Abstract
Songbirds that occur across the diverse types of North American rangelands constitute many families within the Order Passeriformes, and hundreds of species. Most are declining, and many are considered potential indicator species for rangeland ecosystems. We synthesized information on the natural and life history, habitat requirements, conservation status, and responses to management of songbirds associated with North American grasslands and sagebrush steppe, two of the most geographically extensive types of rangelands. We provide a more targeted examination of the habitat associations and management considerations for two focal species, the grassland-obligate grasshopper sparrow (Ammodramus savannarum) and sagebrush-obligate Brewer’s sparrow (Spizella breweri). Grassland- and sagebrush-obligate species rely on expansive stands of grasslands and sagebrush, respectively, and we discuss how key ecological processes and rangeland management approaches—grazing, fire, and mechanical treatments—influence rangeland songbirds. Rangeland management practices can affect breeding songbirds considerably, primarily through the resultant structure and composition of vegetation, which influences the availability of preferred nesting substrates, refugia from predators, and foraging success. Optimal management strategies to limit negative consequences to rangeland songbirds will depend on the target species and local topoedaphic and climatic conditions. The maintenance of large, contiguous patches of native habitats and restoration of previously degraded areas will help facilitate the population persistence of rangeland-associated songbirds. Maintaining structural heterogeneity of habitats within landscapes, moreover, can facilitate local species diversity. Information pertaining to periods outside of the nesting stage is severely lacking for most species, which is concerning because effective management necessitates understanding of threats and limiting factors across the full annual life cycle. Moreover, information on disease effects and prevalence, the effects of a changing climate, and how both may interact with management strategies, also comprise key gaps in knowledge.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Brewer’s sparrow
- Conservation
- Grasshopper sparrow
- Grassland songbirds
- Habitat
- Management
- Sagebrush songbirds
1 Life/Natural History and Population Dynamics
The songbird species that inhabit North American rangelands have relatively fast life histories, with first breeding attempts typically occurring in the first year of adulthood. The distributions of some species are restricted (e.g., Baird’s sparrow [Centronyx bairdii]; Green et al. 2020), whereas the distributions of other species span multiple continents (e.g., horned larks [Eremophila alpestris]; Beason 2020). Most rangeland songbirds are migratory, and territorial on breeding grounds. Primary foods include arthropods during the breeding season and seeds during the winter. The annual life cycle of rangeland-associated songbirds can be classified as nesting, post-fledging, fall migration, over-wintering, and spring migration.
1.1 Nesting
Songbird males establish breeding territories shortly after arriving on breeding grounds in spring. Males often have elaborate courtship songs, and many combine songs with aerial displays. Song dialects can vary regionally, and males of some species (e.g., Brewer’s sparrow [Spizella breweri] and grasshopper sparrow [Ammodramus savannarum]) have different song types for pre- and post-pairing. Most species are socially monogamous at least within a breeding season, though some such as the bobolink (Dolichonyx oryzivorus) and dickcissel (Spiza americana) are polygynous, a mating system in which the desirable males will pair with more than one female (Renfrew et al. 2020; Temple 2020). Even for socially monogamous populations, extra-pair paternity can be common (e.g., Danner et al. 2018).
Nest placement is variable, with some species nesting on the ground amidst vegetation, and others within shrubs or trees. Nest structures typically are open or domed cups, constructed with sticks, grasses, forbs, and/or sedges, and lined with finer material such as rootlets, mammal hair or feathers of other species. Females lay one egg per day until clutch completion, and clutch sizes vary from approximately 2–7 eggs. Eggs develop and remain viable within a specific range of temperatures regulated by incubation (Deeming 2001). Incubation is conducted primarily by females, although males contribute in some species, and females are sometimes provisioned with food on the nest by their mates. Incubation periods typically range from 10 to 13 days for open-cup nesters, whereas cavity nesters incubate for longer periods. The incubation for the juniper titmouse (Baeolophus ridgwayi), for example, is approximately 17 days (Cicero et al. 2020). Nestlings are altricial and highly dependent upon parental care for food and thermoregulation. Nestling periods range from 8 to 14 days for most species, and young are almost always fed by both parents.
Songbird nest survival varies across habitat conditions, sites, and years. The primary source of nesting mortality is predation from a wide variety of species including snakes, rodents, mustelids, canids, domesticated or feral cats (Felis catus), raccoons (Procyon lotor), raptors, shrikes (Lanius spp.) and even ungulates including deer (Odocoileus spp.,) and elk (Cervus canadensis) (Pietz and Granfors 2000; Renfrew and Ribic 2003; Hethcoat and Chalfoun 2015a; Lyons et al. 2015). Many songbirds also experience brood parasitism by the brown-headed cowbird (Molothrus ater) (Shaffer et al. 2019a), though some species such as sage thrashers (Oreoscoptes montanus) remove cowbird eggs from their nests (Reynolds et al. 2020). Other causes of nesting failures include extreme weather events, such as snowstorms or hail (Hightower et al. 2018), and anthropogenic activities.
1.2 Post-fledging
Songbird nestlings typically depart nests before they are fully capable of flight. Mortality from predation or inclement weather during the early post-fledging period can therefore be high for most if not all species (e.g., Fisher and Davis 2011; Hovick et al. 2011). Fledglings are fed by parents for at two least weeks after leaving the nest, achieving adult body mass within about a month (Jones et al. 2018). Family groups likely rely on habitats with sufficient cover to shelter young from predators and the elements (Fisher and Davis 2011). Unfortunately, the post-fledging period for many songbirds rarely is studied (Davis and Fisher 2009; Ribic et al. 2018, 2019), and estimates of post-fledging habitat use and survival are lacking. Where studied, estimates of fledgling survival range from 26 to 36% (Yackel Adams et al. 2006; Berkeley et al. 2007; Hovick et al. 2011; Young et al. 2019). Nestling body condition and wing development, which vary with food availability and provisioning rates, tend to be positively related to post-fledging survival (Yackel Adams et al. 2006; Jones et al. 2017; Jones and Ward 2020).
1.3 Non-breeding
Most songbirds inhabiting North American rangelands during the breeding season are migratory, although some populations inhabiting southern areas are year-round residents. Adults typically complete a full molt of their feathers towards the end of the nesting season, and migrants often form large, single, or mixed-species flocks for southward migration. The length of migration distances ranges from short to long, with many species over-wintering in the southwestern U.S. and northern Mexico (e.g., chestnut-collared longspur [Calcarius ornatus]), and others that migrate to South America (e.g., bobolink and dickcissel). Flocks periodically use migratory stopover habitats to forage and rest. Over-wintering migrants tend to use habitats similar in structure to their breeding habitats (Igl and Ballard 1999; Hovick et al. 2014).
1.4 Survival and Sources of Mortality
Songbird nests are depredated by a wide variety of species (see Nesting section). For most songbird species, much less is known about predator species and rates of predation during the post-fledging, migratory, and over-wintering periods, though many species of raptors (e.g., accipters, falcons) are known to kill adult songbirds (Lima 2009). Fledglings are consumed by raptors, corvids, shrikes, snakes, and mammals (Yackel Adams et al. 2006; Berkeley et al. 2007; Hovick et al. 2011; Young et al. 2019). Free-ranging domestic and feral cats kill billions of songbirds in North America each year (Loss et al. 2013a). Other sources of adult mortality of songbirds include collisions with buildings, vehicles, guy wires extending from communication towers, and wind turbines (Longcore et al. 2012; Loss et al. 2013b; Erickson et al. 2014).
1.5 Seasonal Movements and Dispersal
Movement and dispersal data are rare for most rangeland songbirds. Historically, the logistical challenges of safely radio-tracking very small birds were an impediment. Recent technological advances, however, have enabled the manufacture of smaller, lighter transmitters and light-level geolocators that record the movements and locations of small birds across time upon recapture. Soon after independence, immature birds join post-breeding flocks of adults, leave their natal area, and begin moving with pre-migratory flocks (e.g., Temple 2020). An understanding of the connectivity between the breeding grounds and particular migration routes or over-wintering areas is lacking for most grassland and sagebrush songbird species. Site fidelity, or the repeated return, to breeding sites varies across species, habitats and locations. Juveniles sometimes return to the general area where they were born (natal philopatry; e.g., Renfrew et al. 2020). Some species appear to be facultatively nomadic on breeding grounds between years (e.g., chestnut-collared longspur and lark bunting [Calamospiza melanocorys]), likely as an evolved response to shifting habitat suitability associated with the unpredictable influences of fire, drought, and the movements and grazing of bison (Bison bison) herds (Green et al. 2019).
1.6 Population Dynamics
Whereas offspring mortality during the nesting (e.g., Kerns et al. 2010; Hethcoat and Chalfoun 2015b; Verheijen et al. 2022) and post-fledging (e.g., Young et al. 2019) periods can be high, a lack of research encompassing the full annual life cycle constrains an understanding of which life stages tend to be most limiting for rangeland songbirds (Marra et al. 2015). Because most songbirds have relatively fast life histories, the influence of reproductive success (clutch size, nest survival, post-fledging survival) on population growth likely is high (Saether and Bakke 1997). Nest density also may influence population growth, as avian productivity within an area is the product of per capita nest survival and density (Pulliam et al. 2021). Moreover, carryover effects from over-wintering grounds and migratory stopover sites can affect the timing and reproductive success of songbirds via the interaction between arrival times and food availability, and the condition of adults at the onset of nesting (Bayly et al. 2016).
2 Current Species and Population Status
Most populations of songbirds that breed within North American rangelands are declining, some drastically, concomitant with broad-scale habitat loss and alteration (Table 12.1; Rosenberg et al. 2019; Sauer et al. 2020). For example, of 34 species of New World sparrows, which include scrub-successional, aridland, and grassland species, 17 exhibited significant declines and 27 had negative trend estimates (Sauer et al. 2013). Moreover, numbers of grassland and aridland birds declined by an estimated 55% and 23%, respectively, during 1970–2017 (Rosenberg et al. 2019).
3 Population Monitoring
There is no monitoring program devoted specifically to rangeland songbirds, though many populations are monitored as part of broader efforts. The North American Breeding Bird Survey (BBS) and Christmas Bird Count (see Chap. 11) are used frequently to assess the status and general trends of rangeland songbirds (Table 12.1). Laurent et al. (2012) provide details on these and other national and regional programs, such as the Strategic Multi-scale Grassland Bird Population Monitoring Protocol (SMGBPM) and Monitoring Avian Productivity and Survivorship (MAPS). The SMGBPM uses counties as management units and was developed because of the concern that BBS may underestimate grassland bird numbers in some areas. MAPS utilizes a network of mist-netting efforts and mark-recapture analyses to assess demographic parameters including annual survival and productivity of North American birds over time. Citizen-science programs include eBird, which is an online database of bird observations, and NestWatch, which focuses on reproductive success. Finally, several facilitated databases, including the Avian Knowledge Network, store data that land managers, scientists, and others can access for research and conservation (Laurent et al. 2012).
Monitoring programs vary in their degree of statistical rigor, spatial inference, and limitations. Selection of monitoring data on which to base research or management decisions should therefore depend on the desired metrics (e.g., occupancy, distribution, abundance trends over time, productivity, species richness) and precision. The BBS, for example, was established in 1966 and has been valuable for documenting general population trends of over 400 North American bird species. Surveys, however, are conducted as annual roadside routes, which may under-sample species sensitive to human infrastructure. Current protocols also do not account for potential spatiotemporal differences in the probability of detecting birds. The Integrated Monitoring in Bird Conservation Regions, coordinated by the Bird Conservancy of the Rockies, incorporates randomized sampling and was designed to provide robust estimates of avian occupancy and density across time and multiple spatial scales.
4 Habitat Associations
Songbird species inhabiting the grasslands and arid shrublands of North America (see Table 12.1 for representative species) include habitat specialists (or “obligates”) and those that are open-country generalists. The thick-billed longspur (Rhynchophanes mccownii), for example, is a shortgrass-prairie specialist with a restricted distribution (With 2021). By contrast, the western meadowlark (Sturnella neglecta) inhabits a wide variety of open habitat types and agricultural fields throughout the entire western portion of North America (Davis and Lanyon 2020). The assemblage of songbird species that occupies a given site varies by geographic location, vegetation type, habitat structure, and extent of habitat degradation (Fig. 12.1).

Source photograph credits include Jack Parlapiano (titmouse), Tayler Scherr (thrasher), Rick Bohn (chestnut-collared longspur), Dan Casey (thick-billed longspur), Dave Lambeth (bobolink), Sarah McIntire (juniper), and Anna Chalfoun (sagebrush). Other plant photos drawn from open sources
Artistic rendering of representative songbird and plant species within five of the major rangeland types in North America. Plant species from left to right include western juniper (Juniperus occidentalis), big sagebrush (Artemisia tridentata), buffalo grass (Bouteloua dactyloides), blue grama (Bouteloua gracilis), and big bluestem (Andropogon gerardii). Assemblages are organized by relative longitude. Artwork by Bethann Merkle.
