Abstract
A pilot single-stage setup with the reactor volume of 20 m3 was constructed for ammonium removal from the filtrate of thermophilically digested sludge. The setup was operated at temperatures of 20–37 ℃, dissolved oxygen concentrations of 0.1–0.7 mg/L, pH of 5.7–8.5, hydraulic retention time of 12–36 h, and filtrate output of up to 30 m3/day. The efficiency of nitrogen removal was 75–90%, nitrogen load was 0.9–1.1 kg N/(m3·day), and the specific volumetric nitrogen removal capacity of the reactor reached 0.8–1.0 kg N/(m3·day). The sludge retained activity at low pH (5.7) and enhanced nitrite concentration (up to 250 mg/L). A correlation was established between conductivity reduction of the treated liquid and nitrogen removal efficiency, and the formula for calculation of ammonium concentration using the conductivity was proposed.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
- Autotrophic anaerobic ammonium oxidation (Anammox)
- Anammox bacteria
- Filtrate of anaerobic digesters
- Nitrogen removal
- Wastewater
- Wastewater treatment plants
Introduction
At Moscow wastewater treatment plants the sludge is stabilized by anaerobic thermophilic digestion (52–55 ℃) followed by thickening and dewatering of digested sludge. Significant degradation of volatile suspended solids (VSS) under thermophilic conditions results in high levels of ammonium (up to 700 mg N-NH4/L) in liquid phase (filtrate) of digested sludge. This filtrate forms recycle water flow and increases the load of ammonium on the biological stage of wastewater treatment by up to 50%. Increasing ammonium concentrations in the liquid phase of digested sludge cause technological and economic problems.
The traditional nitrification/denitrification technology is inefficient for ammonium removal from the filtrate of industrial anaerobic digesters, since such filtrate has a low biochemical oxygen demand (BOD)/N ratios (0.7–1.7). The technologies based on microbial process of autotrophic anaerobic ammonium oxidation by nitrite are presently being introduced for treatment of wastewater with high concentrations of ammonium and low BOD values [7, P. 9]. These technologies combine two autotrophic processes: partial nitrification, which involves oxidation of half of ammonium to nitrite, and anaerobic ammonium oxidation by nitrite to molecular nitrogen (anammox) [1, 5, P. 8, 9]. The best-known anammox technologies applied at wastewater treatment plants worldwide are SHARON-ANAMMOX, DEMON, Canon, ANITA-Mox, DeAmmon, OLAND, etc. [8, P. 9]. High efficiency of nitrogen removal, the absence of organic carbon demand, low sludge yield, and 2–3 times lower nitrogen removal costs are the advantages of anammox technologies [6, 14, P. 9]. There are two principal schemes of anammox implementation: two-stage systems where processes of nitrification and anammox are spatially separated in different serially connected reactors, and single-stage systems consisting of one reactor where both processes go simultaneously. Single-stage systems are widely used due to their advantages over two-stage systems, particularly their higher potential rates of nitrogen removal [14, P. 9].
Joint Stock Company (JSC) Mosvodokanal is the largest company in Russia servicing Moscow wastewater treatment plants. The JSC Mosvodokanal in collaboration with Winogradsky Institute of Microbiology (The Federal Research Centre “Fundamentals of Biotechnology” of Russian Academy of Sciences) and within the framework of the agreement with Ministry of Education and Science of the Russian Federation has developed the first Russian anammox-based technology for ammonium removal from the filtrate of industrial anaerobic digesters and tested it in pilot-scale setup. The goal of the present work was the development, start-up, and optimisation of a pilot-scale single-stage setup for ammonium removal from the filtrate of digested sludge.
