmission can result in erratic differences between event-mean throughfall and open-precipitation δPrecip. A synthesis of interception studies (Allen et al. 2017) shows that, on average, the net-precipitation below forest canopies was mostly between 0.2 ‰ lighter to 0.6 ‰ heavier in δ18O than open precipitation (the mean long-term enrichment was 0.19 ‰ δ18O); while individual-event differences can be much larger than these mean values, those short-term variations are erratic. Accordingly, because individual events are small compared to the storage in soils, event-level effects due to interception are likely inconsequential to variations in δSource and the coarse-resolution signals inferred from tree rings. Nonetheless, enrichment is observed on average over long time scales and thus would represent a small systematic bias if ignored.
Resources to aid in predicting δPrecip inputs to a location are widely available, although subsequent infiltration and uptake processes need to be considered to predict δSource. Databases of δPrecip and volumes are available for thousands of locations with monthly or better resolution through the IAEA’s Global Network of Isotopes in Precipitation (GNIP; Aggarwal et al. 2011), and the Water Isotope Database (waterisotopes.org; Putman and Bowen 2019). Additionally, the National Ecological Observatory Network (NEON) provides a rapidly growing public dataset containing time-series of biweekly δPrecip for sites spanning the United States; however, none are yet long time series, which are especially useful (see Sect. 18.5 and Fig. 18.4). Country-specific precipitation-isotope monitoring networks also exist for Switzerland, Austria, Germany, and elsewhere. Global interpolated data products are also available; for example, see those generated from the Water Isotope Database, which can be queried online (see IsoMap http://isomap.org and the Online Isotopes in Precipitation Calculator, http://wateriso.utah.edu/waterisotopes/) or downloaded as maps (Bowen et al. 2014). Another option is to develop predictive isoscape maps for the specific study domain (Bowen 2010). In addition to the raw isotope values provided by GNIP and elsewhere, Allen et al. (2019a) provides fitted sine coefficients that describe δPrecip seasonality at > 600 sites globally, along with various site-characteristic covariates. Another promising source of δPrecip estimates, especially for interpreting isotope paleo-proxy records, is isotope-enabled circulation models that output global spatiotemporal variations in δPrecip over past centuries as estimated from sea surface temperatures (Dee et al. 2015; Konecky et al. 2019). Any of these data products can be useful for constraining δSource estimates in areas where δPrecip data are not available.
3 From precipitation to Soils and Other Subsurface Storages
Nearly all water used by plants originates from precipitation, whether it be from fog, rain or snow. This precipitation is available to plants from different pools within ecosystems. Two predominant pools are water held under tension in soils (the vadose zone), and water held under pressure in groundwater (the saturated zone). Each can be complex water storages that vary spatially and temporally in isotopic values. Water enters these pools by infiltrating locally or flowing in from upslope areas. In most cases, the local infiltration of rainfall and snowmelt provides water to plants. In other cases, shallow groundwater can directly supply trees. These isotopically diverse subsurface waters largely determine the δ2H and δ18O in tree rings; therefore, considering how water resides, mixes, percolates, and evaporates in the subsurface is critical to relating precipitation patterns to plant-tissue isotope ratios.
Two aspects mainly affect the isotope ratios of the water in the vadose zone: (1) the transport, mixing and storage of precipitation with varying isotopic composition within the vadose zone; and (2) the changes of water isotope ratios caused by fractionation during evaporation from soil. Here, we introduce the processes that result in variability in subsurface pore-water isotope ratios. Understanding such processes (e.g., soil–water turnover times or typical depths of evaporative fractionation) can aid in understanding which precipitation may be stored in a soil during the growing season and to what degree soil waters might be evaporatively enriched in heavy isotopes.
3.1 Water Transport and Mixing
Given that precipitation and δPrecip are highly variable, understanding how precipitation travels through the subsurface is key to knowing which precipitation (and associated isotope ratios) will be accessible to plants. Water transport in the unsaturated zone occurs by various mechanisms, with characteristics that can be abstractly characterized as well-mixed flow, uniform translatory (piston) flow (Hewlett and Hibbert 1967) or non-uniform preferential flow (Beven and Germann 1982); in reality, aspects of all of these flow behaviors are relevant.
Subsurface waters are rarely well mixed (Penna and Meerveld 2019), even though well-mixed assumptions are often implicit to common representations of soil and groundwater systems (e.g., Lawrence, et al. 2011). In hypothetical well-mixed systems, a single isotope ratio could be assumed for an entire storage pool, representing the mixture of new inputs and prior storages; in that scenario, the fraction of water extracted by plants, as well as that draining downward from soils, are both unbiased representations of the stored precipitation. This means that more recent precipitation will dominate the storage, and the ‘age’ of the water in storage will match the turnover time (storage/flux). This transport scenario will yield spatially uniform soil–water isotope ratios that vary in time, mimicking a damped and lagged seasonal precipitation isotope cycle. Such assumptions may not apply for soil or shallow groundwater (Penna and Meerveld 2019), but may better apply for large groundwater aquifers that store a mixture of many years or decades of past precipitation; in those conditions, values are relatively spatially and temporally uniform (Vogel and Van Urk 1975), supporting accurate estimates of δSource waters if it is known that trees are using groundwater.
An older conceptual model of soil–water infiltration is that of uniform translatory flow (also known as plug flow or piston flow), where newly infiltrating precipitation pushes down the previous precipitation stored in soil pores; however, this model has decreased in popularity since the majority of soil isotopic data does not fit this model (Fig. 18.5). This uniform-flow mechanism leads to depth-stratified soil waters, with the shallowest soils containing the most recent inputs (e.g., Fig. 18.5a) and increasingly older precipitation with depth. If newly infiltrated water entirely replaces previously stored water (“piston flow”), (a) no water in the profile will be older than the turnover time and (b) the temporal variations in δPrecip would be preserved by depth in soil water isotope profiles (Fig. 18.5a), and (c) tree water isotope ratios would look similar to that of long-term mean precipitation if soils are deep enough to hold years of precipitation and roots take up soil water uniformly across the rooting profile (Sect. 18.4). Alternatively, if trees predominantly use shallow soil water, δSource would approximate rainfall that fell recently (i.e., usually during the growing season). The rate that this oscillating signal propagates downward (e.g., Fig. 18.5a) depends on how much precipitation falls and how much the soils store. In reality, because flow is never entirely uniform, variation in soil water isotope ratios typically decrease substantially with depth due to mixing (dispersion) during percolation (e.g., Thomas et al. 2013; Barbecot et al. 2018; also see Fig. 18.5).
Example profiles of soil–water δ18O (left) and lc-excess (right) for (a and b) a vineyard near Freiburg, Germany, (c and d) a humid, cold, continental pine forest in southeastern Canada, and (e and f) a Mediterranean pine forest in the Pyrenees, Spain. For c–f, the error bars represent ±2 SD and the data show beginning, the middle and end of the growing season. All vertical colored and black lines represent weighted seasonal or annual averages of the δ18O for the sites. For e and f, both the so-called “mobile” (circles) and “bulk” (squares) water values are shown. For further details, see (Sprenger et al. 2016a, 2018b, 2019)
With non-uniform preferential flow, infiltrating precipitation follows the route of least resistance; new inputs differentially bypass storages to yield an isotopically heterogeneous subsurface (Brooks et al. 2010; Sprenger et al. 2018a, 2019). The more that infiltrating water bypasses storages, the more likely it is that soil matrix water will reflect older precipitation that resides across seasons and disproportionately supplies plants (often referred to ecohydrologic separation; Brooks et al. 2010). How much water flows in preferential flow paths versus trickling through the matrix can depend on soil texture, structure, wetness (Weiler and Naef 2003), biological activity (Schaik et al. 2014), and input amount or intensity (Nimmo 2011). With preferential flow, the refilling of soil water stores may occur periodically, resulting in, for example, the under-representation of precipitation that fell at higher intensities (Jasechko and Taylor 2015; Allen et al. 2019b) or when soils are wet (Brooks et al. 2010; Sprenger et al. 2019). These preferential flows have been observed through showing that (a) waters quickly passing through soils are more similar to recent precipitation than is the entirety of water in soils (including finer pores), and (b) the waters stored in the soil matrix are older than they would be if well-mixed or uniform-flow conditions dominated (Fig. 18.5c; see Berghuijs and Allen 2019). Conventional wisdom in hydrology is increasingly gravitating towards assuming that soil–water transport is highly preferential, especially in forest soils (Flury et al. 1994; Nimmo 2011), and thus δSource can be potentially biased towards reflecting precipitation from specific times or events when recharge occurs.
To estimate δSource, researchers should not only consider these soil–water transport concepts but also the general empirical patterns seen in soil–water isotope studies. Real-world soil water transport does not solely reflect any one of these processes, even if we should expect evidence of preferential flows in shallower soils and mixing in deeper soils. These dynamic subsurface flow processes affect the pattern of subsurface isotope ratios in ways that are difficult to predict; this is an active area of research in ecohydrology, with recent progress in generalizing patterns across climates (Sprenger et al. 2016b; Allen et al. 2019b) and across inter-annual climatic variations (Berkelhammer et al. 2020). In general, the isotope ratios of soil water change over the growing season from lower isotope ratios in spring to higher in summer and fall (Fig. 18.5b, c). That is to say, early growing season soil waters are typically lighter than average δPrecip, because they reflect storage of cold-season precipitation, and (at least in shallow soils) later growing season soil waters are typically heavier than average δPrecip (Figs. 18.5). Late growing-season shallow soil water is not only isotopically heavier because of the heavier isotopic composition of summer rainfall (e.g., compare the red and blue vertical lines in Fig. 18.5), it also results from progressive evaporative fractionation (described in the next section). Deeper soils tend to reflect a more wintry signature, even in regions with summer precipitation (Ehleringer et al. 1991; Williams and Ehleringer 2000; Martin et al. 2018; Allen et al. 2019b). Woody plant xylem frequently expresses this lighter isotope ratio associated with deeper or older precipitation.