The habitat preferences of rangeland songbirds evolved based on the conditions most associated with successful survival and reproduction (Nelson et al. 2020). Such preferences often are scale-dependent (Chalfoun and Martin 2007; Lipsey et al. 2017; Box 12.2). Several species may inhabit the same area but primarily nest or forage within more differentiated niches (Grinnell 1917). Such differences likely arose to limit competition for nest sites and food. Within mixed-grass prairie, for example, the nest sites of sympatric songbirds are distributed across microhabitat gradients ranging from shorter, sparser vegetation (e.g., thick-billed longspur) to taller, denser grasses (e.g., western meadowlark; Fig. 12.1). The vertical and horizontal partitioning of nest sites within an area may benefit the reproductive success of co-occurring songbird species by reducing predator search efficiency (Martin 1993). The maintenance of microhabitat heterogeneity within landscapes is therefore a key management consideration. Boxes 12.1 and 12.2 provide more in-depth descriptions of habitat associations for two focal species, a grassland-obligate songbird (grasshopper sparrow) and a sagebrush-steppe obligate (Brewer’s sparrow).
5 Rangeland Management
The primary ecological processes and management practices that influence rangeland songbirds are grazing, fire, and mowing, whereas mechanical management practices include the application of herbicides and pesticides, mowing, chaining, and discing (reviewed in Shaffer and DeLong 2019). Some management interventions, such as bison grazing and fire, often are geared towards mimicking historical disturbance regimes (see Chaps. 6 and 8), whereas mechanical management practices may be used to produce similar outcomes but within faster time frames. These management approaches may be used singly or in combination (e.g., patch-burn grazing). Ecological processes and management practices influence local avian biodiversity primarily through their effect on vegetation structure and composition. A management approach will have variable outcomes depending on timing, intensity, and frequency. Timing, or seasonality, refers to when during the year a management approach is applied. Intensity refers to the degree to which a management approach is applied. In terms of fire, intensity is the amount of heat produced (Chap. 6), whereas for grazing, intensity refers to the number of grazing animals and length of time grazing occurs, or how much biomass is removed. Frequency refers to how often ecological processes or management practices have been applied, either within or among seasons (Chap. 4).
Management approaches depend on goals, and outcomes often are site- or species-specific. Management guidance for individual species is summarized in the accounts constituting Johnson et al. (2019). Thorough coverage of management approaches for grasslands bird species can be found in Sample and Mossman (1997), whereas management considerations pertaining to sagebrush species can be found in Paige and Ritter (1999) and Walker et al. (2020). The two case studies in this chapter (Boxes 12.1 and 12.2) illustrate the complexity of the decisions involved in the application of ecological processes and management practices that maintain and create habitat for specific songbird species.
5.1 Grazing
Direct effects of livestock grazing on rangeland songbird species are rare and include trampling of eggs, nestlings, or adults, and in some cases apparent predation (Nack and Ribic 2005; Bleho et al. 2014). Nest destruction by livestock generally increases with grazing intensity during the nesting season, though for some species of songbirds, the creation of habitat via grazing may offset the minimal nest losses (Owens and Myres 1973; Bleho et al. 2014). Indirect effects of livestock grazing include alteration in vegetation structure (e.g., decreased litter cover, increased bare ground) and composition (e.g., dominance of some plant species over others). Such changes can lead to altered insect food availability or nest predation risk (Johnson et al. 2012). Indirect effects of grazing therefore tend to be more impactful than direct effects in influencing whether grazed rangelands comprise high-quality nesting habitat (Cody 1985; Martin and Possingham 2005). Additional indirect effects of grazing may include increased nest parasitism by brown-headed cowbirds or increased nest predator populations, via the addition of water, feed, and carcasses (Goguen and Mathews 1999, 2000; Coates et al. 2016). However, parasitism rates within grasslands also are influenced by the landscape matrix within which pastures are located. Brown-headed cowbirds may be less likely to parasitize nests of grassland songbirds where tree cover on the landscape is greater and nests of woodland species are readily available as alternative cowbird hosts (Pietz et al. 2009; Hovick and Miller 2013).
Livestock grazing can be used to manipulate vegetation to create desired conditions for rangeland songbirds (Derner et al. 2009; Bleho et al. 2014). The effects of grazing on vegetation, however, can be highly variable and affected by grazing regime (Chap. 4), livestock characteristics (species, breed, sex, age, and genetic factors), precipitation (amount, seasonality), current vegetation structure and composition, soil characteristics, historical land use, and presence and types of other disturbances (Briske et al. 2008; Sliwinski and Koper 2015; Lipsey and Naugle 2017). Thus, prior to implementing a grazing system within any given year to obtain a desired vegetation structure (e.g., habitat outcome; Pulliam et al. 2020, 2021), the characteristics of that grazing system may need to be modified based on the region’s expected precipitation and other aforementioned factors.
Long- and short-term monitoring of the effects of grazing on vegetation structure is important in terms of assessing the effects of grazing on avian abundance, community composition, and reproduction (Pulliam et al. 2021). Short-term effects include the reduction of herbaceous cover or height of vegetation, which can affect songbird species dependent on litter and grass cover for nest concealment. Long-term effects can manifest as altered composition of plant species or reduced vegetation productivity of a site (Briske et al. 2008). For example, repeated livestock grazing can affect shrub and tree establishment, thereby affecting songbird species dependent on non-herbaceous vegetation (Bock et al. 1993). Accordingly, rangeland songbird species may respond differently to grazing-induced changes over time (e.g., Johnson et al. 2011; Sliwinski and Koper 2015). Most species that are affected negatively by grazing are those that are dependent on relatively dense herbaceous ground cover or heavy shrub cover for nesting and foraging. The responses of species in sagebrush and montane coniferous habitats to livestock grazing, however, remain understudied.
Albeit not yet well understood, native grazers such as bison and prairie dogs (Cynomys spp.) may influence songbirds and their habitats differently than livestock (Allred et al. 2011a). The abundance of vesper and grasshopper sparrows in Montana were more abundant in pastures grazed year-round by bison compared with those that were grazed seasonally by cattle, although the abundance of 7 other songbird species and diversity measures did not vary by grazing type (Boyce et al. 2021). The grazing and fossorial activities of prairie dogs have played an influential role in the maintenance and composition of grassland and arid shrubland communities and can facilitate co-occurring bird species (Duchardt et al. 2019, 2021; Chap. 15).
5.2 Fire
As with grazing, the direct effects of fire on songbird species include the destruction of nests and young. Indirect effects involve altered vegetation characteristics as influenced by the timing, intensity, and frequency of fire applications (Chap. 6). In most rangeland systems, fire will reduce the biomass of live and dead herbaceous vegetation and shrub or tree cover, depending on fire intensity, and stimulate regrowth of herbaceous species through nutrient recycling (Sample and Mossman 1997). Responses of rangeland songbirds to fire are a function of each species’ preferences for the resultant post-burn vegetation conditions, and responses may change with time since fire as vegetation recovers. Grassland songbirds occur in fire-evolved ecosystems that historically had more frequent fires than sagebrush ecosystems (Chap. 6). Prescribed fire, therefore, is applied more frequently in grasslands to maintain songbird habitat than in shrubsteppe.
The timing of prescribed burning is an important consideration. Prescribed fire applied outside of the breeding season precludes the destruction of nests and allows for vegetation regrowth before the nesting season (Higgins 1986; Sample and Mossman 1997). Spring burns, however, can be most effective at suppressing the spread of invasive plant species by damaging plants during a vulnerable growth stage (Shaffer and DeLong 2019).
The consideration of historical fire-return intervals within regions and rangeland types is critical for the maintenance of songbird habitat. For example, in low-elevation, xeric sagebrush habitats, the invasion of nonnative annual grasses such as cheatgrass (Bromus tectorum) increases fuel loads, fire frequency, extent, and severity; and reduces shrub cover, which affects habitat suitability for shrub-dependent birds (Knick et al. 2005; Pilliod et al. 2017). Even in fire-dependent grasslands, fire-return intervals shortened relative to historical regimes can result in changes in the composition and structure of vegetation, with resultant reduced habitat quality for some grassland songbird species (Zimmerman 1997; Reinking 2005; With et al. 2008). For example, annual fires can eliminate the residual cover used as avian nesting substrates. Conversely, lengthened fire-return intervals, and especially the suppression of wildfires, may cause the expansion of woody vegetation into previously vast expanses of grassland and high-elevation sagebrush steppe (Grant and Murphy 2005; Noson et al. 2006; Anderson and Steidl 2019).
5.3 Mowing
Mowing uniformly reduces vegetation height, woody vegetation, and litter (Herkert et al. 1996; Sample and Mossman 1997). Mowing can therefore be implemented as a management tool for some grassland songbirds that prefer such conditions, both within the current harvest year (Mabry and Harms 2020) and occasionally the subsequent year (Igl and Johnson 2016). However, mowing can have direct negative effects on ground-nesting songbirds if conducted during the breeding season because nests may be abandoned or destroyed, or incubating adults, eggs, nestlings, and recently fledged young may be killed (Bollinger et al. 1990). Indirect effects of mowing include the reduction of invertebrate populations that serve as important prey for breeding birds (Zalik and Strong 2008). Plant species composition also can be affected over longer time scales with repeated mowing (Sample and Mossman 1997; Allen et al. 2001).
Effects of haying on songbirds depend on the timing and frequency of disturbance. Traditional hayland practices employed by agricultural producers aim to maximize the amount and quality of forage and typically involve an early initial cutting and one or more subsequent harvests that coincide with the avian breeding cycle, which can negatively affect avian reproductive success. Fields that are mowed multiple times within a breeding season and with short intervals between mowing may therefore cause complete avian reproductive failure (Rodenhouse et al. 1995). Conservation-focused haying strategies aim to avoid negative effects on birds by conducting operations after the nesting period (after mid-July or August, depending on location), haying periodically but not annually, and leaving portions of fields un-mowed (Shaffer and DeLong 2019).
5.4 Managing for Heterogeneity
A primary goal of livestock producers is to facilitate livestock growth via the maximal consumption of vegetation, which depending on management can decrease vegetation heterogeneity (variation in plant species composition and structure; Chaps. 8 and 9). Such practices can promote the dominance of a few plant species that are valuable to domesticated livestock but do not necessarily facilitate biological diversity. Traditional grazing systems (Chap. 4) wherein beef production is a primary objective, and without the use of fire, can therefore be insufficient in providing the vegetation heterogeneity required to support a diverse local suite of grassland birds (Sliwinski et al. 2019, 2020). In some situations, increasing the habitat heterogeneity within the overall landscape or region for biodiversity may entail managing for conditions that are rare or absent in surrounding areas.
In some grassland ecosystems of the Great Plains, patch-burn grazing, also known as pyric-herbivory, has been promoted as an alternative rangeland management strategy that aims to increase vegetation heterogeneity and avian and vegetation biodiversity while maintaining profitability for livestock producers (Fuhlendorf and Engle 2001; Allred et al. 2011b; Neilly et al. 2016). Patch-burn grazing entails shifting mosaics of burned patches designed to influence grazing distribution and increase vegetation heterogeneity (Fuhlendorf et al. 2006). Where fire is not a feasible management option, local habitat heterogeneity can be enhanced by herding, strategic placement of salt, minerals, or fencing, or alteration of stocking rates and season of use (Scasta et al. 2015; Sliwinski et al. 2019). The extent to which grazing may be used to increase local habitat heterogeneity will depend also on the spatial and habitat use of cattle, which tend to vary with factors such as topography, soils, water, and stocking rate (Bailey 2005; Rivero et al. 2021; Chap. 4). Cattle tend to decrease habitat selectivity under high stocking rates, which can increase habitat homogeneity (Rivero et al. 2021).
In sagebrush steppe, habitat heterogeneity within a landscape that provides for the entire suite of songbirds may be facilitated through shifts in the relative dominance of woody versus herbaceous vegetation, and promoting both sagebrush and other shrub species in patches of various heights (Knick et al. 2008; Hanser and Knick 2011; Miller et al. 2017). Heterogeneity in plant structure and composition in sagebrush-dominated systems can be influenced by grazing management (Veblen et al. 2014) or reintroduction of fire into communities (e.g., mountain big sagebrush) that have experienced prolonged fire exclusion both of which can help maintain plant diversity (Manier and Hobbs 2006; Davies and Bates 2020). If sagebrush-obligate songbirds (Brewer’s sparrow; sagebrush sparrow, Artemisiospiza nevadensis; sage thrasher, Oreoscoptes montanus) are of primary management interest, the prioritization of areas with relatively tall shrubs (50–200 cm) and high (greater than > 25%) shrub cover is paramount (Chalfoun and Martin 2007; Martin and Carlson 2020; Reynolds et al. 2020). Given the extensive loss of sagebrush habitat range-wide, and the agricultural value of areas within the sagebrush steppe consisting of more mesic, well-drained soils, such conditions have become rare (Knick et al. 2008).
