Abstract
Purpose
Intensive care unit (ICU)-acquired infections (IAI) result in increased hospital and ICU stay, costs and mortality. To date, no biomarker has shown sufficient evidence and ease of application in clinical routine for the identification of patients at risk of IAI. We evaluated the association of the systemic mRNA expression of two host response biomarkers, CD74 and IL10, with IAI occurrence in a large cohort of ICU patients.
Methods
ICU patients were prospectively enrolled in a multicenter cohort study. Whole blood was collected on the day of admission (D1) and on day 3 (D3) and day 6 (D6) after admission. Patients were screened daily for IAI occurrence and data were censored after IAI diagnosis. mRNA expression levels of biomarkers were measured using RT-qPCR. Fine and Gray competing risk models were used to assess the association between gene expression and IAI occurrence.
Results
A total of 725 patients were analyzed. At least one IAI episode occurred in 137 patients (19%). After adjustment for shock and sepsis status at admission, CD74 and IL10 levels were found to be significantly associated with IAI occurrence [subdistribution hazard ratio (95% confidence interval) 0.67 (0.46–0.97) for CD74 D3/D1 expression ratio and 2.21 (1.63–3.00) for IL10 at D3]. IAI cumulative incidence was significantly different between groups stratified according to CD74 or IL10 expression (Gray tests p < 0.001).
Conclusion
Our results suggest that two immune biomarkers, CD74 and IL10, could be relevant tools for the identification of IAI risk in ICU patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intensive care unit (ICU) patients are particularly exposed to secondary infections, namely ICU-acquired infections (IAI). Figures up to 30% have been reported in high-income countries [1]. Pooled cumulative incidence densities of catheter-related blood-stream infections, urinary catheter-related infections, and ventilator-associated pneumonia were 3.5 [95% confidence interval (CI) 2.8–4.1] per 1000 central line-days, 4.1 (3.7–4.6) per 1000 urinary catheter-days, and 7.9 (5.7–10.1) per 1000 ventilator-days, respectively. IAI result in prolonged hospital stay, long-term disability, increased resistance of microorganisms to antimicrobials, massive additional financial burden for health systems, and excess deaths. The pathophysiology of IAI occurrence has been based on the presence of invasive devices such as intubation and catheters. Recently, acquired immunodeficiency as observed following severe insults has received increasing interest [2]. Biomarkers targeting critically ill patients at high risk of IAI may be useful to identify those with immune alterations; they could benefit from specific preventive procedures [3] or from new immunostimulating drugs [4]. However, no biomarker has so far been found sufficiently robust for use in clinical routine. In a recent prospective study in ICU septic patients at admission, no gene was differentially expressed between patients who subsequently developed IAI and those who did not [5].
The objective of our study, undertaken in a large cohort of ICU patients, was to confirm the association of two markers of immune status with IAI occurrence: CD74 and interleukin-10 (IL-10). We have previously reported that the expression of CD74, the invariant chain involved in MHC class II molecules transport, was correlated with monocyte HLA-DR (mHLA-DR) [6], associated with IAI occurrence [7]. We also recently observed a higher mRNA expression level of IL-10, a key cytokine involved in anti-inflammatory response [8], in pediatric ICU patients who developed secondary infections [9]. As the availability of fully automated molecular platforms enables their use in clinical routine, the clinical interest of two potential biomarkers for IAI risk assessment in ICU patients was thus investigated through their systemic mRNA expression level.
Materials and methods
Study population
Patients aged >18 years were prospectively enrolled from December 2009 through June 2011 in six French ICUs (two medical and four surgical, from three hospitals) [10]. The study protocol was approved by the local ethics committee (Centres d’investigation Clinique IRB# 5044) which waived the need for informed consent from patients and/or next of kin (analyses performed on leftover blood). According to the French law at this date, patients or legal representatives were informed about the study and about their right to refuse to participate.
A cohort of 19 healthy volunteers from Etablissement Français du Sang (EFS) (median age 41 [35–50] years, male 42%) was used as control. Informed consent was obtained from blood donors, according to EFS standardized procedures.
