Abstract
Cross-sectional imaging modalities including ultrasound (US), magnetic resonance (MR) imaging, and computed tomography (CT) play a critical role in the diagnosis of diseases of the biliary tract and gallbladder. Biliary imaging for evaluation of obstructive jaundice seeks to diagnose the level of obstruction and its cause. The identification of dilated bile ducts necessitates evaluation for strictures or filling defects, which is best performed with thin-section CT or T2-weighted MR cholangiography (MRCP). Choledocholithiasis with accompanying cholecystolithiasis is the most common cause of biliary obstruction and modern thin-section three dimensional MR cholangiography is highly sensitive and specific for the detection of stones. Recognition of gallbladder wall thickening and surrounding edema may be seen in acute cholecystitis, identification of bile duct wall thickening raises concern for cholangitis or malignancy. Primary sclerosing cholangitis and secondary sclerosing cholangitis such as recurrent pyogenic cholangitis, and IgG4 related sclerosing disease can develop chronic progressive cholestatic liver disease characterized by inflammation, fibrosis, and stricture of the bile ducts. Familiarity with the typical clinical and radiologic appearances of various etiologies of cholangiopathies and biliary malignancies is also important for accurate image interpretation.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
FormalPara Learning Objectives-
To discuss typical imaging features of common cholangiopathies.
-
To discuss the imaging diagnosis of premalignant tumors of the gallbladder and bile duct.
-
To explain classification systems of cholangiocarcinoma.
-
To discuss the strengths and weaknesses of imaging modalities in the diagnostic work-up of biliary malignancies.
-
Cross-sectional imaging of biliary disease often requires a multimodality imaging approach.
-
Sclerosing cholangitis represents a diseases spectrum disease of inflammation, fibrosis, and doctoral strictures, which can be classified as primary and secondary sclerosing cholangitis.
-
Although tissue biopsy needed for the definitive diagnosis of many of biliary strictures or mass -like lesions, certain imaging characteristics such as thickened wall, long-segment involvement, asymmetry, indistinct outer margin, luminal irregularity, hyperenhancement relative to the liver parenchyma may favor a malignant cause.
-
Biliary malignancies demonstrate arterial phase enhancement with persistent enhancement into the portal venous phase, due greater proportion of fibrotic tissue within these neoplasms.
8.1 Biliary Tract
The biliary tree is a common site of both benign diseases, such as choledocholithiasis or congenital malformations, and malignant diseases predominantly in the form of cholangiocarcinoma (CCA). Since benign diseases are often predisposing factors for the development of malignancy, especially if accompanied by chronic biliary inflammation, this chapter will emphasize benign entities that may require further long-term surveillance or surgical intervention. When repeated bouts of inflammation occur in the biliary tree, chronic injury leads to cellular proliferation and eventual survival of mutated cells and eventually bile duct neoplasia and malignancy [1]. While CCA can arise from epithelial cells anywhere along the biliary tree, they are most common at sites that harbor the highest density of peribiliary glands, which contain hepatic stem/progenitor cells (HPCs). These are located at branching points including the cystic duct, biliary confluence, and periampullary region [2, 3].
8.1.1 Normal Anatomy and Variants
The biliary tree is divided into intrahepatic and extrahepatic segments, with the right and left hepatic ducts usually draining the right and left hepatic lobes. The right and left ducts join at the biliary confluence, which forms the superior portion of the extrahepatic biliary tree. The common hepatic duct extends below the biliary confluence while the common bile duct starts below the insertion of the cystic duct. On imaging, when the insertion of the cystic duct is not visible, the extrahepatic duct is simply referred to as the common duct.
The right hepatic duct is usually formed as the confluence of the right anterior and right posterior ducts, which drain the anterior and posterior sectors respectively. The most common variant biliary anatomy consists of the posterior right hepatic duct draining into the left hepatic duct [4]. If this variant is not recognized, it can be led to bile duct injury for a living donor or during hepatic tumor resections. A biliary trifurcation is another common variant, with the posterior right, anterior right, and left hepatic duct joining at the confluence.
8.1.2 Congenital Biliary Anomalies
8.1.2.1 Choledochal Cysts and Anomalous Pancreatobiliary Ductal Junction
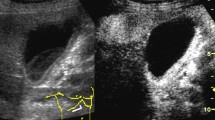
Choledochal cysts describe pathologic dilatations of the biliary tract and are classified by their anatomic location [5]. MRCP is the imaging modality of choice to identify choledochal cysts and the exact type of abnormality. The incidence of biliary malignancy, most commonly adenocarcinoma, varies with the type of choledochal cyst, with the highest among Todani Type I at 68%, followed by Type IV (21%), and below 10% for the remaining [6]. Surgical resection is thus indicated whenever possible, to reduce the risk of malignancy. Cholangiocarcinoma in the setting of a choledochal cyst presents as an intracystic soft tissue mass or irregular thickening of the cyst wall (Fig. 8.1).