6 Disease
The effects of disease on rangeland songbirds are poorly studied. West Nile Virus has been detected in several rangeland-inhabiting species including the bobolink, brown-headed cowbird, black-chinned sparrow (Spizella atrogularis), field sparrow (Spizella pusilla), lark sparrow (Chondestes grammacus), pinyon jay (Gymnorhinus cyanocephalus), and Savannah sparrow (Passerculus sandwichensis) (Centers for Disease Control 2016). Avian pox viruses have been recorded for sagebrush sparrow and Savannah sparrow (Martin and Carlson 2020; Wheelwright and Rising 2020). Songbirds are affected by outbreaks of salmonellosis, which has a high mortality rate; however, the extent to which this disease affects rangeland songbirds in particular is largely unknown. Species that congregate in flocks and are exposed to contaminated feces appear to be most at risk. Some rangeland songbirds may therefore be vulnerable, including those that use feeders or roost in groups. Brown-headed cowbirds seem to be particularly at risk and may serve as a reservoir for salmonellosis, possibly influenced by their association with cattle (Tizard 2004).
Parasites, such as bird blowflies (Protocalliphora spp. and Trypocalliphora braueri), are widespread in songbirds and can inflict serious harm. Effects of blowflies have included reduced nestling survival and fledging success for sage thrashers (Howe 1992), reduced tarsi length for sagebrush sparrow nestlings (Peterson et al. 1986), and retarded feather growth for Savannah sparrow nestlings (Bedard and McNeil 1979). Detrimental effects of ectoparasites on songbird nestlings can be ameliorated by increased food availability and feeding rates by adults, but exacerbated by environmental conditions that decrease foraging opportunities (e.g., adverse weather; Howe 1992; De Lope et al. 1993; Tripet and Richner 1997). Finally, rangeland songbirds also may experience anemia from haematophagous parasites, to a largely unknown extent (Boyd 1951).
7 Ecosystem Threats
7.1 Habitat Conversion and Alteration
The biggest collective threat to rangeland songbird species is habitat loss, fragmentation, and degradation. Large and rapid declines in grassland and aridland species often are linked to the loss and alteration of habitat on breeding grounds (Sauer et al. 2013; Rosenberg et al. 2019). Historically, agricultural practices, and particularly cropland agriculture, have been the greatest causes of native grassland and sagebrush loss in North America (Knick et al. 2003; Rosenberg et al. 2019). Urban development and sprawl in exurban areas, and development for energy resources, have caused further habitat loss and fragmentation (Marzluff and Ewing 2001; Northrup and Wittemyer 2013). The spread of invasive plant species and woody encroachment also causes degradation in habitat quality for songbird species (Archer et al. 2017).
7.2 Energy Development
Portions of North American rangelands coincide with on-going energy extraction, including oil, natural gas, and wind (Northrup and Wittemyer 2013). Effects of oil and gas development on rangeland songbirds include reduced abundance, altered habitat use, and reduced reproductive success (Gilbert and Chalfoun 2011; Kalyn Bogard and Davis 2014; Thompson et al. 2015; Chalfoun 2021 and references therein). Habitat alteration associated with energy development activities can alter trophic dynamics among wildlife species and result in decreased reproduction or survival. In Wyoming’s sagebrush steppe, for example, the nest success of three sagebrush-obligate songbird species decreased with adjacent surface disturbance from natural gas development (Hethcoat and Chalfoun 2015a, b). Nest failures were attributed primarily to increased abundance of rodent nest predators that were attracted to the re-seeded areas surrounding well pads, pipelines and roads (Sanders and Chalfoun 2018).
Activities associated with energy development simultaneously alter many characteristics within landscapes in addition to the footprint, including human activity, noise, and lighting. Yet, the specific mechanisms underlying avian responses are rarely tested or understood (Jones et al. 2015; Chalfoun 2021; but see Bernath-Plaistad and Koper 2016; Mejia et al. 2019). Wind facilities can cause both direct (mortality due to turbine strikes; Allison et al. 2019) and indirect (reduced reproductive success, avoidance of suitable habitat; Mahoney and Chalfoun 2016; Shaffer and Buhl 2016; Shaffer et al. 2019b) effects on rangeland songbirds. Solar installations are increasing in parts of the western U.S. and may pose additional management challenges (Loss 2016).
7.3 Invasive Species
Invasive plant species can affect rangeland songbird habitat in a myriad of ways. Many species of rangeland songbirds occur in areas that contain non-native plants, and use them for various activities including nesting or perching (e.g., Ruehmann et al. 2011; Nelson et al. 2017). Evaluation of the extent to which such use has negative consequences for songbirds, however, has implications for the growth of avian populations (e.g., Ruehmann et al. 2011; Nelson et al. 2018). Moreover, a few species of invasive plants, including cheatgrass, can exert such influence that they change the overall functioning of ecosystems and substantially eliminate or alter songbird habitat (Brooks et al. 2004; Knick et al. 2005; Coffman et al. 2014; Bestelmeyer et al. 2018).
The effects of invasive plants on songbirds include the alteration of habitat structure or composition that can influence habitat use, movements, abundance, survival, or reproductive success in a context- and species-specific manner (Stoleson and Finch 2001; Hovick and Miller 2013; Nelson et al. 2017; Stinson and Pejchar 2018). The abundance of songbirds in the northern mixed-grass prairie, for example, decreased slightly or remained the same with exotic grass encroachment (Pulliam et al. 2020). The cover of exotic grass, however, co-varied with herbaceous biomass. Areas with high leafy spurge (Euphorbia esula) in North Dakota decreased the breeding densities of some, but not all, species of grassland songbirds (Scheiman et al. 2003). Similarly, patterns of occurrence of songbirds in Saskatchewan between native pastures and those partially comprised of crested wheatgrass (Agropyron cristatum) were mixed (Davis and Duncan 1999). Relationships between songbird reproductive success and invasive plants generally have been neutral or positive (Stinson and Pejchar 2018, but see Lloyd and Martin 2005). Other indirect effects include altered prey availability, because native plants typically support more abundant and diverse invertebrate assemblages (Hickman et al. 2006; Litt et al 2014) which can influence reproductive parameters such as nestling growth (Lloyd and Martin 2005). However, the nestling mass of Botteri’s sparrows (Peucaea botterii) and several other species of grassland songbirds was unaffected by invasive grasses (Jones and Bock 2005; Kennedy et al. 2009).
Examples of invasive woody plant species include eastern redcedar (Juniperus virginiana) in southern grasslands (Archer et al. 2017), Pinus spp. and Juniperus spp. in sagebrush communities (Knick et al. 2014), and willow (Salix spp.) and aspen (Populous tremuloides) in northern grasslands (Grant et al. 2004). Woody encroachment alters both the vertical and horizontal characteristics of vegetation communities, and in some cases results in monocultures with little to no understory (Frost and Powell 2011; Archer et al. 2017; Nackley et al. 2017). These vegetation changes often cause avian species turnover and shifts in avian community composition (Grant et al. 2004; Anderson and Steidl 2019). Changes in habitat quality can occur within grasslands with woody encroachment via altered nest predation and brood parasitism rates, and decreased food availability and quality (Archer et al. 2017). In the Great Plains, for example, eastern red cedar encroachment has increased the habitat fragmentation of remnant grassland patches, with resultant decreases in the abundance of rangeland songbirds, at least partially to increased rates of nest predation (Coppedge et al. 2001; Engle et al. 2003). Similar fragmentation effects and reduced avian abundances have occurred in areas where western juniper (Juniperus occidentalis) has expanded into sagebrush steppe (Noson et al. 2006).
7.4 Climate Change
By one estimate, 53% of North American bird species are projected to lose more than half of their current geographic range across three scenarios of climate change by the end of the century (Langham et al. 2015). Grassland habitats and birds are expected to be particularly affected by climate change. Nearly half (42%) of grassland breeding bird species were deemed highly vulnerable under a scenario of a 3.0 °C increase in global mean temperature (Wilsey et al. 2019). Sagebrush songbirds similarly have been deemed threatened with respect to changing climate (Fleishman et al. 2014; National Audubon Society 2014; Nixon et al. 2016).
Spatial and temporal variation in precipitation and temperature influence the occurrence, distribution, and reproductive success of rangeland songbird species (Rotenberry and Wiens 1991; Shaffer and DeLong 2019). Years with moderate moisture and temperatures tend to lead to the highest reproductive output for rangeland songbirds, with implications for increasing variation in precipitation regimes (Ludlow et al. 2014; Conrey et al. 2016; Ruth and Skagen 2018). Increasing intensity of storms, such as those producing hail, can result in local mortality of young and adults tending nests (Carver et al. 2017; Hightower et al. 2018). Moreover, increasing temperatures and drought frequency in the western U.S. will likely decrease the productivity of nesting birds (Skagen and Yackel Adams 2012), especially in areas with higher habitat loss (Zuckerberg et al. 2018). Mismatches between the timing of peak availability of invertebrate prey and peak nesting activity also are likely to continue to become more common with a changing climate, which can lower reproductive success (Lany et al. 2016).
8 Conservation and Management Actions
8.1 Reversing the Loss and Fragmentation of Native Grasslands and Shrublands
One aspect central to all wildlife conservation is the necessity to maintain large and relatively intact landscapes, most of which are at least partially composed of private lands. Landscape protection therefore necessitates broad coalitions and partnerships (e.g., Chap. 27).
8.2 Habitat Management
Given the complexities of the short- and long-term effects of management activities on vegetation and birds in rangelands, and differences in preferred habitat across species, a universal approach to managing rangelands for songbirds does not exist (Duchardt et al. 2019, Shaffer and DeLong 2019). The management practices that facilitate the habitat needs of one species will not necessarily meet the needs of others. Ideal management prescriptions will therefore depend upon specific goals. Because some songbird species are more imperiled than others, a focus on managing for the species of highest conservation concern may be warranted in some scenarios (Herkert et al.1996). Alternatively, management might focus on sensitive species with limited breeding ranges, and whose core breeding ranges occur within the land manager’s jurisdiction. Management suggestions pertaining to individual species can be found in Shaffer and DeLong (2019) and Boxes 12.1 and 12.2 herein. The maintenance of heterogeneity within landscapes can provide the requisite microhabitat diversity for the success of individual songbird species and support a variety of species (Engle et al. 2003; Powell 2006). Patches (e.g., sandy draws) within landscapes consisting of tall shrubs and/or higher shrub cover, for example, support the highest breeding densities of sagebrush-obligate songbird species (Chalfoun and Martin 2007; Williams et al. 2011), and other declining species such as the loggerhead shrike (Lanius ludovicianus).
Other factors that influence the effectiveness of management for songbird habitat are regional differences in dominant vegetation types (e.g., warm-season or cool-season grasses), rangeland health (degree of degradation and level of biotic diversity), microclimate, and soil type (Shaffer and DeLong 2019). The previous and current land uses of a management unit also warrant consideration. Rangeland management for the conservation of birds may include ongoing maintenance of extant or degraded native grasslands or shrublands, and restoration of areas that had been converted for another use (e.g., agricultural production) to a more native state. Emulating historic, natural disturbances that resulted in a mosaic of habitats and vegetation structure can facilitate habitat heterogeneity and avian diversity. Resource managers may need to experiment with combinations of management tools at different sites with varying soil moisture conditions to maintain the array of habitats required to facilitate the biotic diversity of rangeland ecosystems (Ryan 1990).
Given limited resources for conservation, the premise is that management geared towards a single habitat specialist with large home-range requirements, such as the greater sage-grouse (Centrocercus urophasianus), can simultaneously protect other co-occurring species of concern often is appealing. The efficacy of relying on such “umbrella species” (Caro 2010), however, partly depends upon the spatial scale at which management is implemented. At broad spatial scales, the reduction of habitat loss and fragmentation certainly may benefit some co-occurring species (Carlisle et al. 2018a). At finer scales, however, the specific resource needs of the umbrella and sympatric species can diverge, and targeted management actions for the umbrella species may be detrimental to other species (Hanser and Knick 2011; Carlisle and Chalfoun 2020). For example, the experimental reduction of sagebrush cover to benefit sage-grouse during the brood-rearing stage led to complete loss of nesting habitat for sagebrush-obligate songbirds (Carlisle et al. 2018b).
Finally, the need to consider the on-going influence of shifting climatic regimes on vegetation and songbird species will be critical for the long-term success of management actions. Adaptive management strategies that accommodate the shrinking and shifting distributions of climate-sensitive species may be one effective mechanism (Langham et al. 2015).
9 Research and Management Needs
The remaining informational gaps and research needs for rangeland songbirds are extensive, as most have not received the same level of prioritization as many game species. Experimental and longer-term studies would help clarify the specific habitat factors, disturbances, and management interventions that most affect songbird responses and the underlying mechanisms (Chalfoun 2021). The further development of tools to mitigate the effects of energy development on songbirds is merited (Sanders and Chalfoun 2018; Shaffer et al. 2019b), which will necessitate mechanistic understanding of the effects of different types of energy development on songbird species. Efforts to restore habitats to pre-disturbance conditions and protect native ecosystems most at risk of conversion for new energy extraction will be paramount.
A better understanding of how and why songbird abundance and community composition change in areas affected by invasive plant species and woody-plant encroachment would be useful. The development of statistically rigorous (e.g., Before-After Control-Impact) studies of rangeland songbird species in relation to specific management prescriptions within rangeland types would clarify optimal management approaches. Experimental designs that account for the independent contributions of potentially confounding variables, such as the effects of burning versus grazing, also would be fruitful. Improved understanding of the effects of ecological processes and mechanical management practices on avian abundance and productivity at scales relevant to management (e.g., grazing allotments) would further clarify optimal management approaches for songbird management (Pulliam et al. 2021). Potential carryover effects of management activities, such as grazing across years, also would provide more holistic understanding (Johnson et al. 2011). In addition, a better understanding of the influence of multiple stressors, including interactions between changing climatic conditions and their effects on songbird habitats, will be critical for the effective management of rangeland songbirds into the future.
