Abstract
Purpose
Detailed knowledge on the normative growth of the spine is of great relevance in the prenatal diagnosis of its abnormalities. The present study was conducted to compile age-specific reference data for vertebra C4 and its three ossification centers in human fetuses.
Materials and methods
With the use of CT (Biograph mCT), digital image analysis (Osirix 3.9) and statistical analysis (Wilcoxon signed-rank test, Kolmogorov–Smirnov test, Levene’s test, Student’s t test, one-way ANOVA, post hoc RIR Tukey test, linear and nonlinear regression analysis), the normative growth of vertebra C4 and its three ossification centers in 55 spontaneously aborted human fetuses (27 males, 28 females) aged 17–30 weeks was examined.
Results
Significant differences in neither sex nor laterality were found. The height and transverse and sagittal diameters of the C4 vertebral body increased logarithmically as: y = −3.866 + 2.225 × ln(Age) ± 0.238 (R 2 = 0.69), y = −7.077 + 3.547 × ln(Age) ± 0.356 (R 2 = 0.72) and y = −3.886 + 2.272 × ln(Age) ± 0.222 (R 2 = 0.73), respectively. The C4 vertebral body grew linearly in cross-sectional area as y = −7.205 + 0.812 × Age ± 1.668 (R 2 = 0.76) and four-degree polynomially in volume as y = 14.108 + 0.00007 × Age4 ± 6.289 (R 2 = 0.83). The transverse and sagittal diameters, cross-sectional area and volume of the ossification center of the C4 vertebral body generated the following functions: y = −8.836 + 3.708 × ln(Age) ± 0.334 (R 2 = 0.76), y = −7.748 + 3.240 × ln(Age) ± 0.237 (R 2 = 0.83), y = −4.690 + 0.437 × Age ± 1.172 (R 2 = 0.63) and y = −5.917 + 0.582 × Age ± 1.157 (R 2 = 0.77), respectively. The ossification center-to-vertebral body volume ratio gradually declined with age. On the right and left, the neural ossification centers showed the following growth: y = −19.601 + 8.018 × ln(Age) ± 0.369 (R 2 = 0.92) and y = −15.804 + 6.912 × ln(Age) ± 0.471 (R 2 = 0.85) for length, y = −5.806 + 2.587 × ln(Age) ± 0.146 (R 2 = 0.88) and y = −5.621 + 2.519 × ln(Age) ± 0.146 (R 2 = 0.88) for width, y = −9.188 + 0.856 × Age ± 2.174 (R 2 = 0.67) and y = −7.570 + 0.768 × Age ± 2.200 (R 2 = 0.60) for cross-sectional area, and y = −13.802 + 1.222 × Age ± 1.872 (R 2 = 0.84) and y = −11.038 + 1.061 × Age ± 1.964 (R 2 = 0.80) for volume, respectively.
Conclusions
The morphometric parameters of vertebra C4 and its three ossification centers show no sex differences. The C4 vertebral body increases logarithmically in height and both sagittal and transverse diameters, linearly in cross-sectional area, and four-degree polynomially in volume. The three ossification centers of vertebra C4 grow logarithmically in both transverse and sagittal diameters, and linearly in both cross-sectional area and volume. The age-specific reference intervals for evolving vertebra C4 may be useful in the prenatal diagnosis of congenital spinal defects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Advances in ultrasound devices have facilitated the assessment of most fetal structures and improved the prenatal diagnostics [1, 16, 25, 30]. Both CT and MRI examinations of the vertebral column are often superior to ultrasonography for evaluation of spinal anomalies [6, 8, 12, 15, 19, 21]. Detailed knowledge on the normative growth of the spine is relevant for diagnosing its abnormalities [12, 15, 23, 30, 33] and skeletal dysplasias [29].
The typical cervical vertebra is approximately 1/2 and 2/3 of the height of the lumbar and thoracic vertebrae, respectively [2]. Any vertebra ossifies from the three primary ossification centers, one existing in the vertebral body and one occurring in each neural process [2–4, 22]. Developmental pathways of the appearance of ossification centers for the neural processes and vertebral bodies evolve completely independently of each other in a definite topographical sequence [2]. Therefore, ossification of vertebral bodies starts with the thoracolumbar junction to proceed bi-directionally in both cranial and caudal directions [24]. The three ossification pathways of the neural processes have been postulated: firstly, originating in the thoracolumbar, cervico-thoracic, and superior cervical regions [4]; secondly, originating in the mid-thoracic spinal region [22]; and thirdly, originating in the superior cervical region [3].