Inclusion and exclusion criteria
The criteria for inclusion were a prediction of ICU length of stay of at least 2 days and the presence of Systemic Inflammatory Response Syndrome (SIRS) [11] due or not to an infectious ongoing process. The exclusion criteria were a preexisting immunodepression, including recent chemotherapy or immunosuppressive treatment, high dose (>5 mg/kg of equivalent prednisolone for a duration >5 days) or prolonged (0.5 mg/kg equivalent prednisolone >30 days) corticosteroid treatment, and aplasia (circulating neutrophils <500 cells/mm3), primary immune deficiency, and extracorporeal circulation the month before ICU admission.
Data collection
The following data were collected: age, gender, severity measured by the Simplified Acute Physiological Score (SAPS) II at ICU admission [12] and the Sequential Organ Failure Assessment (SOFA) score over the first 24 h after admission (D1), at day 3 (D3) and day 6 (D6) [13], and diagnosis category (medical, surgical). The chronic health status was defined using the Charlson score comorbidities [14]. Site of infection was recorded at admission for septic patients. The adequacy of initial antimicrobial treatment was assessed according to sensitivity or to local protocol. Length of stay and survival were measured at ICU discharge, at 28 days after admission and at hospital discharge. Shock status was defined by the administration of vasopressors.
ICU-acquired infection
During ICU stay, patients were screened daily for exposure to invasive device (intubation, indwelling urinary catheter, and central venous line) and for four major IAI occurrence: pulmonary, urinary tract, primary bloodstream, and catheter-related infections, according to the definitions used by the European Centre for Disease Control and Prevention (ECDC) [15]. Only the first IAI was considered in our analysis. Clinicians in charge of the patients and study nurses were blinded for the biomarker data.
Sample collection
Peripheral whole blood from ICU patients or healthy volunteers was collected in PAXgene™ Blood RNA tubes (PreAnalytix, Hilden, Germany). Samples were stabilized at least 4 h at room temperature after collection and frozen at −80 °C following the manufacturer’s guidelines. For ICU patients, blood was collected at D1 (within 12 h after admission), D3, and D6 after ICU admission. Biomarker data obtained after IAI occurrence were censored.
RNA extraction, reverse transcription and quantitative PCR
Total RNA was extracted from whole blood. Samples with RNA integrity number ≤6 were excluded due to poor quality RNA. Total RNA was reverse-transcribed into cDNA and expression levels of CD74 and IL10 were quantified using quantitative-real time polymerase chain reaction (Supplementary Method 1 and Supplementary Table 1).
Statistical analysis
Comparisons between IAI and no IAI patient clinical characteristics were performed with Chi square or Fisher’s exact tests for qualitative variables and Mann–Whitney or t tests for quantitative variables. Since ICU discharge and death are in competition with the event of interest (IAI), further analyses were performed within the competing risks framework. Cumulative incidence curves of IAI events since ICU admission were represented and Gray tests performed. Multivariate analyses of IAI incidence according to biomarkers expression, adding shock and sepsis status at admission as covariates, were performed with the Fine and Gray model [16]. Models were performed for each time-point of biomarker measurement individually and for the ratio between two time-points. For each biomarker, as four tests were performed, the Family Wise Error Rate was 18%. IAI cumulative incidence curves based on biomarker expression were calculated using a threshold maximizing the Youden index. The level of significance was set at 5%. Results were described with a 95% confidence interval. Statistical analysis were computed with R software v3.0.0. The R package ‘cmprsk’ was used for competing risks calculation.
Results
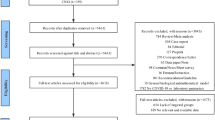
A total of 725 ICU patients were analyzed (Supplementary Fig. 1). At D3 and D6, samples were analyzed for 487 and 324 patients, respectively. At admission, 64% of patients presented with a shock of any origin, 70% with an infection, and 50% of infected patients met septic shock criteria (Table 1). The most frequent primary infection was respiratory community-acquired (64%; Supplementary Table 2). Overall day-28 mortality rate was 28%. Early mortality, defined as death occurring before D6, was high, involving 106 patients (56%).
