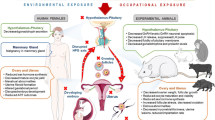
Abstract
Environmental pollution is a global issue caused by the introduction of chemical, biological, and physical substances in the environment. The short- and long-term effects of these substances on human reproduction are a present matter of concern, especially in the developed countries. Several mechanisms may be involved in reproductive damage caused by environmental pollutants including hormone-mediated effects, oxidative stress, and direct genetic damage. In recent times, big efforts have been made for the identification of the harmful pollutants, their mechanism of interaction with human fertility, tolerable limits, and possible solutions to minimize the human exposure. In this chapter, we summarize the current knowledge on the impact of environmental pollutants on human reproduction, with a special focus on their mechanisms of action.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Infertility
- Reproductive health
- Pollutants
- Environment
- Chemical exposure
- Oxidative stress
- Endocrine disruptors
4.1 Introduction
4.1.1 Recent Trends in Human Reproduction
Human reproductive health (defined as a state of physical, emotional, mental, and social well-being in relation to sexuality) is a matter of great concern in the new millennium. Over the past half century, there has been a growing trend toward delayed motherhood in the developed countries. In 2019, 29.4 years was the mean age of women at birth of the first child in European Union. The lowest mean age at birth of the first child was observed in Bulgaria (26.3 years), while the highest values were found in Italy (31.3 years) [1].
Postponement of parenthood has decreased the total fertility rates in almost all the European countries, with a mean fertility rate per woman of 1.53 in 2019 (ranging from 1.86 live births per woman in France to less than 1.3 in Italy, Spain, and Malta). Overall, the number of live births in Europe has decreased by 50% approximately, in the last 50 years. In parallel to the decreased global fertility rates, the chance of a couple remaining involuntarily childless has dramatically increased. A systematic analysis of 277 health surveys conducted by the World Health Organization (WHO) found that 25% of couples were affected by infertility in 2012 [2]. Infertility is defined as “the failure to achieve a pregnancy after 12 months or more of regular unprotected sexual intercourse.” The rise in infertility has been accompanied by the rapid diffusion of assisted reproductive technologies (ARTs) worldwide. Over 9 million in vitro fertilization (IVF) children have been born, and over 2.5 million cycles are performed annually, resulting in over 500,000 deliveries every year. Although ARTs have an increasing contribution to the overall birth rate, they can only partially compensate for the drop in fertility rates in the developed countries [2].
The risk of infertility increases with advancing age. The relationship between age and fertility is particularly significant for the female gender, in which a decline in fertility occurs early. By the age of 30, female fertility starts to decline. The decline becomes more rapid once women reach their mid-30s. By the age of 45, the majority of women are infertile. The key reasons for age-related infertility include reduced number and competence of oocytes due to aging insults, resulting in a higher risk of embryo aneuploidies. With respect to males, a significant reduction of fertility is described around the age of 40–45 years, mainly due to worsening of sperm number and motility. Therefore, in general, increasing parental age reduces the overall chances of pregnancy and increases the risk of spontaneous miscarriage [3].
In addition to parental age, there are many causes of infertility including female factors (e.g., ovulation disorders, tubal disease, endometriosis, uterine abnormalities, and reduced ovarian reserve) and male problems (e.g., varicocele, obstructive disorders, and testicle insufficiency), while 20–30% of cases are idiopathic [4]. With respect to age-independent infertility factors, an alarming phenomenon was recently described in males, also defined as “the male infertility crisis.” Such a crisis refers to a steady annual decline of 1.4% in sperm counts from 1970 (with an overall decline of 52.4% in the last 40 years) to date. The causes of this phenomenon are partly unexplained. However, it is a common belief among scientists that some environmental factors may have played a role in determining a general worsening of human reproductive health.
Environmental pollutants are chemical, biological, and physical substances introduced in the environment as a result of human activities. The short- and long-term effects of these substances on human reproduction are a present matter of concern, especially in the developed countries. Several mechanisms may be involved in reproductive damage caused by environmental pollutants including hormone-mediated effects, oxidative stress, and direct genetic damage. In this chapter, we introduce the most relevant aspects inherent to the relationship between environment pollutants and human reproductive health.
4.1.2 Environmental Pollutants
Environmental pollution is the contamination of the environment (air, water, and land) with substances originated from man’s activities such as urbanization, industrialization, mining, and exploration. It represents the world’s greatest problem faced by humans and a major cause of human morbidity and mortality. According to the recent analyses, environmental pollution may account for 9 million deaths each year worldwide.
Over the last decades, numerous environmental pollutants were established as potential risk factors for various acute and chronic diseases in humans. Although the pathogenic effects of pollutants are often evaluated individually, their action is simultaneous and cumulative if one considers the countless possible sources of exposure [5].
The detrimental effects of pollutants on human fertility are supposed to vary based on the age of the exposed subject. Theoretically, the earlier the exposure (i.e., from intrauterine life to adolescence), the greater the resulting reproductive damage. However, the adults are not exempt from sequelae.
In addition, as an individual’s intensity of exposure increases, so the severity of the damage will increase.
The mechanisms of interaction between pollutants and reproductive health can be academically divided into three classes:
-
1.
