Abstract
Monoclonal antibody (mAb) therapy has successfully been introduced as treatment of several lymphomas and leukemias. However, solid tumors reduce the efficacy of mAb therapy because of an immune-suppressive tumor micro-environment (TME), which hampers activation of effector immune cells. Pro-inflammatory cytokine therapy may counteract immune suppression in the TME and increase mAb efficacy, but untargeted pro-inflammatory cytokine therapy is limited by severe off-target toxicity and a short half-life of cytokines. Antibody-cytokine fusion proteins, also referred to as immunocytokines, provide a solution to either issue, as the antibody both acts as local delivery platform and increases half-life. The antibody can furthermore bridge local cytotoxic immune cells, like macrophages and natural killer cells with tumor cells, which can be eliminated after effector cells are activated via the cytokine. Currently, a variety of different antibody formats as well as a handful of cytokine payloads are used to generate immunocytokines. However, many potential formats and payloads are still left unexplored. In this review, we describe current antibody formats and cytokine moieties that are used for the development of immunocytokines, and highlight several immunocytokines in (pre-)clinical studies. Furthermore, potential future routes of development are proposed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Following the first description of monoclonal antibodies (mAbs) in 1975 [1], it took only eleven years to develop the first FDA-approved mAb-based immunotherapy. The drug named Muromonab was used to prevent transplanted kidney rejection by targeting CD3 on T cells [2]. Unfortunately, a plethora of side-effects was associated with this therapy, including but not limited to cytokine release syndrome (CRS), central nervous system complications and thrombosis, which ultimately resulted in voluntary withdrawal from the US market by its manufacturer [3]. Some side-effects have been attributed to the murine nature of the mAb. In some cases, anaphylaxis was induced. Additionally, efficiency of these antibodies was reduced, because injection of murine antibodies led to the development of human-anti-mouse-antibodies (HAMAs). As such, the murine origin of mAbs became a major obstacle to achieve efficient new mAb therapies [4]. To overcome this, chimeric, humanized or fully human mAbs have been developed [5]. In chimeric mAbs, the constant regions are of human origin, while in humanized antibodies, the framework regions of the variable regions are also human.
As a result, several effective mAb therapies have been developed, including anti-CD20 mAb therapies to treat several B-cell malignancies and autoimmune diseases (e.g. Rituximab, Ofatumumab) and checkpoint inhibitors (e.g. Pembrolizumab (αPD1), Ipilimumab (αCTLA-4) [6,7,8,9]. Nonetheless, other mAb cancer therapies are not effective as a mono-therapy [7]. A possible solution is the combination of mAb therapy with other cancer therapies, such as co-treatment with radio- or chemotherapy. Traditionally, both therapies were administered separately, but nowadays also radionuclide-antibody-conjugates (RACs) and antibody–drug-conjugates (ADCs) are increasingly developed [10, 11]. Furthermore, antibodies have the ability to engage the immune system. They can activate the complement pathway, but additionally can bridge cytotoxic effector cells with cancer cells, through their Fc tail [12]. Unfortunately, many tumors have an immunosuppressive tumor micro-environment (TME) [13, 14], which dampens effective activation of the immune system. As such, combining mAb therapy with agents that focus on changing the tumor micro-environment (i.e. antibody-cytokine-conjugates) may also represent an attractive way to improve efficacy of antibody therapy, and is the focus of this review.
The immune-suppressive tumor micro-environment
Solid tumors establish their immunosuppressive TME via a variety of mechanisms. A large part of the immune suppression is mediated through tumor-associated regulatory immune cells such as tumor-associated macrophages (TAMs), tumor-associated neutrophils (TANs), FoxP3 + regulatory T cells (Tregs), and myeloid-derived suppressor cells (MDSCs) [15, 16]. In the TME, tumor-associated regulatory immune cells secrete various immunosuppressive cytokines, such as interleukin 10 (IL-10) and transforming growth factor beta (TGF-β), which inhibit immune cell activation. Additionally, they express immune-suppressive surface molecules such as programmed death ligand 1 (PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), which also induce inhibitory signaling in immune cells [17, 18].
Cytokines with a pro-inflammatory action may counteract immunosuppression in the TME. Several cytokines have been identified as suitable candidates. For instance, IL-2 is important for activation, differentiation and maintenance of cytotoxic effector cells, such as cytotoxic T cells (CTLs), natural killer (NK) cells and macrophages, but can also directly act on tumor cells by inducing apoptosis [19, 20]. Recombinant IL-2 monotherapies have already been tried in patients with metastatic melanoma or metastatic renal carcinoma in the eighties with mild success [21, 22]. Only some patients benefited due to the toxicity that occurred at higher doses, limiting the efficacy of the therapeutic dose. Treatment of metastatic renal cell carcinoma and melanoma with systemic IL-2 therapy specifically increased the number of tumor-infiltrating lymphocytes (TILs) [23,24,25].
Additionally, IL-12 is a heterodimeric cytokine, composed out of p35 and p40 subunits, and is involved in communication between innate and adaptive immune responses [26]. IL-12 is primarily secreted by antigen-presenting cells after infection. It stimulates interferon gamma (IFN-γ) production by T and NK cells, and shifts CD4+ T cell differentiation toward a Th1 phenotype [27]. MHC-I presentation as well as NK cell and T cell proliferation is stimulated as well, whereas Tregs are inhibited by IL-12 [26, 28]. Moreover, IL-12 also has anti-angiogenic activities [29].
Tumor necrosis factor alpha (TNF-α) is also a robust regulator of immune cells [30]. It binds to the TNF receptor 2 (TNF-R2), that is mostly expressed by immune cells, and promotes migration of CTLs and NK cells [31, 32]. In addition, TNF-α can also be directly cytotoxic to tumor cells by binding to TNF-R1, which is expressed on most tissues. TNF-α acts on endothelial cells to increase vascular permeability and mediates hemorrhagic necrosis [33], which can eradicate the tumor mass by damaging tumor-associated vasculature. Furthermore, TNF-α is able to stimulate upregulation of cell adhesion molecules such as E-selectin, ICAM-1 and VCAM-1 by endothelial cells [34]. This favors influx of pro-inflammatory leukocytes to the cancer site [35].
Thus, systemic pro-inflammatory cytokine therapy represents an attractive option to activate the immune system, but, is unfortunately notorious for its toxicity, as delivery is untargeted [36, 37]. Hence, by conjugating cytokines to mAbs, efficacy and safety of both therapies might be increased. On the one hand toxicity of cytokine treatment may be lowered, as cytokines are specifically delivered at the cancer site. On the other hand mAb therapy may become more effective as effector immune cells will be simultaneously activated by both cytokine and antibody [38, 39]. Antibody-cytokine-conjugates, otherwise known as armed-antibodies or immunocytokines, were originally developed as a vehicle for the local delivery of lymphotoxin or TNF-α to solid tumors in an attempt to increase their half-life without dramatically increasing toxicity [40, 41]. Over the years, various innovations in both the fields of antibody engineering and cytokine therapy have allowed the development of immunocytokines with a plethora of antibody formats and an increased number of their potential armaments. This wide variety of antibody formats and payload moieties allows for great flexibility in designing an immunocytokine to treat a specific disease or to overcome a specific problem.
In this review we provide an overview of the antibody formats and cytokine moieties that are currently used for the development of immunocytokines. Furthermore, potential future routes of innovation are addressed.