Anomalous pancreaticobiliary ductal junction describes a congenital anomaly where the common bile duct and pancreatic duct join before the duodenal wall, leading to abnormal outflow of bile and pancreatic secretions. The abnormal flow of secretions leads to chronic inflammation of the biliary epithelium and a higher predisposition to bile duct malignancy [7]. The risk of malignancy from pancreaticobiliary maljunction varies with the presence or absence of biliary dilatation [8]. While choledochal cysts frequently coexist with a pancreaticobiliary maljunction, the latter is not always present.
8.1.3 Pathologic Conditions
8.1.3.1 Choledocholithiasis
Ultrasound is often used as a first line imaging modality for assessment of right upper quadrant pain, but its sensitivity for biliary stones is only 21–63%, due to challenges in obtaining an acoustic window, especially for the common bile duct [9]. CT is also commonly used for assessment of right upper quadrant pain, but its sensitivity for bile duct stones varies depending on their composition. Calcified gallstones are easiest to detect on CT (Fig. 8.2) while radiolucent stones are hard to distinguish from surrounding bile. Biliary stones are better detected by MR cholangiography, which has a very high sensitivity, especially for detection of stones in the common bile duct. On MRCP, stones are dark on T2 and have variable T1 signal, often appearing T1 hyperintense.
8.1.3.2 Cholangitis
8.1.3.2.1 Suppurative Cholangitis
Choledocholithiasis can lead to acute bacterial cholangitis, which is demonstrated on imaging by diffuse concentric wall thickening of the bile duct with associated periductal edema and mural enhancement. Arterial phase hyperenhancement of the adjacent parenchyma is often seen and reflects inflammation of the affected liver.
8.1.3.2.2 Pyogenic Cholangitis
In endemic areas, such as Southeast Asia, recurrent pyogenic cholangitis (RPC) is a manifestation of chronic parasitic infections in the bile ducts from parasites, including Clonorchis sinensis and Ascaris lumbricoides. The repeated bouts of cholangitis lead to multifocal strictures and superinfection with bacteria, and subsequent stone formation. A characteristic “arrowhead” appearance has been described, due to abrupt tapering of the peripheral ducts [10]. The risk of CCA is increased, given the chronic inflammatory milieu, and tends to arise in atrophic segments or ducts with heavy stone burden.
8.1.3.2.3 Primary Sclerosing Cholangitis
Primary sclerosing cholangitis (PSC) is an idiopathic and chronic inflammatory disease of the bile ducts, is presumed to be autoimmune and has a strong association with inflammatory bowel disease. On imaging, multifocal areas of bile duct narrowing are identified, with intervening normal or mildly dilated ducts, yielding an overall beaded appearance of the bile ducts [11]. Given the presence of chronic inflammation and high likelihood of developing bile duct malignancy, these patients are followed by MRCP to identify new or worsening irregular high-grade stricture. Because these malignancies are often periductal infiltrating CCA, imaging may not demonstrate an obvious mass, but focal biliary wall thickening is present. Intrahepatic mass-forming CCA can also occur in PSC, and the appearance and contrast to periductal infiltrating CCA is further described below.
8.1.3.3 IgG4 Cholangitis
IgG4 sclerosing cholangitis (SC) is commonly found in association with autoimmune pancreatitis (AIP), occurring in about 60–80% of patients with AIP [12]. In these cases, the most commonly involved segment is in the pancreatic head. IgG4 SC can also occur without concurrent AIP. On imaging, the bile duct can be focally or diffusely thickened, with upstream biliary dilatation (Fig. 8.3). PSC occurs more commonly in younger patients and is more often multifocal. When a single extrahepatic bile duct stricture is present, it can be challenging to distinguish from cholangiocarcinoma. Radiologists should seek concurrent pancreatic or extrapancreatic diseases to raise the possibility of IgG4 SC.
