Abstract
The effects of climate change on food production, together with the economic downturn caused by the COVID-19 pandemic, have challenged the true resilience of current global food systems, which has been further constrained with the escalation of global conflicts. As a result, people around the globe are living in a nutritional and health crisis. Providing sustainable, healthy, and nutritious foods to a growing population will require a shift to more plant-based diets that align more closely with national dietary guidelines. Although often minimized in dietary recommendations, taste is among the key determinants to consuming healthful diets. This chapter reviews how umami, through its taste and physiological functions, supports and has the potential to further enhance the acceptance of foods that align with human and planetary health.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
Suboptimal diets on top of social and environmental challenges have resulted in a global nutritional crisis (Independent Expert Group, 2021). Current trends of unhealthy and unsustainable food production, together with an estimated growth in population to about 10 billion by 2050, represent a threat to food security, the health of people, and the health of the planet (Pörtner et al., 2022; Willett et al., 2019). For the shift to sustainable food systems that provide a healthy, nutritious, and sustainable diet, experts have evaluated the best available evidence (Afshin et al., 2019) and identified a need to increase ingestion of plant-based foods and to reduce consumption of animal-based foods (Willett et al., 2019). Therefore, the amount and kinds of foods we choose to eat have an impact not only on our health but also on the environment (Springmann et al., 2016).
The challenge is to make consumption of plant-based foods more appealing. The sense of taste has great relevance to nutrition: it helps identify and support ingestion of nutrients, improving the probability of survival (Mattes, 2021). In this context, the pleasant taste conferred by umami compounds could play a key role in increasing the palatability of plant-based foods and, more specifically, local plant-based crops that are part of the traditional cuisine in each region.
8.1 Diet, Health, and the Environment
The Global Burden of Disease Study 2017 showed an association between the quantity and type of foods we eat and our health (Afshin et al., 2019). The relevance of nutrition for our health has been strongly reinforced in the last 2 years with the COVID-19 pandemic (Pörtner et al., 2022; Willett et al., 2019): individuals infected with SARS-CoV-2 that presented age-associated preexisting comorbidities, such as noncommunicable diseases (NCDs) like diabetes and obesity (Afshin et al., 2019; Michael A Clark, 2019), had a higher risk of mortality (Antos, 2021; Ssentongo, 2020; Centers for Disease Control and Prevention, 2022). In fact, the pre-COVID-19 prevalence and incidence of these chronic diseases, which are indicators of poor metabolic health (but not diseases exclusive to modern humans), have increased and continue to be the leading cause of total lives lost (Thompson, 2013; WHO, 2020a). In 2019, before the COVID-19 pandemic, seven out of ten deaths were the result of NCDs, with cardiovascular diseases at the top of the list—the most frequent chronic illnesses in an aging population (Tubiello et al., 2021).
Suboptimal diets were estimated to pose a higher risk of mortality from NCDs. Inadequate consumption of fruits, vegetables, nuts, seeds, and whole grains and excess consumption of sodium have caused 11 million deaths and 255 million disability-adjusted life-years (DALYs), a combination of years of life lost due to premature mortality (YLLs) and years of healthy life lost due to living with disability (YLDs) (WHO, 2020b). Eating at least 400 grams of fruits and vegetables (including legumes, nuts, and whole grains), more unsaturated fats, and less than 5 grams of salt per day throughout the life course was found to prevent all forms of malnutrition and lower the risk of NCDs (WHO, 2020c).
And, it so happens that these foods not only are more beneficial for our health but also have a lower impact on the environment (Chai, 2019; Clark, 2019; Springmann, 2016; Tilman & Clark, 2014): the composition of the diet strongly affects the emission of greenhouse gases. For instance, the production of plant-based foods releases a lower volume of greenhouse gases than does producing animal-based foods. However, selecting foods solely on their environmental impact may not automatically maximize human health (Tilman & Clark, 2014).
Thus, we have a diet-health-environment dilemma, in which no region of the world meets the recommended consumption of health-promoting foods, unhealthy foods are overconsumed, and unsustainable food systems are causing anthropogenic changes to Earth’s climate.
8.1.1 Shift of Global Traditional Diets
As nations become more affluent, especially low- and middle-income countries, traditional diets based on coarse starchy crops with complex carbohydrates (e.g., cereals, roots, and tubers) are replaced by refined starches, added sugars, vegetable oils, and animal fats, and plant proteins are replaced by such animal products as eggs, meats, and dairy (Drewnowski & Poulain, 2018). These changes in dietary patterns, supported by greater wealth and higher food affordability, are known as nutrition transition (Drewnowski & Popkin, 1997). However, the health and environmental costs seem to be hidden in inexpensive global staple grain and sugar crops, as shown by the increase in the burden of malnutrition (Fanzo, 2018; Tilman & Clark, 2014). A “Western/unhealthy” pattern has been described as having a high intake of refined grains and sweets, meat, and soft drinks (Murakami et al., 2018).
8.1.2 Umami and the Japanese Dietary Pattern
According to the WHO (2019), Japan has the longest average life expectancy in the world, which has been partially attributed to the Japanese dietary pattern. Assessed as the Japanese Dietary Index, the Japanese dietary pattern has been associated with a lower risk of all causes of death and of cardiovascular and heart disease mortality (Abe et al., 2020; Matsuyama et al., 2021). A higher adherence to this diet was associated with a longer life and longer disability-free survival (Abe et al., 2020; Zhang et al., 2019). Experts have known for some time that evaluating dietary patterns, in which foods are eaten in combination, rather than listing foods in isolation, gives better guidance for diet quality (Reedy et al., 2014). Common foods in the Japanese dietary pattern, such as seaweeds, fish, green and yellow vegetables, and green tea, contain myriad beneficial nutrients and phytochemicals, which are suspected to have a cumulative effect.