Antibody formats
Intact IgG
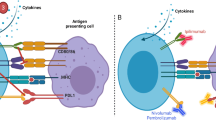
Immunocytokine formats can generally be divided into two categories: i.e. intact IgG or based on antibody fragments. Using intact IgG as delivery platform has several advantages (Table 1). Antibodies contain two light- and two heavy chains (Fig. 1A). Both light- and heavy chains have a variable domain (VL and VH), which together allow specific antigen binding. The light chain contains a single constant region (CL), while the heavy chain comprises three constant regions (CH1, CH2 and CH3). The first advantage of this format is the half-life of an IgG antibody, which is around 21 days in serum. This relatively long half-life is mediated by its large size (150 kDa), which reduces renal clearance rates [42, 43]. Additionally, antibodies of the IgG isotype are able to bind to the neonatal Fc receptor (FcRn), which prevents degradation of IgG in endothelial lysosomes and recycles them back into circulation [44]. Second, the dual variable regions of intact IgG grants immunocytokines based on this format high avidity for its specific target, which allows for high rates of retention at the site of interest [45]. High retention rates combined with long half-life provide IgG antibodies with a large window of activity. Third, IgG antibodies bind to Fc gamma receptors (FcγR’s), which are among others expressed on monocytes, macrophages and NK cells. This can lead to antibody-dependent cellular cytotoxicity (ADCC) or antibody-dependent cellular phagocytosis (ADCP) of the tumor cell, depending on the effector cell. Internalization of the immune complex also leads to degradation of the antibodies, reducing half-life. Monocytes and macrophages mostly kill via phagocytosis, while NK cells secrete perforins and granzymes, resulting in apoptosis [46, 47].
Schematic representation of antibody formats used in immunocytokines. Variable domain light chain (VL), constant region light chain (CL), variable domain heavy chain (VH), constant region heavy chain (CH), immunoglobulin G (IgG), antigen-binding fragment (F(ab′)), single chain variable fragment (scFv), fragment crystallizable region (Fc)
However, not all FcγR’s are activating receptors. FcγRIIb, for example, contains an immunoreceptor tyrosine-based inhibitory motif (ITIM) and is commonly seen as an immune regulator for IgG-mediated activation of various immune cells [48]. In addition, FcγRIIb can also be expressed in a soluble form or cleaved from its transmembrane domain, allowing it to regulate immune responses through binding of immune complexes [49, 50].
Furthermore, high density opsonization of a tumor cell with IgG will attract C1q proteins, starting the complement cascade, which can lead to cell lysis via the membrane attack complex or phagocytosis by effector cells [51,52,53]. However, using an intact IgG antibody format also has some limitations. Its large size may hamper extravasation from vasculature and penetration into the tumor mass, preventing efficient localization at the site of cancer [54]. Additionally, if normal cells have (low) expression of the target antigen, binding of intact IgG may lead to ADCC or ADCP in healthy tissues. This, combined with the long half-life and high tissue retention may lead to higher toxicity in patients [55].
Fragment-based formats
To improve tumor penetration, other antibody-based formats have been developed, such as F(ab’) fragments (Fig. 1B). Composed of the variable domains attached to the CH1 and CL subregions of the heavy and light chains, F(ab′) is essentially one truncated arm of an intact IgG antibody. The advantage of F(ab′) fragments is their smaller size (48 kDa) compared to the intact IgG format (150 kDa), which allows for increased extravasation and tumor penetration [56]. However, because the F(ab′) format only has a single antigen-binding domain, the avidity for its target is decreased. This disadvantage is not present in F(ab′)2 fragments, which are a variant in which two F(ab′) fragments are connected through a linker, restoring high avidity and retention at the site of interest (Fig. 1C). Furthermore, these fragments do not contain an Fc region, preventing binding of activating and inhibitory FcɣR’s, as well as FcRn, decreasing their half-life significantly. Decreased half-life in addition to the lack of Fc-mediated functions strongly reduce the off-target toxicity of these fragments.
The smallest fragment-based format is a single-chain variable fragment (scFv), consisting of the two variable domains of the heavy and light chain connected together with a short linker (Fig. 1D). scFv-based fragments have a molecular weight of approximately 27 kDa and are about six times smaller compared to intact IgG. This allows easier extravasation from the vasculature and higher penetration into the tumor mass. However, the small size and lack of Fc tail results in rapid renal clearance and a short half-life [42, 57, 58]. Like F(ab′) fragments, scFv’s only have a single antigen binding site, leading to decreased avidity to its target and a shorter retention time at the site of interest [42, 59, 60]. As such, this format is better suited for payload delivery than to induce tumor killing. Similar to the F(ab′) fragments, a short half-life in addition to the lack of any Fc-mediated functions strongly reduce off-target toxicity.
To overcome the decreased avidity of scFv’s, Dia- and Tribodies have been developed (Fig. 1E, F) [61, 62]. A Diabody is a dimeric variant of the scFv, while the Tribody combines the Diabody format with an additional F(ab′) fragment. With more antigen-binding moieties, Dia- and Tribodies each have increased avidity while still possessing increased tumor penetration capacity compared to intact IgG formats. The scFv-Fc format was developed to increase the capabilities of scFv to induce ADCC and ADCP (Fig. 1G). scFv-Fc molecules are composed of two scFv fragments that are directly attached to the CH2 and CH3 subregions of IgG heavy chains. As such, it is in essence an intact IgG format, which misses the constant regions of the fab arms, CL and CH1. This grants increased avidity to its target compared to scFv, restores Fc-mediated tumor-killing capabilities and increases half-life [63]. Nonetheless, restoration of these functions comes at the cost of decreased tumor penetration due to a larger size (106 kDa) and increased off-target toxicity if the target is expressed in healthy tissue.
Immunocytokines in (pre-)clinical trials
There is a good number of interesting immunocytokines currently in clinical development (Table 2). In the next paragraphs, we will briefly discuss the key players currently in (pre-) clinical development.
IL-2 is a key cytokine for the differentiation and activation of cytotoxic effector cells, including CTLs, NK cells and macrophages. However, systemic treatment with IL-2 results in severe toxicity [36].
Immunocytokines with an IL-2 moiety increased leukocyte infiltration within the TME of murine tumors. Strong NK cell induced antitumor immunity was observed, leading to necrosis and decreased tumor cell proliferation without the high rates of toxicity associated with untargeted IL-2 therapy [64,65,66,67].
Several IL-2 immunocytokines have therefore been tested in clinical studies, with promising results. One of these immunocytokines is hu14.18-IL-2. This immunocytokine is based on the hu14.18 antibody clone, which is a humanized antibody against GD2, a tumor antigen which is overexpressed in neuroblastoma as well as melanoma and highly restricted on normal tissues with the exception of the immune-privileged cerebellum and peripheral nerves. It has an intact IgG format, with IL-2 molecules genetically linked at the C-terminal of both heavy chains (Fig. 2A). Fusing two IL-2 molecules has the advantage of increased immune modulation compared to immunocytokines with only one IL-2 molecule. The intended method of action for this immunocytokine is to locally activate NK cells and CTLs through the IL-2 payload, and induce ADCC via the functional IgG Fc region. The activation of CTLs makes this immunocytokine an excellent candidate for combination with other therapies aimed at inducing adaptive immune responses, such as vaccination or immune checkpoint inhibitors [68, 69]. In a phase I/II clinical trial, increased numbers of NK cells were found in circulation [70]. Isolated patient NK cells (before treatment), were more effective in inducing ADCC in the presence of hu14.18-IL-2. Moreover, significantly less side effects of hu14.18-IL-2 treatment were observed when compared with untargeted IL-2 therapy, which was confirmed in another clinical study [71]. Consequently, significantly higher dosage can be given, which likely improves efficacy of IL-2 treatment [72]. Intravenous treatment with hu14.18-IL-2 after complete resection of stage III or IV melanoma resulted in prolonged tumor-free survival of patients with high risk of recurrence [73].
