Abstract
The ash species Fraxinus platypoda is the dominant canopy tree species at Ooyamazawa riparian forest. I investigated flowering, seed production, germination, seedling survival and growth, and structural measures in F. platypoda. Flowering and seed production demonstrated a clear masting pattern over a 28-year period. The seeds of F. platypoda germinated in most environments, but seedling survival was regulated by microtopographic factors. I suggest that gap formation processes may be vital to the establishment of canopy-height individuals. The overall forest structure and spatial distribution of F. platypoda suggested that all individuals of this species occurring at Ooyamazawa regenerated simultaneously following a large-scale disturbance event 200 years ago. Since this event occurred, saplings of F. platypoda have regenerated in canopy gaps. It is probable that F. platypoda succeeds as a dominant species in riparian forests by regenerating in response to disturbance at multiple scales.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Advanced sapling
- Dioecy
- Disturbance regime
- Flowering
- Germination
- Life history
- Microtopography
- Reproductive strategy
- Seed production
- Seedling
1 Introduction
Native riparian forests are distributed along the Ooyamazawa stream in the Chichibu Mountains of the Kanto region, central Japan. In these forests, Fraxinus platypoda Oliv. (Oleaceae) is one of the dominant canopy tree species, coexisting with Pterocarya rhoifolia and Cercidiphyllum japonicum. The Chichibu Mountains harbor a very complex topography characterized by steep slopes, with tree diversity distributed among the resulting microhabitats. For example, Tsuga sieboldii and Chamaecyparis obtusa forests are distributed along ridges, Fagus crenata and Fagus japonica on mountain slopes, and F. platypoda, P. rhoifolia, and C. japonicum in valleys (Maeda and Yoshioka 1952; Tanaka 1985). The regeneration mechanisms of T. sieboldii, F. crenata, and F. japonica have been extensively studied by Suzuki (1979, 1980, 1981a, b), Nakashizuka and Numata (1982a, b) and Nakashizuka (1983, 1984a, b), and Ohkubo et al. (1988, 1996), respectively. These researchers examined forest regeneration within the context of gap dynamics theory. Gaps are open spaces within the forest canopy layer formed by die back, trunk breakage, and uproot ed trees. In general, gaps occur on a small scale and are not often accompanied by soil disturbance, with the exception of uprooted trees.
On the other hand, disturbance s within riparian zones vary in type, frequency, magnitude, and size compared to canopy gap formation on hillslopes. In steeper mountain regions, valley floor landforms are sculpted by fluvial process es and a variety of mass soil movement processes from tributaries and adjacent hillslopes (Gregory et al. 1991).
The ash species, Fraxinus platypoda, is a late successional species in riparian habitats of cool temperate forest s distributed along the Pacific coast of Japan. This species is distributed from Tochigi Prefecture in the north to Miyazaki Prefecture in the south (Fig. 2.1). Kisanuki et al. (1992) and Ann and Oshima (1996) examined regeneration mechanisms within a gap dynamics framework in mixed forests of F. Platypoda and P. rhoifolia. However, the regeneration of F. platypoda may also be related to large-scale natural disturbances such as debris flow s and landslide s in the riparian zone . In this chapter, I present long-term research related to the life history and regeneration process of F. platypoda with respect to natural disturbances within the riparian zone.
Distribution of F. platypoda. Modified after Kawahara et al. (2009)
2 Study Species
Fraxinus platypoda is a deciduous canopy species that can reach up to 40 m in height and 150 cm in diameter at breast height (DBH, 130 cm) (Fig. 2.2). This species is well adapted to stream disturbance s that vary in frequency and size, and it dominates forests in riparian zones (Sakio 1997).
Branching occurs above the trunk of the tree. Most individuals have a single trunk and rarely exhibit sprouts, similar to species such as P. rhoifolia and C. japonicum. Leaves are impari-pinnate compound, consisting of 7–9 leaflets, and leaf length is about 25–35 cm and decussate-opposite (Fig. 2.3). The base of the petiole markedly bulges and holds the stem. Open hairs occur along the middle vein on the back of the leaf, but others are hairless. The apical leaflet is oblong-ellipsoid oblanceolate in shape, and is 8–20 cm in length and 3–7 cm in width, with a small petiole of 1–2 cm in length. The side leaflets lack a petiole, and the base is wedged with fine serrations. Twigs are thick, gray-brown, and hairless, and many are oval lenticel. The pith is thick. Branches grow rapidly in early spring and stop growing in June (Sakio 1993). The root system is concentrated in the shallow part of the ground surface. Fraxinus platypoda has medium to large-diameter straight roots and horizontal root s and is classified as a deep root type. Fine roots are dense, but root hairs are rare (Karizumi 1979). The species is easily uprooted by scouring running water. In saplings, sedimentation by soil and sand easily produces adventitious root s (Sakio 2002).