The Japanese dietary pattern traditionally known as washoku (a traditional dietary culture of Japan) is not the only one that lowers the risk of all causes of morbidity and mortality, but it is the only one that includes a specific taste as part of its traditional heritage: the umami taste. The guiding principles of washoku, designated a UNESCO intangible cultural heritage in 2013, explain that the basic structure of a customary Japanese meal includes a distinctive flavor that results from the combination in the mouth of the taste, smell, and the tactile sensation of each ingredient (Ninomiya, 2016). The core flavor of many Japanese recipes is the umami taste from dashi soup stock. The extraction of umami substances when preparing the stock from traditional ingredients—dried kelp, dried bonito, or dried shiitake mushrooms—in combination with such products as vinegar, miso, or soy sauce intensifies the flavor of seasonal and fresh local ingredients (Kumakura, 2015). The style of eating small portions of a large variety of seasonal foods, including fish and abundant vegetables, and the effective use of umami taste seem to be the basic elements that promote positive health outcomes from washoku (San Gabriel et al., 2018).
The modern Japanese diet has transitioned to include high intakes of refined grains (white rice and white flour), as well as vegetables, seaweeds, soybean products, fish, and green tea, together with low consumption of whole grains, nuts, processed meats, and soft drinks (Micha et al., 2015; Murakami et al., 2018). Although there are still lower rates of morbidity and mortality from coronary artery disease in Japan than in other regions, this seems to be supported by higher consumption of plant and marine food and decreased intake of refined carbohydrates and animal fat.
In fact, the Japanese dietary pattern has been changing since the end of World War II, becoming more diversified and Westernized. The consumption of total fats and oils has increased threefold from 1960 to 2005, and the consumption of animal products (meat, poultry, milk, and dairy) increased fourfold, while the intake of white rice was reduced by half in this period. During these 45 years, the mean intake of fish and beans augmented slightly, and vegetable consumption remained constant (Tada et al., 2011). The estimated percentage of energy derived from fat in the Japanese diet has transitioned from 7.0% in 1946 to 26.6% in 2000, together with a gradual decrease on salt intake from 13.7 g/day in 1976 to 10.6 g/day in 2006. Overall, the Westernization of the Japanese dietary style has increased the number of people with higher atherosclerotic risk by augmenting the rates of obesity, dyslipidemia, and hyperinsulinemia with impaired glucose intolerance, especially among the younger population in Japan (Tada et al., 2011).
Murakami et al. (2018) used principal component analysis to identify three major dietary patterns through the Japanese National Health and Nutrition Survey from 2003 to 2015: a “plant food and fish” pattern, a “bread and dairy” pattern, and an “animal food and oil” pattern. They found an apparent continuation of the Westernization of the Japanese diet: a gradual decrease in consumption of foods in the plant food and fish pattern and a significant increase in consumption of foods in the bread and dairy and the animal food and oil patterns (summarized in Table 8.1). Interestingly, the plant food and fish pattern, which seems to be closer to the recommended “healthy/prudent” pattern, also showed a higher use of salt-based seasonings (Murakami et al., 2018). Without forgetting the need to decrease the intake of salt at the population level, experts still express the need to promote a high consumption of plant and fish foods while discouraging the consumption of refined carbohydrate and animal fat for better health outcomes (Tada et al., 2011). In this context, returning to the core flavors of washoku based on the umami taste of traditional ingredients could help halt the speed of Westernization of the Japanese diet and move it more toward the plant food and fish pattern while helping reduce excess salt intake with the use of the umami seasoning (see Chap. 4) (San Gabriel et al., 2018).
8.2 The Significance of Umami Taste in Food Choice
The sensory cues of foods before, during, and after eating direct our selection of foods and are at the center of palatability. Smell and taste drive palatability, depending on our hedonic evaluation of food (Drewnowski, 1997; Gervis et al., 2022; McCrickerd and and Forde, 2016; Yeomans, 1998). But while orthonasal exposure to odors through the nose combined with retronasal odors arising from the mouth provides cues about the food itself in anticipation of eating, taste seems to play a clearer role in sensing nutrients during and after ingestion of foods (Boesveldt & de Graaf, 2017). Often cited as the “nutritional gatekeeper” of the body, the sense of taste has a prominent role in voluntary food ingestion because it helps us choose what to consume and how efficiently these foods will be digested and metabolized (Breslin, 2013). In combination with smell and the tactile sensation from the texture of foods, taste produces flavors and drives a primal response of “acceptable” or pleasantness, when we detect nutrients that we consider safe to ingest, or “unacceptable” or unpleasant, when we estimate that something could be toxic. And this distinction between toxic and nutritious appears to be qualitatively defined by taste, the oral perception of chemicals in food (Breslin, 2013). Unique types of taste cells express specific receptors to detect only one of the five basic tastes: sweet, sour, bitter, salty, or umami (Chandrashekar et al., 2006). The perception of these taste qualities results from the presence in foods of hydrophilic sugars, acids, alkaloids, salts, and amino acids that dissolve in saliva and activate distinctive receptors in taste cells, the same receptors that are used to sense molecular changes in our internal milieu (Bachmanov et al., 2014; Vincis & Fontanini, 2019). The gastrointestinal tract continues detecting the presence of nutrients and harmful compounds via the same taste receptors, but in this case food substances evoke not conscious taste sensations but metabolic responses (Breslin, 2013; San Gabriel & Uneyama, 2013; Steinert & Beglinger, 2011). The brain integrates taste signals from the gustatory system with other nontaste modalities such as texture, temperature, odor, and even visceral and homeostatic signals that help us contextualize food experiences (Vincis & Fontanini, 2019). This is why tastes and flavors that are connected to nutrients and calories will become more pleasurable over time than those flavors that we associate with feeling ill from previous experience (Breslin, 2013). The integration of food signals drives our interpretation of whether a meal is pleasant and ultimately influences our eating behavior (Khan et al., 2021; Rolls, 2009; Small, 2012).
In the case of umami, the taste of monosodium glutamate (MSG) when presented alone is not pleasant (Beauchamp, 2009; Okiyama & Beauchamp, 1998), but functional brain imaging shows that the combination of MSG with a consonant savory smell such as a vegetable odor induces higher signals of pleasantness in brain cortical regions where taste and olfactory signals converge. Thus, some describe umami as a “rich and delicious flavor” (McCabe & Rolls, 2007). In fact, among the five basic tastes, the savory umami taste of the amino acids glutamate and aspartate in combination with 5′-ribonucleotides is one of the tastes we perceive as pleasant in a food context, and these umami compounds are widespread among many of the foods we eat daily (Breslin, 2013; Ninomiya, 1998).
