Abstract
After the accident at Fukushima Daiichi Nuclear Power Plant (FNPP) in March 2011, high contamination levels in the environment suggest possible effects of radiation on nonhuman biota. In order to understand the effect on wild animals and plants, field investigations were conducted in the ex-evacuation zone where ambient dose-rate was particularly high. For the purpose of biomonitoring of the radiation effect, coniferous trees have been demonstrated to be suitable indicator organisms because of their high radiosensitivity, which was revealed decades ago by experiments using gamma irradiation facilities. Subsequently, radiosensitive damages in conifers became real after the Chernobyl nuclear power plant (CNPP) accident in 1986, where local coniferous species showed distinct biological damage in the radioactively contaminated areas. This review outlines the results obtained from the past radiation experiments and cases in surrounding forests after the CNPP accident for radiation effect studies of coniferous trees. By referring to them, the author explains the present situation and problems of the investigation on coniferous trees after the FNPP accident.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Following the accident at Fukushima Daiichi Nuclear Power Plant (FNPP) in March 2011, there are concerns about radionuclide contamination in the forests around FNPP. In addition to the distribution and dynamics of radionuclides in the forest environment, much attention has been paid to the biological consequences of radiation emitted from radionuclides on animals and plants that inhabit the forest. To understand the impact on such wild animals and plants, examinations have been carried out in heavily contaminated areas within the ex-evaluation zone where “it is expected that the residents have difficulties in returning for a long time (Ministry of Economy, Trade and Industry)” since 2011 [1]. Some of the examinations were made on coniferous tree as a possible indicator organism of the radiation effect, because this group of plants has been evaluated to have radiosensitive characteristics.
With regard to the radiation effect in coniferous trees, research that takes environmental impacts into consideration has been conducted in the past. These were conducted primarily as irradiation experiments using outdoor γ-ray irradiation facilities (gamma fields). Many gamma fields existed in Europe and the United States (US) from the 1950s to the 1970s, with the first gamma field established in the US Brookhaven National Laboratory in 1948 [2]. In Japan, research was focused on the current gamma field at the Radiation Breeding Division at the National Agriculture and Food Research Organization [3]. In the gamma field of US and Canada, the changes in trees were monitored for ten or more years after placing a cesium (Cs) radiation source within a forest [4, 5]. However, a conventional study on radiation biology that primarily focused on observing the growth and morphological changes in coniferous trees has been almost complete, and since then, the current number of radiation biology researches on coniferous trees in Europe and US has been declining rapidly.
Meanwhile, unlike in Western Europe, US and Japan, radiation impact research on coniferous trees continued to take place beyond the 1980s in USSR. In the accident at the Mayak Production Association in 1957 and the Chernobyl Nuclear Power Plant (CNPP) accident in 1986, there were actual radiation injuries in coniferous trees within the natural environment and many studies investigating the impact of the accidents. In recent years, due to the growing interest in environmental protection, the influence of radiation on the living environment has begun to gain global attention. This has led to efforts on the issue by international authorities that included publication of reports reviewing the investigations into the radiation effect on forest plants in USSR. Beginning with the 1994 report by the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) [6], the accident report of the International Atomic Energy Agency (IAEA) on the CNPP accident provides a detailed review on the impact of radiation on coniferous trees, to demonstrate how organisms in the environment have been affected [7].
This review outlines the results obtained from irradiation experiments in the past and the cases observed in the surrounding forests after the CNPP accident, as radiation impact research of coniferous trees. By referring to these, the author explains the present situation and issues associated with the investigation on coniferous trees following the FNPP accident.
2 Investigations Using Irradiation Facilities on Coniferous Trees
2.1 Radiation Hypersensitivity Observed at the Lethal Dose/Dose-Rate on Coniferous Trees
Higher plant species show great differences in radiation sensitivity, which has been demonstrated by researches in US using gamma fields. Differences in radiosensitivity of various plant species were compared for growth inhibition and mortality after acute radiation exposure [8, 9]. In woody plants, 28 species (12 broad-leaved and 16 coniferous trees) were studied on the lethal dose 2 years after acute irradiation [9]. Although the lethal dose in the broad-leaved trees spanned a wide range between 2 and 20 kR (20 and 200 Gy), the coniferous trees showed distinctively lower lethal doses between 0.8 and 1.6 kR (8 and 16 Gy).