At least one IAI episode occurred in 19% of patients, with a median onset of the first IAI episode of 10 [6–18] days (Table 2). The most common IAIs were pneumonia (63%), and urinary tract infections (20%). All pneumonia except one were ventilator-associated pneumonia. Almost all patients had at least one invasive device at admission (99%). Significant differences between IAI and no IAI patients were observed for SOFA at D1, trauma, sepsis at admission, treatments and major interventions during ICU stay (Table 1). Of note, lymphocyte count was not different between IAI and IAI-free patients at D1, D3 or D6 (Supplementary Table 2).
There was no difference regarding IAI onset time-course until day 10 between patients with and without shock at admission, but patients with shock had a final slightly higher IAI incidence (21% in shock patients vs. 16% in no shock patients, Gray test p = 0.194; Supplementary Fig. 2a). We observed larger differences when considering sepsis at admission: IAI occurred more frequently, and more rapidly in non-sepsis patients (26%) than in septic patients (16%, Gray test p = 0.002; Supplementary Fig. 2b). IAI patients had higher invasive device exposure duration until IAI occurrence (Supplementary Table 2). ICU and hospital lengths of stay, as well as total duration of invasive device exposure (intubation, urinary and central venous catheters), were higher for IAI patients as compared to IAI-free patients,
As early as at admission, and until D6, we observed significant differences in CD74 and IL10 mRNA expression levels between ICU patients and healthy volunteers, with lower CD74 and higher IL10 expression levels in patients compared to controls (Mann–Whitney test p < 0.001; Supplementary Fig. 3).
After adjustment for shock and sepsis status at admission, significant association with IAI occurrence was found for both CD74 and IL10 (Fig. 1; Supplementary Table 3). At admission, a higher CD74 mRNA expression level was associated with an increased risk of IAI occurrence [sdHR 1.51 (95% CI 1.11–2.06), p = 0.008]. At D3 and D6 there was no significant association with IAI occurrence. Interestingly, a significant association was found for the evolution of mRNA expression between D1 and D3: a higher D3/D1 ratio was associated with a decreased risk of IAI occurrence [sdHR 0.67 (0.46–0.97), p = 0.033].
Adjusted Fine and Gray subdistribution hazard ratios and 95 confidence intervals for IAI occurrence in multivariate analysis. For each individual time-points, models were computed adjusting for shock and sepsis status at admission as covariates (see Supplementary Table 3 for details). For IL10 mRNA levels, subdistribution hazard ratios are expressed for an increase of 0.1 unit. ICU intensive care unit, IAI ICU-acquired infection
A significant association between IL10 mRNA level and IAI occurrence was found at D3 and D6, with increased expression associated with increased IAI occurrence [sdHR 2.21 (1.63–3.00), p < 0.001 and 2.09 (1.41–3.10), p < 0.001, for D3 and D6, respectively]. The evolution between D1 and D3 was also significantly informative [sdHR 1.04 (1.02–1.06), p < 0.001].
CD74 mRNA expression was correlated with mHLA-DR measured at D3 by flow cytometry on a subset of patients (Spearman r = 0.72; Supplementary Fig. 4). We observed no correlation of CD74 or IL10 mRNA expression levels with cell count or severity scores (data not shown).
We assessed IAI cumulative incidence in patient groups stratified on CD74 and IL10 mRNA expression levels, using thresholds maximizing the Youden index. Cumulative incidence curves of IAI were significantly different between groups stratified on CD74 (Gray test p < 0.001; Fig. 2a). Patients exhibiting a CD74 D3/D1 mRNA ratio below 1.238, i.e. patients with a stable level between D1 and D3, had 1.8 (95% CI 1.2–2.7)-fold more IAI than those with a higher ratio, i.e. patients with an increase of CD74 level between D1 and D3 (62/231, 27% of IAI vs. 38/256, 15%). We also obtained a significant difference of IAI occurrence in groups stratified on IL10 mRNA level (Gray test p < 0.001; Fig. 2b), with IAI occurrence 2.7 (95% CI 1.8–4.0)-fold higher in patients with IL10 D3 level above 0.039 compared to patients with IL10 expression level below this threshold (66/206, 32% of IAI vs. 34/281, 12%).