8.3 The Especially Human Taste of Umami
Umami receptors (described in Chaps. 1 and 2) are the means to sense the proteogenic amino acids. However, the high sensitivity of the umami receptor T1R1 + T1R3 for glutamate is very specific to humans. It is hypothesized that in early evolution, the sense of taste may have helped our ancestor hominids identify nutritious foods (Breslin, 2013; Toda et al., 2021). Modern humans lean toward the consumption of more digestible animal proteins instead of plant proteins, but for meats to acquire strong umami flavors, it is necessary to prepare or ferment them (Mouritsen & Styrbeak, 2020). Thus, in periods of low food availability, our ancestors may have had to depend on less digestible and less palatable wild plants (Milton, 2000).
A recent study that combined functional, behavioral, phylogenetic, and ecological methods has found that the umami receptor in primates has evolved at least three times in parallel with the dietary transition from insects to leaves as typical sources of protein (Toda et al., 2021). The T1R1 + T1R3 receptor in primates whose diet depends mostly on insects is most potently activated by 5′-ribonucleotides, whereas in primates whose diet mostly depends on leaves, glutamate evokes the more significant response. According to the analysis of Toda et al. (2021), glutamate is one of the most abundant free amino acids in insects and plants, but plants have lower 5′-ribonucleotide content than do insects. This is because plants do not have muscle tissue, which is a source of ATP, the precursor for free 5′-ribonucleotides (Mouritsen & Styrbeak, 2020). Together with evidence from other studies, it seems that the modern human T1R1 + T1R3 receptor evolved from early mammals to perceive 5′-ribonucleotides present in insect-based diets but later evolved higher sensitivity to glutamate to facilitate the consumption of leafy plants.
However, plants also contain many bitter metabolites that bind to a family of mainly broadly tuned bitter taste receptors, the T2R receptors (Adler et al., 2000; Behrens et al., 2007; Chandrashekar et al., 2000). Because plants contain many bitter metabolites, by considering how bitter taste functions, it is easier to understand the role of umami taste in modulating the noxious bitterness of leafy greens that are so important to reduce the risk of diet-related chronic diseases (Afshin et al., 2019; Drewnowski & Gomez-Carneros, 2000).
8.3.1 Peculiarities of Bitter Taste
Based on their agonist spectra, human T2Rs can be broadly, intermediately, or narrowly tuned (Behrens & Meyerhof, 2013). Bitter taste receptors are thought to function as warning sensors that prevent humans from ingesting noxious food molecules, responding to a vast range of compounds known to be bitter to humans. Twenty-five bitter taste receptor genes in humans (hTAS2Rs) have been identified that encode for the G-protein-coupled receptor family of type 2 taste receptors (T2Rs) (Behrens et al., 2007). They have been described as “chemosensory sentinels” because they alert us to potential threats and trigger defensive responses in the oral cavity and beyond (Harmon et al., 2021). Bitter taste receptors are also found in chemosensory cells of extraoral tissues such as the gut and airways that mediate responses from ingested or inhaled substances (Deshpande et al., 2010; Wooding & Ramirez, 2022). T2Rs are preeminent in the rejection of potentially harmful compounds that at high levels can be harmful and even fatal, making us avoid intensely bitter-tasting toxins. This is why humans normally react by spitting, evading, or vomiting strong bitter substances (Breslin, 2013). However, many bitter compounds at low levels have medicinal properties (Bayer et al., 2021). Therefore, compounds that activate T2Rs are structurally diverse and include different drugs that can be potentially toxic (Mennella et al., 2013).
The work of Meyerhof et al. (2010) indicates that the perception of most bitter compounds is not a simple association between an agonist and a specific receptor but, rather, a complex interaction with a wide set of TAS2Rs (Wooding et al., 2021). In addition, there are large individual differences in the sensitivity for bitter taste compounds. The oldest known example refers to the genetic ability to taste two bitter compounds, phenylthiocarbamide (PTC) and 6-n-propylthiouracil (PROP) (Tepper et al., 2009). Around 70–75% of individuals worldwide taste PTC and PROP as moderately to intensely bitter—they are considered “tasters”—whereas the rest (around 28%) are “nontasters”: they are taste-blind to both compounds (Kim & Drayna, 2004). Psychophysical studies have shown that the population can be divided into three distinctive groups: nontasters (30%), medium tasters (50%), and supertasters (20%) (Bartoshuk et al., 1994). This is explained by three single-nucleotide polymorphisms of the human gene TAS2R38 that cause substitution of three amino acids at positions P49A, A262V, and V296I. These substitutions produce two common haplotypes, the taster variant (PAV) and the nontaster variant (AVI) (Tepper et al., 2009). PROP-sensitive individuals are carriers of the dominant allele PAV/PAV (the supertasters) or PAV/AVI (the medium tasters), whereas PROP-insensitive individuals are carriers of the recessive allele AVI/AVI. Others have shown that the relative expression level for PAV TASR38 mRNA among heterozygous individuals differs widely and correlates with how they rate the bitterness intensity of both PROP and broccoli juice, which contains PTC-like glucosinolates (Lipchock et al., 2013).
These differences in the hTAS2R38 gene affect nutritional and health outcomes. The hTAS2R38 protein is considered the antithyroid-toxin receptor—it detects thiourea moieties such as PTC-like glucosinolates, which are dietary goitrogens present in many vegetables, including broccoli. One theory is that sensitivity to the bitterness of PROP has been conserved as a mechanism to protect humans against the excessive consumption of dietary goitrogens, especially in environments with low-iodine soils, where ingestion of plant glucosinolates may aggravate endemic goiter (Sandell & Breslin, 2006). Infants and children are thought to be more sensitive to the effect of glucosinolates, which may explain their innate aversion to bitterness, to prevent the risk of consuming toxic compounds (Breslin, 2013; Mennella & Bobowski, 2015).
