Abstract
B-type lamins are fundamental components of the nuclear lamina, a complex structure that acts as a scaffold for organization and function of the nucleus. Lamin B1 and B2, the most represented isoforms, are encoded by LMNB1 and LMNB2 gene, respectively. All B-type lamins are synthesized as precursors and undergo sequential post-translational modifications to generate the mature protein. B-type lamins are involved in a wide range of nuclear functions, including DNA replication and repair, regulation of chromatin and nuclear stiffness. Moreover, lamins B1 and B2 regulate several cellular processes, such as tissue development, cell cycle, cellular proliferation, senescence, and DNA damage response. During embryogenesis, B-type lamins are essential for organogenesis, in particular for brain development. As expected from the numerous and pivotal functions of B-type lamins, mutations in their genes or fluctuations in their expression levels are critical for the onset of several diseases. Indeed, a growing range of human disorders have been linked to lamin B1 or B2, increasing the complexity of the group of diseases collectively known as laminopathies. This review highlights the recent findings on the biological role of B-type lamins under physiological or pathological conditions, with a particular emphasis on brain disorders and cancer.
Graphical abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lamins are type V intermediate filament proteins located beneath the inner nuclear membrane, where they form a high-ordered meshwork named nuclear lamina [1, 2]. The nuclear lamina provides structural support to the nucleus, contributing to nuclear shape and mechanical stability and supporting and regulating chromatin organization.
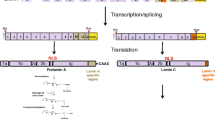
In mammals, lamins are divided into A- and B-types, based on their sequence homologies (Fig. 1).
Domain organization of the main human lamin isoforms. The α-helical rod domain comprises four segments, 1A, 1B, 2A, 2B, which are separated by linker segments. The C-terminal tail domain contains a nuclear localization signal (NLS), an immunoglobulin domain (Ig-like), and a conserved CAAX box, which undergoes farnesylation
LMNA gene, mapping on chromosome 1q11-q2, gives rise to different splicing isoforms: Lamin A, lamin C (the two major isoforms), lamin A ∆10 and lamin C2 [3]. Lamin A is translated as a precursor protein named prelamin A which is post-translationally modified. In particular, ZMPSTE24 metalloproteinase recognizes and cleaves the last 18 C-terminal aminoacids of farnesylated prelamin A, thus producing mature lamin A [3].
The two major B-type lamins, lamins B1 and B2, are encoded by different genes (LMNB1 and LMNB2), located on chromosomes 5q23.2-q31.3 and 19p13.3, respectively (Fig. 1). LMNB2 gene also encodes the minor isoform lamin B3, a germ cell-specific isoform that results from alternative splicing of the LMNB2 gene [1]. All B-type lamins are translated as prelamins, requiring extensive post-translational modifications of the carboxy-terminal-CAAX box, which is subjected to farnesylation, carboxymethylation and partial cleavage to generate lamin B1 and lamin B2 mature forms [1]. The endoprotease involved in B-type lamin cleavage is the Ras-converting enzyme 1 (Rce1) [4]. While lamin B2 farnesylation is not essential for physiological development, this is not true for lamin B1 [5].
While A-type lamins are also present in the nucleoplasm as apparently freely diffusible molecules [6], B lamins are preferentially associated to the nuclear membrane, probably due to their permanently farnesylated state [7, 8]. Indeed, type-B lamins are also located in the nucleoplasm although they were less abundant and more static than type-A [7, 9, 10].
The different disassembly and assembly properties of A- and B-type lamins during mitosis suggest that lamins may be processed in different locations within the nuclear environment and that they form separate networks in the nuclear lamina [1, 8]. Indeed, the different lamin isoforms shape spatially separate but interacting overlapping filament meshwork [7, 9, 10]. Compared to lamin A and C, lamins B have a higher edge length and edge connectivity and in particular, lamin B1 has more edges per face than lamin B2 [9, 11].
The expression pattern is different between A- and B-type lamins. B lamins are expressed in most cell types, independently of their differentiation state, whereas lamin A/C is not expressed in embryonic stem cells, but in most differentiated cells [12].
B-type lamins are not mutually redundant during embryogenesis, being required for normal tissue development, in particular of the central nervous system (CNS) [13], and seem to be non-essential in some tissues, such as epidermis and liver [14]. Of note, lamin A (but not lamin C) is expressed at low levels in most neural and neuroendocrine cells, while lamin A/C is expressed upon stimulation in hematopoietic cells.
Regarding the protein structural properties, B-type lamin tetramers feature elastic properties, which allow significant deformation of the nuclear envelope [10, 11], while lamin A/C confers deformation-resistant viscous stiffness to nuclei [15]. Notably, a recent study by Wintner et al. showed that both lamin A/C and lamin B contribute to nuclear stiffness, while viscosity is specified mostly by lamin A [16].
Importantly, lamins A and B have different binding partners at the nuclear envelope and inside the nucleus, which is reflected in their involvement in a plethora of different intracellular pathways that affect both cytoskeleton and chromatin functional organization [7, 17, 18]. B1 and B2 have been shown to bind 23 and 7 protein partners, respectively [18], while they also bind chromatin at specific sequences called lamina-associated domains or LADs [19,20,21].
In this review, we will summarize and discuss the updated knowledge about the role of B-type lamins in physiological and pathological conditions. Considering the heterogenous and controversial field of lamin B-related diseases, the rarity of most of these pathologies, and non-completely understood mechanisms, this review is aimed at providing an overview of different disorders tethered by shared biological actors.
Role of B-type lamins in cellular homeostasis
B-type lamins are involved in a wide range of nuclear functions, including structural support to the nucleus, regulation of chromatin and DNA replication, transcription and DNA repair [1]. In addition, lamins B are implicated in several cellular processes, including cell cycle regulation, cellular senescence, DNA damage, and tissue development.
Cellular senescence
Lamin B1 is involved in cellular senescence, a cellular decline characterized by permanent cell cycle arrest and by a proinflammatory secretory phenotype (SASP) [22]. Lamin B1 loss causes alterations of nuclear shape and low lamin B1 levels are associated with geroconversion of cells, as it has been demonstrated in murine models and human tissues [23, 24].