A lack of information about the demography of most rangeland songbird species across the full annual life cycle (i.e., outside of the nesting period), and which life stage(s) are most affected by habitat changes and the most limiting to population growth greatly hampers understanding of ideal management allocation (Marra et al. 2015). The post-fledging survival, migratory routes, key stopover areas, over-wintering locations, and annual survival of most grassland and sagebrush songbird populations remain unknown, partly because of historic limitations on tracking technologies that could be deployed safely on small birds. Because most rangeland-associated songbirds leave the nest prior to being capable of sustained flight (Yackel Adams et al. 2006), habitat requirements, and rates and causes of mortality during the post-fledgling period may be particularly important to understand for threat and population assessments (Yackel Adams et al. 2006; Davis and Fisher 2009; Hovick et al. 2011). Lack of knowledge about the movements and cause-specific mortality of many grassland and shrubsteppe songbird species during migration and winter also inhibits understanding of the relative influence of the breeding versus non-breeding periods on annual survival and therefore population growth (Fletcher et al. 2006). Finally, conditions and processes during particular life stages can carry-over into subsequent stages (Akresh et al. 2021), albeit to an unknown extent for most songbirds inhabiting North American rangelands.
Study of the prevalence and effects of disease (e.g., salmonellosis), endoparasites, and ectoparasites (e.g., blowflies) on the condition and vital rates of rangeland songbirds is in its infancy. Fairly high blow fly loads have been observed on some nestlings in Montana and Wyoming, which can result in partial or complete mortality of the brood (A. Chalfoun, personal observation). Whether particular conditions such as energy development, livestock grazing, or weather influence the susceptibility of songbirds to disease or parasites, and whether such changes scale up to influence populations, remains unclear.
Finally, the importance of understanding and acknowledging the contribution of native peoples’ role in wildlife management, and the incorporation of indigenous and local knowledge into management policies, has been emphasized recently by scholars and U.S. legislators (Lam et al. 2020). Such information is rarely incorporated into rangeland management plans, yet such knowledge offers historical insights that may complement and enrich contemporary approaches to sustainable use of landscapes and encourage practices that are more culturally inclusive and holistic (Lam et al. 2020).
Box 12.1. Grasshopper Sparrow (Ammodramus savannarum)
Habitat Associations
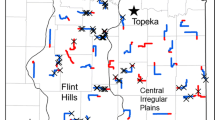
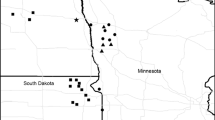
The grasshopper sparrow is a grassland-obligate songbird with a trans-coastal distribution, whose highest breeding densities occur throughout the Great Plains (Vickery 2020; Fig. 12.2). Grasshopper sparrows breed in a variety of native habitats including prairies, desert grasslands, savannahs, and sagebrush steppe, and in nonnative habitats such as planted grasslands (Shaffer et al. 2021). Throughout the grasslands of the Great Plains, grasshopper sparrows tend to avoid areas with woody vegetation (Bakker et al. 2002; Patten et al. 2006; Herse et al. 2018), where the species is reported to be area sensitive [that is, to show a preference for larger extents of grassland; reviewed in Ribic et al. (2009) and Shaffer et al. (2021)]. Within sagebrush steppe, however, the species is more tolerant of shrubby habitats that include native bunchgrasses in the understory (Holmes and Miller 2010; Earnst and Holmes 2012). Vegetation structure likely influences the habitat decisions of grasshopper sparrows to a greater extent than plant species’ composition (Henderson and Davis 2014; Shaffer et al. 2021), as grasshopper sparrows will nest within areas dominated by nonnative grasses such as Kentucky bluegrass (Poa pratensis) and smooth brome (Bromus inermis) (Grant et al. 2004; Shaffer et al. 2021). Structural attributes of vegetation associated with grasshopper sparrow occupancy include intermediate height (8–166 cm), moderate density (4–80 cm visual obstruction reading), moderately deep litter (≤ 9 cm), low-to-moderate bare ground (≤ 38% bare ground), and low shrub cover (< 35%) (Shaffer et al. 2021). This narrative will focus on management approaches to benefit grasshopper sparrows breeding in grassland habitats and will not discuss management approaches for other ecosystems such as sagebrush.
Rangeland Management
Management approaches that maintain open expanses of grassland benefit the grasshopper sparrow. Typical management approaches for the grasshopper sparrow involve grazing-only or a combination of burning and grazing (Shaffer et al. 2021). Optimal management strategies vary across the species’ range depending on how the resultant habitat structure and composition comports with the species’ habitat requirements.
The effects of grazing on grasshopper sparrows depend on local factors such as rangeland type, climate, topoedaphic conditions, and landscape composition (Lipsey and Naugle 2017; Vold et al. 2019; Davis et al. 2021). Further considerations include the timing of grazing, grazing intensity (e.g., stocking rate and duration), and livestock type, all of which may influence the abundance and reproductive success of grasshopper sparrows (Shaffer and DeLong 2019; Shaffer et al. 2021). Appropriate intervals between management treatments depend on rangeland type; for example, mesic prairies regenerate litter more rapidly (1–3 years) than dry prairies (4–6 years) and sooner in southern than northern prairies (Swengel 1996). In tallgrass prairies, moderate-to-heavy grazing reduces vegetation biomass and curtails woody encroachment (Ahlering and Merkord 2016). In mixed-grass prairies, light-to-moderate grazing that reduces vegetation height and density and creates patchy areas is compatible with the habitat needs of the grasshopper sparrow, however, heavy grazing can reduce litter depth and cover to unsuitable levels (Shaffer et al. 2021). Nest densities in Palouse prairie decreased with cattle stocking rates, and no nests were found in pastures with the highest stocking rates of 43.2 animal unit months (46% vegetation utilization) (Johnson et al. 2011). Grazing studies within desert grasslands have been very localized (Ruth 2015), and even light grazing can be detrimental if vegetation becomes too short and open for grasshopper sparrow use (Bock and Webb 1984; Bock et al. 1984, 1993). Heavy grazing in arid grasslands tends to reduce already sparse bunchgrass cover and exclude grasshopper sparrows (Bock and Webb 1984).
In contrast to earlier studies in mixed-grass prairies that suggested that rotational grazing systems may achieve desired vegetation heterogeneity for birds (Shaffer et al. 2021), rest-rotation grazing in northern mixed-grass prairies failed to increase grasshopper sparrow abundance, relative to traditional season-long or summer-rotation grazing (Vold et al. 2019). Similarly, rotational grazing systems in the Nebraska Sandhills (Sliwinski et al. 2019, 2020), shortgrass prairies (Davis et al. 2020), and tallgrass prairies (Temple et al. 1999) did not convey population benefits to the grasshopper sparrow. Rotational grazing systems may fail to create sufficient vegetation heterogeneity to be ecologically relevant to bird communities (Sliwinski et al. 2020), especially when other factors such as year, ecological site (Davis et al. 2020), and stocking rate (Sliwinski et al. 2019; Vold et al. 2019) can have greater effects on bird abundance than grazing system. Livestock type also may influence avian abundance, as grasshopper sparrows were more abundant in Montana pastures grazed by bison than in pastures grazed by cattle (Boyce et al. 2021) and equally as abundant in cattle- and bison-grazed pastures in Colorado (Wilkins et al. 2019).
Fire as a management strategy for grasshopper sparrows is more common within Great Plains grasslands than desert grasslands, where burns usually are the result of wildfires (Shaffer et al. 2021). Similar to grazing, the response of grasshopper sparrows to burning will depend on how vegetation structure is affected, which may vary locally by climate, ecosystem or habitat type, type of burn (e.g., prescribed burn versus wildfire), season, frequency, and intensity (Shaffer and DeLong 2019; Shaffer et al. 2021). The effects of burning-only management on the abundance of grasshopper sparrows in mixed-grass and tallgrass prairies have been varied (Madden et al. 1999; Grant et al. 2010; Byers et al. 2017). In desert grasslands, fires that destroy shrubs may be detrimental, as small shrubs are used by grasshopper sparrows as thermal refugia during extremely hot temperatures (Ruth et al. 2020).
The combination of burning and grazing is a common management approach implemented throughout the Great Plains. Geographic variation in management objectives and approaches, study designs, and timing of the application of burning and grazing, however, make a statement of broad generalizations about the effect on grasshopper sparrow abundance or success difficult (Shaffer et al. 2021). Burning and grazing approaches predominate in northern mixed-grass prairies (e.g., Richardson et al. 2014), shortgrass prairies (e.g., Augustine and Derner 2015), tallgrass prairies (e.g., Fuhlendorf et al. 2006), and sand sagebrush grasslands (Holcomb et al 2014). The patch-burn grazing strategy explained in this chapter’s main section is advocated primarily for tallgrass prairies, with a focus on the Flint Hills (Fuhlendorf and Engle 2001). Examinations between the effects of the traditional burning and grazing approach in the Flint Hills (that is, annual early-spring burns followed immediately by grazing) and the patch-burn grazing approach have yielded variable results on grasshopper sparrow abundance and productivity (Shaffer et al. 2021).
Box 12.2. Brewer’s Sparrow (Spizella breweri)
Habitat Associations
The Brewer’s sparrow often is referred to as a sagebrush-obligate (Rich et al. 2005), along with the sagebrush sparrow (Artemisiospiza nevadensis) and sage thrasher (Orescoptes montanus), although Brewer’s sparrows occasionally inhabit other shrubby habitats. Brewer’s sparrows are migratory, and over-winter in the southwestern U.S. and Mexico (Knick et al. 2014; Valencia-Herverth et al. 2018). The breeding range of Brewer’s sparrows covers most of the extent of North American shrubsteppe, from southern British Columbia, Alberta and Saskatchewan in Canada, south to southern California, southern Nevada, and northern Arizona and New Mexico (Walker et al. 2020; Fig. 12.2).
Brewer’s sparrows prefer and are found in the highest breeding densities within landscapes composed of relatively high cover of sagebrush (> 30%) and taller shrubs (Rotenberry and Wiens 1980; Chalfoun and Martin 2007; Walker et al. 2020). Because Brewer’s sparrows primarily sing, forage and nest within the shrub layer (e.g., Rotenberry and Wiens 1998; Fig. 12.3), the attributes of the shrub layer are paramount. Brewer’s sparrows may be entirely absent from areas with shrub cover ≤ 8% (Walker et al. 2020). Habitat preferences and quality, moreover, are scale-dependent. At smaller spatial scales, Brewer’s sparrows preferred and had higher reproductive success in territories and nest patches (5-m radius) with higher shrub density, and particularly densities of potentially suitable nest shrubs (Chalfoun and Martin 2007, 2009). Brewer’s sparrows prefer to forage and place nests in shrubs with live canopies (Peterson and Best 1985; Rotenberry and Wiens 1998; Chalfoun and Martin 2009; Fig. 12.3).
Rangeland Management
As a near sagebrush-obligate, management actions that remove or decrease big sagebrush (Artemisia tridentata) cover, including burning (Bock and Bock 1987; Knick et al. 2005; Noson et al. 2006), the application of herbicides (Best 1972; Schroeder and Sturges 1975; Kerley and Anderson 1995), and mechanical treatments (Castrale 1982; Carlisle et al. 2018b) tend to eliminate or reduce the local abundance of the Brewer’s sparrow (Walker et al. 2020). Because big sagebrush does not re-sprout after fire, frequent fires increase the cover of annual grasses and reduce sagebrush cover which decreases habitat for sagebrush-obligates including the Brewer’s sparrow (Knick et al. 2003). Burning also can negatively affect sagebrush songbirds by promoting the spread of nonnative weeds and the subsequent conversion of shrubsteppe habitats to nonnative annual grasslands. The planting of nonnative grasses following sagebrush removal hinders recolonization by sagebrush and delays or prohibits the recovery of Brewer’s sparrow habitat (Reynolds and Trost 1980; McAdoo et al. 1989). Insecticide treatments during the nesting period have the potential to reduce arthropod prey and thereby alter Brewer’s sparrow habitat use and productivity (Howe et al. 1996).
Management activities that reduce coniferous encroachment into sagebrush habitats have shown positive effects on sagebrush songbirds (Crow and van Riper 2010). However, habitat treatments traditionally geared towards the reduction of the sagebrush canopy and enhancement of herbaceous understories, thought to benefit the greater sage-grouse (Centrocercus urophasianus) and the Gunnison sage-grouse (Centrocercus minimus) during the brood rearing period, usually negatively affect sagebrush-obligate songbirds. Mechanical treatments (e.g., roller chopping, disking, mowing,) in Colorado significantly decreased densities of Brewer’s sparrows (Lukacs et al. 2015). Moreover, experimental evaluation of mowing effects in central Wyoming resulted in the complete loss of nesting habitat for Brewer’s sparrows and sage thrashers (Carlisle et al. 2018b).