Many cruciferous plants of the family Brassicaceae, including such species as broccoli, Brussels sprouts, cabbage, cauliflower, watercress, radish, kale, and mustard, contain PTC-like glucosinolates (Sandell & Breslin, 2006). The amount of these bioactive compounds present in these vegetables has been shown to be a major barrier for acceptance and consumption of cruciferous vegetables (Drewnowski & Gomez-Carneros, 2000). However, PROP tasters rated as unpleasant not only the taste of cruciferous cultivated vegetables but also tastes of other foods like coffee and grapefruit. Therefore, bitter taste sensitivity to PROP seems to generalize to other bitter compounds, which may influence the foods we choose to eat. And as a result of food preference, variations of TAS2R38 appear to be related to adiposity and vegetable consumption (Duffy et al., 2010; Tepper et al., 2009).
However, not all substances that are toxic taste bitter, nor are all bitter compounds toxic. In fact, associations between bitter taste sensitivity and various health measures, such as tobacco use, body mass index, glucose homeostasis, and susceptibility to respiratory infections, have also been found (Dotson et al., 2008; Lee et al., 2012). And it seems that bitter taste acceptance is a behavior we can learn, so taste education could play an important role in children’s future health (Beckerman et al., 2017; Mennella et al., 2016).
8.3.2 Interaction of Umami Compounds and Bitter Receptors
In the case of umami taste, human psychophysical studies have shown that umami substances appear to suppress the bitterness of various compounds (Keast & Breslin, 2003; Keast & Breslin, 2002; Kemp & Beauchamp, 1994; Yamaguchi, 1998). In vitro assays revealed that MSG, inosine 5′-monophosphate (IMP), L-theanine, and umami peptides behave as antagonists of various bitter taste receptors, such as the salicin bitter taste receptor hTAS2R16 and the caffeine receptors hTAS2R43 and hTAS2R46, also confirmed psychophysically (Kim et al., 2015, 2017). But the potency to suppress bitterness does not seem to correlate to umami intensity, since each compound interacts differently with bitter taste receptors (Kim et al., 2015; Rhyu et al., 2020). Another bitter taste receptor significantly inhibited by umami compounds is hTAS2R14 (Okuno et al., 2020), the receptor activated by the tea catechins (−)-epigallocatechin gallate and (−)-epicatechin gallate, which are partly responsible for the bitterness of tea (Yamazaki et al., 2013), and by several phenolic compounds from extravirgin olive oil (Cui et al., 2021), whose bitterness indicates the presence of phenolic compounds.
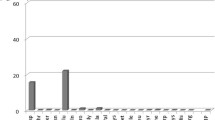
Altogether, it seems that umami compounds can block the activation of some bitter taste receptors, but its efficiency is complicated, partially due to the complex interaction of bitter compounds with TAS2Rs, due to either the broadly tuned (promiscuous) bitter receptors or the intermediately or narrowly tuned (selective) receptors (Bayer et al., 2021; Behrens & Meyerhof, 2013). As shown in Fig. 8.1, the index of promiscuity (number of substances that activate the receptor divided by the total compounds of their data set of food TAS2R agonists) is high for TAS2R14, TAS2R1, TAS2R46, and TAS2R4 and low for TAS2R31, TAS2R20, and TAS2R50. This means that the ability of umami compounds to block the activation of TASR14 or TAS2R46 may not seem specific, whereas umami compounds are probably more selective antagonists for that more narrowly tuned receptors TAS2R43 and TAS2R16. If umami compounds could antagonize the activation of most of the promiscuous TAS2Rs, we could say that umami inhibits the bitterness of most of the bitterness of the food components found in plants. However, as yet there are not enough molecular studies of other broadly tuned TAS2Rs to assume that this is the case.
Index of bitter taste receptor promiscuity estimated by the number of food agonists to an individual TAS2R divided by the total number of compounds in the data set. (Modified from Bayer et al. (2021). Asterisks indicate the bitter receptors for which umami compounds were able to suppress the bitterness signal in in vitro assays)
8.4 The Function of Umami for the Consumption of Plants
The importance of consuming vegetables relies on the growing evidence that vegetables provide not only nutrients and dietary fiber but also phytochemicals (nonnutritive bitter phenolic compounds) that exert various physiological mechanisms, have antioxidant activities, and modulate the immune system (Bayram et al., 2018; Drewnowski & Gomez-Carneros, 2000). These nonnutritive organic constituents, which are very diverse structurally—including phenolic compounds, carotenoids, glucosinolates, alkaloids, terpenes, and peptides—are thought to significantly contribute to the health benefit of plant-based diets (Bayer et al., 2021; Cicero et al., 2017). Yet these are also the molecules that give the astringent and bitter sensory attributes of vegetables by binding to the human T2R bitter taste receptors (Soares et al., 2018; Sterneder et al., 2021) and the reason that vegetables elicit complex perceptual stimuli (Duffy et al., 2010).
8.4.1 Umami, Salt, and Vegetable Consumption
Few sensory studies to date have evaluated the effects of umami on the perception of bitterness in foods and mixed meals, despite anecdotal evidence suggesting a benefit. As indicated in Chap. 4, there is a growing understanding of the impact of umami and specifically MSG on consumer liking and acceptance of reduced-sodium foods. Studies in that context provide some indication, at least directionally, that activating the umami receptor via the presence of glutamate attenuates the perception of bitterness within a mixed meal. For example, Halim et al. (2020) presented participants with three versions—normal salt, reduced salt, reduced salt with MSG—of four different mixed meals: roasted vegetables (carrots and eggplant), quinoa, yogurt-based dip, and pork cauliflower (with onion, peas, and carrots). The study sought to test the hypothesis that, with addition of MSG, reduced-salt versions of mixed dishes would be equally as liked as full-salt versions of the same dishes. In addition to assessing overall liking and liking of appearance, flavor, and texture/mouthfeel, several sensory characteristics were evaluated by a check-all-that-apply scale, which included “bitter” in addition to “deliciousness,” “flavorful,” “balanced,” “bland,” “rancid,” “fresh,” and “savory.” Results showed that, directionally, the attribute of “bitter” was lower in the MSG version of the roasted vegetables, quinoa, and pork cauliflower dishes. Penalty analysis showed that bitter was a negative driver of liking overall; therefore, a shift in less perception of bitter may lead to higher liking.