Depletion of lamin B1 in senescent cells may be associated with changes in histone methylation [25, 26], including the reduction of repressive histone mark H3K27me3, and the activation of different SASP genes [26].
Freund and colleagues reported that, in human cells, reduced lamin B expression mediated by the p53 and pRb pathways is related to cellular senescence [24]. Authors observed that p53 and pRb activation is sufficient to reduce lamin B1 expression independently of known hallmarks of ageing, such as activation of DNA damage response (DDR), p38 mitogen-activated protein kinase (MAPK), nuclear factor-κB (NF-κB) and reactive oxygen species (ROS) [24]. On the other hand, in mice, lamin B1 deficiency is linked to an increase of ROS production through a dysregulation of different Oct-1-dependent genes that are involved in oxidative stress response [27].
A few years later, Dreesen et al. showed that the single lamin B1 depletion is not sufficient to trigger senescence; rather, it is likely that lamin B1 depletion modulates senescence when accompanied by additional stress, such as sparse growth [28]. The same authors observed that lamin B1 overexpression impairs proliferation and culminates in cellular senescence, with these effects being rescued by telomerase or inactivation of p53 [28].
One possible explanation of these apparently conflictual results could be related to lamin A/C expression. Indeed, cells with low levels of lamin A/C are more sensitive to lamin B1 overexpression, displaying impaired proliferation, increased DNA damage, and senescence [29]. It is also conceivable that a range of lamin B1 amount is required to keep cells in a healthy condition, while altered LMNB1 expression leads to senescence. Of note, it has been also shown that different senescence-inducing conditions as oncogene expression or oxidative stress elicit different effects on lamin B1 levels [30].
Cell cycle regulation
B-type lamins play important roles in cell cycle regulation and cellular proliferation. In fact, B-type lamins are required to maintain chromatin condensation in interphase nuclei, and loss of lamin B1 is related to prolonged S phase [31]. Moreover, it has been demonstrated in cancer cells that a decrease of lamin B1 levels leads to a reduction of cell proliferation and deficiencies in DNA synthesis, resulting in the accumulation of cells in early S phase, probably due to a stalling of replication forks [32]. Indeed, it seems that lamin B1 may play an important role in the replication initiation, such as assembly of replicative complex, and in fork progression, especially during the elongation phase [32].
DNA damage
It has been reported that lack of lamin B1 in cancer cells causes chromosomal instability and persistent DNA damages [32]. These damages were revealed by the numerous spontaneous γH2AX and 53BP1 foci, suggesting an accumulation of double-strand-breaks (DSBs). Depletion of lamin B1 induces variations of several repair proteins, including 53BP1, BRCA1, RAD50, and DNA-PKcs, NBS1, and RAD51 [32].
Moreover, lamin B1 seems to control the homologous recombination (HR) repair systems via its interaction with RAD51. It has been hypothesised that this interaction stabilizes RAD51 inhibiting its proteasomal degradation [33].
Tissue development
Lamins B are ubiquitously expressed with high expression in different tissues as bone marrow, blood, gastrointestinal tract, and brain. As anticipated above, lamins B play a fundamental role in the normal development of different tissues, especially of the CNS [13, 23]. It follows that mutations in LMNB genes or alterations in lamin B levels have deleterious consequences and are linked to several CNS diseases.
First studies performed in murine models showed that lamin B1 plays a key role in embryonic development and organogenesis of brain [13, 34]. LMNB1 knockout mice show perinatal lethality and are characterized by lower brain dimension, abnormal layering and apoptosis of cortical neurons [34].
In addition, lamin B1 is essential for dendrite differentiation in primary mouse cortical neurons [35], while lamin B2 localizes in axons and prevents axonal degeneration by maintaining mitochondrial function, axonal integrity and nuclear migration [36].
Loss of lamin B1 causes hypothalamic abnormalities, results in a decrease of cerebellar dimensions due to irregular neural migration [37], and may be related to nuclear membrane ruptures in migrating neurons, accompanied by DNA damage and cell death [38].
It has been recently reported that lamin B1 is downregulated with age in mouse hippocampal neural stem cells [39]. Importantly, restoring lamin B1 levels is sufficient to rescue proliferation deficits in aged mouse hippocampus, improving stem cell proliferation and CNS neurogenesis [39].
All the results support the thesis that a fine control of lamin B1 levels is critical for brain development and maintenance and a balanced expression of lamin B1 in neurons must be guarantee. Indeed, the overexpression of lamin B1 in the mouse CNS is related to abnormal neuronal activity, microglial reaction, astrogliosis and myelin abnormalities, including reduced synthesis of myelin in oligodendrocytes [40, 41]. Nevertheless, the specific mechanisms by which lamin B1 controls brain differentiation are still to be clarified.
Lamin B-related laminopathies
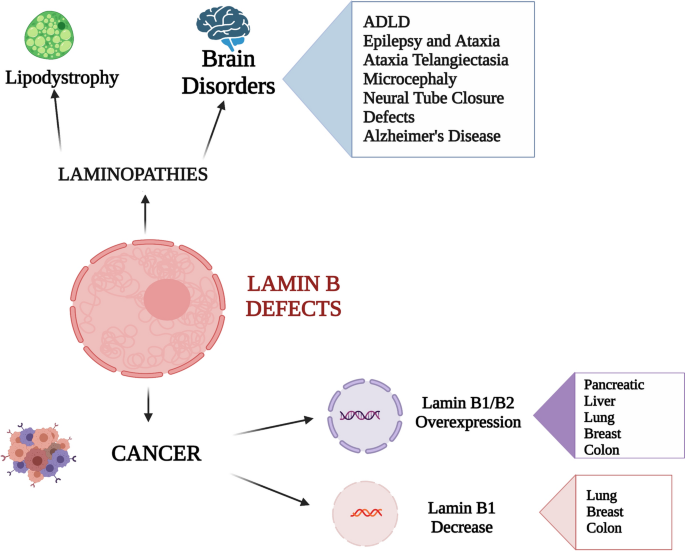
Mutations in lamins or lamin-related genes lead to several human diseases called laminopathies that include muscle, metabolic, neuronal and ageing-related diseases. Even though most of the mutations are in the LMNA gene, mutations in LMNB1 and LMNB2 genes have been reported. The lower number of LMNB-related diseases compared to lamin A-related disorders is probably due to the fact that B-type lamins are required in embryonic and fetal development and gene mutations are often lethal. Moreover, lamin B expression levels are critical for protein functionality and weak changes in lamin B expression levels can lead to different disease, as reported below (Table 1).