Rigorous investigations of the effects of grazing regimes on the Brewer’s sparrows have been limited. The abundance of Brewer’s sparrows did not differ between rest-rotation versus season-long grazing treatments in Montana (Golding and Dreitz 2017). However, Brewer’s sparrow abundance decreased with the highest grazing treatment during a study in southern Idaho and northern Utah, which corresponded with lower shrub cover and higher cover of exotic annuals (Bradford et al. 1998). Brewer’s sparrows tend to be less affected by moderate grazing compared with grassland songbirds that are more reliant on the herbaceous understory (Bock et al. 1993).
References
Ahlering MA, Merkord CL (2016) Cattle grazing and grassland birds in northern tallgrass prairie. J Wildl Manage 80:643–654. https://doi.org/10.1002/jwmg.1049
Akresh ME, King DI, Marra PP (2021) Hatching date influences winter habitat occupancy: examining seasonal interactions across the full annual cycle in a migratory songbird. Ecol Evol 11:9241–9253. https://doi.org/10.1002/ece3.750
Allen AW, Cade BS, Vandever MW (2001) Effects of emergency haying on vegetative characteristics within selected Conservation Reserve Program fields in the northern Great Plains. J Soil Water Conserv 56:120–125. https://www.jswconline.org/content/56/2/120
Allison TD, Diffendorfer JE, Baerwald EF, Beston J, Drake D, Hale A, Hein C, Huso MM, Loss S, Lovich JE, Strickland D, Williams K, Winder V (2019) Impacts to wildlife of wind energy siting and operation in the United States. Issues in Ecol, Report No. 21. https://www.esa.org/wp-content/uploads/2019/09/Issues-in-Ecology_Fall-2019.pdf
Allred BW, Fuhlendorf SD, Hamilton RG (2011) The role of herbivores in Great Plains conservation: comparative ecology of bison and cattle. Ecosphere 2:1–17. https://doi.org/10.1890/ES10-00152.1
Allred BW, Fuhlendorf SD, Engle DM, Elmore RD (2011) Ungulate preference for burned patches reveals strength of fire-grazing interaction. Ecol Evol 1:132–144. https://doi.org/10.1002/ece3.12
Anderson EM, Steidl RJ (2019) Woody plant encroachment restructures bird communities in semiarid grasslands. Biol Cons 240:108276. https://doi.org/10.1016/j.biocon.2019.108276
Archer SR, Anderson EM, Predick KI, Schwinning S, Steidl RJ, Woods SR (2017) Woody plant encroachment: causes and consequences. In: Briske D (ed) Rangeland systems: processes, management, and challenges. Springer Series on Environmental Management, pp 25–84. https://doi.org/10.1007/978-3-319-46709-2
Augustine DJ, Derner JD (2015) Patch-burn grazing management, vegetation heterogeneity, and avian responses in a semi-arid grassland. J Wildl Manage 79:927–936. https://doi.org/10.1002/jwmg.909
Bailey DW (2005) Identification and creation of optimum habitat conditions for livestock. Rangel Ecol Manag 58:109–118. https://doi.org/10.2111/03-147.1
Bakker KK, Naugle DE, Higgins KF (2002) Incorporating landscape attributes into models for migratory grassland bird conservation. Conserv Biol 16:1638–1646. https://doi.org/10.1046/j.1523-1739.2002.01328.x
Barbour MG, Billings WD (2000) North American terrestrial vegetation. Cambridge University Press, Cambridge
Bayly NJ, Gomez C, Hobson KA, Rosenberg KV (2016) Prioritizing tropical habitats for long-distance migratory songbirds: an assessment of habitat quality at a stopover site in Colombia. Avian Conserv Ecol 11:article 5. https://doi.org/10.5751/ACE-00873-110205
Beason RC (2020) Horned lark (Eremophila alpestris). In: Billerman SM (ed) Birds of the World, vers.1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.horlar.01
Bedard J, McNeil JN (1979) Protocalliphora hirudo (Diptera: Calliphoridae) infesting Savannah sparrow, Passerculus sandwichensis (Aves: Fringillidae), in eastern Quebec. Can Entomol 111:111–112. https://doi.org/10.4039/Ent111111-1
Berkeley LI, McCarty JP, Wolfenbarger LL (2007) Postfledging survival and movement in dickcissels (Spiza americana): implications for habitat management and conservation. Auk 124:396–409. https://doi.org/10.1093/auk/124.2.396
Bernath-Plaistad J, Koper N (2016) Physical footprint of oil and gas infrastructure, not anthropogenic noise, reduces nesting success of some grassland songbirds. Biol Conserv 204:434–441. https://doi.org/10.1016/j.biocon.2016.11.002
Best LB (1972) First-year effects of sagebrush control on two sparrows. J Wildl Manage 36:534–544
Bestelmeyer BT, Peters DPC, Archer SR, Browning DM, Okin GS, Schooley RL, Webb NP (2018) The grassland-shrubland regime shift in the southwestern United States: misconceptions and their implications for management. Bioscience 68:678–690. https://doi.org/10.1093/biosci/biy065
Bleho BI, Koper N, Machtans CS (2014) Direct effects of cattle on grassland birds in Canada. Conserv Biol 28:724–734. https://doi.org/10.1111/cobi.12259
Bock CE, Webb B (1984) Birds as grazing indicator species in southeastern Arizona. J Wildl Manage 48:1045–1049. https://doi.org/10.2307/3801466
Bock CE, Bock JH (1987) Avian habitat occupancy following fire in a Montana shrubsteppe. Prairie Naturalist 19:153–158
Bock CE, Bock JH, Kenney WR, Hawthorne VM (1984) Responses of birds, rodents, and vegetation to livestock exclosure in a semidesert grassland site. J Range Manage 37:239–242. https://doi.org/10.2307/3899146
Bock CE, Saab VA, Rich TD, Dobkin DS (1993) Effects of livestock grazing on Neotropical migratory landbirds in western North America. In: Finch DM, Stangel PW (eds) Status and management of Neotropical migratory birds. U.S. Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station, General Technical Report RM-229. Fort Collins, Colorado, pp 296–309
Bollinger EK, Bollinger PB, Gavin TA (1990) Effects of hay-cropping on eastern populations of the bobolink. Wilson Bull 18:142–150. https://www.jstor.org/stable/3782128
Boyce AM, Shamon H, Kunkel KE, McShea WJ (2021) Grassland bird diversity and abundance in the presence of native and non-native grazers. Avian Cons Ecol 16:article 13. https://doi.org/10.5751/ACE-01944-160213
Boyd EM (1951) The external parasites of birds: a review. Wilson Bull 63:363–369
Bradford DF, Franson SE, Neale AC, Heggem DT, Miller GR, Canterbury GE (1998) Bird species assemblages as indicators of biological integrity in Great Basin rangeland. Environ Monit Assess 49:1–22. https://doi.org/10.1023/A:1005712405487
Briske DD, Derner JD, Brown JR, Fuhlendorf SD, Teague WR, Havstad KM, Gillen RL, Ash AJ, Willms WD (2008) Rotational grazing on rangelands: reconciliation of perception and experimental evidence. Rangel Ecol Manag 61:3–17. https://doi.org/10.2111/06-159R.1
Brooks ML, D’Antonio CM, Richardson DM, Grace JB, Keeley JE, DiTomaso JM, Hobbs RJ, Pellant M, Pyke D (2004) Effects of invasive alien plants on fire regimes. Bioscience 54:677–683. https://doi.org/10.1641/00063568(2004)054[0677:EOIAPO]2.0.CO;2
Byers CM, Ribic CA, Sample DW, Dadisman JD, Guttery MR (2017) Grassland bird productivity in warm season grass fields in southwest Wisconsin. Am Midl Nat 178:47–63. https://doi.org/10.1674/0003-0031-178.1.47
Carlisle JD, Chalfoun AD (2020) The abundance of greater sage-grouse as a proxy for the abundance of sagebrush-associated songbirds in Wyoming, USA. Avian Cons Ecol 15:article 16. https://doi.org/10.5751/ACE-01702-150216
Carlisle JD, Keinath DA, Albeke SE, Chalfoun AD (2018a) Identifying holes in the greater sage-grouse conservation umbrella. J Wildl Manage 82:948–957. https://doi.org/10.1002/jwmg.21460
Carlisle JD, Chalfoun AD, Smith KT, Beck JL (2018b) Nontarget effects on songbirds from habitat manipulation for greater sage-grouse: implications for the umbrella species concept. Condor Ornithol Appl 120:439–455. https://doi.org/10.1650/CONDOR-17-200.1
Caro TM (2010) Conservation by proxy: indicator, umbrella, keystone, flagship, and other surrogate species. Island Press, Washington, DC
Carver AR, Ross JD, Augustine DJ et al (2017) Weather radar data correlate to hail-induced mortality in grassland birds. Remote Sens Ecol Conserv 3:90–101. https://doi.org/10.1002/rse2.41
Castrale JS (1982) Effects of two sagebrush control methods on nongame birds. J Wildl Manage 46:945–952. https://doi.org/10.2307/3808227
Centers for Disease Control (2016) Species of dead birds in which West Nile virus has been detected, United States, 1999–2016. https://www.cdc.gov/westnile/resources/pdfs/birdspecies1999-2016.pdf
Chalfoun AD (2021) Responses of vertebrate wildlife to oil and natural gas development: patterns and frontiers. Curr Landscape Ecol Rep 6:71–84. https://doi.org/10.1007/s40823-021-00065-0
Chalfoun AD, Martin TE (2007) Assessments of habitat preferences and quality depend on spatial scale and metrics of fitness. J Appl Ecol 44:983–992. https://doi.org/10.1111/j.1365-2664.2007.01352.x
Chalfoun AD, Martin TE (2009) Habitat structure mediates predation risk for sedentary prey: experimental tests of alternative hypotheses. J Anim Ecol 78:497–503. https://doi.org/10.1111/j.1365-2656.2008.01506.x
Cicero C, Pyle P, Patten MA (2020) Juniper titmouse (Baeolophus ridgwayi). In: Rodewald PG (ed) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.juntit1.01
Coates PS, Brussee BE, Howe KB et al (2016) Landscape characteristics and livestock presence influence common ravens: relevance to greater sage-grouse conservation. Ecosphere 7:e01203. https://doi.org/10.1002/ecs2.1203
Cody ML (1985) Habitat selection in grassland and open country birds. In: Cody ML (ed) Habitat selection in birds. Academic Press, New York, pp 191–226
Coffman JM, Bestelmeyer BT, Kelly JF, Wright TF, Schooley RL (2014) Restoration practices have positive effects on breeding bird species of concern in the Chihuahuan desert. Restor Ecol 22:336–344. https://doi.org/10.1111/rec.12081
Conrey RY, Skagen SK, Yackel Adams AA, Panjabi AO (2016) Extremes of heat, drought and precipitation depress reproductive performance in shortgrass prairie passerines. Ibis 158:614–629. https://doi.org/10.1111/ibi.12373
Coppedge BR, Engle DM, Masters RE, Gregory MS (2001) Avian response to landscape change in fragmented southern Great Plains grasslands. Ecol Appl 11:47–59. https://doi.org/10.1890/1051-0761(2001)011[0047:ARTLCI]2.0.CO;2
Crow C, van Riper IIIC (2010) Avian community responses to mechanical thinning of a pinyon-juniper woodland: specialist sensitivity to tree reduction. Nat Areas J 30:191–201. https://doi.org/10.3375/043.030.0206
Danner JE, Small DM, Ryder TB, Lohr B, Masters BS, Gill DE, Fleischer RC (2018) Temporal patterns of extra-pair paternity in a population of grasshopper sparrows (Ammodramus savannarum) in Maryland. Wilson J Ornithol 130:40–51. https://doi.org/10.1676/16-049.1
Davies KW, Bates JD (2020) Re-introducing fire in sagebrush steppe experiencing decreased fire frequency: does burning promote spatial and temporal heterogeneity? Int J Wildland Fire 29:686–695. https://doi.org/10.1071/WF20018
Davis SK, Duncan DC (1999) Grassland songbird occurrence in native and crested wheatgrass pastures of southern Saskatchewan. In: Vickery PD, Herkert JR (eds) Ecology and conservation of grassland birds of the Western Hemisphere. Stud Avian Biol 19:211–218
Davis SK, Lanyon WE (2020) Western meadowlark (Sturnella neglecta). In: Poole AF (ed) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.wesmea.01
Davis SK, Fisher RJ (2009) Post-fledging movements of Sprague’s pipit. Wilson J Ornithol 121:198–202. https://doi.org/10.1676/08-025.1
Davis KP, Augustine DJ, Monroe AP, Derner JD, Aldridge CL (2020) Adaptive rangeland management benefits grassland birds utilizing opposing vegetation structure in the shortgrass steppe. Ecol Appl 30:e02020. https://doi.org/10.1002/eap.2020
Davis KP, Augustine DJ, Monroe AP, Aldridge CL (2021) Vegetation characteristics and precipitation jointly influence grassland bird abundance beyond the effects of grazing management. Ornithol Appl 123:1–15. https://doi.org/10.1093/ornithapp/duab041
