Abstract
Tree-ring studies in the Mediterranean Basin and in regions characterized by Mediterranean climates are scarce because of a lack of old trees and difficulties related to the clear identification of individual rings.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
1 Introduction
Tree-ring studies in the Mediterranean Basin and in regions characterized by Mediterranean climates are scarce because of a lack of old trees and difficulties related to the clear identification of individual rings. Old trees are lacking because most regions with a Mediterranean climate have long histories of human activity; logging, grazing, and human-induced fires have occurred over millennia. Annual rings are difficult to identify in some woody plant species because highly variable climatic conditions can lead to the formation of intra-annual density fluctuations, which hamper the cross-dating of tree-ring series (Cherubini et al. 2013). Stable isotopes may help in identifying intra-annual density fluctuations (De Micco et al. 2007) and in understanding the physiological processes behind tree-ring formation, carbon uptake, and water use (Battipaglia et al. 2010a, 2014a).
Most of the tree-ring stable isotope studies carried out on Mediterranean tree and shrub species include the use of both carbon and oxygen stable isotopes (Table 21.1), which help to reconstruct past climate and retrospectively assess tree responses to the environment (McCarrol and Loader 2004). The values of the isotopic ratios reflect the extent to which the heavier isotope is discriminated compared to the lighter one during the physical and chemical processes involved in the synthesis of plant organic matter (Farquhar et al. 1989).
In this chapter, we highlight the importance of stable isotope research in Mediterranean ecosystems and explain the link between the morphological and functional characteristics of Mediterranean species and the climatic and environmental adaptations that have occurred over millennia.
2 Mediterranean Climates
Mediterranean climates occur around the world: in California, central Chile, western and southwestern Australia, southwestern South Africa, and in the Mediterranean Basin. As most tree-ring studies have been carried out in the Mediterranean, we focus on this region in this chapter. In a typical Mediterranean climate, winters are wet and rather mild, although frost and cold stress can occur (Larcher 2000; Mitrakos 1980; Terradas and Save 1992). Rainfall starts in September and continues until April, with a total precipitation amount of 400–1200 mm (Pignatti 2008). Summers are hot and dry; rainfall events are rare, and the season is often characterized by an extremely dry period, the so-called summer drought. These general features can vary in different coastal areas of the Mediterranean Basin depending on their elevation and local geomorphological and soil features, but the summer drought is always present (see Fig. 21.1). The main factors leading to the formation of Mediterranean-type climates are air circulation, latitude, topography, and the surface temperature of the surrounding water.
a Map of the Mediterranean Basin showing locations of stable isotope tree-ring studies. The main findings of these studies are summarized in Table 21.1. Background map showing Mediterranean climates according to Köppen classification. b Two Walter and Lieth climate diagrams depicting the climate of two typical Mediterranean areas. Lower diagram: Capri (an island in the Tirrenic sea); upper diagram: Tramonti, at a higher altitude (period 1960–2010). c A few examples of Mediterranean species. Clockwise from top left: Pinus halepensis Mill of southern Spain; Quercus ilex L from southern Italy; Euphorbia dendroides L.: winter habitus and summer habitus
2.1 Mediterranean Climate and Vegetation
Mediterranean plants are adapted to this climate and cope with summer drought through several changes at the phenological, morphological, physiological,
or biochemical levels (Pignatti 2008). These adaptations influence all relevant ecophysiological processes, i.e., transpiration and carbon assimilation, as well as the isotopic signal imprinted in plant organic matter. Hence, the isotopic composition of the wood material of Mediterranean trees reflects their functionality and adaptation strategies. To cope with drought stress, two main strategies are observed in Mediterranean plants: avoidance and tolerance (Pignatti 2008).
Many species survive summer drought by avoiding it through summer dormancy. This strategy is adopted by annual and perennial herbs and by woody species. Annual herbs have a very short life cycle that starts in late winter so that fruits are mature at the beginning of summer. Seeds then remain dormant until conditions are optimal for successful growth. Perennial herbaceous species often have a geophyte life-form, in which the above-ground parts of the plants (leaves, stems) are shed at the beginning of summer and the individual remains dormant by means of a perennating below-ground organ (bulb, tuber, rhizome). Some woody species also have summer dormancy, shedding all their leaves (e.g. Euphorbia dendroides L.) at the end of spring or summer. Several species, such as Cistus, change their leaves according to the season and are characterised by the alternation of a winter and a summer leaf habitus.
Plants without summer dormancy present tolerance attributes, which help them to face summer drought. Many species have a low leaf surface area, e.g., the needle-like leaves of Rosmarinus officinalis L. or Juniperus phoenicea L. subsp. phoenicea, whose juvenile needle-like leaves turn into scale-like leaves that are densely arranged around the stem in older branches. Other species have schlerophyllous leaves, with very dense mesophyll and thick cuticles to constrain water loss. Regulation of stomatal activity is a good strategy to cope with summer drought: stomatal closure can reduce transpiration rate and water loss. Several species count on stomata, which are sunken into the leaf surface and protected by dense, short trichomes, to reduce water loss. The lower leaf blades of R. officinalis, Quercus ilex L., and Olea europaea L. are examples of this feature. Other plants without summer dormancy tolerate high solar radiation by means of a reflecting cuticle (e.g., Myrtus communis L. subsp. communis) or long (often white) hairs on the upper leaf blade (e.g., Centaurea cineraria L. subsp. cineraria) that protect inner tissues from excessive radiation.
The adaptation mechanisms of Mediterranean plants are often linked with human activity, since their long association with humans has left its mark across much of the landscape. Human activities have influenced not only the type of community and forest composition, but also their main characteristics.
Four major types of vegetation can be recognized in the Mediterranean-European region according to the bioclimate type and the elevation (Quézel and Médail 2003; Rivas-Martínez et al. 2007; Médail 2008). First, a thermo-Mediterranean vegetation rises from sea level to ~200–500/800 m a.s.l. and is dominated by sclerophyllous communities (e.g., Olea europaea, Ceratonia siliqua, Chamaerops humilis, Pistacia lentiscus, Pinus halepensis, Pinus brutia, etc.). A meso-Mediterranean vegetation takes over between ~100–500/1000 m a.s.l. and is dominated by sclerophyllous forests (Quercus ilex, Quercus suber, and Q. coccifera) or Pinus halepensis/P. brutia forests in drought-prone forest systems. This then transitions to a supra-Mediterranean vegetation between ~500–1500/1800 m a.s.l., with deciduous oaks forests (e.g. Acer, Carpinus, Ostrya, Quercus, and Sorbus) in the more humid areas. Finally, a mountain-Mediterranean vegetation belt between ~1500–2000 m a.s.l. includes Fagus and deciduous and semi-deciduous Quercus forests, as well as several coniferous forests with Pinus nigra, Pinus sylvestris, firs (Abies alba and most of the Mediterranean Abies spp.), and even cedar (Cedrus libani s.l.) on Cyprus (Médail et al. 2019).
3 Mediterranean Wood Formation in Mediterranean Tree Species
The Mediterranean basin has an extension of more than 2 million square kilometers, and it is the largest of the world’s five mediterraneanc-climate regions, as well as the world’s second largest biodiversity hotspot (Myers et al. 2000).
The Mediterranean region is considered to be one of the climate-change hotspots, being a transition zone between temperate and arid and tropical regions (Diffenbaugh and Giorgi 2012). It has experienced an increase in drought frequency in the past three decades, and forecasts predict increased irregularity of the intra-annual precipitation pattern, and increasing temperature in the next decades (IPCC 2017). Water availability is considered the key factor driving ecophysiological processes in Mediterranean vegetation. It influences cambial activity, photosynthetic rates, dry matter production, phenology, allocation of carbon to roots and leaves, and overall growth activity of woody plants (Margaris and Papadogianni 1977; Lo Gullo and Salleo 1988; Orshan 1989; Gratani 1995; Salleo et al. 1997; Davis et al. 1999).
In Mediterranean ecosystems, tree species are adapted to cope with the so-called “double stress” of summer drought and winter low temperatures (Mitrakos 1980; Cherubini et al. 2003). These stressors trigger the formation of Intra Annual Density Fluctuations (IADFs) (Campelo et al. 2007; Battipaglia et al. 2016a; De Micco et al. 2016), which are caused by the interruption of the normal course of growth during the growing season. When favorable growth conditions resume, growth resumes, resulting in a change in wood density (Tingley 1937; Schulman 1938). In Mediterranean regions, this happens irregularly in space (at different sites at the same time), time (in different years at the same site), and among species (as well as individual trees). This ability to stop and restart growth within a growth season is indicative of the high plasticity of Mediterranean species, whose wood functional traits enable them to preserve hydraulic conductivity throughout the different seasons, achieving a trade-off between hydraulic efficiency and safety (see Beeckman 2016).