3.2 Evaporation Fractionation of Soil Water
Just as in leaves (Chaps. 10 and 11), evaporation from soils results in the enrichment of heavy isotopes in the residual soil water because light isotopes are preferentially evaporated (Gat and Gonfiantini 1981); however, unlike in leaves, a constant resupply of water rarely exists to move up and replace the evaporated fraction. Evaporation results in heavy isotopes accumulating in the residual soil water until new precipitation mixes with or displaces the enriched pool. Thus, soil water will often be more enriched in heavy isotopes relative to the original precipitation water (Fig. 18.2). These effects are very prominent in shallow soil–water samples (Fig. 18.6), although, the most evaporatively enriched soil–water pools are generally of small volume and not necessarily a major contributor to plants (discussed further in Sect. 18.4). Nonetheless, studies inferring uptake depths or precipitation sources should consider potential alterations in δSource attributable to evaporation (Tang and Feng 2001).
Soil–water isotope values compared to the estimated seasonal precipitation inputs at those sites. All soil–water values are from the growing season, when possible separated as early (April-June) versus late (July–September) growing season values. Summer and winter precipitation are defined relative to the solar cycle (thus, for the tropical sites, winter is not necessarily colder). “Shallow” includes depths from 5–30 cm; “Deep” includes depths from 30–100 cm (and to 300 cm for the Nebraska and Luxembourg sites). The studies are as follows: Arid shrubland in China (Zhou et al. 2011), Arid woodlands in Arizona (Snyder and Williams 2000), mesic woodland in Nebraska (Eggemeyer et al. 2009), Floodplain forest in Switzerland (Bertrand et al. 2014), Mediterranean oaks in Portugal (Kurz-Besson et al. 2006), sites in Luxembourg (Sprenger et al. 2016c), Subtropical forest in southern China (Rong et al. 2011), humid montane forest in North Carolina (Berry et al. 2014), a Humid conifer forest (Brooks et al. 2010), and a montane tropical rainforest (Goldsmith et al. 2012). Precipitation data are calculated using the online isotope in precipitation calculator: http://wateriso.utah.edu/waterisotopes/. Note that precipitation estimates can be imprecise; for example, in the Goldsmith et al. study, positive δ18O values for summer rainfall are common. Also, note that the most evaporative enriched soil waters are unlikely to be a substantial source to plants because there is usually minimal water content in these locations
Kinetic fractionation of stable isotopes of water leads to sources deviating from the meteoric water line (Fig. 18.2) because of differences in the relative diffusivities between 2H1HO and 1H2O compared to 1H218O and 1H216O (Horita et al. 2008). Until recently, deviations from meteoric water lines were mostly quantified using d-excess (δ2H – 8 × δ18O), where 8 is the ratio of equilibrium fractionation factors of O and H, defining the slope of the global meteoric water line (Dansgaard 1964); however, local meteoric water lines (LMWLs) often deviate from a slope of 8 (Putman et al. 2019). Thus, variation in d-excess does not imply evaporation because precipitation inputs along a LMWL with slopes different from eight will also vary in d-excess, regardless of any subsequent evaporation. Accordingly, the line-conditioned excess (lc-excess), defined relative to LMWLs (lc-excess = δ2H – slopeLMWL × δ18O – interceptLMWL) is more appropriate for inferring terrestrial evaporation (Landwehr and Coplen 2006). Shallow soil lc-excess values can be well below zero, even in humid climates (Sprenger et al. 2016). Low lc-excess values are especially common following extended warm periods without rain (Sprenger et al. 2018b), e.g., in summer and early autumn (Fig. 18.5d). Although few tree ring isotopic studies have used both δ2H and δ18O from plant tissue in conjunction (but see Voelker et al. 2014), this area of research is advancing because this dual-isotope approach can provide new insights into past evaporative conditions (see Chaps. 10 and 11). Alternatively, evaporation effects in dual isotope space can also be ‘compensated’ by assuming theoretical evaporation lines (which can be notably different than empirical ‘evaporation’ lines) and relating soil and plant waters to their precipitation sources (Benettin et al. 2018; Bowen et al. 2018), for insights into how soil–water is recharged by precipitation.
3.3 Predicting Soil Water Isotope Ratios from δPrecip
Methods for predicting the complex interplay between newly infiltrating precipitation and previously stored water (that is often evaporatively fractionated) could support more accurate uses of tree-ring isotope ratios. Hydrologists use both mechanistic simulation models and statistical, “lumped” models to represent soil water processes. Parameterizing those models is data intensive, requiring measurements of soil water or outflow isotope ratios (e.g., Braud et al. 2005; Haverd and Cuntz 2010; Stumpp et al. 2012; Mueller et al. 2014; Sprenger et al. 2015) to capture the dominant mixing processes (Sect. 3.1), towards generating simulated rooting-zone water isotope time series. While such models can be effective, requiring data-driven parametrizations undermines their usefulness with long tree-ring isotopic records. Given that soil water is an ensemble of fractions of previous precipitation events, soil–water can be estimated as a distribution of those past events (Groh et al. 2018; Benettin et al. 2019); thus transfer functions capturing a statistical relationship between inputs and outputs may be more practical than mechanistic models and outperform estimates implying that soils contain an even mixture of annual precipitation. Otherwise, syntheses of growing season soil–water isotopes measurements show some generalities that aid in constraining expectations.
Soil water δ18O generally deviates from local δPrecip across a wide range of sites (as shown here; Fig. 18.6). Shallow soil–water is isotopically heavier than the typical seasonal precipitation inputs, especially in arid regions, demonstrating strong evaporative enrichment. This pattern diminishes from arid to humid (left to right), with exceptions occurring in sites with extended dry seasons (e.g., during the dry summer in Portugal and following the dry winter in Mexico; Fig. 18.6). In regions where the growing season involves more continuous evaporation, soil water δ18O in the late growing season is usually higher than early growing season δ18O (Fig. 18.6); thus, changes in soil water δ18O throughout a season can be confounded with changes in depth of uptake. Importantly, these highly enriched shallow soil waters should be scarce and under high tension after undergoing significant evaporation, and thus they are not likely to be the primary water source to trees; furthermore, most tree radial growth occurs near the beginning of the growing season (Zweifel et al. 2006; Swidrak et al. 2013), often prior to the development of strong evaporation signatures in soil water isotope ratios. Hence, tree source waters recorded in tree ring δcellulose are unlikely to have δSource values that are as high as late-season shallow soil water values (Fig. 18.6).
In wetter regions, evaporation effects are less prominent and thus isotope ratios in soils tend to be bounded by seasonal δPrecip; however, conditions need to be considered on a case-by-case basis. For example, the Swiss floodplain forest (Fig. 18.6) is likely influenced by a riparian aquifer, explaining its lack of evaporative signal; in general, groundwater tends to be dominated by winter precipitation (Jasechko 2019), and in this case, groundwater is likely sourced from higher elevation precipitation (Bertrand et al. 2014). Another prominent feature across these sites is that deeper soil waters are typically isotopically lighter, because they generally experience less evaporation and water with this evaporated signal only partially trickles downward (i.e., preferential flow), and less variable because of mixing with stored preferential flow; furthermore, the conventional wisdom is that winter precipitation tends to percolate deeper because surface soils are drying less during summer. The deeper soil waters are mostly enveloped by the typical summer and winter δPrecip; notable exceptions are the driest site, where deeper soil waters may still show strong evaporative enrichment, and the tropical rainforest in Mexico, where the original study (Goldsmith et al. 2012) reports higher precipitation δ18O than those predicted by the Online Isotopes in Precipitation Calculator (waterisotopes.org). While using these precipitation predictions provides consistency across studies, δPrecip predictions can be uncertain across topographically complex regions, especially when predicting δPrecip in individual years (Sect. 18.2).
These patterns shown (Fig. 18.5) are explainable, albeit not necessarily a-priori predictable. Nonetheless, our first-order theoretical expectation should be that with larger fluxes (i.e., precipitation inputs) or shallower rooting zones (smaller storages), soil–water turnover times are higher and thus they should more reflect in-phase precipitation isotope values (i.e., growing season). This general pattern is shown in xylem water collected in a snapshot sample across >100 sites in Switzerland, defining steep climate and elevation gradients (Fig. 18.7a). While estimated δPrecip decreases near monotonically with elevation, xylem water δ18O does not; these measurements highlight the influence of climate gradients on δSource. Deviations of xylem water (and thus soil–water available to those plants) from mean annual δPrecip increase with elevation and precipitation amount (Fig. 18.7b), which mostly co-vary in Switzerland; thus, the drier region trees used out-of-season precipitation whereas the wet-region trees use growing-season precipitation, demonstrating the higher turnover in wetter regions. This one example demonstrates how considering the relationship between more precipitation and higher turnover times can guide evaluations of δSource variations across sites.
a Annual precipitation and xylem water δ18O versus elevation and b xylem water δ18O differences from precipitation δ18O versus mean-annual precipitation amount in Switzerland; elevation and annual precipitation are largely collinear here. Panel a shows decreasing δ18O of precipitation with increasing elevation, which is true for most locations on Earth. However, xylem-water (mid-summer values) δ18O did not decrease with elevation. This probably relates to the greater precipitation amounts in higher elevations driving shorter soil–water turnover times, such that the water stored in soils is more in-phase with current precipitation (i.e., wetter climate soils store more summer precipitation in summer). Panel b shows the relationship between xylem-water δ18O at the same sites, but as deviations from annual precipitation δ18O which range from −3 to 5 ‰ δ18O; in wetter areas, this δ18O deviation increases (r = 0.59 and Spearman’s ρ = 0.47, p < 0.001), consistent with the greater soil–water turnover. Considering these relationships allows for avoiding the systematic biases that can result from assuming that xylem water equals annual precipitation across climatologically distinct sites
4 Roots and Uptake Patterns
Patterns in root water uptake add another layer of uncertainty to assumptions about how δxylem relates to annual δPrecip. Earlier, we discussed how soils do not behave like sponges that store and perfectly reflect the isotopic composition of annual rainfall. Similarly, the water that roots take up does not perfectly reflect the isotopic composition of the average soil water. In this section, we highlight how roots take up water, and how those processes influence δSource.
