Abstract
Objectives
We investigated the efficacy of intensive rosuvastatin therapy plus 7-day dual antiplatelet therapy (DAPT) in reducing stroke recurrence for patients with acute ischemic stroke (AIS) and compared subgroups of patients.
Methods
We enrolled patients with AIS whose time of onset to medication was ≤ 72 h, and the baseline scores of NIHSS (bNIHSS) were 0–10. The patients received intensive rosuvastatin therapy plus 7-day DAPT with aspirin and clopidogrel (study group) or rosuvastatin plus single antiplatelet therapy (SAPT, control group). The primary outcomes were recurrence of ischemic stroke, bleeding, statin-induced liver injury, and statin-associated myopathy (SAM) within 90 days. We also performed a subgroup analysis to assess the heterogeneity of the two therapy regimens in reducing recurrent stroke.
Results
Recurrent stroke occurred in 10 patients in the study group and 42 patients in the control group (hazard ratio [HR], 0.373, 95% confidence interval [CI], 0.178–0.780; P = 0.009). Bleeding events occurred in 9 patients in the study group and 14 patients in the control group (HR, 1.019; 95%CI, 0.441–2.353; P = 0.966). Statin-induced liver injury and SAM were not recorded. Intensive rosuvastatin plus 7-day DAPT was generally effective in reducing the risk of recurrent stroke, except in the subgroup with bNIHSS ≤ 2. The therapy was particularly efficient in the elderly, male, high-bNIHSS, and hypertension, diabetes, and hyperlipidemia subgroups, with P < 0.02.
Conclusions
Without increasing bleeding and statin-associated adverse events, intensive rosuvastatin therapy plus 7-day DAPT significantly reduced the risk of recurrent stroke, especially for subgroups with high-risk factors. Clinical trial registration. China Clinical Trial Registration Center (ChiCTR1800017809).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Disorder of lipid metabolism, dysfunction of endothelial cells, and aggregation of many inflammatory factors are the external conditions of arterial thrombosis [1, 2], while platelet activation is the internal factor [3]. All these factors interact with and promote each other [4], eventually leading to acute coronary syndrome (ACS) and acute ischemic stroke (AIS).
Intensive statin therapy is commonly combined with dual antiplatelet therapy (DAPT) in patients with ACS after percutaneous coronary intervention, stent implantation [5, 6], and AIS [7, 8]. This combination treatment can significantly reduce the risk of recurrent thrombosis after stent implantation [9] and recurrent ischemic stroke after AIS [10, 11]. However, there are few reports on the efficacy of this regimen among different subgroups of patients with AIS. Thus, we investigated the risks and benefits of intensive rosuvastatin therapy plus 7-day DAPT in reducing recurrent ischemic stroke for patients with mild to moderate AIS and compared subgroups.
Materials and methods
Patients
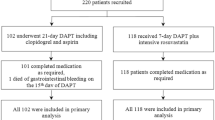
We recruited patients aged 18 years or older who were admitted to the Emergency Department of our hospital from October 2016 to December 2019. Computed tomography (CT) and magnetic resonance imaging (MRI) of the head was used to confirm the new focal infarction lesions within 72 h after the onset. Their National Institutes of Health Stroke Scale (NIHSS) scores at registration were 0–10. We excluded patients with intravenous thrombolysis/arterial thrombectomy or anticoagulation treatments, patients undergoing menstruation or pregnancy, and patients preparing for pregnancy within 3 months. In total, 331 patients with AIS met the above criteria, including 204 patients in the control group and 127 patients in the study group according to the therapy regimens. The Medical Ethics Committee of the China Rehabilitation Research Center approved this study (Ethics approval number: 2018–022-1). All patients provided written informed consent.
Study design and treatment
We stratified the patients based on their baseline demographic characteristics (Table 1). They received one of the two therapy regimens on a voluntary basis (DAPT + intensive rosuvastatin therapy for the study group and single antiplatelet therapy (SAPT) + rosuvastatin for the control group). The follow-up time was 90 days. The primary outcomes were a new ischemic stroke, bleeding events, and the adverse events statin-induced liver injury or statin-associated myopathy (SAM) within 90 days; the secondary outcome was the heterogeneity of the two therapy regimens in reducing recurrent ischemic stroke among subgroups of patients with AIS.