IADFs make it difficult to clearly identify individual annual rings; as a result, the tree rings of Mediterranean species are used less often for dendroecological purposes less than those of plants growing in temperate environments. However, a variety of recent research techniques have provided valuable information about the short-term growth variability of Mediterranean trees and shrubs. These include the intensive monitoring of stem radial variation using dendrometers (Sánchez-Costa et al. 2015), the assessment of cambial phenology (Camarero et al. 2010; Vieira et al. 2017), the measurement of xylem anatomical traits (De Micco et al. 2019; Carvalho et al. 2015; Pacheco et al. 2016), stable isotopes within tree rings (on tree-ring sub-sections, Klein et al. 2005; in continuum by laser ablation, Battipaglia et al. 2010a, b; 2014a) and a combination of methodologies (Zalloni et al. 2018, 2019; Balzano et al. 2018; Castagneri et al. 2018).
4 Carbon and Oxygen Stable Isotopes in Tree Rings of Mediterranean Species
4.1 Carbon Isotopes
Mediterranean species, which grow in climates characterized by summer drought and wet periods during the growing season, show wood structure adjustments that are mostly related to the need to maintain high conductivity when water is available, and to prevent xylem embolism when dry conditions occur (Cherubini et al. 2003; De Micco and Aronne 2012; Meinzer et al. 2010; Sperry et al. 2008). Tree-ring formation is therefore directly influenced by these adjustment processes and carbon stable isotopes can investigate the link between xylem hydraulic properties and the related physiological mechanisms.
Water shortage negatively influences carboxylation rates by reducing the CO2 concentration in the gaseous spaces within the plant (Ci) due to stomata closure, causing a decrease in carbon isotope discrimination (Farquhar et al. 1989; Scheidegger et al. 2000). Many studies conducted in Mediterranean climates have shown that plants growing under water stress (stress induced by low soil water content and high evaporative atmospheric demand) produce tree-rings with higher δ13C (Ferrio et al. 2003; Battipaglia et al. 2010a; Maseyk et al. 2011). However, leaf water availability ultimately influences tree-ring δ13C. This availability is a consequence not only of soil water content, but also of the physical structure and hydraulic resistance along the plant xylem and the way the plant transports and uses water (e.g. leaf phenology) (Masle and Farquhar 1988; Warren and Adams 2000). Further, tree δ13C is influenced by the leaf-to-air vapour pressure deficit (VPD), which is the driving force for transpiration. Studies on isotope-climate relationships have shown that mean annual precipitation controls carbon discrimination (Δ13C) in several forest ecosystem types (Schulze et al. 1998; Diefendorf et al. 2010; Kohn 2010), as well as in tree rings of a large range of Mediterranean species (Shestakova et al. 2019; del Castillo et al. 2013). These studies generally suggest a stronger influence of precipitation on Δ13C in drier environments (Warren et al. 2001).
4.2 Carbon Isotope Discrimination as a Proxy for iWUE
Carbon isotope composition has been used to calculate intrinsic water-use efficiency (iWUE) (see Chap. 17) in Mediterranean species (Ehleringer et al. 1993; Altieri et al. 2015; Battipaglia et al. 2016b; Dawson and Ehleringer 1993; Moreno-Gutiérrez et al. 2012). iWUE is key to the survival of Mediterranean species under drought conditions (Farquar et al. 1989); according to Medrano et al. (2009), a high iWUE can be considered an adaptive trait of Mediterranean species. Inter- and intra-species interactions influence iWUE, and iWUE can vary with inter- and intra-annual climate variability, stand density, and tree size (Forrester 2015). Within and across tree species, variations in iWUE reveal a continuous ecophysiological gradient of plant water-use strategies ranging from “profligate/opportunistic” (low iWUE) to “conservative” (high iWUE) (Moreno-Gutierrez et al. 2012). Several factors can affect iWUE at individual and stand levels, such as tree age and height (Francey and Farquhar 1982; Farquhar et al. 1982, Bert et al. 1997; McDowell et al. 2011a, b; Brienen et al. 2017), site density and characteristics (Battipaglia et al. 2010a; Zalloni et al. 2018), forest management, climate, and increasing atmospheric CO2 (Silva and Horward 2013, De Micco et al. 2019).
Both network studies and case studies have highlighted a general increase in iWUE in Mediterranean species (Linares et al. 2009; Di Matteo et al. 2010; Linares et al. 2011; Peñuelas et al. 2011; González-Muñoz et al. 2015; Brito et al. 2016; Fernández-de-Uña et al. 2016; Paris et al. 2018; Shestakova et al. 2019). All studies agree that the increase in iWUE does not translate into growth enhancement in response to increasing atmospheric CO2. However, it is not yet clear why this occurs because it is extremely difficult to disentangle the influence of single factors on iWUE variation and to quantify the extent to which drought overrides a positive CO2 fertilization effect (see Peñuelas et al. 2011). Moreover, iWUE can vary due to both photosynthetic and stomatal conductance rates, since both affect the ratio between CO2 partial pressure in leaf intercellular space and in the atmosphere. To better understand the role of assimilation on iWUE, several authors have coupled tree-ring δ13C values with wood δ18O values (the dual isotope approach; Chap. 16, paragraph 5).
4.3 Oxygen Isotopes as a Proxy for Source Water
Oxygen isotopes can provide valuable information about changes in source water, which is typically either precipitation or groundwater (Dansgaard, 1964). No fractionation occurs when water is taken up by the roots (Wershaw et al. 1966), so the δ18O of xylem water can be used to investigate plant water source use in a range of environmental conditions (Sternberg and Swart 1987; Ehleringer and Dawson 1992; Dawson et al. 1998a, b). Very few studies, however, have focused on Mediterranean environments (Sargeant and Singer 2016; Singer et al. 2013), where water sources are quite complex and subject to strong seasonal fluctuations. The interface between the influx of marine water and the freshwater table depends strongly on climatic factors and on the exploitation of groundwater for urban or rural purposes, both of which influence the quality of water available for plant communities.
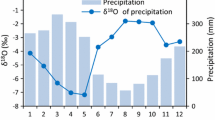
The isotopic composition of rainfall is typically subject to seasonal patterns that are influenced by geographic and climatic factors. Generally, autumn–winter precipitation has a more negative isotopic signature (ranging between ∼−7.8 and −6.0 ‰) than spring–summer precipitation (ranging between ∼−4.0 and +0.8 ‰) (Alessio et al. 2004). Consequently, the freshwater table is labelled by meteoric water, and primarily by the abundant autumn–winter and spring precipitation events (Wu et al. 1996). In contrast, soil water in the shallow layers depends on spring–summer precipitation. Further, water occurring 10–50 cm below the dry soil surface is subject to evaporative enrichment and therefore attains less negative or even positive δ18O values (Craig and Gordon 1965; Allison and Leaney 1982; Yakir et al. 2000). A further source of water is the marine water table, which is typically characterized by enriched values of δ18O (∼0 ‰).
Thus, the δ18O composition of tree rings will reflect the isotopic signature of the tree’s water source: rings will have a depleted δ18O signature if trees take up water from the groundwater table (Dawson and Ehleringer 1998), and an enriched δ18O signature if trees take up water from the vadose zone. Indeed, water in the vadose zone is generally linked to recent precipitation and is subject to high evaporative enrichment of δ18O (Dawson and Ehleringer 1998). These source-water signatures will be different for individual rings within a given species depending on the annual position of the water table and the degree of mixing between precipitation, surface flow, and groundwater during periods of xylem uptake (McCarroll and Loader 2004). It should also be noted that considerable fractionation occurs at the leaf/needle level, where lighter isotopes are preferentially lost via transpiration, leading to an enrichment in plant tissue δ18O of up to 20‰ compared to soil water (Craig and Gordon 1965; Saurer et al. 1998; McCarroll and Loader 2004; Barbour 2007; Cernusak et al. 2016). Indeed, it has been demonstrated that the source water signal can be modified by large changes in evaporative enrichment in drought-adapted Mediterranean species. These species tightly regulate transpiration through their stomata (Ferrio and Voltas, 2005), an ability that is thought to be a functional adaptation to drought (Pallary et al. 1995; Battipaglia et al. 2009).
Singer et al. (2013) and Sargeant and Singer (2016) used annual tree-ring δ18O analyses to interpret plant water use at various temporal and spatial scales as a function of regional hydrology and climate. Both studies underlined the great plasticity of plants as evinced by their ability to take up different sources of water and to modulate their root depth to take up water from different compartments in order to avoid competition. This promising approach needs to be exploited further, as it could provide important information regarding the ability of plants to survive drought events cooperatively (Altieri et al. 2015).
5 Application of Carbon and Oxygen Isotopes in Tree-Rings of Mediterranean Species
The carbon and oxygen composition of tree-rings has been used for elucidating whether plant functional responses are related to stomata control of water losses (gs) or to varying assimilation rates (A), since δ18O shared a dependence on gs with δ13C, but is thought to be independent of variation in A (Scheidegger et al. 2000; Grams et al. 2007; Roden and Farquhar 2012).
Indeed, even if the interpretation of the double model δ13C—δ18O is not straightforward (Roden and Siegwolf 2012), and may sometimes be hampered by changes in the source water isotopes (Gessler et al. 2014), it can still provide important information when applied in strongly water-limited ecosystems, such as the Mediterranean (Ripullone et al. 2009; Moreno-Gutiérrez et al. 2012; Voltas et al. 2013; Gessler et al. 2014; Altieri et al. 2015; Battipaglia et al. 2016a). In the next paragraphs, we will illustrate specific examples of a double δ13C—δ18O model that is relevant for the Mediterranean region.
