Abstract
Traumatic brain injury remains prevalent in children, particularly within the adolescent age group. In severe injury, the priority of treatment is to stabilise the patient initially and prevent the evolution of brain swelling and secondary ischaemia using tiers of medical therapy. The final stage of intervention for such patients is a decompressive craniectomy. Here in, we identify the current evidence for performing decompressive crainectomy in children including the results from the RESCUEicp study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The greatest clinical challenge after a traumatic brain injury is to minimise secondary injury to the brain [1]. The complex, dynamic changes that occur in the brain’s physiology and chemistry can lead to progressive swelling in the brain. This results in reduced blood flow, limited oxygenation and ultimately poor outcome [2,3,4,5]. Advances in multi-modality monitoring parameters and intensive care management have allowed for an improvement in understanding of the optimal physiological targets in adults after a traumatic brain injury [6]. This has helped to guide both medical and surgical intervention in these patients with improving outcomes. Nevertheless, these benefits have been slower to translate into paediatric care after a traumatic brain injury (TBI). As such, the optimal strategies of both medical and surgical intervention remain widely debated [7].
Intracranial hypertension can occur in up to 65% of patients after a severe paediatric TBI, moreover, raised intracranial pressure (ICP) accounts for over half of all TBI-related deaths [8, 9]. The total time that ICP is elevated to greater than 20 mmHg correlates strongly with outcome [10]. Therapeutic interventions for reducing ICP include reducing the intracranial contents by removing CSF, reducing blood volume, or brain volume, by reducing cerebral metabolic demands or by increasing cranial volume by decompressive craniectomy (DC).
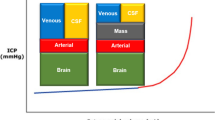
Cranial decompression is regarded as the final stage of intracranial hypertension management with its efficacy debated widely in the context of both paediatric and adult TBI [11,11,12,13,14,15,16,17,18,19,20,22]. The procedure can greatly improve brain compliance and improve compensatory reserve (Fig. 1) [23, 24]. As a consequence, improved cerebral blood flow and cerebral perfusion pressure (CPP) will augment brain tissue oxygenation (PbtO2).
This figure shows an example monitoring trace of a patient with intracranial hypertension as a result of a traumatic brain injury. The trace demonstrates a sustained plateau of intracranial pressure (blue) that lasts for around 20 min. This is associated with a reduced cerebral perfusion pressure (red), and as a result, the brain’s cerebral autoregulation is non-compliant (green). These are key features that can occur with severe traumatic brain injury and if recurrent would demonstrate potential benefit in undertaking a decompressive craniectomy
The classical surgical options include unilateral hemicraniectomies [13, 14, 18, 25] and bifrontal hemicraniectomies [16, 22, 26]. However, less commonly bilateral hemicraniectomies [14, 18], circumferential craniectomies [27] and bilateral temporal craniotomies [15, 19] have been implemented all of which involve the excision of large sections of the skull +/− duraplasty.
Indications
The indications for a DC in a child are not uniformly agreed on. Younger patients generally have a better outcome, nevertheless, given the implications of the procedure caution should be encouraged with patient selection [12, 21, 28]. Features such as brainstem injury and central herniation would generally exclude a patient from such a procedure because of the pre-disposition to poor outcome [29, 30]. The post-resuscitation GCS is perhaps the most accurate assessment [31, 32]. The presence of radiological features is often helpful in determining the necessity to decompress. Midline shift of the brain on computed tomography (CT) is highly prognostic in children with the degree of shift being inversely related to outcome [33]. Interestingly, in adults, where an absence or compression of the basal cisterns is predictive of poor outcome [34], in the paediatric population, patent basal cisterns do not exclude high ICP [35] and extra caution should be observed (Fig. 2). Recent evidence suggests that optic nerve sheath diameter (ONSD) is particularly accurate in predicting current ICP in paediatric patients [36]. Regardless, the most accurate assessment one can make is that of invasive intracranial pressure and cerebral perfusion pressure [10, 37, 38].