To date, little has been known in the medical literature on morphometric values for cervical vertebrae in human fetuses [2, 24, 28], and the quantitative analysis of vertebral ossification centers has not been reported yet. Among other cervical vertebrae, we have specifically looked at the C4 vertebra, being a typical mid-cervical one. Its growth patterns will be useful in further understanding the development of adjacent vertebrae, in both proximal and distal directions. For this reason, to supplement fragmentary information about the dimensions of the C4 vertebra and its ossification centers, our objectives were set to examine the following:
-
age-specific reference intervals for height, transverse and sagittal diameters, cross-sectional area, and volume of its vertebral body;
-
age-specific reference intervals for transverse and sagittal diameters, cross-sectional area, and volume of its three ossification centers;
-
the best-fit growth curves for each parameter examined;
-
the relative growth of the ossification center within the vertebral body (the ossification center-to-vertebral body volume ratio).
Materials and methods
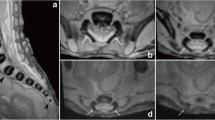
The present study included 55 ethnically homogenous human fetuses (27 males, 28 females) aged 17–30 weeks, of Caucasian racial origin (Table 1), which had been derived from spontaneous abortions or stillbirths in the years 1989–2001 because of placental insufficiency. Gestational ages were determined from measurements of the fetal crown–rump length [14]. No attempt was done to encourage fetal donation. The use of the fetuses for research was accepted by the University Research Ethics Committee (KB 275/2011). None of the fetuses demonstrated visible malformations. For preservation, all specimens were immersed in 10 % neutral buffered formalin solution. The fetuses underwent CT examinations with the reconstructed slice width option of 0.4 mm and 128 slices were acquired simultaneously by Biograph mCT (Siemens). The CT scans obtained were recorded in DICOM formats (Fig. 1a), with possibility to create three-dimensional reconstructions and the morphometric analysis of structures examined. Measurements of the vertebral column could be performed only after identifying vertebra C4. Next, DICOM formats were assessed using digital image analysis of Osirix 3.9 (Fig. 1b–d) with estimating linear (sagittal and transverse diameters, height, length, width), two-dimensional (cross-sectional area), and three-dimensional (volume) parameters of vertebra C4. The contouring procedure of each C4 vertebral body and the three ossification centers was outlined with a cursor and recorded.
The five following parameters of the C4 vertebral body were evaluated for each fetus:
1. height (in mm), corresponding to the distance between the superior and inferior borderlines of the vertebral body (in sagittal projection),
2. transverse diameter (in mm), corresponding to the distance between the left and right borderlines of the vertebral body (in transverse projection),
3. sagittal diameter (in mm), corresponding to the distance between the anterior and posterior borderlines of the vertebral body (in sagittal projection),
4. cross-sectional area (in mm2), traced around the vertebral body (in transverse projection), and
5. volume (in mm3).
In addition, the 12 following parameters of the three ossifications centers were assessed for each fetus: within the vertebral body (6–9):
6. transverse diameter (in mm), corresponding to the distance between the left and right borderlines of the ossification center (in transverse projection),
7. sagittal diameter (in mm), corresponding to the distance between the anterior and posterior borderlines of the ossification center (in sagittal projection),
8. cross-sectional area (in mm2), traced around the ossification center (in transverse projection),
9. volume (in mm3), and within the right and left neural processes (10–17):
10, 11. right and left lengths (in mm), corresponding to the distance between the anterior and posterior borderlines of the ossification center (in transverse projection),
12, 13. right and left widths (in mm), corresponding to the distance between the left and right borderlines of the ossification center (in transverse projection),
14, 15. right and left cross-sectional areas (in mm2), traced around the ossification center (in transverse projection),
16, 17. right and left volumes (in mm3).