As in acute irradiation experiments, hypersensitivity of coniferous trees to radiation was observed also in chronic irradiation experiments [10, 11]. The mean lethal dose-rate after irradiating six species of coniferous trees for 3 years was 22.9 R/day (229 mGy/day), which was approximately 10% of the mean lethal dose-rate in seven species of broad-leaved trees. Chronic irradiation experiments in the Japanese gamma field have also been conducted on Japanese coniferous trees such as Japanese cedar (Cryptomeria japonica), Japanese cypress (Chamaecyparis obtusa), Japanese red pine (Pinus densiflora) and Japanese black pine (Pinus thunbergii), with the lethal dose-rate between 10 and 20 R/day (100 and 200 mGy/day) having been observed after 2 years of irradiation [12].
Furthermore, coniferous trees exposed to radiation over an extended period of time tend to accumulate radiation injuries, and the lethal dose-rate declines with the irradiation period [4]. In pitch pine (Pinus rigida), as γ-irradiation was extended from 8 to 10 years, the lethal dose-rate decreased from >10.6 R/day (over 106 mGy/day) to 5.3 R/day (53 mGy/day), while the 50% lethal dose-rate decreased from 3.1 R/day (31 mGy/day) to 3.0 R/day (30 mGy/day) [4]. In balsam fir (Abies balsamea), which is thought to be particularly sensitive to radiation, the 50% lethal dose-rate with 11 years of γ-irradiation decreased down to 28.5 mGy/day and tended to decrease even further as the irradiation period was extended [5].
Radiation hypersensitivity seen in coniferous trees corresponds to cellular traits unique to this plant group. Coniferous trees have a wide variety of species (about 500 species) from the temperate zone to the subarctic zone around the world, but what is common to them all is a very large genome size (the amount of DNA per genome) [13], which results in a very large volume of cell nucleus. Therefore, even when exposed to radiation of the same absorbed dose, the energy of radiation absorbed per cell nucleus or one chromosome in coniferous trees is larger than that of a broad-leaved tree with smaller genome size and cell nucleus volume. To that end, Sparrow et al. explained that the susceptibility of coniferous trees to radiation influences at the cellular level is responsible for radiation sensitivity of coniferous trees at the organismal level [8, 9, 14].
2.2 Various Radiation Injuries in Coniferous Trees
In plants, various radiation injuries also occur when they are subjected to chronic irradiation below the lethal dose-rate. The minimum dose-rate at which such radiation injury occurs correlates positively with the lethal dose-rate of the plant species. Accordingly, coniferous trees with the low lethal dose-rate can be injured even at a relatively low exposure dose-rate [15]. In pitch pine, the radial growth of the tree trunk decreased after several years of irradiation at 4 R/day (40 mGy/day) [4], and the length of needles was halved after 10 years of irradiation at a mean dose-rate of 3 R/day (30 mGy/day), while minor shrinkage of needles was observed even at 1.5 R/day (15 mGy/day) of irradiation [4]. A decrease in tree growth was obviously detectable in pitch pine within several years of irradiation at 2 R/day (20 mGy/day) [14].