Additionally, a variant (IC35) was developed due to dose-limiting toxicity that was experienced when the hu14.18-IL2 immunocytokine was used clinically. In this variant, the payload was genetically fused to the light chains instead of the C-terminal of the heavy chains using the same linker as the original [74]. It was anticipated that this would result in decreased interaction of IL-2 payloads with their receptors due to increased steric hindrance of the antibody, which, in turn, would decrease off-target toxicity. Pre-clinical data showed that IC35 had decreased affinity for βɣIL2R as well as reduced ability to induce IL-2-mediated proliferation in multiple cell lines, supporting the hypothesis.
L19-IL-2, which targets extradomain-B (EDB; i.e. alternatively spliced fibronectin) represents another widely tested IL-2 immunocytokine. EDB is primarily expressed in embryos and on tumor vasculature [75, 76]. Choosing a TAA that is not expressed on the tumor cells itself, but on the tumor vasculature changes the therapy dynamics, allowing for a different approach for tumor elimination. L19-IL-2 has a non-covalent homodimeric scFv format, allowing for more efficient penetration into the TME as well as a genetic fusion of two IL-2 molecules via (SSSSG)3 linkers (Fig. 2B). Similar to hu14.18-IL-2, L19-IL-2 can locally activate NK cells and CTLs via the IL-2 payload, but lacks the capacity to induce ADCC due to the missing Fc region. Nonetheless, without a functional Fc region, off-target toxicity of the immunocytokine is also reduced. NK cells were the main effector cells for induction of tumor cell killing in a pre-clinical pancreatic tumor model [64]. Furthermore, L19-IL-2 monotherapy induced tumor growth delay of 8 days in a Ramos lymphoma xenograft model, whereas combination with Rituximab (anti-CD20 mAb) therapy resulted in completely tumor eradication [77]. No irreversible toxicity was developed when patients with metastatic renal carcinoma were treated with L19-IL-2 in a Phase I/II trial [78]. L19-IL-2 treatment, in combination with chemotherapy (dacarbazine) lead to an increased number of partial responses and decreased progressive disease in patients with metastatic melanoma [79, 80].
L19-IL-2 has also been extensively tested in combination with L19-TNF, an immunocytokine that is composed of the same scFv targeting EBD, but genetically linked with a different cytokine, i.e. TNF, using (SSSSG)3 linkers (Fig. 2C). L19 scFv does not homodimerize, but trimerizes due to the trimerization motifs present in the TNF payload. As a result, a functional, trimerized immunocytokine has three scFv moieties and one active TNF homotrimer. This increases binding avidity and decreases off-target toxicity through increased retention at the tumor site. Combining L19-IL-2 and L19-TNF treatments resulted in complete remission in murine F9 carcinoma models, while the individual monotherapies were unable to completely eradicate tumors [81]. Similar results were found in a murine myeloma model, in which the combination treatment eradicated 58% of tumors [82]. The combination of L19-IL-2 and L19-TNF was also tested in a phase II clinical trial, in which this treatment showed remarkable efficacy in eradicating non-injected melanoma lesions (7/13 lesions) [83].
Other TNF-based immunocytokines have been developed, but have not reached clinical stage yet. Nevertheless, pre-clinical results of the scFv23/TNF immunocytokine were encouraging. The scFv23 fragment-based immunocytokine, with a Diabody format, targets HER-2/neu, which is overexpressed in approximately 30% of breast cancers (Fig. 2D). The scFv23/TNF is genetically fused to a single TNF monomer using a GGGGS linker, similar to the L19-TNF immunocytokine. As such, it also trimerizes at the target site. The putative mode of action is sensitization of HER-2/neu overexpressing breast cancer cells to TNF-therapy and subsequent induction of apoptosis through the direct effects of TNF on tumor cells. HER-2/neu overexpressing breast cancer cells are typically resistant to the cytotoxic effects of TNF. However, treatment with scFv23/TNF increased expression of the TNFR1 by 5- to sevenfold and effectively sensitized the cells to TNF-induced apoptosis [84]. This principle was confirmed when scFv23/TNF was tested on HER-2/neu overexpressing pancreatic cancer cell lines in combination with various chemotherapeutics. Cell lines that were originally resistant to TNF-induced apoptosis, were killed in equal measure by scFv23/TNF monotherapy when compared to conventional chemotherapeutic agents like 5-fluorouracil or etoposide [85].
Additionally, IL-12-based immunocytokines have also been evaluated in clinical studies. The NHS-IL-12 immunocytokine is based on intact IgG and has two IL-12 heterodimers genetically fused to its heavy chain C-terminals via a flexible (GGGGS)3 linker. The variable domains target DNA/histone complexes, which are not present extracellularly during homeostasis (Fig. 2E). By contrast, DNA/histone complexes are often found at sites of necrosis such as a necrotic tumor core. When tumor necrosis was induced via local irradiation in a human rhabdomyosarcoma xenograft mouse model, increased localization of NHS-IL-12 was observed at the site of the tumor [86]. Since IL-12 plays an important role in crosstalk between the innate and the adaptive immune system, the intended mechanism of action for this immunocytokine is to stimulate antigen-presenting cells, CD8+ T cells and NK cells, thereby initiating robust antitumor adaptive immune responses. When MC38 tumor-bearing mice were treated with various doses of NHS-IL-12, increased serum IFN-ɣ levels were found as well as splenic DC maturation [87]. Additionally, treatment of subcutaneous LLC, B16 and MC38 tumors in athymic mice led to significant growth reduction in all cases. A phase I clinical trial showed that all dose-limiting responses were transient and levels of circulating activated NK and NKT cells were increased after treatment [88]. A phase I/II clinical study investigating the combination treatment with M7824, an anti-PD-L1 antibody carrying a TGF-β trap (the extracellular domain of human TGFβRII), has been initiated (NCT04303117).
Current challenges and routes of future antibody development
Improvement of immunocytokines can be achieved in two ways, i.e. innovation of the antibody moiety or of the payload. Adaptation of the antibody might represent the easier route, since many advancements or concepts in the field of antibody engineering have already been made. Any attempts to improve the efficacy or to decrease side-effects of mAb therapy can practically be directly translated into the field of immunocytokines.
Point mutations
Simple amino acid mutations were shown to have significant effects on the efficacy and/or pharmacokinetics of antibodies [89]. However, they have not been applied in the field of immunocytokines yet. On the one hand, point mutations can be used to increase the effector function of immunocytokines, such as immune cell and complement activation via the Fc tail. FcyRIII is involved in the initiation of ADCP or ADCC by monocytes and macrophages or NK cells. Via extensive mapping of the binding site of IgG Fc receptors, amino acid mutations were identified that increased binding of FcyRIIIA, leading to a twofold increase in ADCC [90]. The triple mutation Ser293Asp/Ala330Leu/Ile332Glu increased both ADCP and ADCC [91]. The same mutation also enhanced the efficiency of NK cell-mediated serial killing [92]. The initial step of the classical complement cascade is binding of C1q to the CH2 region of IgG [93]. Residue mutations that increase the binding of this first step may therefore prove beneficial to the efficacy of the antibody and immunocytokines. As examples, Lys326Ala/Glu333Ala or Lys326Met/Glu333Ser mutations increased complement-dependent-cytotoxicity (CDC) without interfering with the antibody’s ability to induce ADCC [94]. Another application of residue mutations is optimization of the FcRn-binding region. One such mutation increased the half-life by 3,2- or 3,onefold of bevacizumab (IgG1) or cetuximab (IgG1) respectively [95]. A longer half-life increases the therapeutic potential of therapies and reduces the cost of treatment. However, making amino acid mutations in the FcRn-binding region of antibodies might have adverse effects on its capabilities to mediate ADCC, which also has to be addressed [96].