IgG4 sclerosing cholangitis. (a) A 60-year-old male with irregularly thickened extrahepatic bile duct on coronal CT with contrast. (b) 3D MRCP MIP image of narrowed common hepatic duct and moderate intrahepatic biliary dilatation. (c) Resolved bile duct wall thickening and biliary dilatation after treatment with steroids
8.1.3.4 Neoplasms of the Biliary System
8.1.3.4.1 Benign Tumors of the Bile Ducts
8.1.3.4.1.1 Hamartomas and adenomas
Biliary hamartomas, which are also known as von Meyenburg complexes, are benign tumor composed of disorganized bile ducts and ductules best seen on T2-weighted imaging as innumerable cystic appearing T2 hyperintense lesions throughout the liver, usually between 1 and 5 mm [13]. On CT, these are usually too small to characterize as cystic and can mimic metastatic disease. Bile duct adenomas are usually indistinguishable from hamartomas but are usually encountered as a solitary subcentimeter hypodense lesion.
8.1.3.4.1.2 Biliary Intraepithelial Neoplasm and Intraductal Papillary Neoplasms of the Bile Ducts
Chronic inflammation can lead to the appearance of biliary intraepithelial neoplasm (BilIN), a precursor lesion often found in association with RPC and PSC. It is also present in a majority of cases of CCA and is particularly common with extrahepatic CCA [14]. As a microscopic lesion, it is generally not seen on imaging. On the other hand, intraductal papillary neoplasms of the bile duct (IPNB) can appear as a macroscopic lesion on imaging, with variable appearance depending on the tumor growth, its location, and the degree of mucin production [8]. A papillary mass can be visible once the tumor grows to a sufficient size. On T2-weighted MRCP sequences, a papillary lesion appears dark compared to the surrounding T2 hyperintense bile; pre and post contrast imaging, however, shows clear enhancement, distinguishing these from biliary stones or sludge. When mucin production is high, the bile ducts may appear dilated either focally or throughout a segment, both above and below the level of the tumor. This is a distinguishing characteristic that is a result of excess mucin production. An alternative appearance of IPNB is the cystic form, due to focal aneurysmal dilatation of the involved bile duct, also due to excessive mucin production. IPNB can progress to an intraductal growing CCA, as described below [15].
8.1.3.4.1.3 Mucinous Cystic Neoplasms
According to WHO classification (5th edition), mucinous cystic neoplasms (MCNs), formerly referred to as biliary cystadenomas/cystadenocarcinomas, are cyst-forming epithelial neoplasms lined by cuboidal, columnar, or flattened mucin-producing epithelium overlying ovarian like-stroma (OLS), without biliary communication. The presence of OLS is the key distinguishing feature [16]. These tumors occur almost exclusively in mildly aged women. The vast majority of MCNs are benign, and malignant ones tend to occur in older patients. On imaging, they tend to by multilocular with associated septations and calcifications and are more common in the left hepatic lobe. On MRI, they are T2 hyperintense similar to cysts, but have more variable T1 internal signal due to the presence of hemorrhage or proteinaceous contents. While calcifications and mural nodules are associated with malignancy, imaging cannot reliably distinguish between from malignant MCNs, so these tumors are often resected when the diagnosis is suspected.
8.1.3.4.2 Malignant Tumors of the Bile Ducts
8.1.3.4.2.1 Cholangiocarcinoma
CCA are categorized by their location as either intrahepatic or extrahepatic, with the latter beginning at the biliary confluence. For radiologists, the morphologic classification as mass-forming, periductal infiltrating, or intraductal growing is more helpful because of their distinct imaging patterns on cross-sectional imaging.
Mass-forming CCA arise more commonly in patients with chronic hepatitis, especially those with cirrhosis and hepatitis B [17]. They form the majority of intrahepatic CCA and present as a lobulated mass, often with a targeted enhancement pattern, as defined by the American College of Radiology Liver Imaging Reporting and Data Systems. This pattern corresponds pathologically to the presence of a cellular periphery in the tumor that often show arterial phase hyperenhancement and washout, along with a fibrotic/desmoplastic center that shows delayed hyperenhancement (Fig. 8.4). While capsular retraction and peripheral biliary dilatation are distinguishing features of mass-forming CCA, these features are not always present [18]. Some mass-forming CCA are predominantly hypovascular and may overlap in appearance with metastatic disease from a gastrointestinal primary malignancy. A dominant liver mass with satellite lesions may help clue the radiologist to the possibility of a primary liver tumor.