Water flows from soils into plants along a passive water potential gradient known as the soil–plant atmosphere continuum (SPAC). Water enters trees primarily through their non-suberized fine roots along an apoplastic route, and minimally through a symplastic route where water is actively transported across cell membranes. Water traveling along the SPAC apoplastically does not isotopically fractionate (Dawson and Ehleringer 1991, 1993), albeit analytical errors can occur and soil and plant water are not always identical in a given instance in time (Vargas et al. 2017; Penna et al. 2018; Barbeta et al. 2020). However, plants that do use symplastic transport for water uptake, such as salt tolerant plants that exclude salts from uptake pathways, have shown isotopic fractionation of 2H during water uptake (Lin and Sternberg 1993; Ellsworth and Williams 2007). Many salt-excluding species have a highly developed Casparian strip which impedes apoplastic water movement into the endodermis, forcing water through symplastic routes crossing cell membranes. The symplastic route of water movement allows for exclusion of unwanted molecules such as salts, but costs energy for active transport. The energy required for disassociating hydrogen bonding of individual water molecules is greatest for water molecules containing 2H, compared to other isotopologues of water (Clark and Fritz 1997). As a result, water within stems of salt tolerant species has lower δ2H values relative to the source water, without having measurable differences in δ18O (Lin and Sternberg 1993; Ellsworth and Williams 2007).
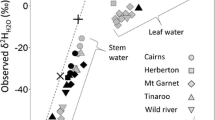
For the majority of tree species with apoplastic water uptake, water within the xylem approximates a spatially integrated mixture of soil water from a plant’s population of fine unsuberized roots, proportional to their uptake rates. Therefore, the origin of water within the xylem will depend on a plant’s root distribution and water uptake rates. Roots decline in density asymptotically with soil depth, with the majority of plant roots occurring within the upper 30 cm of the soil profile (Gale and Grigal 1987; Jackson et al. 1996; Warren et al. 2005). For tree species, the percentage of roots in the upper 30 cm varies from a high of 83% in boreal forests, to a low of 52% in temperate coniferous forests (Jackson et al. 1996). However, variation between sites was highly dependent on soil texture, water availability, and depth to water table, permafrost, or rock. In general, roots are deeper in seasonally dry regions and shallower in regions where soils remain wet (Fan et al. 2017). Thus, in humid regions, not only do soils hold waters sourced from recent, growing-season precipitation (Sect. 18.3), trees are more shallowly rooted and likely use shallower water which should generally be composed of more recent precipitation (Figs. 18.7 and 18.8). In contrast, drier regions likely contain a long-duration mixture of past precipitation and trees are more deeply rooted and thus likely use deeper waters that have dampened and lagged expressions of δPrecip. Xylem water from deeply rooted trees should rarely express the highly evaporated waters observed in shallow soils in the same sites (Figs. 18.7 and 18.8). Some roots can extend down far below the soil bedrock interface allowing them to access deep water reserves (Canadell et al. 1996). Thus while the majority of roots are located in the top of the soil profile, rooting depths can extend below 50 m allowing trees access to many isotopically different water pools.
Mean growing season xylem-water isotope values as a deviation from annual precipitation δ18O, plotted against site mean annual precipitation amount; these are data from the same sites as those in Fig. 18.6, minus the Luxembourg sites because they lacked xylem water measurements. Note that xylem water does not reflect evaporative enrichment above δPrecip as strongly as does soil water (Fig. 18.6) because trees often use deeper waters, although the high variability in the driest site likely results from evaporation effects. Xylem water in wetter sites is generally more enriched compared mean precipitation (r = 0.67, p < 0.05), in contrast with the expected effects of evaporative enrichment being greater in drier sites; this presumably reflects the presence of shallower roots and more in-growing-season precipitation in soils of wetter regions
In some cases, a subset of roots can account for the majority of water uptake, which can explain dissimilarities between physical root distributions and isotope-inferred uptake depths. If all roots were equally taking up water, then the proportion of water acquired from different depths would match the distribution of fine roots; however, root water uptake rates vary with depth depending on root hydraulic properties, soil moisture content and soil water potential. When soils are at field capacity and both soil water content and plant water potential are high, small gradients in water potential are associated with relatively large fluxes of water. The majority of root water uptake during these periods is from the upper soil layer where rooting density is high. As soils dry, equivalent drops in soil water potential yield smaller and smaller volumes of water since soil moisture release curves relating water potential to water content are non-linear (Selker et al. 1999). Root water uptake shifts to deeper layers as the soil moisture content in the upper soils decreases (Warren et al. 2005; Brooks et al. 2006). The ability of roots to extract water also varies by species and with depth in the soil. Meinzer et al. (2007) found that relative uptake per root area increased with rooting depth for both Douglas-fir and western hemlock. Plants with roots extending far beyond the soil can access rock water and deep water aquifers which can sometimes make up a substantial portion of xylem water even though only a small proportion of roots extend to those depths (Schwinning 2010; Oshun et al. 2016), regardless of shallow root abundances. Phreatophytes–plants using groundwater–often have deep taproots reaching water tables which can account for a large portion of their water uptake, even though the taproot is a small portion of their rooting system (Burgess et al. 2000; Hultine et al. 2003a, b). Thus, roots at deeper depths are not only likely to be rooted in soils or substrates that maintain moisture for longer, these deep roots can transport more water, compensating for the relatively fewer roots at deeper depths in soil profiles. Ultimately, the isotopic composition of xylem water of an individual tree will reflect the integrated isotopic composition of soil water accessed by that tree’s roots, weighted by those root’s water uptake rates.
Isotopic approaches has been used extensively to understand the dynamics of root water uptake, and niche partitioning of soil water resources by plants within a community (e.g., Ehleringer et al. 1991; Meinzer et al. 2007; Goldsmith et al. 2012; Guo et al. 2018). Inferring uptake depth using isotopes requires known isotopic variation within the soil either spatially, temporally or both (see Sect. 18.3). Generally, spatial variation in soil water isotopic composition is measured along a depth profile, but spatial variation within a soil layer is also likely (e.g., Goldsmith et al. 2019), and the isotopic composition of water being extracted by roots may not match the bulk isotopic composition of soil water within a layer. Where exactly root tips are located within a layer (e.g., within or around soil pedons) will influence water potential and content they experience, and which water pool they can take up. Spatial variation in all of these factors is complicated even when soils are relatively uniform (McCully 1999; Carminati et al. 2010). While water flows passively along a water potential gradient, roots are alive and the rhizosphere around roots are filled with living organisms that can excrete compounds that can change water potentials (McCully 1999). Additionally, mycorrhiza have been shown to transport water that is taken up by plants (Warren et al. 2008). Understanding these living interactions and how they influence water traveling along the SPAC has implications for the isotopic composition of xylem water; this is an active area of research.
Another complicating factor in determining the source of water uptake within the soil is hydraulic redistribution. Hydraulic redistribution is the passive movement of soil water from one region of the soil to another via roots along a water potential gradient (Caldwell and Richards 1989; Caldwell et al. 1998; Neumann and Cardon 2012). When plants are transpiring, the water potential gradient within the plant drives water flow to the leaves and the atmosphere. However, if transpiration stops, e.g., during the night, then water can flow along potential gradients within the soil. Generally, water will flow from moist soils at depth to the dryer surface soils, and this process was originally described as hydraulic lift. However, water can flow downward via roots after rains (Ryel 2004), as well as laterally (Brooks et al. 2002, 2006), prompting the more generalized term of hydraulic redistribution. Hydraulic redistribution would act to mute isotopic variation within the soil profile, but the volumes of water that are released to the soil are small compared to the volume of water within the soil (Neumann and Cardon 2012). For example, at 5% soil water content, 10 cm of soil would hold 5 mm of water, but rates of hydraulic redistribution are around 0.02 mm within 10 cm of soil per night, several orders of magnitude smaller than the water content (Warren et al. 2007). In addition, the water excreted from the root during the night is the first water to be taken up as transpiration begins again, so isotopic differences between redistributed water and the soil water would not accumulate over time. Thus, the resulting isotopic shifts in soil water are unlikely to be detectable unless a strong isotopic label is used (Brooks et al. 2002, 2006), implying that they are unlikely to strongly influence tree-ring isotope applications.
Roots extract and integrate water from different pores and depths, and thus δSource variations are dampened compared to those of individual soil samples. Nonetheless, temporal variation in soils and xylem water sources can manifest in δSource varying spatially and temporally, introducing challenges to tree-ring δ2H and δ18O interpretations. Furthermore, xylem water concurrently sampled from individual trees or branches in the same site can vary by 1–2 ‰ δ18O (Goldsmith et al. 2019). Thus, uncertainties and variations in source waters (and thus δSource) should always be assumed. Nonetheless, plant-rooting characteristics are a tertiary control over δSource, because roots can only extract water from that which is input to the system as precipitation and held in the subsurface rooting zone (i.e., the primary and secondary controls). Therefore, discussion of how rooting depth relates to δSource depends on some understanding of δPrecip, its variations, and how it resides in soils.