5.1 Carbon and Oxygen Isotopes and Forest Dieback in the Mediterranean Basin
Forest vulnerability is reported to be increasing worldwide; forest dieback episodes have been recorded for different species in all biomes (Allen et al. 2015; Adams et al. 2017; Hartmann et al. 2018) and particularly for tree and shrub species in Mediterranean ecosystems. Manifested by a loss in tree vigour (leaf shedding, canopy and shoot dieback) and growth declines, these dieback cases reveal the high vulnerability of forest ecosystems. Increased tree mortality seems to be a response to the rapid rise in temperature and associated drying trends (Camarero et al. 2015; Colangelo et al. 2017). In the Mediterranean, trends of increasing temperature and altered precipitation patterns lead to higher probabilities of extreme events, such as heat waves and fires (IPPC 2014). Species-, age- and microsite-specific plant responses to changing climatic conditions are responsible for the degree of adaptation and survival under limiting conditions. These factors also determine interspecific competition, and thus vegetation dynamics.
The main mechanisms triggering tree decline and mortality have been identified as carbon starvation and hydraulic failure, but the relative importance of the two processes and their link are not clear yet (McDowell et al. 2008, Sala 2009, Sala et al. 2010, Gruber et al. 2010, Sevanto et al. 2014, Gaylord et al. 2015, Hartmann 2015, Rowland et al. 2015, Salmon et al. 2015; Adams et al. 2017). Plants experience hydraulic failure when more water is lost by transpiration than a plant can take up through its root. This creates high xylem water tension and results in progressive cavitation and conductivity loss of the xylem (Sperry et al. 1998; McDowell et al. 2008; Sevanto et al. 2014; Salmon et al. 2015). This process may be most relevant during very severe, short-term droughts (McDowell et al. 2008). On the other hand, carbon starvation occurs when plants close their stomata to prevent desiccation under drought conditions (McDowell et al. 2008). Low water supply leads to a reduction of photosynthetic activity, which, when coupled with the depletion of non-structural carbohydrates (NSC), can induce a negative carbon balance, leading to so-called starvation (McDowell et al. 2008; McDowell and Sevanto 2010; Hartmann 2015; Salmon et al. 2015).
Gessler et al. (2018) developed a conceptual model, based on the Scheidegger model (Scheidegger et al. 2000, see Chap. 16), to assess the mechanisms of drought-induced tree mortality. Using a synchronic approach, they investigated the tree-ring δ13C and δ18O data for different species and found that an increase in δ13C and δ18O in dying trees as compared to surviving ones indicates low photosynthetic activity and stomatal conductance. These conditions can lead to a slow or continuous decline in growth, possibly followed by carbon starvation-induced death. On the other hand, the long-term reduction of iWUE in dying trees seems to be associated with increased growth and higher stomatal conductance, inducing possible hydraulic failure in dying trees. However, those findings show a huge variability among species and sites. This variability is linked to the complex processes regulating growth and, in particular, the fractionation processes that take place during the mobilization of photosynthetic assimilates from the leaf to the wood (Gessler et al. 2014). Indeed, it has been demonstrated that trees may use remobilized carbohydrates (primarily starch) for the formation of tree rings. These stored carbohydrates may have been produced months or even years earlier (Gessler et al. 2014). This is especially possible in Mediterranean biomes, where the growing season is very long (Castagneri et al. 2018) and where xylogenesis can be interrupted more than once during the year (Balzano et al. 2019, 2018). In this case, a multidisciplinary approach is required to disentangle the different processes underpinning changes in growth and iWUE, and to better understand the causes of tree diebacks (Cailleret et al. 2017; Colangelo et al. 2017; Camarero et al. 2015, 2019).
5.2 Carbon and Oxygen Isotopes and IADF
Intra-annual density fluctuations (IADFs) are abrupt changes in density within a tree ring, and are frequently found in Mediterranean species as a response to seasonal climate fluctuations between dry and wet periods (Bräuning 1999; Campelo et al. 2007; Cherubini et al. 2003; Rigling et al. 2001; Schulman 1938; Tingley 1937; De Micco et al. 2016). Although IADF frequency is related to tree age, size, and tree-ring width, as well as to genetic and site conditions, it mainly depends on drought. IADF frequency is the result of wood functional trait adjustments to preserve hydraulic conductivity under Mediterranean “double stress” conditions (De Micco et al. 2016). The ability to adapt to highly seasonal Mediterranean conditions may depend on a species´ capacity to adjust cambial activity in order to cope with the prevailing environmental conditions. Trees that are not able to quickly adjust their wood traits to respond to climate are more vulnerable to drought (Martinez-Meier et al. 2008). Nevertheless, the question of whether these adaptations result from a hydraulic structure adjustment to avoid stressful conditions or to take advantage of favorable ones is still open (Battipaglia et al. 2016a).
Carbon and oxygen stable isotopes have been used both separately and together to study IADF formation in Pinus pinaster (De Micco et al. 2007), Arbutus unedo (Battipaglia et al. 2010a; b), Erica arborea (Battipaglia et al. 2014a), Quercus ilex, and Pinus pinea (Zalloni et al. 2018, 2019), and to infer information about IADF functionality. In particular, the position of IADF within the rings is linked with several microclimatic factors and varies within the same species according to the soil water availability, reflects different δ13C signals (Battipaglia et al. 2010a). An increase in δ13C is almost always found in the first half of the ring (in the so-called E-IADF band of latewood-like cells in the earlywood; see De Micco et al. 2016) as a consequence of stomatal closure under drought stress, when the tree reduces its vulnerability to cavitation, with a low hydraulic conductivity (De Micco et al. 2007, Battipaglia et al. 2010a, 2014a). A decrease in δ13C is typically found in the second half of the ring in the so-called L-IADF (earlywood-like cells in the latewood) and corresponds with a reactivation of the cambium and of photosynthetic activity due to late-summer or early-autumn water availability following summer dormancy (Battipaglia et al. 2014a). The importance of the position of IADFs and their link to a common ecophysiological process was analysed using a network approach to examine δ13C and δ18O in several species (P. pinea from Italy, Pinus halepensis from Spain and Slovenia; P. pinaster from Portugal, Larix decidua from Poland, and L. decidua xkaempferi from Austria). For all the different sites and species, the types of IADF and their positions had the same isotopic signals in terms of carbon and oxygen. E-IADFs presented δ13C and δ18O in the range of −23 ± 0.6‰ and 32 ± 1‰, respectively; L-IADFs had δ13C and δ18O values of −27 ± 0.7‰ and 29 ± 0.5‰, respectively (Battipaglia et al. 2016a). These results should be extended to a larger database in order to assess how the isotopic composition of IADFs could help with dating problematic samples and support the interpretation of phenomena that trigger the formation of IADFs in the Mediterranean environment.
5.3 Carbon and Oxygen Isotopes and Fire
Fire has been a frequent and important disturbance in Mediterranean forest ecosystems throughout the Holocene (Heinselman 1981; FAO, State of Mediterranean Forest 2018), shaping landscapes and determining vegetation distribution and dynamics. Tree rings have been used to reconstruct fire history and to determine the effects of fire on forest productivity. In recent years, carbon and oxygen isotopes measured in tree rings have proved to be a useful tool for understanding the complex ecophysiological processes that occur in Mediterranean tree species following a wildfire or prescribed burning (Beghin et al. 2011; Battipaglia et al. 2014b, c, 2016b, 2019; De Micco et al. 2014; Valor et al. 2018; Niccoli et al. 2019). Because conifers are widespread in the Mediterranean Basin and highly prone to fire damage, many studies have focused on conifer species, including Pinus sylvestris (Beghin et al., 2011), Pinus halepensis (Battipaglia et al. 2014b; c; Valor et al. 2018), Pinus pinea (Battipaglia et al. 2016b), and Pinus pinaster (Niccoli et al. 2019). Stable isotopes studies indicate that tree response and their potential to recover over the short- and long-term depends on fire severity. Strong wildfires produce a simultaneous increase in tree-ring δ13C and δ18O, suggesting a strong reduction in stomatal conductance and assimilation rate, which are often linked to crown damage (Battipaglia et al. 2014b; Niccoli et al. 2019). When fire severity is low to moderate, the effect on trees is less pronounced; often, the tree rings only report a small variation in δ13C, with no change of δ18O. In these cases, a reduction in tree growth is largely due to the reduced photosynthetic capacity of the burned trees (Battipaglia et al. 2016b). Following prescribed burning s, however, trees show a decrease in both δ13C and δ18O, indicating a possible favorable effect of reduced competition between the surviving plants. In addition to reducing competition, prescribed burning seems to lead to a release of nutrients, which can also stimulate the growth of surviving trees (Battipaglia et al. 2014c, 2016b; Valor et al. 2018; Niccoli et al. 2019).