Intrahepatic mass-forming cholangiocarcinoma. (a) A 78-year-old female with left intrahepatic cholangiocarcinoma on T1-weighted fat saturated imaging showing rim arterial phase hyperenhancement, a targeted imaging feature. (b) Persistent hyperenhancement on delayed hepatobiliary phase with capsular retraction along the anterior liver surface
Periductal infiltrating CCA are usually extrahepatic in location. They are referred to as Klatskin tumors if they involve the biliary confluence. The tumors cause biliary dilatation upstream above the level of biliary stricture, which is accompanied by focal bile duct wall thickening (Fig. 8.5). However, the tumors are often ill-defined and hard to delineate in their entirety, even with optimal CT and MR techniques. As a result, the degree of ductal involvement is often underestimated. For radiologists, reporting the degree of vascular involvement is just as critical, with contact of the hepatic arterial and portal venous anatomy often determining the likelihood of resectability. Vascular contact can be described as absent, abutment (up to 180°), or encasement (180° or more). A structured reporting form has been proposed by the Korean Society of Abdominal Radiology to describe relevant preoperative findings for CCA with the goal of future validation [19].
8.1.3.4.2.2 Metastatic Disease
Metastatic disease to the bile ducts is extremely rare, with colorectal cancer being more common than other cancers such as lung, breast, gallbladder, testicular, prostate, pancreas, melanoma, and lymphoma [20] (Fig. 8.6).
8.2 Gallbladder
8.2.1 Normal Anatomy
Being positioned along the undersurface of the liver in the plane of the interlobar fissure between the right and left hepatic lobes, the gallbladder is physiologically tubular in structure with a cross-sectional diameter of up to 5 cm and a normal wall thickness of 1–3.5 mm, dependent on luminal distention [21, 22].
The bile-filled lumen of the gallbladder measures water-isodensity (0–20 Hounsfield Units) on CT and water-isointense signal characteristics on T2-weighted MR imaging; formation and retention of sludge may create layering or smooth gradients of MR intensity/CT attenuation, resulting in a parfait-like appearance. Vicarious excretion of CT contrast material from prior contrast-enhanced CT imaging through gastrointestinal uptake as well as utilization of hepatocyte-specific contrast materials in hepatic MR imaging may alter the imaging appearance of bilious fluid on contrast-enhanced CT as well as MR imaging [23, 24].
8.2.2 Congenital Variants and Anomalies
8.2.2.1 Agenesis of the Gallbladder
Agenesis of the gallbladder, a rare malformation (0.01–0.2% in autopsy series), results from a developmental failure of the caudal division of the primitive hepatic diverticulum or failure of vacuolization. It may result in formation of extrahepatic and intrahepatic gallstones in up to 50% of patients [21, 25].
8.2.2.2 Duplication of the Gallbladder
Duplication of the gallbladder, an equally rare malformation (0.02% in autopsy series), is characterized by a longitudinal septum, dividing the gallbladder cavity, and each cavity draining through its own cystic duct. Developmental it is the consequence of an incomplete revacuolization of the primitive gallbladder and has to be differentiated from gallbladder folds, a bilobed gallbladder, a choledochal cysts, or a gallbladder diverticulum [26].
8.2.2.3 Phrygian Cap of the Gallbladder
The most common anomaly of the gallbladder is a Phrygian cap configuration through septations of body and the distal fundus and may be seen in up to 6% of patients [21, 27].
8.2.2.4 Diverticula of the Gallbladder, Multiseptate Gallbladder, and Ectopic Gallbladder
True gallbladder diverticula are congenital in nature and contain all three muscle layers; pseudodiverticula are usually associated with adenomyomatosis and contain little or no smooth muscle layers in their walls. (Pseudo)Diverticula can occur throughout the gallbladder wall [21, 29].
Septations throughout the gallbladder creating communicating chambers may lead to stasis of bile and formation of gallstones [28].
Various locations of the gallbladder have been described, in particular an intrahepatic location of the gallbladder, which is entirely surrounded by hepatic parenchyma. Intrahepatic subcapsular locations may particularly complicate the diagnosis of an acute cholecystitis as secondary signs of inflammation may be subtle or masked entirely. Shrinkage of the liver in patients with cirrhosis, as well as patients with chronic obstructive pulmonary disease may show gallbladders interposed between liver surface and diaphragm [21, 30].
8.2.3 Pathologic Conditions
8.2.3.1 Gallstones
In cross-sectional imaging, the appearance of gallstones is primarily based on composition and size; most gallstones contain various admixtures of bile pigment, cholesterol, and calcium. Larger proportions of calcium may render gallstones radiodense on CT imaging, while less calcium may potentially lead to entirely radiolucent gallstones. While pure cholesterol stones may be lower in CT density than surrounding bile, central inclusions may contain gas mostly consist of nitrogen.