Adult-onset autosomal dominant leukodystrophy (ADLD)
Adult-onset Autosomal Dominant Leukodystrophy (ADLD) is characterized by accumulation of lamin B1 caused by tandem duplications involving the LMNB1 gene or deletions upstream of the LMNB1 gene [42, 43]. ADLD is an extremely rare disease characterized by late onset neurological features, such as progressive myelin loss in the CNS and fatal outcome [42]. In the majority of ADLD cases, the first clinical manifestations are related to autonomic dysfunction, followed by ataxia and cognitive impairment that reflects pyramidal and cerebellar involvement.
Several studies demonstrated that LMNB1 gene duplication is related to defects of brain development and to a severe demyelinating phenotype [42]. Of note, lamin B1 has to be finely tuned during brain development, with a peak at birth and followed by a successive gradual decrease [44].
Lamin B1 overexpression affects inner nuclear membrane proteins, chromatin organization, and nuclear pore transport, causing defects in oligodendrocytes differentiation [44, 45]. LMNB1 duplication is also related to inhibition of myelin-specific genes and to the activation of glial fibrillary acidic protein (GFAP) transcription [44].
An increase of lamin B1 expression is also related to an untimely block of oligodendrocytes’ differentiation characterized by an alteration of the myelin proteolipid protein (PLP), the major myelin membrane lipoprotein [44]. Importantly, different types of mutations in PLP1 gene leads to demyelination in a group of disorders known as hypomyelinating leukodystrophies (Pelizaeus-Merzbacher diseases) [46], while PLP overexpression causes oligodendrocytes death [47].
Different mouse models have been developed to better understand the biological mechanisms that link lamin B1 overexpression and demyelination in ADLD patients. In 2013, Heng et al. generated a BAC transgenic mouse model carrying additional copies of murine wild-type lamin B1 (Lmnb1BAC) that recapitulates several features of ADLD [40]. The same group created transgenic mice overexpressing Lmnb1 in different CNS cell lineages, demonstrating that lamin B1 overexpression in oligodendrocytes is sufficient for the onset of histopathological, molecular, and behavioural deficits characteristic of ADLD (not in neurons or astrocytes). Moreover, they reported that lamin B1 overexpression induces motor deficits, aberrant myelin formation, axonal degeneration, and demyelination with a marked decrease of PLP1, due to reduced occupancy of the transcription factor Yin Yang 1 (Yy1) at the promoter [40].
A following independent study generated an ADLD derived oligodendrocyte-specific transgenic mouse overexpressing lamin B1 [41]. This interesting study demonstrated a molecular link between lamin B1 and lipid synthesis in oligodendrocytes and a reduction of myelin-enriched lipids associated to myelin destruction at spinal cord level [41].
Significant insights into the pathophysiology of ADLD have been provided by Bartoletti-Stella and al. that investigated how lamin B1 duplication may impact the whole-genome expression profile, in tissues derived from ADLD patients [48]. Authors reported that duplication of LMNB1 affects transcription and alternative splicing of several genes associated with the immune system, neuronal and skeletal development. Among these genes, raver2, a RNA-binding protein (RBP) that modulates the splicing repressor polypyrimidine tract binding protein (PTB), is significantly increased [48].
Although the demyelination is one of the most significant aspects of ADLD, the molecular and functional mechanisms underlying this pathology have not been fully elucidated. About this, Bartoletti-Stella et al. suggest that ADLD could be considered as a “spliceopathy” caused by increased levels of lamin B1 and raver2 and that the characteristic demyelination could be the result of increased expression, during adulthood, of the embryonic isoform of the PLP1 protein, which has a crucial role in myelin maintenance. Of note, the pivotal role of raver2 in brain development was previously demonstrated in the brains of Lmnb1Δ/Δ embryos, where a dysregulation of raver2 may cause abnormal development, with reduced brain size and impaired corticogenesis [13, 34, 49].
Recent studies indicated that also astrocytes may be pivotal in the development of ADLD. In particular, our research group demonstrated that several nuclear alterations are found in astrocytes, but not in oligodendrocytes, that overexpress lamin B1 [50]. These astrocytes are characterized by reduction in leukaemia inhibitory factor (LIF) secretion which, in turn, leads to down-regulation of the pro-survival Janus kinase-signal transducer and activator of transcription protein 3 (Jak/Stat3) and phosphatidylinositol-3 phosphate kinase (PI3K)/Akt signalling pathways. Increased production of ROS is found in primary dermal fibroblasts from ADLD patients, demonstrating that they may play an additional role in the pathogenesis of ADLD [50]. This effect is consistent with previous studies showing altered nuclear dynamics of the transcription factor Oct-1 in ADLD dermal fibroblast [51]. Oct-1, a main player in stress response, is sequestered at the nuclear envelope by overexpressed lamin B1, which causes altered stress response [51]. The involvement of muscle tissue in ADLD due to impairment of myosin heavy chain 7 expression downstream of Oct-1 dysregulation has been also suggested [51].
A link between lamin B1 overexpression and signalling pathway alterations has been recently supported by our last article, reporting that lamin B increase leads to inactivation of glycogen synthase kinase (GSK)3β, but not the upregulation of β-catenin targets and to a reduction of astrocyte survival in vitro [52]. Interestingly, astrocytes overexpressing lamin B1 show increased immunoreactivity for both GFAP and vimentin together with NF-κB phosphorylation and c-Fos increase, suggesting astrocytes reactivity and substantial cellular activation [52].
