Abstract
Throughout northern Africa, evidence for an intensification of wild grass gathering is reflected in Holocene archaeological contexts. However, both the recovery of macrobotanical assemblages and the specificity of their taxonomic classification are heavily influenced by food processing and post-depositional conditions. In contrast, inflorescence phytoliths provide high levels of taxonomic specificity and preserve well in most archaeological contexts. This study analyses the in situ morphology of inflorescence phytoliths from modern specimens of nine wild C4 grass species commonly observed in ethnographic studies and recovered in seed assemblages from archaeological contexts across northern Africa. Morphological differences in Interdigitate phytoliths within the fertile florets of six Paniceae species enabled differentiation between them. The morphological parameters established in this study provide an additional resource for archaeological and palaeoecological analyses using phytoliths, which demonstrates the effectiveness of applying this method to African wild grass species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although the systematic use of wild grasses has diminished over the last few centuries, they have nonetheless formed a key component of non-agrarian subsistence throughout global history (Harlan 1975; Harris 1984; Nesbitt 1997). In northern Africa, 19th century European explorers frequently documented extensive market economies revolving around the management and consumption of an array of wild grasses (Barth 1857; Duveyrier 1864; Heuglin 1869; Nachtigal 1881). Across the central Sahel region, from Nigeria to Sudan, for example, dozens of species known locally as kreb were routinely collected and extensively traded (Barth 1857; Nachtigal 1881). While published accounts continued to remark upon these subsistence systems during the 20th century (Chevalier 1932; Nicholaisen 1963; Jardin 1967; Tubiana and Tubiana 1977; Harlan 1989a), the consumption of wild grasses has become increasingly associated with famine and considered marginal to modern subsistence (Batello et al. 2004). Despite this diminished economic role, wild grasses are still a dietary staple amongst northern African subsistence farmers and pastoral communities (Batello et al. 2004).
Based on contemporary observations and ethnographic accounts, Harlan (1989a) defined three broad ecological zones within northern Africa with extensive cultural use of specific wild grass species: desert (Aristida pungens, Cenchrus biflorus and Panicum turgidum), swamp (Brachiaria, Echinochloa, Oryza and Paspalum spp.) and savannah (Dactyloctenium, Digitaria, Echinochloa, Eleusine, Eragrostis, Latipes, Oryza, Panicum, Pennisetum, Setaria and Sorghum spp.) (Fig. 1). Within these areas, variations in species selection reflect both local availability and cultural preferences (Barth 1857; Nachtigal 1881; Harlan 1989a; Batello et al. 2004). Of particular interest is the highly varied complex of savannah grasses collectively known in the central Sahel as kreb (Harlan 1989a). Species such as Dactyloctenium aegyptium, Panicum laetum and various Eragrostis spp. are frequently described as components of kreb in ethnographic accounts, however, numerous other taxa are also routinely associated with this complex of grasses (Barth 1857; Nachtigal 1881; Harlan 1989a; Batello et al. 2004). These species are predominately xerophytes and mesophytes in the Panicoideae or Chloridoideae subfamilies, but also hydrophytic taxa, including various Brachiaria, Echinochloa, Paspalum and Oryza spp. used in areas with seasonally standing water (Harlan 1989a; Batello et al. 2004).

adapted from Harlan 1989a) and distribution of Holocene archaeological sites with genus or species level evidence of wild grasses belonging to these groups. The median age of the earliest phase of occurrence for each site is represented; for the list of sites and references see ESM 1
Map of northern Africa indicating broad regions of recent wild grass use (desert, savannah/kreb and swamp,
Numerous ethnographers have noted the importance of kreb to human communities, observable through the routine management and protection of natural grass stands from overgrazing (Barth 1857; Nachtigal 1881; Batello et al. 2004). The Sahelian kreb species form dense stands during the wet season and are easily harvested by transhumant pastoralists and subsistence farmers as they mature in the early dry season in October–November (Nachtigal 1881). Hydrophytes, like wild Oryza barthii, O. staminata or wetland panicoids, are normally available for gathering later and are more easily reached as floods recede well into the dry season by January (Barth 1857; Batello et al. 2004). While methods vary between regions and communities, these dehiscent grass seeds are often gathered by sweeping a basket or container through ripe stands (Barth 1857; Nicholaisen 1963; Harlan 1989a; Batello et al. 2004). The grains can be consumed by people as seed cakes without the removal of the husk by winnowing (Nicholaisen 1963). The preparation of flat breads, porridge, flour, gruel, couscous and beer, on the other hand, typically involves processing by parching, pounding and winnowing of the grains (Harlan 1989a).
Initially inferred through the frequent recovery of grindstones, direct archaeobotanical remains of these species are now consistently found in northern African Holocene deposits (Fig. 1; Harlan 1989a). Evidence for regional intensification of wild grass gathering coincides with the increasing environmental instability of the middle Holocene (Clark 1976; Harlan 1989a; Radomski and Neumann 2011; Mercuri et al. 2018; Fuller et al. 2019). Regionally diverse selection practices are evident in middle and late Holocene archaeological deposits, with numerous species displaying phenotypic modifications derived from intensive management strategies, including Echinochloa and Panicum spp. in southwest Libya (Mercuri et al. 2018). While cultural management of some taxa diminished over time, prolonged selection pressures on other grasses resulted in the complete domestication of Sorghum bicolor (sorghum) in the Sudan (Winchell et al. 2017; Beldados 2019; Barron et al. 2020), Pennisetum glaucum (pearl millet) in the western Sahel (Manning et al. 2011; Fuller et al. 2021), as well as less well-documented processes for Oryza glaberrima (African rice), Eleusine coracana (finger millet), Eragrostis tef (teff) and Digitaria exilis (fonio) (Harlan 1989b; D’Andrea 2008; Giblin and Fuller 2011; Champion et al. 2021).
The archaeological identification of African wild grasses is typically achieved from the recovery of their charred or desiccated macrobotanical remains (Wasylikowa et al. 1997; Barakat and Fahmy 1999; Fahmy 2001; Mercuri and Garcea 2007; Murray et al. 2007; Pelling 2007; Thanheiser 2011; McDonald 2016; Mercuri et al. 2018; Brass et al. 2019) and analysis of spikelet and seed impressions within pottery (Munson 1976; Costantini et al. 1982; Magid 1984, 1989; Stemler 1990; Klee et al. 2000; Fuller et al. 2007, 2021; Beldados 2019; Barron et al. 2020). Microbotanical techniques, including analyses of pollen (Mercuri 1999, 2008; Mercuri and Garcea 2007; Florenzano et al. 2016), starch granules (Buckley et al. 2014; Madella et al. 2014) and particularly phytoliths (Neumann et al. 2009; Radomski and Neumann 2011; Madella et al. 2014; Out et al. 2016; Ryan 2018) have also been used to successfully detect the presence of various grass taxa at archaeological sites.
While species level identifications are often possible with exceptionally well preserved desiccated macrobotanical remains (Mercuri et al. 2018), the identification of charred material is usually more taxonomically limited and largely dictated by conditions of processing, state of charring and post-depositional taphonomy. Chaff remains (glumes, lemmas and paleas) from large-seeded domestic cereals are often preserved in archaeological contexts, however, charring typically destroys smaller and less robust chaff (Wilson 1984; Harvey and Fuller 2005; Märkle and Rösch 2008; Stevens et al. 2021). The small seed size (generally < 2 mm) of economic wild grasses significantly influences the recovery of charred material with diagnostic features required for confident species level identifications (Harvey and Fuller 2005; Märkle and Rösch 2008; Weber and Fuller 2008).