Representative image of paediatric patients with raised intracranial pressure. a Fourteen-year-old patient with acute subdural haematoma (ASDH), opening ICP 32 mmHg. b Seven-year-old patient with diffuse axonal injury (DAI), opening ICP 38 mmHg. c Twelve-year-old patient with ASDH and DAI opening pressure 35 mmHg. All patients demonstrate open basal cisterns despite pathologically raised ICP
Early decompression was thought to be related to a better outcome. That said, premature intervention may be unnecessary and must be balanced against the knowledge that a delayed intervention can result in irreversible injury and increased risk of neurological damage. However, the DECRA trial, which was published in 2011, recruited 155 adult patients with severe diffuse TBI and moderate intracranial hypertension. Patients were randomised within the first 72 h following TBI if their ICP exceeded 20 mmHg for >15 min—continuously or intermittently within a 1-h period, and if they did not respond to optimised first-tier ICP-lowering interventions. The two arms of the trial were bifrontal DC and standard medical management or standard medical management alone. Patients in the surgical arm had a higher rate of unfavourable outcomes (70 versus 51%; OR 2.21; 95% CI 1.14–4.26; p = 0.02) [12]. DC when used appropriately can be lifesaving but may come at the expense of severe neurological impairment.
A stepwise approach to care should be implemented without delay. Only when maximal medical therapy has been exhausted should DC be considered. The initial management of patients should encompass elevation of the head to 30 degrees, sedatives both with and without paralytics, adjustment of ventilatory settings to maintain PaCO2 at 30–35 mmHg, cooling to at least normothermia with a number of centres arguing that hypothermia is also beneficial [39], maintenance of hyperosmolar euvolaemia, correcting hyperglycaemia, supporting CPP with vasopressors and CSF diversion.
Specifically in children, evidence to support thresholds for ICP and CPP is limited. The general consensus is that an ICP greater than 20 mmHg and a CPP below 50 mmHg are consistent with poor outcome [10, 37, 38]. ICP in the low twenties is generally accepted so long that CPP and PbtO2 are well maintained. Further work is required to provide age-specific or even weight-specific pressure thresholds.
Current practice in paediatrics
Generally, the evidence describing the benefits of DC in paediatric TBI patients is limited. Eight class III studies were reviewed for the publication of paediatric TBI management guidelines [40]. The studies vary in terms of the inclusion criteria and also in the intervention performed.
Decompressive craniectomy for control of ICP
A misconception with the implementation of DC is that it is used to reduce the ICP from pre-operative values. Whilst this often results, this is not the primary aim. Rather, the aim is to lower medically intractable ICP such that minimal therapeutic intervention is required to optimise post-operative pressures [40]. Similar to adults, the emphasis of paediatric decompression is with early intervention. [41] describes a complete survival rate of seven patients who presented “massive” bilateral or unilateral swelling, compressed supratentorial swelling and perimesencephalic cisterns. Patients were treated with fronto-temporal DC all within 70 min from ictus. The initial ICP exceeded 45 mmHg in all patients, with six of seven achieving ICP of <20 mmHg post-operatively. The final patient was controlled well with medical therapy.
In contrast, [42] favoured the presence of prolonged ICP plateaus to determine which patients would benefit from surgery. Children who had a sustained ICP of over 20 mmHg for over 30 min were treated with a unilateral DC with duraplasty. In five of six patients, ICP was appropriately controlled in the sixth patient uncontrolled ICP promoted a return to surgery for a contralateral procedure.
[43] described a case series of 23 patients with a TBI under 20 years who underwent DC for initial mass lesion requiring evacuation or elevated ICP that was refractory to medical management. Bifrontal/biparietal craniectomies with duraplasty and sectioning of the falx were the most common performed within the series. Unilateral DC was utilised in the presence of a mass lesion or unilateral swelling. Ten patients underwent an early DC with 11 having a later DC. The mean ICP was significantly reduced from 30 to 18 mmHg post-operatively. All but two patients had medically managed ICP after decompression. [44] observed similar findings in patients who had either mass lesions removed or primary brain swelling decompressed, selected for having an ICP >25 mmHg or evidence of herniation on CT. Of those that underwent decompression, only one required management of sustained ICP elevations post-operatively. [45] described using bifrontal/biparietal craniotomies with duraplasty to control ICP in four of the five patients in his study.