On the other hand, when Fc tail-initiated functions in fact cause harm, amino acid mutations can also be used to reduce off-target toxicity. Over-activation of immune cells or activation of immune cells in healthy tissues can result in significant damage, precluding clinical development. Additional off-target effects can include depletion of immune cells. Either tumor-infiltrating immune cells are purposefully targeted by bispecific antibodies for activation or immune cells express the same target receptors present on tumor cells (e.g. CD20 on B cells). In either case, opsonization of immune cells can mark them for Fc-mediated killing via NK cells or macrophages, resulting in depletion and immune-compromised patients. A potential solution is to use an antibody format that lacks the Fc region, such as scFv and F(ab′) fragments, decreasing the half-life as well as retention at the site of disease, which may diminish side effects. Silencing of Fc regions by residue mutations represents an alternative approach, which retains the long half-life of FcRn-binding antibodies, but abolishes the possible off-target toxicity mediated by activation of immune cells or complement via the Fc region. The most commonly used mutations in this approach are the Leu234Ala and Leu235Ala (LALA) mutations, which for instance abrogated binding of the IgG1 isotype OKT3 antibodies to FcyRI, FcyRIIa, and FcyRIIIa [97,98,99].
Glyco-engineering
It is also possible to change the pharmacokinetics of immunocytokines using glyco-engineering, which is regulating post-translational glycosylation of proteins as opposed to changing the genetic code in the case of residue mutations. Increased formation of a bisecting GlcNAc structure in N-glycans, through the expression of b(1,4)-N-acetylglucosaminyltransferase-III in IgG1-producing cells, increased FcyRIII-mediated ADCC by 10- to 20-fold due to increased affinity of the antibody’s Fc region [100, 101]. Increased galactosylation of antibodies enhanced the capabilities of IgG1 antibodies to induce CDC [102]. The same effect was observed for IgG3 antibodies, while increased galactosylation did not affect the ability to induce CDC of IgG2 and IgG4 isotypes [103]. Several amino acid mutations also had effect on the glycosylation profile, which proved effective in the silencing of ADCC and/or CDC capabilities of antibodies. Substitution of asparagine at position 297 by either alanine, glutamine, or glycine removed this glycosylation site and dramatically decreased the ADCC and CDC capabilities of IgG1 and IgG3 antibodies [104,105,106,107].
Bi-specific formats
Bi-specific antibodies (BsAbs) can be used to recruit effector cells populations, which are normally not effectively engaged by IgG-based antibody therapy. For instance, CTLs (CD8 T cells) are not involved in antibody therapy as they lack Fc receptors. As CD8 cells have great capacity to kill tumor cells, they could significantly add to antibody-based therapy efficacy. T cell activating BsAbs generally target the CD3 receptor in combination with a tumor-associated antigen (TAA) [108,109,110]. Bispecific T cell Engagers (BiTEs), Dual-Affinity Re-Targeting proteins (DARTs) and DuoBody® formats have been described. The BiTE format simply consists of two scFv’s that target different antigens, linked together as a single chain (Fig. 3A). The DART format was designed to overcome some of the limitations of BiTEs. It consists of two separate proteins, in which one contains the VH against antigen 1 and the VL of antigen 2, while the other contains the VH against antigen 2 and the VL of antigen 1 (Fig. 3B). This format allows the heterodimerization that naturally occurs in IgG to form a functional bispecific antibody. The DuoBody® format is an intact IgG format that consists of a complete IgG molecule with two different antigen binding domains (Fig. 3C). This is achieved by generating two separate parental antibodies and performing a Fab-arm exchange using DuoBody® technology [111].
Alternatively, neutrophils are the most abundant cytotoxic effector cells in the circulation. It was demonstrated by us and others that engaging the IgA Fc receptor (FcαRI) on neutrophils effectively induced neutrophil recruitment and tumor cell killing [112,113,114]. However, IgA has shorter half-life (± 6–7 days) compared to IgG. Recently a DuoBody® IgG1 BsAb was developed of which one of the variable domains binds a tumor antigen, while the other variable domain binds FcαRI [115]. This BsAb was able to recruit neutrophils (via the FcαRI arm) and NK cells and macrophages (via the IgG1 Fc tail) as effector cells in a B16F10 melanoma mouse model [115]. Similar effects can be achieved by genetically fusing IgG and IgA Fc regions or by creating a chimeric variant of the two regions (IgGA) [116,117,118].
Bi-specific formats can also be used to further increase activation of immune cells sensitive to FcɣR stimulation. NK cells, for example, can bind IgG, but are further activated by agonistic antibodies that activate a co-receptor [119]. Primary targets for agonistic activation by antibodies include NKG2D, 2B4 and NKp30 [120,121,122]. BsAb immunocytokines may be used to further activate effector cells of interest. However, bispecific formats can also be used to increase specificity for the tumor by targeting two tumor antigens instead of one. Several tumor antigens that are used for mAb therapy are overexpressed in tumors, but also expressed in healthy tissues, leading to off-target toxicity. Targeting immunocytokines toward two antigens that are overexpressed in tumor tissue may reduce off-target toxicity [123].
Tumor-associated antigen expression in patients
The translation of target-specificity from pre-clinical to clinical models is a serious challenge. Commonly, pre-clinical models are used that either strongly overexpress a (TAA) or express a human TAA that is not found in the used animals. In the first instance, the expression of TAA might be artificially inflated compared to clinical tumors, and care must be taken to ensure that the therapy is still sufficiently specific to prevent off-target toxicity when the TAA expression is lower. Similarly, in the case of expressing a human TAA in an animal model, unwanted off-target toxicity may be missed as the target is not expressed in normal tissues. Additionally, using a human TAA in an animal model will increase the immunogenicity of the tumor, possibly confounding the therapeutic effect in patients.
Current challenges and routes of future payload development
Alternative payloads
Most cytokine payloads that are currently tested in a clinical setting are pro-inflammatory, aiming to activate or stimulate immune cells [45]. Alternatively, anti-inflammatory cytokines may be considered. Previously, a Cetuximab-based immunocytokine with IL-10 as payload (CmAb(IL-10)2) was developed, which is classically considered as an anti-inflammatory cytokine [124]. Interestingly, treatment with CmAb(IL-10)2 in several murine tumor models, resulted in superior tumor growth reduction compared to treatment with Cetuximab alone. CmAb(IL-10)2 mediated its effects by hindering IFN-ɣ-induced cell death of tumor-infiltrating CD8+ T cells. This effect was even more pronounced when CmAb(IL-10)2 treatment was combined with checkpoint inhibitors such as anti- PD-L1 and anti-CTLA-4 mAbs.
Although counterintuitive, these data demonstrate that anti-inflammatory cytokines can also (indirectly) induce pro-inflammatory effects. This concept was previously proposed in a review in which it was described that the inflammatory effect of cytokines depends on multiple factors, such as dosage, target cells and experimental models. As such, cytokines cannot be defined in a black-and-white manner like pro- or anti-inflammatory, and it might therefore be worthwhile to further investigate immunocytokines with classically anti-inflammatory cytokines as payload [125].