In contrast to these macroremains, inorganic phytoliths are highly resilient to degradation in most sedimentary environments (Piperno 2006). Formed through the uptake, polymerisation and deposition of mono-silicic acid within the plant cell walls, lumen and intercellular spaces, phytoliths from archaeological contexts usually represent the in situ decay of plant material (Piperno 2006). The archaeological recovery and identification of phytoliths enables reconstructions of human use of plants and local vegetation composition during site formation. Since the 1990s, phytoliths have been increasingly used in African archaeology as environmental proxies and in reconstructions of human use of plants (for example, Barboni et al. 1999; Mercader et al. 2000, 2010, 2011; Fahmy and Magnavita 2006; Barboni and Bremond 2009; Neumann et al. 2009, 2017; Eichhorn et al. 2010; Radomski and Neumann 2011; Wadley et al. 2011, 2020; Novello et al. 2012; Madella et al. 2014, 2016; Novello and Barboni 2015; Out et al. 2016; Collura and Neumann 2017; Esteban et al. 2017; Murungi 2017; Murungi et al. 2017; Out and Madella 2017; Ryan 2018).
While various phytolith taxonomies have been established for the African flora, current phytolith classification indices for wild African grasses remain largely restricted to disarticulated short cell morphotypes (Fahmy 2008; Mercader et al. 2010; Radomski and Neumann 2011; Neumann et al. 2017; Bourel and Novello 2020).
Researchers have recently demonstrated the high taxonomic potential of articulated phytoliths from grass inflorescence bracts (glumes, lemmas and paleas), particularly Interdigitate morphotypes within the lemma and palea of the fertile floret (Lu et al. 2009; Madella et al. 2014; Out et al. 2016; Zhang et al. 2017; Bhat et al. 2018; Ge et al. 2018, 2020). To date, archaeological studies of Interdigitate phytoliths have mainly focused on establishing classification criteria for Eurasian domesticated millets and related wild Panicoideae taxa, with combined morphological and morphometric parameters enabling differentiation of closely related species (Lu et al. 2009; Zhang et al. 2011, 2017, 2018; Kealhofer et al. 2015; Yang et al. 2015; Weisskopf and Lee 2016; Bhat et al. 2018; Ge et al. 2018, 2020). Originally described as an ‘interdigitating’ phytolith pattern by Parry and Hodson (1982), Interdigitate phytoliths are situated between the epidermal cuticle layer and the Elongate cells on the abaxial (outer) surface of the fertile floret. In contrast to Elongate morphotypes, Interdigitate phytoliths are typified by a thin layer of cells that, based on archaeological recovery, seem to be fused together and do not disarticulate into individual cells (Lu et al. 2009; Madella et al. 2014; Out et al. 2016; Weisskopf and Lee 2016; Zhang et al. 2017; Deng et al. 2018; Luo et al. 2019; An et al. 2021). Rather, published photographs of this morphotype in archaeological assemblages suggest that Interdigitate phytolith layers fracture when damaged, preserving fused segments consisting of multiple cells with anatomical characteristics in situ (Lu et al. 2009; Madella et al. 2014; Out et al. 2016; Weisskopf and Lee 2016; Zhang et al. 2017; Deng et al. 2018; Luo et al. 2019; An et al. 2021).
Initial classifications of Interdigitate phytoliths primarily focused on using undulation type and level (amplitude/length of projections along the lateral cell margins) combined with shape variables (height/width ratios) to differentiate Panicum miliaceum and Setaria italica (Lu et al. 2009; Weisskopf and Lee 2016). Subsequent studies have examined a wider array of taxa, with an emphasis on differentiating domesticates from closely related wild species (Zhang et al. 2011, 2018; Kealhofer et al. 2015; Ge et al. 2018, 2020). Zhang et al. (2018) explored the link between the frequency of level III undulations in Interdigitate phytoliths, grain size and growing conditions by comparing samples of P. miliaceum with wild P. repens and P. ruderale from different environmental regions. The results demonstrated a strong correlation, irrespective of growing environment, between greater numbers of Interdigitate phytoliths with level III undulations and a reduction in grain length/width ratio in domesticated P. miliaceum (Zhang et al. 2018). This suggests a correlation between Interdigitate undulation amplitude and seed size, which is primarily under genetic control (Sadras 2007). However, other studies have indicated that growing conditions may also influence Interdigitate size (Ge et al. 2018) and increase intraspecific variability of height/width shape ratios derived from Interdigitate phytoliths (Zhang et al. 2011; Kealhofer et al. 2015). While phytolith shape variables are less plastic and therefore more reliable than size variables (Portillo et al. 2020), researchers have recommended the analysis of multiple populations to account for intraspecific variation linked to growing environment (Ball and Brotherson 1992; Zhang et al. 2011; Kealhofer et al. 2015; Out and Madella 2016, 2017). Furthermore, studies of interspecific variation between numerous closely related taxa indicate that taxonomic identifications based on Interdigitate shape ratios may be limited to genus level (Kealhofer et al. 2015; Ge et al. 2020). Interestingly, a recent study of Interdigitate phytoliths by Ge et al. (2020) suggests that the comparison of multiple morphological characteristics (undulation type and amplitude, shape of terminal margins and shape of the central cell body) as well as Papillate presence, could achieve similar classification levels to morphometric shape ratios, and in some cases, species classifications (Ge et al. 2020).
Despite these advances in the identification of inflorescence phytoliths from small-seeded wild grasses, taxonomic criteria for African species based on articulated phytoliths, and particularly Interdigitate morphotypes, is limited. While preliminary studies on African grasses have indicated the potential in establishing comprehensive classification parameters, these have primarily focused on glumes rather than lemmas or paleas (Madella et al. 2016) and identifying select Interdigitate morphotypes recovered from archaeological contexts (Madella et al. 2014; Out et al. 2016; Ryan 2018). As such, there remains a need for the development of dedicated articulated inflorescence classification criteria for a wide range of taxa in order to differentiate wild African grass species. To help address this gap, this study analyses the in situ morphological characteristics of inflorescence phytoliths from nine species commonly found in archaeological deposits across northern Africa.
Materials and methods
Selected species
Nine wild grass species, representing seven genera associated with the kreb complex, were selected for this initial study (Table 1). These genera often occur together in Holocene archaeological assemblages from across northern Africa including the Tadrart Acacus sites of southwest Libya (Wasylikowa 1992; Mercuri 2008; Mercuri et al. 2018), the Dhar Tichitt sites of south central Mauritania (Munson 1976), the Early Khartoum sites of Central Sudan (Magid 1989; Stemler 1990) and Nabta Playa (Wasylikowa et al. 1997), as well as later Iron Age cities at Jenné-jeno (McIntosh and McIntosh 1979) and Essouk (Nixon et al. 2011). The specimens analysed in this study were sourced from existing reference collections (Table 1, Fig. 2). Subsamples were processed for phytoliths at the University of Queensland (UQ) and stored in their archaeobotanical reference collection. Three spikelets from a single reference accession of each species were dissected, oxidised and analysed to account for variation within the panicles. It is unclear, however, if the selected spikelets in each accession originated from one plant or several that were collected together in the field. Furthermore, with only one reference accession analysed per species, we cannot explore any variation that might be genetic, geographical or ecological. Rather, the focus of this preliminary study was on determining broad morphological variation between key species within the kreb complex.