Endocrine-disrupting chemicals (EDCs): The EDCs are “exogenous chemicals, or mixture of chemicals, that interfere with any aspect of hormone action.” This action can be distinguished in transitory or permanent depending on the time of exposure. This mechanism is typical of polycyclic aromatic hydrocarbons (PAHs) and heavy metals (Cu, Pb, Zn, etc.) contained in particulate matter (PM), especially from diesel exhaust. EDCs can influence ovarian reserve by acting mainly on the aryl hydrocarbon receptor (AhR) or estrogen receptors (ERs). After binding the exogenous ligand, AhR translocates toward the nucleus; it associates with a nuclear receptor and is able to bind to DNA sequences and modulate gene transcription. AhR induces Bax synthesis, namely a proapoptotic factor contributing to follicular atresia [6]. In addition, diesel exhaust particles contain substances with estrogenic, antiestrogenic, and antiandrogenic activities that can affect gonadal steroidogenesis and gametogenesis. ERs play a crucial role during the early phase of folliculogenesis in humans, as they are increasingly expressed from the primordial stage onward [7]. Moreover, they are consistently expressed by oocytes in human fetal ovaries whatever the follicular stage [8]. In different mammals, estrogens can interfere with primordial follicle formation: in a positive manner in primates and bovines and in a negative manner in mice [8].
-
2.
Induction of oxidative stress: Reactive oxygen species (ROSs) are normally balanced in the organism. However, in case of augmented ROS production or reduced ROS metabolism, oxidative stress occurs. In this situation, the ovarian function can be negatively influenced, because ROS may lead to antral follicle apoptosis [9]. In this respect, solid data found that oxidative stress markers are increased in patients with primary ovarian insufficiency (POI) syndrome [10]. This mechanism is demonstrated for nitrogen oxide (NO2), ozone (O3), or PM (through the heavy metals and PAHs they contain) pollutant.
-
3.
Modifications of DNA: Molecules can create alterations to the DNA chain through the formation of DNA adducts. This type of interaction can lead to modifications in gene expression. Moreover, the exposure to pollutants could cause epigenetic modifications on the three-dimensional DNA structure, due to alterations in DNA methylation. If these modifications affect the germ line in a nonmodifiable way, the mutation will be transmitted to the offspring [11].
4.1.2.1 Air Pollution
Air pollution has been considered for decades as a cause of concern for human fertility. Particulate matter (PM) and ground-level ozone (O3) are Europe’s most troubling pollutants, followed by benzo(a)pyrene (BaP). The main sources of these pollutants are transport and energy chains followed by the industries. The correlation between PM levels in the atmosphere and infertility has been largely investigated. It was found that every increase of 10 μg/m3 in PM2.5 concentration was associated with a 22% fecundability decrease (95% confidence interval [CI] = 6–35%) [12]. A further confirmation comes from an observational study with over 36,000 nurses, which showed a direct association between infertility and the proximity of residence to a main road. Hazard ratio (HR) for infertility when living close to major roads compared with farther was 1.11 (CI = 1.02–1.20). The authors therefore concluded that air pollution has a potentially harmful effect on fertility. In addition, the HR for every 10 μg/m3 increase in cumulative PM2.5–10 among women with primary infertility was 1.10 (CI = 0.96–1.27), and similarly, it was 1.10 (CI: 0.94–1.28) for those with secondary infertility [13].
4.1.2.2 Polycyclic Aromatic Hydrocarbons
Polycyclic aromatic hydrocarbons (PAHs) are a class of common environmental pollutants found in water, air, soil, and plants. They can be released by the natural sources; however, the vast majority derives from vehicular emissions, coal-burning plants, and the production and use of petroleum-derived substances. Exposure to PAHs has been associated with the onset of cancer and other diseases, including infertility. Their deleterious action on female reproductive system comes from their ability to interact with the pituitary–ovarian axis, causing alterations on the ovarian physiology and function [14].
4.1.2.3 Pesticides
Pesticides have wide applications in agriculture, especially with growing mass production for commercial export. At toxic doses, they cause oxidative stress due to a direct damage to antioxidant defense system. These substances have been associated with a variety of reproductive issues in males (e.g., germ cell apoptosis, hypotestosteronemia, and asthenozoospermia) and females (e.g., oligoanovulation, impaired folliculogenesis, follicular atresia, implantation defects, and endometriosis) and with obstetrical complications including spontaneous abortions and fetal malformations. For all these reasons, which will be extensively debated, pesticides directly affect fertility and reproductive physiology of the organism [15].
4.1.2.4 Parabens
Parabens are chemical substances used as preservatives in foods, cosmetics, and pharmaceutical products. These substances are endocrine disruptors that mainly act by mimicking the sex hormones, resulting in reproductive imbalance in both males and females [16]. In males, parabens were shown to affect total sperm count, semen motility, and morphology. In females, toxic exposure to parabens (as assessed through an increase in urinary propylparaben) was associated with short menstrual cycle length, low antral follicle count, and high cycle Day 3 follicle-stimulating hormone (FSH) level. Low antral follicle count and high FSH levels are markers of diminished ovarian reserve and reduced success rates of fertility treatments [17]. Other studies showed a variety of hormonal abnormalities after parabens exposure. In particular, increased levels of butylparaben were associated with lower levels of endogenous estradiol levels, altered thyroid hormone levels, and shifts in estradiol/progesterone ratios in women [18].