Another cytokine subset worth looking into is the chemokine family. Chemokines form a complex signaling network with their receptors that allow for the chemotaxis of specific cell subsets toward a gradient of chemokines [126]. In spite of a lot of promiscuity in the chemokine network, there are some chemokines that specifically (or rather preferentially) attract cell subsets. Using immunochemokines to create a local chemokine gradient in the tumor can therefore attract specific immune cells of interest. This may allow for specific skewing of immune responses, which could be of great value for immunotherapy. Recent findings have shown that the tumor manipulates the chemokine milieu to its own advantage by attracting immune cells that are converted into tumor-associated regulatory immune cells [127]. Using chemokines as payload might provide an opportunity to rebalance the chemokine milieu and the immune cell infiltrate.
However, several aspects need to be taken into account when developing immunochemokines. First, the N-terminus of chemokines plays a key role in the interaction with their respective receptors [128, 129]. Fusing chemokines to an antibody might interfere with that interaction. Similarly, chemokines are usually presented on glycosaminoglycans (GAGs) to circulating immune cells by endothelial cells [130, 131]. Having a chemokine presented via an antibody instead of GAGs may potentially severely impair the ability of chemokine receptors to recognize and bind the chemokine moiety of an immunochemokine. Both of these problems might be solved by attaching the chemokine via a cleavable linker. After cleavage, the chemokine is released from the antibody moiety. This allows the chemokine to be presented on GAGs, while also freeing up its N-terminus for interaction with chemokine receptors. Cleavable linkers can be cleaved by matrix metalloproteinases (MMPs) secreted specifically by tumor cells [132], which prevents systemic release of the chemokine. After cleavage, the antibody moiety of the immunochemokine is still able to bind target receptors and mediate tumor killing through its Fc tail.
Potency matched dual-cytokines
A handful of immunocytokines have already proven their efficacy in clinical trials when used as a mono-therapy [73, 79, 80, 88]. However, some immunocytokines, which carry multiple different payloads have been developed. These dual-cytokine immunocytokines have currently only been tested in in vitro and pre-clinical models [133,134,135,136,137,138]. With the cytokine repertoire’s scale and variety, many combinations can be made which could have synergetic effects in vivo. Nonetheless, currently cytokine combinations have been limited to IL-2/TNF, IL-2/TNFR2 and IL-2/TRAIL, which all show remarkable promise.
When designing dual-cytokine immunocytokines, it is important to realize that most cytokines have a different effective dose. When both cytokines are fused to an antibody, the amount of cytokines will be averaged, which may result in an ineffective dose of one of the cytokines. Alternatively, too much of one cytokine may lead to toxicity. Changing the affinity of one of the cytokines for their receptor to match the other cytokine may overcome this issue [135].
Reduction of payload-mediated toxicity
Generally, off-target toxicity is mediated through the Fc region of the antibody moiety. However, toxicity might also be mediated through the payload of an immunocytokine. To overcome this challenge, multiple methods can be used.
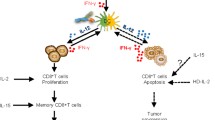
One method to reduce payload-mediated toxicity can be applied if the active form of the cytokine in question is a multimer. In the case of the L19-TNF immunocytokine for example, TNF-α is only active in its homo-trimeric form [139]. Developing an immunocytokine with a TNF-α homo-trimer as payload is possible, but might induce toxicity when injected systemically. Developing an immunocytokine with only a monomer as payload may represent a solution, as the active TNF-α homo-trimer is only formed when the immunocytokine localizes to the tumor (Fig. 4) [81]. A similar approach may be used for an IL-12 payload. IL-12 is a heterodimer composed of a p35 and p40 subunit. Constructing two immunocytokines with the p35 and p40 payloads respectively will ensure that the active form of IL-12 is only formed at the site of localization [140].
A relatively recent development in toxicity reduction is protease-cleavable epitope masking. The basic principle behind this technique is to link an epitope-masking molecule to the antibody via a linker that is cleavable by MMPs, which are specifically or highly expressed at tumor sites [141]. Using this technique, immune-active antibody moieties, such as Fc regions, binding domains, receptor agonistic variable domains or payloads, can be kept in an inactive state until the antibody reaches the tumor [142,143,144]. Once arrived, tumor-specific proteases cleave the linker, releasing the masking molecule from the antibody and converting it to an active state.
Similar to including point mutations to reduce antibody-mediated toxicity, this technique has also been successfully applied to cytokines [145, 146]. These mutated cytokines are referred to as Activation-by-Targeting Cytokines (AcTakines). By mutating amino acid residues involved in receptor-ligand interactions, the affinity of the cytokine for its receptor can be reduced, which leads to decreased signaling at lower concentrations, such as in the circulation or in non-targeted tissues. However, increased concentrations of the immuncytokine in tumors are sufficiently high to enhance avidity to a level which allows efficient signaling.
Alternatively, small molecule inhibitors have been used to temporarily inhibit the respective signaling pathway of the cytokine receptor [147]. This attenuates payload-mediated toxicity in the circulation. As the immunocytokine localizes at the tumor site, its payload becomes active once small molecules have been cleared from the body, reducing off-target toxicity.
Conclusion
Immunocytokines provide a robust and versatile platform for the treatment of cancer. The flexibility of both antibody platforms and payload moieties ensures that immunocytokines can be developed to suit the specific characteristics of each type of cancer. Additionally, there is room for significant innovation and improvement of all aspects of immunocytokines, which may increase their efficacy even further. Thus, we anticipate exciting new developments in this field in the next few years.
Data availability
All relevant data can be found in the original references.
References
Kohler G, Milstein C (1975) Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256(5517):495–497
Todd PA, Brogden RN (1989) Muromonab CD3. A review of its pharmacology and therapeutic potential. Drugs 37(6):871–899
Sgro C (1995) Side-effects of a monoclonal antibody, muromonab CD3/orthoclone OKT3: bibliographic review. Toxicology 105(1):23–29
Vaickus L, Foon KA (1991) Overview of monoclonal antibodies in the diagnosis and therapy of cancer. Cancer Invest 9(2):195–209
LoBuglio AF et al (1989) Mouse/human chimeric monoclonal antibody in man: kinetics and immune response. Proc Natl Acad Sci U S A 86(11):4220–4224
Salles G et al (2017) Rituximab in B cell hematologic malignancies: a review of 20 years of clinical experience. Adv Ther 34(10):2232–2273
