Abstract
Adipocytes are the most abundant cells within the adipose tissue and are the cell type responsible for the tissue dynamic metabolic and endocrine activity. Under energy surplus conditions, the adipocyte is able to suffer hypertrophy in order to accommodate energy in form of lipids. Simultaneously, new adipocytes are differentiated through a complex and specific process, known as adipogenesis. While this process seems clear for white adipocytes in white adipose tissue, brown adipocytes and brown adipose tissue have distinct characteristics and function. Brown adipocytes are not related with fat accumulation but rather with thermogenesis, a process defined by a rapidly oxidization of lipids in order to produce heat. Additionally, a class of beige adipocytes, which are inducible thermogenic adipocytes originating from white adipose tissue and phenotypically distinct from both, have been described though how these are originated and which are the main functions are still matters of discussion. Interestingly, the induction of thermogenesis seems to improve insulin resistance, adiposity and hyperlipidemia. Thus, inducing the browning of white adipocytes to beige adipocytes is thought to be promising to improve the common metabolic disorders, such as obesity or metabolic syndrome. This chapter focuses on the specific signaling and regulatory control of adipocyte functions, particularly adipogenesis and adipocyte browning. Emerging insights of these processes are herein discussed, as promising therapeutic targets for obesity and other common metabolic disorders.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
15.1 Introduction
The adipose tissue (AT) is a metabolically active organ that acts as the main energy repository in the human body and as an endocrine organ able to synthesize several biologically active molecules that regulate metabolic homeostasis. The AT fulfils several functions, which may vary among fat depots due to its size, distribution, and heterogeneity according to the molecular, morphological, and metabolic profiles [1]. In humans, there are two main types of AT, white AT (WAT) and brown AT (BAT), with relevant differences in morphology and function. In the human body, BAT is mainly located in the supraclavicular, periadrenal, and paravertebral regions [2] and its relative proportion decreases since birth into adulthood [3]. Although BAT is also able to store energy in form of fat, the main activity of the tissue lies in heat production or thermogenesis [4]. In contrast, WAT contribution for thermogenesis is nearly irrelevant, but presents much broader physiological functions. Despite WAT being widely distributed in the human body, 80% of total body fat is located subcutaneously, with the main fat depots in the abdominal and femoral-gluteal regions. The remaining 20% are located around the visceral organs, predominantly in proximity to abdominal visceral AT, such as mesenteric and omental, and around the kidney in the retroperitoneal region [5]. By involving organs and infiltrating tissues, the WAT not only offers mechanical protection, but also plays an important role in the regulation of the body temperature, acting as a thermal insulator [4, 6]. Furthermore, WAT accomplishes multiple other functions, such as immune, endocrine, and regenerative [7, 8].
The AT is mainly constituted by adipocytes. Adipocytes are very unique cells due to their morphology and functions. Adipocytes are responsible for the dynamic activity of the AT, conferring metabolic and endocrine activity to the tissue. The process of cellular differentiation into adipocytes, or adipogenesis, is a specific and finely regulated process which differs in WAT and BAT. While white adipocytes are the “classic” adipocytes, whose main function is to accumulate energy in form of lipids in large lipid droplets, brown adipocytes are rather distinct. Brown adipocytes are described as being smaller in comparison to white adipocytes, with relatively abundant cytoplasm, numerous small lipid droplets of different sizes and numerous mitochondria that produce heat by fatty acids oxidation [4]. More recently, a third type of adipocytes was identified, being characterized as an intermediate morphology between brown and white adipocytes. These adipocytes also have a thermogenic activity and a high number of mitochondria, thus have been termed as beige or brite adipocytes. In this chapter, the specific signaling and regulatory control of adipocytes will be discussed, particularly white and brown adipogenesis, thermogenesis, and the interchange between the two adipocyte phenotypes with focus on WAT browning and beige adipocytes development.
15.2 White Adipocyte Signaling
15.2.1 White Adipogenesis
Adipose tissue mass accumulation is associated with adipocyte cell hypertrophy and increased number of white adipocytes. Whenever there is an energy surplus, excess energy is predominantly accumulated in the form of lipids in white adipocytes, which hypertrophy as the lipid droplets size increase. This WAT buffering activity is suggested as an adaptive response to energy excess, which protects other tissues from lipotoxicity [9]. The maintenance of WAT homeostasis includes simultaneous cell hyperplasia, a phenomena by which adipocyte precursors, or preadipocytes, proliferate and differentiate into mature adipocytes [10]. Thus, this process denominated adipogenesis culminates with the formation of new adipocyte cells and consists of a two phase process (Fig. 15.1). The first step towards the differentiation into the adipocyte lineage consists in the generation of preadipocytes from mesenchymal stem cells (MSCs). The second phase includes the terminal differentiation of preadipocytes into functional mature adipocytes, including morphologic alterations and the formation of lipid droplets.
Schematic illustration of white adipogenesis. White adipogenesis is described as two-step process, starting with the proliferation and commitment of white preadipocytes and culminating with the terminal differentiation to mature white adipocytes. The differentiation process starts with the expression of two CCAAT/enhancer-binding proteins (C/EBP), C/EBPB and C/EBPD, which directly induce the expression of C/EBPA and peroxisome proliferator-activated receptor gamma (PPARG), the most important transcriptional regulators of adipogenesis. Then, C/EBPA and PPARG directly induce self-expression in a positive feedback mechanism, activating several downstream adipocyte-specific genes
As mature adipocytes are not able to proliferate in vivo, the regeneration and hyperplasia of the WAT is thought to be dependent on the proliferative capacity of a pool of precursor cells, the preadipocytes [11]. In general, the term “preadipocyte” is widely used to describe a progenitor cell that is responsible for the formation of mature adipocytes [12]. A crucial property of preadipocytes is the ability to proliferate and differentiate into mature adipocytes in order to maintain the AT homeostasis throughout the lifespan of a living organism [13]. These cells are also characterized as a committed cell population destined to proliferate and differentiate only into the adipose-lineage, although morphologically undistinguished from its progenitors [14, 15]. Preadipocytes are thought to arise from MSCs and more specifically from human adipose-derived stem cells (ADSCs). In fact, ADSCs were first identified in 2002 as a subpopulation of multipotent self-renewing cells isolated from WAT that are morphologically and phenotypically similar to the MSCs [16]. Then, ADSCs were isolated, cultured, and termed human multipotent adipose-derived stem cells (hMADSCs) [17]. As hMADSCs maintain the capacity to enter the adipose lineage and to differentiate into cells that present characteristics highly similar to native human adipocytes, these are considered as a faithful model to study human AT physiology [18,19,20]. One of the major focus in adipocyte biology research concerns the identification of distinct cellular intermediates between ASCs and fully functional adipocytes, although there is a lack of specific biomarkers to identify and isolate these cells. However, preadipocytes isolated from different AT present different characteristics in terms of gene expression profiles, proliferation, differentiation, and consequently signaling pathways [21]. Furthermore, adipocytes from different fat depots also present different functional properties, contributions towards energy homeostasis and thus different behaviors during metabolic disease scenarios [22, 23]. However, the molecular mechanisms underlying these AT regional-dependent differences remain unknown.
The second and last phase of adipogenesis is the terminal differentiation, where preadipocytes acquire a phenotype of mature adipocytes. Mature white adipocytes are cells specialized in the synthesis, storage, and mobilization of lipids, which are accumulated in the lipid droplet. This lipid droplet occupies the majority of the adipocyte cytoplasm and consequently the nucleus is found at the periphery of the cell [24]. The terminal differentiation consists of a cascade of transcriptional events which culminates in the formation of mature adipocytes. It is thought that the differentiation process starts with the expression of two CCAAT/enhancer-binding proteins (C/EBP), C/EBPB and C/EBPD, which directly induce the expression of C/EBPA and peroxisome proliferator-activated receptor gamma (PPARG), the most important transcriptional regulators of adipogenesis [25, 26]. Then, C/EBPA and PPARG directly induce its own expression in a positive feedback mechanism [27] and activate several downstream adipocyte-specific genes that are necessary for adipocyte function, including fatty acid binding protein/adipose protein 2 (FABP/aP2), insulin receptor, glucose transporters (GLUTs), acetyl-coA carboxylase, fatty acid synthase, and glycerol-3-phosphate dehydrogenase (GPDH) [28]. Although the factors that control proliferation, commitment, and differentiation of preadipocytes are still poorly characterized, some important regulatory pathways were already identified (Fig. 15.2) and will be discussed on the subsequent topics.

Abbreviations ACTRII—activin type II receptor; C/EBPA—CCAAT/enhancer-binding protein alpha; C/EBPB—CCAAT/enhancer-binding protein beta; C/EBPD—CCAAT/enhancer-binding protein delta; DLK1—delta-like 1 homolog; ERK—extracellular signal-regulated kinase; FGF-1—fibroblast growth factor 1; FGF1R—fibroblast growth factor 1 receptor; FGF-2—fibroblast growth factor 2; FZD—frizzled receptor; HH—hedgehog; LRP5/6—low-density lipoprotein-receptor-related protein 5 and 6 co-receptors; PPARG—peroxisome proliferator-activated receptor gamma; PTC—patched receptor; Smo—smoothened protein; TACE—tumor necrosis alpha converting enzyme; TGF-B—transforming growth factor beta; TGFBR—transforming growth factor beta receptor; TNF-A—tumor necrosis factor alpha; Wnt—wingless-type MMTV integration site; B-Cat—Beta-catenin
Schematic illustration of white adipogenesis regulation. Several factors regulate adipogenesis, leading to preadipocytes proliferation and inhibiting terminal differentiation. Upon ceasing the proliferative stimuli, preadipocytes commit to the terminal differentiation process. Arrows indicate activation or stimulation in the direction of arrowheads. Crossbars indicate suppression or inhibition.
15.2.2 White Adipogenesis Regulation
15.2.2.1 Peroxisome Proliferator-Activated Receptor Gamma (PPARG)
Inducing the expression of PPARG, the major transcriptional regulator of adipogenesis, is sufficient to induce terminal adipocyte differentiation. In fact, it is thought that it is not possible to induce preadipocytes terminal differentiation in the absence of PPARG [29]. Moreover, all essential signaling pathways in adipogenesis seems to converge on the regulation of PPARG expression or activity [30]. There are two isoforms of PPARG (PPARG1 and PPARG2) that are generated by alternative splicing and both are induced during adipogenesis [31]. The adipogenic activity of both PPARG isoforms differs and PPARG2 was found to be more efficient in promoting terminal differentiation. Both PPARG1 and PPARG2 were observed to induce adipocyte differentiation in Pparg knockout fibroblasts, although at higher efficiency for PPARG2 [32]. Additionally, it was demonstrated that 3T3-L1 cells, a cell line originally established from primary murine embryonic fibroblasts able to differentiate into adipocytes, would undergo adipocyte differentiation in the presence of exogenous PPARG2 but not PPARG1 despite the inhibition of Pparg1 and Pparg2 genes by zin-finger proteins [33]. However, studies in animal knockout models yielded contradictory results, which adds complexity and discussion to the topic. For an instance, PPARG2 knockout mice were shown to have decreased AT size and impaired adipogenesis in one study [34], while other authors observed normal AT size and adipocyte morphology in these mice although insulin resistance was reported [35]. Thus, while PPARG2 is important for adipogenesis in vivo, is hypothesized not to be absolutely required when PPARG1 is normally expressed. Additionally, PPARG is essential for the maintenance of adipocytes differentiated state. Inhibition of PPARG in mature 3T3-L1 adipocytes led to de-differentiation with loss of lipid accumulation and reduced expression of adipocyte specific markers [36]. Moreover, in vivo inducible knockout of Pparg led to apoptosis in differentiated adipocytes and stimulated new adipocytes generation [37]. These studies highlight the complexity in PPARG signaling for adipocytes differentiation and physiology.