4.1.2.5 Perfluorooctanesulfonate (PFOS)/Perfluorooctanoate (PFOA)
Perfluorinated chemicals are substances widely used in everyday items such as food packaging, pesticides, clothing, upholstery, carpets, and personal care products. They have been extensively studied, and recent findings led to the acquaintance that they could be associated with infertility in women and men.
It was found that women who had higher levels of perfluorooctanoate (PFOA) and perfluorooctanesulfonate (PFOS) in their blood took longer to achieve a pregnancy than women with lower levels. The researchers divided the women’s levels of PFOS/PFOA into four quartiles and found that, compared with women with the lowest levels of exposure, the likelihood of infertility increased by 70–134% for women in the higher three quartiles of PFOS exposure and by 60–154% for women in the higher three quartiles of PFOA exposure [19]. In men, the recent studies have suggested that PFOA/PFOS exposure can lead to hypotestosteronemia and reduction in semen quality.
The PFOS/PFOA role in male and female infertility is now under the spotlight, deserving an appropriate discussion in a later section.
Considering the wide distribution of different pollutant molecules in the environment and the difficulty in approaching their debate, we consider eligible an academic and a systematic approach. For this reason, here we present two main examples of environmental pollutants and how they could affect human reproductive health.
4.1.3 A Striking Example: The Role of Bisphenol
Bisphenol-A, 2,2-bis(4-hydroxyphenyl) propane (BPA), is one of the most investigated bisphenols. It is largely found in polycarbonate resin mainly used for plastic bags, bottles, baby battles and packaging, coated tins, particularly food and drink cans, and microwave ovenware. More than 90% of the overall exposure to BPA is diet being it a constituent of food containers and packaging, because it can leach into food products, especially after heating. The exposure to BPA occurring through the dermal absorption by the handling of thermal paper or by the application of cosmetics, together with air inhalation and dust and dental material ingestion, represents only the 5% of BPA exposure [20, 21]. In accordance with the Chapel Hill BPA consensus statement, “low BPA doses” have been considered in human epidemiological studies below the reference dose of tolerable daily intake (TDI), corresponding to 0.05 mg/kg (50 μg/kg) body weight/day as established by the United States Environmental Protection Agency (EPA). According to the World Health Organization (WHO) and the Food and Agriculture Organization (FAO) of the United Nations, in Europe, it has been estimated that BPA daily intake is around 0.2 μg/kg bw/day in breast-fed babies and around 11 g/kg bw/day in formula-fed babies for which feeding polycarbonate bottles were used. The estimated daily intake for adults is around 1.5 g/kg bw/day [20, 21].
4.1.3.1 Bisphenol Pathophysiological Impact
BPA acts through a dual mechanism. First, it acts as endocrine-disrupting chemical, thus affecting hormone synthesis, metabolism, and function [22]. In several in vivo and in vitro studies, it has been demonstrated the high affinity of bisphenol-A for estrogen receptors (ERs), having an estrogen-mimicking behavior and consequently stimulating estrogen function [20, 21, 23]. Therefore, BPA has been supposed to be involved in many diseases of female reproductive system [24, 25], due to its property to stimulate ER-dependent gene expression involved in the pathophysiology of female reproductive system [26,27,28,29]. Indeed, BPA has a conformational structure that confers the ability to bind both ER alpha (ERα) and ER beta (ERβ), although, according to the in vivo models, the affinity of BPA for ER is 1000-fold to 10,000-fold less than the affinity of 17β-estradiol (E2) [30].
Second, BPA has a slow action on genomic pathways interacting with nuclear ER and regulating several gene expressions; in addition, BPA has a rapid action through nongenomic pathways, activating, for example, the kinase signaling cascades or modulating the calcium flux through the cell’s walls [31]. Genomic and nongenomic mechanisms can be triggered by the low- and high-dose exposure of BPA [32].
Taking together these modalities of action, BPA has a different effect if the exposure occurs during prenatal, perinatal, or postnatal period. Deleterious effects are more critical during perinatal exposure, causing dysregulation of hypothalamic–pituitary–ovarian (HPO) axis in babies and adults, with a precocious maturation of the axis through a damage of gonadotropin-releasing hormone (GnRH) pulsatility, gonadotropin signaling, and sex steroid hormone production. Further, a transmission from the pregnant woman to the developing fetus or child through the placenta and breast milk (during gestation and lactation) was also demonstrated, causing BPA-related diseases [33, 34].
4.1.3.2 Bisphenol A and Infertility
Increasing evidence has suggested that BPA might contribute to the pathogenesis of female and male infertility. The hypothetical impact of BPA on natural conception has been investigated in several observational studies [6, 35,36,37]. The number of subjects with detectable BPA levels (limit of detection [LOD] of assay: 0.5 ng/mL) was higher in infertile than in fertile women [6] and above all in infertile women who live in metropolitan areas [36]. Humans and rodents share the same regulation of reproductive system by the hypothalamic–pituitary–ovarian (HPO) axis. Hypothalamus releases gonadotropin-releasing hormone (GnRH) in rhythmic pulses; the pituitary gland secretes follicle-stimulating hormone (FSH) and luteinizing hormone (LH); the ovary releases sex hormones, including estradiol and progesterone, controlling the function of reproductive system. Most studies were performed on rat models with variation regarding to the exact timing of toxic exposure during their development. The findings of studies conducted on animal models pointed out that the deleterious effect of BPA could vary depending on doses, administration route, window of exposure, and animal models.