The high signal intensity of bile on T2-weighted images allows the better delineation of hypointense gallstones compared to T1-weighted sequences. While cholesterol stones are usually hypointense in appearance on T1-weighted images, pigment stones tend to have higher signal intensities; central areas of T2 hyperintensity usually corresponds to fluid-filled clefts [21, 31, 32].
8.2.3.2 Acute Cholecystitis
An obstruction of either the gallbladder neck or the cystic duct may lead to increased intraluminal pressures and eventually results in an inflammation of the gallbladder wall. Gallstones lodged in the neck of the gallbladder or the cystic duct leading to biliodynamic obstruction as well as pressure-induced mucosa ischemia and mucosal injury are the preeminent reason for acute cholecystitis. Ultrasound, CT, and MRI may show distinct features of acute calculous cholecystitis, such as cholecystolithiasis, gallbladder wall thickening, the pericholecystic fluid and inflammation, thickened bile, an indistinct interface between gallbladder wall and liver capsule and potentially gallbladder perforation. Gallbladder perforations can be subdivided into acute, subacute, and chronic scenarios; a subacute perforation with surrounding abscess is the most frequently encountered type of gallbladder perforation. The use of hepatobiliary contrast agents in MR imaging may provide additional functional information about cystic duct patency [21, 33, 34].
In emphysematous cholecystitis and additional vascular compromise of the cystic artery is hypothesized to accelerate the development of gas-forming organisms in the resultant anaerobic environment with eventual penetration of gas into the gallbladder wall. A more frequent occurrence in diabetic patients, as well as the male population with an acalculous gallbladder potentially hints at a separate pathogenesis in contrast to calculous cholecystitis [35].
Inflammation causing ulceration of the mucosal lining and subsequent necrosis may lead to hemorrhagic cholecystitis. The intraluminal hematoma may be seen on CT and MRI may, however, be difficult to differentiate from high intensity/density bile. An accompanying perforation of the gallbladder wall may lead to hemoperitoneum [36].
Coexisting cardiovascular disease predispositions patients with acute cholecystitis to develop gangrenous wall segments. Intraluminal membranes and irregularity of the gallbladder wall intermittently perforated and potentially surrounded by a pericholecystic abscess are key imaging features.
8.2.3.3 Acalculous Cholecystitis
In approximately 5% of all patients with acute cholecystitis, no intraluminal stones can be found. Long stays in intensive care units and abdominal trauma may lead to increased viscosity and subsequent stasis of bile eventually leading to obstruction and mucosa ischemia [21, 37].
8.2.3.4 Chronic Cholecystitis
Repetitive mucosal trauma through pre-existing gallstones as well as recurrent episodes of acute cholecystitis events may contribute to the poorly understood pathogenesis of this fairly common disease. A florid inflammatory response to irritations may also indicate a genetic predisposition. While cross-sectional imaging of chronic cholecystitis may not substantially differ from acute cholecystitis, the greatest difference appears to be a contracted state of the gallbladder in chronic cholecystitis compared to the acute scenario. A decreased gallbladder ejection fraction is oftentimes associated with chronic cholecystitis [21].
Microperforations through mucosal ulcerations as well as ruptured Rokitansky-Aschoff sinuses may lead to penetration of bile into the gallbladder wall, resulting in the formation of xanthogranulomas representing the hallmark of xanthogranulomatous cholecystitis. Gallstones are almost always present, and an irregular configuration of the gallbladder wall is frequently observed. Xanthogranulomatous lesions in the wall may potentially also lead to abscess formations. These may appear hypodense on contrast-enhanced CT imaging as well as hyperintense nodules on T2-weighted MR imaging. Differentiation from gallbladder cancer may be challenging; however, a patent mucosal lining/luminal surface is more indicative of xanthogranulomatous cholecystitis [21].
Impaction of gallstones inside the cystic duct with subsequent compression of the common hepatic duct, and resultant inflammation are mechanisms leading to Mirizzi syndrome. A fairly low insertion of the cystic duct into the common hepatic duct may represent a predisposition. Differentiating the inflammatory origin of the stricture of the common hepatic duct from a neoplastic process may be challenging, the lack of lymphadenopathy, as well as a distinct focal mass may be helpful secondary signs. Erosion of gallstones through the gallbladder wall directly into the adjacent bowel via a cholecystoenteric fistula is the most common mechanism to form a gallstone ileus, in particular, involving the distal ileum [21, 38].