Foliar uptake is an alternative uptake pathway that bypasses root and soil processes. The uptake of water on leaf surfaces, typically occurring under foggy, humid conditions (Simonin et al. 2009; Gerlein-Safdi et al. 2018) when plants have lower water potentials (Goldsmith et al. 2013), represents a potentially different isotopic signature. These are generally small supplements of water (Gotsch et al. 2014), and identifying their magnitude is difficult because the isotopic effects of foliar uptake are also not always distinguishable from the back diffusion into stomata that occurs with transpiration (Goldsmith et al. 2017; Lehmann et al. 2017; also see Chaps. 10 and 11). Foliar uptake effects on tree-ring isotope ratios are not well described, but if boundary layer vapor isotope ratios are roughly in equilibrium with precipitation (see Fiorella et al. 2019), foliar uptake may not have a substantial effect on δSource; this is because the fluxes are small and the effects match those of the back diffusion already accounted for in leaf-atmosphere fractionation representations (Chap. 10).
5 Applications and Practical Considerations
5.1 Assuming δSource Values to Study Leaf-Climate Interactions
Knowing the value of δSource is critical for many applications using tree-ring δ18O (and δ2H) because much of the theory is based on isotopic shifts relative to δSource and is calculated as Δcellulose ≈ δcellulose − δSource (Barbour 2007). Often Δcellulose is used to infer leaf-atmosphere interactions, because transpiration and vapor exchange with the atmosphere alter the isotopic ratio of leaf water in systematic ways (Chaps. 10 and 11). As a result, Δcellulose is used as a proxy for climate and leaf physiology, after accounting for the many mixing and fractionation processes that occur during the incorporation of leaf water into cellulose (see Chaps. 10, 13, and 16). Thus, accurate knowledge or predictions of δSource through time are key to accurate Δcellulose estimates and interpretations.
A common assumption for estimating δSource to calculate Δcellulose of an annual increment is that δSource is equal to long-term mean δPrecip at a given location. Indeed, trees integrate soil water both temporally and spatially so that the annual value of δSource is often bounded by the annual range of δPrecip inputs to a site (unless the trees use shallow soil waters that are especially fractionated by evaporation). Soils could possibly be an unbiased mixture of a year’s precipitation, but in most scenarios (especially in more humid regions), the rooting-zone in soils can only hold a small fraction of a year’s precipitation: e.g., consider that most roots are within the upper 30 cm of soil (Gale and Grigal 1987), which could hold a maximum of 90 mm of precipitation if the field capacity is 30%. In contrasting settings with deep roots, water may only occasionally percolate to the deeper depths that disproportionately supply trees. Thus, where evaporation ratios or seasonal variation in δPrecip is large, δSource could deviate (systematically) from long-term mean δPrecip by several ‰ δ18O or tens of ‰ δ2H.
Assuming δSource is always equal to long-term mean δPrecip, and thereby also assuming that δPrecip is constant over time (Fig. 18.4) can confound interpretations of inter-annual variations in Δcellulose. To mitigate temporal uncertainties, where possible, one can:
-
Select sites where soil or xylem water isotope ratios have been measured over years of contrasting meteorological conditions to test how dry or wet years affect the soil–water or xylem-water isotope ratios, e.g., to rule out their influence on signals that are assumed to be due to Δcellulose. For measuring soil water, bulk samples (e.g., taken with an auger) should be used because suction lysimeters sample water that is more likely to drain quickly and not contribute to transpiration. For xylem water, twigs or cores should be collected. Both soils and xylem material should be sampled a few times throughout the growing season and extracted water by cryogenic vacuum distillation (see the discussion on sampling in Penna et al. 2018).
-
Select sites where inter-annual variations in δSource are small, e.g., because δPrecip variations are small, or trees use streamwater or groundwater with a less dynamic stable isotope signature (e.g. Ulrich et al. 2019). Interestingly, these site conditions for stable δSource contrast with the droughty edaphic sites where tree-ring widths are more responsive to temperature, precipitation, or humidity, and thus droughty sites are often preferred for dendroclimatological applications.
-
Select sites where long records of δPrecip exist, or where magnitudes of inter-annual variations in mean δPrecip can be constrained using publicly available δPrecip datasets, which should provide a basis for estimations of uncertainties in assuming constant δSource.
-
If there are no means of estimating δPrecip and δSource variability, then uncertainties in δSource should be assumed and interpreted accordingly. While values of assumed uncertainty cannot be universally prescribed, Figs. 18.4 and 18.8 show potential ranges of inter-annual δPrecip variability and δSource- δPrecip differences that can occur.
Assuming δSource is always equal to long-term mean δPrecip can confound interpretations of spatial variations in Δcellulose across climate gradients. For example, deviations of δSource from mean annual δPrecip can depend on climate (Fig. 18.7). To mitigate spatial uncertainties associated with δSource, where possible, one can:
-
Select sites where trees have predictable sources with relatively stable isotope ratios (e.g., obligate phreatophytes), and measure their xylem water isotopic values.
-
Select sites where precipitation, xylem and/or soil water isotopic data are available or can be measured to estimate how δSource relates to δPrecip.
-
Select sites with similar climates, soils, and species to where past studies have estimated the relationship of δSource to mean δPrecip.
-
Search online isotopic databases for δPrecip data and streamwater isotope time series (i.e. IAEA Global Network of Isotopes in Rivers, GNIR, Water Isotope Database wiDB, waterisotopes.org, etc.). These data can be used to estimate the mean isotope ratio of evapotranspired waters (see Eq. 21 in Kirchner and Allen, 2020).
-
If there is no way to estimate or constrain how δSource differs from mean annual δPrecip, then δSource can be assumed to have uncertainties of magnitudes reflecting the seasonal amplitude in δPrecip (Fig. 18.3).
Assuming δSource is always equal to long-term mean δPrecip can confound interpretations of species or individual-tree differences in Δcellulose, even at the same site. Many species and individuals use different water pools in the subsurface (e.g., due to differences in rooting depths). To mitigate inter species uncertainties, where possible, one can:
-
Select sites where all species use groundwater or soil waters δ18O is relatively homogenous (which should involve measuring xylem water δ18O to document the similarity between species).
-
Measure xylem water δ18O over a growing season to benchmark expected differences between δSource and δPrecip for each species. This assumes that measurements of δPrecip are available or are also measured.
-
Select sites where rooting depth information is available for the species of interest. Insights into the relative rooting depths can be used to understand how δSource may differ in time for different trees at the same site (e.g., a tree that uses shallow water exclusively may experience more inter-annual variation in δSource than does one that uses deeper soil water with less variation in δ18O).
-
Assume that trees within a single site vary substantially, and the variation is not always attributable to species. The mean within-plot range was 1.8 ‰ for the xylem-water samples in Fig. 18.7. Without some information on δSource or rooting depth, it may be impossible to interpret species or individual differences in Δcellulose.
5.2 Inferring the Source of Water from Tree Rings Isotope Ratios
Another application of tree-ring δ18O ratios is identifying the water sources used by trees. Often questions pertain to inferring changes in sources of water to trees (e.g., by depth, or between soil water, stream water and groundwater), across species, climates, landscapes, or time (e.g., (Sargeant et al. 2020; Miller et al. 2006; Sarris et al. 2013; Saurer et al. 2016; Sargeant and Singer 2016). All of these questions require first accounting for the fractionation undergone from source-water to cellulose, a sequence known to vary with meteorological conditions, species, and canopy position (this is the subject of Chaps. 10, 11 and 13); it should also be noted that constraining the leaf-atmosphere exchange fractionation sequence described in Chaps. 10, 11, and 13 also assumes knowledge of the isotopic ratio of ambient atmospheric water vapor, which can be out of equilibrium with local water sources (Fiorella et al. 2019).
After accounting for leaf-level fractionation effects, this application assumes that residual variations in cellulose δ18O reflect variations in δSource. Indeed δsource can be substantially different between pools: shallow soil waters are often isotopically heavier (than deeper soil water, river water, or groundwater), because they are influenced more by evaporative fractionation, and they are also more likely to be sourced from recent growing season precipitation that is isotopically heavier than winter precipitation in most extra-tropical regions; Fig. 18.3). Alternatively, deeper soils, groundwater, and streamwater tend to be isotopically lighter than shallow soil water because they recharge more in colder seasons or often from higher elevations (Sect. 18.3). However, interpretations of source waters from cellulose need to consider the following possible confounding factors.
First, it needs to be recognized that predicting source water isotope ratios from cellulose requires estimating leaf-water evaporative enrichment and fractionation processes that occur with sugar assimilation. These processes can be uncertain and their effect size can be similar or greater in magnitude than the effects of δSource variation (Song et al. 2014). As a starting point to constrain those processes, inter-annual variations in temperature and relative humidity are key data, which can be applied in models to constrain expected inter-annual variability attributable to leaf fractionation processes (Sargeant et al. 2020; Cernusak et al. 2016). These processes (described in Chap. 11–13) must be constrained prior to estimating source waters from cellulose.
Second, inter-annual variations within a water source can confound the ability to separate between the use of different water sources over time. For example, source differentiation will be confounded if (a) the range of isotope ratios in those sources vary among years (e.g., shallow soils are isotopically lighter in years with less summer precipitation because they are not diluted by summer rain’s higher δPrecip), (b) values of δPrecip are anomalously high or low in a given year, (c) the growing seasons and thus water uptake seasons differed among years such that the rings reflect larger proportions of uptake during either spring (light, less evaporated) or late summer (heavy, more evaporated). To mitigate uncertainties in interpreting source water signals, where possible, one can:
-
Select sites where seasonal and inter-annual isotope variations are smaller, such as lower elevation or more coastal regions, but where the potential sources have large isotopic differences. If the goal is to identify inter-annual differences in use of groundwater relative to soil water, select sites where groundwater is sourced by rain from much higher elevations so that its influence can be clearly distinguished (e.g., Oerter et al. 2019).