Morphological and functional changes in the reproductive system due to BPA can impair female fertility. BPA is able to inhibit androgen function by binding androgen receptors (ARs) [38], resulting in altered ovarian steroidogenesis [39,40,41,42,43,44] and folliculogenesis [41, 45, 46]. Moreover, BPA can influence endometrial receptivity, resulting in impaired embryo implantation [47,48,49,50].
Males are not exempt from reproductive damage due to BPA. In the postpubertal male, BPA is able to interfere with sex hormone synthesis, expression, and function of the respective receptors, resulting in reduced libido and ejaculatory defects. The effects are more detrimental during in utero exposure, as BPA was found to cause a variety of defects including feminization, atrophy of testes and epididymis, increased prostate size, and alteration of adult sperm parameters.
4.1.3.2.1 The Reproductive Organ Impairment in Females
The effects of BPA on ovarian, oviduct, and uterus morphology and functions in humans are still unclear, although different authors suggested that BPA exposure can affect in utero morphogenesis and the reproductive function in adults. Most of the experimental studies have been performed on mice; for both humans and rodents, the ovarian development is a dynamic process consisting in the growth of the ovary and establishment of the finite pool of primordial follicles, occurring predominantly during the embryonic period [51]. The functional alterations of ovary, which can be the cause or consequence of alterations of ovarian morphology, include mainly the impairment of folliculogenesis, beyond the impairment of steroidogenesis and sex hormones production.
The BPA action on the ovary and on the sex hormone secretion has been investigated in female pups and adult animals during different phases of the estrus cycle. Indeed, in rat and mouse female offspring perinatally, prenatally, and postnatally exposed to oral, gavage, and subcutaneous administration of low and high BPA doses, increased circulating E2 levels have been recorded [40, 42, 52, 53]. In addition to the regulation of sex hormone synthesis, the exposure to low BPA doses increased messenger and protein expression of FSH receptor (FSHR) too in the ovarian tissue of female adult rats [54].
4.1.3.2.2 The Folliculogenesis Impairment
BPA may interfere with multiple molecular processes and pathways involved in folliculogenesis. Indeed, it has been shown that BPA enhances ER messenger expression in ovary and, through the binding to ER, induces epigenetic modifications, in particular DNA hypomethylation, of genes involved in oocyte maturation, with a consequent acceleration of the transformation of the primordial to primary follicles [55]. The administration of low BPA doses in ovaries explanted from mouse female pups inhibited germ cell nest breakdown and enhanced primordial follicle recruitment, by decreasing the expression of Ki-67, Fas, Bac, Bax, and Caspase 3 and 8; increasing the expression of Bcl2; and activating phosphoinositide-3-kinase (PI3K)/Akt pathway. Low BPA doses accelerated follicle development with an increase in antral follicle growth [46], while high BPA doses selectively inhibited antral follicle growth [42, 45, 56]. Low BPA doses’ effects on antral follicles were found to be associated with high methylation level of several maternally and paternally imprinted genes [46], whereas high BPA doses’ effects on antral follicles were found to be mediated by interference with the expression of genes involved in cell cycle progression (increased expression of cyclin-dependent kinase 4 (CDK4) and cyclin E1 (CCNE1) and decreased expression of cyclin D2 (CCND2), and apoptosis (increased expression of p53, Bcl-2, and Bax) [57].
However, the disruption of folliculogenesis seems to be a reversible process depending on the timing of BPA exposure. Indeed, when BPA exposure occurs in adults, the damage appears to be transient (with a reduction in the number of antral follicles), but a restoration of pre-exposure conditions few weeks later is generally observed. On the contrary, when the exposure to BPA occurs during the postnatal period, a persistent disruption of folliculogenesis is noticed (i.e., a decreased number of primordial follicles and an increased number of atretic follicles persisting in adulthood) [58]. Alarmingly, a recent study on a cohort of women undergoing IVF reported that higher urinary BPA levels were associated with lower antral follicle count, raising concerns for a possible accelerated follicle loss [59].
4.1.3.2.3 The Altered Embryo Implantation
Experimental ex vivo studies on uterus confirmed that exposure to BPA affected uterine function, particularly interfering with uterine receptivity and embryo implantation. An ex vivo study, conducted on uterus of female adult mice treated with BPA in the first 3 days of pregnancy, demonstrated that high BPA doses delayed the transfer of embryos to the uterus, damaged blastocyst development before implantation, and inhibited embryo implantation. Regarding the putative mechanism, high BPA dose exposure induces a dose-dependent increase of endothelial nitric oxide synthase (eNOS) protein expression in trophoblast cells, the cells forming the outer layer of a blastocyst, with a consequent induction of excess nitrogen monoxide (NO), which might represent one of the causal factors involved in embryo implantation [47].
Unfavorable embryo implantation was also observed in female adult mice exposed to low BPA dose treatment, in which ex vivo analysis of uterus showed reduced decidual cells surrounding the attached embryo and an increased percentage of intrauterine hemorrhage, due to the shedding and collapse of the endometrium [55].