15.2.2.2 CCAAT/Enhancer-Binding Protein (C/EBP) Family
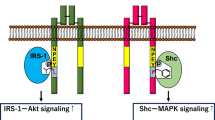
The C/EBP family is constituted by important regulators of adipocyte differentiation. Indeed, the C/EBP family participates in a signaling cascade where C/EBPB and C/EBPD induce the expression of C/EBPA and thus terminal differentiation of adipocytes. Although C/EBPB and C/EBPD can compensate each other in case of loss or inhibition, there is evidence that both factors are not absolutely needed for adipogenesis. A study with knockout mice for C/EBPB or C/EBPD shown that lack of these factors reduces AT size. In addition, double-knockout for C/EBPB and C/EBPD led to a greater reduction of the AT mass. However, the levels of C/ebpa and Pparg mRNA in adipocytes from double-knockout mice remain normal, which raised the hypothesis that absence of C/EBPB or C/EBPD could result only in abnormal lipogenesis and not adipogenesis inhibition [38]. These findings point out that C/EBPB and C/EBPD may not be essential for adipogenesis in vivo as other signaling pathways can produce similar effects. On the other hand, C/EBPA is known to have a much more important role in adipogenesis. For instance, inducing C/EBPA in vitro is sufficient to trigger differentiation of preadipocytes into mature adipocytes [39]. Moreover, mice where the C/ebpa locus was replaced by C/ebpb present reduced AT size [40], while knockout mice for C/ebpa depict an almost absence of WAT [39]. Besides, the expression of C/EBPA in differentiated adipocytes is also important for insulin sensitivity [41]. In this study, it is proposed that C/EBPA triggers the expression of GLUT4 and insulin-dependent glucose transport. In the absence of C/EBPA, GLUT4 is not expressed and cells are rendered insensitive to insulin. Nonetheless, C/EBPA needs the presence of PPARG to efficiently promote adipogenesis since the expression of C/EBPA in knockout fibroblasts for Pparg cannot induce adipocyte differentiation [42]. In addition, C/EBPB needs PPARG to induce the expression of C/EBPA and start the terminal differentiation pathway [43]. Hence, C/EBPs family members, with especial focus in C/EBPA, are indeed important but PPARG expression is crucial for adipogenesis.
15.2.2.3 Fibroblasts Growth Factors (FGF)
One of the most important signaling pathways regulating adipogenesis concerns the autocrine/paracrine role of fibroblast growth factors (FGF). Undifferentiated hMADSCs with reduced FGF-2 secretion were observed to proliferate more slowly [44]. On the other hand, hMADSCs supplemented with FGF-2 would recover and proliferate much faster illustrating that this signaling pathway is essential for proliferation in these cells. FGF-2 has a high affinity for FGF type 1 receptor (FGF1R), which activates extracellular signal-regulated kinase (ERK) 1/2 pathway, leading to cellular proliferation [45]. In fact, hMADSCs treated with a specific FGF1R inhibitor presented a reduced differentiation potential [44, 46]. These findings suggested that FGF-2 is secreted by hMADSCs in order to regulate their proliferative potential through FGF1R/ERK signaling pathway. Additionally, FGF-2 plays a role in preadipocytes differentiation. FGF-2 effects were reported to be dose-dependent, where FGF-2 can function as either a positive or negative adipogenic factor [47]. FGF-2 at concentrations lower than 2 ng/ml promoted differentiation of hMADSCs, while concentrations higher than 10 ng/ml inhibited the differentiation. These effects are also mediated by the FGF1R/ERK signaling pathway, where high concentrations of FGF-2 are able to sustain ERK phosphorylation and lead PPARG phosphorylation, consequently blocking terminal differentiation. Furthermore, FGF-2 levels in WAT of diet induced obese mice were observed to be lower than those from normal diet mice, indicating that FGF-2 expression levels could have a reverse correlation with the fat tissues size. Thus, FGF-2 was hypothesized to play a protective role in obesity and metabolic disease. These findings are further supported by another study conducted in AT of morbidly obese patients who underwent bariatric surgery or lean controls who underwent cholecystectomy [48]. ASCs were obtained from WAT of these individuals and the worse metabolic scenarios were accompanied by a reduced ASCs proliferation rate and decreased FGF-2 secretion. Together, these results suggest that FGF-2 and WAT hyperplasia play a protective role against metabolic syndrome. However, not only FGF-2 but also FGF-1 plays a role in adipogenesis [46]. In fact, FGF-1 seems to be involved in the regulation of FGF-2. FGF-1 treated hMADSCs were reported to have reduced FGF-2 levels by 80%. Moreover, FGF-2 knockout hMADSCs presented decreased proliferation and increased adipogenic genes expression [49]. Thus, while FGF-2 seems to be essential for preadipocytes proliferation, FGF-1 seems to downregulate its secretion, promoting differentiation. For instance, obese individuals also present increased expression of FGF-1 in WAT, which supports the hypothesis of a protective role for FGF-1/2 [50]. Interestingly, in obese individuals were subjected to weigh loss FGF-1 levels were reported to remain high, which may constitute a permanent alteration due to obesity. Nevertheless, further studies are needed to test that hypothesis.
15.2.2.4 Activin A and Transforming Growth Factor Beta (TGF-B) Family
Activin A is a member of the transforming growth factor beta (TGF-B) family and is also secreted by undifferentiated hMADSCs. In fact, activin A was proposed as a biomarker of undifferentiated hMADSCs [51]. Similarly to FGF-2, activin A secretion promotes hMADSCs proliferation and blocks differentiation in an autocrine/paracrine fashion. Activin A effects are mediated via activin type II receptor, which activates Smad2 in order to increase cellular proliferation and inhibit C/EBPB [52]. Thus, while activin A secretion is increased, hMADSCs will proliferate but will not differentiate whereas activin A inhibition leads to hMADSCs differentiation. Interestingly, increased levels of activin A were also found in the WAT of obese individuals [51]. When expressed in high levels, activin A was also reported to act as a profibrotic agent, stimulating the transformation of preadipocytes into myofibroblasts [53, 54]. In fact, AT fibrosis in the context of human obesity limits adipocyte hypertrophy, with beneficial effects on systemic metabolism [55]. Therefore, activin A acts together with FGF-1/2 to constitute a complex and finely regulated signaling pathway in adipogenesis. However, the mechanisms that regulate the increased secretion of FGF-2 and activin A by preadipocytes and subsequent inhibition with terminal differentiation still need to be clarified. There is some evidence that WAT microenvironment is the main responsible for this modulation and that not only preadipocytes and adipocytes regulate adipogenesis. For instance, macrophages isolated from the AT of obese individuals are reported to produce factors that stimulated the expression of FGF-2 and activin A in hMADSCs [51]. Indeed, tumor necrosis factor alpha (TNF-A) is one of the factors secreted by macrophages reported to increase FGF-2 and activin A expression in hMADSCs [20]. Obesity and AT mass accumulation are linked with inflammation and macrophages infiltration [56]. Thus, macrophages are thought to be essential for adipogenesis and WAT homeostasis.
Besides activin A, other members of the TGF-B family are reported to regulate adipogenesis. In fact, TGF-B itself is mostly inhibitory [57]. Studies using 3T3 cell lines (fibroblast cell line) showed that TGF-B increases preadipocyte proliferation, while inhibiting differentiation through Smad3 signaling [58]. However, other studies reported that this inhibitory effect is restricted to the first 40 h due to TFG-B type I and II receptors downregulation during differentiation [58, 59]. The activation of the Smad3 pathway is similar to Smad2, decreasing the expression of PPARG and C/EBPA due to the physical inhibition of C/EBPB and C/EBPD [60]. Besides, Smad3 signaling also involves up-regulation of Wnt signaling pathway factors [61], which will be addressed in further detail in the next section.
Bone morphogenetic proteins (BMPs), are another subtype of growth factor members of the TGF-B family, which also regulate ASCs adipogenesis in an autocrine and dose-dependent manner. For instance, BMP4 is probably the most notorious member of the BMP family in terms of proadipogenic effects [57]. BMP4 activates Smad1/5/8 and positively regulates PPARG expression, whereas BMP4 inhibition leads to reduced lipid droplets size [62]. Moreover, BMP4 has a role in preadipocytes proliferation. While in high doses significantly reduces cell proliferation, in low doses reduces the number of apoptotic cells and increases proliferation [63]. Interestingly, BMP4 promotes differentiation of brown preadipocytes into white adipocytes, reducing the expression of brown-specific proteins. In addition, obese individuals with type 2 diabetes mellitus present higher levels of BMP4 [64]. These studies highlight a possible role for BMP in adipocytes physiology and disease mechanisms associated with diabetes and obesity, but further studies are needed to unveil the mechanisms controlled by this pathway.
15.2.2.5 Wingless-Type MMTV Integration Site (Wnt) Family
Wingless-type MMTV integration site (Wnt) family members are highly conserved signaling glycoproteins with autocrine/paracrine action, known to for regulating tissue homeostasis and remodeling [25]. Wnts exhibit two distinct signaling pathways, denominated “canonical” and “noncanonical”. The canonical pathway is linked with the transcriptional factor Beta-catenin. In sum, Wnts binds to frizzled receptors (FZD) and low-density lipoprotein-receptor-related protein-5 or protein-6 co-receptors (LRP5/6) blocking the ubiquitination and proteasomal degradation of Beta-catenin, which in turn is hypophosphorylated and translocated to the nucleus to bind lymphoid-enhancer-binding factor/T-cell-specific transcription factor (LEF/TCF) and induce the expression of Wnt target genes [65]. The Wnt canonical signaling pathway has an antiadipogenic activity. In support of this hypothesis, are the repeated observations showing that Wnt pathway blocking leads to spontaneous adipocyte differentiation [66,67,68]. Furthermore, LRP5/6 activation inhibits terminal differentiation, whereas LRP5/6 inactivation exerts the opposite effect [69,70,71]. In fact, Wnt1 represses terminal differentiation through downregulation of PPARG and C/EBPA [66]. In addition, Wnt10b, previously known as Wnt12, is also described as an important antiadipogenic factor and an adipogenic switch in adipogenesis. Wnt10b is another member of the Wnt family whose gene is clustered with Wnt. Wnt10b is highly expressed in preadipocytes, which also stabilizes Beta-catenin and inhibits terminal differentiation. In addition, Wnt10b inhibition also promoted preadipocyte differentiation [72]. Thus, Wnt canonical signaling pathway is hypothesized to be an important molecular switch regulating adipogenesis, which is suppressed until the pathway is no longer activated. In fact, PPARG and Beta-catenin are mutual antagonists. The activation of PPARG decreases Beta-catenin levels due to its degradation through proteasomal complexes [73]. Therefore, once preadipocytes receive the trigger to initiate the PPARG positive feedback mechanism, become committed to terminal differentiation. On the other hand, the noncanonical pathway is related with intracellular Ca2+ release, which activates the phosphatase calcineurin and the Ca2+-sensitive kinases Ca2+/calmodulin-dependent kinase II (CAMKII) and protein kinase C [74]. Contrastingly, the noncanonical pathway can either promote adipogenesis through antagonism of the canonical pathway or inhibit adipogenesis in a similar fashion to the canonical pathway. For instance, Wnt5b increases during adipogenesis and promotes terminal differentiation through inhibition of Beta-catenin [75]. However, Wnt5a is reported to inhibit terminal differentiation by activating Beta-catenin and suppressing the activity of PPARG, thus inducing terminal differentiation, via a pathway unrelated to Ca2+ [76]. Altogether, these findings illustrate the complexity of the signaling intrinsic to adipogenesis and further studies are needed.