Mazzarella L, Guida A, Curigliano G (2018) Cetuximab for treating non-small cell lung cancer. Expert Opin Biol Ther 18(4):483–493
McDermott J, Jimeno A (2015) Pembrolizumab: PD-1 inhibition as a therapeutic strategy in cancer. Drugs Today (Barc) 51(1):7–20
Joshi SS, Maron SB, Catenacci DV (2018) Pembrolizumab for treatment of advanced gastric and gastroesophageal junction adenocarcinoma. Future Oncol 14(5):417–430
Hafeez U et al (2020) Antibody-drug conjugates for cancer therapy. Molecules 25(20):4764
Peltek OO et al (2019) Current outlook on radionuclide delivery systems: from design consideration to translation into clinics. J Nanobiotechnology 17(1):90
Goydel RS, Rader C (2021) Antibody-based cancer therapy. Oncogene 40(21):3655–3664
Tang H, Qiao J, Fu YX (2016) Immunotherapy and tumor microenvironment. Cancer Lett 370(1):85–90
Frankel T, Lanfranca MP, Zou W (2017) The role of tumor microenvironment in cancer immunotherapy. Adv Exp Med Biol 1036:51–64
Fridlender ZG et al (2009) Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 16(3):183–194
Zhou J et al (2020) Tumor-associated macrophages: recent insights and therapies. Front Oncol 10:188
Kuang DM et al (2009) Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J Exp Med 206(6):1327–1337
Ruffell B et al (2014) Macrophage IL-10 blocks CD8+ T cell-dependent responses to chemotherapy by suppressing IL-12 expression in intratumoral dendritic cells. Cancer Cell 26(5):623–637
Malek TR, Castro I (2010) Interleukin-2 receptor signaling: at the interface between tolerance and immunity. Immunity 33(2):153–165
Waters RS et al (2018) The effects of interleukin-2 on immune response regulation. Math Med Biol 35(1):79–119
Atkins MB et al (1999) High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol 17(7):2105–2116
Klapper JA et al (2008) High-dose interleukin-2 for the treatment of metastatic renal cell carcinoma : a retrospective analysis of response and survival in patients treated in the surgery branch at the National Cancer Institute between 1986 and 2006. Cancer 113(2):293–301
Rosenberg SA et al (1998) Durability of complete responses in patients with metastatic cancer treated with high-dose interleukin-2: identification of the antigens mediating response. Ann Surg 228(3):307–319
Rosenberg SA (2014) IL-2: the first effective immunotherapy for human cancer. J Immunol 192(12):5451–5458
Shaker MA, Younes HM (2009) Interleukin-2: evaluation of routes of administration and current delivery systems in cancer therapy. J Pharm Sci 98(7):2268–2298
Trinchieri G et al (1992) Natural killer cell stimulatory factor (NKSF) or interleukin-12 is a key regulator of immune response and inflammation. Prog Growth Factor Res 4(4):355–368
Tugues S et al (2015) New insights into IL-12-mediated tumor suppression. Cell Death Differ 22(2):237–246
Trinchieri G (2003) Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat Rev Immunol 3(2):133–146
Voest EE et al (1995) Inhibition of angiogenesis in vivo by interleukin 12. J Natl Cancer Inst 87(8):581–586
Pasche N, Neri D (2012) Immunocytokines: a novel class of potent armed antibodies. Drug Discov Today 17(11–12):583–590
Probst P et al (2017) Sarcoma eradication by doxorubicin and targeted TNF relies upon CD8(+) T cell recognition of a retroviral antigen. Cancer Res 77(13):3644–3654
Almishri W et al (2016) TNFalpha augments cytokine-induced NK Cell IFNgamma production through TNFR2. J Innate Immun 8(6):617–629
Borsi L et al (2003) Selective targeted delivery of TNFalpha to tumor blood vessels. Blood 102(13):4384–4392
Fukumura D et al (1995) Tumor necrosis factor alpha-induced leukocyte adhesion in normal and tumor vessels: effect of tumor type, transplantation site, and host strain. Cancer Res 55(21):4824–4829
Halin C et al (2003) Synergistic therapeutic effects of a tumor targeting antibody fragment, fused to interleukin 12 and to tumor necrosis factor alpha. Cancer Res 63(12):3202–3210
Baldo BA (2014) Side effects of cytokines approved for therapy. Drug Saf 37(11):921–943
Leonard JP et al (1997) Effects of single-dose interleukin-12 exposure on interleukin-12-associated toxicity and interferon-gamma production. Blood 90(7):2541–2548
Halin C et al (2002) Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat Biotechnol 20(3):264–269
Ronca R et al (2009) Delivering cytokines at tumor site: the immunocytokine-conjugated anti-EDB-fibronectin antibody case. Immunobiology 214(9–10):800–810
Gillies SD et al (1991) Expression of genetically engineered immunoconjugates of lymphotoxin and a chimeric anti-ganglioside GD2 antibody. Hybridoma 10(3):347–356
Hoogenboom HR, Volckaert G, Raus JC (1991) Construction and expression of antibody-tumor necrosis factor fusion proteins. Mol Immunol 28(9):1027–1037
Thurber GM, Schmidt MM, Wittrup KD (2008) Factors determining antibody distribution in tumors. Trends Pharmacol Sci 29(2):57–61
Li Z, Krippendorff BF, Shah DK (2017) Influence of molecular size on the clearance of antibody fragments. Pharm Res 34(10):2131–2141
Pyzik M et al (2019) The neonatal Fc receptor (FcRn): a misnomer? Front Immunol 10:1540
Hutmacher C, Neri D (2019) Antibody-cytokine fusion proteins: biopharmaceuticals with immunomodulatory properties for cancer therapy. Adv Drug Deliv Rev 141:67–91
Paul S, Lal G (2017) The molecular mechanism of natural killer cells function and its importance in cancer immunotherapy. Front Immunol 8:1124
Gul N et al (2014) Macrophages eliminate circulating tumor cells after monoclonal antibody therapy. J Clin Invest 124(2):812–823
Pricop L et al (2001) Differential modulation of stimulatory and inhibitory Fc gamma receptors on human monocytes by Th1 and Th2 cytokines. J Immunol 166(1):531–537
Esposito-Farese ME et al (1995) Membrane and soluble Fc gamma RII/III modulate the antigen-presenting capacity of murine dendritic epidermal Langerhans cells for IgG-complexed antigens. J Immunol 155(4):1725–1736
Sautes C et al (1991) Soluble Fc gamma receptors II (Fc gamma RII) are generated by cleavage of membrane Fc gamma RII. Eur J Immunol 21(1):231–234
Heemskerk N, van Egmond M (2018) Monoclonal antibody-mediated killing of tumour cells by neutrophils. Eur J Clin Invest 48(Suppl 2):e12962
Daha NA et al (2011) Complement activation by (auto-) antibodies. Mol Immunol 48(14):1656–1665
Sarma JV, Ward PA (2011) The complement system. Cell Tissue Res 343(1):227–235
Strohl WR (2015) Fusion proteins for half-life extension of biologics as a strategy to make biobetters. BioDrugs 29(4):215–239
Gillies SD et al (2002) Improved circulating half-life and efficacy of an antibody-interleukin 2 immunocytokine based on reduced intracellular proteolysis. Clin Cancer Res 8(1):210–216
Read T et al (2015) Label-free Fab and Fc affinity/avidity profiling of the antibody complex half-life for polyclonal and monoclonal efficacy screening. Anal Bioanal Chem 407(24):7349–7357
Yokota T et al (1992) Rapid tumor penetration of a single-chain Fv and comparison with other immunoglobulin forms. Cancer Res 52(12):3402–3408
Schneider EL et al (2016) Approach for half-life extension of small antibody fragments that does not affect tissue uptake. Bioconjug Chem 27(10):2534–2539
Batra SK et al (2002) Pharmacokinetics and biodistribution of genetically engineered antibodies. Curr Opin Biotechnol 13(6):603–608
