Abstract
The human circadian system drives 24-h rhythms in all aspects of biochemistry, physiology and behaviour. This rhythmicity ensures that the right function occurs at the right time, and separates incompatible functions. Rhythmic synchronisation is key to overall health and wellbeing, and light input from the day-night cycle is the primary ‘zeitgeber’ or entraining agent. Coordination of internal clocks promotes positive mood, optimum alertness and performance, and ensures sleep quality essential for learning and memory. Indoor light intensity is rarely sufficient to stabilise daily entrainment, while even an hour outdoors in the morning can significantly enhance cognitive function, academic achievement, and physical condition.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Day-night cycle
- Light as major synchronising agent (zeitgeber)
- Circadian rhythms
- Importance of sleep
- Light and learning
- Light and mood
1 Introduction
Outdoor-based learning: an interesting new concept for a chronobiologist. But immediately I recognised the relevance of the field of biological rhythms to the understanding of a basic factor of the outdoor environment, taken for granted yet not completely analysed: daylight. Here a short biology lesson will show why outdoor light can be crucial for high quality learning, via neural mechanisms that control a broad range of behaviour, physiology and endocrine function, and support stable mood and alertness during the day and consolidated sleep at night.
2 Daylight
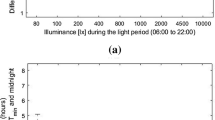
Let us begin with detailing the parameter of outdoor daylight availability. Daylight follows a predictable pattern of light intensity and spectral changes day by day throughout the year, with twilight transitions of dawn and dusk. The daily and annual pattern is specific for a given geographic location, and Fig. 1 shows the complexity of daylight at a latitude of 50°N (e.g. Champagne in France).
It is extraordinary that the human eye can register this enormous range of light intensity from a starlight minimum of 0.0003 lx to sun overhead at ca. 100,000 lx (Fig. 2). Subjectively, we experience the brightness of skylight compressed on a log scale, but our physiology tracks the signal in exquisite detail.
Daylight is the primary geophysical signal to which all life on earth has evolved. Chronobiology is the science of daily (circadian), monthly, tidal, and seasonal rhythms that are related to the regular and predictable movements of sun and moon (Hastings et al., 2008). Internal temporal organisation ensures that the right function occurs at the right time (and separates incompatible functions)—within individuals, between people, and between species. The most obvious rhythms are those linked to the 24-h day-night cycle (Cajochen et al., 2010; Hastings et al., 2008). These circadian rhythms range from behavioural patterns in the general population (more accidents in the second half of the night) to the individual (the daily sleep-wake cycle and core body temperature), as well as metabolic rhythms that occur in each organ and cell. Circadian rhythms are not merely a response to the environmental time cues of light and dark, they are endogenous, that is, they are driven by molecular clock genes that tick at a frequency ~24 h (Hastings et al., 2008). Nearly all living organisms—from bacteria to plants, insects and mammals—have evolved astonishingly similar molecular timing systems (Dunlap & Loros, 2017): they guide plants to time photosynthesis, allow monarch butterflies to navigate thousands of miles, cue hamsters to hibernate, and maintain health status in humans—as long as they live in sync with their biological clocks.
3 The Human Circadian System
Within the biological clock, which lies in the hypothalamic suprachiasmatic nuclei (SCN), a clock gene network encodes circadian periodicity of approximately, but not exactly, 24 h (Hastings et al., 2008). The circadian system in the brain needs to receive information about the consistent external day-night cycle (Cajochen et al., 2010), and light is the major synchronising agent (or ‘zeitgeber’). Photic input from the eyes to the SCN is transduced via specialised circadian photoreceptors called intrinsic photosensitive retinal ganglion cells (ipRGCs), which contain a blue-wavelength-sensitive photopigment, melanopsin (Hankins et al., 2008); these transmit the signal directly from the retina to the brain via the retinohypothalamic tract (Fig. 3).
The classical cone photoreceptors, which mediate daytime vision—color, movement, shape, and edges—and rod photoreceptors, which enable us to see in dim light and near-darkness, have secondary interactions with the non-visual ipRGC system (Hankins et al., 2008). Neuronal firing patterns in the SCN convey the day-night information to many regions of the brain, driving day-night rhythms in biochemistry, physiology, and behaviour. The variety of non-visual functions directly influenced by this pathway ranges from pupil size, pineal hormone melatonin secretion, mood, vigilance, and cognitive performance, to sleep and learning.