de Lope F, González G, Pérez JJ, Møller AP (1993) Increased detrimental effects of ectoparasites on their bird hosts during adverse environmental conditions. Oecologia 95:234–240. https://doi.org/10.1007/BF00323495
Deeming DC (2001) Avian incubation: environment, behavior and evolution. Oxford University Press, London
Derner JD, Lauenroth WK, Stapp P, Augustine DJ (2009) Livestock as ecosystem engineers for grassland bird habitat in the western Great Plains of North America. Rangel Ecol Manag 62:111–118. https://doi.org/10.2111/08-008.1
Duchardt CJ, Augustine DJ, Beck JL (2019) Threshold responses of grassland and sagebrush birds to patterns of disturbance created by an ecosystem engineer. Landsc Ecol 34:895–909. https://doi.org/10.1007/s10980-019-00813-y
Duchardt CJ, Porensky LM, Pearse IS (2021) Direct and indirect effects of a keystone engineer on a shrubland-prairie food web. Ecology 102:e03195. https://doi.org/10.1002/ecy.3195
Earnst SL, Holmes AL (2012) Bird-habitat relationships in Interior Columbia Basin shrubsteppe. Condor 14:15–29. https://doi.org/10.1525/cond.2009.080109
Engle DM, Fuhlendorf SD, Coppedge BR (2003) Conservation priorities on fragmented, homogenized Great Plains landscapes. In: Fore S (ed) Proceeding of the 18th North American Prairie Conference, pp 1–6
Erickson WP, Wolfe MM, Bay KJ, Johnson DH, Gehring JL (2014) A comprehensive analysis of small-passerine fatalities from collision with turbines at wind energy facilities. PLoS ONE 9:e107491. https://doi.org/10.1371/journal.pone.0107491
Fisher RJ, Davis SK (2011) Post-fledging dispersal, habitat use, and survival of Sprague’s pipits: are planted grasslands a good substitute for native? Biol Conserv 144:263–271. https://doi.org/10.1016/j.biocon.2010.08.024
Fleishman E, Thomson JR, Kalies EL, Dickson BG, Dobkin DS, Leu M (2014) Projecting current and future location, quality, and connectivity of habitat for breeding birds in the Great Basin. Ecosphere 5: article 82. https://doi.org/10.1890/ES13-00387.1
Fletcher RJ Jr, Koford RR, Seaman DA (2006) Critical demographic parameters for declining songbirds breeding in restored grasslands. J Wildl Manage 70:145–157. https://doi.org/10.2193/0022-541X(2006)70[145:CDPFDS]2.0.CO;2
Frost JS, Powell LA (2011) Cedar infestation impacts avian communities along the Niobrara River Valley, Nebraska. Restor Ecol 19:529–536. https://doi.org/10.1111/j.1526-100X.2009.00618.x
Fuhlendorf SD, Engle DM (2001) Restoring heterogeneity on rangelands: ecosystem management based on evolutionary grazing patterns. Bioscience 51:625–632. https://doi.org/10.1641/0006-3568(2001)051[0625:RHOREM]2.0.CO;2
Fuhlendorf SD, Harrell WC, Engle DM, Hamilton RG, Davis CA, Leslie DM Jr (2006) Should heterogeneity be the basis for conservation? Grassland bird response to fire and grazing. EcolAppl 16:1706–1716. https://doi.org/10.1890/10510761(2006)016%5B1706:SHBTBF%5D2.0.CO;2
Gilbert MG, Chalfoun AD (2011) Energy development affects populations of sagebrush songbirds in Wyoming. J Wildl Manage 75:816–824. https://doi.org/10.1002/jwmg.123
Goguen CB, Mathews NE (1999) Review of the causes and implications of the association between cowbirds and livestock. In: Hahn DC, Hall LS, Morrison ML et al (eds) Research and management of the brown-headed cowbird in western landscapes. Stud Avian Biol 18:10–17
Goguen CB, Mathews NE (2000) Local gradients of cowbird abundance and parasitism relative to livestock grazing in a western landscape. Conserv Biol 14:1862–1869. https://doi.org/10.1111/j.1523-1739.2000.99313.x
Golding JD, Dreitz VJ (2017) Songbird response to rest-rotation and season-long cattle grazing in a grassland sagebrush ecosystem. J Environ Manage 204:605–612. https://doi.org/10.1016/j.jenvman.2017.09.044
Grant TA, Murphy RK (2005) Changes in woodland cover on prairie refuges in North Dakota, USA. Nat Areas J 25:359–368
Grant TA, Madden E, Berkey GB (2004) Tree and shrub invasion in northern mixed-grass prairie: implications for breeding grassland birds. Wildl Soc Bull 32:807–818. https://doi.org/10.2193/0091-7648(2004)032[0807:TASIIN]2.0.CO;2
Grant TA, Madden E, Shaffer TL, Dockens JS (2010) Effects of prescribed fire on vegetation and passerine birds in northern mixed-grass prairie. J Wildl Manage 74:1841–1851. https://doi.org/10.2193/2010-006
Green AW, Pavlacky DC Jr, George TL (2019) A dynamic multi-scale occupancy model to estimate temporal dynamics and hierarchical habitat use for nomadic species. Ecol Evol 9:793–803. https://doi.org/10.1002/ece3.4822
Green MT, Lowther PE, Jones SL, Davis SK, Dale BC (2020) Baird's sparrow (Centronyx bairdii). In: Poole AF, Gill FB (eds) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.baispa.01
Grinnell J (1917) The niche-relationships of the California thrasher. Auk 34:427–433. https://doi.org/10.2307/4072271
Hanser SE, Knick ST (2011) Greater sage-grouse as an umbrella species for shrubland passerine birds: a multiscale assessment. In: Knick S, Connelly JW (eds) Ecology, conservation, and management of grouse. Stud Avian Biol 38:475–488
Henderson AE, Davis SK (2014) Rangeland health assessment: a useful tool for linking range management and grassland bird conservation? Rangel Ecol Manag 67:88–98. https://doi.org/10.2111/REM-D-12-00140.1
Herkert JR, Sample DW, Warner RE (1996) Management of midwestern grassland landscapes for the conservation of migratory birds. In: Thompson III FR (ed) Management of midwestern landscapes for the conservation of Neotropical migratory birds. U.S. Department of Agriculture, Forest Service, North Central Forest Experiment Station, General Technical Report GTR-NC-187. St. Paul, Minnesota, pp 89–116
Herse MR, With KA, Boyle WA (2018) The importance of core habitat for a threatened species in changing landscapes. J Appl Ecol 55:2241–2252. https://doi.org/10.1111/1365-2664.13234
Hethcoat MG, Chalfoun AD (2015) Toward a mechanistic understanding of human-induced rapid environmental change: a case study linking energy development, nest predation, and predators. J Appl Ecol 52:1492–1499. https://doi.org/10.1016/j.biocon.2015.02.009
Hethcoat MG, Chalfoun AD (2015) Energy development and avian nest survival in Wyoming, USA: a test of a common disturbance index. Biol Conserv 184:327–334. https://doi.org/10.1016/j.biocon.2015.02.009
Hickman KR, Farley GH, Channell R, Steier JE (2006) Effects of old world bluestem (Bothriochloa ischaemum) on food availability and avian community composition within the mixed-grass prairie. Southwest Nat 51:524–530. https://doi.org/10.1894/0038-4909(2006)51[524:EOOWBB]2.0.CO;2
Higgins KF (1986) A comparison of burn season effects on nesting birds in North Dakota mixed-grass prairie. Prairie Nat 18:219–228
Hightower JN, Carlisle JD, Chalfoun AD (2018) Nest mortality of sagebrush songbirds due to a severe hailstorm. Wilson J Ornithol 130:561–567. https://doi.org/10.1676/17-025.1
Holcomb ED, Davis CA, Fuhlendorf SD (2014) Patch-burn management: implications for conservation of avian communities in fire-dependent sagebrush ecosystems. J Wildl Manage 78:848–856. https://doi.org/10.1002/jwmg.723
Holmes AL, Miller RF (2010) State-and-transition models for assessing grasshopper sparrow habitat use. J Wildl Manage 74:1834–1840. https://doi.org/10.2193/2009-417
Hovick TJ, Miller JR (2013) Broad-scale heterogeneity influences nest selection by brown-headed cowbirds. Landsc Ecol 28:1493–1503. https://doi.org/10.1007/s10980-013-9896-7
Hovick TJ, Miller JR, Koford RR, Engle DM, Debinski DM (2011) Post-fledging survival of grasshopper sparrows in grasslands managed with fire and grazing. Condor 113:429–437. https://doi.org/10.1525/cond.2011.100135
Hovick TJ, Elmore RD, Fuhlendorf SD (2014) Structural heterogeneity increases diversity of non-breeding grassland birds. Ecosphere 5:1–13. https://doi.org/10.1890/ES14-00062.1
Howe FP (1992) Effects of Protocalliphora braueri (Diptera: Calliphoridae) parasitism and inclement weather on nestling sage thrashers. J Wildl Dis 28:141–143. https://doi.org/10.7589/0090-3558-28.1.141
Howe FP, Knight RL, McEwen LC, George TL (1996) Direct and indirect effects of insecticide applications on growth and survival of nestling passerines. Ecol Appl 6:1314–1324. https://doi.org/10.2307/2269609
Igl LD, Ballard BM (1999) Habitat associations of migrating and overwintering grassland birds in southern Texas. Condor 101:771–782. https://doi.org/10.2307/1370064
Igl LD, Johnson DH (2016) Effects of haying on breeding birds in CRP grasslands. J Wildl Manage 80:1189–1204. https://doi.org/10.1002/jwmg.21119
Johnson TN, Kennedy PL, DelCurto T, Taylor RV (2011) Bird community responses to cattle stocking rates in a Pacific Northwest bunchgrass prairie. Agric Ecosyst Environ 144:338–346. https://doi.org/10.1016/j.agee.2011.10.003
Johnson TN, Kennedy PL, Etterson MA (2012) Nest success and cause-specific nest failure of grassland passerines breeding in prairie grazed by livestock. J Wildl Manage 76:1607–1616. https://doi.org/10.1002/jwmg.437
Johnson DH, Igl LD, Shaffer JA, DeLong JP (2019) The effects of management practices on grassland birds. U.S. Geological Survey Professional Paper 1842. https://doi.org/10.3133/pp1842
Jones ZF, Bock CE (2005) The Botteri’s sparrow and exotic Arizona grasslands: an ecological trap or habitat regained? Condor 107:731–741. https://doi.org/10.1093/condor/107.4.731
Jones TM, Ward MP (2020) Pre- to post-fledging carryover effects and the adaptive significance of variation in wing development for juvenile songbirds. J Anim Ecol 89:2235–2245. https://doi.org/10.1111/1365-2656.13285
Jones NF, Pejchar L, Kiesecker JM (2015) The energy footprint: how oil, natural gas, and wind energy affect land for biodiversity and the flow of ecosystem services. Bioscience 65:290–301. https://doi.org/10.1093/biosci/biu224
Jones TM, Ward MP, Benson TJ, Brawn JD (2017) Variation in nestling body condition and wing development predict cause-specific mortality in fledgling dickcissels. J Avian Biol 48:439–447. https://doi.org/10.1111/jav.01143
Jones TM, Brawn JD, Ward MP (2018) Development of activity rates in fledgling songbirds: when do young birds begin to behave like adults? Behaviour 155:337–350. https://doi.org/10.1163/1568539X-00003492
Kalyn Bogard HJ, Davis SK (2014) Grassland songbirds exhibit variable responses to the proximity and density of natural gas wells. J Wildl Manage 78:471–482. https://doi.org/10.1002/jwmg.684
Kennedy PL, DeBano SJ, Bartuszevige AM, Lueders AS (2009) Effects of native and non-native grassland plant communities on breeding passerine birds: implications for restoration of northwest bunchgrass prairie. Restor Ecol 17:515–525. https://doi.org/10.1111/j.1526-100X.2008.00402.x
Kerley LL, Anderson SH (1995) Songbird responses to sagebrush removal in a high elevation sagebrush steppe ecosystem. Prairie Naturalist 27:129–146
Kerns CK, Ryan MR, Murphy RK, Thompson FR III, Rubin CS (2010) Factors affecting songbird nest survival in northern mixed-grass prairie. J Wildl Manage 74:257–264. https://doi.org/10.2193/2008-249
Knick ST, Dobkin DS, Rotenberry JT, Schroeder MA, Vander Haegen WM, van Riper IIIC (2003) Teetering on the edge or too late? Conservation and research issues for avifauna of sagebrush habitats. Condor 105:611–634. https://doi.org/10.1650/7329
Knick ST, Holmes AL, Miller RF (2005) The role of fire in structuring sagebrush habitats and bird communities. In: Saab VA, Powell HDW (eds) Fire and avian ecology in North America. Stud Avian Biol 30:63–75
Knick ST, Rotenberry JT, Leu M (2008) Habitat, topographical, and geographical components structuring shrubsteppe bird communities. Ecography 31:389–400. https://doi.org/10.1111/j.0906-7590.2008.05391.x
Knick ST, Leu M, Rotenberry JT, Hanser SE, Fesenmyer KA (2014) Diffuse migratory connectivity in two species of shrubland birds: evidence from stable isotopes. Oecologia 174:595–608. https://doi.org/10.1007/s00442-013-2791-8