Pavlinkova G et al (2000) Pharmacokinetics and biodistribution of a light-chain-shuffled CC49 single-chain Fv antibody construct. Cancer Immunol Immunother 49(4–5):267–275
Adams GP et al (2006) Avidity-mediated enhancement of in vivo tumor targeting by single-chain Fv dimers. Clin Cancer Res 12(5):1599–1605
Wu AM et al (1996) Tumor localization of anti-CEA single-chain Fvs: improved targeting by non-covalent dimers. Immunotechnology 2(1):21–36
Unverdorben F et al (2016) Pharmacokinetic properties of IgG and various Fc fusion proteins in mice. MAbs 8(1):120–128
Wagner K et al (2008) The targeted immunocytokine L19-IL2 efficiently inhibits the growth of orthotopic pancreatic cancer. Clin Cancer Res 14(15):4951–4960
Neal ZC et al (2004) Enhanced activity of hu14.18-IL2 immunocytokine against murine NXS2 neuroblastoma when combined with interleukin 2 therapy. Clin Cancer Res 10(14):4839–4847
Gutbrodt KL et al (2013) Antibody-based delivery of interleukin-2 to neovasculature has potent activity against acute myeloid leukemia. Sci Transl Med 5(201):201ra118
Lode HN et al (2000) Amplification of T cell-mediated immune responses by antibody-cytokine fusion proteins. Immunol Invest 29(2):117–120
Aiken TJ et al (2022) Mechanism of effective combination radio-immunotherapy against 9464D-GD2, an immunologically cold murine neuroblastoma. J Immunother Cancer 10(5):e004834
Baniel CC et al (2020) In situ vaccine plus checkpoint blockade induces memory humoral response. Front Immunol 11:1610
King DM et al (2004) Phase I clinical trial of the immunocytokine EMD 273063 in melanoma patients. J Clin Oncol 22(22):4463–4473
Ribas A et al (2009) Phase I/II open-label study of the biologic effects of the interleukin-2 immunocytokine EMD 273063 (hu1418-IL2) in patients with metastatic malignant melanoma. J Transl Med 7:68
Albertini MR et al (2012) Phase II trial of hu14.18-IL2 for patients with metastatic melanoma. Cancer Immunol Immunother 61(12):2261–2271
Albertini MR et al (2018) Pilot trial of the hu14.18-IL2 immunocytokine in patients with completely resectable recurrent stage III or stage IV melanoma. Cancer Immunol Immunother 67(10):1647–1658
Perez Horta Z et al (2019) Human and murine IL2 receptors differentially respond to the human-IL2 component of immunocytokines. Oncoimmunology 8(6):e1238538
Fukuda T et al (2002) Mice lacking the EDB segment of fibronectin develop normally but exhibit reduced cell growth and fibronectin matrix assembly in vitro. Cancer Res 62(19):5603–5610
Kaspar M, Zardi L, Neri D (2006) Fibronectin as target for tumor therapy. Int J Cancer 118(6):1331–1339
Schliemann C et al (2009) Complete eradication of human B cell lymphoma xenografts using rituximab in combination with the immunocytokine L19-IL2. Blood 113(10):2275–2283
Johannsen M et al (2010) The tumour-targeting human L19-IL2 immunocytokine: preclinical safety studies, phase I clinical trial in patients with solid tumours and expansion into patients with advanced renal cell carcinoma. Eur J Cancer 46(16):2926–2935
Eigentler TK et al (2011) A dose-escalation and signal-generating study of the immunocytokine L19-IL2 in combination with dacarbazine for the therapy of patients with metastatic melanoma. Clin Cancer Res 17(24):7732–7742
Weide B et al (2019) A phase II study of the L19IL2 immunocytokine in combination with dacarbazine in advanced metastatic melanoma patients. Cancer Immunol Immunother 68(9):1547–1559
Schwager K et al (2013) The immunocytokine L19-IL2 eradicates cancer when used in combination with CTLA-4 blockade or with L19-TNF. J Invest Dermatol 133(3):751–758
Menssen HD et al (2018) Antibody-based delivery of tumor necrosis factor (L19-TNFalpha) and interleukin-2 (L19-IL2) to tumor-associated blood vessels has potent immunological and anticancer activity in the syngeneic J558L BALB/c myeloma model. J Cancer Res Clin Oncol 144(3):499–507
Danielli R et al (2015) Intralesional administration of L19-IL2/L19-TNF in stage III or stage IVM1a melanoma patients: results of a phase II study. Cancer Immunol Immunother 64(8):999–1009
Lyu MA, Rosenblum MG (2005) The immunocytokine scFv23/TNF sensitizes HER-2/neu-overexpressing SKBR-3 cells to tumor necrosis factor (TNF) via up-regulation of TNF receptor-1. Mol Cancer Ther 4(8):1205–1213
Lyu MA, Kurzrock R, Rosenblum MG (2008) The immunocytokine scFv23/TNF targeting HER-2/neu induces synergistic cytotoxic effects with 5-fluorouracil in TNF-resistant pancreatic cancer cell lines. Biochem Pharmacol 75(4):836–846
Eckert F et al (2016) Enhanced binding of necrosis-targeting immunocytokine NHS-IL12 after local tumour irradiation in murine xenograft models. Cancer Immunol Immunother 65(8):1003–1013
Fallon J et al (2014) The immunocytokine NHS-IL12 as a potential cancer therapeutic. Oncotarget 5(7):1869–1884
Strauss J et al (2019) First-in-human phase I trial of a tumor-targeted cytokine (NHS-IL12) in subjects with metastatic solid tumors. Clin Cancer Res 25(1):99–109
Saunders KO (2019) Conceptual approaches to modulating antibody effector functions and circulation half-life. Front Immunol 10:1296
Shields RL et al (2001) High resolution mapping of the binding site on human IgG1 for Fc gamma RI, Fc gamma RII, Fc gamma RIII, and FcRn and design of IgG1 variants with improved binding to the Fc gamma R. J Biol Chem 276(9):6591–6604
Lazar GA et al (2006) Engineered antibody Fc variants with enhanced effector function. Proc Natl Acad Sci U S A 103(11):4005–4010
Romain G et al (2014) Antibody Fc engineering improves frequency and promotes kinetic boosting of serial killing mediated by NK cells. Blood 124(22):3241–3249
Hughes-Jones NC, Gardner B (1979) Reaction between the isolated globular sub-units of the complement component C1q and IgG-complexes. Mol Immunol 16(9):697–701
Idusogie EE et al (2001) Engineered antibodies with increased activity to recruit complement. J Immunol 166(4):2571–2575
Zalevsky J et al (2010) Enhanced antibody half-life improves in vivo activity. Nat Biotechnol 28(2):157–159
Ghetie V et al (1997) Increasing the serum persistence of an IgG fragment by random mutagenesis. Nat Biotechnol 15(7):637–640
Wines BD et al (2000) The IgG Fc contains distinct Fc receptor (FcR) binding sites: the leukocyte receptors Fc gamma RI and Fc gamma RIIa bind to a region in the Fc distinct from that recognized by neonatal FcR and protein A. J Immunol 164(10):5313–5318
Xu D et al (2000) In vitro characterization of five humanized OKT3 effector function variant antibodies. Cell Immunol 200(1):16–26
Lund J et al (1991) Human Fc gamma RI and Fc gamma RII interact with distinct but overlapping sites on human IgG. J Immunol 147(8):2657–2662
Umana P et al (1999) Engineered glycoforms of an antineuroblastoma IgG1 with optimized antibody-dependent cellular cytotoxic activity. Nat Biotechnol 17(2):176–180
Davies J et al (2001) Expression of GnTIII in a recombinant anti-CD20 CHO production cell line: expression of antibodies with altered glycoforms leads to an increase in ADCC through higher affinity for FC gamma RIII. Biotechnol Bioeng 74(4):288–294
Dekkers G et al (2017) Decoding the human immunoglobulin G-glycan repertoire reveals a spectrum of Fc-receptor- and complement-mediated-effector activities. Front Immunol 8:877
Peschke B et al (2017) Fc-Galactosylation of human immunoglobulin gamma isotypes improves C1q binding and enhances complement-dependent cytotoxicity. Front Immunol 8:646