6 Nitrogen Stable Isotopes
Nitrogen availability is an important factor limiting productivity in Mediterranean ecosystems (Noy-Meir 1973; Gutierrez and Withford 1987; Lloret et al. 1999) and therefore influencing wood formation. Tree-ring δ15N is mainly influenced by the isotopic ratio of the available nitrogen sources (Nadelhoffer and Fry 1994; Peñuelas and Estiarte 1997; Evans 2001, Stewart et al. 2001). Thus, tree-ring δ15N allows for the reconstruction of the nitrogen source and the relative activity of the different biogeochemical processes that affect the δ15N of nitrogen compounds taken up by trees in the surrounding ecosystem (i.e., mineralization, nitrification, denitrification and, NO3 leaching; see Chap. 12). However, other fractionation events that occur in plants during assimilation processes can also influence tree-ring δ15N and complicate interpretation of the nitrogen sources (Evans 2001). Indeed, tree-ring δ15N may also depend on nitrogen reabsorption or re-translocation (Kolb and Evans 2002) or fractionation by different mycorrhizal associations (Michelsen et al. 1998; Craine et al. 2009). The incorporated tree-ring δ15N signature thus results from an integration of nitrogen sources and internal transformations. These complications may explain why only a limited number of studies have been carried out in Mediterranean ecosystems, most of which have focused on understanding the effects of climate change on carbon and nitrogen cycles (Peñuelas et al. 1996; Alfaro-Sanchez et al. 2016).
Tree-ring δ15N has also been used to assess changes in nitrogen availability due to (i) variations in atmospheric nitrogen deposition and (ii) their influence on ecosystem nitrogen dynamics, since the nitrogen isotope ratio in compounds produced as a result of anthropogenic activities may be significantly different from the natural background δ15N in the soil (Freyer 1991). Guerrieri et al. (2009, 2011) and Battipaglia et al. (2010b) showed that increases in nitrogen deposition from the atmosphere or from fertilization are reflected in tree-ring δ15N signals as a result of source δ15 N (e.g., NOx, NHx forms, or NH4+ vs NO3−) and the processes occurring during different phases of soil biogeochemical processes (i.e., losses caused by NO3− leaching, denitrification, and NH3 volatilization; see also Chap. 12). Changes in δ15N in annual tree-rings reflect the causes of variation in the tree’s iWUE due to climatic or anthropogenic impacts, suggesting that an increase in nitrogen input from the atmosphere (Guerrieri et al. 2011) or from consociation from nitrogen-fixing species could led to an increase of trees iWUE under a scenario of reduction in precipitation, such as in Mediterranean area (Guerrieri et al. 2011; Battipaglia et al. 2017). Measurement of δ15N of the different possible sources, and a better understanding of the fractionation processes in the different species, is needed for a proper interpretation of tree-ring δ15N data.
7 Conclusions and Outlook
Tree-ring isotope studies in the Mediterranean Basin have increased in the last years for two main reasons. First, there is general agreement regarding the importance of reliable information about how Mediterranean trees are responding to a changing climate. Such information informs assessments of the future state of such vulnerable forests, as well as their role in carbon sequestration and ecosystem services. Second, stable isotopes help to identify the ecological behavior and vulnerability of Mediterranean tree and shrub species, information that can improve management options. Further research is needed to improve our knowledge about the xylem plasticity of the different Mediterranean species. This will help pinpoint how cambial activity affects the functional processes of trees and their responses to climate change. The combination of multiple approaches, including stable isotopes, xylogenesis, wood anatomy, and long-term growth and isotope-based gas exchange data, may provide more insight on how plants optimize growth and minimize costs.
References
Adams HD, Zeppel MJB, Anderegg WRL, Hartmann H, Landhäusser SM, Tissue DT, Huxman TE, Hudson PJ, Franz TE, Allen CD, Anderegg LD, Allen CD (2017) A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nature Ecol Evol 1(9):1285–1291. https://doi.org/10.1038/s41559-017-0248-x
Aguilera M, Espinar C, Ferrio JP, Pérez G, Voltas J (2009) A map of autumn precipitation for the third millennium BP in the eastern Iberian Peninsula from charcoal carbon isotopes. J Geochem Explor 102(3):157–165. https://doi.org/10.1016/j.gexplo.2008.11.019
Aguilera M, Ferrio JP, Araus JL, Tarrús J, Voltas J (2011) Climate at the onset of western Mediterranean agriculture expansion: evidence from stable isotopes of sub-fossil oak tree rings in Spain. Palaeogeogr, Palaeoclimatol, Palaeoecol 299(3–4):541–551. https://doi.org/10.1016/j.palaeo.2010.11.026
Alfaro-Sánchez R, Camarero JJ, Sánchez-Salguero R, Sangüesa-Barreda G, De Las HJ (2016) Post-fire aleppo pine growth, C and N isotope composition depend on site dryness. Trees—Struct Funct 30(3):581–595. https://doi.org/10.1007/s00468-015-1342-9
Allen CD, Breshears DD, McDowell NG (2015) On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 6(8). https://doi.org/10.1890/ES15-00203.1
Alessio GA, de Lillis M, Brugnoli E, Lauteri M (2004) Water sources and water-use efficiency in Mediterranean coastal dune vegetation. Plant Boil 6:350–357. https://doi.org/10.1055/s-2004-820882
Allison GB, Leaney FW (1982) Estimation of isotopic exchange parameters, using constant-feed pans. J Hydrol 55:151–161
Altieri S, Mereu S, Cherubini P, Castaldi S, Sirignano C, Battipaglia LC, G, (2015) Tree-ring carbon and oxygen isotopes indicate different water use strategies in three Mediterranean shrubs at Capo Caccia (Sardinia, Italy). Trees—Struct Funct 29(5):1593–1603. https://doi.org/10.1007/s00468-015-1242-z
Balzano A, Čufar K, Battipaglia G, Merela M, Prislan P, Aronne G, De Micco V (2018) Xylogenesis reveals the genesis and ecological signal of IADFs in Pinus pinea L. and Arbutus unedo L. and. Ann Botany 121(6):1231–1242. https://doi.org/10.1093/aob/mcy008
Balzano A, Battipaglia G, De Micco V (2019) Wood-trait analysis to understand climatic factors triggering intra-annual density-fluctuations in co-occurring Mediterranean trees. IAWA J 40(2):241–258. https://doi.org/10.1163/22941932-40190220
Barbeta A, Peñuelas J (2017) Increasing carbon discrimination rates and depth of water uptake favor the growth of Mediterranean evergreen trees in the ecotone with temperate deciduous forests. Glob Change Biol 23(12):5054–5068. https://doi.org/10.1111/gcb.13770
Barbour MM (2007) Stable oxygen isotope composition of plant tissue: a review. Funct Plant Biol 34(2):83. https://doi.org/10.1071/fp06228
Bartolomei P (1995) Solar flare particle effects and seasonal radiocarbon variations in tree rings of the Northern and Southern Hemispheres. Radiocarbon 37(2):593–598. https://doi.org/10.1017/S0033822200031088
Battipaglia G, Jäggi M, Saurer M, Siegwolf RTW, Cotrufo MF (2008) Climatic sensitivity of δ18O in the wood and cellulose of tree rings: results from a mixed stand of Acer pseudoplatanus L. and Fagus sylvatica L. Palaeogeogr Palaeoclimatol Palaeoecol 261(1–2):193–202. https://doi.org/10.1016/j.palaeo.2008.01.020
Battipaglia G, Saurer M, Cherubini P, Siegwolf RTW, Cotrufo MF (2009) Tree rings indicate different drought resistance of a native (Abies alba mill.) and a nonnative (Picea abies (L.) karst.) species co-occurring at a dry site in southern Italy. Forest Ecol Manage 257(3):820–828. https://doi.org/10.1016/j.foreco.2008.10.015
Battipaglia G, De Micco V, Brand WA, Linke P, Aronne G, Saurer M, Cherubini P (2010a) Variations of vessel diameter and δ13C in false rings of Arbutus unedo L. reflect different environmental conditions. New Phytol 188(4):1099–1112. https://doi.org/10.1111/j.1469-8137.2010.03443.x
Battipaglia G, Marzaioli F, Lubritto C, Altieri S, Strumia S, Cherubini P, Cotrufo MF (2010b) Traffic pollution affects tree-ring width and isotopic composition of Pinus pinea. Sci Total Environ 408(3):586–593. https://doi.org/10.1016/j.scitotenv.2009.09.036