Finally, [46] described similar findings in a case series of 23 patients under 2 years of age who underwent surgery as a result of non-accidental injury. Decision to decompress was based purely on the ICP. Of the nine patients who were decompressed, DC lowered the mean ICP from 54.9 to 11.9 mmHg. Surgery in all patients was performed within 24 h of injury.
Decompressive craniotomy for improving outcome
The impact of decompression on the outcome of children with intracranial hypertension is unclear. In one prospective study, [19] reported a favourable trend of the effect of decompression on ICP and outcome in a small pilot clinical trial of decompressive craniectomy in 27 children (13 of whom were treated with decompression). In addition, several case series have been published that collectively describe very early application of either unilateral or bilateral decompression in 11 paediatric cases [7, 42]. In some of these cases, decompression was the therapeutic choice prior to maximal medical management. Specifically, [7] describes Glasgow Outcome Scores of 4–5 at 40-month follow-up with [42] citing similar results at 6 months in seven of 11 patients.
A retrospective study on 23 patients who underwent decompression for diffuse axonal injury or contusions reported survival in 16 patients, 13 with favourable outcome [43]. Mannitol, hypertonic saline, neuromuscular blockade and controlled ventilation (targeting a PaCO2 of ~35 mmHg) were used as the medical management strategies, and surgery was undertaken if ICP remained >20 mmHg on this regimen.
Decompression has been reported to have a negative impact in two studies [44, 47]. Managed using the Lund approach five of eight patients displayed significant neurological deterioration at the time of surgery. Nevertheless, all but one survived with three patients having a full neurological recovery.
In one of the larger studies, 51 patients underwent decompressive craniectomy, mainly for evacuation of a mass lesion [44]. Five cases were performed for the refractory ICP >25 mmHg and the sixth for clinical herniation. Five of the six patients died.
RESCUEicp
This international, multicentre, parallel-group, superiority, randomised trial assessed the comparative effectiveness of craniectomy versus advanced medical management (with the option of barbiturates), for patients with severe and refractory intracranial hypertension. The trial included patients aged 10 to 65 with a TBI and raised intracranial pressure (>25 mmHg for 1 to 12 h, despite stage 1 and 2 measures). In total, 408 patients were randomised, of whom 56 were ≤18 years and 16 patients were ≤16. The primary analysis showed a significant between-group difference in the extended Glasgow Outcome Scale (GOS-E) distribution and a substantial reduction in mortality with surgery. The pre-specified sensitivity analysis dichotomized at upper severe disability (independent at home) or better was significant at 12 months (i.e. 45.4% of the patients in the surgical group were at least independent at home, as compared with 32.4% of patients in the medical group; p = 0.01). Furthermore, we estimated that treating 100 patients with craniectomy as opposed to medical treatment would result in 22 more survivors of whom, at 12 months, almost 60% will be at least independent at home.
Complications
Frequently after decompression, the refractory swelling that results from a hyperaemic state can cause strangulation and herniation, particularly if the craniectomy is of insufficient size.
The most frequent complication that arises from decompressive surgery is disruption of CSF dynamics. In adults, hydrocephalus occurs between 14 and 29% of patients and hygromas in 26% [48,48,50]. Other complications include cerebral ischemia, infection, wound dehiscence, seizures, syndrome of the trephined and secondary brain injury as a result of an unprotected brain.
Hydrocephalus as a result of the trauma must be distinguished from that as a result of surgery. The resulting increase in pressure can promote CSF leak from the skin and predispose to infection. Options for treating hydrocephalus include external ventricular drainage, ventriculoperitoneal shunting or lumbar drainage if the cisterns allow. Frequently when the bone is replaced, hydrocephalus ex vacuo may resolve with only a subcohort of patients requiring a shunt after cranioplasty.