In particular, the injury that is noticeably observed even at low dose-rates is the growth of the tip of the tree shoot (apical growth), associated with cell division and elongation in apical meristematic tissues, and formation of buds and reproductive organs. The annual growth in height of Japanese larch (Larix kaempferi) begun to be suppressed at 2 R/day (20 mGy/day), and its 50% reduced dose-rate was estimated to be 2.5–4.5 R/day (25–45 mGy/day) [16]. In pitch pine, the higher the dose-rate, the lower the growth of leader shoot, the greater the growth of lateral sprouts, and the lower was the number of lateral buds formed newly [17]. In balsam fir, the amount of shoot elongation and the number of buds formed were significantly decreased by γ-irradiation of 19 mGy/day for 3 years [5]. Furthermore, in Taxus media (Taxus media), the number of buds formed decreased significantly after irradiation at 3.75 R/day (37.5 mGy/day) for 1 year, while irradiation at a higher dose-rate of 12.5 R/day (125 mGy/h) decreased the number of buds formed down to 0.5% of the control [18]. Prior to the decrease in the number of buds formed, changes in apical meristematic tissues were observed to form; specifically necrosis of apical initial cells developed in the meristem at 22 days after the start of irradiation at 3.75 R/day (37.5 mGy/day). In white spruce (Picea glauca), the disappearance of apical initial cells was observed even in the lowest dose-rate of 0.5 R/day (5 mGy/day) after being exposed to a total dose of 52.7 R (527 mGy) from spring through to autumn [19]. In pitch pine, changes in the apical meristem were also observed after 2 months of chronic exposure at 5 R/day (50 mGy/day) [17]. In these injured meristem, regeneration of meristematic tissue was often observed to replace the necrotic apical meristem [17,18,19].
In the case of reproductive organs, the number of mature seeds in pitch pine decreased down to 10% of the control, after 9 years of irradiation at 3.5 R/day (35 mGy/day) [12]. Additionally, around 50% of the pollen aborted at 7 R/day (70 mGy/day), and germination of pollen decreased to 50% of normal at 3–5 R/day (30–50 mGy/day) [20]. Male flowers tended to shorten in length at 0.4 R/day (4 mGy/day), and such change became significant at 3.7 R/day (37 mGy/day) [20].
3 Radiation Injuries to Coniferous Trees Due to the Chernobyl Nuclear Power Plant (CNPP) Accident
In the CNPP accident in the spring of 1986, radioactive plumes contaminated a wide range of surrounding forests rich in subarctic coniferous trees. The results of the examination conducted by the former Soviet Union (USSR) on the radiation effect of forest plants have been summarized in the report by IAEA [7]. It has been shown that radiation injuries, including withering and death, occurred in forest trees contaminated with radionuclides in the Chernobyl exclusion zone (CEZ), particularly in coniferous trees.
The main coniferous trees that constitute forests in this area are Scots pine (Pinus sylvestris) and Norway spruce (Picea abies), both of which showed clear biological injuries. In “lethal zone” (4 km2), which suffered the highest degree of contamination, all of these coniferous trees died and were called “red forest” from the color of the dead trees. In areas with lower level of radiation contamination, the coniferous injuries reduced, and in “sublethal zone” (38 km2), there were some individual tree deaths or deaths of most growth points. Reproductive suppression and morphological changes were observed in the “medium damage zone” (120 km2), while even the “minor damage zone” showed growth, reproduction and morphological disturbances. In contrast, radiation injury to broad-leaved trees such as white birch (Betula platyphylla) that were mixed with these coniferous trees was relatively small, and even in the “red forest” where all the coniferous trees had died, only partial injuries were observed.
Radiation injuries, such as death of coniferous trees, were observed within 2–3 weeks after the CNP accident and progressed from the spring to the summer in that year. For such injuries, the contribution of radiation exposure in the short term in the early stages of the accident was large, and it was thought that the majority was caused by β-particles from radionuclides directly adhering to the trees. According to the dose estimation of radiation exposure of trees, the reproductive capacity in Scots pine was affected by receiving a total dose of 2000 mGy or more at subacute exposure; growth retardation and morphological damage occurred at 1000 mGy or more; cytogenetic damage was estimated to have been detectable at 500 mGy or more; and there were no visible damages until 5 years after the accident at doses under 100 mGy [21]. In Norway spruce, the radiation sensitivity was higher than that of Scots pine, and the absorbed dose to cause needle, bud, and stem malformations was estimated to be 700–1000 mGy.