In a continuous effort to minimize measurements and observer bias, all the measurements were performed by one researcher (M.B). Each measurement was repeated three times under the same conditions, but at different times, and the mean of the three was finally used. The findings obtained were subjected to statistical analysis. The intra-observer variation was assessed by the Wilcoxon signed-rank test. All the parameters examined were plotted versus gestational age to construct their growth dynamics. The relative growth, both at the vertebral body and its ossification center, was expressed as the sagittal-to-transverse diameter ratios and the ossification center-to-vertebral body volume ratio. The data obtained was checked for normality of distribution using the Kolmogorov–Smirnov test and homogeneity of variance with the use of Levene’s test. As a consequence of the statistical analysis, Student’s t test was used to examine the impact of sex on the values obtained. In order to examine sex differences, we tested possible differences between the five following age groups: 17–19, 20–22, 23–25, 26–28, and 29–30 weeks. Next, we checked sex differences for the whole examined group, without taking into account gestational age. To check whether variables changed significantly with age, the one-way ANOVA test and the post hoc RIR Tukey test were used for the five age groups mentioned above. Linear and nonlinear regression analysis was used to derive the best-fit curve for each parameter studied versus gestational age, with estimating coefficients of determination (R 2) between each parameter and fetal age.
Results
No statistically significant differences were observed in assessing intra-observer reproducibility of the spinal measurements. In addition, no significant difference was found in the values of the parameters studied according to sex, so the morphometric values for vertebra C4 (Table 2) and its ossification centers (Tables 3, 4) have been summarized for both sexes. By contrast, a statistically significant (P = 0.0000) increase in values of all the measurements was accompanied by advancing gestational age. The numerical data correlated to gestational age presented differentiated growth dynamics, expressed by specific best-fit growth curves (Figs. 2, 3 and 5–8).
The size of the C4 vertebral body has been shown in Table 2. The values of the vertebral body height rose from 2.07 to 3.81 ± 0.21 mm for fetuses aged 17 and 30 weeks, respectively. With advancing gestational age, an increase in height (Fig. 2a) followed logarithmically as y = −3.866 + 2.225 × ln(Age) ± 0.238 (R 2 = 0.69). Between ages of 17 and 30 weeks, the transverse diameter of the vertebral body (Fig. 2b) attained the values from 2.90 to 5.31 ± 0.28 mm, in accordance with the logarithmic function: y = −7.077 + 3.547 × ln(Age) ± 0.356 (R 2 = 0.72). During the duration of the study period, the values of sagittal diameter of the vertebral body (Fig. 2c) increased logarithmically from 2.77 to 3.95 ± 0.24 mm, following the formula: y = −3.886 + 2.272 × ln(Age) ± 0.222 (R 2 = 0.73). Consequently, at ages of 17 and 30 weeks, the growth velocities (mm per week) for height and transverse and sagittal diameters of the vertebral body gradually declined with advancing fetal age (P < 0.01), from 0.13 to 0.08 mm, 0.20 to 0.22 mm, and 0.13 to 0.08 mm, respectively. The relative growth of the C4 vertebral body was not proportionate, since the transverse diameter grew much faster than the sagittal diameter. This was expressed by the decrement of the sagittal-to-transverse diameter ratio (Fig. 2d) from 0.84 ± 0.07 to 0.77 ± 0.06 (P < 0.01). The values of cross-sectional area of the vertebral body (Fig. 3a) ranged from 7.50 to 18.33 ± 1.70 mm2 in fetuses aged 17 and 30 weeks respectively, and generated the linear function y = −7.205 + 0.812 × Age ± 1.668 (R 2 = 0.76). During that time the volumetric growth of the vertebral body (Fig. 3b), from 15.53 to 72.43 ± 9.46 mm3, modeled the four-degree polynomial regression y = 14.108 + 0.00007 × Age4 ± 6.289 (R 2 = 0.83).