Therapy regimens
Patients in the study group received DAPT + intensive rosuvastatin therapy: aspirin (Bayer, 100 mg per tablet) 100 mg/d with an initial dose of 300 mg for 90 days, clopidogrel (Sanofi, 75 mg per tablet) 75 mg/d with an initial dose of 75–300 mg determined based on the clinical symptoms for 7 days, plus rosuvastatin (Nanjing Chia Tai-Tianqing Pharmaceutical Co., Ltd, 10 mg per tablet), 20 mg/d for 21 days, and then 10 mg/d for 90 days in total. Patients in the control group received SAPT + rosuvastatin: aspirin (Bayer, 100 mg per tablet) 100 mg/d or clopidogrel (Sanofi, 75 mg per tablet) 75 mg/d for 90 days, plus rosuvastatin (Nanjing Chia Tai-Tianqing Pharmaceutical Co., Ltd, 10 mg per tablet) 10 mg/d for 90 days.
According to previous studies, aspirin and clopidogrel consistently reduced recurrent vascular events [17] or recurrent ischemic stroke events [18, 19] for stroke patients within 1 year. For a few patients in the control group who were intolerant to aspirin, we used clopidogrel 75 mg/d instead of aspirin 100 mg/d due to its lower gastric toxicity [17], which had no significant effect on the outcome.
Assessment criteria
We assessed focal neurological deficits by assessing the bNIHSS, which ranges from 0 to 43, with higher scores indicating worse deficits [20]. Because there were few patients with bNIHSS above 10 in our Emergency Department, and the therapy regimens in this study were very poor for them, we only registered patients with bNIHSS ≤ 10 in this study.
Recurrent ischemic stroke—the aggravation of existing clinical symptoms or the emergence of new focal neurological deficit symptoms within 90 days after the first treatment—was confirmed by CT and MRI scans of the head, which showed obviously enlarged original lesions or new ischemic lesions. Bleeding events included intracranial and gastrointestinal mucosal hemorrhage, which were confirmed by head CT and gastric contents analysis or fecal occult blood test, respectively, within 90 days after treatment. According to the global use of streptokinase and tissue plasminogen activator to treat coronary occlusion (GUSTO) [21], the severity of bleeding was classified as mild, moderate, or severe. Statin-induced liver injury and SAM were defined as a more than three-fold increase in the normal upper limit levels of transaminase (alanine transferase [ALT] or aspartate transferase [AST]) [22] and creatine kinase (CK) [23] within 90 days.
Statistical analysis
This study adopted an incomplete randomized controlled trial design, with Type I Error α = 0.05 and power of test (1 − β) = 0.85. The PASS 15.0 software (NCSS, LLC, Kaysville, UT, USA) was used to estimate the sample size. According to a meta-analysis by Kwok et al. [24], SAPT reduced the recurrence rate of ischemic stroke by 52%. In line with the CHANCE trial [25], DAPT actually reduced AIS recurrent stroke by nearly 84%, which is significantly more than SAPT. With these studies in mind, we concluded that we needed a total sample size of 312 cases, with a control group to study group size ratio of around 3:2. We effectively recruited 331 patients, with 204 patients in the control group and 127 in the research group.
SPSS 25.0 statistical software (IBM Corporation, Armonk, NY, USA) was used for data analysis. We expressed measurement data as the median (M) and inter-quartile range (IQR) from the rank-sum test results and expressed count data as % from the χ2 test results. A Cox proportional hazards model was used to evaluate differences in the recurrent ischemic stroke events and bleeding events within 90 days between the two groups. We compared the ALT, AST, lactate dehydrogenase (LDH), and CK levels before and 2 weeks after therapy using a rank sum test. Finally, we assessed the heterogeneity of the two therapy regimens of this study in reducing recurrent ischemic stroke by performing a subgroup analysis. P < 0.05 was considered statistically significant for the first three statistical analyses, and P < 0.02 was considered statistically significant for the subgroup analysis.
Results
Baseline data between the two groups
Baseline demographic characteristics were well balanced between the two groups with all P > 0.05 among age; gender; systolic and diastolic blood pressure at registration; OMT; bNIHSS; previous medical histories; and the levels of ALT, AST, LDH, and CK before and 2 weeks (14 ± 3 days) after therapy (Table 2).