15.2.2.6 Delta-Like 1 Homolog (DLK1/Pref-1)
Delta-like 1 homolog (DLK1), also known as preadipocyte factor 1 (Pref-1), is a transmembrane glycoprotein that can be cleaved by tumor necrosis alpha converting enzyme (TACE), originating a biologically active soluble form [77, 78]. DLK1 is highly expressed by proliferating preadipocytes and is suppressed during differentiation [79]. In fact, several studies reported that high DLK1 levels in preadipocytes inhibits differentiation [80,81,82] while reduced levels enhances differentiation [83]. More recently, DLK1 knockout mice were shown to have enhanced preadipocyte proliferation, which was associated with a reduced membrane-bound form of DLK1 whereas a soluble form of DLK1 had no effect on preadipocyte proliferation but in the terminal differentiation process [84]. Thus, it was hypothesized that the membrane form and soluble form of DLK1 are likely to have distinct functions and targets [85]. Although DLK1 is considered essential for adipogenic regulation, these results remain controversial since DLK1 interaction molecules remain to be identified.
15.2.2.7 Hedgehog (Hh) Pathway
Hedgehog (Hh) signaling pathway also plays a role in adipogenesis. Hh binds to the Patched receptor (Ptc), which cease the suppressor effect on smoothened protein (Smo) [86, 87]. Smo activates a signaling cascade that results in the stabilization of the transcription factors Gli1 and Gli2, inducing the transcription of Hh target genes [88]. Moreover, Hh signaling pathway seems to interact with the ERK pathway [89] thus suggesting an association with the aforementioned pathways. In fact, Hh basal levels are needed for human MSCs proliferation [90]. Additionally, Smo inhibition by cyclopamine, thus inhibiting the Hh signaling, decreased human MSC proliferation and led to cell cycle arrest, which was associated with a decrease in cyclin A expression and consequently an increase in the active form of retinoblastoma protein (pRb) expression. Conversely, the ability to differentiate human MSCs was not affected, although another study observed that Hh decreases during the differentiation step and that Smo activation impaired preadipocytes terminal differentiation through downregulation of PPARG and C/EBPA [91]. More recently, Hh was demonstrated to inhibit not only adipocyte differentiation but also lipogenesis in adipocytes in vitro, which in turn improved the metabolic condition of diet-induced obese mice [92].
15.2.2.8 Zinc-Finger Proteins
Zinc-finger proteins are crucial molecules involved in adipogenesis regulation. Kruppel-like factors (KLF) family members are zinc-finger proteins, known for playing numerous roles in the regulation of apoptosis, proliferation, and differentiation [93]. In fact, several members of the KLF family are described as adipogenic regulators. KLF15 is known to promote differentiation and the expression of adipocyte-related genes in preadipocytes, such as GLUT4 [94, 95]. KLF4 stimulates the expression of C/EBPB [96], while KLF5 is induced during terminal differentiation by C/EBPB and C/EBPD, which in turn induces the expression of PPARG [97]. Like KLF5, KLF9 binds directly to the PPARG promoter besides also activating PPARG indirectly by binding to C/EBPA [98]. In addition, KLF6 promotes adipogenesis by inhibiting DLK1 [99]. However, some KLFs depict antiadipogenic effects, such as KLF2 that suppresses PPARG expression [100, 101]. Thus, a shift in KLFs expression during terminal differentiation is hypothesized to occur, with KLF2 downregulation that is replaced by the other proadipogenic family members [97]. Similarly to KLFs, GATA zinc fingers found in the GATA-family transcription factors are also known to modulate adipogenesis. GATA2 and GATA3 are expressed in preadipocytes and both inhibit PPARG and C/EBPs expression. In addition, GATA2 and GATA3 expression decreases during terminal differentiation [102, 103]. There is evidence that GATA3 expression is induced by the Wnt/Beta-catenin pathway [104], however the detailed mechanisms underlying this pathway regulation in preadipocytes are still unknown.
15.3 Brown Adipocyte Signaling
15.3.1 Brown Adipogenesis
Brown adipocytes are the main cellular constituent of BAT. Brown adipocytes are morphologically distinct from white adipocytes, as besides depicting a “brown” color that derives from the large number of mitochondria present in the cell cytoplasm, also present a high number of small lipid droplets. The size of brown adipocytes lipid droplets allows to increase the droplet surface area, which was hypothesized to promote the high rate of metabolite exchange with the mitochondria that is necessary for the thermogenic activity [105]. Brown adipocytes arise from brown preadipocytes, although the pathways underlying brown preadipocytes differentiation are not fully understood. In dedicated BAT depots, brown preadipocytes are known to originate from cells residing in the dermomyotome and express specific transcription factors, including paired-box protein 7 (PAX7), engrailed 1 (EN1), and myogenic factor 5 (MYF5) [106]. Yet, different brown adipocytes subpopulations were described in different BAT depots, which despite not expressing all these factors are still able to differentiate into mature brown adipocytes. Indeed, both brown and white adipocytes from different fat depots were shown to exhibit different gene expression profiles and functional characteristics [107].
In a similar process as depicted by white adipocytes, brown adipocytes can expand in size through lipid accumulation leading to hypertrophy or increase in cell number leading to BAT hyperplasia. However, the main stimulus for brown adipocyte expansion is not energy surplus as for white adipocytes but is instead regulated by the needs to maintain body temperature. In thermoneutral conditions sympathetic stimuli are reduced, thus decreasing brown adipocytes thermogenic activity and increasing lipid accumulation. Several lipid droplets are then combined into a single lipid droplet, leading the brown adipocyte into a hypertrophic state [107]. On the other hand, in a cold environment, thermogenesis is activated and both brown adipocytes cells and BAT sizes decrease. In conditions of persistent cold environment exposure, brown adipogenesis is induced [108, 109]. Therefore, brown adipogenesis and thermogenic activity are highly dependent on the cold stimulus.
15.3.1.1 Brown Adipogenesis Regulation
In general, brown and white adipogenesis share the same signaling cascade, highlighting PPARG and C/EBP family members’ role in adipocytes and lipids metabolism. However, some brown adipocyte-specific transcription factors have been identified (Fig. 15.3). Positive regulatory domain containing protein 16 (PRDM16) is described as a transcriptional factor that acts as the main switch of brown adipogenesis. PRDM16 is not only part of a transcriptional complex with C/EBPB and PPARG [110, 111], but has also the ability to induce uncoupling protein 1 (UCP1) and peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1A) expression, resulting in the consequent thermogenic phenotype of brown adipocytes [112, 113]. Moreover, genetic deletion of euchromatic histone lysine methyltransferase 1 (EHMT1), an essential component of PRDM16 complex, decreases brown adipocytes terminal differentiation [114]. Conversely, PRDM16 deletion in brown preadipocytes was found not to affect brown terminal differentiation, which suggests the existence of compensatory pathways [115]. These pathways could be mediated by other PRDM family members, which can induce brown adipogenesis in vivo. One of these members is PRDM3, a PRDM family member that presents a high homology with PRDM16 and forms a complex with EHMT1 and C/EBPB to induce PPARG expression [116]. Thus, EHMT1 is highlighted as a crucial enzymatic regulator of brown adipogenesis. Additionally, early B-cell factor 2 (EBF2) is a transcription factor essential for brown adipogenesis. EBF2 is highly and specifically expressed in brown preadipocytes and adipocytes [117, 118]. EBF2 suppresses the expression of several transcription factors, ensuring preadipocytes commitment to the brown adipocyte terminal differentiation [119]. Zinc-finger protein 516 (ZFP516) expression is also reported to induce PRDM16 and brown adipogenesis [120, 121].

Abbreviations: C/EBPB—CCAAT/enhancer-binding protein beta; cAMP—cyclic adenosine monophosphate; EBF2—early B-cell factor 2; EN1—engrailed 1; IRF4—interferon regulatory factor 4; MYF5—myogenic factor 5; PAX7—paired-box protein 7; PGC1A—proliferator-activated receptor gamma coactivator 1 alpha; PKA—protein kinase A; PPARG—peroxisome proliferator-activated receptor gamma; PRDM16—Positive regulatory domain containing protein 16; UCP1—uncoupling protein 1; ZFP516—zinc-finger protein 516
Schematic illustration of brown adipogenesis regulation and thermogenesis. Brown adipogenesis is triggered with the expression of PRDM16, leading to the thermogenic phenotype and the expression of UCP1.
15.3.2 Thermogenesis
Brown adipocytes are able to rapidly exhibit a high oxidative respiration and substrate oxidation capacity due to the high thermogenic potential and UCP1 expression, a protein that acts as a respiration uncoupler. UCP1 is only found in significant amounts in the mitochondria of thermogenic adipocytes and is described as identical to mitochondrial ADP/ATP carriers [122]. The mechanism of thermogenesis was first proposed by Mitchell and later developed by Nicholls [123]. Briefly, mitochondrial respiratory chain complexes I, III, and IV act as proton pumps, generating a proton gradient where protons are transported from the mitochondria matrix to the intermembrane space. ATP-synthase consume the protons in order to produce ATP from the phosphorylation of ADP, in an endothermic reaction. However, in the presence of an uncoupler protein such as UCP1, which is an inner mitochondrial membrane protein that acts as a proton channel, protons re-entry the mitochondrial matrix from the intermembrane space, collapsing the proton gradient and activating the respiratory chain to compensate the decreased membrane potential. Since the ATP-synthase is no longer able to consume protons in order to produce ATP from ADP, the energy derived from oxidation is then dissipated as heat.