In contrast, although the number of studies on the longer-term chronic radiation effect is limited, it has been reported that Scots pine planted after the cutting of the dead trees in the “red forest” showed delayed morphological changes several years later, which has led to abnormal branching of the trunk (disappearance of apical dominance) [22]. In the area where such a morphological change has occurred in 50% of individual trees, the radiation dose-rate of trees was estimated to be 0.96 mGy/day [22].
4 Changes in Forests After the Fukushima Daiichi Nuclear Power Plant (FNPP) Accident
Around FNPP, there were a number of warm temperate forests on the land where radionuclides flowed after the accident in March 2011 were deposited. In the forests of this region, coniferous trees such as Japanese red pine (Pinus densiflora), Japanese cedar (Cryptomeria japonica), Japanese cypress (Chamaecyparis obtusa), and Japanese fir (Abies firma) are mixed with broad-leaved trees like Konara oak (Quercus serrata). These coniferous trees have been mainly examined in forests in the ex-evacuation zone [1]. During the first investigation conducted within the ex-evacuation zone of the FNPP accident in November 2011, around 8 months after the accident, external signs of radiation injuries such as yellowing of the leaves of trees and morphological abnormalities were not observed, even in the most contaminated forest around 3 km west of FNPP [1, 23]. This indicates that large-scale radiation injury in coniferous trees due to subacute radiation exposure did not occur after the FNPP accident, unlike after the CNPP accident. However, to investigate the mid- and long-term effect on trees by radiation, examination within the ex-evacuation zone was continued.
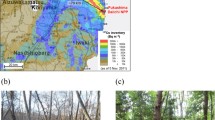
In an examination in January 2015, around 4 years after the accident, the tree morphology in the Japanese fir population naturally occurring in the forest was observed [24]. In each of the three experimental sites within the ex-evacuation zone and one control site that was quite distant, plots of 800–1200 m2 were established, and all young fir trees (height of 40 cm–5 m, around 100–200 trees in each plot) were observed (Fig. 8.1). In the Japanese fir population in the sites with particularly high ambient dose-rate in the ex-evacuation zone, a significant increase in the frequency of morphological changes was observed compared to the population in the control site with low ambient dose-rate (Fig. 8.2). Moreover, it was found that the frequency was higher depending on the ambient dose-rate in each site. Whereas Japanese fir trees usually have one trunk extending vertically, trees with morphological changes were characterized by branching defects caused by deletion of the leader shoot. When the position of branching defect was identified for each individual tree, a significant increase in the frequency of branching defect became apparent in the part that elongated after the accident between 2012 and 2013, compared to before the accident in 2010 (Fig. 8.3). Taking into account that, as shown in past γ-irradiation experiments and the CNPP accident, coniferous trees like Japanese fir have high radiation sensitivity, these results suggest the possibility that radiation contributed to morphological changes in the Japanese fir trees in area near FNPP.
Map showing the observation sites. A red star indicates the location of Fukushima Daiichi Nuclear Power Plant. The base map was modified from the Digital Japan Portal Web Site (Geospatial Information Authority of Japan; GSI). Airborne monitoring results on 2013.11.19; the “Extension site of distribution map of radiation dose, etc.,/Digital Japan” [25] was used as the source of the distribution of radiation dose
Relative frequency of main axis defects in Japanese fir trees from different sites. The defects were counted by the observation of five annual whorls from the top of the trees. The figure in parenthesis indicates ambient dose-rate at each observation site. Bonferroni-corrected p-values are presented using Chi-square tests with df = 1
Relative frequency of deleted leader shoot in the annual whorls of the main axis in Japanese fir trees from different sites. The deletions were counted by the observation of five annual whorls from the top of the trees. Years indicate the time that the whorl branches sprouted out in the spring. Different letters indicate statistically significant differences compared to the whorls of 2010 in each site
Similar changes to coniferous trees have been reported in Japanese red pine as well [26]. As a result of investigating the morphology of young Japanese red pine in eight sites within Fukushima Prefecture (including the ex-evacuation zone) between 2014 and 2016, disappearance of apical dominance (i.e., branching of trunk) was observed as a typical morphological change. The incidence rate of morphological changes correlated with the dose-rate of radiation that the tree population was exposed to, and the morphological changes manifested within 4 years from the start of exposure. These were similar to the morphological changes observed in the Scots pine within the ex-evacuation zone following the CNPP accident [22], which suggested that radiation is the cause of morphological changes.