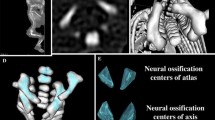
The size of the ossification center of the C4 vertebral body has been presented in Table 3, while Fig. 4 presents the three ossification centers of vertebra C4 within its body (1), and right (2) and left (3) neural processes in fetuses aged 17, 22, 26, and 30 weeks, respectively. During the analyzed period, the transverse (Fig. 5a) and sagittal (Fig. 5b) diameters of the ossification center of the vertebral body grew logarithmically from 1.60 to 3.74 ± 0.44 mm, and from 1.32 to 3.37 ± 0.12 mm, according to the following models: y = − 8.836 + 3.708 × ln(Age) ± 0.334 (R 2 = 0.76) and y = − 7.748 + 3.240 × ln(Age) ± 0.237 (R 2 = 0.83), respectively. As a result, the growth dynamics for transverse and sagittal diameters decreased with gestational age, from 0.21 to 0.13 mm per week, and from 0.19 to 0.11 mm per week (P < 0.01), respectively. During the study period, the sagittal-to-transverse diameter ratio of the ossification center (Fig. 5c) increased from 0.86 ± 0.04 to 0.88 ± 0.11 (P < 0.05). The cross-sectional area of the ossification center (Fig. 5d) increased linearly from 3.70 mm2 in fetuses aged 17 weeks to 10.23 ± 3.14 mm2 in fetuses aged 30 weeks, according to the function: y = −4.690 + 0.437 × Age ± 1.172 (R 2 = 0.63). Similarly, the volumetric growth of the ossification center (Fig. 6a), from 4.67 to 13.45 ± 3.20 mm3, followed linearly as y = −5.917 + 0.582 × Age ± 1.157 (R 2 = 0.77).
The volumetric growth of the C4 vertebral body and its ossification center (Fig. 6b) is presented in a relative manner by the ossification center-to-vertebral body volume ratio. As shown in Fig. 6c, its value gradually decreased from 0.23 ± 0.04 to 0.21 ± 0.03 during the study period (P < 0.05).
The size of ossification centers of the neural processes has been listed in Table 4. Although the right–left differences for the entire group were not statistically significant, the results have already been presented separately for each neural process, because of their great inter-individual variability. The ossification center of the neural process grew in length from 3.40 to 7.55 ± 0.32 mm on the right (Fig. 7a), and from 3.49 to 7.38 ± 0.30 mm on the left (Fig. 7b), in correspondence with the logarithmic functions: y = −19.601 + 8.018 × ln (Age) ± 0.369 (R 2 = 0.92) and y = −15.804 + 6.912 × ln(Age) ± 0.471 (R 2 = 0.85), respectively. Its width increased from 1.66 to 2.67 ± 0.27 mm on the right (Fig. 7c), and from 1.62 to 2.59 ± 0.32 mm on the left (Fig. 7d), following the logarithmic functions: y = −5.806 + 2.587 × ln(Age) ± 0.146 (R 2 = 0.88) and y = −5.621 + 2.519 × ln(Age) ± 0.146 (R 2 = 0.88), respectively. The cross-sectional area of the ossification center for the neural process revealed an increase from 6.40 to 18.05 ± 7.36 mm2 on the right (Fig. 8a), and from 4.60 to 15.03 ± 5.60 mm2 on the left (Fig. 8b), as the linear functions: y = −9.188 + 0.856 × Age ± 2.174 (R 2 = 0.67) and y = −7.570 + 0.768 × Age ± 2.200 (R 2 = 0.60), respectively. The growth in volume of the right (Fig. 8c) and left (Fig. 8d) ossification centers of the neural processes varied from 8.37 to 21.53 ± 3.15 mm3, and from 6.17 to 19.83 ± 2.44 mm3, respectively, following the linear functions: y = −13.802 + 1.222 × Age ± 1.872 (R 2 = 0.84), and y = −11.038 + 1.061 × Age + 1.964 (R 2 = 0.80).
Discussion
The spine starts to ossify in the 8th gestational week [4] and from the 9th week it can be monitored ultrasonographically. In fetuses aged 11 weeks, ossification centers are detectable within the T2–L2 vertebral bodies and the C1–L1 neural processes [3]. Histological studies showed mineralization in much younger specimens than radiological observations [4]. The ossification timing was found to be significantly earlier in females than in males [31]. Vertebra S5 is just one example of this, because ossification centers were identified in its body and neural processes in 42.9 and 28.6 % of the females, respectively, and in no one male at the same gestational age [31]. In this aspect, our findings do not correspond with the existing literature, because no statistically significant sex differences were found in the material under examination. The possible explanation to this may be partly attributed either to the great inter-individual variability of the fetuses examined or to the different methods used.