Brown adipocytes are known to produce heat in a process denominated non-shivering thermogenesis. In general, thermogenesis occurs by stimulation of the sympathetic nervous system in response to cold exposure. Whenever there are no requirements to produce extra heat, brown adipocytes remain quiescent and UCP1 is inhibited by purine nucleotides [124]. In fact, inactive brown adipocytes are morphologically similar to white adipocytes, but still preserving the specific genetic and metabolic identity [125]. On the other hand, upon cold stress, norepinephrine is released activating the beta-3 adrenergic receptors and increasing cyclic adenosine monophosphate (cAMP), which results in cAMP-driven protein kinase A (PKA) signaling activation. This signaling pathway culminates in lipolysis, the process where triacylglycerol stored in lipid droplets is hydrolyzed in free fatty acids and glycerol, and increased expression of PGC1A and UCP1 [126, 127]. PGC1A has a crucial role in thermogenesis regulation in brown adipocytes, although interestingly the knockout of PGC1A in brown preadipocytes does not affect brown adipogenesis [128, 129]. PGC1A is highly expressed upon cold stress and PRDM16 induction by cAMP signaling, which results in thermogenic genes activation, such as UCP1 [113]. In addition, cAMP signaling also activates interferon regulatory factor 4 (IRF4), which interacts with PGC1A to form a complex and induces the expression of UCP1 [130]. However, some proteins were identified as inhibitors of PGC1A expression or activity. Receptor-interacting protein 140 (RIP140), together with liver X receptor (LXR) are able to suppress the effects of PGC1A [131, 132]. Steroid receptor co-activator 2 (SRC2) and twist-related protein 1 (TWIST1) also suppress PGC1A effects [133, 134].
Conversely, recent studies have shown that as long as exogenous free fatty acids are available these may directly increase UCP1 expression, thus brown adipocytes might not be fully dependent of brown adipocytes lipolysis [127, 135, 136]. In addition, brown adipocytes are able to perform lipogenesis, where free fatty acids are synthesized from circulating glucose [137], besides the ability to also use acyl-carnitines to sustain thermogenesis [138]. Interestingly, upon cold exposure, brown adipocytes depict both catabolic and anabolic reactions by performing thermogenesis and lipogenesis. Due to this paradoxical activity, it is hypothesized that lipogenesis may have other metabolic implications since several intermediates may also act as signaling agents [107, 139].
15.4 White Adipose Tissue Browning and Beige Adipocytes
15.4.1 Beige Adipocytes Development
Studies with 18F-fluodeoxyglucose PET-CT imaging allowed to identify several metabolic and thermogenic active adipose depots in the abdominal and subcutaneous regions in adult human individuals. These studies led to the conclusion that WAT was not completely white [140,141,142]. Indeed, a second type of UCP1-expressing and thermogenic adipocytes were found in WAT. These adipocytes were named brite (brown-like in white) or beige adipocytes. The morphology of beige adipocytes is somewhat intermediary between white and brown adipocytes and can be found within WAT depots mainly upon cold exposure [106]. Thus, when the formation of beige adipocytes is induced, the phenomenon of WAT browning can be observed. Interestingly, each WAT depot has a different susceptibility to browning. While subcutaneous WAT seems to possess a high capacity to undergo a browning process upon cold exposure, the same does not occur in visceral WAT [143]. Even when WAT browning is artificially induced, visceral WAT is not able to reach the browning ability of subcutaneous WAT, which further highlights the molecular differences between fat depots.
The origin of beige adipocytes could be explained by two different theories, although not mutually exclusive (Fig. 15.4). The first line of thought hypothesized that beige adipocytes arise from a precursor cell pool through beige adipogenesis [144]. Beige adipocytes are reported to originate from a preadipocyte population expressing platelet-derived growth factor receptor A (PDGFRA) and stem cells antigen 1 (SCA1) or from smooth muscle-like precursors expressing myosin heavy chain 11 (MYH11) [106]. On the other hand, another theory defends that beige adipocytes are originated in conditions of thermogenic needs from pre-existing mature adipocytes that undergo a trans-differentiation process, known as adipocyte browning [145, 146]. The expression of PRDM16 in preadipocytes upon cold exposure triggers the beige-linked genes expression and beige adipocytes development while suppressing WAT-selective genes [112]. In fact, both hypotheses are possible, but the regulation of the mechanisms leading to each specific route are still unclear. In addition, beige adipocytes were shown to share some activity and differentiation regulation with brown adipocytes, although whether there are beige-specific transcription factors remains to be elucidated. In support of both theories, beige adipocyte differentiation can be induced in experimental conditions using two different approaches. The first method consists in animal models exposure to severely low temperatures (4–6 °C), which is thought to be the closest approach to physiological conditions. Low temperature exposure induces UCP1-expressing beige adipocytes in WAT either by beige adipogenesis [147] or through browning of mature white adipocytes [145]. The second method consists in using beta-3 adrenergic agonists to stimulate white adipocytes, which mimics the effects of cold exposure and induces the process of adipocyte browning [148]. Interestingly, treatment with beta-3 adrenergic agonists only functions in mature white adipocytes in vitro as inhibition of this pathway in vivo does not prevents the browning induced by cold stimulus, which suggests that browning induction may be dependent on multiple signaling pathways. Indeed, several other conditions were reported to induce beige adipocytes development, such as physical exercise, cancer cachexia, and peripheral tissue injury [149, 150]. Thus, beige adipocytes development seems to be stimulated not only as a thermogenic need, but also to contribute to energy homeostasis.

Abbreviations MYH11—myosin heavy chain 11; A—platelet-derived growth factor receptor A; PRDM16—PR domain containing protein 16; SCA1—stem cells antigen 1; UCP1—uncoupling protein 1
Schematic illustration of beige adipocytes differentiation. Upon cold exposure, beige adipocytes differentiate from beige preadipocytes or through transdifferentiation of mature white adipocytes. Beige adipocytes express PRDM16 and present a brown phenotype with heat production through thermogenesis. However when the cold stimulus ceases, beige adipocytes are able to return to a white adipocyte phenotype.
A distinctive characteristic of beige adipocytes is that the thermogenic ability is inducible and reversible depending on the environmental conditions, as when the cold stress ceases, beige adipocytes are slowly replaced by white adipocytes both in vitro and in vivo [108, 151]. These findings suggest that beige adipocytes are only maintained during the presence of the stimuli and upon its withdrawal, these revert to the white adipocytes’ phenotype. This short-term maintenance and beige-to-white adipocyte conversion or “adipocyte whitening” was attributed to the loss of mitochondrial biogenesis and activity upon the withdrawal of external stimuli [149]. It was hypothesized that adipocyte whitening is linked with mitophagy, a selective form of autophagy responsible for mitochondria degradation. The blockade of the mitophagy pathway prevents adipocyte whitening even upon external stimuli removal and thus leads to the prolongation of the thermogenic activity of beige adipocytes. Moreover, this thermogenic activity was associated with higher energy expenditure and protection against diet-induced obesity [151]. As autophagy pathways are dysregulated in AT of obese and diabetic patients [152] and adipocyte whitening seems to be faster in obese subjects [151], detailed understanding of these pathways could be highly relevant for the development of new pharmacological targets for obesity treatment.
15.4.2 Unconventional Thermogenesis of Beige Adipocytes
UCP1 has been described as the main thermogenic protein in AT thermogenesis. However, it was reported that UCP1 knockout mice exposed to cold still exhibited higher respiration rates in WAT [153]. Moreover, chronic treatment with beta-3 adrenergic receptor agonists in the same animal model also increased the respiration of WAT [154]. Therefore, this implies the existence of UCP1-independent thermogenic pathways. In fact, a specific UCP1-independent thermogenic pathway in beige adipocytes was noticed [155]. This novel and unconventional pathway involves ATP-dependent Ca2+ cycling by sarco/endoplasmatic reticulum Ca2+-ATPase 2b (SERCA2b) and ryanodine receptor 2 (RyR2). This specific thermogenic pathway is only possible in beige adipocytes due to the high capacity of ATP generation by glycolysis and tricarboxylic acid metabolism. These findings also led the authors to state that this novel pathway could be a potential pharmacological target to improve metabolic health in the elderly population, known to possess a low number of UCP1-positive adipocytes.
15.5 Conclusions and Future Perspectives
White, brown, and beige adipocyte differentiation and function are regulated by unique and complex signaling processes. Although some pathways were already identified, the search for factors that regulate adipogenesis just reached the light of the day. While the C/EBP family and PPARG are the main regulators of both white and brown adipogenesis, other transcription factors and signaling proteins that regulate in a direct way and/or upstream/downstream the terminal differentiation signaling cascade are still missing in the big signaling puzzle. In this sense, the new technology of CRISPR-Cas9 may be revolutionary for genetic studies and further extend the knowledge on key adipocyte regulators. Moreover, emerging evidence also highlights the role of miRNAs signaling and epigenetic regulation during adipogenesis and metabolic dysregulation scenarios. Compelling data concerning adipocyte-selective miRNAs that modulate adipocyte proliferation and differentiation [156, 157] point out a considerable new trend in adipocyte biology research. Additionally, the high cellular heterogeneity among adipocytes from different or even within fat depots makes it imperative to characterize their origin and distinct signaling pathways. Nonetheless, WAT browning and inducible thermogenesis is a compelling treatment approach for metabolic diseases such as obesity and type 2 diabetes. A better understanding of the pathways concerning beige adipogenesis and adipocyte browning may point out to promising novel drugs. Given the increasing prevalence and the high health and economic impact of obesity and obesity related disorders, new insights concerning beige adipogenesis and adipocyte browning must be set as a priority in the years to come.