Recurrent ischemic stroke
Within 90 days, 52 patients underwent recurrent ischemic stroke: 10 (7.87%) in the study group and 42 (20.60%) in the control group. The study group had a 62% lower risk of recurrent ischemic stroke than the control group (hazard ratio [HR] for the study group vs. control group, 0.373, 95% confidence interval [CI], 0.178–0.780, P = 0.009). This result shows that intensive rosuvastatin therapy plus 7-day DAPT was far superior to rosuvastatin plus SAPT in reducing the risk of recurrent ischemic stroke within 90 days (Table 3 and Fig. 1).
Bleeding events
A total of 9 patients (7.09%) in the study group and 14 patients (6.86%) in the control group reported bleeding events. A Cox proportional hazards model revealed no significant difference between the two groups (HR, 1.019; 95% CI, 0.441–2.353; P = 0.966), suggesting that intensive rosuvastatin therapy plus 7-day DAPT did not increase the risk of bleeding compared with rosuvastatin plus SAPT (Table 3).
Statin-induced liver injury or SAM
None of the patients showed an increase superior to three-fold in the levels of ALT, AST, or CK. The ALT, AST, LDH, and CK levels remained stable before therapy and after 2 weeks (14 ± 3 days) among the groups (P > 0.05), except for the CK levels in the control group, which were significantly lower after 2 weeks of treatment (P < 0.001) (Table 4). These findings showed that the two regimens did not increase the risk of statin-induced liver injury or SAM.
Heterogeneity in reducing recurrent ischemic stroke among subgroups
To explore the heterogeneity of the two different therapy regimens in reducing the risk of recurrent ischemic stroke for AIS patients within 90 days, we performed a subgroup analysis according to the aforementioned stratification. The intensive rosuvastatin therapy plus 7-day DAPT always effectively reduced the risk of recurrent ischemic stroke, with the HR value of each subgroup located on the left side of the invalid line, except the subgroup with bNIHSS ≤ 2. It was particularly efficient in these subgroups with high-risk factors such as the elderly, hypertension, diabetes, hyperlipidemia, or prior-stroke, non-antiplatelet treatment, and high-bNIHSS (3–10 points), while it was less efficient in female, OMT > 48 h, and prior atrial fibrillation subgroups (Fig. 2).
Discussion
In this study, compared with rosuvastatin plus SAPT, the intensive rosuvastatin therapy plus 7-day DAPT significantly reduced the risk of recurrent stroke within 90 days for patients with mild to moderate AIS, without increasing adverse events, such as bleeding, statin-induced liver injury, or SAM. The reduction was particularly significant in the subgroups with high-risk factors (the elderly [> 68 years old], hypertension, diabetes, hyperlipidemia, prior-stroke, non-antiplatelet treatment, and high bNIHSS [3–10 points]), but not significant in female, OMT > 48 h, and prior atrial fibrillation subgroups. These results should be interpreted by considering the characteristics of the therapy regimens in this study, namely, the strong inhibition of platelet aggregation and reduction of thrombosis by DAPT and the multiple effects of intensive statin therapy.
Previous study showed that statins, especially fat-soluble statins, commonly caused statin-induced liver injury and SAM, along with a three-fold or more increase in ALT, AST, or CK levels [26]. However, as a water-soluble statin, rosuvastatin only enters hepatocytes through special channel proteins on the cell membrane, rarely causing rosuvastatin-induced liver injury and SAM [27, 28]. DAPT can increase the risk of bleeding [29,30,31], although this is most likely to occur after 1 week of treatment [31]. Therefore, intensive rosuvastatin therapy plus 7-day DAPT significantly reduced the risk of ischemic stroke recurrence in patients with mild to moderate AIS, without increasing bleeding, statin-induced liver injury, and SAM.
In the elderly subgroup (> 68 years old), the study therapy reduced the risk of stroke recurrence more than the control therapy did, which may be related to elderly patients’ high risk of stroke [32], low self-healing ability [33], and higher dependence on effective interventions. In the male subgroup, the treatment effect of the study group was significantly better than that of the control group, while. However, there was no statistically significant difference in the female subgroup, which can be ascribed to the older age, higher prevalence of risk factors [34], higher disability severity, and worse prognosis [35,36,37,38,39] for female patients when the ischemic stroke occur, leading to the failure of various interventions. Moreover, the incidence rate of ischemic stroke was significantly higher in men than in women [39], which led to a relatively large sample size for the male subgroup and made it easier to obtain statistically significant results.