Tao MH, Morrison SL (1989) Studies of aglycosylated chimeric mouse-human IgG. Role of carbohydrate in the structure and effector functions mediated by the human IgG constant region. J Immunol 143(8):2595–2601
Kanda Y et al (2007) Comparison of biological activity among nonfucosylated therapeutic IgG1 antibodies with three different N-linked Fc oligosaccharides: the high-mannose, hybrid, and complex types. Glycobiology 17(1):104–118
Mimura Y et al (2001) Role of oligosaccharide residues of IgG1-Fc in Fc gamma RIIb binding. J Biol Chem 276(49):45539–45547
Lund J et al (1990) A protein structural change in aglycosylated IgG3 correlates with loss of huFc gamma R1 and huFc gamma R111 binding and/or activation. Mol Immunol 27(11):1145–1153
Feldmann A et al (2012) Novel humanized and highly efficient bispecific antibodies mediate killing of prostate stem cell antigen-expressing tumor cells by CD8+ and CD4+ T cells. J Immunol 189(6):3249–3259
Loffler A et al (2003) Efficient elimination of chronic lymphocytic leukaemia B cells by autologous T cells with a bispecific anti-CD19/anti-CD3 single-chain antibody construct. Leukemia 17(5):900–909
Wu MR et al (2015) B7H6-specific bispecific T cell engagers lead to tumor elimination and host antitumor immunity. J Immunol 194(11):5305–5311
Labrijn AF et al (2013) Efficient generation of stable bispecific IgG1 by controlled Fab-arm exchange. Proc Natl Acad Sci U S A 110(13):5145–5150
Gruijs M, Sewnath CAN, Egmond MV (2021) Therapeutic exploitation of neutrophils to fight cancer. Semin Immunol, 101581
van der Steen L et al (2009) Immunoglobulin A: Fc(alpha)RI interactions induce neutrophil migration through release of leukotriene B4. Gastroenterology 137(6):2018-29.e1–3
Valerius T et al (1997) FcalphaRI (CD89) as a novel trigger molecule for bispecific antibody therapy. Blood 90(11):4485–4492
Heemskerk N et al. (2021) Augmented antibody-based anticancer therapeutics boost neutrophil cytotoxicity. J Clin Invest 131(6)
Kelton W et al (2014) IgGA: a “cross-isotype” engineered human Fc antibody domain that displays both IgG-like and IgA-like effector functions. Chem Biol 21(12):1603–1609
Borrok MJ et al (2015) Enhancement of antibody-dependent cell-mediated cytotoxicity by endowing IgG with FcalphaRI (CD89) binding. MAbs 7(4):743–751
Chintalacharuvu KR et al (2001) Hybrid IgA2/IgG1 antibodies with tailor-made effector functions. Clin Immunol 101(1):21–31
Chester C, Fritsch K, Kohrt HE (2015) Natural killer cell immunomodulation: targeting activating, inhibitory, and co-stimulatory receptor signaling for cancer immunotherapy. Front Immunol 6:601
Semeraro M et al (2015) Clinical impact of the NKp30/B7-H6 axis in high-risk neuroblastoma patients. Sci Transl Med 7(283):283ra55
Garcia-Cuesta EM et al (2015) NKG2D is a key receptor for recognition of bladder cancer cells by IL-2-activated NK cells and BCG promotes NK cell activation. Front Immunol 6:284
Nakajima H et al (1999) Activating interactions in human NK cell recognition: the role of 2B4-CD48. Eur J Immunol 29(5):1676–1683
Huang S et al (2020) Bispecific antibodies targeting dual tumor-associated antigens in cancer therapy. J Cancer Res Clin Oncol 146(12):3111–3122
Qiao J et al (2019) Targeting tumors with IL-10 prevents dendritic cell-mediated CD8(+) T cell apoptosis. Cancer Cell 35(6):901-915.e4
Cavaillon JM (2001) Pro- versus anti-inflammatory cytokines: myth or reality. Cell Mol Biol (Noisy-le-grand) 47(4):695–702
Griffith JW, Sokol CL, Luster AD (2014) Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu Rev Immunol 32:659–702
Mollica Poeta V et al (2019) Chemokines and chemokine receptors: new targets for cancer immunotherapy. Front Immunol 10:379
Szpakowska M et al (2018) Different contributions of chemokine N-terminal features attest to a different ligand binding mode and a bias towards activation of ACKR3/CXCR7 compared with CXCR4 and CXCR3. Br J Pharmacol 175(9):1419–1438
Kleist AB et al (2016) New paradigms in chemokine receptor signal transduction: moving beyond the two-site model. Biochem Pharmacol 114:53–68
Graham GJ, Handel TM, Proudfoot AEI (2019) Leukocyte adhesion: reconceptualizing chemokine presentation by glycosaminoglycans. Trends Immunol 40(6):472–481
Crijns H, Vanheule V, Proost P (2020) Targeting chemokine-glycosaminoglycan interactions to inhibit inflammation. Front Immunol 11:483
Mohammadi M et al (2020) A highly potential cleavable linker for tumor targeting antibody-chemokines. J Biomol Struct Dyn 40:1–11
De Luca R et al (2019) A novel fully-human potency-matched dual cytokine-antibody fusion protein targets carbonic anhydrase IX in renal cell carcinomas. Front Oncol 9:1228
De Luca R et al (2018) A novel dual-cytokine-antibody fusion protein for the treatment of CD38-positive malignancies. Protein Eng Des Sel 31(5):173–179
De Luca R, Neri D (2018) Potentiation of PD-L1 blockade with a potency-matched dual cytokine-antibody fusion protein leads to cancer eradication in BALB/c-derived tumors but not in other mouse strains. Cancer Immunol Immunother 67(9):1381–1391
De Luca R et al (2017) Potency-matched dual cytokine-antibody fusion proteins for cancer therapy. Mol Cancer Ther 16(11):2442–2451
Padutsch T et al (2019) Superior Treg-expanding properties of a novel dual-acting cytokine fusion protein. Front Pharmacol 10:1490
Schanzer JM et al (2006) Antitumor activity of a dual cytokine/single-chain antibody fusion protein for simultaneous delivery of GM-CSF and IL-2 to Ep-CAM expressing tumor cells. J Immunother 29(5):477–488
Daub H et al (2020) The trimer to monomer transition of Tumor Necrosis Factor-Alpha is a dynamic process that is significantly altered by therapeutic antibodies. Sci Rep 10(1):9265
Venetz D et al (2016) Targeted reconstitution of cytokine activity upon antigen binding using split cytokine antibody fusion proteins. J Biol Chem 291(35):18139–18147
Lucchi R, Bentanachs J, Oller-Salvia B (2021) The masking game: design of activatable antibodies and mimetics for selective therapeutics and cell control. ACS Cent Sci 7(5):724–738
Chen IJ et al (2017) Selective antibody activation through protease-activated pro-antibodies that mask binding sites with inhibitory domains. Sci Rep 7(1):11587
Elter A et al (2021) Protease-activation of fc-masked therapeutic antibodies to alleviate off-tumor cytotoxicity. Front Immunol 12:715719
Erster O et al (2012) Site-specific targeting of antibody activity in vivo mediated by disease-associated proteases. J Control Rel 161(3):804–812
Dakhel S et al (2021) An attenuated targeted-TNF localizes to tumors in vivo and regains activity at the site of disease. Int J Mol Sci 22(18):10020
Van Den Eeckhout B et al. (2021) Selective IL-1 activity on CD8(+) T cells empowers antitumor immunity and synergizes with neovasculature-targeted TNF for full tumor eradication. J Immunother Cancer 9(11)
Dakhel S et al (2019) Targeted enhancement of the therapeutic window of L19-TNF by transient and selective inhibition of RIPK1-signaling cascade. Oncotarget 10(62):6678–6690
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
The first draft of the manuscript was written by DYG and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gout, D.Y., Groen, L.S. & van Egmond, M. The present and future of immunocytokines for cancer treatment. Cell. Mol. Life Sci. 79, 509 (2022). https://doi.org/10.1007/s00018-022-04514-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04514-9