Finally, a recent study has achieved to abrogate the ADLD-specific phenotypes with the use of siRNA treatment (non-duplicated allele by ASP-siRNA used may reduce the amount of LMNB1 without excessively downregulating the gene) in ADLD fibroblasts, in murine oligodendrocytes overexpressing human LMNB1, and neurons directly reprogrammed from patients' fibroblasts, providing an effective therapeutic strategy for ADLD treatment [53].
Lipodystrophies
Lipodystrophies are a heterogeneous group of genetic or acquired diseases characterized by defects affecting formation, distribution, and maintenance of adipose tissue. Lipodystrophic patients show a loss of fat in specific district or in the whole body. Mutations in LMNB2 may cause susceptibility to acquired partial lipodystrophy. Indeed, enrichment of heterozygous LMNB2 variants in acquired partial lipodystrophy patients has been described [54, 55].
Lamin B-related neuronal disorders
Several lamin B-related laminopathies are frequently confined to CNS. Unlike lamin A, which is physiologically expressed at low levels in the brain, LMNB1 gene is highly expressed and small fluctuations in the expression levels seem to have remarkable molecular and functional consequences for the CNS cells [29, 56]. Of note, both lamin B1 and B2 are required during brain development as they regulate nuclear migration during neurogenesis of the CNS.
In murine and human models, indeed, mutations or alterations of lamin B expression are related to different neuronal diseases (as reported below). Mice carrying homozygous deletions of Lmnb1 and Lmnb2 genes die at birth and display a neuronal phenotype resembling lissencephaly [13, 37, 57]. Moreover, Lmnb2-null mice showed abnormal development of the cerebral cortex and cerebellum [34, 57].
-
(a)
Epilepsy and ataxia
Variants of LMNB2 have been described in a family with progressive myoclonic epilepsy and ataxia [58]. Homozygous missense mutation in LMNB2 has been found in patients with autosomal recessive progressive myoclonus epilepsy with early ataxia syndrome, probably caused by a mistaken neuronal migration.
-
(b)
Ataxia telangiectasia
Similar to ADLD, elevated levels of lamin B1 have also been reported in patients with ataxia telangiectasia (AT), an autosomal-recessive disorder characterized by neurological defects, including demyelination of the CNS [30]. AT is caused by mutations of ataxia telangiectasia mutated (ATM) that controls early steps during DNA damage response signalling and could contribute to cellular senescence [28, 59]. The complex relationship between elevated lamin B1 levels and impaired DNA damage response has been previously reported.
-
(c)
Microcephaly
Microcephaly has been related to dominant lamin B1 and B2 variants [60, 61]. In human neurons, where lamin B1 is more abundant, impaired nuclear envelope integrity can result in a spectrum of negative consequences that ultimately lead to microcephaly in affected individuals. These variants appear to interfere with the normal assembly of the nuclear envelope, altering the conformation of lamin B1, weakening the nuclear lamina, impairing nucleokinesis and leading to decreased neuronal survival [38].
Furthermore, the alteration of nuclear envelope integrity, as observed for these different variants, influences the survival and migration of neural precursors, leading to a marked reduction of brain size.
-
(d)
Neural tube closure defects
A LMNB1 polymorphic variant has been related to neural tube closure defects, including spina bifida and anencephaly [62]. This variant displays a decreased stability of the nuclear lamina, resulting in increased nuclear dysmorphology, altered cell cycle progression and premature cellular senescence.
-
(e)
Alzheimer’s disease
A marked reduction of lamins B expression has been reported in cortical neurons of Alzheimer’s disease (AD) patients [63]. In particular, in neurons of AD patients, pathological tau leads to the stabilization of actin filaments, disrupting the Linkers of the nucleoskeleton to the cytoskeleton (LINC) complex and decreasing lamin B. The consequences are the relaxation of constitutive heterochromatin and the activation of cell cycle in post-mitotic neurons, causing the death of the neurons [64]. The involvement of lamin B in AD onset suggests that this widespread neurodegenerative disorder may be considered as an aged-related laminopathy [64, 65].
Cancer
The aberrant localization or expression of lamins B has been related to cancer development, aggressiveness and metastasis, with lamin B being misregulated in a wide variety of cancers, probably because of the deformation of nuclear morphology caused by lamins B alterations [66] (Table 2). Indeed, cancer cells are subjected to changes of nuclear shape that favour the capability of cells to migrate and hence to promote metastasis. Given that lamins provide structural and mechanical strength as well as stiffness to the nucleus, their role in tumour development is now under growing and attentive examination, even if it is still under investigations whether lamins B misexpression modulate the ability of cancer cells to metastasize by altering the nucleus mechanical properties or rather by acting directly on cell proliferation, signalling, and differentiation.
It has been demonstrated that lamin B1 and lamin B2 are up- and down-regulated in several cancer cells and, accordingly, play different roles in cancer onset and development (Table 2). However, a particular interest should be addressed to lamin B1 expression, because there are some contradictory results when we take into account different types of tumour. While some study reported that increased lamin B1 expression is related to a more aggressive and invasive phenotype (in pancreatic and liver cancer [67, 68]) and importantly, that overexpression of lamin B1 is accompanied by a bad prognosis (in renal and gastric cancers [69, 70]), a decrease of lamin B expression has been also associated with greater aggressiveness and bad prognosis (in lung and breast cancer [71, 72]). These contradicting results suggest that the numerous functions that lamins B carry out in the cell may have different effects in cancers that arise from different cell types. Moreover, the interactions of lamin B with other binding proteins and signalling downstream components have been found in several tumour cells, contributing to the cancer development.
A possible involvement of lamin B in cancer onset could be related to the relationship between lamin B and oxidative stress, which is in turn connected with aggressiveness and metastasis [73]. In fact, it has been demonstrated that lamin B1 is overexpressed under oxidative stress conditions [30]. Another explanation of the role of lamin B in carcinogenesis could be that the overexpression of lamin B1 can hasten cell migration through the inhibition of the formation of a perinuclear actin rim that typically limits cell migration rate, as recently demonstrated [74].
However, further investigations to unravel the specific role of lamins B alterations in the different forms of cancer are needed, hence providing new insights in better understanding the cancerogenic processes regulated by variations of these type of lamins.