-
To account for inter-annual cellulose isotopic variations attributable to inter-annual variations in δPrecip, select regions where inter-annual δPrecip was measured, or estimate likely ranges of inter-annual δPrecip to see whether they could explain tree-ring variations.
-
To rule out the possibility that inter-annual isotope signatures change because the mixtures of seasonal precipitation in soils, groundwater, or streams are changing, examine inter-seasonal precipitation amount variations (and δPrecip seasonal amplitude) to see whether their effects on weighted-mean annual δPrecip are large enough to explain inter-annual variations in ring isotope ratios (e.g., during the calibration period when data are available). These data are more widely available than records of inter-annual variation in δPrecip.
-
To rule out the influence of variations in the radial growth season, use dendrometer records of radial growth phenology to indicate how growing season varies; this issue may not matter if intra-annual (e.g., early-wood versus late-wood) isotope variations are small anyway.
Third, confounding factors in inferring site-and-species differences in source can arise, for example, when (a) comparing across soils or landscapes where water turnover times differ due to storage capacity or climate (e.g., a soil in a very wet region might hold recent precipitation whereas one in a dry site may contain several previous years of precipitation) (b) comparing across soils where infiltration processes differ and include substantial bypass flow (e.g., allowing more recent precipitation to show up lower in soil profiles or in stream or groundwater sources), or (c) species differ in growth and water-uptake seasons such that isotopic differences might reflect trees taking up water from the same pools but at different times. To mitigate uncertainties, where possible, one can:
-
Select trees that co-occupy sites (or at least occupying sites with similar climates and soils) to rule out the effects of soils differences in species comparisons.
-
Use multi-seasonal xylem water measurements to show that potential sources across sites similarly respond to precipitation inputs and seasonal climate variations, to rule out the possibility that tree-ring isotope variations are due to soil and δPrecip dynamics, rather than changes in sources used by trees.
And last, confounding factors in inferring intra-annual variations (i.e., from sub-annual slices of cellulose) in sources can arise, for example when (a) water that was enriched due to evaporation from stems over winter (Treydte et al. 2014) is incorporated in early radial growth, (b) sources change isotopically throughout the growing season, confounding estimates of the relative use of each source, (e.g., rain dilutes evaporatively enriched soils resulting in profiles that are inverse to the more typical ones seen in growing seasons; Figs. 18.5 and 18.6). To mitigate uncertainties, where possible, one can:
-
Interpret the earliest early-wood isotope values cautiously to avoid misinterpreting the effects of evaporative enrichment throughout the dormant seasons or of stored carbohydrates that contain source information from previous years.
-
Unless the potential end-members are consistently very distinct, use a calibrated physical model to account for the source-water dynamics and interactions; these finer scale dynamics should be considered when making finer-resolution inferences from sub-annual isotopic variations.
6 Conclusions
Geographic, vertical, and temporal variations in the isotopic composition of potential water sources to plants should be considered when interpreting tree-ring δ18O or δ2H. While tree rings provide opportunity for studying these variations, there are many potential confounding factors. When interpreting tree-ring isotope ratios, the relative influences of source water variations versus climate and physiology effects on leaf-water enrichment (Chaps. 10 and 11) should both be considered; there are only few circumstances where spatiotemporal variability in source water isotopic composition can be ignored. There are many reasons why one ring, one tree, or one site may have more enriched δ18O or δ2H than another, and these could result in confounded interpretations; Table 18.1 demonstrates this challenge by offering a list of hypothetical explanations for heavier-than-normal δ18O in a ring. Nonetheless, by recognizing the full suite of controlling processes (Sects. 18.2, 18.3, and 18.4), many potential confounding factors can be eliminated when interpreting data (Sect. 18.5). Accounting for source-water isotope variations involves first examining the average and seasonal precipitation inputs to a site (Figs. 18.3 and 18.4), then considering how fast soil–water pools may turnover and how evaporation may affect their isotope ratios (Figs. 18.5, 18.6, 18.7), and last, considering which soil-waters will be taken up by trees (Figs. 18.7 and 18.8). Fortunately, soil–water-transport and plant-uptake dynamics both tend to correspond with climate, allowing source water to be partially predictable and interpretable. Understanding these processes and moving beyond assuming that source-waters isotopic compositions are constant or equal to that of long-term mean precipitation is an important step towards improved interpretation of tree-ring isotope ratios.
References
Aemisegger F, Spiegel J, Pfahl S et al (2015) Isotope meteorology of cold front passages: a case study combining observations and modeling. Geophys Res Lett 42:5652–5660
Aggarwal PK, Araguas-Araguas L, Groening M et al (2010) Global hydrological isotope data and data networks. In: West JB, Bowen GJ, Dawson TE, Tu KP (eds) Isoscapes: understanding movement, pattern, and process on earth through isotope mapping, pp 33–50
Aggarwal PK, Froehlich K, Gonfiantini R (2011) Contributions of the International Atomic Energy Agency to the development and practice of isotope hydrology. Hydrogeol J 19:5–8. https://doi.org/10.1007/s10040-010-0648-3
Allen ST, Jasechko S, Berghuijs WR, et al (2019a) Global sinusoidal seasonality in precipitation isotopes. Hydrol Earth Syst Sci Discuss, 1–23. https://doi.org/10.5194/hess-2019-61
Allen ST, Kirchner JW, Braun S et al (2019b) Seasonal origins of soil water used by trees. Hydrol Earth Syst Sci 23:1199–1210. https://doi.org/10.5194/hess-23-1199-2019
Allen ST, Keim RF, Barnard HR et al (2017) The role of stable isotopes in understanding rainfall interception processes: a review. WIREs Water 4:n/a-n/a. https://doi.org/10.1002/wat2.1187
Anderson WT, Bernasconi SM, McKenzie JA et al (2002) Model evaluation for reconstructing the oxygen isotopic composition in precipitation from tree ring cellulose over the last century. Chem Geol 182:121–137. https://doi.org/10.1016/S0009-2541(01)00285-6
Anderson WT, Bernasconi SM, McKenzie JA, Saurer M (1998) Oxygen and carbon isotopic record of climatic variability in tree ring cellulose (Picea abies): an example from central Switzerland (1913–1995). J Geophys Res Atmos 103:31625–31636. https://doi.org/10.1029/1998JD200040
Barbecot F, Guillon S, Pili E, et al (2018) Using water stable isotopes in the unsaturated zone to quantify recharge in two contrasted infiltration regimes. Vadose Zone J 17:170170. https://doi.org/10.2136/vzj2017.09.0170
Barbeta A, Gimeno TE, Clavé L et al (2020) An explanation for the isotopic offset between soil and stem water in a temperate tree species. New Phytol. https://doi.org/10.1111/nph.16564
Benettin P, Queloz P, Bensimon M et al (2019) Velocities, residence times, tracer breakthroughs in a vegetated Lysimeter: a multitracer experiment. Water Resour Res 55:21–33. https://doi.org/10.1029/2018WR023894
Benettin P, Volkmann THM, von Freyberg J et al (2018) Effects of climatic seasonality on the isotopic composition of evaporating soil waters. Hydrol Earth Syst Sci 22:2881–2890. https://doi.org/10.5194/hess-22-2881-2018
Berghuijs WR, Allen ST (2019) Waters flowing out of systems are younger than the waters stored in those same systems. Hydrol Process. https://doi.org/10.1002/hyp.13569
Berkelhammer M, Still CJ, Ritter F et al (2020) Persistence and plasticity in conifer water-use strategies. J Geophys Res Biogeosci 125:e2018JG004845. https://doi.org/10.1029/2018JG004845
Berkelhammer MB, Stott LD (2008) Recent and dramatic changes in Pacific storm trajectories recorded in δ18O from Bristlecone Pine tree ring cellulose. Geochem Geophys Geosyst 9. https://doi.org/10.1029/2007GC001803
Berry ZC, Hughes NM, Smith WK (2014) Cloud immersion: an important water source for spruce and fir saplings in the southern Appalachian Mountains. Oecologia 174:319–326. https://doi.org/10.1007/s00442-013-2770-0
Bertrand G, Masini J, Goldscheider N et al (2014) Determination of spatiotemporal variability of tree water uptake using stable isotopes (δ18O, δ2H) in an alluvial system supplied by a high-altitude watershed, Pfyn forest, Switzerland. Ecohydrology 7:319–333. https://doi.org/10.1002/eco.1347