At the early stage of cerebral infarction, a large part of the brain tissue is still in the ischemic penumbra due to the incomplete rupture of the lipid plaque and relatively mild inflammatory storm [4]. Therefore, the earlier effective intervention measures are given, the more dormant brain cells are saved, and the more obvious clinical symptoms are relieved, which could explain why the effect of the subgroup of OMT ≤ 48 h surpassed that of the subgroup with OMT > 48 h.
The NIHSS can not only quantify the symptoms and signs of focal neurological deficit [40] but also accurately reflect the volume of cerebral infarction within a certain infarct volume [41]. The lower the score, the lighter the symptoms, and the better the prognosis. Study showed that AIS patients with a bNIHSS < 3 had a good prognosis and were not affected by intravenous thrombolysis [16]. This is consistent with our results in the subgroup with bNHISS ≤ 2, independent of the therapy regimens. However, in the subgroup with bNHISS of 3–10, the study treatment was significantly more efficient than the control treatment, indicating that the intensive rosuvastatin therapy plus 7-day DAPT regimen was more beneficial to the subgroup with high bNIHSS (3–10).
Many studies have shown that AIS patients with high-risk factors [42], such as hypertension [43,44,45], diabetes [46, 47], hyperlipidemia [48], and prior-stroke [49], have a significantly higher risk of recurrent stroke. The study also showed that statins can reduce the incidence of serious cardiovascular and cerebrovascular events by 20–30% in patients with high-risk factors [50], which is consistent with our results. Moreover, the study treatment reduced the risk of recurrent stroke significantly more than the control treatment did in the subgroups with hypertension, diabetes, hyperlipidemia, and prior-stroke. Additionally, the study treatment did not reduce the risk of recurrent stroke any more than the control treatment did in the atrial fibrillation subgroup, which may be related to the fact that thrombus comes from the heart [51, 52], and the best treatment regimen for these patients is anticoagulation agents [53].
Aspirin and clopidogrel are the most common antiplatelet drugs. Aspirin has an irreversible inhibitory effect on platelet aggregation through acetylated platelet cyclooxygenase, while clopidogrel, as an adenosine diphosphate receptor inhibitor, inhibits platelet aggregation. For patients who took antiplatelet agents regularly and still had AIS, the possible cause was aspirin resistance [54] or clopidogrel resistance [55], which may explain that the study treatment can only reduce the risk of recurrent stroke significantly in patients of the subgroup of non-antiplatelet, but not in patients of the subgroup of prior-antiplatelet.
Limitations
This study was a single-center study with small sample size and incomplete randomized controlled design. These characteristics inevitably led to some weaknesses in the research results, which need to be confirmed by future large sample size and multi-center clinical studies.
Conclusion
Compared with rosuvastatin plus SAPT, the intensive rosuvastatin therapy plus 7-day DAPT with aspirin and clopidogrel significantly reduced the risk of recurrent ischemic stroke within 90 days for patients with mild to moderate AIS, without increasing bleeding, statin-induced liver injury, or SAM. These effects were particularly significant in the subgroups with high-risk factors such as elderly patients (> 68 years old), patients with hypertension, diabetes, hyperlipidemia, prior-stroke, non-antiplatelet treatment, and high bNHISS scores (3–10).
Data availability
All the required data about the study are present in the manuscript. We are happy to provide additional data if the reviewer or the editor requires further data.