Assessment of the fetal vertebral column in both transverse and parasagittal planes is an integral part of routine ultrasound scanning [24]. The cervical spine length has previously been reported to be linear [22], parabolic [2], or exponential [24] when related to advancing gestational age. Bagnall et al. [2] showed that in fetuses aged 8–26 weeks, the cervical spine length grew parabolically from 9 to 27 mm, being precisely expressed by the quadratic function y = −10.28 + 107.98 × Age − 67.35 × Age2 (R = 0.90, Age—in years) with a negative coefficient of power 2 causing a gradually decreasing growth rate. Although the entire presacral spine showed slowed down growth, both the cervical and lumbar parts slowed down to approximately half the growth rate of the thoracic part [2]. Therefore, in fetuses at the age of 8 and 26 weeks, the cervical part of the spine was about 60 % that of the thoracic part. Furthermore, the length of the “average” cervical unit (vertebra plus disc) at 26 weeks of gestation attained the value of 3.9 mm. Dimeglio et al. [9, 10] presented the longitudinal growth of the cervical spine from birth to maturity. At birth, its length measured 3.7 cm, and grew approximately by 9 cm to reach the adult size of 12–13 cm. It should be emphasized that the cervical spine doubled its length around 6 years of age and gained about 3.5 cm during the pubertal growth spurt. In our opinion, the aforementioned numerical data support that in children and adolescents, the cervical spine shows slowing down of its lengthwise growth, maybe even in a quasi-logarithmic fashion. Thus, the evolving vertebra seems to grow in the same manner both in fetuses and children. In addition, intervertebral discs accounted for the cervical spinal length, approximately 30 % at birth and 22 % at maturity [9, 10]. As reported by Tulsi [28], the heights of all cervical vertebrae continued to increase until adulthood by 39–45 % between 2–4 and 17–19 years.
In the present study, the height and transverse and sagittal diameters of the C4 vertebral body did not create linear, quadratic, or exponential functions on nomograms. In fact, we proved that the best-fit growth models were the following logarithmic functions: y = −3.866 + 2.225 × ln (Age) ± 0.238 for its height, y = −7.077 + 3.547 × ln (Age) ± 0.356 for its transverse diameter, and y = −3.886 + 2.272 × ln (Age) ± 0.222 for its sagittal diameter. As a consequence, their growth velocity gradually decreases with age, as previously reported by Bagnall et al. [2]. According to Tulsi [28], between 2–4 years and adulthood, the transverse and sagittal diameters increased by 6 % (6–12 %) and 33 % (20–33 %), respectively. In the material under examination, the vertebral body did not show a proportionate evolution because the sagittal-to-transverse diameter ratio declined from 0.84 ± 0.07 to 0.77 ± 0.06 during the duration of the analyzed period. Since both the transverse and sagittal diameters of the vertebral body increased logarithmically, its cross-sectional area being approximately a product of these two diameters computed the linear fashion y = −7.205 + 0.812 × Age ± 1.668.
The overall growth rate of the vertebral body was best expressed by measuring its volume [28]. Schild et al. [24] presented three-dimensional sonographic volume calculation of the T12–L5 vertebral bodies in fetuses aged 16–37 weeks. Their growth in volume varied in correspondence (P < 0.01) with exponential functions. It is noteworthy that in the material under examination, the vertebral body volume varied from 15.53 to 72.43 ± 9.46 mm3, with the best-fit model for volume presented by the four-degree polynomial function y = 14.108 + 0.00007 × Age4 ± 6.289. This model may probably result from multiplying the three values for height and transverse and sagittal diameters, each changing logarithmically. Postnatally, an increase in volume of the cervical vertebrae by 58–68 % was reported between 2–4 and 17–19 years, but without any regression models [28].