Beven K, Germann P (1982) Macropores and water flow in soils. Water Resour Res 18:1311–1325. https://doi.org/10.1029/WR018i005p01311
Bowen GJ (2010) Statistical and geostatistical mapping of precipitation water isotope ratios. Isoscapes. Springer, Dordrecht, pp 139–160
Bowen GJ (2008) Spatial analysis of the intra-annual variation of precipitation isotope ratios and its climatological corollaries. J Geophys Res 113:D05113. https://doi.org/10.1029/2007JD009295
Bowen GJ, Cai Z, Fiorella RP, Putman AL (2019) Isotopes in the water cycle: regional- to global-scale patterns and applications. Annu Rev Earth Planet Sci 47:453–479
Bowen GJ, Liu Z, Vander Zanden HB et al (2014) Geographic assignment with stable isotopes in IsoMAP. Methods Ecol Evol 5:201–206. https://doi.org/10.1111/2041-210X.12147
Bowen GJ, Putman A, Brooks JR et al (2018) Inferring the source of evaporated waters using stable H and O isotopes. Oecologia, 1–15. https://doi.org/10.1007/s00442-018-4192-5
Braud I, Bariac T, Gaudet JP, Vauclin M (2005) SiSPAT-Isotope, a coupled heat, water and stable isotope (HDO and H218O) transport model for bare soil. Part I. Model description and first verifications. J Hydrol 309:277–300. https://doi.org/10.1016/j.jhydrol.2004.12.013
Brooks JR, Barnard HR, Coulombe R, McDonnell JJ (2010) Ecohydrologic separation of water between trees and streams in a Mediterranean climate. Nat Geosci 3:100–104. https://doi.org/10.1038/ngeo722
Brooks JR, Meinzer FC, Coulombe R, Gregg J (2002) Hydraulic redistribution of soil water during summer drought in two contrasting Pacific Northwest coniferous forests. Tree Physiol 22:1107–1117. https://doi.org/10.1093/treephys/22.15-16.1107
Brooks JR, Meinzer FC, Warren JM et al (2006) Hydraulic redistribution in a Douglas-fir forest: lessons from system manipulations. Plant, Cell Environ 29:138–150. https://doi.org/10.1111/j.1365-3040.2005.01409.x
Burgess SSO, Pate JS, Adams MA, Dawson TE (2000) Seasonal water acquisition and redistribution in the Australian Woody Phreatophyte, Banksia prionotes. Ann Bot 85:215–224. https://doi.org/10.1006/anbo.1999.1019
Caldwell MM, Dawson TE, Richards JH (1998) Hydraulic lift: consequences of water efflux from the roots of plants. Oecologia 113:151–161. https://doi.org/10.1007/s004420050363
Caldwell MM, Richards JH (1989) Hydraulic lift: water efflux from upper roots improves effectiveness of water uptake by deep roots. Oecologia 79:1–5. https://doi.org/10.1007/BF00378231
Canadell J, Jackson RB, Ehleringer JB et al (1996) Maximum rooting depth of vegetation types at the global scale. Oecologia 108:583–595. https://doi.org/10.1007/BF00329030
Carminati A, Moradi AB, Vetterlein D et al (2010) Dynamics of soil water content in the rhizosphere. Plant Soil 332:163–176. https://doi.org/10.1007/s11104-010-0283-8
Cernusak LA, Barbour MM, Arndt SK et al (2016) Stable isotopes in leaf water of terrestrial plants. Plant Cell Environ 39:1087–1102. https://doi.org/10.1111/pce.12703
Chimner RA, Cooper DJ (2004) Using stable oxygen isotopes to quantify the water source used for transpiration by native shrubs in the San Luis Valley, Colorado U.S.A. Plant Soil 260:225–236
Clark ID, Fritz P (1997) Environmental isotopes in hydrogeology. CRC Press, Boca Raton
Coplen TB, Neiman PJ, White AB et al (2008) Extreme changes in stable hydrogen isotopes and precipitation characteristics in a landfalling Pacific storm. Geophys Res Lett 35:L21808. https://doi.org/10.1029/2008GL035481
Coplen TB, Neiman PJ, White AB, Ralph FM (2015) Categorisation of northern California rainfall for periods with and without a radar brightband using stable isotopes and a novel automated precipitation collector. Tellus B Chem Phys Meteorol 67:28574. https://doi.org/10.3402/tellusb.v67.28574
Craig H (1961) Isotopic Variations in meteoric waters. Science 133:1702–1703. https://doi.org/10.1126/science.133.3465.1702
Craig H, Gordon LI (1965) Deuterium and oxygen-18 variations in the ocean and the marine atmosphere. In: Tongiorgi E (ed) Proceedings of a conference on stable isotopes in oceanographic studies and paleotemperatures, Spoleto, Italy
Danis PA, Masson-Delmotte V, Stievenard M et al (2006) Reconstruction of past precipitation δ18O using tree-ring cellulose δ18O and δ13C: A calibration study near Lac d’Annecy, France. Earth Planet Sci Lett 243:439–448. https://doi.org/10.1016/j.epsl.2006.01.023
Dansgaard W (1953) The abundance of O18 in atmospheric water and water vapour. Tellus 5:461–469
Dansgaard W (1954) The O18-abundance in fresh water. Geochim Cosmochim Acta 6:241–260
Dansgaard W (1964) Stable isotopes in precipitation. Tellus 16:436–468. https://doi.org/10.1111/j.2153-3490.1964.tb00181.x
Dawson TE, Ehleringer JR (1991) Streamside trees that do not use stream water. Nature 350:335–337
Dawson TE, Ehleringer JR (1993) Isotopic enrichment of water in the “woody” tissues of plants: Implications for plant water source, water uptake, and other studies which use the stable isotopic composition of cellulose. Geochim Cosmochim Acta 57:3487–3492. https://doi.org/10.1016/0016-7037(93)90554-A
Dee S, Noone D, Buenning N et al (2015) SPEEDY-IER: A fast atmospheric GCM with water isotope physics. J Geophys Res Atmos 120:73–91. https://doi.org/10.1002/2014JD022194
Eggemeyer KD, Awada T, Harvey FE et al (2009) Seasonal changes in depth of water uptake for encroaching trees Juniperus virginiana and Pinus ponderosa and two dominant C4 grasses in a semiarid grassland. Tree Physiol 29:157–169. https://doi.org/10.1093/treephys/tpn019
Ehleringer JR, Phillips SL, Schuster WSF, Sandquist DR (1991) Differential utilization of summer rains by desert plants. Oecologia 88:430–434. https://doi.org/10.1007/BF00317589
Ellsworth PZ, Williams DG (2007) Hydrogen isotope fractionation during water uptake by woody xerophytes. Plant Soil 291:93–107. https://doi.org/10.1007/s11104-006-9177-1
Evans MN (2007) Toward forward modeling for paleoclimatic proxy signal calibration: a case study with oxygen isotopic composition of tropical woods. Geochem Geophys Geosyst, 8. https://doi.org/10.1029/2006GC001406
Fan Y, Miguez-Macho G, Jobbágy EG et al (2017) Hydrologic regulation of plant rooting depth. PNAS 114:10572–10577. https://doi.org/10.1073/pnas.1712381114
Feng X, Faiia AM, Posmentier ES (2009) Seasonality of isotopes in precipitation: a global perspective. J Geophys Res Atmosp 114. https://doi.org/10.1029/2008JD011279
Fiorella RP, West JB, Bowen GJ (2019) Biased estimates of the isotope ratios of steady-state evaporation from the assumption of equilibrium between vapour and precipitation. Hydrol Process 33:2576–2590. https://doi.org/10.1002/hyp.13531
Fischer BMC, van Meerveld HJ (Ilja), Seibert J (2017) Spatial variability in the isotopic composition of rainfall in a small headwater catchment and its effect on hydrograph separation. J Hydrol 547:755–769. https://doi.org/10.1016/j.jhydrol.2017.01.045
Flury M, Flühler H, Jury WA, Leuenberger J (1994) Susceptibility of soils to preferential flow of water: a field study. Water Resour Res 30:1945–1954. https://doi.org/10.1029/94WR00871
Gale MR, Grigal DF (1987) Vertical root distributions of northern tree species in relation to successional status. Can J for Res 17:829–834. https://doi.org/10.1139/x87-131
Gat JR (1996) Oxygen and hydrogen isotopes in the hydrologic cycle. Annu Rev Earth Planet Sci 24:225–262. https://doi.org/10.1146/annurev.earth.24.1.225
Gat JR, Gonfiantini R (1981) Stable isotope hydrology. Deuterium and oxygen-18 in the water cycle
Gat JR, Tzur Y (1968) Modification of the isotopic composition of rainwater by processes which occur before groundwater recharge. In: Isotope hydrology, proceedings of symposium on Vienna 1966. International Atomic Energy Agency, pp 49–60
Gedzelman SD, Lawrence J (1990) The isotopic composition of precipitation from two extratropical cyclones. Mon Weather Rev 118:495–509
Gedzelman SD, Lawrence J, Gamache J et al (2003) Probing hurricanes with stable isotopes of rain and water vapor. Mon Weather Rev 131:1112–1127
Gerlein-Safdi C, Gauthier PPG, Caylor KK (2018) Dew-induced transpiration suppression impacts the water and isotope balances of Colocasia leaves. Oecologia 187:1041–1051. https://doi.org/10.1007/s00442-018-4199-y
Goldsmith GR, Allen ST, Braun S et al (2019) Spatial variation in throughfall, soil, and plant water isotopes in a temperate forest. Ecohydrology 12:e2059. https://doi.org/10.1002/eco.2059
Goldsmith GR, Lehmann MM, Cernusak LA et al (2017) Inferring foliar water uptake using stable isotopes of water. Oecologia 184:763–766. https://doi.org/10.1007/s00442-017-3917-1
Goldsmith GR, Matzke NJ, Dawson TE (2013) The incidence and implications of clouds for cloud forest plant water relations. Ecol Lett 16:307–314. https://doi.org/10.1111/ele.12039
Goldsmith GR, Muñoz-Villers LE, Holwerda F et al (2012) Stable isotopes reveal linkages among ecohydrological processes in a seasonally dry tropical montane cloud forest. Ecohydrology 5:779–790. https://doi.org/10.1002/eco.268