Chronic inflammatory changes of the gallbladder wall may lead to dystrophic calcifications associated with thick fibrous tissue layers of the gallbladder wall, indicating a porcelain gallbladder. The porcelain gallbladder is frequently associated with gallbladder carcinoma [39].
8.2.3.5 Hyperplastic Cholecystosis
A benign proliferation of normal gallbladder wall tissue characterizes this non-inflammatory condition. A deposition of cholesterol-laden macrophages into the lamina propria of the gallbladder wall may lead to the formation of cholesterol polyps and cholesterolosis. Due to their small size, these polyps are best seen on ultrasound imaging [21, 40].
A hypertrophy of the muscular wall with corresponding mucosal overgrowth, formation of intramural diverticula and sinus tracts, then called Rokitansky-Aschoff sinuses, is the hallmark of this disease. Detection of a thickened gallbladder wall in addition to small cystic spaces on CT and MR imaging helps to differentiate adenomyomatosis from gallbladder cancer [40].
8.2.3.6 Gallbladder Neoplasms
Benign neoplasms of the gallbladder are rare and usually represent adenomas, which are incidentally, (0.3–0.5%) found during cholecystectomies [21].
During the 6th or 7th decades of life with a female predilection of up to 3:1, gallbladder carcinomas, histopathologically usually presenting as adenocarcinomas; however, adenosquamous, squamous, or neuroendocrine carcinomas can also be found may arise from the gallbladder wall. Predisposing factors associated with gallbladder carcinoma are gallstones (75% of patients with gallbladder carcinomas have gallstones), porcelain gallbladder, genetic factors as well as pancreatobiliary ductal unions (reflux of pancreatic juice into the common bile duct leading to chronic inflammation). On cross-sectional imaging, either a mass is visualized invading the gallbladder fossa or the mass is noted to fill most of the enlarged and deformed gallbladder. Invasion of surrounding structures, in particular the liver, the hepatoduodenal ligament, the right hepatic flexure, or the duodenum is frequently observed. Lymphatic spread to the regional and distant lymph nodes is very common; hematogenous metastasis are usually found in the liver, peritoneal seeding is also fairly common. Biliary obstruction may be observed in up to 50% of patients [21, 41, 42].
Secondary lymphoma to the gallbladder may be seen in disseminated lymphomatous stages, lymphoma involving the gallbladder is extremely rare [43].
Metastases to the gallbladder have been described, malignant melanoma being the most common cause of metastatic tumors, accounting for more than 50% of all cases of gallbladder metastases [44].
8.3 Conclusion
Knowledge of various diseases of the gallbladder and biliary tract in combination with careful inspection of the imaging appearances is of paramount importance for correct interpretation of biliary studies. Offering a succinct set of differential diagnoses for various cholangiopathies is important because specific management pathways exist and prognosis can differ according to the type of underlying disease. Cross-sectional imaging studies play an essential roles in the diagnosis and treatment planning as well as visualization of disease evolution of patients with biliary malignancies and multimodality and multiparametric imaging approaches can provide complementary information in evaluating the tumor extent and resectability.
Take-Home Messages
-
An appreciation of the pathologic basis of diseases of the gallbladder and biliary tract, combined with careful inspection of the imaging appearances, is vital for the correct interpretation of biliary studies.
-
Differential diagnosis of various cholangiopathies is important because specific management exists and prognosis can differ according to the type of disease.
-
Cross-sectional imaging studies play an essential role in the diagnosis and treatment planning as well as visualization of disease evolution of patients with biliary malignancies and multimodality and multiparametric imaging approaches can provide complementary information in evaluating the tumor extent and resectability.
References
Labib PL, Goodchild G, Pereira SP. Molecular pathogenesis of cholangiocarcinoma. BMC Cancer. 2019;19(1):185.
Cardinale V, Carpino G, Reid L, Gaudio E, Alvaro D. Multiple cells of origin in cholangiocarcinoma underlie biological, epidemiological and clinical heterogeneity. World J Gastrointest Oncol. 2012;4(5):94–102.
Alpini G, McGill JM, Larusso NF. The pathobiology of biliary epithelia. Hepatology. 2002;35(5):1256–68.
Catalano OA, Singh AH, Uppot RN, Hahn PF, Ferrone CR, Sahani DV. Vascular and biliary variants in the liver: implications for liver surgery. Radiographics. 2008;28(2):359–78.
Kim OH, Chung HJ, Choi BG. Imaging of the choledochal cyst. Radiographics. 1995;15(1):69–88.
Todani T, Tabuchi K, Watanabe Y, Kobayashi T. Carcinoma arising in the wall of congenital bile duct cysts. Cancer. 1979;44(3):1134–41.