The impairment of uterine receptivity and the unfavorable embryo implantation can be addressed by the BPA capability to increase uterine luminal area, enhancing the uterine luminal epithelial cell height, and capability to affect E2 and heart and neural crest derivatives expressed 2 (P/HAND2) pathways [56]. Indeed, BPA-exposed uterine epithelial and stromal tissues showed a marked suppression of E2 and P receptor expression and P receptor downstream target gene, HAND2. These factors enhance the activation of fibroblast growth factor and mitogen‑activated protein kinase (MAPK) signaling in the epithelium, thus contributing to aberrant proliferation, lack of uterine receptivity, and impaired embryo implantation [47,48,49,50].
In conclusion, prenatal, perinatal, and postnatal BPA exposure may influence the following: (1) ovarian development, by reducing the breakdown of germ cell nest, as a consequence of deregulated expression of apoptotic genes; (2) oviduct morphology; (3) folliculogenesis, by downregulating the expression of cell cycle regulatory genes and steroidogenic enzymes, leading to increased follicle apoptosis and premature ovarian insufficiency; and (4) uterine receptivity and embryo implantation.
4.1.3.3 To Strengthen the Concept: The Role of Phthalates
Phthalates are chemical substances that are mainly used as plasticizers in disposable and non-disposable products. Since their widespread use in factories and environment, knowledge on their potential effects on human health is of paramount importance. Notably, different disorders were described in connection to phthalates exposure, including infertility [60].
Chemically, phthalates are esters of 1,2-benzene dicarboxylic acid with a structure that varies according to the number of side chains. Side chains can be composed by dialkyl, alkyl, or aryl groups. Physically, phthalates are colorless, oily, and odorless substances, with a low solubility in water, which is inversely correlated to the chain length. Conversely, phthalic acid derivatives are more soluble in organic solvents [60,61,62].
Regarding their role in human pathology, phthalates are classified as endocrine disruptors (EDs), namely specific substances hampering the hormonal balance in males and females. Regarding males, phthalates can affect their reproductive function with different mechanisms. First, they can interfere with male reproductive system development [63]. Second, they can induce testicular dysgenesis syndrome (TDS) being responsible for decreased testis weight, spermatogenesis dysfunction, and external genital malformations (shortened anogenital distance, hypospadias, and cryptorchidism) [64]. Third, they can be responsible for male puberty dysfunction [65]. Fourth, phthalates can induce cancer in male reproductive organs [66].
In females, a putative negative effect of phthalates on ovarian function has been hypothesized. Studies on mice showed a considerable reduction of antral follicle development [67] after phthalate exposure, up to follicle exhaustion and premature ovarian failure (POF). Perhaps, the antiestrogenic activity of phthalates may be involved in POF [68]. When the exposure occurs in postnatal age, the natural onset of female puberty can be anticipated or delayed [69, 70]. In addition, exposure to phthalate during pregnancy may somehow result in spontaneous miscarriage and other obstetric complications [71]. Finally, also in women, a role of phthalates in carcinogenesis of the genital tract cannot be excluded [72].
References
EUROSTAT. Last accessed 12 Feb 2022. https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Fertility_statistics
Mascarenhas MN, Flaxman SR, Boerma T, Vanderpoel S, Stevens GA. National, regional, and global trends in infertility prevalence since 1990: a systematic analysis of 277 health surveys. PLoS Med. 2012;9(12):e1001356. https://doi.org/10.1371/journal.pmed.1001356. Epub 2012 Dec 18. PMID: 23271957; PMCID: PMC3525527.
Gnoth C, Godehardt E, Frank-Herrmann P, Friol K, Tigges J, Freundl G. Definition and prevalence of subfertility and infertility. Hum Reprod. 2005 May;20(5):1144–7. https://doi.org/10.1093/humrep/deh870.
Bellver J, Donnez J. Introduction: infertility etiology and offspring health. Fertil Steril. 2019;111(6):1033–5. https://doi.org/10.1016/j.fertnstert.2019.04.043. PMID: 31155112
Park SK, Tao Y, Meeker JD, Harlow SD, Mukherjee B. Environmental risk score as a new tool to examine multi-pollutants in epidemiologic research: an example from the NHANES study using serum lipid levels. PLoS One. 2014;9(6):e98632. Published 2014 Jun 5. https://doi.org/10.1371/journal.pone.0098632.
Caserta D, Bordi G, Ciardo F, Marci R, La Rocca C, Tait S, et al. The influence of endocrine disruptors in a selected population of infertile women. Gynecol Endocrinol. 2013;29(5):444–7.
Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, Gore AC. Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev. 2009;30:293–342. https://doi.org/10.1210/er.2009-0002.
Fernandez M, Bianchi M, Lux-Lantos V, Libertun C. Neonatal exposure to bisphenol a alters reproductive parameters and gonadotropin releasing hormone signaling in female rats. Environ Health Perspect. 2009;117(5):757–62.
Luderer U. Ovarian toxicity from reactive oxygen species. Vitam Horm. 2014;94:99–127. https://doi.org/10.1016/B978-0-12-800095-3.00004-3.
Tokmak A, Yildirim G, Sarikaya E, Cinar M, Bogdaycioglu N, Yilmaz FM, et al. Increased oxidative stress markers may be a promising indicator of risk for primary ovarian insufficiency: a cross-sectional case control study. Rev Bras Ginecol Obstet. 2015;37:411–6.
Nilsson E, Larsen G, Manikkam M, Guerrero-Bosagna C, Savenkova MI, Skinner MK. Environmentally induced epigenetic transgenerational inheritance of ovarian disease. PLoS One. 2012;7:e36129. https://doi.org/10.1371/journal.pone.0036129.