-
(a)
Pancreatic cancer
Aberrant expression of lamin B1 has been identified in pancreatic cancer cells and related to a more aggressive phenotype with worse prognosis [67]. For this reason, it has been suggested that the increase of lamin B1 could be considered as a novel biomarker for pancreatic cancer and as a therapeutic target [67]. On the contrary, the decrease of lamin B1 reduces the proliferation, invasion, and tumorigenicity of pancreatic cancer cells [67]. This is particularly interesting as there are several inhibitors of pre-lamin B1 processing at the level of farnesylation or methylation, which could be explored as modulators of lamin B1 levels.
-
(b)
Liver cancer
It has reported that lamin B1 expression increases in hepatocellular carcinoma (HCC) cells [68]. Moreover, it has been recently reported that also lamin B2 is involved in HCC cancer development promoting cell proliferation, migration, and invasion in HCC cell lines and in primary HCC cells [75].
-
(c)
Lung cancer
Recently, a link between lung cancer onset and lamins B has been demonstrated. Deletion of one copy of LMNB1 gene in mice and in human is related [72]. Lower levels of lamin B1 expression, indeed, in patients affected by lung cancer favour metastasis through epigenetic mechanism [72]. In particular, lamin B1 acts as onco-suppressor recruiting the polycomb repressive complex 2 (PRC2) and repressing genes involved in cell migration, including RET and p38 MAPK signalling axis components. From this perspective, lamin B1 plays a key role in lung cancer progression, providing a molecular link between altered nuclear morphology, aberrant epigenetic patterning, and malignant phenotype.
Furthermore, Zhang et al. reported that the overexpression of lamin B2 promotes migration of non-small cell lung cancer by increasing H3K9me2 level, thus eliciting E-cadherin gene silencing [76].
-
(d)
Breast cancer
In breast cancer, different studies have reported that both a decreased expression of lamin B1 [71] and an increase of lamin B2 [77] are related to bad outcome, confirming the importance to strictly maintain proper B-type lamin levels.
-
(e)
Neoplasms of the gastrointestinal tract
Decrease of lamin B1 expression is linked to different type of gastrointestinal tumours, including colon carcinomas, adenomas and gastric cancers [70, 78].
Lamin B receptor diseases
Another spectrum of pathologies connected to lamin B alterations involve mutations in the lamin B receptor (LBR). The LBR is an inner nuclear membrane protein that binds lamin B proteins (both lamin B1 and B2). Two different functions have been attributed to LBR. The nucleoplasmatic domain interacts with chromatin and has a structural function, whereas its transmembrane domains exhibit sterol reductase activity, a crucial function of the cholesterol synthesis metabolic pathway (Further details on LBR in the review of Nikolakaki et al. [79]).
Mutations of LBR have been related to two rare disease: Greenberg dysplasia and Pelger–Huet anomaly; the former is a lethal disease characterized by skeletal dysplasia, fetal hydrops, short limbs and abnormal chondro-osseous calcification [80], the latter is an autosomal dominant pathology, characterized by altered shape of nuclei in neutrophil and eosinophil granulocytes [81, 82], which is asymptomatic in the heterozygous state and causes variable degrees of epilepsy, developmental delay and skeletal abnormalities in homozygotes [82].
Conclusions and perspectives
Growing amount of evidence shows that B-type lamins play an important role in laminopathies and cancer. Besides the clear involvement of B-type lamins in CNS diseases and the fact that LMNA-linked disorders do not affect brain functionality, several aspects of lamin B-related pathophysiology highlight the specificity of B-type lamins in cellular and organism homeostasis with respect to A-type lamins. The pathogenesis of ADLD, the main lamin B-linked disease, has been so far linked to increased ROS levels and altered lipid metabolism leading to demyelination [83]. The association of lamin B2 with APL further supports a role for B-type lamins in lipid and adipose tissue metabolism. Yet, the molecular mechanisms can be recapitulated into a few pathways involving chromatin anchorage at specific sites (lamina-associated domains, LADs), a group of repressed sequences that could be reactivated in the presence of altered lamin B levels. Elevated ROS levels caused by increased lamin B1-Oct-1 interaction and altered nuclear dynamics has been also suggested as a pathogenetic pathway for ADLD and ADLD pathogenesis has been further linked to the onset of aberrant splicing events elicited by elevated lamin B1 levels [27, 51].
A clear input for further research that could also open therapeutic perspectives is the well corroborated evidence that lamin B amount is a determinant of protein functionality. In fact, while increased lamin B1 levels are linked to ADLD, reduced lamin B1 amount is associated with ageing processes. On the other hand, depending on the trigger, cellular senescence can be also linked to increased lamin B1 amount [30], a condition recently linked to sequestering of the DNA damage repair factor 53BP1 and altered DNA damage response [84].
Even in cancer cells, levels of B-type lamins are altered and a relationship between cancer aggressiveness and lamin amount can be established in a tumour-specific way. These observations imply that drugs affecting lamin B1 post-translational processing, including isoprenylcysteinecarboxyl methyltransferase (ICMT) or RCE1 inhibitors could be explored for their effect on lamin B stability in view of therapeutic approaches [85,86,87].
Availability of data and materials
Not applicable.