5 Perspectives for Future Study
The investigations on the coniferous trees within the ex-evacuation zone of the FNPP accident did not show the kind of large-scale radiation injuries observed after the subacute radiation exposure at the initial stages following the CNPP accident. On the other hand, the fact that morphological changes were observed more frequently in shoots of Japanese fir and Japanese red pine near FNPP suggests the influence of chronic exposure. However, it is important to note that the observed morphological changes in coniferous trees are not necessarily radiation-specific, and similar changes may also be caused by environmental and physiological factors. Therefore, in order to clarify the relationship between radionuclide contamination and morphological changes, it is necessary to evaluate accurately the difference in exposed radiation dose of coniferous trees and various environmental factors in each area of investigation, to specifically elucidate the process by which the morphological changes developed.
Additionally, in order to verify the morphological changes in coniferous trees due to radiation exposure, it is important to make comparisons with data from irradiation experiments that used experimental facilities like gamma fields. Existing data from irradiation experiments conducted on coniferous trees indicate that radiation sensitivity differs depending on the coniferous species and depending on the injuries such as lethality, growth inhibition, effects on reproductive function, etc. Therefore, irradiation experiments on the specific coniferous species (Japanese fir and Japanese red pine) should be considered, under conditions similar to their growing environments, to investigate if the same morphological change as observed near FNPP occurs (Fig. 8.4).
To reproduce the radiation exposure situation in the verification experiment at the irradiation facility, it is also important to estimate as accurately as possible the radiation dose that the coniferous trees were actually exposed to in the investigated area. By estimating the radiation dose of the coniferous trees in the investigated area and comparing it with the irradiation dose at the radiation facility that causes morphological changes in the coniferous trees, it will become possible to elucidate the causal relationship between the incidence of morphological changes in the investigated area and radiation exposure. For this reason, it is an important challenge to create a dose evaluation model to reproduce complicated exposures that coniferous tree tissues receive from various radiation sources in the environment and then reconstruct the change in the exposed dose of coniferous trees after the FNPP accident based on actual measurements of the radionuclide concentration in the trees and their environment.
6 Remarks
The contents concerning morphological abnormalities posted here are based on “Morphological defects in native Japanese fir trees around the Fukushima Daiichi Nuclear Power Plant” by Y. Watanabe et al., licensed under Creative Commons.
References
Ministry of Environment Results of measurements after monitoring wild animals and plants. https://www.env.go.jp/jishin/monitoring/results_r-wl.html. Accessed 20 Mar 2018
Sparrow AH (1966) Research uses of the gamma field and related radiation facilities at Brookhaven national laboratory. Radiat Bot 6:377–405
Kawara K (1963) Introduction of a gamma field in Japan. Radiat Bot 3:175–177
Sparrow AH, Schairer LA, Woodwell GM (1965) Tolerance of Pinus rigida trees to a ten-year exposure to chronic gamma irradiation from cobalt-60. Radiat Bot 5:7–22
Dugle JR (1986) Growth and morphology in Balsam fir: effects of gamma radiation. Can J Bot 64:144–192
United Nations Scientific Committee on the Effects of Atomic Radiation (1996) Sources and effects of ionizing radiation; report to the general assembly. United Nations, New York