Sample preparation and analysis
A wet ashing protocol modified from Lu et al. (2009) and Ge et al. (2020) was applied for this study. Each spikelet was separated into individual components (lower glume, upper glume, sterile lemma, sterile palea, fertile lemma and fertile palea), depending on the physiology of each species (Fig. 3). Individual spikelet components were cleaned with distilled water and placed in 10 ml centrifuge tubes. For most spikelet components, cell lignin and cytoplastic material were oxidised with 5 ml of 9.8 M (30%) hydrogen peroxide (H2O2) in a 60 °C water bath for ~ 24 h. Higher lignification and silicification in the glumes or fertile florets of some species required substitution of H2O2 with 5.6 M (30%) nitric acid (HNO3) for ~ 6 h to fully oxidise them. Trials with stronger nitric acid concentrations (> 30%) caused increased fracturing of the cuticle and abaxial phytolith layers, hindering observation of articulated morphology. Chemical oxidisation was conducted in a fume hood, with reaction completion observable through full specimen transparency and loss of buoyancy. While most wet ashing procedures use centrifugation to rinse specimens after oxidisation (Piperno 2006), this was substituted with a series of rinses with distilled water in glass petri dishes to retain articulated silica structures (Ge et al. 2020). Distilled water was used as a slide mounting medium as it enabled easy reorientation of specimens to focus on specific cell layers and areas, such as abaxial/adaxial. An Olympus BX50 light microscope was used at 400 × , equipped with a MIchrome 5 pro camera and using Mosaic v. 2.0 software.
Phytolith nomenclature
Following previous studies (Lu et al. 2009; Ge et al. 2018, 2020), phytolith classifications focused on the abaxial layer (the surface facing away from the seed), as the adaxial (towards the seed) layers of the inner epidermis are comprised of undiagnostic hypoderm fibres and Elongate entire phytoliths. Phytolith types are classified and described according to the International Code for Phytolith Nomenclature (ICPN 1.0 and 2.0) (Madella et al. 2005; Neumann et al. 2019). Short cell phytoliths (Fig. 4) were classified using previously published African grass Bilobate, Saddle, Rondel and Cross morphologies (Fahmy 2008; Mercader et al. 2010; Radomski and Neumann 2011; Novello et al. 2012; Novello and Barboni 2015; Neumann et al. 2017). Elongate classifications also followed common descriptions for projections along the lateral margins (clavate, dentate, entire and sinuate) (Madella et al. 2005; Piperno 2006; Ball et al. 2009) (Fig. 4).
Photographic examples of grass phytolith nomenclature for short and long cells used in this study. BIL, Bilobate; CRO, Cross; SAD, Saddle; RON, Rondel; ar, arched, conc, concave; conv, convex; sq, squat; st, short tabular; ta, tabular; th, thin; tz; trapeziform base. ELO, Elongate; CLA, clavate; DET, dentate; ENT entire; SIN, sinuate. ACU, Acute bulbosus. Sorghum Elongate pattern shows distinctive arrangement of Elongate clavate and Bilobate short tabular rounded morphotypes found in the sessile (unstalked) glumes
Previous studies have classified Interdigitate phytoliths from the abaxial layers as epidermal long cells (Lu et al. 2009; Zhang et al. 2011), inflorescence long cells (Kealhofer et al. 2015), husk phytoliths (Deng et al. 2018), silica skeletons (Madella et al. 2014; Weisskopf and Lee 2016; An et al. 2021) and silica layers (Ge et al. 2018). However, these terminologies are also typically used to classify articulated sheets of Elongate phytoliths (Rosen 1992; Ball et al. 2009). While the anatomical position of inflorescence phytolith layers can sometimes be preserved in archaeological assemblages, with Interdigitate phytoliths overlaying Elongate phytoliths, these morphotypes represent separate cell layers and can occur independently in archaeological assemblages (Parry and Hodson 1982; Ge et al. 2020). Therefore, following the recommendations of Ge et al. (2020), Interdigitate phytoliths were classified independently of Elongate ones. While Ge et al. (2020) proposed ‘Interdigitating’ as the formal morphotype, we use the term ‘Interdigitate’ in accordance with botanical terminology, derived from the Latin adjective digitatus for radiating digit-like projections (Stearn 1983).
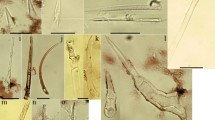
In accordance with existing studies, morphological characteristics of Interdigitate phytoliths (Fig. 5) were further classified according to differences in: 1, undulation pattern of the lateral margin projections (Λ-type, β-type or η-type), which were further classified according to the level of amplitude (higher levels relate to increased undulation length and complexity) (Fig. 6); 2, terminal margin pattern (repand/sinuolate = slightly wavy, articulate = jointed, articulate/brachiate = branching); and 3, Papillate cell (attached, detached or absent) (Ge et al. 2020). The use of these descriptors assists in avoiding confusion with those used for Elongate phytoliths. Previous studies have also referred to the terminal margins of Interdigitate morphotypes as ‘junctions’ (Zhang et al. 2018), ‘wavy fingers’ (Lu et al. 2009), or ‘connections’ (Ge et al. 2020), however, the use of ‘terminal margin’ (Fig. 6) to denote the characteristics along the shorter cell axis easily differentiates from ‘undulation’ (connecting projections along the lateral cell margin) and follows ICPN guidelines. Morphometric analysis of Interdigitate phytolith shape variables such as height/width ratio was not done in this initial study, as previous studies have shown the potential limitations of these parameters based on single reference accessions (Ball and Brotherson 1992; Kealhofer et al. 2015; Out and Madella 2017). Other phytoliths of lesser taxonomic value observed in the present study consisted of Papillate cells in the non-fertile florets and Acute bulbosus (hair cell) morphotypes (Fig. 4). Additionally, stomata were present in all species with distinct lateral nerves, and cork cells adjoined short cells in the glumes of all species; however, these are of little taxonomic value.

modified from Ge et al. 2020). Arrows indicate area of interest for each morphological characteristic
Morphological characteristics of Interdigitate phytolith layers observed in this study (terminology
Results
All spikelet components were successfully analysed for each species, with the exception of glumes from Dactyloctenium aegyptium and the uppermost sterile floret, non-flowering and flowering glumes of Eleusine indica. These lower bracts were not present in our reference specimens as the floret of these species disarticulates easily. For the purposes of this study, this was not considered an issue, as existing publications suggest that phytoliths in the fertile floret are of higher taxonomic value (Lu et al. 2009; Ge et al. 2018, 2020). As noted in “Sample preparation and analysis”, a stronger oxidising agent was required to process robust spikelet components including sessile (unstalked) glumes of S. bicolor ssp. verticilliflorum as well as the fertile florets of Echinochloa colona, Setaria sphacelata and the three Panicum spp. Increased cell lignin and silica deposition within these components is linked to enhanced seed protection (Hodson and Sangster 1988; Kumar et al. 2017). This mainly occurs on the abaxial surface, with pronounced silicification observable macroscopically with thick spikelet components and ornate epidermal patterns (Fig. 3). Within each species, morphotype occurrence was stable across the three analysed spikelets.
Variations in phytolith occurrence between inflorescence bracts
Glumes
For most of the analysed species, the glumes were thin and papery with reduced silicification compared to the fertile florets (Table 2). Short cell and Elongate phytoliths in the glumes tended to match those found in the sterile florets of all species and were of limited taxonomic value (note that a complete photographic taxonomic key for each species is presented in ESM 2). As previously reported (Radomski and Neumann 2011), Rondel morphotypes were only observed in the upper glume of Panicum turgidum. Acute bulbosus morphotypes were found in relatively greater quantity in the glumes of all species compared to the florets, with density increasing towards the top of each glume. In contrast to the other species, the sessile glumes of Sorghum bicolor ssp. verticilliflorum were strongly lignified, with Elongate clavate and Bilobate short tabular morphotypes covering the abaxial surface. This observed phytolith pattern within the sessile glumes matched previous publications (Madella et al. 2016; Ge et al. 2020) and displayed the greatest taxonomic specificity for S. bicolor ssp. verticilliflorum (Fig. 4).