References
Burke B, Stewart CL (2013) The nuclear lamins: flexibility in function. Nat Rev Mol Cell Biol 14(1):13–24. https://doi.org/10.1038/nrm3488
Xie W, Burke B (2016) Lamins. Curr Biol 26(9):R348–R350. https://doi.org/10.1016/j.cub.2016.01.055
Chiarini F et al (2019) The cutting edge: The role of mTOR signaling in laminopathies. Int J Mol Sci. https://doi.org/10.3390/ijms20040847
Maske CP et al (2003) A carboxyl-terminal interaction of lamin B1 is dependent on the CAAX endoprotease Rce1 and carboxymethylation. J Cell Biol 162(7):1223–1232. https://doi.org/10.1083/jcb.200303113
Jung HJ et al (2013) Farnesylation of lamin B1 is important for retention of nuclear chromatin during neuronal migration. Proc Natl Acad Sci U S A 110(21):E1923–E1932. https://doi.org/10.1073/pnas.1303916110
Moir RD et al (2000) Nuclear lamins A and B1: different pathways of assembly during nuclear envelope formation in living cells. J Cell Biol 151(6):1155–1168. https://doi.org/10.1083/jcb.151.6.1155
Shimi T et al (2008) The A- and B-type nuclear lamin networks: microdomains involved in chromatin organization and transcription. Genes Dev 22(24):3409–3421. https://doi.org/10.1101/gad.1735208
Dechat T, Adam SA, Goldman RD (2009) Nuclear lamins and chromatin: when structure meets function. Adv Enzyme Regul 49(1):157–166. https://doi.org/10.1016/j.advenzreg.2008.12.003
Shimi T et al (2015) Structural organization of nuclear lamins A, C, B1, and B2 revealed by superresolution microscopy. Mol Biol Cell 26(22):4075–4086. https://doi.org/10.1091/mbc.E15-07-0461
Nmezi B et al (2019) Concentric organization of A- and B-type lamins predicts their distinct roles in the spatial organization and stability of the nuclear lamina. Proc Natl Acad Sci U S A 116(10):4307–4315. https://doi.org/10.1073/pnas.1810070116
Figueiras E et al (2019) Phasor-assisted nanoscopy reveals differences in the spatial organization of major nuclear lamina proteins. Biochim Biophys Acta Mol Cell Res 1866(12):118530. https://doi.org/10.1016/j.bbamcr.2019.118530
Dechat T et al (2010) Nuclear lamins. Cold Spring Harb Perspect Biol 2(11):a000547. https://doi.org/10.1101/cshperspect.a000547
Coffinier C et al (2011) Deficiencies in lamin B1 and lamin B2 cause neurodevelopmental defects and distinct nuclear shape abnormalities in neurons. Mol Biol Cell 22(23):4683–4693. https://doi.org/10.1091/mbc.E11-06-0504
Yang SH et al (2011) An absence of both lamin B1 and lamin B2 in keratinocytes has no effect on cell proliferation or the development of skin and hair. Hum Mol Genet 20(18):3537–3544. https://doi.org/10.1093/hmg/ddr266
Lammerding J et al (2006) Lamins A and C but not lamin B1 regulate nuclear mechanics. J Biol Chem 281(35):25768–25780. https://doi.org/10.1074/jbc.M513511200
Wintner O et al (2020) A unified linear viscoelastic model of the cell nucleus defines the mechanical contributions of lamins and chromatin. Adv Sci (Weinh) 7(8):1901222. https://doi.org/10.1002/advs.201901222
Maraldi NM et al (2011) Laminopathies and lamin-associated signaling pathways. J Cell Biochem 112(4):979–992. https://doi.org/10.1002/jcb.22992
Simon DN, Wilson KL (2013) Partners and post-translational modifications of nuclear lamins. Chromosoma 122(1–2):13–31. https://doi.org/10.1007/s00412-013-0399-8
Ahanger SH et al (2021) Distinct nuclear compartment-associated genome architecture in the developing mammalian brain. Nat Neurosci 24(9):1235–1242. https://doi.org/10.1038/s41593-021-00879-5
Meuleman W et al (2013) Constitutive nuclear lamina-genome interactions are highly conserved and associated with A/T-rich sequence. Genome Res 23(2):270–280. https://doi.org/10.1101/gr.141028.112
Zheng X, Kim Y, Zheng Y (2015) Identification of lamin B-regulated chromatin regions based on chromatin landscapes. Mol Biol Cell 26(14):2685–2697. https://doi.org/10.1091/mbc.E15-04-0210
Campisi J, d’Adda di Fagagna F (2007) Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 8(9):729–740. https://doi.org/10.1038/nrm2233
Shimi T et al (2011) The role of nuclear lamin B1 in cell proliferation and senescence. Genes Dev 25(24):2579–2593. https://doi.org/10.1101/gad.179515.111
Freund A et al (2012) Lamin B1 loss is a senescence-associated biomarker. Mol Biol Cell 23(11):2066–2075. https://doi.org/10.1091/mbc.E11-10-0884
Sadaie M et al (2013) Redistribution of the Lamin B1 genomic binding profile affects rearrangement of heterochromatic domains and SAHF formation during senescence. Genes Dev 27(16):1800–1808. https://doi.org/10.1101/gad.217281.113
Shah PP et al (2013) Lamin B1 depletion in senescent cells triggers large-scale changes in gene expression and the chromatin landscape. Genes Dev 27(16):1787–1799. https://doi.org/10.1101/gad.223834.113
Malhas AN, Lee CF, Vaux DJ (2009) Lamin B1 controls oxidative stress responses via Oct-1. J Cell Biol 184(1):45–55. https://doi.org/10.1083/jcb.200804155
Dreesen O et al (2013) Lamin B1 fluctuations have differential effects on cellular proliferation and senescence. J Cell Biol 200(5):605–617. https://doi.org/10.1083/jcb.201206121
Jung HJ et al (2012) Regulation of prelamin A but not lamin C by miR-9, a brain-specific microRNA. Proc Natl Acad Sci U S A 109(7):E423–E431. https://doi.org/10.1073/pnas.1111780109
Barascu A et al (2012) Oxidative stress induces an ATM-independent senescence pathway through p38 MAPK-mediated lamin B1 accumulation. EMBO J 31(5):1080–1094. https://doi.org/10.1038/emboj.2011.492
Camps J et al (2014) Loss of lamin B1 results in prolongation of S phase and decondensation of chromosome territories. FASEB J 28(8):3423–3434. https://doi.org/10.1096/fj.14-250456