3 Reproductive Traits
3.1 Flower
Sexual expression of F. platypoda is not obvious. The species exhibits two morphological types: one with male flower s and the other with hermaphrodite flower s (Fig. 2.4). Both flowers lack a perianth. Male flowers have one stamen with a pair of anther s, while hermaphrodite flowers have one pistil with a pair of anthers. Whether the male portion of hermaphrodite flowers of F. platypoda is functional is unknown. However, sexual expression of F. platypoda may be functionally considered to be androdioecy , as the pollen of hermaphrodite flowers exhibits germination ability. Because the breeding characteristics of F. platypoda are not clearly understood, I treat the sexual expression of F. platypoda as dioecy , i.e., female trees and male trees, in this chapter.
The timing of flowering in F. platypoda differs depending on altitude but, typically, occurs in mid-April (700 m a.s.l) to mid-May (1500 m a.s.l.). In the Ooyamazawa riparian forest (1500 m a.s.l.), flowering occurs from the beginning of May to mid-May. However, the exact timing fluctuates annually.
Flowering data collected over 28 years have demonstrated clear fluctuations of flower values for F. platypoda in the core research plot (Fig. 2.5). The numbers of female and male trees in the core plot (0.54 ha) were 26 and 20, respectively. The rank of flowering for female and male trees was scored from 1 to 5 by observation using binoculars. The average flowering rank of all individuals was 3.22 for females and 4.11 for males; values were significantly higher in males. The coefficient of variation (CV) was 0.48 for females and 0.28 for males; values were significantly higher in females. In this forest, although the flowering interval was 2–3 years, the interval changed over the 28 years. The flowering of the two sexes exhibited clear synchronization until 2002, but synchronization ceased thereafter. After 2002, more males have flowered every year, while females have retained a distinct interval. The change in flowering fluctuation after 2002 may have been driven by external factors, such as climate warming.
3.2 Seed Production
After flowering, fruit (samara ) of F. platypoda continues to grow, causing abortion . The pericarp finishes growing at the end of August, at which point seeds begin to grow. Oven-dried matured fruit of F. platypoda weighed 144 ± 24 mg, and the dry weight of seeds was 80 ± 17 mg (Fig. 2.6; Sakio et al. 2002). Seeds mature in mid-October and are dispersed by wind and stream water in November (Fig. 2.7). Many mature seeds occur in mast year s, but a large number of seeds are empty or insect-damaged during non-mast years.
Fruits of F. platypoda in the stream in autumn (Sakio 2008)
Clear fluctuations in seed production and flowering of F. platypoda occurred over the 28 years (Fig. 2.8). Because seed production was strongly positively correlated with the extent of flowering, the former is presumed to be regulated by the latter (Fig. 2.9).
4 Germination
The seeds of F. platypoda germinate from the end of June to mid-July. Current-year seedling s are found on litter , gravel, mineral soil, and fallen logs, except after non-mast years. As long as the light environment is not very strong, current-year seedlings of F. platypoda only have cotyledon s (Fig. 2.10), and the true leaf does not expand during the germination year. The germination site of F. platypoda is not strongly restricted by the soil and light environment, unlike C. japonicum, whose germination sites are limited. F. platypoda does not exhibit seed dormancy . The seeds of F. platypoda produced in autumn germinate in the early summer of the following year and do not germinate thereafter.
5 Seedling Survival
The seeds of F. platypoda are randomly dispersed. Therefore, the distribution of current-year seedling s of F. platypoda is not affected by microenvironments such as soil and light conditions. However, the distribution pattern of seedlings of F. platypoda changes with the growth of seedlings, shifting from random or uniform distribution to aggregated throughout growth (Fig. 2.11). Small-sized seedlings (height < 20 cm) tend to be distributed around the active channel (Figs. 2.11 and 2.12), while the distribution of larger seedlings (20 cm ≤ height < 1 m) is more closely related to the microtopography than to canopy gap s. These larger seedlings tend to be aggregated in abandoned channel s. On hillslopes where the forest floor vegetation is dense, F. platypoda seedlings disappear after several years due to the effects of shade. The mean longevity of F. platypoda seedlings in various environments is 1.19 ± 0.58 years (Sakio et al. 2002). On the other hand, seedlings near active channels exhibit long life spans and high density due to the absence of forest floor vegetation (Fig. 2.11). However, seedling communities can be destroyed by flooding, often caused by large typhoon s. Gravel deposit s formed during flooding events can serve as new seedling establishment sites. When the seedling community stabilizes due to channel fluctuations, it continues to grow into a large-sized seedling community (20 cm ≤ height).