Lam DPM, Hinz E, Lang DJ, Tengo M, von Wehrden H, Martin-López B (2020) Indigenous and local knowledge in sustainability transformations research: a literature review. Ecol Soc 25: article 3. https://doi.org/10.5751/ES-11305-250103
Langham GM, Schuetz JG, Distler T, Soykan CU, Wilsey C (2015) Conservation status of North American birds in the face of future climate change. PLoS ONE 10:e0135350. https://doi.org/10.1371/journal.pone.0135350
Lany NK, Ayres MP, Stange EE, Sillett TS, Rodenhouse NL, Holmes RT (2016) Breeding timed to maximize reproductive success for a migratory songbird: the importance of phenological asynchrony. Oikos 125:656–666. https://doi.org/10.1111/oik.02412
Laurent EJ, Bart J, Giocomo J, Harding S, Koch K, Moore-Barnhill L, Mordecai R, Sachs E, Wilson T (2012) A field guide to southeast bird monitoring programs and protocols. Southeast Partners in Flight. http://SEmonitoringguide.sepif.org
Lima SL (2009) Predators and the breeding bird: behavioral and reproductive flexibility under the risk of predation. Biol Rev 84:485–513. https://doi.org/10.1111/j.1469-185X.2009.00085.x
Lipsey MK, Naugle DE (2017) Precipitation and soil productivity explain effects of grazing on grassland songbirds. Rangel Ecol Manag 70:331–340. https://doi.org/10.1016/j.rama.2016.10.010
Lipsey MK, Naugle DE, Nowak J, Lukacs PM (2017) Extending utility of hierarchical models to multi-scale habitat selection. Divers Distrib 23:783–793. https://doi.org/10.1111/ddi.12567
Litt AR, Cord EE, Fulbright TE, Schuster GL (2014) Effects of invasive plants on arthropods. Conserv Biol 28:1532–1549. https://doi.org/10.1111/cobi.12350
Lloyd JD, Martin TE (2005) Reproductive success of chestnut collared longspurs in native and exotic grassland. Condor 107:363–374. https://doi.org/10.1650/7701
Longcore T, Rich C, Mineau P, MacDonald B, Bert DG, Sullivan LM, Mutrie E, Gauthreaux SA Jr, Avery ML, Crawford RL, Manville AM II, Travis ER, Drake D (2012) An estimate of avian mortality at communication towers in the United States and Canada. PLoS ONE 7:e34025. https://doi.org/10.31371/journal.pone.0034025
Loss SR (2016) Avian interactions with energy infrastructure in the context of other anthropogenic threats. Condor 118:424–432. https://doi.org/10.1650/condor-16-12.1
Loss SR, Will T, Marra PP (2013a) The impact of free-ranging domestic cats on wildlife of the United States. Nat Commun 4:article 1396. https://doi.org/10.1038/ncomms2380
Loss SR, Will T, Marra PP (2013b) Estimates of bird collision mortality at wind facilities in the contiguous United States. Biol Conser 168:201–209. https://doi.org/10.1016/j.biocon.2013.10.007
Ludlow SM, Brigham RM, Davis SK (2014) Nesting ecology of grassland songbirds: effects of predation, parasitism, and weather. Wilson J Ornithol 126:686–699. https://doi.org/10.1676/13-176.1
Lukacs P, Seglund A, Boyle S (2015) Effects of Gunnison sage-grouse habitat treatment efforts on associated avifauna and vegetation structure. Avian Conserv Ecol 10:article 7. https://doi.org/10.5751/ACE-00799-100207
Lyons TP, Miller JR, Debinski DM, Engle DM (2015) Predator identity influences the effect of habitat management on nest predation. Ecol Appl 25:1596–1605. https://doi.org/10.1890/14-1641
Mabry CM, Harms TM (2020) Impact of delayed mowing on restoring populations of grassland birds of conservation concern. Ecol Restor 38:77–82
Madden E, Hansen AJ, Murphy RK (1999) Influence of prescribed fire history on habitat and abundance of passerine birds in northern mixed-grass prairie. Can Field Nat 113:627–640
Mahoney A, Chalfoun AD (2016) Reproductive success of horned lark and McCown's longspur in relation to wind energy infrastructure. Condor: Ornithol Appl 118:360–375. https://doi.org/10.1650/CONDOR-15-25.1
Manier DJ, Hobbs NT (2006) Large herbivores influence the composition and diversity of shrubsteppe communities in the Rocky Mountains, USA. Oecologia 146:641–651. https://doi.org/10.1007/s00442-005-0065-9
Marra PP, Cohen EB, Loss SR, Rutter JE, Tonra CM (2015) A call for full annual cycle research in animal ecology. Biol Lett 11:e20150552. https://doi.org/10.1098/rsbl.2015.0552
Martin TG (1993) Nest predation and nest sites: new perspectives on old patterns. Bioscience 43:523–532
Martin TG, Possingham HP (2005) Predicting the impact of livestock grazing on birds using foraging height data. J Appl Ecol 42:400–408. https://doi.org/10.1111/j.1365-2664.2005.01012.x
Martin JW, Carlson BA (2020) Sagebrush sparrow (Artemisiospiza nevadensis). In: Poole AF (ed) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.sagspa1.01
Marzluff J, Ewing K (2001) Restoration of fragmented landscapes for the conservation of birds: a general framework and specific recommendations for urbanizing landscapes. Restor Ecol 9:280–292. https://doi.org/10.1046/j.1526-100X.2001.009003280.x
McAdoo JK, Longland WS, Evans RA (1989) Nongame bird community responses to sagebrush invasion of crested wheatgrass seedings. J Wildl Manage 53:494–502. https://doi.org/10.2307/3801155
Mejia EC, McClure CJW, Barber JR (2019) Large-scale manipulation of the acoustic environment can alter the abundance of breeding birds: evidence from a phantom natural gas field. J Appl Ecol 56:2091–2101. https://doi.org/10.1111/1365-2664.13449
Miller RA, Bond L, Migas PN, Carlisle JD, Kaltenecker GS (2017) Contrasting habitat associations of sagebrush-steppe songbirds in the intermountain west. Western Birds 48:35–55
Nack JL, Ribic CA (2005) Apparent predation by cattle at grassland bird nests. Wilson Bull 117:56–62
Nackley LL, West AG, Skowno AL, Bond WJ (2017) The nebulous ecology of native invasions. Trends Ecol Evol 32:814–824. https://doi.org/10.1016/j.tree.2017.08.003
National Audubon Society (2014) Audubon’s birds and climate change report: a primer for practitioners. Vers. 1.2. National Audubon Society, New York. http://climate.audubon.org/sites/default/files/Audubon-Birds-Climate-Report-v1.2.pdf
Neilly H, Vanderwal J, Schwarzkopf L (2016) Balancing biodiversity and food production: a better understanding of wildlife response to grazing will inform off-reserve conservation on rangelands. Rangel Ecol Manag 69:430–436. https://doi.org/10.1016/j.rama.2016.07.007
Nelson SB, Coon JJ, Duchardt CJ, Fischer JD, Halsey SJ, Kranz AJ, Parker CM, Schneider SC, Swartz TM, Miller JR (2017) Patterns and mechanisms of invasive plant impacts on North American birds: a systematic review. Biol Invasions 19:1547–1563. https://doi.org/10.1007/s10530-017-1377-5
Nelson SB, Coon JJ, Duchardt CJ, Miller JM, Debinski DM, Schacht WH (2018) Contrasting impacts of invasive plants and human-altered landscape context on nest survival and brood parasitism of a grassland bird. Landsc Ecol 33:1799–1813. https://doi.org/10.1007/s10980-018-0703-3
Nelson SB, Coon JJ, Miller RJ (2020) Do habitat preferences improve fitness? Context-specific adaptive habitat selection by a grassland songbird. Oecologia 193:15–26. https://doi.org/10.1007/s00442-020-04626-8
Nixon AE, Fisher RJ, Stralberg D, Bayne EM, Farr DR (2016) Projected responses of North American grassland songbirds to climate change and habitat availability at their northern range limits in Alberta, Canada. Avian Conserv Ecol 11:1–14. https://doi.org/10.5751/ACE-00866-110202
Northrup JM, Wittemyer G (2013) Characterising the impacts of emerging energy development on wildlife, with an eye towards mitigation. Ecol Lett 16:112–125. https://doi.org/10.1111/ele.12009
Noson AC, Schmitz RA, Miller RF (2006) Influence of fire and juniper encroachment on birds in high-elevation sagebrush steppe. West N Am Nat 66:343–353. https://doi.org/10.3398/1527-0904(2006)66[343:IOFAJE]2.0.CO;2
Owens RA, Myres MT (1973) Effects of agriculture upon populations of native passerine birds of an Alberta fescue grassland. Can J Zool 51:697–713
Paige C, Ritter SA (1999) Birds in a sagebrush sea: managing sagebrush habitats for bird communities. Partners in Flight Western Working Group, Boise, Idaho. https://partnersinflight.org/resources/birds-in-a-sagebrush-sea/. Accessed 1 Aug 2022
Partners in Flight (2021) Avian conservation assessment database, vers. 2021. https://partnersinflight.org/what-we-do/science/databases/
Patten MA, Shochat E, Reinking DL, Wolfe DH, Sherrod SK (2006) Habitat edge, land management, and rates of brood parasitism in tallgrass prairie. Ecol Appl 16:687–695. https://doi.org/10.1890/10510761(2006)016%5B0687:HELMAR%5D2.0.CO;2
Peterson KL, Best LB (1985) Brewer’s sparrow nest-site characteristics in a sagebrush community. J Field Ornithol 56:23–27
Peterson KL, Best LB, Winter BM (1986) Growth of nestling sage sparrows and Brewer’s sparrows. Wilson Bull 98:535–546
Pietz PJ, Granfors DA (2000) Identifying predators and fates of grassland passerine nests using miniature video cameras. J Wildl Manage 64:71–87. https://doi.org/10.2307/3802976
Pietz PJ, Buhl DA, Shaffer JA, Winter M, Johnson DH (2009) Influence of trees in the landscape on parasitism rates of grassland passerine nests in southeastern North Dakota. Condor 111:36–42. https://doi.org/10.1525/cond.2009.080012
Pilliod DS, Welty JL, Arkle RS (2017) Refining the cheatgrass-fire cycle in the Great Basin: precipitation timing and fine fuel composition predict wildfire trends. Ecol Evol 7:8126–8151. https://doi.org/10.1002/ece3.3414
Powell AFLA (2006) Effects of prescribed burns and bison (Bos bison) grazing on breeding bird abundance in tallgrass prairie. Auk 123:183–197. https://doi.org/10.1642/00048038(2006)123%5B0183:EOPBAB%5D2.0.CO;2
Pulliam JP, Somershoe S, Sather M, McNew LM (2020) Habitat targets for imperiled grassland birds in northern mixed-grass prairie. Rangel Ecol Manag 73:511–519. https://doi.org/10.1016/j.rama.2020.02.006
Pulliam JP, Somershoe S, Sather M, McNew LM (2021) Nest density drives productivity in chestnut-collared longspurs: implications for grassland bird conservation. PLoS ONE 16:e0256346. https://doi.org/10.1371/journal.pone.0256346
Reinking DL (2005) Fire regimes and avian responses in the central tallgrass prairie. In: Saab VA, Powell HDW (eds) Fire and avian ecology in North America. Stud Avian Biol 30:116–126
Renfrew R, Strong AM, Perlut NG, Martin SG, Gavin TA (2020) Bobolink (Dolichonyx oryzivorus). In: Rodewald PG (ed) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.boboli.01
Renfrew RB, Ribic CA (2003) Grassland passerine nest predators near pasture edges identified on videotape. Auk 120:371–383. https://doi.org/10.1642/0004-8038(2003)120[0371:GPNPNP]2.0.CO;2
Reynolds TD, Trost CH (1980) The response of native vertebrate populations to crested wheatgrass planting and grazing by sheep. J Range Manag 33:122–125. https://doi.org/10.2307/3898425
Reynolds TD, Rich TD, Stephens DA (2020) Sage thrasher (Oreoscoptes montanus). In: Poole AF, Gill FB (eds) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.sagthr.01
Ribic CA, Koford RR, Herkert JR (2009) Area sensitivity in North American grassland birds: patterns and processes. Auk 126:233–244. https://doi.org/10.1525/auk.2009.1409
Ribic CA, Ng CS, Koper N et al (2018) Diel fledging patterns among grassland passerines: relative impacts of energetics and predation risk. Auk 135:1100–1112. https://doi.org/10.1642/AUK-17-213.1
Ribic CA, Rugg DJ, Koper N et al (2019) Behavior of adult and young grassland songbirds at fledging. J Field Ornithol 90:143–153. https://doi.org/10.1111/jofo.12289
Rich TD, Wisdom M J, Saab VA (2005) Conservation of priority birds in sagebrush ecosystems. In: Ralph CJ, Rich TD (eds) Bird conservation implementation and integration in the Americas: Proceedings of the 3rd international partners in flight conference, 2002 March 20–24, Asilomar, California, vol 1. U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station, General Technical Report PSW-GTR-191. Albany, California, pp 589–606
Richardson AN, Koper N, White KA (2014) Interactions between ecological disturbances—burning and grazing and their effects on songbird communities in northern mixed-grass prairies. Avian Conserv Ecol 9:article 5. https://doi.org/10.5751/ACE-00692-090205