Lemmas
Phytolith types in the sterile lemmas were similar to those in the glumes within all species, with slightly more developed projections on Elongate morphotypes (Table 2). The fertile lemma of Pennisetum divisum had a weakly silicified Interdigitate layer. All other Paniceae species had morphologically robust fertile lemmas with a strongly silicified Interdigitate phytolith layer on the abaxial surface (Fig. 5). While there is an Elongate layer underlying the Interdigitate phytoliths in the fertile lemma and palea of these species, this layer was not described, as the anatomical articulation of the abaxial layers prevent direct observation. Eleusine indica fertile lemmas had Saddle squat morphotypes which are commonly found in chloridoid grasses (Twiss et al. 1969). Dactyloctenium aegyptium was characterised by Elongate phytoliths with pronounced dentate projections (Fig. 4) and Saddle thin morphotypes.
Paleas
In general, paleas displayed similar phytolith morphologies to lemmas, however, sterile paleas tended to have reduced phytolith abundance in comparison to sterile lemmas (Table 2). The fertile and sterile paleas of Sorghum bicolor ssp. verticilliflorum were weakly silicified, displaying Elongate entire/sinuate and Acute bulbosus morphotypes of low taxonomic value. As observed in the fertile lemma, an Interdigitate phytolith layer was present in all Paniceae species, though in Pennisetum divisum and the sterile palea of Panicum laetum it was only weakly silicified.
Classification of Interdigitate morphotypes
In the inflorescence bracts of all species, the phytoliths were dominated by Elongate and short cell morphotypes. However, while select morphotypes were of higher taxonomic interest (for example, Elongate dentate in Dactyloctenium aegyptium and the Elongate/Bilobate arrangement in Sorghum bicolor ssp. verticilliflorum), anatomical distribution revealed significant variation within the individual bracts of each species in addition to overlap between species. While there is significant overlap in short cell and Elongate morphologies, species with an Interdigitate phytolith layer between the cuticle and Elongate layer (Echinochloa colona, Panicum anabaptistum, P. laetum, P. turgidum, Pennisetum divisum and Setaria sphacelata) have increased taxonomic potential (Table 3). Comparison of multiple morphological traits including undulation pattern, terminal margins and Papillate presence and form (Fig. 5) enabled confident taxonomic discrimination of these six species (see ESM 2 for anatomical variations in Interdigitate morphology within each species). The central cell shape of Interdigitate morphotypes was rectangular in all species analysed. As observed in previous studies, Interdigitate taxonomic specificity is greatest at the centre of the fertile lemma or palea, with undulation amplitude reduced to only basic undulation towards the margins (Figs. 6, 7; Lu et al. 2009; Zhang et al. 2011, 2018; Bhat et al. 2018; Ge et al. 2018, 2020). With the exception of Pennisetum divisum, the floral bracts of these species also have involute margins (rolled inwards) characterised by reduced undulation and significant morphological overlap between species (Fig. 7).
Echinochloa: As noted in other studies of Echinochloa spp. (Yang et al. 2015; Weisskopf and Lee 2016; Ge et al. 2018), E. colona Interdigitate phytoliths have characteristic β-type undulations with articulate terminal margins and Papillate cells attached to the central body. Level III undulations were only observed in the centre of the fertile lemma and palea. Level IV undulations were not present in the specimens analysed.
Panicum: Papillate cells were absent in the fertile florets of all three Panicum species. Interdigitate phytoliths in P. anabaptistum were characterised by η-type level I undulations with repand/sinuolate terminal margins (Fig. 8a). P. turgidum also had η-type level I-II undulations, with more articulate terminal margins than those of P. anabaptistum (Fig. 8b). Undulation patterns in P. laetum were η-type level II-III with intricate articulate/brachiate terminal margins (Fig. 8c). Weakly silicified Interdigitate patterns with basic η-type undulations and Papillate cells attached to the central cell body were observed in the sterile palea of P. laetum.
Variations in Interdigitate morphology in the middle of the fertile palea of three Panicum species. a, Panicum anabaptistum, η-type level I undulations, repand/sinuolate terminal margins; b, Panicum turgidum, η-type level II undulations, articulate terminal margins; c, Panicum laetum, η-type level II-III undulations, articulate/brachiate terminal margins. Papillate cells were absent in all three species. Scale bar (20 µm) applies to all panels
Pennisetum: Weakly silicified Interdigitate morphotypes were observed in P. divisum. These were characterised by η-type undulations with large Papillate cells detached from the central cell body. While undulation amplitude is similar to Panicum anabaptistum and other previously published species with η-type undulations, for example, Panicum miliaceum, P. ruderale and P. repens (Lu et al. 2009; Zhang et al. 2018), the undulations in Pennisetum divisum are slightly more crenate (broader) in shape towards the top of the fertile lemma and palea. These features, combined with the presence of Papillate cells detached from the central cell body, may enable taxonomic differentiation from other species.
Setaria: S. sphacelata has Λ-type undulation patterns with large Papillate cells attached to the central cell body similar to reported S. verticillata morphologies (Bhat et al. 2018). Within S. sphacelata, Λ-type level III undulations were only observed in the centre of the fertile palea (Fig. 6). Terminal margins were repand/sinuolate within the fertile lemma and articulate within the fertile palea. Pronounced Papillate cells matched the ornate epidermal patterns on the abaxial surface of the fertile lemma and were less pronounced in the fertile palea.
Discussion
Interdigitate phytoliths were present in the fertile floret of six of the nine species analysed in this study. In accordance with previous publications, presence was restricted to Paniceae grasses. As such, they were not observed in Dactyloctenium aegyptium, Eleusine indica and Sorghum bicolor ssp. verticilliflorum, which are in the Cynodonteae and Andropogoneae tribes respectively. While the analysis of a single accession of each species restricts confidence in species level classifications, within the context of this study all analysed species could be differentiated from each other by multiple morphological parameters (Tables 2, 3). However, observed Interdigitate morphologies share similarities with published parameters for closely related taxa within the same genus (Lu et al. 2009; Zhang et al. 2011, 2018; Yang et al. 2015; Weisskopf and Lee 2016; Bhat et al. 2018; Ge et al. 2018). The Interdigitate phytolith layer observed in Echinochloa colona matches previously published criteria for this species, with β-type undulations and small Papillate cells attached to the central cell body (Yang et al. 2015; Ge et al. 2018). Likewise, Interdigitate patterns in the three Panicum spp. analysed in this study were morphologically similar to published descriptions of Panicum miliaceum, P. ruderale and P. repens, with η-type undulations and no Papillate cells (Lu et al. 2009; Zhang et al. 2018). While there was overlap in undulation amplitude between the three analysed Panicum spp., the terminal margin morphology varied between species. Weakly silicified Interdigitate phytoliths with η-type undulations were also observed in Pennisetum divisum, however in contrast to Panicum spp., these had pronounced Papillate cells. Interdigitate Λ-type undulations in Setaria sphacelata were morphologically similar to S. verticillata (Bhat et al. 2018) and differed from other published Setaria spp. with Ω-type undulations (Lu et al. 2009; Zhang et al. 2011; Kealhofer et al. 2015; Ge et al. 2020). Further analyses of other accessions for each species as well as a wider range of species within each genus may enable these patterns to be refined.