Abbreviations
- ACTRII:
-
Activin type II receptor
- ADSC:
-
Adipose-derived stem cell
- AT:
-
Adipose tissue
- BAT:
-
Brown adipose tissue
- C/EBPA:
-
CCAAT/enhancer-binding protein alpha
- C/EBPB:
-
CCAAT/enhancer-binding protein beta
- C/EBPD:
-
CCAAT/enhancer-binding protein delta
- CAMKII:
-
Ca2+/calmodulin-dependent kinase II
- cAMP:
-
Cyclic adenosine monophosphate
- DLK1:
-
Delta-like 1 homolog
- EBF2:
-
Early B-cell factor 2
- EHMT1:
-
Euchromatic histone lysine methyltransferase 1
- EN1:
-
Engrailed 1
- ERK:
-
Extracellular signal-regulated kinase
- FABP/aP2:
-
Fatty acid binding protein/adipose protein 2
- FGF:
-
Fibroblast growth factor
- FGF1R:
-
FGF type 1 receptor
- FZD:
-
Frizzled receptor
- GLUT:
-
Glucose transporter
- GPDH:
-
Glycerol-3-phosphate dehydrogenase
- Hh:
-
Hedgehog
- hMADSC:
-
Human multipotent adipose-derived stem cell
- IRF4:
-
Interferon regulatory factor 4
- KLF:
-
Kruppel-like factors
- LEF/TCF:
-
Lymphoid-enhancer-binding factor/T-cell-specific transcription factor
- LRP5/6:
-
Low-density lipoprotein-receptor-related protein 5 and 6 co-receptors
- LXR:
-
Liver X receptor
- MSC:
-
Mesenchymal stem cell
- MYF5:
-
Myogenic factor 5
- MYH11:
-
Myosin heavy chain 11
- PAX7:
-
Paired-box protein 7
- PDGFRA:
-
Platelet-derived growth factor receptor A
- PGC1A:
-
Proliferator-activated receptor gamma coactivator 1-alpha
- PKA:
-
Protein kinase A
- PPARG:
-
Peroxisome proliferator-activated receptor gamma
- pRb:
-
Retinoblastoma protein
- PRDM16:
-
Positive regulatory domain containing protein 16
- Pref-1:
-
Preadipocyte factor 1
- Ptc:
-
Patched receptor
- RIP140:
-
Receptor-interacting protein 140
- RyR2:
-
Ryanodine receptor 2
- SCA1:
-
Stem cells antigen 1
- SERCA2b:
-
Sarco/endoplastmatic reticulum Ca2+-ATPase 2b
- Smo:
-
Smoothened protein
- SRC2:
-
Steroid receptor co-activator 2
- TACE:
-
Tumor necrosis alpha converting enzyme
- TGF:
-
Transforming growth factor
- TNF-A:
-
Tumor necrosis factor alpha
- TWIST1:
-
Twist-related protein 1
- UCP:
-
Uncoupling protein
- WAT:
-
White adipose tissue
- Wnt:
-
Wingless-type MMTV integration site
- ZFP516:
-
Zinc-finger protein 516
References
Ibrahim MM (2010) Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev 11(1):11–18. https://doi.org/10.1111/j.1467-789X.2009.00623.x
Cristancho AG, Lazar MA (2011) Forming functional fat: a growing understanding of adipocyte differentiation. Nat Rev Mol Cell Biol 12(11):722. https://doi.org/10.1038/nrm3198
Sacks H, Symonds ME (2013) Anatomical locations of human brown adipose tissue: functional relevance and implications in obesity and type 2 diabetes. Diabetes 62(6):1783–1790. https://doi.org/10.2337/db12-1430
Saely CH, Geiger K, Drexel H (2012) Brown versus white adipose tissue: a mini-review. Gerontology 58(1):15–23. https://doi.org/10.1159/000321319
Tsiloulis T, Watt MJ (2015) Exercise and the regulation of adipose tissue metabolism. Progr Mol Biol Transl Sci 135:175–201. https://doi.org/10.1016/bs.pmbts.2015.06.016
Fonseca-Alaniz MH, Takada J, Alonso-Vale MI, Lima FB (2007) Adipose tissue as an endocrine organ: from theory to practice. Jornal de Pediatria (Rio de Janeiro) 83(5 Suppl):S192–S203. https://doi.org/10.2223/JPED.1709
Coelho M, Oliveira T, Fernandes R (2013) Biochemistry of adipose tissue: an endocrine organ. Arch Med Sci 9(2):191–200. https://doi.org/10.5114/aoms.2013.33181
Thomou T, Tchkonia T, Kirkland JL (2010) Cellular and molecular basis of functional differences among fat depots. In: Adipose tissue in health and disease, pp 21–47. https://doi.org/10.1002/9783527629527.ch2
Chavey C, Lagarrigue S, Annicotte J, Fajas L (2013) Emerging roles of cell cycle regulators in adipocyte metabolism. In: Bastard J-P, Fève B (eds) Physiology and physiopathology of adipose tissue. Springer, Paris, pp 17–25. https://doi.org/10.1007/978-2-8178-0343-2_2
Nishimura S, Manabe I, Nagasaki M, Hosoya Y, Yamashita H, Fujita H, Ohsugi M, Tobe K, Kadowaki T, Nagai R, Sugiura S (2007) Adipogenesis in obesity requires close interplay between differentiating adipocytes, stromal cells, and blood vessels. Diabetes 56(6):1517–1526. https://doi.org/10.2337/db06-1749
Spalding KL, Arner E, Westermark PO, Bernard S, Buchholz BA, Bergmann O, Blomqvist L, Hoffstedt J, Näslund E, Britton T (2008) Dynamics of fat cell turnover in humans. Nature 453(7196):783. https://doi.org/10.1038/nature06902
Zuk P (2013) Adipose-derived stem cells in tissue regeneration: a review. ISRN Stem Cells 2013. https://doi.org/10.1155/2013/713959
Cawthorn WP, Scheller EL, MacDougald OA (2012) Adipose tissue stem cells meet preadipocyte commitment: going back to the future. J Lipid Res 53(2):227–246. https://doi.org/10.1194/jlr.R021089
Rosen ED, Spiegelman BM (2014) What we talk about when we talk about fat. Cell 156(1–2):20–44. https://doi.org/10.1016/j.cell.2013.12.012
Sarantopoulos CN, Banyard DA, Ziegler ME, Sun B, Shaterian A, Widgerow AD (2018) Elucidating the preadipocyte and its role in adipocyte formation: a comprehensive review. Stem Cell Rev Rep 14(1):27–42. https://doi.org/10.1007/s12015-017-9774-9
Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13(12):4279–4295
Rodriguez A-M, Pisani D, Dechesne CA, Turc-Carel C, Kurzenne J-Y, Wdziekonski B, Villageois A, Bagnis C, Breittmayer J-P, Groux H (2005) Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse. J Exp Med 201(9):1397–1405
Bezaire V, Mairal A, Ribet C, Lefort C, Girousse A, Jocken J, Laurencikiene J, Anesia R, Rodriguez AM, Ryden M, Stenson BM, Dani C, Ailhaud G, Arner P, Langin D (2009) Contribution of adipose triglyceride lipase and hormone-sensitive lipase to lipolysis in hMADS adipocytes. J Biol Chem 284(27):18282–18291. https://doi.org/10.1074/jbc.M109.008631
Poitou C, Divoux A, Al F, Tordjman J, Hugol D, Aissat A, Keophiphath M, Henegar C, Sp C, Clément K (2009) Role of serum amyloid a in adipocyte-macrophage cross talk and adipocyte cholesterol efflux. J Clin Endocrinol Metab 94(5):1810–1817. https://doi.org/10.1210/jc.2008-2040
Mohsen-Kanson T, Wdziekonski B, Villageois P, Hafner A-L, Lay N, Martin P, Zaragosi L-E, Billon N, Plaisant M, Peraldi P (2013) Development of adipose cells. In: Physiology and physiopathology of adipose tissue. Springer, pp 3–16
Tchkonia T, Lenburg M, Thomou T, Giorgadze N, Frampton G, Pirtskhalava T, Cartwright A, Cartwright M, Flanagan J, Karagiannides I (2007) Identification of depot-specific human fat cell progenitors through distinct expression profiles and developmental gene patterns. Am J Physiol-Endocrinol Metab 292(1):E298–E307. https://doi.org/10.1152/ajpendo.00202.2006
Fried SK, Lee MJ, Karastergiou K (2015) Shaping fat distribution: new insights into the molecular determinants of depot-and sex-dependent adipose biology. Obesity 23(7):1345–1352. https://doi.org/10.1002/oby.21133
Pellegrinelli V, Carobbio S, Vidal-Puig A (2016) Adipose tissue plasticity: how fat depots respond differently to pathophysiological cues. Diabetologia 59(6):1075–1088. https://doi.org/10.1007/s00125-016-3933-4
Tordjman J (2013) Histology of adipose tissue. In: Physiology and physiopathology of adipose tissue. Springer, pp 67–75
Christodoulides C, Lagathu C, Sethi JK, Vidal-Puig A (2009) Adipogenesis and WNT signalling. Trends Endocrinol Metab 20(1):16–24
Carrageta DF, Dias TR, Alves MG, Oliveira PF, Monteiro MP, Silva BM (2018) Anti-obesity potential of natural methylxanthines. J Funct Foods 43:84–94. https://doi.org/10.1016/j.jff.2018.02.001
Park BO, Ahrends R, Teruel MN (2012) Consecutive positive feedback loops create a bistable switch that controls preadipocyte-to-adipocyte conversion. Cell Rep 2(4):976–990. https://doi.org/10.1016/j.celrep.2012.08.038
Eisenstein A, Ravid K (2014) G protein-coupled receptors and adipogenesis: a focus on adenosine receptors. J Cell Physiol 229(4):414–421
Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM (2000) Transcriptional regulation of adipogenesis. Genes Dev 14(11):1293–1307
Rosen ED, MacDougald OA (2006) Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol 7(12):885. https://doi.org/10.1038/nrm2066
Zhu Y, Qi C, Korenberg JR, Chen X-N, Noya D, Rao MS, Reddy JK (1995) Structural organization of mouse peroxisome proliferator-activated receptor gamma (mPPAR gamma) gene: alternative promoter use and different splicing yield two mPPAR gamma isoforms. Proc Natl Acad Sci USA 92(17):7921–7925
Mueller E, Drori S, Aiyer A, Yie J, Sarraf P, Chen H, Hauser S, Rosen ED, Ge K, Roeder RG (2002) Genetic analysis of adipogenesis through peroxisome proliferator-activated receptor γ isoforms. J Biol Chem 277(44):41925–41930
Ren D, Collingwood TN, Rebar EJ, Wolffe AP, Camp HS (2002) PPARγ knockdown by engineered transcription factors: exogenous PPARγ2 but not PPARγ1 reactivates adipogenesis. Genes Dev 16(1):27–32
Zhang J, Fu M, Cui T, Xiong C, Xu K, Zhong W, Xiao Y, Floyd D, Liang J, Li E (2004) Selective disruption of PPARγ2 impairs the development of adipose tissue and insulin sensitivity. Proc Natl Acad Sci USA 101(29):10703–10708
Medina-Gomez G, Virtue S, Lelliott C, Boiani R, Campbell M, Christodoulides C, Perrin C, Jimenez-Linan M, Blount M, Dixon J (2005) The link between nutritional status and insulin sensitivity is dependent on the adipocyte-specific peroxisome proliferator-activated receptor-γ2 isoform. Diabetes 54(6):1706–1716
Tamori Y, Masugi J, Nishino N, Kasuga M (2002) Role of peroxisome proliferator-activated receptor-γ in maintenance of the characteristics of mature 3T3-L1 adipocytes. Diabetes 51(7):2045–2055