Rivero MJ, Grau-Campanario P, Mullan S et al (2021) Factors affecting site use preference of grazing cattle Studied from 2000 to 2020 through GPS tracking: a review. Sensors 21:article 2696. https://doi.org/10.3390/s21082696
Rodenhouse NL, Best LB, O’Connor RJ et al (1995) Effects of agricultural practices and farmland structures. In: Martin TE, Finch DM (eds) Ecology and management of neotropical migratory birds. Oxford University Press, New York, pp 269–293
Rosenberg KV, Dokter AM, Blancher PJ et al (2019) Decline of the North American avifauna. Science 366:120–124. https://doi.org/10.1126/science.aaw1313
Rotenberry JT, Wiens JA (1980) Habitat structure, patchiness, and avian communities in North American steppe vegetation—a multivariate analysis. Ecology 61:1228–1250. https://doi.org/10.2307/1936840
Rotenberry JT, Wiens JA (1991) Weather and reproductive variation in shrubsteppe sparrows—a hierarchical analysis. Ecology 72:1325–1335. https://doi.org/10.2307/1941105
Rotenberry JT, Wiens JA (1998) Foraging patch selection by shrubsteppe sparrows. Ecology 79:1160–1173. https://doi.org/10.1890/0012-9658(1998)079[1160:FPSBSS]2.0.CO;2
Ruehmann MB, Desmond MJ, Gould WR (2011) Effects of smooth brome on Brewer’s sparrow nest survival in sagebrush steppe. Condor 113:419–428. https://doi.org/10.1525/cond.2011.100022
Ruth JM (2015) Status Assessment and Conservation Plan for the grasshopper sparrow (Ammodramus savannarum). U.S. Fish and Wildlife Service, Lakewood. Vers. 1.0. https://digitalcommons.unl.edu/usfwspubs/471/
Ruth JM, Skagen SK (2018) Reproductive response of Arizona grasshopper sparrows to weather patterns and habitat structure. Condor 120:596–616. https://doi.org/10.1650/CONDOR-17-128.1
Ruth JM, Talbot WA, Smith EK (2020) Behavioral response to high temperatures in a desert grassland bird: use of shrubs as thermal refugia. West N Am Nat 80:265–275. https://doi.org/10.3398/064.080.0215
Ryan MR (1990) A dynamic approach to the conservation of the prairie ecosystem in the Midwest. In: Sweeney JM (ed) Management of dynamic ecosystems. The Wildlife Society, North Central Section, West Lafayette, Indiana
Saether BE, Bakke O (1997) Avian life history variation and contribution of demographic traits to the population growth rate. Ecology 81:642–653. https://doi.org/10.2307/177366
Sample DW, Mossman MJ (1997) Managing habitat for grassland birds—A guide for Wisconsin Wisconsin Department of Natural Resources, Madison, Wisconsin
Sanders LE, Chalfoun AD (2018) Novel landscape elements within natural gas fields increase densities but not fitness of an important songbird nest predator. Biol Conser 228:132–141. https://doi.org/10.1016/j.biocon.2018.10.020
Sauer JR, Link WA, Fallon JE, Pardieck DL, Ziolkowski DJ (2013) The North American breeding bird survey 1966–2011: summary analysis and species accounts. North American Fauna 79:1–32. https://doi.org/10.3996/nafa.79.0001
Sauer JR, Link WA, Hines JE (2020) The North American breeding bird survey, analysis results 1966–2019: U.S. Geological Survey data release. https://doi.org/10.5066/P96A7675
Scasta JD, Thacker ET, Hovick TJ, Engle DM, Allred BW, Fuhlendorf SD, Weir JR (2015) Patch-burn grazing (PBG) as a livestock management alternative for fire-prone ecosystems of North America. Renew Agric Food Syst 31:550–567. https://doi.org/10.1017/S1742170515000411
Scheiman DM, Bollinger EK, Johnson DH (2003) Effects of leafy spurge infestation on grassland birds. J Wildl Manage 67:115–121. https://doi.org/10.2307/3803067
Schroeder MH, Sturges DL (1975) The effects on the Brewer’s sparrow of spraying big sagebrush. J Range Manag 28:294–297
Shaffer JA, Buhl DA (2016) Effects of wind-energy facilities on breeding grassland bird distributions. Conserv Biol 30:59–71. https://doi.org/10.1111/cobi.12569
Shaffer JA, DeLong JP (2019) The effects of management practices on grassland birds: an introduction to North American grasslands and the practices used to manage grasslands and grassland birds. In: Johnson DH, Igl LD, Shaffer JA, DeLong JP (eds) The effects of management practices on grassland birds, chap. A. U.S. Geological Survey Professional Paper 1842. https://doi.org/10.3133/pp1842A
Shaffer JA, Igl LD, Johnson DH (2019a) The effects of management practices on grassland birds—rates of brown-headed cowbird (Molothrus ater) parasitism in nests of North American grassland birds. In: Johnson DH, Igl LD, Shaffer JA, DeLong JP (eds) The effects of management practices on grassland birds, chap. PP. U.S. Geological Survey Professional Paper 1842. https://doi.org/10.3133/pp1842PP
Shaffer JA, Loesch CR, Buhl DA (2019) Estimating offsets for avian displacement effects of anthropogenic impacts. Ecol Appl 29:e01983. https://doi.org/10.1002/eap.1983
Shaffer JA, Igl LD, Johnson DH, Sondreal ML, Goldade CM, Nenneman MP, Wooten TL, Euliss BR (2021) The effects of management practices on grassland birds—grasshopper sparrow (Ammodramus savannarum). In: Johnson DH, Igl LD, Shaffer JA, DeLong JP (eds) The effects of management practices on grassland birds, chap. GG. U.S. Geological Survey Professional Paper 1842. https://doi.org/10.3133/pp1842GG
Skagen SK, Yackel Adams A (2012) Weather effects on avian breeding performance and implications of climate change. Ecol Appl 22:1131–1145. https://doi.org/10.1890/11-0291.1
Sliwinski MS, Powell LA, Schacht WH (2019) Grazing systems do not affect bird habitat on a Sandhills landscape. Rangel Ecol Manag 72:136–144. https://doi.org/10.1016/j.rama.2018.07.006
Sliwinski MS, Powell LA, Schacht WH (2020) Similar bird communities across grazing systems in the Nebraska Sandhills. J Wildl Manage 84:802–812. https://doi.org/10.1002/jwmg.21825
Sliwinski MS, Koper N (2015) Managing mixed-grass prairies for songbirds using variable cattle stocking rates. Rangel Ecol Manag 68:470–475.https://doi.org/10.1016/j.rama.2015.07.010
Stinson LT, Pejchar L (2018) The effects of introduced plants on songbird reproductive success. Biol Invasions 20:1403–1416. https://doi.org/10.1007/s10530-017-1633-8
Stoleson SH, Finch DM (2001) Breeding bird use of and nesting success in exotic Russian olive in New Mexico. Wilson Bull 113:452–455
Swengel SR (1996) Management responses of three species of declining sparrows in tallgrass prairie. Bird Conserv Int 6:241–253. https://doi.org/10.1017/S0959270900003130
Temple SA (2020) Dickcissel (Spiza americana). In: Poole AF, Gill FB (eds) Birds of the world, vers. 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.dickci.01
Temple SA, Fevold BM, Paine LK, Undersander DJ, Sample DW (1999) Nesting birds and grazing cattle—accommodating both on midwestern pastures. In: Vickery PD, Herkert JR (eds) Ecology and conservation of grassland birds of the Western Hemisphere. Stud Avian Biol 19:196–202
Thompson SJ, Johnson DH, Niemuth ND, Ribic CA (2015) Avoidance of unconventional oil wells and roads exacerbates habitat loss for grassland birds in the North American Great Plains. Biol Conser 192:82–90. https://doi.org/10.1016/j.biocon.2015.08.040
Tizard I (2004) Salmonellosis in wild birds. Semin Avian Exotic Pet Med 13:50–66. https://doi.org/10.1053/j.saep.2004.01.008
Tripet F, Richner H (1997) Host responses to ectoparasites: food compensation by parent blue tits. Oikos 78:557–561. https://doi.org/10.2307/3545617
Valencia-Herverth J, Garrido D, Valencia-Herverth R (2018) Contributions to the knowledge of the distribution of birds in the state of Hidalgo, Mexico. Southwest Nat 63:83–87. https://doi.org/10.1894/0038-4909.63.83
Veblen KE, Pyke DA, Aldridge CL, Casazza ML, Assal TJ, Farinha MA (2014) Monitoring of livestock grazing effects on Bureau of Land Management land. Rangel Ecol Manag 67:68–77. https://doi.org/10.2111/REM-D-12-00178.1
Verheijen BHF, Erickson AN, Boyle WA, Leveritte KS, Sojka JL, Spahr LA, Williams EJ, Winnicki SK, Sandercock BK (2022) Predation, parasitism, and drought counteract the benefits of patch-burn grazing for the reproductive success of grassland songbirds. Ornithol Appl 124:1–22. https://doi.org/10.1093/ornithapp/duab066
Vickery PD (2020) Grasshopper sparrow (Ammodramus savannarum) In: Poole AF, Gill FB (eds) Birds of the world, vers 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.graspa.01
Vickery PD, Tubaro PL, da Silva JMC, Peterjohn BG, Herkert JR, Cavalcanti RB (1999) Conservation of grassland birds in the Western Hemisphere. In: Vickery PD, Herkert JR (eds) Ecology and conservation of grassland birds of the Western Hemisphere. Stud Avian Biol 19:2–26
Vold ST, Berkeley LI, McNew LB (2019) Effects of livestock grazing management on grassland birds in a northern mixed-grass prairie ecosystem. Rangel Ecol Manag 72:933–945. https://doi.org/10.1016/j.rama.2019.08.005
Walker BL, Igl LD, Shaffer JA (2020) The effects of management practices on grassland birds—Brewer’s sparrow (Spizella breweri breweri). In: Johnson DH, Igl LD, Shaffer JA, DeLong JP (eds) The effects of management practices on grassland birds, chap. AA. U.S. Geological Survey Professional Paper 1842. https://doi.org/10.3133/pp1842AA
Wheelwright NT, Rising JD (2020) Savannah sparrow (Passerculus sandwichensis). In: Poole AF (ed) Birds of the world, vers 1.0. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.savspa.01
Wilkins K, Pejchar L, Garvoille R (2019) Ecological and social consequences of bison reintroduction in Colorado. Conserv Sci Pract 1:e9. https://doi.org/10.1111/csp2.9
Williams MI, Paige GB, Thurow TL, Hild AL, Gerow KG (2011) Songbird relationships to shrub-steppe ecological site characteristics. Rangel Ecol Manage 64:109–118. https://doi.org/10.2111/REM-D-10-00076.1
Wilsey C, Taylor L, Bateman B, Jensen C, Michel N, Panjabi A, Langham G (2019) Climate policy action needed to reduce vulnerability of conservation-reliant grassland birds in North America. Conserv Sci Pract 1:e21. https://doi.org/10.1111/csp2.21
With KA (2021) Thick-billed longspur (Rhynchophanes mccownii). In: Poole AF (ed) Birds of the world, vers. 1.1. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/bow.mcclon.01.1
With KA, King AW, Jensen WE (2008) Remaining large grasslands may not be sufficient to prevent grassland bird declines. Biol Conser 141:3152–3167. https://doi.org/10.1016/j.biocon.2008.09.025
Yackel Adams A, Skagen SK, Savidge JA (2006) Modeling post-fledging survival of lark buntings in response to ecological and biological factors. Ecology 87:178–188. https://doi.org/10.1890/04-1922
Young AC, Cox W, McCarty JP, Wolfenbarger L (2019) Postfledging habitat selection and survival of Henslow’s sparrow: management implications for a critical life stage. Avian Conserv Ecol 14:article 10. https://doi.org/10.5751/ACE-01418-1402
Zalik NJ, Strong AM (2008) Effects of hay cropping on invertebrate biomass and the breeding ecology of Savannah sparrows (Passerculus sandwichensis). Auk 125:700–710. https://doi.org/10.1525/auk.2008.07106
Zimmerman JL (1997) Avian community responses to fire, grazing, and drought in the tallgrass prairie. In: Knopf FL, Samson FB (eds) Ecology and conservation of Great Plains vertebrates. Springer-Verlag, New York, pp 167–180
Zuckerberg B, Ribic CA, McCauley LA (2018) Effects of temperature and precipitation on grassland bird nesting success as mediated by patch size. Conserv Biol 32:872–882. https://doi.org/10.1111/cobi.13089
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Chalfoun, A.D., Johnson, T.N., Shaffer, J.A. (2023). Rangeland Songbirds. In: McNew, L.B., Dahlgren, D.K., Beck, J.L. (eds) Rangeland Wildlife Ecology and Conservation . Springer, Cham. https://doi.org/10.1007/978-3-031-34037-6_12
Download citation
DOI: https://doi.org/10.1007/978-3-031-34037-6_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-34036-9
Online ISBN: 978-3-031-34037-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)