In other words, the distribution of F. platypoda seedlings is regulated by the microtopographic variation in forest floor vegetation . In riparian forests, the light environment depends not only on the presence of canopy gap s but also on gaps in the forest floor vegetation due to stream disturbance . The dynamics of F. platypoda seedlings are thought to be strongly influenced by the latter.
6 Seedling Growth
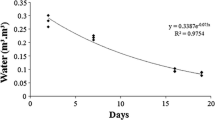
Large-sized seedlings (1 m ≤ height) that have established on stable sites exhibit variation in growth rates depending on the light environment . New shoots of F. platypoda begin to elongate rapidly in early spring and stop growing in June (Sakio 1993). Seedling s under canopy gap s grow faster than seedlings under the canopy (Fig. 2.13) and have more leaves. In many forests, canopy gaps appear to be necessary for the growth of canopy trees (Suzuki 1980, 1981a; Nakashizuka and Numata 1982a, b; Nakashizuka 1983, 1984a). Therefore, even in riparian forests, gap formation via the death of canopy trees may be necessary for seedlings of F. platypoda to grow into the canopy.
Seasonal changes of current shoot lengths of F. platypoda saplings in canopy gaps and under the canopy. Modified after Sakio (1993)
7 Forest Structure and Spatial Distribution
In the 4.71-ha study plot within the Ooyamazawa riparian forest, we observed 418 F. platypoda individuals out of a total of 2214 trees (4 cm ≤ DBH) (Sakio et al. 2002). Canopy tree s, subcanopy trees (10 cm ≤ DBH), and the shrub layer (DBH < 10 cm) accounted for 304, 59, and 55 individuals, respectively. The mean DBH of F. platypoda canopy trees was 56.9 ± 19.0 cm, with a maximum of 140.5 cm (Sakio et al. 2002). The DBH distribution of F. platypoda was continuous from saplings to large canopy trees (Fig. 2.14). F. platypoda exhibited two peaks in DBH distribution: one formed by small trees (DBH < 10 cm) and the other formed by the 40-cm DBH class. These data suggest that F. platypoda maintains sapling banks. The peak of the 40-cm class suggests synchronous regeneration caused by a large-scale disturbance . The relative density of dominant canopy tree s of F. platypoda is high and does not exhibit distinct fluctuations along the stream.
DBH distribution of F. platypoda individuals in the Ooyamazawa riparian plot (4.71 ha). Only individuals over 4 cm in DBH were measured. Modified after Sakio (2008)
In the core plot (60 × 90 m), one peak of F. platypoda individuals occurred within the 40–60-cm DBH class, similar to the pattern observed in the 4.71-ha plot (Sakio 1997). In addition, increment core s of all F. platypoda individuals larger than 4-cm DBH were obtained using an increment borer in November 1998, in the core plot. The age distribution of F. platypoda individuals was continuous from saplings to older canopy-aged trees (Sakio 1997). Figure 2.15 presents the spatial distribution of tree age for F. platypoda in the core plot. The age of most individuals was aggregated around 200 years, but several young aggregated groups also occurred (e.g., small patches A, C, and F). These results suggest that a large-scale disturbance occurred around 200 years ago in the Ooyamazawa riparian forest and that F. platypoda regenerated simultaneously. Since that time, F. platypoda advanced sapling s have regenerated under canopy gaps. Thus, F. platypoda is likely to become an overwhelmingly dominant species within the basin because it can regenerate within sites of large-scale disturbance as well as within small gaps.
8 Conclusion
Fraxinus platypoda produces a large number of seeds once every few years, thus forming young advanced sapling communities within various microtopographic habitats. In particular, advanced sapling communities are formed in gravel deposit s along mountain streams. These sapling communities continue to be regenerated after repeated destruction by mountain stream disturbances and subsequent regeneration on new gravel deposits. After large disturbances, F. platypoda regenerates within all river basins and also fills canopy gap s by advanced saplings when small gaps are formed. Thus, F. platypoda succeeds as a dominant species in riparian forest s by regenerating in response to various scales of disturbances throughout its life history .