Methods
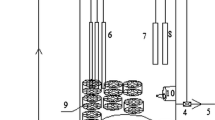
The schematic diagram of the pilot setup is shown in Fig. 1. The filtrate from dewatering centrifuges was passed through the grid (1) for removal of coarse particles and was then collected in the receiving tank (2). From this tank the filtrate was transferred to the 4-m3 primary settler (3), then to the heat exchanger (4), and subsequently to the 20-m3 main bioreactor (5). The setup was designed for carrying out the intended processes of water treatment within a broad range of operating temperatures (from 20 to 37 ℃). The required operating temperature was maintained by the heat exchanger with an electric heater and biologically treated water as a coolant. The bioreactor was a completely stirring-type reactor. The sludge from the secondary settler (8) was also recycled to the reactor. Removal of ammonium and organic matter occurred in the bioreactor. The intended biochemical processes were carried out by suspended flocculated sludge and by the sludge immobilized on a stationary lamellar carrier. The bioreactor contained three blocks of the stationary lamellar carrier, the aeration system providing oxygen for the process, and the large-bubble system of regeneration for removal of excess biomass from the carrier. Air was supplied by a compressor (11). Oxygen concentration in the reactor was maintained at 0.1–0.7 mg/L and monitored using an oxygen sensor.
Hydraulic retention time (HRT) for filtrate was 12–36 h. From the bioreactor the sludge was transferred to a setup for intermediate aeration 2 m3 in volume (6), then to 18-L sludge homogenizer (7), and to the secondary settler (4 m3). Setups for intermediate aeration and homogenizer were required to prevent flotation of the activated sludge in the secondary settler under high nitrogen load. After settling, the treated filtrate was discharged, and return sludge was pumped back into the bioreactor. Some of the sludge (excessive) was removed from the setup.
Concentrations of suspended solids (SS), chemical oxygen demand (COD), BOD, NH4 +, NO2 −, NO3 −, total suspended solids (TSS), and volatile suspended solids (VSS) were measured by common methods [12, P. 9].
Results
The above-described pilot setup with a 20-m3 bioreactor was intended for ammonium removal from the filtrate of dewatering centrifuges of digested sludge from the Lyuberetskie wastewater treatment plant (LWWP), Moscow, Russia. The reactor was inoculated by anammox sludge from the reactor, described earlier [10, 11, P. 9]. During first days of reactor operation filtrate supply was 3–4 m3/d, and then it was gradually increased. The planned filtrate supply rate of 20 m3/day was reached after 70 days of setup operation. Dynamics of nitrogen compounds in the treated filtrate from the pilot setup (Fig. 2), showing a trend of decreasing ammonium and nitrate concentrations, reflects improvements of the technological process. Drastic fluctuations were caused by periodic changes in the operation mode due to adjustment of the systems for aeration, filtrate supply, temperature, etc. The concentrations of ammonium and nitrate were in opposite phases. The general picture indicated the start-up period of the setup.
The setup reached its full capacity, with nitrogen load of 0.9–1.1 kg N/(m3·day) and specific volumetric nitrogen removal capacity of the reactor of 0.8–1.0 kg N/(m3·day) after 12 months of operation. Nitrogen removal was the main target parameter. Nitrogen removal exceeding 70% was considered technologically sufficient (Table 1).
Implementation of the above-described technology resulted in a high efficiency of nitrogen removal and high reliability of the process. Thus, resistance of the activated sludge to elevated nitrite concentration (250 mg/L) was shown. In the single-stage setup, nitrites were formed in the course of the aerobic nitritation process, carried out by ammonium-oxidizing bacteria (AOB) [14, P. 9]. Nitritation is essential for efficient functioning of anammox reactors, since AOB provide anammox bacteria with nitrite, one of the major anammox substrates, which is absent in the digested sludge filtrate. While anammox bacteria have high affinity to both ammonium and nitrite, the anammox process is inhibited by nitrite concentrations over 100 mg/L (by nitrogen) [13, P. 9]. This threshold may be shifted to higher or lower concentrations [2, 3, 15, P. 8, 9] and depends on duration of the inhibitory action of nitrite [4, P. 8]. Recovery of anammox after nitrite removal from the medium indicates reversible inhibition [9, P. 9]. Calculations of the efficiency of nitrogen removal at different nitrite concentrations (Fig. 3) were carried out prior to the reactor reaching its designed capacity, i.e., when it did not reach its maximal output. Since at least 50% of the maximal activity was preserved (within the range from 50 to 90 mg N-NO2/L) at nitrite concentrations up to 175 mg/L, and ~30% of the maximal activity—at 250 mg/L, the technology was highly stable in this respect.