Battipaglia G, De Micco V, Brand WA, Saurer M, Aronne G, Linke P, Cherubini P (2014a) Drought impact on water use efficiency and intra-annual density fluctuations in Erica arborea on Elba (Italy). Plant, Cell Environ 37(2):382–391. https://doi.org/10.1111/pce.12160
Battipaglia G, De Micco V, Fournier T, Aronne G, Carcaillet C (2014b) Isotopic and anatomical signals for interpreting fire-related responses in Pinus halepensis. Trees—Struct Funct 28(4):1095–1104. https://doi.org/10.1007/s00468-014-1020-3
Battipaglia G, Strumia S, Esposito A, Giuditta E, Sirignano C, Altieri S, Rutigliano FA (2014c) The effects of prescribed burning on Pinus halepensis mill. as revealed by dendrochronological and isotopic analyses. Forest Ecol Manage 334:201–208. https://doi.org/10.1016/j.foreco.2014.09.010
Battipaglia G, Campelo F, Vieira J, Grabner M, De Micco V, Nabais C, de Luis M et al (2016a) Structure and function of intra–annual density fluctuations: mind the gaps. Front Plant Sci 7:1–8. https://doi.org/10.3389/fpls.2016.00595
Battipaglia G, Savi T, Ascoli D, Castagneri D, Esposito A, Mayr S, Nardini A (2016b) Effects of prescribed burning on ecophysiological, anatomical and stem hydraulic properties in Pinus pinea L. Tree Physiol 36(8):1–13. https://doi.org/10.1093/treephys/tpw034
Battipaglia G, Pelleri F, Lombardi F, Altieri S, Vitone A, Conte E, Tognetti R (2017) Effects of associating Quercus robur L. and Alnus cordata loisel. on plantation productivity and water use efficiency. Forest Ecol Manage 391:106–114. https://doi.org/10.1016/j.foreco.2017.02.019
Beeckman H (2016) Wood anatomy and trait-based ecology. IAWA J 37:127–151
Beghin R, Cherubini P, Battipaglia G, Siegwolf R, Saurer M, Bovio G (2011) Tree-ring growth and stable isotopes (13C and 15N) detect effects of wildfires on tree physiological processes in Pinus sylvestris L. Trees Struct Funct 25:627–636. https://doi.org/10.1007/s00468-011-0539-9
Bogino SM, Bravo F (2014) Carbon stable isotope-climate association in tree rings of Pinus pinaster and Pinus sylvestris in Mediterranean environments. [Asociación entre el clima y los isótopos estables de carbono en los anillos de crecimiento de Pinus pinaster y Pinus sylvestris en ambientes Mediterráneos]. Bosque 35(2):175–184. https://doi.org/10.4067/S0717-92002014000200005
Bräuning A (1999) Dendroclimatological potential of drought-sensitive tree stands in Southern Tibet for the reconstruction of monsoonal activity. IAWA J 20(3):325–338
Brienen RJW, Gloor E, Clerici S, Newton R, Arppe L, Boom A, Timonen M et al (2017) Tree height strongly affects estimates of water-use efficiency responses to climate and CO2 using isotopes. Nat Commun 8(1). https://doi.org/10.1038/s41467-017-00225-z
Brito P, Grams TEE, Matysssek R, Jimenez MS, Gonzalez-Rodríguez AM, Oberhuber W, Wieser G (2016) Increased water use efficiency does not prevent growth decline of Pinus canariensis in a semi-arid treeline ecotone in tenerife, canary islands (Spain). Ann Forest Sci 73(3):741–749. https://doi.org/10.1007/s13595-016-0562-5
Brooks JR, Coulombe R (2009) Physiological responses to fertilization recorded in tree rings: isotopic lessons from a long-term fertilization trial. Ecolo Appl 19(4):1044–1060. https://doi.org/10.1890/08-0310.1
Bert D, Leavitt SW, Dupouey JL (1997) Variations of wood δ13C and water-use efficiency of Abies alba during the last century. Ecology 78(1997):1588–1596. https://doi.org/10.1890/0012-9658(1997)078[1588:vowcaw]2.0.co
Cailleret M, Jansen S, Robert EMR , deSoto L, Martinez-Vilalta J et al (2017) A synthesis of radial growth patterns preceding tree mortality. Glob Chang Biol 23:1675–1690
Camarero JJ, Olano JM, Parras A (2010) Plastic bimodal xylogenesis in conifers from continental Mediterranean climates. New Phytol 185:471–480
Camarero JJ, Gazol A, Sangüesa-Barreda G, Oliva J, Vicente-Serrano SM (2015) To die or not to die: early-warning signals of dieback in response to a severe drought. J Ecol 103:44–57
Camarero JJ, Sangüesa-Barreda G, Pérez-Díaz S, Montiel-Molina C, Seijo F, López-Sáez JA (2019) Abrupt regime shifts in post-fire resilience of Mediterranean mountain pinewoods are fuelled by land use. Int J Wildland Fire. https://doi.org/10.1071/wf18160
Campelo F, Nabais C, Freitas H, Gutiérrez E (2007) Climatic significance of tree-ring width and intra-annual density fluctuations in Pinus pinea from a dry Mediterranean area in Portugal. Ann for Sci 64:229–238
Carvalho A, Nabais C, Vieira J, Rossi S, & Campelo F (2015) Plastic response of tracheids in Pinus pinaster in a water-limited environment: adjusting lumen size instead of wall thickness. PLoS ONE 10:e0136305.https://doi.org/10.1371/journal.pone.0136305
Castagneri D, Battipaglia G, Von Arx G, Pacheco A, Carrer M (2018) Tree-ring anatomy and carbon isotope ratio show both direct and legacy effects of climate on bimodal xylem formation in Pinus pinea. Tree Physiol 38(8):1098–1109. https://doi.org/10.1093/treephys/tpy036
Cernusak LA, Barbour MM, Arndt SK et al (2016) Stable isotopes in leaf water of terrestrial plants. Plant Cell Environ 39:1087–1102
Cherubini P, Gartner BL, Tognetti R, Bra ̈ker OU, Schoch W, Innes JL, (2003) Identification, measurement and interpretation of tree rings in woody species from Mediterranean climates. Biol Rev Cambridge Philos Soc 78:119–148
Cherubini P, Humbel T, Beeckman H, Gartner H, Mannes D, Pearson C, Schoch W, Tognetti R, Levyadun S (2013) Olive tree-ring problematic dating: a comparative analysis on Santorini (Greece). PLoS ONE 8(1):1–5, e54730
Colangelo M, Camarero JJ, Battipaglia G, Borghetti M, De Micco V, Gentilesca T, Ripullone F (2017) A multi-proxy assessment of dieback causes in a mediterranean oak species. Tree Physiol 37(5):617–631. https://doi.org/10.1093/treephys/tpx002
Craig H, Gordon LI (1965) Deuterium and oxygen 18 variations in the ocean and the marine atmosphere. In: Tongiorgi E (ed) Stable isotopes in oceanographic studies and paleotemperatures. Laboratorio di Geologia Nucleare, Pisa, pp 9–130
Craine JM, Elmore AJ, Aidar MPM, Bustamante M, Dawson TE, Hobbie EA, Kahmen A, Mack MC, McLauchlan KK, Michelsen A et al (2009) Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol 183:980–992
Dansgaard W (1964) Stable isotopes in precipitation. Tellus 16:436–468
Davis MA, Wrage KJ, Reich PB, Tjoelker MG, Schaeffer T, Muermann C (1999) Survival, growth, and photosynthesis of tree seedlings competing with herbaceous vegetation along a water-light-nitrogen gradient. Plant Ecol 145:341–350
Dawson TE (1998) Fog in the california redwood forest: ecosystem inputs and use by plants. Oecologia 117(4):476–485. https://doi.org/10.1007/s004420050683
Dawson TE, Ehleringer JR (1993) Isotopic enrichment of water in the “woody” tissues of plants: implications for plant water source, water uptake, and other studies which use stable isotopic composition of cellulose. Geochim Cosmochim Acta 57:3487–3492
Dawson TE, Ehleringer JR (1998) The role of plants in catchment-level hydraulic processes: insights from stable isotope studies. In: Kendall C, McDonnell JJ (eds) Isotope tracers in catchment hydrology. Elsevier; Amsterdam, pp 165–202
Del Castillo J, Aguilera M, Voltas J, Ferrio JP (2013) Isoscapes of tree-ring carbon-13 perform like meteorological networks in predicting regional precipitation patterns. J Geophys Res: Biogeosci 118(1):352–360. https://doi.org/10.1002/jgrg.20036
De Micco V, Aronne G (2012) Occurrence of morphological and anatomical adaptive traits in young and adult plants of the rare Mediterranean cliff species primula Palinuri Petagna. The Sci World J. https://doi.org/10.1100/2012/471814
De Micco V, Saurer M, Aronne G, Tognetti R, Cherubini P (2007) Variations of wood anatomy and δ13C within-tree rings of coastal pinus pinaster showing intra-annual density fluctuations. IAWA J 28(1):61–74
De Micco V, Battipaglia G, Brand WA, Linke P, Saurer M, Aronne G, Cherubini P (2012) Discrete versus continuous analysis of anatomical and δ 13C variability in tree rings with intra-annual density fluctuations. Trees—Struct Funct 26(2):513–524. https://doi.org/10.1007/s00468-011-0612-4
De Micco V, Battipaglia G, Cherubini P, Aronne G (2014) Comparing methods to analyse anatomical features of tree rings with and without intra- annual-density-fluctuations (IADFs). Dendrochronologia 32:1–6. https://doi.org/10.1016/j.dendro.2013.06.001