Slama R, Bottagisi S, Solansky I, Lepeule J, Giorgis-Allemand L, Sram R. Short-term impact of atmospheric pollution on fecundability. Epidemiology. 2013:871–879. doi:10.1097/EDE.0b013e3182a702c5.
Mahalingaiah S, Hart JE, Laden F, Farland LV, Hewlett MM, Chavarro J, Aschengrau A, Missmer SA. Adult air pollution exposure and risk of infertility in the Nurses' health study II. In: Human Reprod, vol. 31; 2016. p. 638. https://doi.org/10.1093/humrep/dev330.
Ramesh A, Archibong AE, Niaz MS. Ovarian susceptibility to benzo[a]pyrene: tissue burden of metabolites and DNA adducts in F-344 rats. J Toxicol Environ Health A. 2010;73(23):1611–25.
Kumar BJ, Mittal M, Saraf P, Kumari P. Pesticides induced oxidative stress and female infertility: a review. Toxin Reviews. 2018;39:1–13. https://doi.org/10.1080/15569543.2018.14749266.
Jurewicz J, et al. 2017 Human semen quality, sperm DNA damage, and the level of reproductive hormones in relation to urinary concentrations of parabens. J Occup Envrion Med. 2017;59:1034–40.
Smith KW, Souter I, Dimitriadis I, Ehrlich S, Williams PL, Calafat AM, Hauser R. Urinary paraben concentrations and ovarian aging among women from a fertility center. Environ Health Perspect. 2013;121(11–12):1299–305.
Nishihama Y, Yoshinaga J, Iida A, Konishi S, Imai H, Yoneyama M, Nakajima D, Shiraishi H. Association between paraben exposure and menstrual cycle in female university students in Japan. Reprod Toxicol. 2016;63:107–13.
Fei C, McLaughlin JK, Lipworth L, Olsen J. Maternal levels of perfluorinated chemicals and subfecundity. Hum Reprod. 2009;24(5):1200–5. https://doi.org/10.1093/humrep/den490. Epub 2009 Jan 28. PMID: 19176540.
Bisphenol A (BPA)-current state of knowledge and future actions by WHO and FAO, in International Food Safety Authorities Network (INFOSAN) Information Note No. 5/2009–Bisphenol A. 2009.
Konieczna A, Rutkowska A, Rachon D. Health risk of exposure to bisphenol A (BPA). Rocz Panstw Zakl Hig. 2015;66(1):5–11.
Gregoraszczuk EL, Ptak A. Endocrine-disrupting chemicals: some actions of POPs on female reproduction. Int J Endocrinol. 2013;2013:828532.
Fenichel P, Chevalier N, Brucker-Davis F. Bisphenol A: an endocrine and metabolic disruptor. Ann Endocrinol (Paris). 2013;74(3):211–20.
Sifakis S, Androutsopoulos VP, Tsatsakis AM, Spandidos DA. Human exposure to endocrine disrupting chemicals: effects on the male and female reproductive systems. Environ Toxicol Pharmacol. 2017;51:56–70.
Wei M, Chen X, Zhao Y, Cao B, Zhao W. Effects of prenatal environmental exposures on the development of endometriosis in female offspring. Reprod Sci. 2016;23(9):1129–38.
Chen X, Wang Y, Xu F, Wei X, Zhang J, Wang C, et al. The rapid effect of Bisphenol-A on long-term potentiation in hippocampus involves estrogen receptors and ERK activation. Neural Plast. 2017;2017:5196958.
Richter CA, Birnbaum LS, Farabollini F, Newbold RR, Rubin BS, Talsness CE, et al. In vivo effects of bisphenol A in laboratory rodent studies. Reprod Toxicol. 2007;24(2):199–224.
Shi XY, Wang Z, Liu L, Feng LM, Li N, Liu S, et al. Low concentrations of bisphenol A promote human ovarian cancer cell proliferation and glycolysis-based metabolism through the estrogen receptor-alpha pathway. Chemosphere. 2017;185:361–7.
Zhang Y, Wei F, Zhang J, Hao L, Jiang J, Dang L, et al. Bisphenol A and estrogen induce proliferation of human thyroid tumor cells via an estrogen-receptor-dependent pathway. Arch Biochem Biophys. 2017;633:29–39.
Toxicological and health aspects of bisphenol A, In report of joint FAO/WHO expert meeting. 2010. Ottawa.
Shanle EK, Xu W. Endocrine disrupting chemicals targeting estrogen receptor signaling: identification and mechanisms of action. Chem Res Toxicol. 2011;24(1):6–19.
Alonso-Magdalena P, Ropero AB, Soriano S, Garcia-Arevalo M, Ripoll C, Fuentes E, et al. Bisphenol-A acts as a potent estrogen via non-classical estrogen triggered pathways. Mol Cell Endocrinol. 2012;355(2):201–7.
Cao XL, Zhang J, Goodyer CG, Hayward S, Cooke GM, Curran IH. Bisphenol A in human placental and fetal liver tissues collected from greater Montreal area (Quebec) during 1998–2008. Chemosphere. 2012;89(5):505–11.
Lee J, Choi K, Park J, Moon HB, Choi G, Lee JJ, et al. Bisphenol A distribution in serum, urine, placenta, breast milk, and umbilical cord serum in a birth panel of mother-neonate pairs. Sci Total Environ. 2018;626:1494–501.