However, lower bitter with MSG was shown only in the quinoa and the yogurt dip, where overall liking was higher with the MSG versions than with the standard full-salt versions. There were many limitations in this study, most notably that the dishes were not optimized for salt and MSG, and neither dish was considered particularly bitter. Yet, these results suggest that, in a practical setting, umami may favorably affect the perception of bitter, which may relate to acceptance of foods. Additional research, particularly in plant-based foods rich in bitter compounds, is warranted to better understand the relationship between umami and bitter (Fig. 8.2).
8.4.2 Umami Perception and Dietary Patterns
Various studies have examined the association between umami taste sensitivity and food preference and/or food consumption, with mixed results (Fluitman et al., 2021; Gervis et al., 2022; Puputti et al., 2019). Puputti et al. (2019) found that those with a higher sensitivity to umami consumed more vegetables than those least sensitive to umami. Fluitman and colleagues found that poor umami taste sensitivity related to lower adherence to the Mediterranean diet, which is abundant on foods rich in umami compounds (Fluitman et al., 2021; Ninomiya, 1998). However, in the cohort study from Gervis et al. (2022), which defined profiles of the perception of all five basic tastes collectively, older adults with high sensitivity to all tastes but umami had a higher probability of following a vegetable-rich diet than those with low bitter or higher umami perception. The difference could be explained by differences in the cohorts and the type of taste measurements (perception vs. sensitivity). But in the end, umami is implicated in specific dietary patterns, in addition to individual and age differences, so it seems important to clarify the association between umami and overall taste perception and food consumption.
8.5 Conclusion
Taste is often a forgotten element in a sustainable diet, yet it is one of the key drivers for promoting consumption of a nutritious and healthful dietary pattern (Fig. 8.3). Of the five basic tastes, umami in particular is often omitted in discussions toward sustainable food systems that provide healthy, nutritious, and sustainable diets that better promote health for both people and planet. The global population continues to grow and age, increasing the prevalence of such noncommunicable diseases (NCDs) as diabetes and obesity, primary causes of disability and premature deaths worldwide that result from preventable lifestyle and dietary habits. Psychophysical, molecular, and nutritional studies continue to show how umami can block the bitterness of many phytochemicals that are cardioprotective and how umami perception is involved in the preference of healthier dietary patterns. Application of umami compounds, as has been done for centuries in the traditional Japanese diet, can serve as a tool to reduce salt and increase the intake of fresh and local plant-based foods. This aligns with the current trend to increase the ingestion of more sustainable food choices while reducing consumption of animal-based foods.
Steps involved in food preference and food selection, which through the bitter compounds in vegetables ultimately influences cardiovascular health. Umami substances can modulate various steps by suppressing the signaling of bitter receptors, modifying taste perception, and increasing food preference, thus helping regulate food choice and vegetable intake. The effectiveness of umami compounds varies with individual differences and with age: children tend to be more sensitive to the bitterness of food, and some older adults may lose the ability to taste some bitter compounds
References
Abe, S., Zhang, S., Tomata, Y., Tsuduki, T., Sugawara, Y., & Tsuji, I. (2020). Japanese diet and survival time: The Ohsaki cohort 1994 study. Clinical Nutrition, 39, 298–303.
Adler, E., Hoon, M. A., Mueller, K. L., Chandrashekar, J., Ryba, N. J. P., & Zuker, C. S. (2000). A novel family of mammalian taste receptors. Cell, 100, 693–702.
Afshin, A., Sur, P. J., Fay, K. A., Cornaby, L., Ferrara, G., Salama, J. S., Mullany, E. C., Abate, K. H., Abbafati, C., Abebe, Z., et al. (2019). Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the global burden of disease study 2017. Lancet, 11, 14.
Antos, A., Kwong, M. L., Balmorez, T., Villanueva, A., & Murakami, S. (2021). Unusually high risks of COVID-19 mortality with age-related comorbidities: An adjusted meta-analysis method to improve the risk assessment of mortality using the comorbid mortality data. Infectious Disease Reports, 13, 11.
Bachmanov, A. A., Bosak, N. P., Lin, C., Matsumoto, I., Ohmoto, M., Reed, D. R., & Nelson, T. (2014). Genetics of taste receptors. Current Pharmaceutical Design, 20, 2669–2683.
Bartoshuk, L., Duffy, V., & Miller, I. J. (1994). PTC/PROP tasting: Anatomy, psychophysics, and sex effects. Physiology & Behavior, 56, 1165–1171.
Bayer, S., Mayer, A. I., Borgonovo, G., Morini, G., Di Pizio, A., & Bassoli, A. (2021). Chemoinformatics view on bitter taste receptor agonists in food. Journal of Agricultural and Food Chemistry, 69, 13916–13924.
Bayram, B., González-Sarrías, A., Istas, G., Garcia-Aloy, M., Morand, C., Tuohy, K., García-Villalba, R., & Mena, P. (2018). Breakthroughs in the health effects of plant food bioactives: A perspective on Microbiomics, Nutri(epi)genomics, and metabolomics. Journal of Agricultural and Food Chemistry, 66, 10686–10692.
Beauchamp, G. K. (2009). Sensory and receptor responses to umami: An overview of pioneering work. The American Journal of Clinical Nutrition, 90, 723S–727S.
Beckerman, J. P., Tamez, M., Mattei, J., Alike, Q., Lovin, E., & Lovin, E. (2017). The development and public health implications of food preferences in children. Journal of Agricultural and Food Chemistry, 4, 66.
Behrens, M., & Meyerhof, W. (2013). Bitter taste receptor research comes of age: From characterization to modulation of TAS2Rs. Seminars in Cell & Developmental Biology, 24, 215–221.
Behrens, M., Foerster, S., Staehler, F., Raguse, J.-D., & Meyerhof, W. (2007). Gustatory expression pattern of the human TAS2R bitter receptor gene family reveals a Heterogenous population of bitter responsive taste receptor cells. The Journal of Neuroscience, 27, 12630–12640.
Boesveldt, S., & de Graaf, K. (2017). The differential role of smell and taste for eating behavior. Perception, 46, 307–319.
Breslin, P. (2013). An evolutionary perspective on food and human taste. Current Biology, 23, 9.