Imai T, Takakuwa R, Marchand S, Dentz E, Bornert J-M, Messaddeq N, Wendling O, Mark M, Desvergne B, Wahli W (2004) Peroxisome proliferator-activated receptor γ is required in mature white and brown adipocytes for their survival in the mouse. Proc Natl Acad Sci USA 101(13):4543–4547. https://doi.org/10.1073/pnas.0400356101
Tanaka T, Yoshida N, Kishimoto T, Akira S (1997) Defective adipocyte differentiation in mice lacking the C/EBPβ and/or C/EBPδ gene. EMBO J 16(24):7432–7443
Linhart HG, Ishimura-Oka K, DeMayo F, Kibe T, Repka D, Poindexter B, Bick RJ, Darlington GJ (2001) C/EBPα is required for differentiation of white, but not brown, adipose tissue. Proc Natl Acad Sci USA 98(22):12532–12537
Chen S-S, Chen J-F, Johnson PF, Muppala V, Lee Y-H (2000) C/EBPβ, when expressed from the C/ebpα gene locus, can functionally replace C/EBPα in liver but not in adipose tissue. Mol Cell Biol 20(19):7292–7299
El-Jack AK, Hamm JK, Pilch PF, Farmer SR (1999) Reconstitution of insulin-sensitive glucose transport in fibroblasts requires expression of both PPARγ and C/EBPα. J Biol Chem 274(12):7946–7951
Rosen ED, Hsu C-H, Wang X, Sakai S, Freeman MW, Gonzalez FJ, Spiegelman BM (2002) C/EBPα induces adipogenesis through PPARγ: a unified pathway. Genes Dev 16(1):22–26
Zuo Y, Qiang L, Farmer SR (2006) Activation of CCAAT/enhancer-binding protein (C/EBP) α expression by C/EBPβ during adipogenesis requires a peroxisome proliferator-activated receptor-γ-associated repression of HDAC1 at the C/ebpα gene promoter. J Biol Chem 281(12):7960–7967. https://doi.org/10.1074/jbc.M510682200
Zaragosi LE, Ailhaud G, Dani C (2006) Autocrine fibroblast growth factor 2 signaling is critical for self-renewal of human multipotent adipose-derived stem cells. Stem Cells 24(11):2412–2419. https://doi.org/10.1634/stemcells.2006-0006
Prusty D, Park B-H, Davis KE, Farmer SR (2002) Activation of MEK/ERK signaling promotes adipogenesis by enhancing peroxisome proliferator-activated receptor γ (PPARγ) and C/EBPα gene expression during the differentiation of 3T3-L1 preadipocytes. J Biol Chem 277(48):46226–46232
Widberg CH, Newell FS, Bachmann AW, Ramnoruth SN, Spelta MC, Whitehead JP, Hutley LJ, Prins JB (2009) Fibroblast growth factor receptor 1 is a key regulator of early adipogenic events in human preadipocytes. Am J Physiol-Endocrinol Metab 296(1):E121–E131. https://doi.org/10.1152/ajpendo.90602.2008
Kim S, Ahn C, Bong N, Choe S, Lee DK (2015) Biphasic effects of FGF2 on adipogenesis. PLoS One 10(3):e0120073. https://doi.org/10.1371/journal.pone.0120073
Oliva-Olivera W, Coín-Aragüez L, Lhamyani S, Clemente-Postigo M, Torres JA, Bernal-López MR, El Bekay R, Tinahones FJ (2016) Adipogenic impairment of adipose tissue-derived mesenchymal stem cells in subjects with metabolic syndrome: possible protective role of FGF2. J Clin Endocrinol Metab 102(2):478–487. https://doi.org/10.1210/jc.2016-2256
Hutley LJ, Newell FS, Kim Y-H, Luo X, Widberg CH, Shurety W, Prins JB, Whitehead JP (2011) A putative role for endogenous FGF-2 in FGF-1 mediated differentiation of human preadipocytes. Mol Cell Endocrinol 339(1–2):165–171. https://doi.org/10.1016/j.mce.2011.04.012
Mejhert N, Galitzky J, Pettersson AT, Bambace C, Blomqvist L, Bouloumié A, Frayn KN, Dahlman I, Arner P, Rydén M (2010) Mapping of the fibroblast growth factors in human white adipose tissue. J Clin Endocrinol Metab 95(5):2451–2457. https://doi.org/10.1210/jc.2009-2049
Zaragosi LE, Wdziekonski B, Villageois P, Keophiphath M, Maumus M, Tchkonia T, Bourlier V, Mohsen-Kanson T, Ladoux A, Elabd C, Scheideler M, Trajanoski Z, Takashima Y, Amri EZ, Lacasa D, Sengenes C, Ailhaud G, Clement K, Bouloumie A, Kirkland JL, Dani C (2010) Activin a plays a critical role in proliferation and differentiation of human adipose progenitors. Diabetes 59(10):2513–2521. https://doi.org/10.2337/db10-0013
Villageois P, Wdziekonski B, Zaragosi L-E, Plaisant M, Mohsen-Kanson T, Lay N, Ladoux A, Peraldi P, Dani C (2012) Regulators of human adipose-derived stem cell self-renewal. Am J Stem Cells 1(1):42
Divoux A, Tordjman J, Lacasa D, Veyrie N, Hugol D, Aissat A, Basdevant A, Guerre-Millo M, Poitou C, Zucker JD, Bedossa P, Clement K (2010) Fibrosis in human adipose tissue: composition, distribution, and link with lipid metabolism and fat mass loss. Diabetes 59(11):2817–2825. https://doi.org/10.2337/db10-0585
Keophiphath M, Achard V, Henegar C, Rouault C, Clément K, Lacasa D (2009) Macrophage-secreted factors promote a profibrotic phenotype in human preadipocytes. Mol Endocrinol 23(1):11–24. https://doi.org/10.1210/me.2008-0183
Muir LA, Neeley CK, Meyer KA, Baker NA, Brosius AM, Washabaugh AR, Varban OA, Finks JF, Zamarron BF, Flesher CG (2016) Adipose tissue fibrosis, hypertrophy, and hyperplasia: correlations with diabetes in human obesity. Obesity 24(3):597–605. https://doi.org/10.1002/oby.21377
Lauterbach MA, Wunderlich FT (2017) Macrophage function in obesity-induced inflammation and insulin resistance. Pflügers Archiv-Eur J Physiol 469(3–4):385–396. https://doi.org/10.1007/s00424-017-1955-5
Zamani N, Brown CW (2010) Emerging roles for the transforming growth factor-β superfamily in regulating adiposity and energy expenditure. Endocr Rev 32(3):387–403. https://doi.org/10.1210/er.2010-0018
Choy L, Skillington J, Derynck R (2000) Roles of autocrine TGF-β receptor and Smad signaling in adipocyte differentiation. J Cell Biol 149(3):667–682
Ignotz RA, Massague J (1985) Type beta transforming growth factor controls the adipogenic differentiation of 3T3 fibroblasts. Proc Natl Acad Sci USA 82(24):8530–8534
Choy L, Derynck R (2003) Transforming growth factor-β inhibits adipocyte differentiation by Smad3 interacting with CCAAT/enhancer-binding protein (C/EBP) and repressing C/EBP transactivation function. J Biol Chem 278(11):9609–9619
Zhou S, Eid K, Glowacki J (2004) Cooperation between TGF-beta and Wnt pathways during chondrocyte and adipocyte differentiation of human marrow stromal cells. J Bone Miner Res 19(3):463–470. https://doi.org/10.1359/JBMR.0301239
Suenaga M, Matsui T, Funaba M (2010) BMP inhibition with dorsomorphin limits adipogenic potential of preadipocytes. J Vet Med Sci 72(3):373–377. https://doi.org/10.1292/jvms.09-0442
Vicente López MA, Vázquez García MN, Entrena A, Olmedillas Lopez S, García-Arranz M, García-Olmo D, Zapata A (2010) Low doses of bone morphogenetic protein 4 increase the survival of human adipose-derived stem cells maintaining their stemness and multipotency. Stem Cells Dev 20(6):1011–1019. https://doi.org/10.1089/scd.2010.0355
Modica S, Straub LG, Balaz M, Sun W, Varga L, Stefanicka P, Profant M, Simon E, Neubauer H, Ukropcova B (2016) Bmp4 promotes a brown to white-like adipocyte shift. Cell Reports 16(8):2243–2258. https://doi.org/10.1016/j.celrep.2016.07.048
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Ann Rev Cell Dev Biol 20:781–810. https://doi.org/10.1146/annurev.cellbio.20.010403.113126
Ross SE, Hemati N, Longo KA, Bennett CN, Lucas PC, Erickson RL, MacDougald OA (2000) Inhibition of adipogenesis by Wnt signaling. Science 289(5481):950–953
Bennett CN, Ross SE, Longo KA, Bajnok L, Hemati N, Johnson KW, Harrison SD, MacDougald OA (2002) Regulation of Wnt signaling during adipogenesis. J Biol Chem 277(34):30998–31004. https://doi.org/10.1074/jbc.M204527200
Liu J, Farmer SR (2004) Regulating the balance between peroxisome proliferator-activated receptor γ and β-catenin signaling during Adipogenesis A glycogen synthase kinase 3β phosphorylation-defective mutant of β-catenin inhibits expression of a subset of adipogenic genes. J Biol Chem 279(43):45020–45027
Christodoulides C, Laudes M, Cawthorn WP, Schinner S, Soos M, O’Rahilly S, Sethi JK, Vidal-Puig A (2006) The Wnt antagonist Dickkopf-1 and its receptors are coordinately regulated during early human adipogenesis. J Cell Sci 119(12):2613–2620. https://doi.org/10.1242/jcs.02975
Ai M, Holmen SL, Van Hul W, Williams BO, Warman ML (2005) Reduced affinity to and inhibition by DKK1 form a common mechanism by which high bone mass-associated missense mutations in LRP5 affect canonical Wnt signaling. Mol Cell Biol 25(12):4946–4955. https://doi.org/10.1128/Mcb.25.12-4946-4955.2005
Qiu W, Andersen TE, Bollerslev J, Mandrup S, Abdallah BM, Kassem M (2007) Patients with high bone mass phenotype exhibit enhanced osteoblast differentiation and inhibition of adipogenesis of human mesenchymal stem cells. J Bone Miner Res 22(11):1720–1731. https://doi.org/10.1359/jbmr.070721
Krishnan V, Bryant HU, MacDougald OA (2006) Regulation of bone mass by Wnt signaling. J Clin Invest 116(5):1202–1209. https://doi.org/10.1172/JCI28551
Liu J, Wang H, Zuo Y, Farmer SR (2006) Functional interaction between peroxisome proliferator-activated receptor γ and β-catenin. Mol Cell Biol 26(15):5827–5837. https://doi.org/10.1128/MCB.00441-06
Semenov MV, Habas R, MacDonald BT, He X (2007) SnapShot: noncanonical Wnt signaling pathways. Cell 131(7):1378. https://doi.org/10.1016/j.cell.2007.12.011
Kanazawa A, Tsukada S, Kamiyama M, Yanagimoto T, Nakajima M, Maeda S (2005) Wnt5b partially inhibits canonical Wnt/β-catenin signaling pathway and promotes adipogenesis in 3T3-L1 preadipocytes. Biochem Biophys Res Commun 330(2):505–510. https://doi.org/10.1016/j.bbrc.2005.03.007
Mikels AJ, Nusse R (2006) Purified Wnt5a protein activates or inhibits β-catenin–TCF signaling depending on receptor context. PLoS Biol 4(4):e115. https://doi.org/10.1371/journal.pbio.0040115