After reviewing the medical literature on developmental pathways of vertebral ossification centers, we failed to find any data for their dimensions [2–4, 22, 31]. Thus, the present study is the first to provide the literature with completely novel reference values and growth dynamics for length, width, cross-sectional area and volume of the three ossification centers of vertebra C4 in human fetuses. As illustrated in Tables 3, 4 and Fig. 4, the ossification center of the vertebral body offered a sharp contrast, being much larger than that of each neural process. However, it should be emphasized that the growth dynamics for all the three ossification centers of vertebra C4 were similar to each other. As a result, both their transverse and sagittal diameters increased logarithmically, while both their cross-sectional areas and volumes generated straight lines. It is important to note, however, that the sagittal-to-transverse diameter ratio of ossification center of the vertebral body was found to increase with gestational age from 0.86 ± 0.04 to 0.88 ± 0.11. It should also be noted that the vertebral body and its ossification center grew in volume according to the four-degree polynomial (y = 14.108 + 0.00007 × Age4 ± 6.289) and linear (y = − 5.917 + 0.582 × Age ± 1.157) functions, respectively. As a consequence, the relative size of the ossification center gradually declined with age, from 0.23 ± 0.04 at 17 weeks to 0.21 ± 0.03 at 30 weeks of gestation.
As far as the neural processes are concerned, their left and right ossification centers developed symmetrically, with no laterality differences. On the right and left sides, both their lengths (y = −19.601 + 8.018 × ln (Age) ± 0.369, y = −15.804 + 6.912 × ln(Age) ± 0.471) and widths (y = −5.806 + 2.587 × ln(Age) ± 0.146, y = −5.621 + 2.519 × ln(Age) ± 0.146) increased logarithmically. On the other hand, both their cross-sectional areas (y = −9.188 + 0.856 × Age ± 2.174, y = −7.570 + 0.768 × Age ± 2.200) and volumes (y = −13.802 + 1.222 × Age ± 1.872, y = −11.038 + 1.061 × Age + 1.964) generated straight lines. Such morphometric data have not been previously reported, thereby limiting discussion on quantitative anatomy of ossification centers. Ossification progression within the neural processes is of relevance in the diagnosis of neural tube defects [4, 11, 17, 18].
Due to age-specific reference values for vertebra C4, such abnormalities as hemivertebra, butterfly vertebra, block vertebrae, and spina bifida may ultrasonographically be diagnosed and monitored in fetuses [32]. Hemivertebra is characterized by a wedge-shaped vertebra with the absence (aplasia) of one of the two chondrification centers within the vertebral body, resulting in substantial deformity of the spine [15] in its sagittal and coronal alignment. Butterfly vertebra refers to the failure of fusion of two chondrification centers with the persistent notochord separating them [7, 23]. Both hemivertebra and butterfly vertebra may be associated with skeletal anomalies [12], diastematomyelia [20], cardiac, urogenital and gastrointestinal anomalies, and some conditions including Jarcho–Levin, Klippel-Fiel, VATER, VACTERL, and OEIS syndromes [30]. Block vertebrae are the consequence of their mal-segmentation and fusion through neighboring intervertebral discs. Spina bifida is characterized by a midline cleft between the two neural processes [5, 13, 18, 27]. Furthermore, detailed knowledge on the normal growth of spinal ossification centers in fetuses may be helpful in the prenatal diagnosis of skeletal dysplasias (osteochondrodysplasias). Such dysplasias result in both delayed ossification centers and widespread demineralization, typical of osteogenesis imperfecta type II [29], achondrogenesis [26], and thanatophoric dysplasia type I [29]. In infants with life-threatening conditions, inorganic pyrophosphate is accumulated extracellularly, resulting in both rickets and osteomalacia and finally in progressive chest and spine deformity [34].
The main limitation of this study is a relatively narrow fetal age, ranging from 17 to 30 weeks of gestation. Were we to collect a larger fetal sample with a wider age range, it would be possible to improve the growth curves obtained. Another partial limitation may be that all measurements were performed by one observer in a blind fashion. Finally, our results have been presented as if describing a sequential process in one specimen, even though the data were obtained from the cross-sectional study of 55 fetuses.
In summary, this is a cross-sectional study that describes the normative data of fetal vertebra C4 and documents its evolution. Our reference values for vertebra C4 and its three ossification centers may facilitate the diagnosis of many spinal disorders in human fetuses.
Conclusions
-
1.
No sex differences are found in the morphometric parameters of growing vertebra C4 and its three ossification centers.
-
2.
The C4 vertebral body increases logarithmically in height and both sagittal and transverse diameters, linearly in cross-sectional area, and four-degree polynomially in volume.
-
3.
The three ossification centers of vertebra C4 grow logarithmically in both transverse and sagittal diameters, and linearly in both cross-sectional area and volume.
-
4.