Centers for Disease Control and Prevention, People with Certain Medical Conditions. COVID-19., The Centers for Disease Control and Prevention (CDC). (2022). https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html
Chai, B. C., Reidar, J., van der Voort, K., Grofelnik, H. G., Eliasdottir, I. K., & Perez-Cueto, F. J. A. (2019). Which diet has the least environmental impact on our planet? A systematic review of vegan, vegetarian and omnivorous diets. Sustainability, 11, 4110.
Chandrashekar, J., Mueller, K. L., Hoon, M. A., Adler, E., Feng, L., Guo, W., Zuker, C. S., & Ryba, N. J. P. (2000). T2Rs function as bitter taste receptors. Cell, 100, 703–711.
Chandrashekar, J., Hoon, M. A., Ryba, N. J. P., & Zuker, C. S. (2006). The receptors and cells for mammalian taste. Nature, 444, 288–294.
Cicero, A. F. G., Fogacci, F., & Colletti, A. (2017). Food and plant bioactives for reducing cardiometabolic disease risk: An evidence based approach. Food & Function, 8, 2076–2088.
Clark, M. A., Springmann, M., Hill, J., & Tilman, D. (2019). Multiple health and environmental impacts of foods. Proceedings of the National Academy of Sciences of the United States of America, 116, 5.
Cui, M., Chen, B., Xu, K., Rigakou, A., Diamantakos, P., Melliou, E., Logothetis, D. E., & Magiatis, P. (2021). Activation of specific bitter taste receptors by olive oil phenolics and secoiridoids. Scientific Reports, 11, 22340.
Deshpande, D. A., Wang, W. C. H., McIlmoyle, E. L., Robinett, K. S., Schillinger, R. M., An, S. S., Sham, J. S. K., & Liggett, S. B. (2010). Bitter taste receptors on airway smooth muscle bronchodilate by localized calcium signaling and reverse obstruction. Nature Medicine, 16, 1299–1304.
Dotson, C. D., Zhang, L., Xu, H., Shin, Y.-K., Vigues, S., Ott, S. H., Elson, A. E. T., Choi, H. J., Shaw, H., Egan, J. M., Mitchell, B. D., Li, X., Steinle, N. I., & Munger, S. D. (2008). Bitter taste receptors influence glucose homeostasis. PLoS One, 3, e3974.
Drewnowski, A. (1997). Taste preferences and food intake. Annual Review of Nutrition, 17, 237–253.
Drewnowski, A., & Gomez-Carneros, C. (2000). Bitter taste, phytonutrients, and the consumer: A review. The American Journal of Clinical Nutrition, 72, 1424–1435.
Drewnowski, A., & Popkin, B. M. (1997). The nutrition transition: New trends in the global diet. Nutrition Reviews, 55, 31–43.
Drewnowski, A., & Poulain, J.-P. (2018). What lies behind the transition from plant-based to animal protein? AMA Journal of Ethics, 20, E987–E993.
Duffy, V. B., Hayes, J. E., Davidson, A. C., Kidd, J. R., Kidd, K. K., & Bartoshuk, L. M. (2010). Vegetable intake in college-aged adults is explained by Oral sensory phenotypes and TAS2R38 genotype. Chemosensory Perception, 3, 137–148.
Fanzo, J. (2018). Does global goal setting matter for nutrition and health? AMA Journal of Ethics, 20, E979–E986.
Fluitman, K., Hesp, A., Kaihatu, R., Nieuwdorp, M., Keijser, B. J., Ijzerman, R. G., & Visser, M. (2021). Poor taste and smell are associated with poor appetite, macronutrient intake, and dietary quality but not with undernutrition in older adults. The Journal of Nutrition, 151, 605–614.
Gervis, J. E., Fernández-Carrión, R., Chui, K. K., Ma, J., Coltell, O., Sorli, J. V., Asensio, E. M., Ortega-Azorín, C., Pérez-Fidalgo, J. A., Portolés, O., & Lichtenstein, A. H. (2022). Associations between taste perception profiles and empirically derived dietary patterns: An exploratory analysis among older adults with metabolic syndrome. Nutrients, 14, 142.
Halim, J., Bouzari, A., Felder, D., & Guinard, J. (2020). The salt Flip: Sensory mitigation of salt (and sodium) reduction with monosodium glutamate (MSG) in “better-for-you” foods. Journal of Food Science, 85, 2902–2914.
Harmon, C. P., Deng, D., & Breslin, P. A. S. (2021). Bitter taste receptors (T2Rs) are sentinels that coordinate metabolic and immunological defense responses. Current Opinion in Physiology, 20, 70–76.
Independent Expert Group. 2021. 2021 Global Nutrition Report: The state of global nutrition.
Keast, R. S. J., & Breslin, P. A. S. (2002). Modifying the bitterness of selected oral pharmaceuticals with cation and anion series of salts. Pharmaceutical Research, 19, 1019–1026.
Keast, R. S. J., & Breslin, P. A. S. (2003). An overview of binary taste–taste interactions. Food Quality and Preference, 14, 111–124.
Kemp, S. E., & Beauchamp, G. K. (1994). Flavor modification by sodium chloride and monosodium glutamate. Journal of Food Science, 59, 682–686.
Khan, M. S., Spann, R. A., Münzberg, H., Yu, S., Albaugh, V. L., He, Y., Berthoud, H.-R., & Morrison, C. D. (2021). Protein appetite at the Interface between nutrient sensing and physiological homeostasis. Nutrients, 13, 4103.
Kim, U., & Drayna, D. (2004). Genetics of individual differences in bitter taste perception: Lessons from the PTC gene. Clinical Genetics, 67, 275–280.
Kim, M. J., Son, H. J., Kim, Y., Misaka, T., & Rhyu, M.-R. (2015). Umami-bitter interactions: The suppression of bitterness by umami peptides via human bitter taste receptor. Biochemical and Biophysical Research Communications, 456, 586–590.
Kim, Y., Kim, E.-Y., Son, H. J., Lee, J.-J., Choi, Y.-H., & Rhyu, M.-R. (2017). Identification of a key umami-active fraction in modernized Korean soy sauce and the impact thereof on bitter-masking. Food Chemistry, 233, 256–262.
Kumakura, I. (2015). Introduction to Japanese cuisine, nature, history and culutre (1st ed.). Shunhari Initiative.