Wang Y, Sul HS (2006) Ectodomain shedding of preadipocyte factor 1 (Pref-1) by tumor necrosis factor alpha converting enzyme (TACE) and inhibition of adipocyte differentiation. Mol Cell Biol 26(14):5421–5435. https://doi.org/10.1128/MCB.02437-05
Lee YL, Helman L, Hoffman T, Laborda J (1995) dlk, pG2 and Pref-1 mRNAs encode similar proteins belonging to the EGF-like superfamily. In: Identification of polymorphic variants of this RNA. Biochimica et Biophysica Acta (BBA)—Gene structure and expression, vol 1261, no 2, pp 223–232
Garcés C, Ruiz-Hidalgo MJ, Bonvini E, Goldstein J, Laborda J (1999) Adipocyte differentiation is modulated by secreted delta-like (dlk) variants and requires the expression of membrane-associated dlk. Differentiation 64(2):103–114
Baisong M, Ling Z, Li C, Sul HS (2002) Only the large soluble form of preadipocyte factor-1 (Pref-1), but not the small soluble and membrane forms, inhibits adipocyte differentiation: role of alternative splicing. Biochem J 364(1):137–144
Smas CM, Chen L, Sul HS (1997) Cleavage of membrane-associated pref-1 generates a soluble inhibitor of adipocyte differentiation. Mol Cell Biol 17(2):977–988
Smas CM, Sul HS (1993) Pref-1, a protein containing EGF-like repeats, inhibits adipocyte differentiation. Cell 73(4):725–734
Smas CM, Chen L, Zhao L, Latasa M-J, Sul HS (1999) Transcriptional repression of pref-1 by glucocorticoids promotes 3T3-L1 adipocyte differentiation. J Biol Chem 274(18):12632–12641
Mortensen SB, Jensen CH, Schneider M, Thomassen M, Kruse TA, Laborda J, Sheikh SP, Andersen DC (2012) Membrane-tethered delta-like 1 homolog (DLK1) restricts adipose tissue size by inhibiting preadipocyte proliferation. Diabetes:DB_120176. https://doi.org/10.2337/db12-0176
Traustadottir GA, Kosmina R, Sheikh SP, Jensen CH, Andersen DC (2013) Preadipocytes proliferate and differentiate under the guidance of Delta-like 1 homolog (DLK1). Adipocyte 2(4):272–275. https://doi.org/10.4161/adip.24994
Teglund S, Toftgård R (2010) Hedgehog beyond medulloblastoma and basal cell carcinoma. Biochimica et Biophysica Acta (BBA)—Rev Cancer 1805(2):181–208. https://doi.org/10.1016/j.bbcan.2010.01.003
Fernandes-Silva H, Correia-Pinto J, Moura RS (2017) Canonical sonic hedgehog signaling in early lung development. J Dev Biol 5(1):3. https://doi.org/10.3390/jdb5010003
i Altaba AR, Mas C, Stecca B (2007) The Gli code: an information nexus regulating cell fate, stemness and cancer. Trends Cell Biol 17(9):438–447. https://doi.org/10.1016/j.tcb.2007.06.007
Stecca B, Mas C, Clement V, Zbinden M, Correa R, Piguet V, Beermann F, i Altaba AR (2007) Melanomas require HEDGEHOG-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc Natl Acad Sci USA 104(14):5895–5900. https://doi.org/10.1073/pnas.0700776104
Plaisant M, Giorgetti-Peraldi S, Gabrielson M, Loubat A, Dani C, Peraldi P (2011) Inhibition of hedgehog signaling decreases proliferation and clonogenicity of human mesenchymal stem cells. PLoS One 6(2):e16798. https://doi.org/10.1371/journal.pone.0016798
Fontaine C, Cousin W, Plaisant M, Dani C, Peraldi P (2008) Hedgehog signaling alters adipocyte maturation of human mesenchymal stem cells. Stem Cells 26(4):1037–1046. https://doi.org/10.1634/stemcells.2007-0974
Shi Y, Long F (2017) Hedgehog signaling via Gli2 prevents obesity induced by high-fat diet in adult mice. Elife 6. https://doi.org/10.7554/eLife.31649
Moseti D, Regassa A, Kim WK (2016) Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int J Mol Sci 17(1). https://doi.org/10.3390/ijms17010124
Mori T, Sakaue H, Iguchi H, Gomi H, Okada Y, Takashima Y, Nakamura K, Nakamura T, Yamauchi T, Kubota N, Kadowaki T, Matsuki Y, Ogawa W, Hiramatsu R, Kasuga M (2005) Role of Kruppel-like factor 15 (KLF15) in transcriptional regulation of adipogenesis. J Biol Chem 280(13):12867–12875. https://doi.org/10.1074/jbc.M410515200
Gray S, Feinberg MW, Hull S, Kuo CT, Watanabe M, Sen-Banerjee S, DePina A, Haspel R, Jain MK (2002) The Kruppel-like factor KLF15 regulates the insulin-sensitive glucose transporter GLUT4. J Biol Chem 277(37):34322–34328. https://doi.org/10.1074/jbc.M201304200
Birsoy K, Chen Z, Friedman J (2008) Transcriptional regulation of adipogenesis by KLF4. Cell Metab 7(4):339–347. https://doi.org/10.1016/j.cmet.2008.02.001
Oishi Y, Manabe I, Tobe K, Tsushima K, Shindo T, Fujiu K, Nishimura G, Maemura K, Yamauchi T, Kubota N, Suzuki R, Kitamura T, Akira S, Kadowaki T, Nagai R (2005) Kruppel-like transcription factor KLF5 is a key regulator of adipocyte differentiation. Cell Metab 1(1):27–39. https://doi.org/10.1016/j.cmet.2004.11.005
Pei H, Yao Y, Yang Y, Liao K, Wu JR (2011) Kruppel-like factor KLF9 regulates PPARgamma transactivation at the middle stage of adipogenesis. Cell Death Differ 18(2):315–327. https://doi.org/10.1038/cdd.2010.100
Li D, Yea S, Li S, Chen Z, Narla G, Banck M, Laborda J, Tan S, Friedman JM, Friedman SL, Walsh MJ (2005) Kruppel-like factor-6 promotes preadipocyte differentiation through histone deacetylase 3-dependent repression of DLK1. J Biol Chem 280(29):26941–26952. https://doi.org/10.1074/jbc.M500463200
Wu J, Srinivasan SV, Neumann JC, Lingrel JB (2005) The KLF2 transcription factor does not affect the formation of preadipocytes but inhibits their differentiation into adipocytes. Biochemistry 44(33):11098–11105. https://doi.org/10.1021/bi050166i
Banerjee SS, Feinberg MW, Watanabe M, Gray S, Haspel RL, Denkinger DJ, Kawahara R, Hauner H, Jain MK (2003) The Kruppel-like factor KLF2 inhibits peroxisome proliferator-activated receptor-gamma expression and adipogenesis. J Biol Chem 278(4):2581–2584. https://doi.org/10.1074/jbc.M210859200
Tong Q, Dalgin G, Xu H, Ting C-N, Leiden JM, Hotamisligil GS (2000) Function of GATA transcription factors in preadipocyte-adipocyte transition. Science 290(5489):134–138
Tong Q, Tsai J, Tan G, Dalgin G, Hotamisligil GS (2005) Interaction between GATA and the C/EBP family of transcription factors is critical in GATA-mediated suppression of adipocyte differentiation. Mol Cell Biol 25(2):706–715. https://doi.org/10.1128/MCB.25.2.706-715.2005
Wang L, Di L-j (2015) Wnt/β-catenin mediates AICAR effect in increasing GATA3 expression and inhibiting adipogenesis. J Biol Chem:JBC M115:641332. https://doi.org/10.1074/jbc.M115.641332
Benador IY, Veliova M, Mahdaviani K, Petcherski A, Wikstrom JD, Assali EA, Acin-Perez R, Shum M, Oliveira MF, Cinti S, Sztalryd C, Barshop WD, Wohlschlegel JA, Corkey BE, Liesa M, Shirihai OS (2018) Mitochondria bound to lipid droplets have unique bioenergetics, composition, and dynamics that support lipid droplet expansion. Cell Metab 27(4):869–885. https://doi.org/10.1016/j.cmet.2018.03.003
Inagaki T, Sakai J, Kajimura S (2016) Transcriptional and epigenetic control of brown and beige adipose cell fate and function. Nat Rev Mol Cell Biol 17(8):480. https://doi.org/10.1038/nrm.2016.62
Jung SM, Sanchez-Gurmaches J, Guertin DA (2018) Brown adipose tissue development and metabolism. Handb Exp Pharmacol. https://doi.org/10.1007/164_2018_168
Rosenwald M, Perdikari A, Rulicke T, Wolfrum C (2013) Bi-directional interconversion of brite and white adipocytes. Nat Cell Biol 15(6):659–667. https://doi.org/10.1038/ncb2740
Lee YH, Petkova AP, Konkar AA, Granneman JG (2015) Cellular origins of cold-induced brown adipocytes in adult mice. FASEB Journal 29(1):286–299. https://doi.org/10.1096/fj.14-263038
Kajimura S, Seale P, Kubota K, Lunsford E, Frangioni JV, Gygi SP, Spiegelman BM (2009) Initiation of myoblast to brown fat switch by a PRDM16–C/EBP-β transcriptional complex. Nature 460(7259):1154. https://doi.org/10.1038/nature08262
Seale P, Bjork B, Yang W, Kajimura S, Chin S, Kuang S, Scime A, Devarakonda S, Conroe HM, Erdjument-Bromage H (2008) PRDM16 controls a brown fat/skeletal muscle switch. Nature 454(7207):961. https://doi.org/10.1038/nature07182
Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, Ishibashi J, Cohen P, Cinti S, Spiegelman BM (2011) Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 121(1):96–105. https://doi.org/10.1172/JCI44271
Seale P, Kajimura S, Yang W, Chin S, Rohas LM, Uldry M, Tavernier G, Langin D, Spiegelman BM (2007) Transcriptional control of brown fat determination by PRDM16. Cell Metab 6(1):38–54. https://doi.org/10.1016/j.cmet.2007.06.001
Ohno H, Shinoda K, Ohyama K, Sharp LZ, Kajimura S (2013) EHMT1 controls brown adipose cell fate and thermogenesis through the PRDM16 complex. Nature 504(7478):163. https://doi.org/10.1038/nature12652
Harms MJ, Ishibashi J, Wang W, Lim H-W, Goyama S, Sato T, Kurokawa M, Won K-J, Seale P (2014) Prdm16 is required for the maintenance of brown adipocyte identity and function in adult mice. Cell Metab 19(4):593–604. https://doi.org/10.1016/j.cmet.2014.03.007
Ishibashi J, Firtina Z, Rajakumari S, Wood KH, Conroe HM, Steger DJ, Seale P (2012) An Evi1-C/EBPβ complex controls peroxisome proliferator-activated receptor γ2 gene expression to initiate white fat cell differentiation. Mol Cell Biol 32(12):2289–2299. https://doi.org/10.1128/MCB.06529-11
Rajakumari S, Wu J, Ishibashi J, Lim H-W, Giang A-H, Won K-J, Reed RR, Seale P (2013) EBF2 determines and maintains brown adipocyte identity. Cell Metab 17(4):562–574. https://doi.org/10.1016/j.cmet.2013.01.015
Wang W, Kissig M, Rajakumari S, Huang L, Lim H-w, Won K-J, Seale P (2014) Ebf2 is a selective marker of brown and beige adipogenic precursor cells. Proc Natl Acad Sci USA 111(40):14466–14471. https://doi.org/10.1073/pnas.1412685111