Good SP, Mallia DV, Denis EH et al (2014a) High frequency trends in the isotopic composition of Superstorm Sandy. In: Bennington JB, Farmer EC (eds) Learning from the impacts of Superstorm Sandy. Academic Press, pp 41–55
Good SP, Mallia DV, Lin JC, Bowen GJ (2014b) Stable isotope analysis of precipitation samples obtained via crowdsourcing reveals the spatiotemporal evolution of Superstorm Sandy. PLOS ONE 9:e91117. https://doi.org/10.1371/journal.pone.0091117
Gotsch SG, Asbjornsen H, Holwerda F et al (2014) Foggy days and dry nights determine crown-level water balance in a seasonal tropical montane cloud forest. Plant Cell Environ 37:261–272. https://doi.org/10.1111/pce.12151
Groh J, Stumpp C, Lücke A et al (2018) Inverse estimation of soil hydraulic and transport parameters of layered soils from water stable isotope and lysimeter data. Vadose Zone J 17. https://doi.org/10.2136/vzj2017.09.0168
Guo JS, Hungate BA, Kolb TE, Koch GW (2018) Water source niche overlap increases with site moisture availability in woody perennials. Plant Ecol 219:719–735. https://doi.org/10.1007/s11258-018-0829-z
Haverd V, Cuntz M (2010) Soil–Litter–Iso: a one-dimensional model for coupled transport of heat, water and stable isotopes in soil with a litter layer and root extraction. J Hydrol 388:438–455. https://doi.org/10.1016/j.jhydrol.2010.05.029
Helliker BR, Richter SL (2008) Subtropical to boreal convergence of tree-leaf temperatures. Nature 454:511–514. https://doi.org/10.1038/nature07031
Hewlett JD, Hibbert AR (1967) Factors affecting the response of small watersheds to precipitation in humid areas. In: Sopper W, Lull HW (eds) Forest hydrology. Pergamon Press, New York, NY, USA, pp 275–290
Horita J, Rozanski K, Cohen S (2008) Isotope effects in the evaporation of water: a status report of the Craig-Gordon model. Isot Environ Health Stud 44:23–49. https://doi.org/10.1080/10256010801887174
Hultine KR, Cable WL, Burgess SSO, Williams DG (2003a) Hydraulic redistribution by deep roots of a Chihuahuan Desert phreatophyte. Tree Physiol 23:353–360. https://doi.org/10.1093/treephys/23.5.353
Hultine KR, Williams DG, Burgess SSO, Keefer TO (2003b) Contrasting patterns of hydraulic redistribution in three desert phreatophytes. Oecologia 135:167–175. https://doi.org/10.1007/s00442-002-1165-4
IAEA/WMO (2020) Global network of isotopes in precipitation. The GNIP Database. http://www.iaea.org/water. Vienna, Austria
Jackson RB, Canadell J, Ehleringer JR et al (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108:389–411. https://doi.org/10.1007/BF00333714
Jasechko S (2019) Global isotope hydrogeology—review. Rev Geophys. https://doi.org/10.1029/2018RG000627
Jasechko S, Perrone D, Befus KM et al (2017) Global aquifers dominated by fossil groundwaters but wells vulnerable to modern contamination. Nat Geosci 10:425
Jasechko S, Taylor RG (2015) Intensive rainfall recharges tropical groundwaters. Environ Res Lett 10:124015. https://doi.org/10.1088/1748-9326/10/12/124015
Jouzel J, Koster RD, Suozzo RJ, Russell GL (1994) Stable water isotope behavior during the last glacial maximum: a general circulation model analysis. J Geophys Res 99:25791–25801
Kendall C, Caldwell E (1998) Fundamentals of isotope geochemistry. In: Isotope tracers in catchment hydrology (Developments in water science). Elsevier Science, pp 51–86
Kennedy CD, Bataille CP, Liu Z et al (2012) Dynamics of nitrate and chloride during storm events in agricultural catchments with different subsurface drainage intensity (Indiana, USA). J Hydrol 466–467:1–10. https://doi.org/10.1016/j.hydrol.2012.05.002
Kirchner JW, Allen ST (2020) Seasonal partitioning of precipitation between streamflow and evapotranspiration, inferred from end-member splitting analysis. Hydrol Earth Syst Sci 24:17–39. https://doi.org/10.5194/hess-24-17-2020
Konecky B, Dee SG, Noone DC (2019) WaxPSM: a forward model of leaf wax hydrogen isotope ratios to bridge proxy and model estimates of past climate. J Geophys Res Biogeosci 124:2107–2125. https://doi.org/10.1029/2018JG004708
Kurz-Besson C, Otieno D, Lobo do Vale R, et al (2006) Hydraulic lift in cork oak trees in a savannah-type Mediterranean ecosystem and its contribution to the local water balance. Plant Soil 282:361–378. https://doi.org/10.1007/s11104-006-0005-4
Landwehr JM, Coplen TB (2006) Line-conditioned excess: a new method for characterizing stable hydrogen and oxygen isotope ratios in hydrologic systems
Lawrence, DM, Oleson KW, Flanner MG et al (2011) Parameterization improvements and functional and structural advances in Version 4 of the Community Land Model. J Adv Model Earth Syst 3. https://doi.org/10.1029/2011MS00045
Lawrence J, Gedzelman SD (1996) Low stable isotope ratios of tropical cyclone rains. Geophys Res Lett 23:527–530
Lehmann M, Goldsmith G, Schmid L et al (2017) The effect of 18O-labelled water vapour on the oxygen isotope ratio of water and assimilates in plants at high humidity. New Phytol 217:105–116. https://doi.org/10.1111/nph.14788
Lin G, Sternberg L (1993) 31—hydrogen isotopic fractionation by plant roots during water uptake in coastal wetland plants. In: Ehleringer JR, Hall AE, Farquhar GD (eds) Stable isotopes and plant carbon-water relations. Academic Press, San Diego, pp 497–510
Lin W, Domec J-C, Ward EJ et al (2019) Using δ13C and δ18O to analyze loblolly pine (Pinus taeda L.) response to experimental drought and fertilization. Tree Physiol 39:1984–1994. https://doi.org/10.1093/treephys/tpz096
Liu Z, Bowen GJ, Welker JM (2010) Atmospheric circulation is reflected in precipitation isotope gradients over the conterminous United States. J Geophys Res 115:D22120. https://doi.org/10.1029/2010JD014175
Martin J, Looker N, Hoylman Z et al (2018) Differential use of winter precipitation by upper and lower elevation Douglas fir in the Northern Rockies. Glob Change Biol 24:5607–5621. https://doi.org/10.1111/gcb.14435
McCully ME (1999) Root xylem embolisms and refilling. relation to water potentials of soil, roots, and leaves, and osmotic potentials of root xylem sap. Plant Physiol 119:1001–1008. https://doi.org/10.1104/pp.119.3.1001
Meinzer FC, Warren JM, Brooks JR (2007) Species-specific partitioning of soil water resources in an old-growth Douglas-fir–western hemlock forest. Tree Physiol 27:871–880. https://doi.org/10.1093/treephys/27.6.871
Miller DL, Mora CI, Grissino-Mayer HD et al (2006) Tree-ring isotope records of tropical cyclone activity. PNAS 103:14294–14297. https://doi.org/10.1073/pnas.0606549103
Mueller MH, Alaoui A, Kuells C et al (2014) Tracking water pathways in steep hillslopes by δ18O depth profiles of soil water. J Hydrol 519:340–352. https://doi.org/10.1016/j.jhydrol.2014.07.031
Munksgaard NC, Wurster CM, Bass A, Bird MI (2012) Extreme short-term stable isotope variability revealed by continuous rainwater analysis. Hydrol Process 26:3630–3634. https://doi.org/10.1002/hyp.9505
Neumann RB, Cardon ZG (2012) The magnitude of hydraulic redistribution by plant roots: a review and synthesis of empirical and modeling studies. New Phytol 194:337–352. https://doi.org/10.1111/j.1469-8137.2012.04088.x
Nimmo JR (2011) Preferential flow occurs in unsaturated conditions. Hydrol Process 26:786–789. https://doi.org/10.1002/hyp.8380
Oerter EJ, Siebert G, Bowling DR, Bowen G (2019) Soil water vapour isotopes identify missing water source for streamside trees. Ecohydrology 12:e2083. https://doi.org/10.1002/eco.2083
Oshun J, Dietrich WE, Dawson TE, Fung I (2016) Dynamic, structured heterogeneity of water isotopes inside hillslopes. Water Resour Res 52:164–189. https://doi.org/10.1002/2015WR017485
Penna D, Hopp L, Scandellari F et al (2018) Tracing ecosystem water fluxes using hydrogen and oxygen stable isotopes: challenges and opportunities from an interdisciplinary perspective. Biogeosciences 15:6399–6415. https://doi.org/10.5194/bg-2018-286
Penna D, Meerveld HJ (Ilja) van (2019) Spatial variability in the isotopic composition of water in small catchments and its effect on hydrograph separation. Wiley Interdiscip Rev Water 6:e1367. https://doi.org/10.1002/wat2.1367
Pfahl S, Wernli H, Yoshimura K (2012) The isotopic composition of precipitation from a winter storm—a case study with the limited-area model COSMOiso. Atmos Chem Phys 12:1629–1648
Poage MA, Chamberlain CP (2001) Empirical relationships between elevation and the stable isotope composition of precipitation and surface waters: considerations for studies of paleoelevation change. Am J Sci 301:1–15
Price RM, Swart PK, Willoughby HE (2008) Seasonal and spatial variation in the stable isotopic composition (δ18O and δD) of precipitation in south Florida. J Hydrol 358:193–205. https://doi.org/10.1016/j.jhydrol.2008.06.003
Putman AL, Bowen GJ (2019) Technical note: a global database of the stable isotopic ratios of meteoric and terrestrial waters. Hydrol Earth Syst Sci 23:4389–4396. https://doi.org/10.5194/hess-23-4389-2019