4 Light as Zeitgeber
Of prime importance is the characteristic of light as a zeitgeber to shift circadian rhythms earlier or later depending on time of day of exposure (Minors et al., 1991; Roenneberg et al., 2013). Morning light advances the clock earlier; while evening light delays the clock (Minors et al., 1991). Additionally, regular light exposure synchronises and stabilises rhythms: all the cellular and organ clocks depend on daily zeitgeber exposure for good entrainment (Van Someren & Riemersma-Van Der Lek, 2007).
This critical role of light as zeitgeber for humans was discovered using light intensity much higher (1000 lx) than indoors (10–300 lx) (Lewy et al., 1980). In later experiments circadian responses have been observed at lower light intensities, depending on prior light exposure, duration and spectrum of the light source, and with large interindividual variability (Boivin et al., 1994; Danilenko et al., 2000; Phillips et al., 2019). In contrast, clinical applications of light use 10,000 lx (Terman & Terman, 2010), well within the range of full daylight.
Thus, there is a specific range of light intensities that affects human rhythms. In order to maintain stable entrainment—which is not only a prerequisite for good night-time sleep and daytime alertness, but also for mood state, cognition, and neurobehavioural function—a minimum of 1000 lx for 30–60 min per day is considered adequate.
In addition to light, other zeitgebers such as physical exercise (Lewis et al., 2018), mealtimes (Lewis et al., 2020), and to a lesser extent, sleep itself (Danilenko et al., 2003), contribute to entrainment. Social signals (such jobs or school, alarm clocks, social demands) were originally considered the main zeitgebers for humans (Mistlberger & Skene, 2004), but they are now understood to act indirectly on the SCN: they determine the timing and structure of daily habits and thus the pattern and level of exposure to outdoor and indoor light.
5 Seasonality
With the invention of artificial light, humans were able to choose their sleep-wake schedules and lifestyle. In today’s 24/7 society, we no longer follow daylight duration across seasons. Rather, we have come to live under artificially designed constant day length that might be called, functionally, biological darkness. This is because we spend most of the day indoors, where room light—though adequate for vision—is insufficient to regulate our circadian rhythms.
Does seasonality still exist in humans, and if so, what would that mean? Humans do retain neurobiological responses to seasonal changes in daylength, though overt seasonal behaviour is rarely seen (Wirz-Justice, 2018). Reduced exposure to sunlight during the day with more artificial light at night leads to late circadian and sleep timing throughout the year. Natural daylight exposure (e.g. camping) in both summer and winter has been shown to rapidly entrain the biological clock to sunset and sunrise, with earlier timing compared with urban life (Stothard et al., 2017), demonstrating that we are still sensitive to these environmental cues.
Seasonal affective disorder, or winter depression, is an example of vulnerability to shorter daylength in winter. The standard application to treat winter depression is with a light box providing 10′000 lx white light for half-an-hour every morning (Terman & Terman, 2010).
6 Chronotype
Each one of us has an inborn preference to go to bed and wake up at a particular time, when we have the freedom to do so (usually only on holidays). This is called ‘chronotype’ (Horne & Ostberg, 1977), determined primarily by each individual’s clock genes. The well-known description of extreme chronotypes are the early ‘larks’ and late ‘owls’, whereas the majority can be considered ‘doves’ with ‘normal’ sleep timing (Fig. 4).
Although our biology determines chronotype, there are shifts over the life span, probably related to hormonal changes (Roenneberg et al., 2004). Children are mostly larks until puberty, when their sleep timing shifts later and later until about the age of 20. Thereafter sleep shifts slowly earlier until older persons are larks again, with early morning awakening. The discrepancy between internal time and social demands has been labeled ‘social jet lag’ (Wittmann et al., 2006).
Adolescents suffer the most. They have difficulties waking in the morning—thus the timing of classes should be later than most schools allow—and they cannot fall asleep easily (Marx et al., 2017). This leads to alarm-clock-induced shortening of sleep duration, which becomes chronic. Add to that the habit of late-night-use of iPhones and computers with bright white screens that contain a strong blue component which directly stimulates the blue-sensitive ipRGCs in the retina. Such use shifts the clock even later, thereby making falling asleep more difficult (Green et al., 2017). Morning light exposure is therefore extremely important in this age group in order to reset sleep timing earlier (a challenging exercise, but doable). Studies have looked at the effects of indoor bright light to increase students’ alertness in the morning (e.g. Hansen et al., 2005), but of course the simplest solution would be to spend the first lesson of the day outdoors, where daylight provides the natural and sufficient stimulus to wake up the clock (Martinez-Nicolas et al., 2011).