Hygromas usually occur on the ipsilateral side following surgery [48, 49]. Although most resolve spontaneously without intervention on occasion, they can require drainage or CSF diversion [48, 49], in children externalisation of drains is avoided where possible. Percutaneous drainage of epidural CSF collections may also be considered. On the whole replacement of the bone flap will usually improve CSF dynamics.
The extensive scalp incision and bony resection involved in craniectomy combined with lengthy intensive care unit stays and multiple invasive procedures all combined to increase the risk of infection in patients’ post-decompression. In addition to the 3–6% of infection, wound dehiscence is a major concern. Particularly, common in younger children due to the depth of the scalp, the tension expressed by the expanding brain can frequently cause the wound to open.
Seizures occur in both the acute and chronic phases of treatment with persistent seizures requiring medical intervention occurring in up to 5% of patients [48]. The reduction of Bovie cautery when replacing the bone flap has been demonstrated to reduce seizures significantly. Finally, syndrome of the trephined is particularly unique to decompressive procedures [51]. It is characterised by headaches, dizziness, mood changes or seizures as a result of a sunken flap after oedema has resolved. It usually improves after replacement of a bone flap.
Cranioplasty
In patients who undergo full craniectomy, the replacement of a bone flap weeks to months later will be required. The duration between primary surgery and craniooplasty varies between 1 and 12 months during which time there is an increased risk of injury to the unprotected brain [52]. This will be either replacement of the original flap or a synthetic cranioplasty. The original flap can either be stored in a certified tissue bank or in the abdominal subcutaneous wall of the patient. Abdominal storage requires both additional surgery to store and remove the flap and also holds potential risks of resorption, rhabdomyolysis and infection. Nevertheless, it offers a rapid replacement of the bone flap within a few weeks [Zabaty et al. 2015]. Alternatively, a synthetic implant can be used. These can be generated from polyetheretherketone (PEEK), porous polyethylene, acrylic or titanium.
Conclusions
The collective evidence of small case series within the literature suggests that large decompressive surgeries with duraplasty can be effective in paediatric patients with early signs of neurologic deterioration or herniation and in treating intracranial hypertension refractory to medical management. It is proposed that the reversal of such pathology may assist in improving outcomes in critically ill patients who have sustained a severe TBI. Although there is good evidence that DC controls ICP, the operation is associated with complications and the decision to undertake the procedure requires careful assessment and appropriate assessment of parents/guardians.
References
Hutchinson PJ, Jalloh I, Helmy A, Carpenter KL, Rostami E, Bellander BM, Boutelle MG, Chen JW, Claassen J, Dahyot-Fizelier C, Enblad P, Gallagher CN, Helbok R, Hillered L, Le Roux PD, Magnoni S, Mangat HS, Menon DK, Nordström CH, O'Phelan KH, Oddo M, Perez Barcena J, Robertson C, Ronne-Engström E, Sahuquillo J, Smith M, Stocchetti N, Belli A, Carpenter TA, Coles JP, Czosnyka M, Dizdar N, Goodman JC, Gupta AK, Nielsen TH, Marklund N, Montcriol A, O'Connell MT, Poca MA, Sarrafzadeh A, Shannon RJ, Skjøth-Rasmussen J, Smielewski P, Stover JF, Timofeev I, Vespa P, Zavala E, Ungerstedt U (2015) Consensus statement from the 2014 international microdialysis forum. Intensive Care Med 41(9):1517–1528