The age-specific reference intervals for evolving vertebra C4 may be useful in the prenatal diagnosis of congenital spinal defects.
References
Abe S, Suzuki M, Cho KH, Murakami G, Cho BH, Ide Y (2011) CD34-positive developing vessels and other structures in human fetuses: an immunohistochemical study. Surg Radiol Anat 33:919–927
Bagnall KM, Harris PF, Jones PRM (1979) A radiographic study of the human fetal spine. 3. Longitudinal development of the ossification centres. J Anat 128:777–787
Bareggi R, Grill V, Zweyer M, Narducci P, Forabosco A (1994) A quantitative study on the spatial and temporal ossification patterns of vertebral centra and neural arches and their relationship to the fetal age. Ann Anat 176:311–317
de Biasio P, Ginocchio G, Aicardi G, Ravera G, Venturini PL (2003) Ossification timing of sacral vertebrae by ultrasound in the mid-second trimester of pregnancy. Prenat Diagn 23:1056–1059
Chen Y, Zhuang Z, Qi W, Yang H, Chen Z, Wang X, Kong K (2011) A three-dimensional study of the atlantodental interval in a normal Chinese population using reformatted computed tomography. Surg Radiol Anat 33:801–806
Cho KH, Rodríguez-Vázquez JF, Kim JH, Abe H, Murakami G, Cho BH (2011) Early fetal development of the human cerebellum. Surg Radiol Anat 33:523–530
Chrzan R, Podsiadlo L, Herman-Sucharska I, Urbanik A, Bryll A (2010) Persistent notochordal canal imitating compression fracture—plain film, CT and MR appearance. Med Sci Monit 16:76–79
Cui G, Watanabe K, Hosogane N, Tsuji T, Ishii K, Nakamura M, Toyama Y, Chiba K, Lenke LG, Matsumoto M (2012) Morphologic evaluation of the thoracic vertebrae for safe free-hand pedicle screw placement in adolescent idiopathic scoliosis: a CT-based anatomical study. Surg Radiol Anat 34:209–216
Dimeglio A, Bonnel F, Canavese F (2011) Normal growth of the spine and thorax. In: Akbarnia BA, Yazici M, Thompson GH (eds) The growing spine. Springer, Berlin, Heidelberg, pp 13–42
Dimeglio A, Canavese F, Charles YP (2011) Growth and adolescent idiopathic scoliosis: when and how much? J Pediatr Orthop 32(Suppl 1):28–36
Doukas A, Petridis AK (2010) A case of aplasia of the posterior arch of the atlas mimicking fracture: review of the literature. Clin Anat 23:881–882
Goldstein I, Makhoul IR, Weissman A, Drugan A (2005) Hemivertebra: prenatal diagnosis, incidence and characteristics. Fetal Diagn Ther 20:121–126
van der Hof MC, Nicolaides KH, Campbell J, Campbell S (1990) Evaluation of the lemon and banana signs in one hundred thirty fetuses with open spina bifida. Am J Obstet Gynecol 162:322–327
Iffy L, Jakobovits A, Westlake W, Wingate MB, Caterini H, Kanofsky P, Menduke H (1975) Early intrauterine development: I. The rate of growth of Caucasian embryos and fetuses between the 6th and 20th weeks of gestation. Pediatrics 56:173–186
Jalanko T, Rintala R, Puisto V, Helenius I (2011) Hemivertebra resection for congenital scoliosis in young children: comparison of clinical, radiographic, and health-related quality of life outcomes between the anteroposterior and posterolateral approaches. Spine 36:41–49
Jin ZW, Song KJ, Lee NH, Nakamura T, Fujimiya M, Murakami G, Cho BH (2011) Contribution of the anterior longitudinal ligament to ossification and growth of the vertebral body: an immunohistochemical study using the human fetal lumbar vertebrae. Surg Radiol Anat 33:11–18
Kibii JM, Pan R, Tobias PV (2010) Morphometric variations of the 7th cervical vertebrae of Zulu, White, and colored South Africans. Clin Anat 23:399–406
Kumar A, Tubbs RS (2011) Spina bifida: a diagnostic dilemma in paleopathology. Clin Anat 24:19–33