7 Out of Sync
There is growing evidence for long-term health consequences of irregular, inadequate, and poorly timed light-dark cycles that disrupt sleep (Medic et al., 2017; Van Someren & Riemersma-Van Der Lek, 2007; Wirz-Justice et al., 2009). With insufficient daily light exposure—often the case if remaining indoors—our body clocks may be de-synchronised with the day-night cycle (Van Someren & Riemersma-Van Der Lek, 2007). This is most clearly experienced with shift work or transmeridian flight.
No longer being synchronised to the naturalistic dawn–dusk signal can contribute to vulnerability for mood and sleep disorders, and perpetuate or exacerbate a wide variety of clinical symptoms (Van Someren & Riemersma-Van Der Lek, 2007). For example, disrupted and poorly timed sleep has been linked to obesity and development of diabetes (Buxton et al., 2012; Potter et al., 2016). Thus students should learn how to recognise their chronotype (see automated questionnaire on https://chronotype-self-test.info, accessed 13/04/2022), be aware that social jet lag can have profound effects on emotions, performance, and alertness, and use the knowledge about daylight as a synchronising agent to improve their sleep-wake habits. Teachers also should recognise their students’ chronotypes and not blame delayed sleep in adolescents on laziness or rebelliousness—it’s biology! An owl will not perform well early in the day, whether training in the swimming pool or solving algebraic equations.
8 Too Much Light?
Of course, daylight has many effects beyond those related to circadian rhythms, sleep and mood (Wirz-Justice et al., 2020). It is required for Vitamin D synthesis via the skin, and for eye development in childhood (preventing myopia). In contrast, excessive ultraviolet exposure from sunlight is responsible for many skin cancers. An intelligent balance must be sought.
Blue-enhanced bright light sources in group settings such as classrooms and offices may not always be preferred given that some individuals are abnormally light sensitive, and may react with nausea, glare, and headache.
9 Light Outdoors
How then are my observations as a chronobiologist relevant to the advocacy of outdoor learning? Irregular sleep-wake cycles are associated with poorer academic performance and learning (Phillips et al., 2017; Wright et al., 2006). Regular and sufficient daylight exposure has positive effects on the quality and quantity of nighttime sleep. Indoor light lacks the intensity to do this. Conversely, one has to ensure darkness in the bedroom to support stable and deep sleep.
There is some evidence that higher light exposure improves learning, but most of the data are indirect, as reflected by increased alertness, performance, faster reaction time, and memory recall (e.g. Huiberts et al., 2015; Yoshiike et al., 2019). A far larger body of evidence supports the importance of sleep for learning new skills and motor performance. The quantity and quality of sleep affect a person’s memory consolidation, since sleep is a period where the brain turns recent experience into long-term memories (e.g. Ellenbogen et al., 2006; Fattinger et al., 2017; MacDonald & Cote, 2021; Walker, 2009). Finally, sleep plays an important role in brain plasticity as the young human brain develops (Dang-Vu et al., 2006; Kurth et al., 2012).
10 Conclusion
Many physiological and psychological functions are profoundly affected by daylight (and artificial indoor surrogates). All efforts to improve entrainment of rhythms with morning light will also improve alertness and performance during the day and sleep during the night. Good sleep is crucial in the educational environment since it impacts memory, motor skills, learning, mood, behaviour, immunological responses, metabolism, hormone levels, digestive processes, and more.
The growing recognition that sufficient light is important for psychological and somatic wellbeing is leading toward novel lighting solutions in architecture as well as more conscious exposure to natural daylight. In short, sufficient daily light exposure can support overall health, and the most natural and efficacious source thereof is the sun.
Recommended Further Reading
-
1.
Center for Environmental Therapeutics www.cet.org
-
2.
The Daylight Academy www.daylight.academy
-
3.
Society for Light Treatment and Biological Rhythms https://sltbr.org/ (all accessed 13/04/2022)
References
Boivin, D. B., Duffy, J. F., Kronauer, R. E., & Czeisler, C. A. (1994). Sensitivity of the human circadian pacemaker to moderately bright light. Journal of Biological Rhythms, 9, 315–331.
Buxton, O. M., Cain, S. W., O’Connor, S. P., Porter, J. H., Duffy, J. F., Wang, W., Czeisler, C. A., & Shea, S. A. (2012). Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Science Translational Medicine, 4, 129ra43.