Cumulative incidence of first IAI episode in percentage, in populations defined by the CD74 D3/D1 ratio (a) or IL10 at D3 (b). For each biomarker, the threshold was selected to maximize the Youden index (1.238 for CD74 D3/D1 expression ratio and 0.039 for IL10 at D3). For each group, curves were represented until the end of patient follow-up, defined by the occurrence of the first event among IAI, discharge alive without IAI or death without IAI. A significant difference was obtained between the two curves for the CD74 D3/D1 ratio (Gray test p < 0.001) and for IL10 at D3 (Gray test p < 0.001). ICU intensive care unit, IAI ICU-acquired infection
Discussion
In this prospective cohort of 725 ICU patients, 137 patients (19%) developed at least one episode of IAI. The mRNA expression levels of two host immune response biomarkers, CD74 and IL10, were significantly associated with IAI occurrence at different time-points, especially for the CD74 D3/D1 ratio and IL10 at D3 and D6 after admission. This association was observed independently of sepsis and shock status at admission. Patients’ stratification based on either CD74 or IL10 mRNA threshold levels allowed the discrimination of patients with low IAI risk from those who were more likely to develop IAI.
Our cohort considered patients irrespective of sepsis or shock status at admission, with the perspective to appreciate the ability of the markers to identify high risk of IAI in critically ill patients. The rate of 19% of IAI is consistent with the literature [5, 17, 18], regarding the severity of the cohort with a median SAPS II of 56. As previously reported [1], the most frequent IAI was pneumonia, followed by urinary tract infections. Diagnostic criteria of IAI were homogenous in the six ICUs of the same university hospitals group.
At admission, 70% of patients were septic, with 50% of them in septic shock. Obviously, this last group was very heterogeneous. For instance, the prognosis of the 67 trauma patients (9% of the cohort) is not the same as medical patients. A shock state requiring vasopressors was present in 467 patients (including the 255 septic shock patients). The common feature of this ICU cohort is the severity. As previously shown, the prognosis of non-infected ICU patients and severe sepsis was comparable [19]. However, IAI cumulative incidence varied among the groups defined by shock or sepsis status: if there was no difference between shocked and non-shocked patients (septic or not), the onset of IAI differed between septic and non-septic patients. This could be due to the less efficient microbiological IAI diagnosis in the presence of antimicrobials. This implies that IAI definitions must be firmly established and applied. In our study, definitions from the ECDC were used [15].
Another important aspect is data analysis design. Modifications of immunity may either be a cause or a consequence of IAI. It appears essential to censor biomarkers values at the date of IAI diagnosis, as we did in the present study, in order to assess their predictive potential. We used appropriate statistic models, such as competing risks models, in order to avoid results misinterpretation due to the mortality rate.
Recently, several studies have investigated the immunological modifications occurring after a severe aggression, whether or not it is of infectious nature. Depending on several factors such as the intensity of the initial aggression or patient comorbidities, immune response may not return to homeostasis but to an immunosuppressive state [8]. The intensity and duration of this immunosuppression have been found to be associated with organ failure, fatal outcome, and a decreased resistance to pathogens [2, 17]. The prediction of the risk of IAI has been studied in specific populations such as sepsis [7], trauma [20], burn [21], or in general ICU populations [2]. mHLA-DR appears as the most promising candidate biomarker for IAI prediction. However, its determination requires flow cytometers, making its use complex in large multicentric clinical evaluations and, more generally, in everyday clinical practice. mHLA-DR measurement needs to be performed within a few hours, due to the rapid modulation of its expression on cell surfaces and the impossibility of sample storage. Here, CD74 and IL10 were quantified by molecular biology. This technique, commonly used in clinics for viral load determination [22] or infection diagnosis [23], is now available on standardized and automated point of care platforms, ensuring ease of use for clinical routine in ICU. In the present study, as has been previously shown [6, 24, 25], CD74 mRNA was highly correlated with mHLA-DR expression and appears as a good alternative to flow cytometry. From D1, CD74 mRNA expression levels were lower in ICU patients compared to healthy volunteers. In agreement with results obtained for mHLA-DR [26], a decrease of CD74 D3/D1 mRNA expression ratio was significantly associated with IAI occurrence. However, contrary to what was expected based on mHLA-DR data, we observed a significant association between an increase of CD74 mRNA expression at D1 and IAI occurrence. CD74 expression was measured in whole blood, whereas the downregulation of HLA-DR in IAI patients has been mainly observed on monocytes. The higher expression of CD74 in IAI patients at admission could be due to a different regulation in other cell types, such as T lymphocytes [27]. This might also be explained by a different kinetic of expression regulation between mRNA and protein at early time-points.