References
Badimon L, Suades R, Fuentes E et al (2016) Role of platelet-derived microvesicles as crosstalk mediators in atherothrombosis and future pharmacology targets: a link between inflammation, atherosclerosis, and thrombosis. Front Pharmacol 7:293. https://doi.org/10.3389/fphar.2016.00293
Iba T, Levy JH (2018) Inflammation and thrombosis: roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J Thromb Haemost 16(2):231–241. https://doi.org/10.1111/jth.13911
Koupenova M, Clancy L, Corkrey HA et al (2018) Circulating platelets as mediators of immunity, inflammation, and thrombosis. Circ res 122(2):337–351. https://doi.org/10.1161/CIRCRESAHA.117.310795
Montecucco F, Carbone F, Schindler TH (2016) Pathophysiology of ST-segment elevation myocardial infarction: novel mechanisms and treatments. Eur Heart J 37(16):1268–1283. https://doi.org/10.1093/eurheartj/ehv592
Di Sciascio G, Patti G, Pasceri V et al (2009) Efficacy of atorvastatin reload in patients on chronic statin therapy undergoing percutaneous coronary intervention: results of the ARMYDA-RECAPTURE (Atorvastatin for Reduction of Myocardial Damage During Angioplasty) Randomized Trial. J Am Coll Cardiol 54(6):558–565. https://doi.org/10.1016/j.jacc.2009.05.028
Yun KH, Jeong MH, Oh SK et al (2009) The beneficial effect of high loading dose of rosuvastatin before percutaneous coronary intervention in patients with acute coronary syndrome. Int J Cardiol 137(3):246–251. https://doi.org/10.1016/j.ijcard.2008.06.055
Yuan HW, Ji RJ, Lin YJ et al (2018) Intensive versus moderate statin therapy discontinuation in patients with acute ischemic stroke or transient ischemic attack. Clin Ther 40(12):2041–2049. https://doi.org/10.1016/j.clinthera.2018.10.003
Grundy SM, Stone NJ, Bailey AL et al (2019) 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 139(25):e1046–e1081. https://doi.org/10.1161/CIR.0000000000000624
Jang HG, Kim K, Park HW et al (2021) Restenosis of a drug eluting stent on the previous bioresorbable vascular scaffold successfully treated with a drug-coated balloon: a case report. World J Clin Cases 9(3). https://doi.org/10.12998/wjcc.v9.i3.758
O’Donnell MJ, Chin SL, Rangarajan S et al (2016) Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet 388(10046). https://doi.org/10.1016/S0140-6736(16)30506-2
Ridker PM, Revkin J, Amarenco P et al (2017) Cardiovascular efficacy and safety of bococizumab in high-risk patients. N Engl J Med 376(16). https://doi.org/10.1056/NEJMoa1701488
Tan YF, Zhu MJ, Zhou K (2016) Life expectancy, healthy working life expectancy, and postponement of retirement age. Popul J 38(1):26–34. https://doi.org/10.16405/j.cnki.1004-129X.2016.01.003
Hu AG (2015) The core idea of the 13th Five Year Plan is to promote the all-round development of people. Red Flag Manuscript 23:4–6
Liu LP, Duan Chen WQ, WY, et al (2019) Chinese stroke association guidelines for clinical management of cerebrovascular disorder (excerpts) — clinical management of ischemic cerebrovascular disorders. Chin J Stroke 14(07):709–726. https://doi.org/10.3969/j.issn.1673-5765.2019.07.014
Yoo AJ, Romero J, Hakimelahi R et al (2010) Predictors of functional outcome vary by the hemisphere of involvement in major ischemic stroke treated with intra-arterial therapy: a retrospective cohort study. BMC Neurol 10(1):1–11. https://doi.org/10.1186/1471-2377-10-25
Romano JG, Gardener H, Campo-Bustillo I et al (2021) Predictors of outcomes in patients with mild ischemic stroke symptoms: MaRISS. Stroke 52(6):1995–2004. https://doi.org/10.1161/STROKEAHA.120.032809
CAPRIE steering Committee (1996). A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 348(9038):1329–1339. https://doi.org/10.1016/s0140-6736(96)09457-3
Sacco RL, Diener HC, Yusuf S et al (2008) Aspirin and extended-release dipyridamole versus clopidogrel for recurrent stroke. N Engl J Med 359(12):1238–1251. https://doi.org/10.1056/NEJMoa0805002
Xiao WM, Wang SF, Lin XX et al (2009) Comparison of clinical efficacy of clopidogrel and aspirin in preventing recurrence of non cardiogenic cerebral infarction. Guangdong Med J 30(2):288–289. https://doi.org/10.13820/j.cnki.gdyx.2009.02.056
Ghandehari K (2013) Challenging comparison of stroke scales. J Res Med Sci 18(10):906–910
The GUSTO Investigators (1993). An international randomized trial comparing four thrombolytic strategies for acute myocardial infarction. N Engl J Med 329:673–682. https://doi.org/10.1056/NEJM199309023291001