Kamisawa T, Kuruma S, Chiba K, Tabata T, Koizumi S, Kikuyama M. Biliary carcinogenesis in pancreaticobiliary maljunction. J Gastroenterol. 2017;52(2):158–63.
Zulfiqar M, Chatterjee D, Yoneda N, Hoegger MJ, Ronot M, Hecht EM, Bastati N, Ba-Ssalamah A, Bashir MR, Fowler K. Imaging features of premalignant biliary lesions and predisposing conditions with pathologic correlation. Radiographics. 2022;42(5):1320–37.
Yeh BM, Liu PS, Soto JA, Corvera CA, Hussain HK. MR imaging and ct of the biliary tract. RadioGraphics. 2009;29(6):1669–88.
Seo N, Kim SY, Lee SS, et al. Sclerosing cholangitis: clinicopathologic features, imaging spectrum, and systemic approach to differential diagnosis. Korean J Radiol. 2016;17:25–38.
Venkatesh SK, Welle CL, Miller FH, Jhaveri K, Ringe KI, Eaton JE, Bungay H, Arrivé L, Ba-Ssalamah A, Grigoriadis A, Schramm C, Fulcher AS, IPSCSG. Reporting standards for primary sclerosing cholangitis using MRI and MR cholangiopancreatography: guidelines from MR Working Group of the International Primary Sclerosing Cholangitis Study Group. Eur Radiol. 2022;32(2):923–37.
Martínez-de-Alegría A, Baleato-González S, García-Figueiras R, Bermúdez-Naveira A, Abdulkader-Nallib I, Díaz-Peromingo JA, Villalba-Martín C. IgG4-related disease from head to toe. Radiographics. 2015;35(7):2007–25.
Horton KM, Bluemke DA, Hruban RH, Soyer P, Fishman EK. CT and MR imaging of benign hepatic and biliary tumors. Radiographics. 1999;19(2):431–51.
Sibulesky L, Nguyen J, Patel T. Preneoplastic conditions underlying bile duct cancer. Langenbecks Arch Surg. 2012;397(6):861–7.
Park HJ, Kim SY, Kim HJ, Lee SS, Hong GS, Byun JH, Hong SM, Lee MG. Intraductal papillary neoplasm of the bile duct: clinical, imaging, and pathologic features. AJR Am J Roentgenol. 2018;211(1):67–75.
Lee MH, Katabathina VS, Lubner MG, Shah HU, Prasad SR, Matkowskyj KA, Pickhardt PJ. Mucin-producing cystic hepatobiliary neoplasms: updated nomenclature and clinical, pathologic, and imaging features. Radiographics. 2021;41(6):1592–610.
Lee CH, Chang CJ, Lin YJ, Yeh CN, Chen MF, Hsieh SY. Viral hepatitis-associated intrahepatic cholangiocarcinoma shares common disease processes with hepatocellular carcinoma. Br J Cancer. 2009;100(11):1765–70.
Horvat N, Nikolovski I, Long N, Gerst S, Zheng J, Pak LM, Simpson A, Zheng J, Capanu M, Jarnagin WR, Mannelli L, Do RKG. Imaging features of hepatocellular carcinoma compared to intrahepatic cholangiocarcinoma and combined tumor on MRI using liver imaging and data system (LI-RADS) version 2014. Abdom Radiol (NY). 2018;43(1):169–78.
Lee DH, Kim B, Lee ES, Kim HJ, Min JH, Lee JM, Choi MH, Seo N, Choi SH, Kim SH, Lee SS, Park YS, Chung YE. Korean Society of Abdominal Radiology. Radiologic evaluation and structured reporting form for extrahepatic bile duct cancer: 2019 consensus recommendations from the Korean Society of Abdominal Radiology. Korean J Radiol. 2021;22(1):41–62.
Riopel MA, Klimstra DS, Godellas CV, Blumgart LH, Westra WH. Intrabiliary growth of metastatic colonic adenocarcinoma: a pattern of intrahepatic spread easily confused with primary neoplasia of the biliary tract. Am J Surg Pathol. 1997;21(9):1030–6.
Lim JH, Kim KW, Choi D-i. Biliary tract and gallbladder. In: Haaga JR, Boll DT, editors. CT and MRI of the whole body. 6th ed. Philadelphia: Elsevier; 2017. p. 1192–267.
Smathers RL, Lee JK, Heiken JP. Differentiation of complicated cholecystitis from gallbladder carcinoma by computed tomography. AJR Am J Roentgenol. 1984;143:255–9.