Cajochen, C., Chellappa, S., & Schmidt, C. (2010). What keeps us awake?—the role of clocks and hourglasses, light, and melatonin. International Review of Neurobiology, 93, 57–90.
Dang-Vu, T. T., Desseilles, M., Peigneux, P., & Maquet, P. (2006). A role for sleep in brain plasticity. Pediatric Rehabilitation, 9, 98–118.
Danilenko, K. V., Cajochen, C., & Wirz-Justice, A. (2003). Is sleep per se a zeitgeber in humans? Journal of Biological Rhythms, 18, 170–178.
Danilenko, K. V., Wirz-Justice, A., Kräuchi, K., Weber, J. M., & Terman, M. (2000). The human circadian pacemaker can see by the dawn’s early light. Journal of Biological Rhythms, 15, 437–446.
Dunlap, J. C., & Loros, J. J. (2017). Making time: Conservation of biological clocks from fungi to animals. Microbiology Spectrum, 5 (3). https://doi.org/10.1128/microbiolspec.FUNK-0039-2016.
Ellenbogen, J. M., Payne, J. D., & Stickgold, R. (2006). The role of sleep in declarative memory consolidation: Passive, permissive, active or none? Current Opinion in Neurobiology, 16, 716–722.
Fattinger, S., de Beukelaar, T. T., Ruddy, K. L., Volk, C., Heyse, N. C., Herbst, J. A., Hahnloser, R. H. R., Wenderoth, N., & Huber, R. (2017). Deep sleep maintains learning efficiency of the human brain. Nature Communications, 8, 15405.
Green, A., Cohen-Zion, M., Haim, A., & Dagan, Y. (2017). Evening light exposure to computer screens disrupts human sleep, biological rhythms, and attention abilities. Chronobiology International, 34, 855–865.
Hankins, M. W., Peirson, S. N., & Foster, R. G. (2008). Melanopsin: An exciting photopigment. Trends in Neurosciences, 31, 27–36.
Hansen, M., Janssen, I., Schiff, A., Zee, P. C., & Dubocovich, M. L. (2005). The impact of school daily schedule on adolescent sleep. Pediatrics, 115, 1555–1561.
Hastings, M. H., Maywood, E. S., & Reddy, A. B. (2008). Two decades of circadian time. Journal of Neuroendocrinology, 20, 812–819.
Horne, J. A., & Ostberg, O. (1977). Individual differences in human circadian rhythms. Biological Psychology, 5, 179–190.
Huiberts, L. M., Smolders, K. C. H. J., & de Kort, Y. A. W. (2015). Shining light on memory: Effects of bright light on working memory performance. Behavioural Brain Research, 294, 234–245.
Kurth, S., Ringli, M., Lebourgeois, M. K., Geiger, A., Buchmann, A., Jenni, O. G., & Huber, R. (2012). Mapping the electrophysiological marker of sleep depth reveals skill maturation in children and adolescents. NeuroImage, 63, 959–965.
Lewis, P., Korf, H. W., Kuffer, L., Gross, J. V., & Erren, T. C. (2018). Exercise time cues (zeitgebers) for human circadian systems can foster health and improve performance: A systematic review. BMJ Open Sport and Exercise Medicine, 4, e000443.
Lewis, P., Oster, H., Korf, H. W., Foster, R. G., & Erren, T. C. (2020). Food as a circadian time cue – evidence from human studies. Nature Reviews. Endocrinology, 16, 213–223.
Lewy, A. J., Wehr, T. A., Goodwin, F. K., Newsome, D. A., & Markey, S. P. (1980). Light suppresses melatonin secretion in humans. Science, 210, 1267–1269.
MacDonald, K. J., & Cote, K. A. (2021). Contributions of post-learning REM and NREM sleep to memory retrieval. Sleep Medicine Reviews, 59, 101453.
Martinez-Nicolas, A., Ortiz-Tudela, E., Madrid, J. A., & Rol, M. A. (2011). Crosstalk between environmental light and internal time in humans. Chronobiology International, 28, 617–629.
Marx, R., Tanner-Smith, E. E., Davison, C. M., Ufholz, L. A., Freeman, J., Shankar, R., Newton, L., Brown, R. S., Parpia, A. S., Cozma, I., & Hendrikx, S. (2017). Later school start times for supporting the education, health, and well-being of high school students. Cochrane Database of Systematic Reviews, 7, CD009467.
Medic, G., Wille, M., & Eh Hemels, M. (2017). Short- and long-term health consequences of sleep disruption. Nature and Science of Sleep, 9, 151–161.