Interdigitate phytoliths have been successfully recovered from archaeological grave fills (Madella et al. 2014; Out et al. 2016; Ryan 2018; Le Moyne unpublished), dental calculus (Le Moyne unpublished), middens (Yang et al. 2015; Weisskopf and Lee 2016; An et al. 2021), sediments (Zhang et al. 2017; Deng et al. 2018; Luo et al. 2019) and grindstones (Yang et al. 2015). As the occurrence of this morphotype is restricted to the fertile floret of select grass species, its presence within archaeological contexts is strongly associated with crop processing and human food consumption (Harvey and Fuller 2005). Presence also reflects the seasonality of wild grass gathering, with flowering and fruiting expected to occur at the end of the rainy season for savannah and Sahelian grasses or into the dry season for wetland plants like Oryza and some Echinochloa or Paspalum species. Most wild African grasses routinely harvested for subsistence purposes, and particularly kreb species, are freely dehiscent with disarticulation occurring above the glumes during gathering and threshing not required (Nicholaisen 1963). Thus, phytolith assemblages derived from processing by parching, pounding and winnowing of wild Paniceae species would include Interdigitate morphotypes and provide clear indications of food preparation areas (Harvey and Fuller 2005). In contrast, phytolith assemblages derived from the use of these species in animal fodder or for purposes such as thatching would contain higher frequencies of Elongate and short cell phytoliths from the leaves and culms (Harvey and Fuller 2005). In agricultural contexts, these wild grasses often occur as arable weeds with domesticated crops (Harvey and Fuller 2005). Recovery of Interdigitate morphotypes from wild grass species within domestic crop assemblages has the potential to provide high resolution taxonomic information regarding weed species. The inclusion of Interdigitate phytoliths within dental calculus matrices can provide direct evidence for consumption of grasses by an individual, as husk fragments can be expected to contaminate grains even after dehusking. These numerous pathways into the archaeological record, combined with published observations documenting the preservation of valuable taxonomic characteristics, indicate that the recovery and classification of Interdigitate phytoliths can enhance our ability to study the composition of past vegetation communities, seasonality, cultural preferences and human behaviours.
Lastly, the findings reported by Zhang et al. (2018) highlight the potential for examining domestication processes through phenotypic modification of Interdigitate phytoliths. For example, undulation amplitude appears to be correlated with grain length/width ratios (Zhang et al. 2018), which has been shown for some Paniceae species, such as P. miliaceum, to have undergone a marked shift during domestication (Stevens et al. 2021). However, the high panicoid and chloridoid grass diversity in Africa makes this more of a challenge to apply in this context, as does the high diversity of documented grass use. Jardin (1967) catalogued the consumption of over 60 species of small-seeded wild grasses across Africa during the mid 20th century, though many were no longer routinely consumed. Future analyses of inflorescence phytoliths from kreb and other African wild grasses should therefore work towards building comprehensive taxonomic keys for the full suite of economic taxa. Incorporating Aristida, Brachiaria, Cenchrus, Digitaria, Eragrostis, Latipes, Oryza and Paspalum species would assist in determining accurate morphological markers for each genus and potentially refine differentiation between related wild and domesticated species from phytolith assemblages. Additionally, as demonstrated by Ge et al. (2018), the collection and analysis of multiple accessions of each species, grown in different conditions, is necessary to account for phenotypic plasticity. Future studies combining morphological and morphometric analysis of multiple accessions and incorporating a greater number of economic species would potentially enable confident differentiation between species within an African context.
Conclusions
The results presented within this preliminary study demonstrate the taxonomic potential of Interdigitate inflorescence phytoliths in African Paniceae grasses and provide additional means of identifying the presence of these taxa from archaeological contexts. The analysis of phytolith occurrence within individual spikelet components indicates that highly diagnostic morphotypes are restricted to heavily silicified and lignified components. Combined morphological parameters enable classification to genus level for the nine African wild grass species included in this study. Morphological variations in the undulation amplitude and terminal margins of Interdigitate phytoliths as well as the presence or absence of Papillate cells suggest the potential for reliable differentiation between African Paniceae species and particularly Panicum spp., however, morphometric analysis of additional taxa and reference accessions is required to further test this finding. The morphological parameters described in this preliminary study provide an additional means of taxonomic classification for archaeological material, potentially providing another tool by which we can understand different trajectories of wild grass use. Current perceptions of the past economic importance of wild grasses across northern Africa are constrained by analytical limitations. However, it is evident that these plants formed a vital component of past subsistence practices that persisted after the uptake of dedicated agricultural practices until the very recent past.
References
An J, Kirleis W, Zhao C, Jin G (2021) Understanding crop processing and its social meaning in the Xinzhai period (1850–1750 cal BCE): A case study on the Xinzhai site. Veget Hist Archaeobot, China. https://doi.org/10.1007/s00334-021-00851-0
Ball TB, Brotherson JD (1992) The effect of varying environmental conditions on phytolith morphology in two species of grass (Bouteloua curtipendula and Panicum virgatum). Scanning Micro 6:1163–1182
Ball TB, Ehlers R, Standing MD (2009) Review of typologic and morphometric analysis of phytoliths produced by wheat and barley. Breed Sci 59:505–512. https://doi.org/10.1270/jsbbs.59.505
Barakat H, Fahmy AG (1999) Wild grasses as ‘Neolithic’ food resources in the eastern Sahara: a review of the evidence from Egypt. In: van der Veen M (ed) The exploitation of plant resources in ancient Africa. Springer, New York, pp 33–46
Barboni D, Bremond L (2009) Phytoliths of East African grasses: An assessment of their environmental and taxonomic significance based on floristic data. Rev Palaeobot Palynol 158:29–41. https://doi.org/10.1016/j.revpalbo.2009.07.002
Barboni D, Bonnefille R, Alexandre A, Meunier JD (1999) Phytoliths as paleoenvironmental indicators, west side middle awash valley, Ethiopia. Palaeogeogr Palaeoclimatol Palaeoecol 152:87–100. https://doi.org/10.1016/S0031-0182(99)00045-0