De Micco V, Campelo F, De Luis M, Bräuning A, Grabner M, Battipaglia G, Cherubini P (2016) Intra-annual density fluctuations in tree rings: how, when, where, and why? IAWA J 37(2):232–259. https://doi.org/10.1163/22941932-20160132
De Micco V, Carrer M, Rathgeber CBK, Julio Camarero J, Voltas J, Cherubini P, Battipaglia G (2019) From xylogenesis to tree rings: wood traits to investigate tree response to environmental changes. IAWA J 40(2):155–182. https://doi.org/10.1163/22941932-40190246
Diefendorf AF, Mueller KE, Wing SL, Koch PL, Freeman KH (2010) Global patterns in leaf 13C discrimination and implications for studies of past and future climate. Proc Natl Acad Sci 107(13):5738–5743. https://doi.org/10.1073/pnas.0910513107
Diffenbaugh NS, Giorgi F (2012) Climate change hotspots in the CMIP5 global climate model ensemble. Clim Change 114(3–4):813–822. https://doi.org/10.1007/s10584-012-0570-x
Di Matteo G, De Angelis P, Brugnoli E, Cherubini P, & Scarascia-Mugnozza G (2010) Tree-ring Δ13C reveals the impact of past forest management on water-use efficiency in a mediterranean oak coppice in Tuscany (italy). Ann Forest Sci 67(5). https://doi.org/10.1051/forest/2010012
Ehleringer JR, Dawson TE (1992) Water uptake by plants: perspectives from stable isotope composition. Plant, Cell Environ 15(9):1073–1082. https://doi.org/10.1111/j.1365-3040.1992.tb01657.x
Ehleringer JR, Hall AE, Farquhar GD (1993) Introduction: water use in relation to productivity. In: Ehleringer JR, Hall AE, Farquhar GD (eds) Stable isotopes and plant carbon–water relations. Academic Press, New York, pp 3–8
Evans R (2001) Physiological mechanisms influencing plant nitrogen isotope composition. Trends Plant Sci 6:121–126
Farquhar GD, Leary MHO, Berry JA (1982) On the relation- ship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Ann Rev Plant Physiol Plant Mol Biol 40:503–537
Fernández-de-Uña L, Mcdowell NG, Cañellas I, Gea-Izquierdo G (2016) Disentangling the effect of competition, CO2 and climate on intrinsic water-use efficiency and tree growth. J Ecol 104(3):678–690. https://doi.org/10.1111/1365-2745.12544
Fernández-De-Uña L, Rossi S, Aranda I, Fonti P, González-González BD, Cañellas I, Gea-Izquierdo G (2017) Xylem and leaf functional adjustments to drought in Pinus sylvestris and Quercus pyrenaica at their elevational boundary. Front Plant Sci 8.https://doi.org/10.3389/fpls.2017.01200
Ferrio JP, Florit A, Vega A, Serrano L, Voltas J (2003) D13C and tree-ring width reflect different drought responses in Quercus ilex and Pinus halepensis. Oecologia 137(4):512–518. https://doi.org/10.1007/s00442-003-1372-7
Ferrio JP, Voltas J (2005) Carbon and oxygen isotope ratios in wood constituents of Pinus halepensis as indicators of precipitation, temperature and vapour pressure deficit. Tellus 57B:164–173
Forrester DI (2015) Transpiration and water-use efficiency in mixed-species forests versus monocultures: effects of tree size, stand density and season. Tree Physiol 35(3):289–304. https://doi.org/10.1093/treephys/tpv011
Francey RJ, Farquhar GD (1982) An explanation of 13C/12C variations in tree rings. Nature 297:28–31
Freyer HD (1991) Seasonal variation of 15N/14N ratios in atmospheric nitrate species. Tellus B 43(1):30–44. https://doi.org/10.1034/j.1600-0889.1991.00003.x
Gaylord ML, Kolb TE, McDowell NG (2015) Mechanisms of piñon pine mortality after severe drought: a retrospective study of mature trees. Tree Physiol 35:806–816
Gessler A, Ferrio JP, Hommel R, Treydte K, Werner RA, Monson RK (2014) Stable isotopes in tree rings: towards a mechanistic understanding of isotope fractionation and mixing processes from the leaves to the wood. Tree Physiol 34(8):796–818. https://doi.org/10.1093/treephys/tpu040
Gessler A, Cailleret M, Joseph J, Schönbeck L, Schaub M, Lehmann M, Treydte K, Rigling A, Timofeeva G, Saurer M (2018) Drought induced tree mortality—a tree-ring isotope based conceptual model to assess mechanisms and predispositions. New Phytol 219(2):485–490. https://doi.org/10.1111/nph.15154
González-Muñoz N, Linares JC, Castro-Díez P, Sass-Klaassen U (2015) Contrasting secondary growth and water-use efficiency patterns in native and exotic trees co-occurring in inner spain riparian forests. Forest Syst 24(1). https://doi.org/10.5424/fs/2015241-06586
Grams TE, Kozovits AR, Haberle KH, Matyssek R, Dawson TE (2007) Combining d13C and d18O analyses to unravel competition, CO2 and O3 effects on the physiological performance of different-aged trees. Plant, Cell Environ 30:1023–1034
Granda E, Rossatto DR, Camarero JJ, Voltas J, Valladares F (2014) Growth and carbon isotopes of mediterranean trees reveal contrasting responses to increased carbon dioxide and drought. Oecologia 174(1):307–317. https://doi.org/10.1007/s00442-013-2742-4
Gratani L (1995) Structural and ecophysiological plasticity of some evergreen species of the Mediterranean maquis in response to climate. Photosynthetica 31:335–343
Gutierrez JR, Whitford WG (1987) Chihuahuan desert annuals: importance of water and nitrogen. Ecology 68:2032–2045
Guerrieri MR, Siegwolf RTW, Saurer M, Jäggi M, Cherubini P, Ripullone F, Borghetti M (2009) Impact of different nitrogen emission sources on tree physiology as assessed by a triple stable isotope approach. Atmos Environ 43(2):410–418. https://doi.org/10.1016/j.atmosenv.2008.08.042
Guerrieri R, Siegwolf R, Saurer M, Ripullone F, Mencuccini M, Borghetti M (2010) Anthropogenic NOx emissions alter the intrinsic water-use efficiency (WUEi) for quercus cerris stands under Mediterranean climate conditions. Environ Pollut 158(9):2841–2847. https://doi.org/10.1016/j.envpol.2010.06.017
Guerrieri R, Mencuccini M, Sheppard LJ, Saurer M, Perks MP, Levy P, Sutton MA, Borghetti M, Grace J (2011) The legacy of enhanced N and S deposition as revealed by the combined analysis of δ13C, δ18O and δ15N in tree rings. Glob Change Biol 17(5):1946–1962.https://doi.org/10.1111/j.1365-2486.2010.02362.x
Gruber A, Strobl S, Veit B, Oberhuber W (2010) Impact of drought on temporal dynamics of wood formation in Pinus Sylvestris. Tree Physiol 30:490–501
Hartmann H (2015) Carbon starvation during drought‐induced tree mortality—are we chasing a myth? J Plant Hydraul 2(e005):1–5
Hartmann H, Moura CF, Anderegg WRL, Ruehr NK, Salmon Y, Allen CD, O’Brien M et al (2018) Research frontiers for improving our understanding of drought-induced tree and forest mortality. New Phytol 218(1):15–28.https://doi.org/10.1111/nph.15048
Heinselman ML (1981) Fire and succession in the conifer forest of northern North America. In: West DC, Shugart HH, Botkin DB (eds) Forest succession: concepts and application. Springer, New York, pp 374–405
IPCC (2014) Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva
IPCC (2017) IPCC Fifth Assessment Report (AR5) Observed Climate Change Impacts Database, Version 2.01. Palisades, NY: NASA Socioeconomic Data and Applications Center (SEDAC). https://doi.org/10.7927/H4FT8J0X
Klein T, Hemming D, Lin T, Grünzweig JM, Maseyk K, Rotenberg E, Yakir D (2005) Association between tree-ring and needle δ 13 C and leaf gas exchange in pinus halepensis under semi-arid conditions. Oecologia 144(1):45–54. https://doi.org/10.1007/s00442-005-0002-y
Konter O, Holzkämper S, Helle G, Büntgen U, Saurer M, Esper J (2014) Climate sensitivity and parameter coherency in annually resolved δ13C and δ18O from pinus uncinata tree-ring data in the spanish pyrenees. Chem Geol 377:12–19. https://doi.org/10.1016/j.chemgeo.2014.03.021
Kohn MJ (2010) Carbon isotope compositions of terrestrial C3 plants as indicators of (paleo)ecology and (paleo)climate. Proc Natl Acad Sci 107(46):19691–19695. https://doi.org/10.1073/pnas.1004933107
Kolb KJ, Evans RD (2002) Implications of leaf nitrogen recycling on the nitrogen isotope composition of deciduous plant tissues. New Phytol 156:57–64
Larcher W (2000) Temperature stress and survival ability of Mediterranean sclerophyllous plants. Plant Biosyst 134:279–295
Liñán ID, Gutiérrez E, Andreu-Hayles L, Heinrich I, Helle G (2012) Potential to explain climate from tree rings in the south of the iberian peninsula. Climate Res 55(2):119–134. https://doi.org/10.3354/cr01126