Kwan MK, Jeffry A, Chan CY, Saw LB (2012) A radiological evaluation of the morphometry and safety of S1, S2 and S2—ilium screws in the Asian population using three dimensional computed tomography scan: an analysis of 180 pelvis. Surg Radiol Anat 34:217–227
Leug YL, Buton N (2005) Combined diastematomyelia and hemivertebra. A review of the management at a single centre. J Bone Joint Surg 87:1380–1384
Matsumoto M, Okada E, Kaneko Y, Ichihara D, Watanabe K, Chiba K, Toyama Y, Fujiwara H, Momoshima S, Nishiwaki Y, Hashimoto T, Takahata T (2011) Wedging of vertebral bodies at the thoracolumbar junction in asymptomatic healthy subjects on magnetic resonance imaging. Surg Radiol Anat 33:223–228
Noback CR, Robertson GG (1951) Sequences of appearance of ossification centers in the human skeleton during the first five prenatal months. Am J Anat 89:1–28
Patinharayil G, Han CW, Marthya A, Meethall KC, Surendran S, Rudrappa GH (2008) Butterfly vertebra: an uncommon congenital spinal anomaly. Spine 15(33):926–928
Schild RL, Wallny T, Fimmers R, Hansmann M (2000) The size of the fetal thoracolumbar spine: a three-dimensional ultrasound study. Ultrasound Obstet Gynecol 16:468–472
Szpinda M, Daroszewski M, Woźniak A, Szpinda A, Mila-Kierzenkowska C (2012) Tracheal dimensions in human fetuses: an anatomical, digital and statistical study. Surg Radiol Anat 34:317–323
Taner MZ, Kurdoglu M, Taskiran C, Onan MA, Gunaydin G, Himmetoglu O (2008) Prenatal diagnosis of achondrogenesis type I: a case report. Cases J 1:406. doi:10.1186/1757-1626-1-406
Travan L, Saccheri P, Sabbadini G, Crivellato E (2011) Bilateral arcuate foramen associated with partial defect of the posterior arch of the atlas in a medieval skeleton: case report and review of the literature. Looking backward to go forward. Surg Radiol Anat 33:495–500
Tulsi RS (1971) Growth of the human vertebral column: an osteological study. Acta Anat 79:570–580
Ulla M, Aiello H, Cobos MP, Orioli I, García-Mónaco R, Etchegaray A, Igarzábal ML, Otaño L (2011) Prenatal diagnosis of skeletal dysplasias: contribution of three-dimensional computed tomography. Therapy Fetal Diagn 29:238–247
Varras M, Akrivis C (2010) Prenatal diagnosis of fetal hemivertebra at 20 weeks’ gestation with literature review. Int J Gen Med 3:197–201
Vignolo M, Ginocchio G, Parodi A, Torrisi C, Pistorio A, Venturini PL, Aicardi G, de Biasio P (2005) A fetal spine ossification: the gender and individual differences illustrated by ultrasonography. Ultrasound Med Biol 31:733–738
Wax JR, Watson WJ, Miller RC, Ingardia CJ, Pinette MG, Cartin A, Grimes CK, Blackstone J (2008) Prenatal sonographic diagnosis of hemivertebrae: associations and outcomes. J Ultrasound Med 27:1023–1027
Weisz B, Achiron R, Schindler A, Eisenberg VH, Lipitz S, Zalel Y (2004) Prenatal sonographic diagnosis of hemivertebra. J Ultrasound Med 23:853–857
Whyte MP, Greenberg CR, Salman NJ, Bober MB, McAlister WH, Wenkert D, Van Sickle BJ, Simmons JH, Edgar TS, Bauer ML, Hamdan MA, Bishop N, Lutz RE, McGinn M, Craig S, Moore JN, Taylor JW, Cleveland RH, Chanley WR, Lim R, Thacher TD, Mayhew JE, Downs M, Millan JL, Skrinar AM, Crine P, Landy H (2012) Enzyme-replacement therapy in life-threatening hypophosphatasia. N Engl J Med 366:904–913
Conflict of interest
The authors declare that they have no conflict of interest.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Baumgart, M., Szpinda, M. & Szpinda, A. New anatomical data on the growing C4 vertebra and its three ossification centers in human fetuses. Surg Radiol Anat 35, 191–203 (2013). https://doi.org/10.1007/s00276-012-1022-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-012-1022-z