Shinoda K, Luijten IH, Hasegawa Y, Hong H, Sonne SB, Kim M, Xue R, Chondronikola M, Cypess AM, Tseng Y-H (2015) Genetic and functional characterization of clonally derived adult human brown adipocytes. Nat Med 21(4):389. https://doi.org/10.1038/nm.3819
Sambeat A, Gulyaeva O, Dempersmier J, Tharp KM, Stahl A, Paul SM, Sul HS (2016) LSD1 interacts with Zfp516 to promote UCP1 transcription and brown fat program. Cell Rep 15(11):2536–2549. https://doi.org/10.1016/j.celrep.2016.05.019
Dempersmier J, Sambeat A, Gulyaeva O, Paul SM, Hudak CS, Raposo HF, Kwan H-Y, Kang C, Wong RH, Sul HS (2015) Cold-inducible Zfp516 activates UCP1 transcription to promote browning of white fat and development of brown fat. Mol Cell 57(2):235–246. https://doi.org/10.1016/j.molcel.2014.12.005
Ricquier D (2013) Brown adipose tissue: function and development. In: Physiology and physiopathology of adipose tissue. Springer, pp 51–66
Nicholls DG, Locke RM (1984) Thermogenic mechanisms in brown fat. Physiol Rev 64(1):1–64
Nicholls DG (2006) The physiological regulation of uncoupling proteins. Biochimica et Biophysica Acta (BBA)—Bioenergetics 1757(5–6):459–466. https://doi.org/10.1016/j.bbabio.2006.02.005
Roh HC, Tsai LT, Shao M, Tenen D, Shen Y, Kumari M, Lyubetskaya A, Jacobs C, Dawes B, Gupta RK (2018) Warming induces significant reprogramming of beige, but not brown, adipocyte cellular identity. Cell Metab 27(5):1121–1137. e1125. https://doi.org/10.1016/j.cmet.2018.03.005
Sluse FE, Jarmuszkiewicz W, Navet R, Douette P, Mathy G, Sluse-Goffart CM (2006) Mitochondrial UCPs: new insights into regulation and impact. Biochimica et Biophysica Acta (BBA)—Bioenergetics 1757(5–6):480–485. https://doi.org/10.1016/j.bbabio.2006.02.004
Fedorenko A, Lishko PV, Kirichok Y (2012) Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell 151(2):400–413. https://doi.org/10.1016/j.cell.2012.09.010
Uldry M, Yang W, St-Pierre J, Lin J, Seale P, Spiegelman BM (2006) Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab 3(5):333–341. https://doi.org/10.1016/j.cmet.2006.04.002
Lin J, Wu P-H, Tarr PT, Lindenberg KS, St-Pierre J, Zhang C-y, Mootha VK, Jäger S, Vianna CR, Reznick RM (2004) Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1α null mice. Cell 119(1):121–135
Kong X, Banks A, Liu T, Kazak L, Rao RR, Cohen P, Wang X, Yu S, Lo JC, Tseng Y-H (2014) IRF4 is a key thermogenic transcriptional partner of PGC-1α. Cell 158(1):69–83. https://doi.org/10.1016/j.cell.2014.04.049
Hallberg M, Morganstein DL, Kiskinis E, Shah K, Kralli A, Dilworth SM, White R, Parker MG, Christian M (2008) A functional interaction between RIP140 and PGC-1α regulates the expression of the lipid droplet protein CIDEA. Mol Cell Biol 28(22):6785–6795. https://doi.org/10.1128/MCB.00504-08
Wang H, Zhang Y, Yehuda-Shnaidman E, Medvedev AV, Kumar N, Daniel KW, Robidoux J, Czech MP, Mangelsdorf DJ, Collins S (2008) Liver X receptor α is a transcriptional repressor of the uncoupling protein 1 gene and the brown fat phenotype. Mol Cell Biol 28(7):2187–2200. https://doi.org/10.1128/MCB.01479-07
Picard F, Géhin M, Annicotte J-S, Rocchi S, Champy M-F, O’Malley BW, Chambon P, Auwerx J (2002) SRC-1 and TIF2 control energy balance between white and brown adipose tissues. Cell 111(7):931–941
Pan D, Fujimoto M, Lopes A, Wang Y-X (2009) Twist-1 is a PPARδ-inducible, negative-feedback regulator of PGC-1α in brown fat metabolism. Cell 137(1):73–86. https://doi.org/10.1016/j.cell.2009.01.051
Shin H, Ma Y, Chanturiya T, Cao Q, Wang Y, Kadegowda AK, Jackson R, Rumore D, Xue B, Shi H (2017) Lipolysis in brown adipocytes is not essential for cold-induced thermogenesis in mice. Cell Metab 26(5):764–777.e765. https://doi.org/10.1016/j.cmet.2017.09.002
Schreiber R, Diwoky C, Schoiswohl G, Feiler U, Wongsiriroj N, Abdellatif M, Kolb D, Hoeks J, Kershaw EE, Sedej S (2017) Cold-induced thermogenesis depends on ATGL-mediated lipolysis in cardiac muscle, but not brown adipose tissue. Cell Metab 26(5):753–763. e757. https://doi.org/10.1016/j.cmet.2017.09.004
Sanchez-Gurmaches J, Tang Y, Jespersen NZ, Wallace M, Calejman CM, Gujja S, Li H, Edwards YJ, Wolfrum C, Metallo CM (2018) Brown fat AKT2 is a cold-induced kinase that stimulates ChREBP-mediated de novo lipogenesis to optimize fuel storage and thermogenesis. Cell Metab 27(1):195–209. e196. https://doi.org/10.1016/j.cmet.2017.10.008
Simcox J, Geoghegan G, Maschek JA, Bensard CL, Pasquali M, Miao R, Lee S, Jiang L, Huck I, Kershaw EE (2017) Global analysis of plasma lipids identifies liver-derived acylcarnitines as a fuel source for brown fat thermogenesis. Cell Metab 26(3):509–522. e506. https://doi.org/10.1016/j.cmet.2017.08.006
Pietrocola F, Galluzzi L, Bravo-San Pedro JM, Madeo F, Kroemer G (2015) Acetyl coenzyme A: a central metabolite and second messenger. Cell Metab 21(6):805–821. https://doi.org/10.1016/j.cmet.2015.05.014
Yoneshiro T, Aita S, Matsushita M, Kayahara T, Kameya T, Kawai Y, Iwanaga T, Saito M (2013) Recruited brown adipose tissue as an antiobesity agent in humans. J Clin Invest 123(8):3404–3408. https://doi.org/10.1172/JCI67803
van der Lans AA, Hoeks J, Brans B, Vijgen GH, Visser MG, Vosselman MJ, Hansen J, Jörgensen JA, Wu J, Mottaghy FM (2013) Cold acclimation recruits human brown fat and increases nonshivering thermogenesis. J Clin Investig 123(8):3395–3403. https://doi.org/10.1172/JCI68993
Lee P, Smith S, Linderman J, Courville AB, Brychta RJ, Dieckmann W, Werner CD, Chen KY, Celi FS (2014) Temperature-acclimated brown adipose tissue modulates insulin sensitivity in humans. Diabetes:DB_140513. https://doi.org/10.2337/db14-0513
Ohno H, Shinoda K, Spiegelman BM, Kajimura S (2012) PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab 15(3):395–404. https://doi.org/10.1016/j.cmet.2012.01.019
Wang QA, Tao C, Gupta RK, Scherer PE (2013) Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat Med 19(10):1338. https://doi.org/10.1038/nm.3324
Cinti S (2009) Transdifferentiation properties of adipocytes in the adipose organ. Am J Physiol-Endocrinol Metab 297(5):E977–E986. https://doi.org/10.1152/ajpendo.00183.2009
Barbatelli G, Murano I, Madsen L, Hao Q, Jimenez M, Kristiansen K, Giacobino JP, De Matteis R, Cinti S (2010) The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol-Endocrinol Metab 298(6):E1244–E1253. https://doi.org/10.1152/ajpendo.00600.2009
Berry DC, Jiang Y, Graff JM (2016) Mouse strains to study cold-inducible beige progenitors and beige adipocyte formation and function. Nat Commun 7:10184. https://doi.org/10.1038/ncomms10184
Jiang Y, Berry DC, Graff JM (2017) Distinct cellular and molecular mechanisms for β3 adrenergic receptor-induced beige adipocyte formation. eLife 6:e30329. https://doi.org/10.7554/elife.30329
Ikeda K, Maretich P, Kajimura S (2018) The common and distinct features of brown and beige adipocytes. Trends Endocrinol Metab 29(3):191–200. https://doi.org/10.1016/j.tem.2018.01.001
Singh AM, Dalton S (2018) What can ‘Brown-ing’ do for you? Trends Endocrinol Metab 29(5):349–359. https://doi.org/10.1016/j.tem.2018.03.002
Altshuler-Keylin S, Shinoda K, Hasegawa Y, Ikeda K, Hong HM, Kang QQ, Yang YY, Perera RM, Debnath J, Kajimura S (2016) Beige adipocyte maintenance is regulated by autophagy-induced mitochondrial clearance. Cell Metab 24(3):402–419. https://doi.org/10.1016/j.cmet.2016.08.002
Kosacka J, Kern M, Klöting N, Paeschke S, Rudich A, Haim Y, Gericke M, Serke H, Stumvoll M, Bechmann I (2015) Autophagy in adipose tissue of patients with obesity and type 2 diabetes. Mol Cell Endocrinol 409:21–32. https://doi.org/10.1016/j.mce.2015.03.015
Ukropec J, Anunciado RP, Ravussin Y, Hulver MW, Kozak LP (2006) UCP1-independent thermogenesis in white adipose tissue of cold-acclimated Ucp1-/-mice. J Biol Chem 281(42):31894–31908. https://doi.org/10.1074/jbc.M606114200
Granneman J, Burnazi M, Zhu Z, Schwamb L (2003) White adipose tissue contributes to UCP1-independent thermogenesis. Am J Physiol-Endocrinol Metab 285(6):E1230–E1236
Ikeda K, Kang Q, Yoneshiro T, Camporez JP, Maki H, Homma M, Shinoda K, Chen Y, Lu X, Maretich P (2017) UCP1-independent signaling involving SERCA2b-mediated calcium cycling regulates beige fat thermogenesis and systemic glucose homeostasis. Nat Med 23(12):1454. https://doi.org/10.1038/nm.4429
Engin AB (2017) MicroRNA and adipogenesis. In: Obesity and lipotoxicity. Springer, pp 489–509
Shamsi F, Zhang H, Tseng Y-H (2017) MicroRNA regulation of brown adipogenesis and thermogenic energy expenditure. Front Endocrinol 8:205. https://doi.org/10.3389/fendo.2017.00205
Acknowledgments
This work was supported by the Portuguese Foundation for Science and Technology: PTDC/MEC-MET/32151/2017; David F. Carrageta (SFRH/BD/136779/2018); Marco G. Alves (IFCT2015, PTDC/BIM-MET/4712/2014 and PTDC/MEC-AND/28691/2017); Pedro F. Oliveira (IFCT2015 and PTDC/BBB-BQB/1368/2014); UMIB (Pest-OE/SAL/UI02015/2019); and co-funded by FEDER funds through the POCI/COMPETE 2020.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
None to declare.
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2020 The Author(s)
About this chapter
Cite this chapter
Carrageta, D.F., Oliveira, P.F., Monteiro, M.P., Alves, M.G. (2020). Adipocyte Specific Signaling. In: Silva, J.V., Freitas, M.J., Fardilha, M. (eds) Tissue-Specific Cell Signaling. Springer, Cham. https://doi.org/10.1007/978-3-030-44436-5_15
Download citation
DOI: https://doi.org/10.1007/978-3-030-44436-5_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-44435-8
Online ISBN: 978-3-030-44436-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)