Berger MS, Pitts LH, Lovely M, Edwards MSB, Bartkowski HM (1985) Outcome from severe head injury in children and adolescents. J Neurosurg 62:194–199
Juul N, Morris GF, Marshall SB, The Executive Committee of the Interna- tional Selfotel Trial, Marshall LF (2000) Intracranial hypertension and cerebral perfusion pressure: influence on neurological deterioration and outcome in severe head injury. J Neurosurg 92:1–6
Marshall LF, Smith RW, Shapiro HM (1979) The outcome with aggressive treatment in severe head injuries. The significance of intracranial pressure monitoring. J Neurosurg 50:20–25
Pople IK, Mulbauer MS, Sanford RA, Kirk E (1995) Results and complications of intracranial pressure monitoring in 303 children. Pediatr Neurosurg 23:64–67
Czosnyka M, Smielewski P, Kirkpatrick P, Laing RJ, Menon D, Pickard JD (1997) Continuous assessment of the cerebral vasomotor reactivity in head injury. Neurosurgery 41(1):11–7; discussion 17–9
Figaji AA, Fieggen AG, Peter JC (2003) Early decompressive craniotomy in children with severe traumatic brain injury. Childs Nerv Syst 19(9):666–673
Agrawal S, Branco RG (2016) Neuroprotective measures in children with traumatic brain injury. World J Crit Care Med 5(1):36–46
Maas AI, Stocchetti N, Bullock R (2008) Moderate and severe traumatic brain injury in adults. Lancet Neurol 7(8):728–741
Young AM, Donnelly J, Czosnyka M, Jalloh I, Liu X, Aries MJ, Fernandes HM, Garnett MR, Smielewski P, Hutchinson PJ, Agrawal S (2016) Continuous multimodality monitoring in children after traumatic brain injury-preliminary experience. PLoS One 11(3):e0148817
Alexander E, Ball MR, Laster DW (1987) Subtemporal decompression: radiological observations and current surgical experience. Br J Neurosurg 1:427–433
Cooper DJ, Rosenfeld JV, Murray L, Arabi YM, Davies AR, D'Urso P, Kossmann T, Ponsford J, Seppelt I, Reilly P, Wolfe R, DECRA trial investigators (2011) Australian and new Zealand Intensive Care Society clinical trials group. Decompressive craniectomy in diffuse traumatic brain injury. N Engl J Med 364(16):1493–1502
Coplin WM, Cullen NK, Policheria PS, Vinas FC, Wilseck JM, Zafonte RD, Rengachary SS (2001) Safety and feasibility of craniectomy with duraplasty as the initial surgical intervention for severe traumatic brain injury. J Trauma 50:1050–1059
Gaab MR, Rittierodt M, Lorenz M, Heissler HE (1990) Traumatic brain swelling and operative decompression: a prospective investigation. Acta Neurochir Suppl 51:326–328
Gower DJ, Lee KS, McWhorter JM (1988) Role of subtemporal decompression in severe closed head injury. Neurosurgery 23:417–422
Kjellberg RA, Prieto A (1971) Bifrontal decompressive craniotomy for massive cerebral edema. J Neurosurg 34:488–493
Kolias AG, Adams H, Timofeev I, Czosnyka M, Corteen EA, Pickard JD, Turner C, Gregson BA, Kirkpatrick PJ, Murray GD, Menon DK, Hutchinson PJ (2016) Br J Neurosurg 30(2):246–250
Munch E, Horn P, Schurer L, Piepgras A, Torsten P, Smiedeck P (2000) Management of severe traumatic brain injury by decompressive craniectomy. Neurosurgery 47:315–323
Taylor A, Butt W, Rosenfeld J, Shann F, Ditchfield M, Lewis E, Klug G, Wallace D, Henning R, Tibbals J (2001) A randomized trial of very early decompressive craniectomy in children with traumatic brain injury and sustained intracranial hypertension. Childs Nerv Syst 17:154–162
Timofeev I, Santarius T, Kolias AG, Hutchinson PJ (2012) Decompressive craniectomy—operative technique and perioperative care. Adv Tech Stand Neurosurg 38:115–136
Walcott BP, Kahle KT, Simard JM (2013) The DECRA trial and decompressive craniectomy in diffuse traumatic brain injury: is decompression really ineffective? World Neurosurg 79(1):80–81