Chinese Medical Association (2018). Guideline for primary care of dyslipidemias: practice version (2019). Chin J Gen Pract 018(005):417–421. https://doi.org/10.3760/cma,j.issn.1671-7368.2019.05.004
Tournadre A (2020) Statins, myalgia, and rhabdomyolysis. Joint Bone Spine 87(1):37–42. https://doi.org/10.1016/j.jbspin.2019.01.018
Kwok CS, Shoamanesh A, Copley HC et al (2015) Efficacy of antiplatelet therapy in secondary prevention following lacunar stroke: pooled analysis of randomized trials. Stroke 46(4):1014–1023. https://doi.org/10.1161/strokeaha.114.008422
Wang YJ, Wang YL, Zhao XQ et al (2013) Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med 369(1):11–19. https://doi.org/10.1056/NEJMoa1215340
Kiortsis DN, Filippatos TD, Mikhailidis DP et al (2007) Statin-associated adverse effects beyond muscle and liver toxicity. Atherosclerosis 195(1):7–16. https://doi.org/10.1016/j.atherosclerosis.2006.10.001
Li YF (2018) Research and development of statins and their fat solubility and water solubility. Chin J Geriatr Heart Brain Vessel Dis 20(9):1008–1008. https://doi.org/10.3969/j.issn.1009-0126.2018.09.030
Petry NJ, Baye JF, Frear S et al (2022) Progression of precision statin prescribing for reduction of statin-associated muscle symptoms. Pharmacogenomics 23(10):585–596. https://doi.org/10.2217/pgs-2022-0055
Wang D, Gui L, Dong Y et al (2016) Dual antiplatelet therapy may increase the risk of non- intracranial haemorrhage in patients with minor strokes: a subgroup analysis of the CHANCE trial. Stroke Vasc Neurol 1(2):29–36. https://doi.org/10.1136/svn-2016-000008
Bhatia K, Jain V, Aggarwal D et al (2021) Dual antiplatelet therapy versus aspirin in patients with stroke or transient ischemic attack: meta-analysis of randomized controlled trials. Stroke 52(6):e217–e223. https://doi.org/10.1161/STROKEAHA.120.033033
Johnston SC, Easton JD, Farrant M et al (2018) Clopidogrel and aspirin in acute ischemic stroke and high-risk TIA (POINT). N Engl J Med 379(3):215–225. https://doi.org/10.1056/NEJMoa1800410
Lisabeth LD, Brown DL, Zahuranec DB et al (2021) Temporal trends in ischemic stroke rates by ethnicity, sex, and age (2000–2017): the brain attack surveillance in Corpus Christi Project. Neurology 97(22):e2164–e2172. https://doi.org/10.1212/WNL.0000000000012877
Gao SY, Qin YF, Zhang HL et al (2021) Correlation between age and 1-year prognosis of patients with acute cerebral infarction. Chin J Stroke 16(08):810–815. https://doi.org/10.3969/j.issn.1673-5765.2021.08.010
Sarrafzadegan N, Gharipour M, Sadeghi M et al (2017) Metabolic syndrome and the risk of ischemic stroke. J Stroke Cerebrovasc Dis 26(2). https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.09.019
Phan HT, Reeves MJ, Blizzard CL et al (2019) Sex differences in severity of stroke in the INSTRUCT study: a meta-analysis of individual participant data. J Am Heart Assoc 8(1):e010235. https://doi.org/10.1161/JAHA.118.010235
Carcel C, Wang X, Sandset EC et al (2021) Sex differences in treatment and outcome after stroke: pooled analysis including 19,000 participants. Neurology 96(23):1106. https://doi.org/10.1212/WNL.0000000000011946
Kremer C, Gdovinova Z, Bejot Y et al (2022) European stroke organisation guidelines on stroke in women: management of menopause, pregnancy and postpartum. Eur Stroke J. 7(2):I–XIX. https://doi.org/10.1177/23969873221078696
Ali M, van Os HJA, van der Weerd N et al (2022) Sex differences in presentation of stroke: a systematic review and meta-analysis. Stroke 53(2):345–354. https://doi.org/10.1161/STROKEAHA.120.034040
Gu HQ, Wang CJ, Yang X et al (2022) Sex differences in vascular risk factors, in-hospital management, and outcomes of patients with acute ischemic stroke in China. Eur J Neurol 29(1):188–198. https://doi.org/10.1111/ene.15124
Fonarow GC, Saver JL, Smith EF et al (2012) Relationship of National Institutes of Health Stroke Scale to 30-day mortality in medicare beneficiaries with acute ischemic stroke. J Am Heart Assoc 1(1):42–50. https://doi.org/10.1161/JAHA.111.000034
Mishra NK, Albers GW, Christensen S et al (2014) Comparison of magnetic resonance imaging mismatch criteria to select patients for endovascular stroke therapy. Stroke 45(5):1369–1374. https://doi.org/10.1161/STROKEAHA.114.004772