Barron A, Fuller DQ, Stevens C, Champion L, Winchell F, Denham T (2020) Snapshots in time: MicroCT scanning of pottery sherds determines early domestication of sorghum (Sorghum bicolor) in East Africa. J Archaeol Sci 123:105259. https://doi.org/10.1016/j.jas.2020.105259
Barth H (1857) Travels and discoveries in North and Central Africa. Harper and Bros, New York
Batello C, Marzot M, Harouna Toure A (2004) The future is an ancient lake: Traditional knowledge, biodiversity and genetic resources for food and agriculture in lake Chad basin ecosystems. Food & Agriculture Organization of the United Nations (FAO), Rome
Beldados A (2019) Millets in eastern Sudan: An archaeobotanical study. Azania 54:501–515. https://doi.org/10.1080/0067270X.2019.1691844
Bhat MA, Shakoor SA, Badgal P, Soodan AS (2018) Taxonomic demarcation of Setaria pumila (Poir.) Roem. & Schult., S. verticillata (l.) P. Beauv., and S. viridis (l.) P. Beauv. (Cenchrinae, Paniceae, Panicoideae, Poaceae) from phytolith signatures. Front Plant Sci 9:864. https://doi.org/10.3389/fpls.2018.00864
Bourel B, Novello A (2020) Bilobate phytolith size matters for taxonomical and ecological identification of Chad grasses: A case study on 15 species. Rev Palaeobot Palynol 275:104114. https://doi.org/10.1016/j.revpalbo.2019.104114
Brass M, Fuller DQ, MacDonald K et al (2019) New findings on the significance of Jebel Moya in the eastern Sahel. Azania 54:425–444. https://doi.org/10.1080/0067270x.2019.1691845
Buckley S, Usai D, Jakob T, Radini A, Hardy K (2014) Dental calculus reveals unique insights into food items, cooking and plant processing in prehistoric central Sudan. PLoS ONE 9:e100808. https://doi.org/10.1371/journal.pone.0100808
Champion L, Fuller DQ, Ozainne S, Huysecom É, Mayor A (2021) Agricultural diversification in West Africa: an archaeobotanical study of the site of Sadia (Dogon Country, Mali). Archaeol Anthropol Sci 13:60. https://doi.org/10.1007/s12520-021-01293-5
Chevalier A (1932) Ressources végétales du Sahara et de ses confins nord et sud. Musée d’Histoire Naturelle, Paris
Clark JD (1976) Prehistoric populations and pressures favoring plant domestication in Africa. In: Harlan JR (ed) Origins of African plant domestication, 1st edn. De Gruyter, Berlin, pp 67–105. https://doi.org/10.1515/9783110806373.67
Collura LV, Neumann K (2017) Wood and bark phytoliths of West African woody plants. Quat Int 434:142–159. https://doi.org/10.1016/j.quaint.2015.12.070
Costantini L, Fattovich R, Pardini E, Piperno M (1982) Preliminary report of archaeological investigation at the site of Teglinos (Kassala). Nyame Akuma 21:30–33
D’Andrea AC (2008) T’ef (Eragrostis tef) in ancient agricultural systems of highland Ethiopia. Econ Bot 62:547–566. https://doi.org/10.1007/s12231-008-9053-4
Deng Z, Hung H, Fan X, Huang Y, Lu H (2018) The ancient dispersal of millets in southern China: New archaeological evidence. Holocene 28:34–43. https://doi.org/10.1177/0959683617714603
Duveyrier H (1864) Les Touareg du Nord. Challamel Ainé, Paris. Reprint Kraus, Liechtenstein
Eichhorn B, Neumann K, Garnier A (2010) Seed phytoliths in west African Commelinaceae and their potential for palaeoecological studies. Palaeogeogr Palaeoclimatol Palaeoecol 298:300–310. https://doi.org/10.1016/j.palaeo.2010.10.004
Esteban I, De Vynck JC, Singels E et al (2017) Modern soil phytolith assemblages used as proxies for paleoscape reconstruction on the south coast of south Africa. Quat Int 434:160–179. https://doi.org/10.1016/j.quaint.2016.01.037
Fahmy AG (2001) Palaeoethnobotanical studies of the Neolithic settlement in Hidden Valley, Farafra Oasis. Egypt Veget Hist Archaeobot 10:235–246. https://doi.org/10.1007/PL00006935
Fahmy AG (2008) Diversity of lobate phytoliths in grass leaves from the Sahel region, west tropical Africa: Tribe Paniceae. Plant Syst Evol 270:1–23. https://doi.org/10.1007/s00606-007-0597-z
Fahmy AG, Magnavita C (2006) Phytoliths in a silo: Micro-botanical evidence from Zilum (Lake Chad basin), NE Nigeria (c. 500 cal BC). J Biol Sci 6:824–832. https://doi.org/10.3923/jbs.2006.824.832
Florenzano A, Mercuri AM, Altunoz M, Garcea EAA (2016) Palynological evidence of cultural and environmental connections in Sudanese Nubia during the early and middle Holocene. Quat Int 412:65–80. https://doi.org/10.1016/j.quaint.2016.01.001
Fuller DQ, MacDonald K, Vernet R (2007) Early domesticated pearl millet in Dhar Nema (Mauritania): Evidence of crop processing waste as ceramic temper. In: Cappers R (ed) Fields of change: Progress in African archaeobotany. Barkhuis & Groningen University Library, Groningen, pp 71–76
Fuller DQ, Barron A, Champion L, Dupuy C, Commelin D, Raimbault M, Denham T (2021) Transition from wild to domesticated Pearl Millet (Pennisetum glaucum) revealed in ceramic temper at three Middle Holocene sites in northern Mali. Afr Archaeol Rev 38:211–230. https://doi.org/10.1007/s10437-021-09428-8
Fuller DQ, Champion L, Stevens C (2019) Comparing the tempo of cereal dispersal and the agricultural transition: two African and one West Asian trajectory. In: Eichhorn B, Höhn A (eds) Trees, grasses and crops: people and plants in sub-Saharan Africa and beyond. (Frankfurter Archäologische Schriften 37) Habelt, Bonn, pp 119–140
Ge Y, Lu H, Zhang J, Wang C, He K, Huan X (2018) Phytolith analysis for the identification of barnyard millet (Echinochloa sp.) and its implications. Archaeol Anthropol Sci 10:61–73. https://doi.org/10.1007/s12520-016-0341-0
Ge Y, Lu H, Zhang J, Wang C, Gao X (2020) Phytoliths in inflorescence bracts: Preliminary results of an investigation on common Panicoideae plants in China. Front Plant Sci 10:1736. https://doi.org/10.3389/fpls.2019.01736
Giblin JD, Fuller DQ (2011) First and second millennium A.D. agriculture in Rwanda: archaeobotanical finds and radiocarbon dates from seven sites. Veget Hist Archaeobot 20:253–265. https://doi.org/10.1007/s00334-011-0288-0
Harlan JR (1975) Crops and man. American Society for Agronomy, Madison
Harlan JR (1989a) Wild-grass seed harvesting in the Sahara and sub-Sahara of Africa. In: Harris DR, Hillman GC (eds) Foraging and farming: the evolution of plant exploitation. Routledge, London, pp 79–98
Harlan JR (1989b) The tropical African cereals. In: Harris DR, Hillman GC (eds) Foraging and farming: The evolution of plant exploitation. Routledge, London, pp 335–343
Harris DR (1984) Ethnohistorical evidence for the exploitation of wild grasses and forbs: Its scope and archaeological implications. In: van Zeist W, Casparie WA (eds) Plants and ancient man: Studies in palaeoethnobotany. Balkema, Rotterdam, pp 63–69
Harvey EL, Fuller DQ (2005) Investigating crop processing using phytolith analysis: The example of rice and millets. J Archaeol Sci 32:739–752. https://doi.org/10.1016/j.jas.2004.12.010
Heuglin M (1869) Reise in das Gebiet des Weissen Nil und seiner westlichen Zuflüsse in den Jahren 1862–1864. Winter’sche Verlagshandlung, Leipzig
Hodson MJ, Sangster AG (1988) Silica deposition in the inflorescence bracts of wheat (Triticum aestivum). I. Scanning electron microscopy and light microscopy. Can J Bot 66:829–838. https://doi.org/10.1139/b88-121
Jardin C (1967) Lists of foods used in Africa. FAO, Rome
Kealhofer L, Huang F, DeVincenzi M, Kim MM (2015) Phytoliths in Chinese foxtail millet (Setaria italica). Rev Palaeobot Palynol 223:116–127. https://doi.org/10.1016/j.revpalbo.2015.09.004