Linares JC, Delgado-Huertas A, Camarero JJ, Merino J, Carreira JA (2009) Competition and drought limit the response of water-use efficiency to rising atmospheric carbon dioxide in the mediterranean fir Abies pinsapo. Oecologia 161(3):611–624. https://doi.org/10.1007/s00442-009-1409-7
Linares JC, Delgado-Huertas A, Carreira JA (2011) Climatic trends and different drought adaptive capacity and vulnerability in a mixed abies pinsapo-pinus halepensis forest. Clim Change 105(1):67–90. https://doi.org/10.1007/s10584-010-9878-6
Lloret F, Casanovas C, Peñuelas J (1999) Seedling survival of Mediterranean shrubland species in relation to root: shoot ratio, seed size and water and nitrogen use. Funct Ecol 13:210–216
Lo Gullo MA, Salleo S (1988) Different strategies of drought resistance in three Mediterranean sclerophyllous trees growing in the same environmental conditions. New Phytol (cambridge, Gran Bretagna) 108:267–276
Margaris NS, Papadogianni P (1977) Cambial activity in some plants dominating phryganic formations in Greece. Phyton (greece) 36:1–5
Martinez-Meier A, Sanchez L, Pastorino M, Gallo L, Rozenberg P (2008) What is hot in tree rings? The wood density of surviving Douglas-firs to the 2003 drought and heat wave. For Ecol Manage 256(4):837–884
Martínez-Sancho E, Dorado-Liñán I, Gutiérrez Merino E, Matiu M, Helle G, Heinrich I, Menzel A (2018) Increased water-use efficiency translates into contrasting growth patterns of scots pine and sessile oak at their southern distribution limits. Glob Change Biol 24(3):1012–1028. https://doi.org/10.1111/gcb.13937
Masle J, Farquhar GD (1988) Effects of soil strength on the relation of water-use efficiency and growth to carbon isotope discrimination in wheat seedlings. Plant Physiol 86:32–38
Maseyk K, Hemming D, Angert A, Leavitt SW, Yakir D (2011) Increase in water-use efficiency and underlying processes in pine forests across a precipitation gradient in the dry Mediterranean region over the past 30 years. Oecologia 167(2):573–585
McCarroll D, Loader NJ (2004) Stable isotope is tree rings. Quatern Sci Rev 23:771–801
McDowell NG, Sevanto S (2010) The mechanisms of carbon starvation: how, when, or does it even occur at all? New Phytol 186:264–266
McDowell NG, Beerling D, Breshears D, Fisher R, Raffa K, Stitt M (2011) Interdependence of mechanisms underlying climate-driven vegetation mortality. Trends Ecol Evol 26:523–532
McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Sperry J, West A, Williams D, Yepez EA (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? Tansley review. New Phytol 178:719–739
McDowell NG, Bond BJ, Dickman LT, Ryan MG, Whitehead D (2011) Relationships between tree height and carbon isotope discrimination. In: Meinzer FC et al (eds) Size- and age-related changes in tree structure and function. Springer, New York, NY, USA, pp 255–285
Meinzer FC, McCulloh KA, Lachenbruch B, Woodruff DR, Johnson DM (2010) The blind men and the elephant: the impact of context and scale in evaluating conflicts between plant hydrau- lic safety and efficiency. Oecologia 164:287–296
Médail F (2008) Ecosystems: Mediterranean. In: Jørgensen SE, Fath B (eds) Encyclopedia of ecology, vols 3, 5. Elsevier, Oxford, pp 2296–2308
Médail F, Monnet AC, Pavon D, Nikolic T, Dimopoulos P, Bacchetta G, Arroyo J, Barina Z, Albassatneh MC, Domina G, Fady B, Matevski V, Mifsud S, Leriche A (2019) What is a tree in the Mediterranean basin hotspot? A critical analysis. For Ecosyst 6 (2019):17. https://doi.org/10.1186/s40663-019-0170-6
Medrano H, Flexas J, Galmés J (2009) Variability in water use efficiency at the leaf level among Mediterranean plants with different growth forms. Plant Soil 317(1–2):17–29. https://doi.org/10.1007/s11104-008-9785-z
Michelsen AC, Quarmby C, Sleep D, Jonasson S (1998) Vascular plant 15N abundance in heath and forest tundra ecosystems is closely correlated with presence and type of mycorrhizal fungi in roots. Oecologia 115:406–418
Mitrakos KA (1980) A theory for Mediterranean plant life. Acta Oecol 1:245–252
Moreno-Gutiérrez C, Battipaglia G, Cherubini P, Saurer M, Nicolás E, Contreras S, Querejeta JI (2012a) Stand structure modulates the long-term vulnerability of pinus halepensis to climatic drought in a semiarid mediterranean ecosystem. Plant, Cell Environ 35(6):1026–1039. https://doi.org/10.1111/j.1365-3040.2011.02469.x
Moreno-Gutierrez C, Dawson TE, Nicol_as E, & Querejeta JI (2012b) Isotopes reveal contrasting water use strategies among coexisting plant species in a Mediterranean ecosystem. New Phytol 196:489–496
Moreno-Gutiérrez C, Battipaglia G, Cherubini P, Delgado Huertas A, Querejeta JI (2015) Pine afforestation decreases the long-term performance of understorey shrubs in a semi-arid mediterranean ecosystem: a stable isotope approach. Funct Ecol 29(1):15–25. https://doi.org/10.1111/1365-2435.12311
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nadelhoffer KJ, Fry B (1994) Nitrogen isotope studies in forest ecosystems. In: Lajtha K, Michener RH (eds) Stable isotopes in ecology and environmental science. Blackwell, Oxford, pp 22–44
Niccoli F, Esposito A, Altieri S, Battipaglia G (2019) Fire severity influences ecophysiological responses of Pinus pinaster ait. Front Plant Sci 10:539. https://doi.org/10.3389/fpls.2019.00539
Noy-Meir I (1973) Desert ecosystems: environments and producers. Annu Rev Ecol Syst 4:25–51
Pacheco A, Camarero JJ, Carrer M (2016) Linking wood anatomy and xylogenesis allows pinpointing of climate and drought influences on growth of coexisting conifers in continen- tal Mediterranean climate. Tree Physiol 36:502–512. https://doi.org/10.1093/treephys/tpv125
Pallardy SG, Čermák J, Ewers FW, Kaufmann MR, Parker WC, Sperry JS (1995) Water transport dynamics in trees and stands. In: Smith TM, Hinckley TM (eds) Resource physiology of conifers. Academic Press, San Diego, p 301
Paris P, Di Matteo G, Tarchi M, Tosi L, Spaccino L, Lauteri M (2018) Precision subsurface drip irrigation increases yield while sustaining water-use efficiency in mediterranean poplar bioenergy plantations. Forest Ecol Manage 409:749–756. https://doi.org/10.1016/j.foreco.2017.12.013
Peñuelas J, Estiarte M (1996) Trends in plant carbon concentration and plant demand for N throughout this century. Oecologia 109(1):69–73. https://doi.org/10.1007/s004420050059
Peñuelas J, Canadell JG, Ogaya R (2011) Increased water-use efficiency during the 20th century did not translate into enhanced tree growth. Glob Ecol Biogeogr 20(4):597–608. https://doi.org/10.1111/j.1466-8238.2010.00608.x
Quézel P, Médail F (2003) Ecologie et biogéographie des forêts du bassin méditerranéen. Elsevier, Paris
Rigling A, Waldner PO, Forster T, Bräker OU, Pouttu A (2001) Ecological interpretation of tree-ring width and intraannual density fluctuations in pinus sylvestris on dry sites in the central alps and Siberia. Can J Forest Res 31(1):18–31. https://doi.org/10.1139/x00-126
Ripullone F, Guerrieri MR, Saurer M, Siegwolf RTW, Jäggi M, Guarini R, Magnani F (2009) Testing a dual isotope model to track carbon and water gas exchanges in a Mediterranean forest. iForest 2:59–66
Rivas-Martínez S, Asensi A, Garretas B, Valle F, Cano E, Costa M, Luisa López M, Díaz T, Fernández Prieto JA, Llorens L, Del Arco AM, Osorio VE, Pérez L, de Paz P, Wildpret W, Reyes-Betancort J, García Gallo A, Rodríguez O, Acebes J, Gaisberg M, Soriano P (2007) Mapa de series, geoseries y geo-permaseries de vegetazión de España. Itinera Geobot 17:5–436
Roden JS, Siegwolf R (2012) Is the dual isotope conceptual model fully operational? Tree Physiol 32:1179–1182
Roden JS, Farquhar GD (2012) A controlled test of the dual-isotope approach for the interpretation of stable carbon and oxygen isotope ratio variation in tree rings. Tree Physiol 32:490–503
Rowland L, da Costa ACL, Galbraith DR, Oliveira RS, Binks OJ, Oliveira AAR, Pullen AM, Doughty CE, Metcalfe DB, Vasconcelos SS, Ferreira LV, Meir P (2015) Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature.https://doi.org/10.1038/nature15539
Sala A (2009) Lack of direct evidence for the carbon-starvation hypothesis to explain drought-induced mortality in trees. Proc Natl Acad Sci United States of Am 106(26). https://doi.org/10.1073/pnas.0904580106