Putman AL, Fiorella RP, Bowen GJ, Cai Z (2019) A global perspective on local meteoric water lines: meta-analytic insight into fundamental controls and practical constraints. Water Resour Res 55:6896–6910. https://doi.org/10.1029/2019WR025181
Roden J, Siegwolf R (2012) Is the dual-isotope conceptual model fully operational? Tree Physiol 32:1179–1182. https://doi.org/10.1093/treephys/tps099
Rong L, Chen X, Chen X et al (2011) Isotopic analysis of water sources of mountainous plant uptake in a karst plateau of southwest China. Hydrol Process 25:3666–3675. https://doi.org/10.1002/hyp.8093
Ryel RJ (2004) Hydraulic redistribution. In: Esser K, Lüttge U, Beyschlag W, Murata J (eds) Progress in botany: genetics physiology systematics ecology. Springer, Berlin, Heidelberg, pp 413–435
Sargeant CI, Singer MB (2016) Sub-annual variability in historical water source use by Mediterranean riparian trees. Ecohydrol 9:1328–1345. https://doi.org/10.1002/eco.1730
Sargeant CI, Singer MB, Vallet‐Coulomb C (2020) Identification of source-water oxygen isotopes in trees toolkit (ISO-Tool) for deciphering historical water use by forest trees. Water Resour Res n/a. https://doi.org/10.1029/2018WR024519
Sarris D, Siegwolf R, Körner C (2013) Inter- and intra-annual stable carbon and oxygen isotope signals in response to drought in Mediterranean pines. Agric for Meteorol 168:59–68. https://doi.org/10.1016/j.agrformet.2012.08.007
Saurer M, Kirdyanov AV, Prokushkin AS et al (2016) The impact of an inverse climate–isotope relationship in soil water on the oxygen-isotope composition of Larix gmelinii in Siberia. New Phytol 209:955–964. https://doi.org/10.1111/nph.13759
Schwinning S (2010) The ecohydrology of roots in rocks. Ecohydrology 3:238–245. https://doi.org/10.1002/eco.134
Selker JS, McCord JT, Keller CK (1999) Vadose zone processes. CRC Press, Boca Raton
Simonin KA, Santiago LS, Dawson TE (2009) Fog interception by Sequoia sempervirens (D. Don) crowns decouples physiology from soil water deficit. Plant Cell Environ 32:882–892. https://doi.org/10.1111/j.1365-3040.2009.01967.x
Snyder KA, Williams DG (2000) Water sources used by riparian trees varies among stream types on the San Pedro River, Arizona. Agric for Meteorol 105:227–240. https://doi.org/10.1016/S0168-1923(00)00193-3
Song X, Farquhar GD, Gessler A, Barbour MM (2014) Turnover time of the non-structural carbohydrate pool influences δ18O of leaf cellulose. Plant Cell Environ 37:2500–2507. https://doi.org/10.1111/pce.12309
Sprenger M, Erhardt M, Riedel M, Weiler M (2016a) Historical tracking of nitrate in contrasting vineyards using water isotopes and nitrate depth profiles. Agr Ecosyst Environ 222:185–192. https://doi.org/10.1016/j.agee.2016.02.014
Sprenger M, Leistert H, Gimbel K, Weiler M (2016b) Illuminating hydrological processes at the soil-vegetation-atmosphere interface with water stable isotopes. Rev Geophys 54:2015RG000515. https://doi.org/10.1002/2015RG000515
Sprenger M, Seeger S, Blume T, Weiler M (2016c) Travel times in the vadose zone: variability in space and time. Water Resour Res 52:5727–5754. https://doi.org/10.1002/2015WR018077
Sprenger M, Llorens P, Cayuela C et al (2019) Mechanisms of consistently disjunct soil water pools over (pore) space and time. Hydrol Earth Syst Sci 23:2751–2762. https://doi.org/10.5194/hess-23-2751-2019
Sprenger M, Tetzlaff D, Buttle J et al (2018a) Measuring and modeling stable isotopes of mobile and bulk soil water. Vadose Zone J 17. https://doi.org/10.2136/vzj2017.08.0149
Sprenger M, Tetzlaff D, Buttle J et al (2018b) Storage, mixing, and fluxes of water in the critical zone across northern environments inferred by stable isotopes of soil water. Hydrol Process 32:1720–1737. https://doi.org/10.1002/hyp.13135
Sprenger M, Volkmann THM, Blume T, Weiler M (2015) Estimating flow and transport parameters in the unsaturated zone with pore water stable isotopes. Hydrol Earth Syst Sci 19:2617–2635. https://doi.org/10.5194/hess-19-2617-2015
Stumpp C, Stichler W, Kandolf M, Šimůnek J (2012) Effects of land cover and fertilization method on water flow and solute transport in five lysimeters: a long-term study using stable water isotopes. Vadose Zone J 11:0–0. https://doi.org/10.2136/vzj2011.0075
Swidrak I, Schuster R, Oberhuber W (2013) Comparing growth phenology of co-occurring deciduous and evergreen conifers exposed to drought. Flora Morphol Distrib Funct Ecol Plants 208:609–617. https://doi.org/10.1016/j.flora.2013.09.004
Tang K, Feng X (2001) The effect of soil hydrology on the oxygen and hydrogen isotopic compositions of plants’ source water. Earth Planet Sci Lett 185:355–367. https://doi.org/10.1016/S0012-821X(00)00385-X
Thomas EM, Lin H, Duffy CJ et al (2013) Spatiotemporal patterns of water stable isotope compositions at the shale hills critical zone observatory: linkages to subsurface hydrologic processes. Vadose Zone J 12. https://doi.org/10.2136/vzj2013.01.0029
Treydte K, Boda S, Graf Pannatier E et al (2014) Seasonal transfer of oxygen isotopes from precipitation and soil to the tree ring: source water versus needle water enrichment. New Phytol 202:772–783. https://doi.org/10.1111/nph.12741
Ulrich DEM, Still C, Brooks JR et al (2019) Investigating old-growth ponderosa pine physiology using tree-rings, δ13C, δ18O, and a process-based model. Ecology 100:e02656. https://doi.org/10.1002/ecy.2656
van Schaik L, Palm J, Klaus J et al (2014) Linking spatial earthworm distribution to macropore numbers and hydrological effectiveness. Ecohydrology 7:401–408. https://doi.org/10.1002/eco.1358
Vargas AI, Schaffer B, Yuhong L, Sternberg LDSL (2017) Testing plant use of mobile vs immobile soil water sources using stable isotope experiments. New Phytol 215:582–594. https://doi.org/10.1111/nph.14616
Voelker SL, Brooks JR, Meinzer FC et al (2014) Reconstructing relative humidity from plant delta 18O and delta D as deuterium deviations from the global meteoric water line. Ecol Appl 24:960–975
Vogel JC, Van Urk H (1975) Isotopic composition of groundwater in semi-arid regions of southern Africa. J Hydrol 25:23–36. https://doi.org/10.1016/0022-1694(75)90036-0
Warren JM, Brooks JR, Meinzer FC, Eberhart JL (2008) Hydraulic redistribution of water from Pinus ponderosa trees to seedlings: evidence for an ectomycorrhizal pathway. New Phytol 178:382–394. https://doi.org/10.1111/j.1469-8137.2008.02377.x
Warren JM, Meinzer FC, Brooks JR et al (2007) Hydraulic redistribution of soil water in two old-growth coniferous forests: quantifying patterns and controls. New Phytol 173:753–765. https://doi.org/10.1111/j.1469-8137.2006.01963.x
Warren JM, Meinzer FC, Brooks JR, Domec JC (2005) Vertical stratification of soil water storage and release dynamics in Pacific Northwest coniferous forests. Agric for Meterol 130:39–58
Weiguo L, Xiahong F, Yu L et al (2004) δ18O values of tree rings as a proxy of monsoon precipitation in arid Northwest China. Chem Geol 206:73–80. https://doi.org/10.1016/j.chemgeo.2004.01.010
Weiler M, Naef F (2003) An experimental tracer study of the role of macropores in infiltration in grassland soils. Hydrol Process 17:477–493. https://doi.org/10.1002/hyp.1136
Williams DG, Ehleringer JR (2000) Intra- and interspecific variation for summer precipitation use in pinyon–juniper woodlands. Ecol Monogr 70:517–537. https://doi.org/10.1890/0012-9615(2000)070[0517:IAIVFS]2.0.CO;2
Zeng X, Liu X, Evans MN et al (2016) Seasonal incursion of Indian Monsoon humidity and precipitation into the southeastern Qinghai-Tibetan Plateau inferred from tree ring δO18 values with intra-seasonal resolution. Earth Planet Sci Lett 443:9–19. https://doi.org/10.1016/j.epsl.2016.03.011
Zhou Y, Chen S, Song W et al (2011) Water-use strategies of two desert plants along a precipitation gradient in northwestern China. Chin J Plant Ecol 35:789–800
Zweifel R, Zimmermann L, Zeugin F, Newbery DM (2006) Intra-annual radial growth and water relations of trees: implications towards a growth mechanism. J Exp Bot 57:1445–1459. https://doi.org/10.1093/jxb/erj125
Acknowledgements
We thank Holly Barnard, Elizabeth Olson, Margaret Barbour, and Paul Mayer for their thoughtful and detailed reviews of this chapter. This chapter has been subjected to Agency review and has been approved for publication. The views expressed in this paper are those of the author(s) and do not necessarily reflect the views or policies of the U.S. Environmental Protection Agency. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 © The Author(s)
About this chapter
Cite this chapter
Allen, S.T., Sprenger, M., Bowen, G.J., Brooks, J.R. (2022). Spatial and Temporal Variations in Plant Source Water: O and H Isotope Ratios from Precipitation to Xylem Water. In: Siegwolf, R.T.W., Brooks, J.R., Roden, J., Saurer, M. (eds) Stable Isotopes in Tree Rings. Tree Physiology, vol 8. Springer, Cham. https://doi.org/10.1007/978-3-030-92698-4_18
Download citation
DOI: https://doi.org/10.1007/978-3-030-92698-4_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92697-7
Online ISBN: 978-3-030-92698-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)