Klee M, Zach B, Neumann K (2000) Four thousand years of plant exploitation in the Chad basin of northeast Nigeria I: The archaeobotany of Kursakata. Veget Hist Archaeobot 9:223–237. https://doi.org/10.1007/BF01294637
Kumar S, Soukup M, Elbaum R (2017) Silicification in grasses: Variation between different cell types. Front Plant Sci 8:438. https://doi.org/10.3389/fpls.2017.00438
Lu H, Zhang J, Wu N, Liu K-b, Xu D, Li Q (2009) Phytoliths analysis for the discrimination of foxtail millet (Setaria italica) and common millet (Panicum miliaceum). PLoS ONE 4:e4448. https://doi.org/10.1371/journal.pone.0004448
Luo W, Gu C, Yang Y et al (2019) Phytoliths reveal the earliest interplay of rice and broomcorn millet at the site of Shuangdun (ca. 7.3–6.8 ka BP) in the middle Huai River valley. China J Archaeol Sci 102:26–34. https://doi.org/10.1016/j.jas.2018.12.004
Madella M, Alexandre A, Ball T (2005) International code for phytolith nomenclature 1.0. Ann Bot 96:253–260. https://doi.org/10.1093/aob/mci172
Madella M, García-Granero JJ, Out WA, Ryan P, Usai D (2014) Microbotanical evidence of domestic cereals in Africa 7000 years ago. PLoS ONE 9:e110177. https://doi.org/10.1371/journal.pone.0110177
Madella M, Lancelotti C, García-Granero JJ (2016) Millet microremains—an alternative approach to understand cultivation and use of critical crops in prehistory. Archaeol Anthropol Sci 8:17–28. https://doi.org/10.1007/s12520-013-0130-y
Magid AA (1984) Archaeobotanical remains from Shaqadud: Interim note. Nyame Akuma 24:25–27
Magid AA (1989) Exploitation of plants in the eastern Sahel (Sudan), 5,000–2,000 B.C. In: Krzyzaniak L, Kobusuewicz M (eds) Late prehistory of the Nile basin and the Sahara. Poznań Archaeological Museum, Poznań, pp 459–468
Manning K, Pelling R, Higham T, Schwenniger J-L, Fuller DQ (2011) 4500-year old domesticated pearl millet (Pennisetum glaucum) from the Tilemsi valley, Mali: new insights into an alternative cereal domestication pathway. J Archaeol Sci 38:312–322. https://doi.org/10.1016/j.jas.2010.09.007
Märkle T, Rösch M (2008) Experiments on the effects of carbonization on some cultivated plant seeds. Veget Hist Archaeobot 17:257–263. https://doi.org/10.1007/s00334-008-0165-7
McDonald MMA (2016) The pattern of neolithization in Dakhleh Oasis in the eastern Sahara. Quat Int 410:181–197. https://doi.org/10.1016/j.quaint.2015.10.100
McIntosh SK, McIntosh RJ (1979) Initial perspectives on prehistoric subsistence in the inland Niger Delta (Mali). World Archaeol 11:227–243. https://doi.org/10.1080/00438243.1979.9979762
Mercader J, Runge F, Vrydaghs L, Doutrelepont H, Ewango CEN, Juan-Tresseras J (2000) Phytoliths from archaeological sites in the tropical forest of Ituri, Democratic Republic of Congo. Quat Res 54:102–112. https://doi.org/10.1006/qres.2000.2150
Mercader J, Astudillo F, Barkworth M et al (2010) Poaceae phytoliths from the Niassa rift, Mozambique. J Archaeol Sci 37:1953–1967. https://doi.org/10.1016/j.jas.2010.03.001
Mercader J, Bennett T, Esselmont C, Simpson S, Walde D (2011) Soil phytoliths from miombo woodlands in Mozambique. Quat Res 75:138–150. https://doi.org/10.1016/j.yqres.2010.09.008
Mercuri AM (1999) Palynological analysis of the early Holocene sequence. In: di Lernia S (ed) The Uan Afuda cave: Hunter-gatherer societies of central Sahara. Edizioni all’Insegna Del Giglio, Rome, pp 149–181
Mercuri AM (2008) Plant exploitation and ethnopalynological evidence from the Wadi Teshuinat area (Tadrart Acacus, Libyan Sahara). J Archaeol Sci 35:1619–1642. https://doi.org/10.1016/j.jas.2007.11.003
Mercuri AM, Garcea EAA (2007) The impact of hunter/gatherers on the vegetation in the central Sahara during the early Holocene. In: Cappers R (ed) Fields of change: Progress in African archaeobotany. Barkhuis & Groningen University Library, Groningen, pp 87–103
Mercuri AM, Fornaciari R, Gallinaro M, Vanin S, di Lernia S (2018) Plant behaviour from human imprints and the cultivation of wild cereals in Holocene Sahara. Nat Plants 4:71–81. https://doi.org/10.1038/s41477-017-0098-1
Munson PJ (1976) Archaeological data on the origins of cultivation in the Southwestern Sahara and their implications for West Africa. In: Harlan JR (ed) Origins of African plant domestication, 1st edn. De Gruyter, Berlin, pp 187–210. https://doi.org/10.1515/9783110806373.187
Murray MA, Fuller DQ, Cappeza C (2007) Crop production on the Senegal River in the early first millennium AD: preliminary archaeobotanical results from Cubabel. In: Cappers R (ed) Fields of change: Progress in African archaeobotany. Barkhuis & Groningen University Library, Groningen, pp 63–69
Murungi ML, McGlynn G, Lejju JB (2017) Alpine grassland palaeoecology of the Virunga Volcanoes, East Africa: a new phytolith record from Mt. Muhavura Quat Int 434:102–116. https://doi.org/10.1016/j.quaint.2016.01.015
Murungi ML (2017) Phytoliths at Sibudu (South Africa): Implications for vegetation, climate and human occupation during the MSA. Dissertation, University of the Witwatersrand, Johannesburg
Nachtigal G (1881) Sahărâ und Sûdân: Borkû, Kânem, Bornû und Bagirmi, vol I. Weidmann, Berlin
Nesbitt M (1997) Archaeobotanical identification of Near Eastern grass caryopses. Dissertation, University College London, London
Neumann K, Fahmy A, Lespez L, Ballouche A, Huysecom E (2009) The early Holocene palaeoenvironment of Ounjougou (Mali): Phytoliths in a multiproxy context. Palaeogeogr Palaeoclimatol Palaeoecol 276:87–106. https://doi.org/10.1016/j.palaeo.2009.03.001
Neumann K, Fahmy AG, Müller-Scheeßel N, Schmidt M (2017) Taxonomic, ecological and palaeoecological significance of leaf phytoliths in West African grasses. Quat Int 434:15–32. https://doi.org/10.1016/j.quaint.2015.11.039
Neumann K, Strömberg CAE, Ball T, Albert RM, Vrydaghs L, Cummings LS (2019) International code for phytolith nomenclature (ICPN) 2.0. Ann Bot 124:189–199. https://doi.org/10.1093/aob/mcz064
Nicolaisen G (1963) Ecology and culture of the pastoral Tuareg. National Museum, Copenhagen
Nixon S, Murray MA, Fuller DQ (2011) Plant use at an early Islamic merchant town in the West African Sahel: The archaeobotany of Essouk-Tadmakka (Mali). Veget Hist Archaeobot 20:223–239. https://doi.org/10.1007/s00334-010-0279-6
Novello A, Barboni D (2015) Grass inflorescence phytoliths of useful species and wild cereals from sub-Saharan Africa. J Archaeol Sci 59:10–22. https://doi.org/10.1016/j.jas.2015.03.031
Novello A, Barboni D, Berti-Equille L, Mazur J-C, Poilecot P, Vignaud P (2012) Phytolith signal of aquatic plants and soils in Chad, Central Africa. Rev Palaeobot Palynol 178:43–58. https://doi.org/10.1016/j.revpalbo.2012.03.010
Out WA, Madella M (2016) Morphometric distinction between bilobate phytoliths from Panicum miliaceum and Setaria italica leaves. Archaeol Anthropol Sci 8:505–521. https://doi.org/10.1007/s12520-015-0235-6
Out WA, Madella M (2017) Towards improved detection and identification of crop by-products: Morphometric analysis of bilobate leaf phytoliths of Pennisetum glaucum and Sorghum bicolor. Quat Int 434:1–14. https://doi.org/10.1016/j.quaint.2015.07.017