Havrilla TR, Reich NE, Haaga JR, Seidelmann FE, Cooperman AM, Alfidi RJ. Computed tomography of the gallbladder. AJR Am J Roentgenol. 1978;130:1059–67.
Strax R, Toombs BD, Kam J, Rauschkolb EN, Patel S, Sandler CM. Gallbladder enhancement following angiography: a normal CT finding. J Comput Assist Tomogr. 1982;6:766–8.
Al-Fallouji MA. Perforated posterior peptic ulcer associated with gallbladder agenesis and midgut malrotation. Br J Clin Pract. 1983;37:353–6, 358.
Sheng H, Chen G, Yang M, Guan H. A proposed feasible classification of common bile duct duplications based on a newly described variant and review of existing literature. BMC Pediatr. 2022;22(1):647.
Chen X, Yi B. Triple gallbladder. J Pediatr. 2022;247:173–4.
Hopmann P, Tan E, Lo D. Multiseptate gallbladder presenting with biliary colic. J Surg Case Rep. 2022;2022(9):rjac417.
Kochhar R, Nagi B, Mehta SK, Gupta NM. ERCP diagnosis of a gallbladder diverticulum. Gastrointest Endosc. 1988;34:150–1.
Gore RM, Ghahremani GG, Joseph AE, Nemcek AA Jr, Marn CS, Vogelzang RL. Acquired malposition of the colon and gallbladder in patients with cirrhosis: CT findings and clinical implications. Radiology. 1989;171:739–42.
Brink JA, Ferrucci JT. Use of CT for predicting gallstone composition: a dissenting view. Radiology. 1991;178:633–4.
Tsai HM, Lin XZ, Chen CY, Lin PW, Lin JC. MRI of gallstones with different compositions. AJR Am J Roentgenol. 2004;182:1513–9.
Bennett GL, Balthazar EJ. Ultrasound and CT evaluation of emergent gallbladder pathology. Radiol Clin North Am. 2003;41:1203–16.
Paulson EK. Acute cholecystitis: CT findings. Semin Ultrasound CT MR. 2000;21:56–63.
Jacob H, Appelman R, Stein HD. Emphysematous cholecystitis. Am J Gastroenterol. 1979;71:325–30.
Jenkins M, Golding RH, Cooperberg PL. Sonography and computed tomography of hemorrhagic cholecystitis. AJR Am J Roentgenol. 1983;140:1197–8.
Mirvis SE, Vainright JR, Nelson AW, et al. The diagnosis of acute acalculous cholecystitis: a comparison of sonography, scintigraphy, and CT. AJR Am J Roentgenol. 1986;147:1171–5.
Koehler RE, Melson GL, Lee JK, Long J. Common hepatic duct obstruction by cystic duct stone: Mirizzi syndrome. AJR Am J Roentgenol. 1979;132:1007–9.
Kane RA, Jacobs R, Katz J, Costello P. Porcelain gallbladder: ultrasound and CT appearance. Radiology. 1984;152:137–41.
Jutras JA. Hyperplastic cholecystoses; Hickey lecture, 1960. Am J Roentgenol Radium Ther Nucl Med. 1960;83:795–827.
Hamrick RE Jr, Liner FJ, Hastings PR, Cohn I Jr. Primary carcinoma of the gallbladder. Ann Surg. 1982;195:270–3.
Yoshimitsu K, Honda H, Shinozaki K, et al. Helical CT of the local spread of carcinoma of the gallbladder: evaluation according to the TNM system in patients who underwent surgical resection. AJR Am J Roentgenol. 2002;179:423–8.
Mitropoulos FA, Angelopoulou MK, Siakantaris MP, et al. Primary non-Hodgkin’s lymphoma of the gall bladder. Leuk Lymphoma. 2000;40:123–31.
Guida M, Cramarossa A, Gentile A, et al. Metastatic malignant melanoma of the gallbladder: a case report and review of the literature. Melanoma Res. 2002;12:619–25.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Do, R.K., Boll, D.T. (2023). Diseases of the Gallbladder and the Biliary Tree. In: Hodler, J., Kubik-Huch, R.A., Roos, J.E., von Schulthess, G.K. (eds) Diseases of the Abdomen and Pelvis 2023-2026. IDKD Springer Series. Springer, Cham. https://doi.org/10.1007/978-3-031-27355-1_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-27355-1_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-27354-4
Online ISBN: 978-3-031-27355-1
eBook Packages: MedicineMedicine (R0)