Butin-Israeli V et al (2015) Role of lamin b1 in chromatin instability. Mol Cell Biol 35(5):884–898. https://doi.org/10.1128/MCB.01145-14
Liu NA et al (2015) Regulation of homologous recombinational repair by lamin B1 in radiation-induced DNA damage. FASEB J 29(6):2514–2525. https://doi.org/10.1096/fj.14-265546
Vergnes L et al (2004) Lamin B1 is required for mouse development and nuclear integrity. Proc Natl Acad Sci U S A 101(28):10428–10433. https://doi.org/10.1073/pnas.0401424101
Giacomini C et al (2016) Lamin B1 protein is required for dendrite development in primary mouse cortical neurons. Mol Biol Cell 27(1):35–47. https://doi.org/10.1091/mbc.E15-05-0307
Yoon BC et al (2012) Local translation of extranuclear lamin B promotes axon maintenance. Cell 148(4):752–764. https://doi.org/10.1016/j.cell.2011.11.064
Young SG et al (2012) Understanding the roles of nuclear A- and B-type lamins in brain development. J Biol Chem 287(20):16103–16110. https://doi.org/10.1074/jbc.R112.354407
Chen NY et al (2019) An absence of lamin B1 in migrating neurons causes nuclear membrane ruptures and cell death. Proc Natl Acad Sci U S A 116(51):25870–25879. https://doi.org/10.1073/pnas.1917225116
Bin Imtiaz MK et al (2021) Declining lamin B1 expression mediates age-dependent decreases of hippocampal stem cell activity. Cell Stem Cell 28(5):967–977. https://doi.org/10.1016/j.stem.2021.01.015
Heng MY et al (2013) Lamin B1 mediates cell-autonomous neuropathology in a leukodystrophy mouse model. J Clin Invest 123(6):2719–2729. https://doi.org/10.1172/JCI66737
Rolyan H et al (2015) Defects of lipid synthesis are linked to the age-dependent demyelination caused by lamin B1 overexpression. J Neurosci 35(34):12002–12017. https://doi.org/10.1523/JNEUROSCI.1668-15.2015
Padiath QS et al (2006) Lamin B1 duplications cause autosomal dominant leukodystrophy. Nat Genet 38(10):1114–1123. https://doi.org/10.1038/ng1872
Giorgio E et al (2015) A large genomic deletion leads to enhancer adoption by the lamin B1 gene: a second path to autosomal dominant adult-onset demyelinating leukodystrophy (ADLD). Hum Mol Genet 24(11):3143–3154. https://doi.org/10.1093/hmg/ddv065
Lin ST, Fu YH (2009) miR-23 regulation of lamin B1 is crucial for oligodendrocyte development and myelination. Dis Model Mech 2(3–4):178–188. https://doi.org/10.1242/dmm.001065
Dugas JC et al (2006) Functional genomic analysis of oligodendrocyte differentiation. J Neurosci 26(43):10967–10983. https://doi.org/10.1523/JNEUROSCI.2572-06.2006
Inoue K (2019) Pelizaeus-Merzbacher disease: molecular and cellular pathologies and associated phenotypes. Adv Exp Med Biol 1190:201–216. https://doi.org/10.1007/978-981-32-9636-7_13
Bauer J et al (2002) Endoplasmic reticulum stress in PLP-overexpressing transgenic rats: gray matter oligodendrocytes are more vulnerable than white matter oligodendrocytes. J Neuropathol Exp Neurol 61(1):12–22. https://doi.org/10.1093/jnen/61.1.12
Bartoletti-Stella A et al (2015) Messenger RNA processing is altered in autosomal dominant leukodystrophy. Hum Mol Genet 24(10):2746–2756. https://doi.org/10.1093/hmg/ddv034
Kim Y et al (2011) Mouse B-type lamins are required for proper organogenesis but not by embryonic stem cells. Science 334(6063):1706–1710. https://doi.org/10.1126/science.1211222
Ratti S et al (2021) Cell signaling pathways in autosomal-dominant leukodystrophy (ADLD): the intriguing role of the astrocytes. Cell Mol Life Sci 78(6):2781–2795. https://doi.org/10.1007/s00018-020-03661-1
Columbaro M et al (2013) Oct-1 recruitment to the nuclear envelope in adult-onset autosomal dominant leukodystrophy. Biochim Biophys Acta 1832(3):411–420. https://doi.org/10.1016/j.bbadis.2012.12.006
Ratti S et al (2021) Lamin B1 accumulation’s effects on autosomal dominant Leukodystrophy (ADLD): induction of reactivity in the astrocytes. Cells. https://doi.org/10.3390/cells10102566
Giorgio E et al (2019) Allele-specific silencing as treatment for gene duplication disorders: proof-of-principle in autosomal dominant leukodystrophy. Brain 142(7):1905–1920. https://doi.org/10.1093/brain/awz139
Hegele RA et al (2006) Sequencing of the reannotated LMNB2 gene reveals novel mutations in patients with acquired partial lipodystrophy. Am J Hum Genet 79(2):383–389. https://doi.org/10.1086/505885
Gao J et al (2012) A Chinese patient with acquired partial lipodystrophy caused by a novel mutation with LMNB2 gene. J Pediatr Endocrinol Metab 25(3–4):375–377. https://doi.org/10.1515/jpem-2012-0007
Zhang J et al (2011) A human iPSC model of Hutchinson Gilford Progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell 8(1):31–45. https://doi.org/10.1016/j.stem.2010.12.002
Coffinier C et al (2010) Abnormal development of the cerebral cortex and cerebellum in the setting of lamin B2 deficiency. Proc Natl Acad Sci U S A 107(11):5076–5081. https://doi.org/10.1073/pnas.0908790107
Damiano JA et al (2015) Mutation of the nuclear lamin gene LMNB2 in progressive myoclonus epilepsy with early ataxia. Hum Mol Genet 24(16):4483–4490. https://doi.org/10.1093/hmg/ddv171
Hutchison CJ (2012) B-type lamins and their elusive roles in metazoan cell proliferation and senescence. EMBO J 31(5):1058–1059. https://doi.org/10.1038/emboj.2012.39
Cristofoli F et al (2020) De novo variants in LMNB1 cause pronounced syndromic microcephaly and disruption of nuclear envelope integrity. Am J Hum Genet 107(4):753–762. https://doi.org/10.1016/j.ajhg.2020.08.015
Parry DA et al (2021) Heterozygous lamin B1 and lamin B2 variants cause primary microcephaly and define a novel laminopathy. Genet Med 23(2):408–414. https://doi.org/10.1038/s41436-020-00980-3