Whitfield PC, Patel H, Hutchinson PJA, Czosnyka M, Parry D, Menon D, Pickard JD, Kirkpatrick PJ (2001) Bifrontal decompressive craniectomy in the management of posttraumatic intracranial hypertension. Br J Neurosurg 15:500–507
Le Roux P (2015) Intracranial pressure monitoring and management. Translat Res Trauma Brain Inj:315–338
Ahmad FU, Bullock R (2011) Decompressive craniectomy for severe head injury. World Neurosurg 75(3–4):451–453
Carter BS, Ogilvy CS, Candia GJ, Rosas HD, Buonanno F (1997) One year outcome after decompressive surgery for massive nondominant hemispheric infarction. Neurosurgery 40:1168–1176
Yoo D, Kim D, Cho K, Huh P, Park C, Kang J (1999) Ventricular pressure monitoring during bilateral compression with dural expansion. J Neurosurg 91:953–959
Clark K, Nash T, Hutchison GC (1968) The failure of circumferential cranioto—my in acute traumatic cerebral swelling. J Neurosurg 29:367–371
Sahuquillo J, Martínez-Ricarte F, Poca MA (2013) Decompressive craniectomy in traumatic brain injury after the DECRA trial. Where do we stand? Curr Opin Crit Care 19(2):101–106
Mannion RJ, Cross J, Bradley P, Coles JP, Chatfield D, Carpenter A, Pickard JD, Menon DK, Hutchinson PJ (2007) Mechanism-based MRI classification of traumatic brainstem injury and its relationship to outcome. J Neurotrauma 24(1):128–135
Pereyra C, Benito Mori L, Schoon P, Violi D, Jacintho P, Segui G, Losio D, Lugaro M, Benavent G, Prieto M, Strati J, Diaz G (2012) Decompressive craniectomy and brain death prevalence and mortality: 8-year retrospective review. Transplant Proc 44(7):2181–2184
Shimoda K, Maeda T, Tado M, Yoshino A, Katayama Y, Bullock MR (2014) Outcome and surgical management for geriatric traumatic brain injury: analysis of 888 cases registered in the Japan Neurotrauma Data Bank. World Neurosurg 82(6):1300–1306
Bullock MR, Merchant RE, Choi SC, Gilman CB, Kreutzer JS, Marmarou A, Teasdale GM (2002) Outcome measures for clinical trials in neurotrauma. Neurosurg Focus 13(1):ECP1
Gürer B, Kertmen H, Yilmaz ER, Sekerci Z (2013) The surgical outcome of traumatic extra-axial hematomas causing brain herniation in children. Pediatr Neurosurg 49(4):215–222
Miller MT, Pasquale M, Kurek S, White J, Martin P, Bannon K, Wasser T, Li M (2004) Initial head computed tomographic scan characteristics have a linear relationship with initial intracranial pressure after trauma. J Trauma 56(5):967–972 discussion 972-3
Kouvarellis AJ, Rohlwink UK, Sood V, Van Breda D, Gowen MJ, Figaji AA (2011) The relationship between basal cisterns on CT and time-linked intracranial pressure in paediatric head injury. Childs Nerv Syst 27(7):1139–1144
Agrawal S, Brierley J (2012) Optic nerve sheath measurement and raised intracranial pressure in paediatric traumatic brain injury. Eur J Trauma Emerg Surg 38(1):75–77
Brady KM, Shaffner DH, Lee JK, Easley RB, Smielewski P, Czosnyka M, Jallo GI, Guerguerian AM (2009) Continuous monitoring of cerebrovascular pressure reactivity after traumatic brain injury in children. Pediatrics 124(6):e1205–e1212
Lewis PM, Czosnyka M, Carter BG, Rosenfeld JV, Paul E, Singhal N, Butt W (2015) Cerebrovascular pressure reactivity in children with traumatic brain injury. Pediatr Crit Care Med 16(8):739–749
O'Leary R, Hutchinson PJ, Menon D (2016) Hypothermia for intracranial hypertension after traumatic brain injury. N Engl J Med 374(14):1383–1384