The presence of two extremes in Fig. 3 (at 50–90 and 225–260 mg N-NO2/L) indicates heterogeneity of the anammox bacteria population. It is likely represented by two groups of bacteria with different nitrite optima. The optimum nitrite concentration was within the range of 50–100 mg N-NO2/L for one of them and ~250 mg/L for the other. Pyrosequencing of the 16S rRNA gene fragments revealed predominance of the genus Candidatus “Brocadia” in the activated sludge [10, P. 9].
Inverse correlation was revealed between nitrite concentration and pH, i.e., the medium was acidified at elevated nitrite concentrations. The optimal pH for the setup operation was 7.5–8.3 at nitrite concentrations of 50–100 mg/L. The pH minimum was pH 5.7 at 250 mg/L nitrite, and the maximum was pH 8.3–8.5 at less than 50 mg/L nitrite.
The conductivity of the reactor medium, an important factor in technological monitoring, was also investigated. Good correlation was found between relative decrease in conductivity (%) and efficiency of total nitrogen removal (the correlation coefficient was 0.63) (Fig. 4). This parameter may be used for online express analysis of the efficiency of the setup operation.
Conductivity was found to be associated with ammonium concentrations in the inflowing and treated filtrate (the correlation coefficient was 0.92) (Fig. 5). Using the conductivity values, ammonium concentration may be calculated, which is important for controlling the technology:
NH4 concentration (mg N/L) = C × 0.12−159.5,
where C is conductivity of the reactor medium (µS/cm2).
Conclusions
Thus, the reported single-stage pilot setup carries out the target processes of ammonium removal from the filtrate of dewatering centrifuges of digested sludge. Flocculated free-floating sludge is involved in the oxidation of organic matter, sorption of suspended solids, and partial oxidation of ammonium to nitrite. In the inner layers of the biofilm immobilized on the carrier, autotrophic (anammox) and heterotrophic denitrification occurs. The setup carries out the target processes within the temperature range of 20–37 ℃, oxygen concentrations from 0.1 to 0.7 mg/L, and pH 5.7–8.5. At hydraulic retention time of 12–36 h and filtrate output of 14–30 m3/day, nitrogen load is as high as 0.9–1.1 kg N/(m3·day), and specific volume capacity for nitrogen removal is 0.8–1.0 kg N/(m3·day). The activated sludge was found to be stable at elevated nitrite concentration (250 mg/L) and decreased pH value (5.7). Using the conductivity of the medium for control of the efficiency of nitrogen removal and for calculation of ammonium concentrations in the incoming and outcoming filtrate was confirmed. The data obtained will be used for designing an industrial anammox bioreactor at the Lyuberetskie wastewater treatment plants, with a capacity of 18,000 m3/day and nitrogen removal rate of up to 11 t/day.