De Castro SC et al (2012) Lamin b1 polymorphism influences morphology of the nuclear envelope, cell cycle progression, and risk of neural tube defects in mice. PLoS Genet 8(11):e1003059. https://doi.org/10.1371/journal.pgen.1003059
Frost B et al (2014) Tau promotes neurodegeneration through global chromatin relaxation. Nat Neurosci 17(3):357–366. https://doi.org/10.1038/nn.3639
Frost B, Bardai FH, Feany MB (2016) Lamin dysfunction mediates neurodegeneration in tauopathies. Curr Biol 26(1):129–136. https://doi.org/10.1016/j.cub.2015.11.039
Frost B (2016) Alzheimer’s disease: an acquired neurodegenerative laminopathy. Nucleus 7(3):275–283. https://doi.org/10.1080/19491034.2016.1183859
Sakthivel KM, Sehgal P (2016) A novel role of lamins from genetic disease to cancer biomarkers. Oncol Rev 10(2):309. https://doi.org/10.4081/oncol.2016.309
Li L et al (2013) Lamin B1 is a novel therapeutic target of betulinic acid in pancreatic cancer. Clin Cancer Res 19(17):4651–4661. https://doi.org/10.1158/1078-0432.CCR-12-3630
Sun S et al (2010) Circulating Lamin B1 (LMNB1) biomarker detects early stages of liver cancer in patients. J Proteome Res 9(1):70–78. https://doi.org/10.1021/pr9002118
Radspieler MM et al (2019) Lamin-B1 is a senescence-associated biomarker in clear-cell renal cell carcinoma. Oncol Lett 18(3):2654–2660. https://doi.org/10.3892/ol.2019.10593
Yu ZY et al (2020) Lamin B1 deficiency promotes malignancy and predicts poor prognosis in gastric cancer. Neoplasma 67(6):1303–1313. https://doi.org/10.4149/neo_2020_200109N33
Wazir U et al (2013) The clinicopathological significance of lamin A/C, lamin B1 and lamin B receptor mRNA expression in human breast cancer. Cell Mol Biol Lett 18(4):595–611. https://doi.org/10.2478/s11658-013-0109-9
Jia Y et al (2019) Lamin B1 loss promotes lung cancer development and metastasis by epigenetic derepression of RET. J Exp Med 216(6):1377–1395. https://doi.org/10.1084/jem.20181394
Costa A, Scholer-Dahirel A, Mechta-Grigoriou F (2014) The role of reactive oxygen species and metabolism on cancer cells and their microenvironment. Semin Cancer Biol 25:23–32. https://doi.org/10.1016/j.semcancer.2013.12.007
Fracchia A et al (2020) Increased Lamin B1 levels promote cell migration by altering perinuclear actin organization. Cells. https://doi.org/10.3390/cells9102161
Li XN, Yang H, Yang T (2020) miR-122 inhibits hepatocarcinoma cell progression by targeting LMNB2. Oncol Res 28(1):41–49. https://doi.org/10.3727/096504019X15615433287579
Zhang MY et al (2020) Lamin B2 promotes the malignant phenotype of non-small cell lung cancer cells by upregulating dimethylation of histone 3 lysine 9. Exp Cell Res 393(2):112090. https://doi.org/10.1016/j.yexcr.2020.112090
Zhao CC et al (2021) Lamin B2 promotes the progression of triple negative breast cancer via mediating cell proliferation and apoptosis. Biosci Rep. https://doi.org/10.1042/BSR20203874
Moss SF et al (1999) Decreased and aberrant nuclear lamin expression in gastrointestinal tract neoplasms. Gut 45(5):723–729. https://doi.org/10.1136/gut.45.5.723
Nikolakaki E, Mylonis I, Giannakouros T (2017) Lamin B receptor: interplay between structure function and localization. Cells. https://doi.org/10.3390/cells6030028
Waterham HR et al (2003) Autosomal recessive HEM/Greenberg skeletal dysplasia is caused by 3 beta-hydroxysterol delta 14-reductase deficiency due to mutations in the lamin B receptor gene. Am J Hum Genet 72(4):1013–1017. https://doi.org/10.1086/373938
Schreiber KH, Kennedy BK (2013) When lamins go bad: nuclear structure and disease. Cell 152(6):1365–1375. https://doi.org/10.1016/j.cell.2013.02.015
Oosterwijk JC et al (2003) Congenital abnormalities reported in Pelger-Huet homozygosity as compared to Greenberg/HEM dysplasia: highly variable expression of allelic phenotypes. J Med Genet 40(12):937–941. https://doi.org/10.1136/jmg.40.12.937
Padiath QS (2019) Autosomal dominant leukodystrophy: a disease of the nuclear lamina. Front Cell Dev Biol 7:41. https://doi.org/10.3389/fcell.2019.00041
Etourneaud L et al (2021) Lamin B1 sequesters 53BP1 to control its recruitment to DNA damage. Sci Adv. https://doi.org/10.1126/sciadv.abb3799
Chelsky D, Sobotka C, O’Neill CL (1989) Lamin B methylation and assembly into the nuclear envelope. J Biol Chem 264(13):7637–7643
Malhas A et al (2007) Defects in lamin B1 expression or processing affect interphase chromosome position and gene expression. J Cell Biol 176(5):593–603. https://doi.org/10.1083/jcb.200607054
Lau HY et al (2014) An improved isoprenylcysteine carboxylmethyltransferase inhibitor induces cancer cell death and attenuates tumor growth in vivo. Cancer Biol Ther 15(9):1280–1291. https://doi.org/10.4161/cbt.29692
Dong CH et al (2021) LMNB2 promotes the progression of colorectal cancer by silencing p21 expression. Cell Death Dis 12(4):331. https://doi.org/10.1038/s41419-021-03602-1
Funding
The work was supported by: Ministero dell’Istruzione, dell’Università e della Ricerca-PRIN 2017 (to LM and to CE); Fondazione Cassa di Risparmio Bologna (to SR) and Intesa San Paolo Foundation (to LC).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the writing and editing of the article.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Evangelisti, C., Rusciano, I., Mongiorgi, S. et al. The wide and growing range of lamin B-related diseases: from laminopathies to cancer. Cell. Mol. Life Sci. 79, 126 (2022). https://doi.org/10.1007/s00018-021-04084-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-021-04084-2