Out WA, Ryan P, García-Granero JJ, Barastegui J, Maritan L, Madella M, Usai D (2016) Plant exploitation in Neolithic Sudan: A review in the light of new data from the cemeteries R12 and Ghaba. Quat Int 412:36–53. https://doi.org/10.1016/j.quaint.2015.12.066
Parry DW, Hodson MJ (1982) Silica distribution in the caryopsis and inflorescence bracts of foxtail millet [Setaria italica (l.) Beauv.] and its possible significance in carcinogenesis. Ann Bot 49:531–540
Pelling RE (2007) Agriculture and trade amongst the Garamantes and the Fezzanese: 3000 years of archaeobotanical data from the Sahara and its margins. Dissertation, University College London, London
Piperno DR (2006) Phytoliths: A comparative guide for archaeologists and palaeoecologists. AltaMira Press, Lanham
Portillo M, Ball TB, Wallace M et al (2020) Advances in morphometrics in archaeobotany. Environ Archaeol 25:246–256. https://doi.org/10.1080/14614103.2019.1569351
Radomski KU, Neumann K (2011) Grasses and grinding stones: Inflorescence phytoliths from modern West African Poaceae and archaeological stone artefacts. In: Fahmy AG, Kahlheber S, D’Andrea AC (eds) Windows on the African past: Current approaches to African archaeobotany. Africa Magna Verlag, Frankfurt, pp 153–166
Rosen A (1992) Preliminary identification of silica skeletons from Near Eastern archaeological sites An anatomical approach. In Rapp G Jr, Mulholland SC (eds) Advances in archaeological and museum science Phytolith systematics: Emerging issues, vol 1. Springer, Boston, pp 129–147. https://doi.org/10.1007/978-1-4899-1155-1_7
P Ryan 2018 Plants as grave goods: Microbotanical remains (phytoliths) from the white deposits in the graves A Kerma ancien cemetery in the northern Dongola reach, excavations at site H29. (Sudan Archaeological Research Society 22, Eds Welsby DA Archaeopress Oxford 203 206
Sadras VO (2007) Evolutionary aspects of the trade-off between seed size and number in crops. Field Crops Red 100:125–138. https://doi.org/10.1016/j.fcr.2006.07.004
Stearn WT (1983) Botanical Latin: History, grammar, syntax, terminology and vocabulary. David & Charles Publishers, Newton Abbot
Stemler A (1990) Scanning electron microscopic analysis of plant impressions in pottery from the sites of Kaderoe, El Zakiab, Um Direiwa and El-Kadada. Archeol Nil Moyen 4:87–105
Stevens CJ, Shelach-Lavi G, Zhang H, Teng M, Fuller DQ (2021) A model for the domestication of Panicum miliaceum (common, proso or broomcorn millet) in China. Veget Hist Archaeobot 30:21–33. https://doi.org/10.1007/s00334-020-00804-z
Thanheiser U (2011) Island of the blessed: 8000 years of plant exploitation in the Dakhleh Oasis, Egypt. In: Fahmy AG, Kahlheber S, D’Andrea AC (eds) Windows on the African past: Current approaches to African archaeobotany. Africa Magna Verlag, Frankfurt, pp 79–90
Tubiana M-J, Tubiana J (1977) The Zaghawa from an ecological perspective: foodgathering, the pastoral system, tradition, and the development of the Zaghawa of the Sudan and Chad. Balkema, Rotterdam
Twiss PC, Suess E, Smith RM (1969) Morphological classification of grass phytoliths. Soil Sci Soc Am J 33:109–115. https://doi.org/10.2136/sssaj1969.03615995003300010030x
Wadley L, Sievers C, Bamford M, Goldberg P, Berna F, Miller C (2011) Middle Stone Age bedding construction and settlement patterns at Sibudu. South Africa. Science 334(6061):1388–1391. https://doi.org/10.1126/science.1213317
Wadley L, Esteban I, De La Peña P et al (2020) Fire and grass-bedding construction 200 thousand years ago at Border Cave. South Africa Science 369(6505):863–866. https://doi.org/10.1126/science.abc7239
Wasylikowa K (1992) Holocene flora of the Tadrart Acacus area, SW Libya, based on plants macrofossils from Uan Muhuggiag and Ti-n-Torha/Two Caves archaeological sites. Origini 16:125–159
Wasylikowa K, Mitka J, Wendorf F, Schild R (1997) Exploitation of wild plants by the early Neolithic hunter-gatherers of the western desert, Egypt: Nabta playa as a case-study. Antiquity 71:932–941. https://doi.org/10.1017/S0003598X00085835
Weber SA, Fuller DQ (2008) Millets and their role in early agriculture. Pragdhara 18:69–90
Weisskopf AR, Lee G-A (2016) Phytolith identification criteria for foxtail and broomcorn millets: a new approach to calculating crop ratios. Archaeol Anthropol Sci 8:29–42. https://doi.org/10.1007/s12520-014-0190-7
Wiersema JH, Dahlberg J (2007) The nomenclature of Sorghum bicolor (L.) Moench (Gramineae). Taxon 56:941–946. https://doi.org/10.2307/25065876
Wilson DG (1984) The carbonization of weed seeds and their representation in macrofossil assemblages. In: van Zeist W, Casparie WA (eds) Plants and ancient man: Studies in palaoethnobotany. Balkema, Rotterdam, pp 201–206
Winchell F, Stevens CJ, Murphy C, Champion L, Fuller DQ (2017) Evidence for sorghum domestication in fourth millennium BC eastern Sudan: Spikelet morphology from ceramic impressions of the Butana group. Curr Anthropol 58:673–683. https://doi.org/10.1086/693898
Yang X, Fuller DQ, Huan X et al (2015) Barnyard grasses were processed with rice around 10000 years ago. Sci Rep 5:16251. https://doi.org/10.1038/srep16251
Zhang J, Lu H, Wu N, Yang X, Diao X (2011) Phytolith analysis for differentiating between foxtail millet (Setaria italica) and green foxtail (Setaria viridis). PLoS ONE 6:e19726. https://doi.org/10.1371/journal.pone.0019726
Zhang J, Lu H, Jia PW, Flad R, Wu N, Betts A (2017) Cultivation strategies at the ancient Luanzagangzi settlement on the easternmost Eurasian steppe during the Late Bronze Age. Veget Hist Archaeobot 26:505–512. https://doi.org/10.1007/s00334-017-0608-0
Zhang J, Lu H, Liu M, Diao X, Shao K, Wu N (2018) Phytolith analysis for differentiating between broomcorn millet (Panicum miliaceum) and its weed/feral type (Panicum ruderale). Sci Rep 8:13022. https://doi.org/10.1038/s41598-018-31467-6
Acknowledgements
We thank Andrew Fairbairn, Ben Schoville, Pauline Fowler and Jeremy Farr for useful discussions and feedback on the text, and Emilija Nicolosi and Chat Marasinghe for technical support in the laboratory. We are also grateful to the anonymous reviewers for their comments on an earlier draft.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This research was supported by funding from an Australian Government Research Training Program Scholarship, and The University of Queensland School of Social Science. The authors would also like to thank The Australian Institute of Nuclear Science and Engineering (AINSE) Limited for providing financial assistance (Award—PGRA) to enable this research.
Author information
Authors and Affiliations
Contributions
CLM Conceptualization, methodology, formal analysis & investigation, writing—original draft, writing—review & editing. DQF supervision, writing—review & editing. AC conceptualization, supervision, writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by K. Neumann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Le Moyne, C., Fuller, D.Q. & Crowther, A. Microbotanical signatures of kreb: differentiating inflorescence phytoliths from northern African wild grasses. Veget Hist Archaeobot 32, 49–63 (2023). https://doi.org/10.1007/s00334-022-00880-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-022-00880-3