Lee, R. J., Xiong, G., Kofonow, J. M., Chen, B., Lysenko, A., Jiang, P., Abraham, V., Doghramji, L., Adappa, N. D., Palmer, J. N., Kennedy, D. W., Beauchamp, G. K., Doulias, P.-T., Ischiropoulos, H., Kreindler, J. L., Reed, D. R., & Cohen, N. A. (2012). T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. The Journal of Clinical Investigation, 122, 4145–4159.
Lipchock, S. V., Mennella, J. A., Spielman, A. I., & Reed, D. R. (2013). Human bitter perception correlates with bitter receptor messenger RNA expression in taste cells. The American Journal of Clinical Nutrition, 98, 1136–1143.
Matsuyama, S., Sawada, N., Tomata, Y., Zhang, S., Goto, A., Yamaji, T., Iwasaki, M., Inoue, M., Tsuji, I., & Tsugane, S. (2021). Association between adherence to the Japanese diet and all-cause and cause-specific mortality: The Japan public health center-based prospective study. European Journal of Nutrition, 60, 1327–1336.
Mattes, R. D. (2021). Taste, teleology and macronutrient intake. Current Opinion in Physiology, 19, 162–167.
McCabe, C., & Rolls, E. T. (2007). Umami: A delicious flavor formed by convergence of taste and olfactory pathways in the human brain. The European Journal of Neuroscience, 25, 1855–1864.
McCrickerd, K., & Forde, C. G. (2016). Sensory influences on food intake control: Moving beyond palatability. Obesity Reviews, 17, 18–29.
Mennella, J. A., & Bobowski, N. K. (2015). The sweetness and bitterness of childhood: Insights from basic research on taste preferences. Physiology & Behavior, 152, 502–507.
Mennella, J. A., Spector, A. C., Reed, D. R., & Coldwell, S. E. (2013). The bad taste of medicines: Overview of basic research on bitter taste. Clinical Therapeutics, 35, 1225–1246.
Mennella, J. A., Reiter, A. R., & Daniels, L. M. (2016). Vegetable and fruit acceptance during infancy: Impact of ontogeny, genetics, and early experiences. Journal of Agricultural and Food Chemistry, 7, 211S–219S.
Meyerhof, W., Batram, C., Kuhn, C., Brockhoff, A., Elke, C., Bufe, B., Appendino, G., & Behrens, M. (2010). The molecular receptive ranges of human TAS2R bitter taste receptors. Chemical Senses, 35, 157–170.
Micha, R., Khatibzadeh, S., Shi, P., Andrews, K. G., Engell, R. E., & Mozaffarian, D. (2015). Global, regional and national consumption of major food groups in 1990 and 2010: A systematic analysis including 266 country-specific nutrition surveys worldwide. BMJ Open, 5, e008705.
Milton, K. (2000). Back to basics: Why foods of wild primates have relevance for modern human health. Nutrition, 16, 480–483.
Mouritsen, O. G., & Styrbeak, K. (2020). Design and ‘umamification’ of vegetable dishes for sustainable eating. International Journal of Food Design, 5, 9–42.
Murakami, K., Livingstone, M., & Sasaki, S. (2018). Thirteen-year trends in dietary patterns among Japanese adults in the National Health and nutrition survey 2003–2015: Continuous westernization of the Japanese diet. Nutrients, 10, 994.
Ninomiya, K. (1998). Natural occurrence. Food Review International, 14, 177–211.
Ninomiya, K. (2016). Food science of dashi and umami taste. Yakugaku Zasshi, 136, 1327–1334.
Okiyama, A., & Beauchamp, G. K. (1998). Taste dimensions of monosodium glutamate (MSG) in a food system: Role of glutamate in young American subjects. Physiology & Behavior, 65, 177–181.
Okuno, T., Morimoto, S., Nishikawa, H., Haraguchi, T., Kojima, H., Tsujino, H., Arisawa, M., Yamashita, T., Nishikawa, J., Yoshida, M., Habara, M., Ikezaki, H., & Uchida, T. (2020). Bitterness-suppressing effect of umami dipeptides and their constituent amino acids on diphenhydramine: Evaluation by gustatory sensation and taste sensor testing. Chemical & Pharmaceutical Bulletin, 68, 234–243.
Pörtner, H.-O., D. C. Roberts, H. Adams, I. Adelekan, C. Adler, R. Adrian, P. Aldunce, E. Ali, R. Ara Begum, B. Bednar-Friedl, R. Bezner Kerr, R. Biesbroek, J. Birkmann, et. al. (2022). Climate Change 2022: Impacts, Adaptation and Vulnerability Contribution of Working Group II to the IPCC Sixth Assessment Report.
Puputti, S., Hoppu, U., & Sandell, M. (2019). Taste sensitivity is associated with food consumption behavior but not with recalled pleasantness. Food, 8, 444.
Reedy, J., Krebs-Smith, S. M., Miller, P. E., Liese, A. D., Kahle, L., Park, Y., & Subar, A. F. (2014). Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. The Journal of Nutrition, 144, 881–889.
Rhyu, M.-R., Kim, Y., & Misaka, T. (2020). Suppression of hTAS2R16 signaling by umami substances. International Journal of Molecular Sciences, 21, 7045.
Rolls, E. T. (2009). Functional neuroimaging of umami taste: What makes umami pleasant? The American Journal of Clinical Nutrition, 90, 804S–813S.
San Gabriel, A., & Uneyama, H. (2013). Amino acid sensing in the gastrointestinal tract. Amino Acids, 45, 451–461.
San Gabriel, A., Ninomiya, K., & Uneyama, H. (2018). The role of the Japanese traditional diet in healthy and sustainable dietary patterns around the world. Nutrients, 10, 173.
Sandell, M. A., & Breslin, P. A. (2006). Variability in a taste-receptor gene determines whether we taste toxins in food. Current Biology, 16, R792–R794.
Small, D. M. (2012). Flavor is in the brain. Physiology & Behavior, 107, 540–552.
Soares, S., Silva, M. S., Garcia-Estevez, I., Groβmann, P., Brás, N., Brandão, E., & Mateus, N. (2018). Human bitter taste receptors are activated by different classes of polyphenols. Journal of Agricultural and Food Chemistry, 66, 8814–8823.