Kochanek PM, Carney N, Adelson PD, Ashwal S, Bell MJ, Bratton S, Carson S, Chesnut RM, Ghajar J, Goldstein B, Grant GA, Kissoon N, Peterson K, Selden NR, Tasker RC, Tong KA, Vavilala MS, Wainwright MS, Warden CR, American Academy of Pediatrics-Section on Neurological Surgery; American Association of Neurological Surgeons/Congress of Neurological Surgeons; Child Neurology Society; European Society of Pediatric and Neonatal Intensive Care; Neurocritical Care Society; Pediatric Neurocritical Care Research Group; Society of Critical Care Medicine; Paediatric Intensive Care Society UK; Society for Neuroscience in Anesthesiology and Critical Care; World Federation of Pediatric Intensive and Critical Care Societies (2012) Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents—second edition. Pediatr Crit Care Med 13 Suppl 1:S1–82
Hejazi N, Witzmann A, Fae P (2002) Unilateral decompressive craniectomy for children with severe brain injury. Report of seven cases and review of the relevant literature. Eur J Pediatr 161:99–104
Ruf B, Heckmann M, Schroth I, Hügens-Penzel M, Reiss I, Borkhardt A, Gortner L, Jödicke A (2003) Early decompressive craniectomy and duraplasty for refractory intracranial hypertension in children: results of a pilot study. Crit Care 7:R133–R138
Jagannathan J, Okonkwo DO, Dumont AS, Ahmed H, Bahari A, Prevedello DM, Jane JA Sr, Jane JA Jr (2007) Outcome following decompressive craniectomy in children with severe traumatic brain injury: a 10-year single-center experience with long-term follow up. J Neurosurg 106:268–275
Kan P, Amini A, Hansen K, White GL Jr, Brockmeyer DL, Walker ML, Kestle JR (2006) Outcomes after decompressive craniectomy for severe traumatic brain injury in children. J Neurosurg 105:337–342
Rutigliano D, Egnor MR, Priebe CJ, McCormack JE, Strong N, Scriven RJ, Lee TK (2006) Decompressive craniectomy in pediatric patients with traumatic brain injury with intractable elevated intracranial pressure. J Pediatr Surg 41:83–87 discussion 83–87
Cho DY, Wang YC, Chi CS (1995) Decompressive craniotomy for acute shaken/impact baby syndrome. Pediatr Neurosurg 23:192–198
Skoglund TS, Eriksson-Ritzen C, Jensen C, Rydenhag B (2006) Aspects on decompressive craniectomy in patients with traumatic head injuries. J Neurotrauma 23:1502–1509
Kurland DB, Khaladj-Ghom A, Stokum JA, Carusillo B, Karimy JK, Gerzanich V, Sahuquillo J, Simard JM (2015) Complications associated with decompressive craniectomy: a systematic review. Neurocrit Care 23:292–304 Online publication date: 1-Oct-2015
Aarabi B, Hesdorffer DC, Ahn ES, Aresco C, Scalea TM, Eisenberg HM (2006) Outcome following decompressive craniectomy for malignant swelling due to severe head injury. J Neurosurg 104(4):469–479
Schuss P, Borger V, Güresir Á, Vatter H, Güresir E (2015) Cranioplasty and ventriculoperitoneal shunt placement after decompressive craniectomy: staged surgery is associated with fewer postoperative complications. World Neurosurg 84(4):1051–1054
Grantham EG, Landis HP (1948) Cranioplasty and the post-traumatic syndrome January Vol. 5 pp 19–22
Tsang AC, Hui VK, Lui WM, Leung GK (2015) Complications of post-craniectomy cranioplasty: risk factor analysis and implications for treatment planning. J Clin Neurosci 22(5):834–837
Acknowledgements
Adam Young is supported by an NIHR Academic Clinical Fellowship. Angelos Kolias is supported by a Clinical Lectureship, School of Clinical Medicine, University of Cambridge. Peter Hutchinson is supported by a Research Professorship from the National Institute for Health Research (NIHR), the NIHR Cambridge Biomedical Research Centre, a European Union Seventh Framework Program grant (CENTER-TBI; grant no. 602150), and the Royal College of Surgeons of England.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Young, A.M.H., Kolias, A.G. & Hutchinson, P.J. Decompressive craniectomy for traumatic intracranial hypertension: application in children. Childs Nerv Syst 33, 1745–1750 (2017). https://doi.org/10.1007/s00381-017-3534-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-017-3534-7