References
Blackburne, R., Yuan, Z., Keller, J.: Partial nitrification to nitrite using low dissolved oxygen concentration as the main selection factor. Biodegradation 19(2), 303–312 (2008)
Dapena-Mora, A., Fernandez, I., Campos, J.L., Mosquera-Corral, A., Mendez, R., Jetten, M.S.M.: Evaluation of activity and inhibition effects on anammox process by batch tests based on the nitrogen gas production. Enzyme Microb. Technol. 40, 859–865 (2007)
Egli, K., Fanger, U., Alvarez, P., Siegrist, H., van der Meer, J., Zehnder, A.: Enrichment and characterization of an anammox bacterium from a rotating biological contactor treating ammonium-rich leachate. Arch. Microbiol. 175, 198–207 (2001)
Fux, C., Huang, D., Monti, A., Siegrist, H.: Difficulties in maintaining long-term partial nitritation of ammonium-rich sludge digester liquids in a moving-bed biofilm reactor (MBBR). Water Sci. Technol. 49, 53–60 (2004)
Hellinga, C., Schellen, A., Mulder, J., van Loosdrecht, M.C.M., Heijnen, J.J.: The SHARON process: an innovative method for nitrogen removal from ammonium-rich wastewater. Water Sci. Technol. 37(9), 135–142 (1998)
Jetten, M.S.M., Strous, M., Van de Pas-Schoonen, K.T., Schalk, J., Van Dongen, U.G.J.M., Van de Graaf, A.A., Logemann, S., Muyzer, G., van Loosdrecht, M.C.M., Kuenen, J.G.: The anaerobic oxidation of ammonium. FEMS Microbiol. Rev. 22(5), 421–437 (1999)
Kuenen, J.G.: Anammox bacteria: from discovery to application. Nat. Rev. Microbiol. 6, 320–326 (2008)
Lackner, S., Gilbert, E.M., Vlaeminck, S.E., Joss, A., Horn, H., van Loosdrecht, M.C.M.: Full-scale partial nitritation/anammox experiences—an application survey. Water Res. 55, 292–303 (2014)
Lotti, T., van der Star, W.R.L., Kleerebezem, R., Lubello, C., van Loosdrecht, M.C.M.: The effect of nitrite inhibition on the anammox process. Water Res. 46, 2559–2569 (2012)
Mardanov, A.V., Beletskii, A.V., Kallistova, A.Yu., Kotlyarov, R.Yu., Nikolaev, Yu.A., Kevbrina, M.V., Agarev, A.M., Ravin, N.V., Pimenov, N.V.: Dynamics of the composition of a microbial consortium during start-up of a single-stage constant flow laboratory nitritation/anammox setup. Microbiology. 85(6), 681–692 (2016)
Nikolaev, Yu.A., Kozlov, M.N., Kevbrina, M.V., Dorofeev, A.G., Pimenov, N.V., Kallistova, A.Yu., Grachev, V.A., Kazakova, E.A., Zharkov, A.V., Kuznetsov, B.B., Patutina, E.O., Bumazhkin, B.K.: Candidatus “Jettenia moscovienalis” sp. nov., a new species of bacteria carrying out anaerobic ammonium oxidation. Microbiology. 84(2), 256–262 (2015)
Standard methods for the examination of water and wastewater.: American Public Health Association, American Water Works Association, Water Environment Association. In: Rice, E.W., Baird, R.B., Eaton, A.D., Clesceriand, L.S., Bridgewater, L. (eds.). Washington, D.C. 22nd ed. 1496 p (2012)
Strous, M., Kuenen, J.G., Jetten, M.S.M.: Key physiology of anaerobic ammonium oxidation. Appl. Environ. Microbiol. 65, 3248–3250 (1999)
Van Hulle, S.W.H., Vandeweyer, H.J.P., Meesschaert, B.D., Vanrolleghem, P.A., Dejans, P., Dumoulin, A.: Engineering aspects and practical application of autotrophic nitrogen removal from nitrogen rich streams. Chem. Eng. J. 162, 1–20 (2010)
Xiao, Y., Xiao, Q., Xiang, S.: Modeling of simultaneous partial nitrification, anammox and denitrification process in a single reactor. J. Environ. Anal. Toxicol. 4 (2014). doi:10.4172/2161-0525.1000204
Acknowledgements
Research is carried out with the financial support of the state represented by the Ministry of Education and Science of the Russian Federation. Agreement no. 14.607.21.0018, June 5, 2014. Unique project Identifier: RFMEFI60714X0018.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
This chapter is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
Copyright information
© 2018 The Author(s)
About this paper
Cite this paper
Agarev, A.M. et al. (2018). Development of the First Russian Anammox-Based Technology for Nitrogen Removal from Wastewater. In: Anisimov, K., et al. Proceedings of the Scientific-Practical Conference "Research and Development - 2016". Springer, Cham. https://doi.org/10.1007/978-3-319-62870-7_73
Download citation
DOI: https://doi.org/10.1007/978-3-319-62870-7_73
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-62869-1
Online ISBN: 978-3-319-62870-7
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)