Springmann, M., Godfray, H. C., Rayner, M., & Scarborough, P. (2016). Analysis and valuation of the health and climate change cobenefits of dietary change. Proceedings of the National Academy of Sciences of the United States of America, 113, 4146–4151.
Ssentongo, P., Ssentongo, A. E., Heilbrunn, E. S., Ba, D. M., & Chinchilli, V. M. (2020). Association of cardiovascular disease and 10 other pre-existing comorbidities with COVID-19 mortality: A systematic review and meta-analysis. PLoS One, 15.
Steinert, R. E., & Beglinger, C. (2011). Nutrient sensing in the gut: Interactions between chemosensory cells, visceral afferents and the secretion of satiation peptides. Physiology & Behavior, 105, 62–70.
Sterneder, S., Stoeger, V., Dugulin, C. A., Liszt, K. I., Di Pizio, A., Korntheuer, K., Dunkel, A., Eder, R., Ley, J. P., & Somoza, V. (2021). Astringent Gallic acid in red wine regulates mechanisms of gastric acid secretion via activation of bitter taste sensing receptor TAS2R4. Journal of Agricultural and Food Chemistry, 69, 10550–10561.
Tada, N., Maruyama, C., Koba, S., Tanaka, H., Birou, S., Teramoto, T., & Sasaki, J. (2011). Japanese dietary lifestyle and cardiovascular disease. Journal of Atherosclerosis and Thrombosis, 18, 723–734.
Tepper, B. J., White, E. A., Koelliker, Y., Lanzara, C., d’Adamo, P., & Gasparini, P. (2009). Genetic variation in taste sensitivity to 6-n-propylthiouracil and its relationship to taste perception and food selection. Annals of the New York Academy of Sciences, 1170, 126–139.
Thompson, R. C., Allam, A. H., Lombardi, G. P., Wann, L. S., Sutherland, M. L., Sutherland, J. D., Soliman, M. A., Frohlich, B., Mininberg, D. T., Monge, J. M., & Vallodolid, C. M. (2013). Atherosclerosis across 4000 years of human history: The Horus study of four ancient populations. Lancet, 381, 10.
Tilman, D., & Clark, M. (2014). Global diets link environmental sustainability and human health. Nature, 515, 518–522.
Toda, Y., Hayakawa, T., Itoigawa, A., Kurihara, Y., Nakagita, T., Hayashi, M., Ashino, R., Melin, A. D., Ishimaru, Y., Kawamura, S., Imai, H., & Misaka, T. (2021). Evolution of the primate glutamate taste sensor from a nucleotide sensor. Current Biology, 31, 4641–4649.
Tubiello, F. N., Rosenzweig, C., Conchedda, G., Karl, K., Gütschow, J., Xueyao, P., Obli-Laryea, G., Wanner, N., Qiu, S. Y., De Barros, J., Flammini, A., Mencos-Contreras, E., Souza, L., Quadrelli, R., Heiðarsdóttir, H. H., Benoit, P., Hayek, M., & Sandalow, D. (2021). Greenhouse gas emissions from food systems: Building the evidence base. Environmental Research Letter, 16.
Vincis, R., & Fontanini, A. (2019). Central taste anatomy and physiology. Handbook of Clinical Neurology, 164, 187–204.
WHO. (2019). World health statistics 2019: Monitoring health for the SDGs, sustainable development goals. WHO.
WHO. (2020a). Global Health estimates 2020: Deaths by cause, age, sex, by country and by region, 2000–2019. WHO.
WHO. (2020b). Global health estimates: Leading causes of DALYs: Disease burden 2000–2019. https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates/global-health-estimates-leading-causes-of-dalys
WHO. (2020c). Healthy Diet. is this https://www.who.int/news-room/fact-sheets/detail/healthy-diet or is it https://www.who.int/health-topics/healthy-diet#tab=tab_1 ??
Willett, W., Rockström, J., Loken, B., Springmann, M., Lang, T., Vermeulen, S., Garnett, T., Tilman, D., DeClerck, F., & Wood, A. (2019). Food in the anthropocene: The EAT-lancet commission on healthy diets from sustainable food systems. The Lancet, 393, 447–492.
Wooding, S. P., & Ramirez, V. A. (2022). Global population genetics and diversity in the TAS2R bitter taste receptor family Frontiers. Frontiers in Genetics, 13.
Wooding, S. P., Ramirez, V. A., & Behrens, M. (2021). Bitter taste receptors. Evolution, Medicine, and Public Health, 9, 431–447.
Yamaguchi, S. (1998). Badic properties of umami and its effects on food flavor. Food Review International, 14, 139–176.
Yamazaki, T., Narukawa, M., Mochizuki, M., Misaka, T., & Watanabe, T. (2013). Activation of the hTAS2R14 human bitter-taste receptor by (−)-epigallocatechin gallate and (−)-epicatechin gallate. Bioscience, Biotechnology, and Biochemistry, 77, 1981–1983.
Yeomans, M. R. (1998). Taste, palatability and the control of appetite. Proceedings of the Nutrition Society, 57, 609–615.
Zhang, S., Tomata, Y., Sugawara, Y., Tsuduki, T., & Tsuji, I. (2019). The Japanese dietary pattern is associated with longer disability-free survival time in the general elderly population in the Ohsaki cohort 2006 study. The Journal of Nutrition, 149, 1245–1251.
Acknowledgments
A.S. is an employee of Ajinomoto Co., Inc., and T.M.R. is an employee of Ajinomoto Health and Nutrition North America, Inc. The writing of this chapter was supported by the nonprofit International Glutamate Information Service (IGIS). The content does not necessarily represent the opinions of IGIS or the views of the companies but, rather, is the responsibility of the authors.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2024 The Author(s)
About this chapter
Cite this chapter
Gabriel, A.S., Rains, T.M. (2024). Umami Taste as a Component of Healthy Diets. In: San Gabriel, A., Rains, T.M., Beauchamp, G. (eds) Umami. Food and Health. Springer, Cham. https://doi.org/10.1007/978-3-031-32692-9_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-32692-9_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-32691-2
Online ISBN: 978-3-031-32692-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)