Buck Louis GM, Sundaram R, Sweeney AM, Schisterman EF, Maisog J, Kannan K. Urinary bisphenol A, phthalates, and couple fecundity: the longitudinal investigation of fertility and the environment (LIFE) study. Fertil Steril. 2014;101(5):1359–66.
La Rocca C, Tait S, Guerranti C, Busani L, Ciardo F, Bergamasco B, et al. Exposure to endocrine disrupters and nuclear receptor gene expression in infertile and fertile women from different Italian areas. Int J Environ Res Public Health. 2014;11(10):10146–64.
Velez MP, Arbuckle TE, Fraser WD. Female exposure to phenols and phthalates and time to pregnancy: the maternal-infant research on environmental chemicals (MIREC) study. Fertil Steril. 2015;103(4):1011–20 e2.
Lee HJ, Chattopadhyay S, Gong EY, Ahn RS, Lee K. Antiandrogenic effects of bisphenol a and nonylphenol on the function of androgen receptor. Toxicol Sci. 2003;75(1):40–6.
Zhou W, Liu J, Liao L, Han S, Liu J. Effect of bisphenol a on steroid hormone production in rat ovarian theca-interstitial and granulosa cells. Mol Cell Endocrinol. 2008;283(1–2):12–8.
Xi W, Lee CKF, Yeung WSB, Giesy JP, Wong MH, Zhang XW, et al. Effect of perinatal and postnatal bisphenol A exposure to the regulatory circuits at the hypothalamus-pituitary-gonadal axis of CD-1 mice. Reprod Toxicol. 2011;31(4):409–17.
Peretz J, Gupta RK, Singh J, Hernandez-Ochoa I, Flaws JA. Bisphenol a impairs follicle growth, inhibits steroidogenesis, and downregulates rate-limiting enzymes in the estradiol biosynthesis pathway. Toxicol Sci. 2011;119(1):209–17.
Gamez JM, Penalba R, Cardoso N, Bernasconi PS, Carbone S, Ponzo O, et al. Exposure to a low dose of bisphenol A impairs pituitary-ovarian axis in prepubertal rats effects on early folliculogenesis. Environ Toxicol Pharmacol. 2015;39(1):9–15.
Mansur A, Adir M, Yerushalmi G, Hourvitz A, Gitman H, Yung Y, et al. Does BPA alter steroid hormone synthesis in human granulosa cells in vitro? Hum Reprod. 2016;31(7):1562–9.
Peretz J, Flaws JA. Bisphenol A down-regulates rate-limiting Cyp11a1 to acutely inhibit steroidogenesis in cultured mouse antral follicles. Toxicol Appl Pharmacol. 2013;271(2):249–56.
Peretz J, Craig ZR, Flaws JA. Bisphenol A inhibits follicle growth and induces atresia in cultured mouse antral follicles independently of the genomic estrogenic pathway. Biol Reprod. 2012;87(3):63.
Trapphoff T, Heiligentag M, El Hajj N, Haaf T, Eichenlaub-Ritter U. Chronic exposure to a low concentration of bisphenol A during follicle culture affects the epigenetic status of germinal vesicles and metaphase II oocytes. Fertil Steril. 2013;100(6):1758–67 e1.
Pan X, Wang X, Sun Y, Dou Z, Li Z. Inhibitory effects of preimplantation exposure to bisphenol-a on blastocyst development and implantation. Int J Clin Exp Med. 2015;8(6):8720–9.
Xiao S, Diao H, Smith MA, Song X, Ye X. Preimplantation exposure to bisphenol A (BPA) affects embryo transport, preimplantation embryo development, and uterine receptivity in mice. Reprod Toxicol. 2011;32(4):434–41.
Berger RG, Foster WG, de Catanzaro D. Bisphenol-A exposure during the period of blastocyst implantation alters uterine morphology and perturbs measures of estrogen and progesterone receptor expression in mice. Reprod Toxicol. 2010;30(3):393–400.
Varayoud J, Ramos JG, Bosquiazzo VL, Lower M, Munoz-de-Toro M, Luque EH. Neonatal exposure to bisphenol A alters rat uterine implantation-associated gene expression and reduces the number of implantation sites. Endocrinology. 2011;152(3):1101–11.
Grive KJ, Freiman RN. The developmental origins of the mammalian ovarian reserve. Development. 2015;142(15):2554–63.
Naule L, Picot M, Martini M, Parmentier C, Hardin-Pouzet H, Keller M, et al. Neuroendocrine and behavioral effects of maternal exposure to oral bisphenol A in female mice. J Endocrinol. 2014;220(3):375–88.
Fernandez M, Bourguignon N, Lux-Lantos V, Libertun C. Neonatal exposure to bisphenol A and reproductive and endocrine alterations resembling the polycystic ovarian syndrome in adult rats. Environ Health Perspect. 2010;118(9):1217–22.
Zhou J, Qu F, Jin Y, Yang DX. The extracts of Pacific oyster (Crassostrea gigas) alleviate ovarian functional disorders of female rats with exposure to bisphenol a through decreasing FSHR expression in ovarian tissues. Afr J Tradit Complement Altern Med. 2014;11(5):1–7.
Li Q, Davila J, Kannan A, Flaws JA, Bagchi MK, Bagchi IC. Chronic exposure to Bisphenol A affects uterine function during early pregnancy in mice. Endocrinology. 2016;157(5):1764–74.