Minors, D. S., Waterhouse, J. M., & Wirz-Justice, A. (1991). A human phase-response curve to light. Neuroscience Letters, 133, 36–40.
Mistlberger, R. E., & Skene, D. J. (2004). Social influences on mammalian circadian rhythms: Animal and human studies. Biological Reviews of the Cambridge Philosophical Society, 79, 533–556.
Phillips, A. J. K., Clerx, W. M., O’Brien, C. S., Sano, A., Barger, L. K., Picard, R. W., Lockley, S. W., Klerman, E. B., & Czeisler, C. A. (2017). Irregular sleep/wake patterns are associated with poorer academic performance and delayed circadian and sleep/wake timing. Scientific Reports, 7, 3216.
Phillips, A. J. K., Vidafar, P., Burns, A. C., McGlashan, E. M., Anderson, C., Rajaratnam, S. M. W., Lockley, S. W., & Cain, S. W. (2019). High sensitivity and interindividual variability in the response of the human circadian system to evening light. Proceedings of the National Academy of Sciences of the United States of America, 116, 12019–12024.
Potter, G. D. M., Skene, D. J., Arendt, J., Cade, J. E., Grant, P. J., & Hardie, L. J. (2016). Circadian rhythm and sleep disruption: Causes, metabolic consequences and countermeasures. Endocrine Reviews, 37, 584–608.
Roenneberg, T., Kantermann, T., Juda, M., Vetter, C., & Allebrandt, K. V. (2013). Light and the human circadian clock. Handbook of Experimental Pharmacology, 217, 311–331.
Roenneberg, T., Kuehnle, T., Pramstaller, P. P., Ricken, J., Havel, M., Guth, A., & Merrow, M. (2004). A marker for the end of adolescence. Current Biology, 14, R1038–R1039.
Stothard, E. R., McHill, A. W., Depner, C. M., Birks, B. R., Moehlman, T. M., Ritchie, H. K., Guzzetti, J. R., Chinoy, E. D., LeBourgeois, M. K., Axelsson, J., & Wright, K. P., Jr. (2017). Circadian entrainment to the natural light-dark cycle across seasons and the weekend. Current Biology, 27, 508–513.
Terman, M. & Terman, J. S. (2010). Light therapy. In M. H. Kryger, T. Roth & W. C. Dement (eds.), Principles and practice of sleep medicine (5th ed., pp. 1682–1695). Elsevier/Saunders.
Van Someren, E. J. W., & Riemersma-Van Der Lek, R. F. (2007). Live to the rhythm, slave to the rhythm. Sleep Medicine Reviews, 11, 465–484.
Walker, M. P. (2009). The role of slow wave sleep in memory processing. Journal of Clinical Sleep Medicine, 5, S20–S26.
Wirz-Justice, A. (2018). Seasonality in affective disorders. General and Comparative Endocrinology, 258, 244–249.
Wirz-Justice, A., Bromundt, V., & Cajochen, C. (2009). Circadian disruption and psychiatric disorders: The importance of entrainment. Sleep Medicine Clinics, 4, 273–284.
Wirz-Justice, A., Skene, D. J., & Münch, M. (2020). The relevance of daylight for humans. Biochemical Pharmacology, 114304.
Wittmann, M., Dinich, J., Merrow, M., & Roenneberg, T. (2006). Social jetlag: Misalignment of biological and social time. Chronobiology International, 23, 497–509.
Wright, K. P., Jr., Hull, J. T., Hughes, R. J., Ronda, J. M., & Czeisler, C. A. (2006). Sleep and wakefulness out of phase with internal biological time impairs learning in humans. Journal of Cognitive Neuroscience, 18, 508–521.
Yoshiike, T., Honma, M., Ikeda, H., & Kuriyama, K. (2019). Bright light exposure advances consolidation of motor skill accuracy in humans. Neurobiology of Learning and Memory, 166, 107084.
Acknowledgements
Thanks to Michael Terman, colleague and exigent editor, for much clarifying input.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Wirz-Justice, A. (2022). How Daylight Controls the Biological Clock, Organises Sleep, and Enhances Mood and Performance. In: Jucker, R., von Au, J. (eds) High-Quality Outdoor Learning. Springer, Cham. https://doi.org/10.1007/978-3-031-04108-2_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-04108-2_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-04107-5
Online ISBN: 978-3-031-04108-2
eBook Packages: Social SciencesSocial Sciences (R0)