International Atomic Energy Agency (2006) Environmental consequences of the Chernobyl accident and their remediation: twenty years of experience, report of the Chernobyl Forum Expert Group ‘Environment’. IAEA, Vienna
Sparrow RC, Sparrow AH (1965) Relative radiosensitivities of woody and herbaceous spermatophytes. Science 147:1449–1451
Sparrow AH, Rogers AF, Schwemmer SS (1968) Radiosensitivity studies with woody plants. I. Acute gamma irradiation survival data for 28 species and predictions for 190 species. Radiat Bot 8:149–174
Sparrow AH, Schwemmer SS, Klug EE et al (1970) Radiosensitivity studies with woody plants. II. Survival data for 13 species irradiated chronically for up to 8 years. Radiat Res 44:154–177
Sparrow AH, Schwemmer SS, Klug EE et al (1970) Woody plants: changes in survival in response to long-term (8 years) chronic gamma irradiation. Science 169:1082–1084
Ohba K (1964) Studies on radiosensitivity and induction of somatic mutations in forest trees. Gamma Field Symp 3:111–141
Murray BG (1998) Nuclear DNA amounts in gymnosperms. Ann Bot 82:3–15
Baetcke KP, Sparrow AH, Nauman CH et al (1967) The relationship of DNA content to nuclear and chromosome volumes and to radiosensitivity (LD50). Proc Natl Acad Sci USA 58:533–540
Sparrow AH, Woodwell GM (1962) Prediction of the sensitivity of plants to chronic gamma irradiation. Radiat Bot 2:9–26
Murai M, Ohba K (1966) Study on radiosensitivity of forest trees (III) influence of gamma ray irradiation on the growth of Larix Kaempferi larch and cell division in leaves. J Jpn For Soc 48:62–68
Mergen F, Thielges BA (1966) Effects of chronic exposures to Co60 radiation on Pinus rigida seedlings. Radiat Bot 6:203–210
Miksche JP, Sparrow AH, Rogers AF (1962) The effects of chronic gamma irradiation on the apical meristem and bud formation of Taxus media. Radiat Bot 2:125–129
Cecich RA, Miksche JP (1970) The response of white spruce (Picea glauca (Moench) Voss) shoot apices to exposures of chronic gamma radiation. Radiat Bot 10:457–467
Mergen F, Johansen TS (1963) Effect of ionizing radiation on microsporogenesis in Pinus Rigida mill. Radiat Bot 3:321–331
Arkhipov NP, Kuchma ND, Askbrant S et al (1994) Acute and long-term effects of irradiation on pine (Pinus silvestris) stands post-Chernobyl. Sci Total Environ 157:383–386
Yoschenko VI, Kashparov VA, Melnychuk MD et al (2011) Chronic irradiation of scots pine trees (Pinus Sylvestris) in the Chernobyl exclusion zone: dosimetry and radiobiological effects. Health Phys 101:393–408
Watanabe Y, Ichikawa S, Kubota M et al (2012) Effects of radionuclide contamination on forest trees in the exclusion zone around the Fukushima Daiichi Nuclear Power Plant. In: Proceedings of the international symposium on environmental monitoring and dose estimation of residents after accident of TEPCO’s Fukushima Daiichi Nuclear Power Stations, pp 2–14
Watanabe Y, Ichikawa S, Kubota M et al (2015) Morphological defects in native Japanese fir trees around the Fukushima Daiichi Nuclear Power Plant. Sci Rep 5:13232
Ministry of Education, Culture, Sports, Science and Technology Radiation dose distribution map, http://ramap.jaea.go.jp/map/. Accessed 20 Mar 2018
Yoschenko V, Nanba K, Yoshida S et al (2016) Morphological abnormalities in Japanese red pine (Pinus densiflora) at the territories contaminated as a result of the accident at Fukushima Dai-Ichi Nuclear Power Plant. J Environ Radioact 165:60–67
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2020 The Author(s)
About this chapter
Cite this chapter
Watanabe, Y. (2020). Influence of the FNPP Accident on Coniferous Trees: A Review. In: Fukumoto, M. (eds) Low-Dose Radiation Effects on Animals and Ecosystems. Springer, Singapore. https://doi.org/10.1007/978-981-13-8218-5_8
Download citation
DOI: https://doi.org/10.1007/978-981-13-8218-5_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8217-8
Online ISBN: 978-981-13-8218-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)