Ziv-Gal A, Craig ZR, Wang W, Flaws JA. Bisphenol A inhibits cultured mouse ovarian follicle growth partially via the aryl hydrocarbon receptor signaling pathway. Reprod Toxicol. 2013;42:58–67.
Forte M, Mita L, Cobellis L, Merafina V, Specchio R, Rossi S, et al. Triclosan and bisphenol A affect decidualization of human endometrial stromal cells. Mol Cell Endocrinol. 2016;422:74–83.
Lopez-Rodriguez D, Franssen D, Sevrin E, Gerard A, Balsat C, Blacher S, et al. Persistent vs transient alteration of folliculogenesis and estrous cycle after neonatal vs adult exposure to bisphenol A. Endocrinology. 2019;160:2558.
Souter I, Smith KW, Dimitriadis I, Ehrlich S, Williams PL, Calafat AM, et al. The association of bisphenol-A urinary concentrations with antral follicle counts and other measures of ovarian reserve in women undergoing infertility treatments. Reprod Toxicol. 2013;42:224–31.
Benjamin S, Masai E, Kamimura N, Takahashi K, Anderson RC, Faisal PA. Phthalates impact human health: epidemiological evidences and plausible mechanism of action. J Hazard Mater. 2017;340:360–83. https://doi.org/10.1016/j.jhazmat.2017.06.036. Epub 2017 Jun 19. PMID: 28800814
Mikula P, Svobodová Z, Smutná M. Phthalates: toxicology and food safety—a review. Czech J Food Sci. 2005;23:217–23. https://doi.org/10.17221/3394-CJFS.
Staples CA, Peterson DR, Parkerton TF, Adams WJ. The environmental fate of phthalate esters: a literature review. Chemosphere. 1997;35:667–749. https://doi.org/10.1016/S0045-6535(97)00195-1.
Jurewicz J, Radwan M, Sobala W, Ligocka D, Radwan P, Bochenek M. Human urinary phthalate metabolites level and main semen parameters, sperm chromatin structure, sperm aneuploidy and reproductive hormones. Reprod Toxicol. 2013;42:232–41. https://doi.org/10.1016/j.reprotox.2013.10.001.
Sharpe RM, Skakkebaek NE. Testicular dysgenesis syndrome: mechanistic insights and potential new downstream effects. Fertil Steril. 2008;89:e33–8. https://doi.org/10.1016/j.fertnstert.2007.12.026.
Xie C, Zhao Y, Gao L, Chen J, Cai D, Zhang Y. Elevated phthalates’ exposure in children with constitutional delay of growth and puberty. Mol Cell Endocrinol. 2015;407:67–73. https://doi.org/10.1016/j.mce.2015.03.006.
Zhu M, Huang C, Ma X, Wu R, Zhu W, Li X. Phthalates promote prostate cancer cell proliferation through activation of ERK5 and p38. Environ Toxicol Pharmacol. 2018;63:29–33. https://doi.org/10.1016/j.etap.2018.08.007.
Patiño-García D, Cruz-Fernandes L, Buñay J, Palomino J, Moreno RD. Reproductive alterations in chronically exposed female mice to environmentally relevant doses of a mixture of phthalates and alkylphenols. Endocrinology. 2018;159(2):1050–61. https://doi.org/10.1210/en.2017-00614. PMID: 29300862
Gallicchio L, Miller S, Greene T, Zacur H, Flaws J. Premature ovarian failure among hairdressers. Hum Reprod. 2009;24:2636–41. https://doi.org/10.1093/humrep/dep252.
Srilanchakon K, Thadsri T, Jantarat C, Thengyai S, Nosoognoen W, Supornsilchai V. Higher phthalate concentrations are associated with precocious puberty in normal weight Thai girls. J Pediatr Endocrinol Metab. 2017;30:1293–8. https://doi.org/10.1515/jpem-2017-0281.
Newbold RR. Impact of environmental endocrine disrupting chemicals on the development of obesity. Hormones. 2010;9:206–17. https://doi.org/10.14310/horm.2002.1271.
Messerlian C, Wylie BJ, Mínguez-Alarcón L, Williams PL, Ford JB, Souter IC. Urinary concentrations of phthalate metabolites and pregnancy loss among women conceiving with medically assisted reproduction. Epidemiology. 2016;27:879–88. https://doi.org/10.1097/EDE.0000000000000525.
Park MA, Hwang KA, Lee HR, Yi BR, Jeung EB, Choi KC. Cell growth of BG-1 ovarian cancer cells is promoted by di-n-butyl phthalate and hexabromocyclododecane via upregulation of the cyclin D and cyclin-dependent kinase-4 genes. Mol Med Rep. 2012 Mar;5(3):761–6. https://doi.org/10.3892/mmr.2011.712.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Marci, R., Buzzaccarini, G., Wenger, J.M., Vitagliano, A. (2023). Introduction to Environmental Pollutants and Human Reproduction. In: Marci, R. (eds) Environment Impact on Reproductive Health. Springer, Cham. https://doi.org/10.1007/978-3-031-36494-5_4
Download citation
DOI: https://doi.org/10.1007/978-3-031-36494-5_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-36493-8
Online ISBN: 978-3-031-36494-5
eBook Packages: MedicineMedicine (R0)