Sala A, Piper F, Hoch G (2010) Physiological mechanisms of drought-induced tree mortality are far from being resolved. New Phytol 186(2):274–281. https://doi.org/10.1111/j.1469-8137.2009.03167.x
Salleo S, Nardini A, Lo Gullo MA (1997) Is sclerophylly of Mediterranean evergreens an adaptation to drought? New Phytol 135:603–612
Salmon Y, Torres-Ruiz JM, Poyatos R, Martinez-Vilalta J, Meir P, Cochard H, Mencuccini M (2015) Balancing the risks of hydraulic failure and carbon starvation: a twig scale analysis in declining Scots pine. Plant, Cell Environ 38:2575–2588
Sánchez-Costa E, Poyatos R, Sabaté S, (2015) Contrasting growth and water use strategies in four co-occurring Mediterranean tree species revealed by concurrent measurements of sap flow and stem diameter variations. Agric for Meteorol 207:24–37. https://doi.org/10.1016/j.agrformet.2015.03.012
Sargeant CI, Singer MB (2016) Sub-annual variability in historical water source use by mediterranean riparian trees. Ecohydrology 9(7):1328–1345. https://doi.org/10.1002/eco.1730
Sarris D, Siegwolf R, Körner C (2013) Inter- and intra-annual stable carbon and oxygen isotope signals in response to drought in Mediterranean pines. Agric Forest Meteorol 168:59–68. https://doi.org/10.1016/j.agrformet.2012.08.007
Saurer M, Robertson I, Siegwolf R, Leuenberger M (1998) Oxygen isotope analysis of cellulose: an interlaboratory comparison. Anal Chem 70(10):2074–2080. https://doi.org/10.1021/ac971022f
Scheidegger Y, Saurer M, Bahn M, Siegwolf R (2000) Linking stable oxygen and carbon isotopes with stomatal conductance and photosynthetic capacity: a conceptual model. Oecologia 125:350–357. https://doi.org/10.1007/s004420000466
Schulman E (1938) Classification of false annual rings in Monterey pine. Tree-Ring Bull 4:4–7
Sevanto S, McDowell NG, Dickman LT, Pangle R, Pockman WT (2014) How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Environ 37:153–151
Shestakova TA, Aguilera M, Ferrio JP, Gutiérrez E, Voltas J (2014) Unravelling spatiotemporal tree-ring signals in mediterranean oaks: a variance-covariance modelling approach of carbon and oxygen isotope ratios. Tree Physiol 34(8):819–838. https://doi.org/10.1093/treephys/tpu037
Shestakova TA, Camarero JJ, Ferrio JP, Knorre AA, Gutiérrez E, Voltas J (2017) Increasing drought effects on five european pines modulate Δ13C-growth coupling along a Mediterranean altitudinal gradient. Funct Ecol 31(7):1359–1370. https://doi.org/10.1111/1365-2435.12857
Shestakova TA, Voltas J, Saurer M, Berninger F, Esper J, Andreu-Hayles L, Gutiérrez E et al (2019) Spatio-temporal patterns of tree growth as related to carbon isotope fractionation in european forests under changing climate. Glob Ecol Biogeogr. https://doi.org/10.1111/geb.12933
Silva LCR, Horwath WR (2013) Explaining global increases in water use efficiency: why have we overestimated responses to rising atmospheric CO2 in natural forest ecosystems? PLoS ONE 8(1). https://doi.org/10.1371/journal.pone.0053089
Singer MB, Stella JC, Dufour S, Piégay H, Wilson RJS, Johnstone L (2013) Contrasting water-uptake and growth responses to drought in co-occurring riparian tree species. Ecohydrology 6(3):402–412. https://doi.org/10.1002/eco.1283
Sperry JS, Adler FR, Campbell GS, Comstock JP (1998) Limitation of plant water use by rhizosphere and xylem con- ductance: results from model. Plant, Cell Environ 21:347–359
Sperry JS, Meinzer FC, McCulloh KA (2008) Safety and efficiency conflicts in hydraulic architecture: scaling from tissues to trees. Plant Cell Environ 31:632–645
Stemberg L, Swart PK (1987) Utilization of fresh- water and ocean water by coastal plants of southern Florida. Ecology 68:1898–1905
Stewart D, Barnes J, Cote J, Cudeck R, Malthouse E (2001) Factor analysis. J Consum Psychol 10(1–2):75–82. https://doi.org/10.1207/s15327663jcp1001&2_07
Szymczak S, Joachimski MM, Bräuning A, Hetzer T, Kuhlemann J (2011) Comparison of whole wood and cellulose carbon and oxygen isotope series from Pinus nigra ssp. laricio (Corsica/France). Dendrochronologia 29(4):219–226. https://doi.org/10.1016/j.dendro.2011.04.001
Szymczak S, Joachimski MM, Bräuning A, Hetzer T, Kuhlemann J (2012a) A 560 yr summer temperature reconstruction for the western mediterranean basin based on stable carbon isotopes from pinus nigra ssp. laricio (Corsica/France). Climate of the Past 8(5):1737–1749. https://doi.org/10.5194/cp-8-1737-2012
Szymczak S, Joachimski MM, Bräuning A, Hetzer T, Kuhlemann J (2012b) Are pooled tree ring δ13C and δ18O series reliable climate archives?—a case study of Pinus nigra spp. laricio (Corsica/France). Chem Geol 308–309:40–49. https://doi.org/10.1016/j.chemgeo.2012.03.013
Szymczak S, Hetzer T, Bräuning A, Joachimski MM, Leuschner H, Kuhlemann J (2014) Combining wood anatomy and stable isotope variations in a 600-year multi-parameter climate reconstruction from Corsican black pine. Quatern Sci Rev 101:146–158. https://doi.org/10.1016/j.quascirev.2014.07.010
Terradas J, Save R (1992) The influence of summer and winter stress and water relationships on the distribution of Quercus ilex L. Vegetatio 99(100):137–145
Tingley MA (1937) Double growth rings in Red Astrachan. Proc Am Soc Horticultural Sci 34:61
Valor T, Casals P, Altieri S, González-Olabarria JR, Piqué M, Battipaglia G (2018) Disentangling the effects of crown scorch and competition release on the physiological and growth response of Pinus halepensis Mill. using δ13C and δ18O isotopes. For Ecol Manag 424:276–287. https://doi.org/10.1016/j.foreco.2018.04.056
Vieira J, Nabais C, Rossi S, Carvalho A, Freitas H, Campelo F (2017) Rain exclusion affects cambial activity in adult maritime pines. Agric for Meteorol 237:303–310. https://doi.org/10.1016/j.agrformet.2017.02.024
Voelker SL, Meinzer FC, Lachenbruch B, Brooks JR, Guyette RP (2014) Drivers of radial growth and carbon isotope discrimination of bur oak (Quercus macrocarpa michx.) across continental gradients in precipitation, vapour pressure deficit and irradiance. Plant, Cell Environment 37(3):766–779. https://doi.org/10.1111/pce.12196
Voltas J, Camarero JJ, Carulla D, Aguilera M, Oriz A, Ferrio JP (2013) A retrospective, dual-isotope approach reveals individual predispositions to winter-drought induced tree dieback in the southernmost distribution limit of Scots pine. Plant, Cell Environ 36:1435–1448
Warren CR, Adams MA (2000) Water availability and branch length determineδ13C in foliage of Pinus pinaster Tree Physiol 10:637–644
Wershaw RL, Friedman I, Heller SJ, Frank PA (1966) Hydrogen isotope fraction- ation of water passing through trees. In: Hobson GD (ed) Advances in organic geochemistry. Pergamon Press, New York, pp 55–67
Wu J, Zhang R, Yang J (1996) Analysis of rainfall-recharge relationships. J Hydrol 177:143–160
Yakir D, da Sternberg L, S. L. (2000) The use of stable isotopes to study ecosystem gas exchange. Oecologia 123(3):297–311. https://doi.org/10.1007/s004420051016
Zalloni E, Battipaglia G, Cherubini P, Saurer M, & De Micco V (2018) Contrasting physiological responses to mediterranean climate variability are revealed by intra-annual density fluctuations in tree rings of Quercus ilex L. and Pinus pinea L. and. Tree Physiol 38(8):1213–1224. https://doi.org/10.1093/treephys/tpy061
Zalloni E, Battipaglia G, Cherubini P, Saurer M, De Micco V (2019) Wood growth in pure and mixed Quercus ilex l. forests: drought influence depends on site conditions. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00397
Acknowledgements
The authors thank Francesco Niccoli for helping with Figure 21.1 and Erin Gleeson for English editing.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 © The Author(s)
About this chapter
Cite this chapter
Battipaglia, G., Cherubini, P. (2022). Stable Isotopes in Tree Rings of Mediterranean Forests. In: Siegwolf, R.T.W., Brooks, J.R., Roden, J., Saurer, M. (eds) Stable Isotopes in Tree Rings. Tree Physiology, vol 8. Springer, Cham. https://doi.org/10.1007/978-3-030-92698-4_21
Download citation
DOI: https://doi.org/10.1007/978-3-030-92698-4_21
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92697-7
Online ISBN: 978-3-030-92698-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)