IL-10 has emerged as a key regulator of immune response. We observed higher IL10 expression level in patients at admission than in controls, with a progressive decrease over time. This had been already reported in injured patients, in the presence or absence of infection [28]. In the present study, IL10 expression level from D3 after admission was significantly associated with IAI occurrence. These data in adult patients are consistent with our recent results obtained in a small cohort of pediatric ICU patients [9]. Previous studies evaluating IL-10 for IAI prediction gave conflicting results depending on the studied population, time-points and technique of measurement [18, 29, 30]. However, we showed that a single measurement of IL10 mRNA expression at D3 after ICU admission was able to identify patients at high risk of IAI, irrespective of infection and shock status at admission.
Few biomarkers have emerged for IAI prediction, and none has been sufficiently validated to be used in clinical routine. A recent large-scale transcriptomic analysis, performed at a single time-point at admission, failed to identify an immune response signature predictive of IAI occurrence in sepsis patients [5]. This underlines the importance of serial measurement of biomarkers since immunodepression develops several days after injury.
Based on our results, CD74 and IL10 could be considered as markers of an altered immune status. In this context, immunostimulating adjuvant therapies to combat the immunosuppressive state of late sepsis and improve clinical outcomes have become an area of growing interest. A phase III trial is currently under recruitment, using GM-CSF as an immunostimulant (NCT02361528). Several other drugs were or are being evaluated, such as IL-7 (NCT02640807), anti-PDL-1 (NCT02576457) or thymosin [31]. Several protective procedures could be used in patients particularly exposed to IAI due to an altered immune status identified by biomarkers. Selective digestive decontamination is largely used in some countries of northern Europe characterized by a low level of bacterial resistance [32], but not in southern Europe, probably for fear of a large exposure of antimicrobials. In the same way, chlorhexidine bathing [33], impregnated catheters [34], or specific dressings could be used to decrease IAI occurrence in these patients.
Our study has some limitations. First, biomarkers were measured at fixed time-points (D1, D3, D6) while IAI could appear at any time of the ICU stay, as long as the patient is at risk (mainly because of exposure to invasive devices). Daily determination of these parameters could give a better insight on the temporal relationship between IAI and persistence of immune alterations, as illustrated by the better performances of the CD74 D3/D1 mRNA expression ratio. Secondly, as there was no post-discharge follow-up, secondary infections acquired during hospital stay after ICU discharge were not recorded.
In conclusion, we confirm a significant association between IAI occurrence and systemic mRNA expression of CD74 and IL10, two key molecules involved in host immune response. As this was observed independently of sepsis and shock status at admission, these results suggest that these biomarkers may be relevant potential tools for the identification of patients at high risk of secondary infections in general ICU population.
Contributors
All authors were involved in the analysis and interpretation of the data as well as drafting the manuscript or revising it critically for important intellectual content. AL, AP, FV, EP, AF, MAC, VB, GM and JT made substantial contributions to the conception and design of the study and data acquisition. SM, DMB, EP, AF, AP, FV, GM, JT and AL participated actively in statistical analysis and interpretation of the data. All authors read and approved the final manuscript.