References
Ann SW, Oshima Y (1996) Structure and regeneration of Fraxinus spaethiana - Pterocarya rhoifolia forests in unstable valleys in the Chichibu Mountains, central Japan. Ecol Res 11(3):363–370
Gregory SV, Swanson FJ, Mckee WA, Cummins KW (1991) An ecosystem perspective of riparian zones: focus on links between land and water. BioScience 41:540–551
Karizumi N (1979) Illustration of tree roots. Seibundo Shinkousha (in Japanese)
Kawahara T, Watanabe S, Matsui T, Takahashi M (2009) Silvics of Japan Bunpuzu. In: The publishing association of Silvics of Japan (ed) Silvics of Japan I. Japan Forestry Investigation Committee, Tokyo, pp 725–760
Kisanuki H, Kaji M, Suzuki K (1992) Structure and regeneration process of ash (Fraxinus spaethiana Ling.) stands in Chichibu Mountains. Bull Tokyo Univ For 88:15–32 (in Japanese with English summary)
Maeda T, Yoshioka J (1952) Studies on the vegetation of Chichibu Mountain forest. (2) The plant communities of the temperate mountain zone. Bull Tokyo Univ For 42:129–150+3pls (in Japanese with English summary)
Nakashizuka T (1983) Regeneration process of climax beech (Fagus crenata Blume) forests. III. Structure and development processes of sapling populations in different aged gaps. Jpn J Ecol 33:409–418
Nakashizuka T (1984a) Regeneration process of climax beech (Fagus crenata Blume) forests IV. Gap formation. Jpn J Ecol 34:75–85
Nakashizuka T (1984b) Regeneration process of climax beech (Fagus crenata Blume) forests. V. Population dynamics of beech in a regeneration process. Jpn J Ecol 34:411–419
Nakashizuka T, Numata M (1982a) Regeneration process of climax beech forests I. Structure of a beech forest with the undergrowth of Sasa. Jpn J Ecol 32:57–67
Nakashizuka T, Numata M (1982b) Regeneration process of climax beech forests II. Structure of a forest under the influences of grazing. Jpn J Ecol 32:473–482
Ohkubo T, Kaji M, Hamaya T (1988) Structure of primary Japanese beech (Fagus japonica Maxim.) forest in the Chichibu Mountains, central Japan, with special reference to regeneration processes. Ecol Res 3:101–116
Ohkubo T, Tanimoto T, Peters R (1996) Response of Japanese beech (Fagus japonica Maxim.) sprouts to canopy gaps. Vegetatio 124:1–8
Sakio H (1993) Sapling growth patterns in Fraxinus platypoda and Pterocarya rhoifolia. Jpn J Ecol 43(3):163–167 (in Japanese with English summary)
Sakio H (1997) Effects of natural disturbance on the regeneration of riparian forests in a Chichibu Mountains, central Japan. Plant Ecol 132:181–195
Sakio H (2002) Survival and growth of planted trees in relation to debris movement on gravel deposit of a check dam. J Jpn For Soc 84(1):26–32 (in Japanese with English summary)
Sakio H (2008) Coexistence mechanisms of three riparian species in the upper basin with respect to their life histories, ecophysiology, and disturbance regimes. In: Sakio H, Tamura T (eds) Ecology of riparian forests in Japan: disturbance, life history and regeneration. Springer, pp 75–90
Sakio H, Kubo M, Shimano K, Ohno K (2002) Coexistence of three canopy tree species in a riparian forest in the Chichibu Mountains, central Japan. Folia Geobot 37:45–61
Suzuki E (1979) Regeneration of Tsuga sieboldii forest. I. Dynamics of development of a mature stand revealed by stem analysis data. Jpn J Ecol 29:375–386 (in Japanese with English Synopsis)
Suzuki E (1980) Regeneration of Tsuga sieboldii forest. II. Two cases of regenerations occurred about 260 and 50 years ago. Jpn J Ecol 30:333–346 (in Japanese with English Synopsis)
Suzuki E (1981a) Regeneration of Tsuga sieboldii forest. III. Regeneration under a canopy gap with low density of conifer seedlings and a method for estimating the time of gap formation. Jpn J Ecol 31:307–316 (in Japanese with English synopsis)
Suzuki E (1981b) Regeneration of Tsuga sieboldii forest. IV. Temperate conifer forests of Kubotani-Yama and its adjacent area. Jpn J Ecol 31:421–434 (in Japanese with English Summary)
Tanaka N (1985) Patchy structure of a temperate mixed forest and topography in the Chichibu Mountains, Japan. Jpn J Ecol 35:153–167
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2020 The Author(s)
About this chapter
Cite this chapter
Sakio, H. (2020). Fraxinus platypoda . In: Sakio, H. (eds) Long-Term Ecosystem Changes in Riparian Forests. Ecological Research Monographs. Springer, Singapore. https://doi.org/10.1007/978-981-15-3009-8_2
Download citation
DOI: https://doi.org/10.1007/978-981-15-3009-8_2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-3008-1
Online ISBN: 978-981-15-3009-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)