Wang J, Bai L, Shi M et al (2016) Trends in age of first-ever stroke following increased incidence and life expectancy in a low-income Chinese population. Stroke 47(4):929–935. https://doi.org/10.1161/STROKEAHA.115.012466
Robinson TG, Minhas JS, Miller J (2022) Review of major trials of acute blood pressure management in stroke. J Cereb Blood Flow Metab 42(3):404–410. https://doi.org/10.1177/0271678X211004310
Jillella DV, Calder CS, Uchino K et al (2020) Blood pressure and hospital discharge outcomes in acute ischemic stroke patients undergoing reperfusion therapy. J Stroke Cerebrovasc Dis 29(11):105211. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105211
Yannoutsos A, Dreyfuss Tubiana C, Safar ME et al (2017) Optimal blood pressure target in stroke prevention. Curr Opin Neurol 30(1):8–14. https://doi.org/10.1097/WCO.0000000000000407
Lau LH, Lew J, Borschmann K et al (2019) Prevalence of diabetes and its effects on stroke outcomes: a meta-analysis and literature review. J Diabetes Investig 10(3):780–792. https://doi.org/10.1111/jdi.12932
Fadini GP, Cosentino F (2018) Diabetes and ischaemic stroke: a deadly association. Eur Heart J 39(25):2387–2389. https://doi.org/10.1093/eurheartj/ehy033
Jeong W, Joo JH, Kim H et al (2022) Association between statin adherence and the risk of stroke among South Korean adults with hyperlipidemia. Nutr Metab Cardiovasc Dis 32(3):560–566. https://doi.org/10.1016/j.numecd.2021.12.010
Report on stroke prevention and treatment in China Writing Group (2022). Brief report on stroke prevention and treatment in China, 2020. Chin J Cerebrovasec Dis 19(2):136–144. https://doi.org/10.3969/j.issn.1672-5921.2022.02.011
Milionis H, Ntaios G, Korompoki E et al (2020) Statin-based therapy for primary and secondary prevention of ischemic stroke: a meta-analysis and critical overview. Int J Stroke 15(4):377–384. https://doi.org/10.1177/1747493019873594
Wang W, Jiang B, Sun H et al (2017) Prevalence, incidence, and mortality of stroke in China: results from a nationwide population-based survey of 480687 adults. Circulation 135(8):759–771. https://doi.org/10.1161/CIRCULATIONAHA.116.025250
Feigin VL, Roth GA, Naghavi M et al (2016) Global burden of stroke and risk factors in 188 countries, during 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet Neurol 15(9):913–924. https://doi.org/10.1016/S1474-4422(16)30073-4
Kumbhani DJ, Cannon CP, Beavers CJ et al (2021) 2020 ACC expert consensus decision pathway for anticoagulant and antiplatelet therapy in patients with atrial fibrillation or venous thromboembolism undergoing percutaneous coronary intervention or with atherosclerotic cardiovascular disease: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol 77(5):629–658. https://doi.org/10.1016/j.jacc.2020.09.011
Morton M, Kubiak-Balcerewicz K, Sarnowska A et al (2021) Biochemical aspirin resistance in acute stroke patients and its association with clinical factors: a prospective pilot study. Folia Neuropathol 59(3):271–275. https://doi.org/10.5114/fn.2021.109434
Wiśniewski A (2021) Multifactorial background for a low biological response to antiplatelet agents used in stroke prevention. Medicina (Kaunas) 57(1):59. https://doi.org/10.3390/medicina57010059
Acknowledgements
We thank Ph.D Yunlei Wang for careful reading of the manuscript.
Funding
This study was funded by Beijing Municipal Commission of Science and Technology (Grand Numbers: Z181100001718066).
Author information
Authors and Affiliations
Contributions
Study conception or design: TZ and HTL. Acquisition of the data: TD, XML, JMC, XHY. Analysis and interpretation of the data: TD, WH, XML. Drafting and revising the article: TZ and HTL. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
The Medical Ethics Committee of China Rehabilitation Research Center has approved the study (Ethics approval number: 2018–022-1). Written informed consents were obtained from all participants.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, T., Zhang, T., Lu, H. et al. Evaluation and subgroup analysis of the efficacy and safety of intensive rosuvastatin therapy combined with dual antiplatelet therapy in patients with acute ischemic stroke. Eur J Clin Pharmacol 79, 389–397 (2023). https://doi.org/10.1007/s00228-022-03442-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-022-03442-8