References
World Health Organization (2011) Report on the burden of endemic health care-associated infection worldwide. http://apps.who.int/iris/bitstream/10665/80135/1/9789241501507_eng.pdf. Accessed 16 Aug 2016
Conway Morris A, Anderson N, Brittan M et al (2013) Combined dysfunctions of immune cells predict nosocomial infection in critically ill patients. Br J Anaesth 111:778–787. doi:10.1093/bja/aet205
Climo MW, Yokoe DS, Warren DK et al (2013) Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med 368:533–542. doi:10.1056/NEJMoa1113849
Hutchins NA, Unsinger J, Hotchkiss RS, Ayala A (2014) The new normal: immunomodulatory agents against sepsis immune suppression. Trends Mol Med 20:224–233. doi:10.1016/j.molmed.2014.01.002
van Vught LA, Klein Klouwenberg PMC, Spitoni C et al (2016) Incidence, risk factors, and attributable mortality of secondary infections in the intensive care unit after admission for sepsis. JAMA. doi:10.1001/jama.2016.2691
Cazalis M-A, Friggeri A, Cavé L et al (2013) Decreased HLA-DR antigen-associated invariant chain (CD74) mRNA expression predicts mortality after septic shock. Crit Care 17:R287. doi:10.1186/cc13150
Landelle C, Lepape A, Voirin N et al (2010) Low monocyte human leukocyte antigen-DR is independently associated with nosocomial infections after septic shock. Intensive Care Med 36:1859–1866. doi:10.1007/s00134-010-1962-x
Hotchkiss RS, Monneret G, Payen D (2013) Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol 13:862–874. doi:10.1038/nri3552
Peronnet E, Nguyen K, Cerrato E et al (2016) Evaluation of mRNA biomarkers to identify risk of hospital acquired infections in children admitted to paediatric intensive care unit. PLoS ONE 11:e0152388. doi:10.1371/journal.pone.0152388
Friggeri A, Cazalis M-A, Pachot A et al (2016) Decreased CX3CR1 messenger RNA expression is an independent molecular biomarker of early and late mortality in critically ill patients. Crit Care 20:204. doi:10.1186/s13054-016-1362-x
Bone RC, Balk RA, Cerra FB et al (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 101:1644–1655
Le Gall JR, Lemeshow S, Saulnier F (1993) A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2963
Vincent JL, Moreno R, Takala J et al (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Charlson ME, Sax FL (1987) The therapeutic efficacy of critical care units from two perspectives: a traditional cohort approach vs a new case-control methodology. J Chronic Dis 40:31–39
Suetens C, Morales I, Savey A et al (2007) European surveillance of ICU-acquired infections (HELICS-ICU): methods and main results. J Hosp Infect 65(Suppl 2):171–173. doi:10.1016/S0195-6701(07)60038-3
Fine JP, Gray RJ (1999) A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 94:496. doi:10.2307/2670170
Lukaszewicz A-C, Grienay M, Resche-Rigon M et al (2009) Monocytic HLA-DR expression in intensive care patients: interest for prognosis and secondary infection prediction. Crit Care Med 37:2746–2752. doi:10.1097/CCM.0b013e3181ab858a
Gomez HG, Gonzalez SM, Londoño JM et al (2014) Immunological characterization of compensatory anti-inflammatory response syndrome in patients with severe sepsis: a longitudinal study*. Crit Care Med 42:771–780. doi:10.1097/CCM.0000000000000100
Dulhunty JM, Lipman J, Finfer S, Sepsis Study Investigators for the ANZICS Clinical Trials Group (2008) Does severe non-infectious SIRS differ from severe sepsis? Results from a multi-centre Australian and New Zealand intensive care unit study. Intensive Care Med 34:1654–1661. doi:10.1007/s00134-008-1160-2
Cheron A, Floccard B, Allaouchiche B et al (2010) Lack of recovery in monocyte human leukocyte antigen-DR expression is independently associated with the development of sepsis after major trauma. Crit Care 14:R208. doi:10.1186/cc9331
Venet F, Tissot S, Debard A-L et al (2007) Decreased monocyte human leukocyte antigen-DR expression after severe burn injury: correlation with severity and secondary septic shock. Crit Care Med 35:1910–1917. doi:10.1097/01.CCM.0000275271.77350.B6
Gullett JC, Nolte FS (2015) Quantitative nucleic acid amplification methods for viral infections. Clin Chem 61:72–78. doi:10.1373/clinchem.2014.223289
Brendish NJ, Schiff HF, Clark TW (2015) Point-of-care testing for respiratory viruses in adults: the current landscape and future potential. J Infect 71:501–510. doi:10.1016/j.jinf.2015.07.008
Payen D, Lukaszewicz A-C, Belikova I et al (2008) Gene profiling in human blood leucocytes during recovery from septic shock. Intensive Care Med 34:1371–1376. doi:10.1007/s00134-008-1048-1
Pachot A, Monneret G, Brion A et al (2005) Messenger RNA expression of major histocompatibility complex class II genes in whole blood from septic shock patients. Crit Care Med 33:31–38
Gouel-Chéron A, Allaouchiche B, Guignant C et al (2012) Early interleukin-6 and slope of monocyte human leukocyte antigen-DR: a powerful association to predict the development of sepsis after major trauma. PLoS ONE 7:e33095. doi:10.1371/journal.pone.0033095
Tschaikowsky K, Hedwig-Geissing M, Schiele A et al (2002) Coincidence of pro- and anti-inflammatory responses in the early phase of severe sepsis: longitudinal study of mononuclear histocompatibility leukocyte antigen-DR expression, procalcitonin, C-reactive protein, and changes in T-cell subsets in septic and postoperative patients. Crit Care Med 30:1015–1023
Pachot A, Monneret G, Voirin N et al (2005) Longitudinal study of cytokine and immune transcription factor mRNA expression in septic shock. Clin Immunol 114:61–69. doi:10.1016/j.clim.2004.08.015
Sherry RM, Cue JI, Goddard JK, et al (1996) Interleukin-10 is associated with the development of sepsis in trauma patients. J Trauma 40:613-616; discussion 616-617
Giannoudis PV, Smith RM, Perry SL et al (2000) Immediate IL-10 expression following major orthopaedic trauma: relationship to anti-inflammatory response and subsequent development of sepsis. Intensive Care Med 26:1076–1081
Wu J, Zhou L, Liu J et al (2013) The efficacy of thymosin alpha 1 for severe sepsis (ETASS): a multicenter, single-blind, randomized and controlled trial. Crit Care Lond Engl 17:R8. doi:10.1186/cc11932
de Smet AMGA, Bonten MJM, Kluytmans JAJW (2012) For whom should we use selective decontamination of the digestive tract? Curr Opin Infect Dis 25:211–217. doi:10.1097/QCO.0b013e3283507f8c
Derde LPG, Dautzenberg MJD, Bonten MJM (2012) Chlorhexidine body washing to control antimicrobial-resistant bacteria in intensive care units: a systematic review. Intensive Care Med 38:931–939. doi:10.1007/s00134-012-2542-z
Smith RN, Nolan JP (2013) Central venous catheters. BMJ 347:f6570
Acknowledgements
MIP Rea study group
Guillaume Marcotte, Christian Guillaume, Romain Hernu, Sylvie De La Salle, Marie Simon, Thomas Baudry, Elisabeth Cerrato, Emmanuelle Gallet-Gorius, Audrey Larue-Triolet, Christine Alberti-Segui, Nathalie Panel, Marion Provent, Mathieu Page, and Anne Portier. The authors gratefully thank Sophie Blein-Henry for her precious advice on statistical analysis.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
All authors are employees of either bioMerieux (EP, MAC, VB, JT and AP) or the University Hospital of Lyon (FV, DMB, AF, MC, LA, BA, BF, FA, TR, FT, VP, JB, GM, SM, HV and AL). This project is part of Advanced Diagnostics for New Therapeutic Approaches, a program dedicated to personalized medicine, coordinated by Institut Mérieux, and supported by the French public agency BPI (Banque Publique d’Investissement). TR reports non-financial support from Fresenius Medical Care, as well as grants, personal fees and non-financial support from Gambro Baxter, unconnected with the submitted work.
Fundings
This study was funded by University Hospital of Lyon, bioMérieux and Advanced Diagnostics for New Therapeutic Approaches, a program dedicated to personalized medicine, coordinated by Institut Mérieux and supported by the French public agency BPI (Banque Publique d’Investissement). bioMérieux and Lyon University Hospital were co-responsible for the study design, collection of data, interpretation of results, and writing of the report. BPI had no active role in the study.
Additional information
The MIP Rea Study Group members are listed in Acknowledgements section and in ESM1.
Take-home message:
Our study is the first to report a significant association between two host response biomarkers, CD74 and IL10, and IAI occurrence in a large cohort of medico-surgical ICU patients. This suggests that these biomarkers could guide clinicians for IAI prevention by using specific protective procedures, or could help patient stratification in clinical trials evaluating immunostimulating therapies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Peronnet, E., Venet, F., Maucort-Boulch, D. et al. Association between mRNA expression of CD74 and IL10 and risk of ICU-acquired infections: a multicenter cohort study. Intensive Care Med 43, 1013–1020 (2017). https://doi.org/10.1007/s00134-017-4805-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-017-4805-1