Abstract
Purpose
Patients aged > 80 years represent an increasing proportion of colon cancer diagnoses. Selecting patients for elective surgery is challenging because of possibly compromised health status and functional decline. The aim of this retrospective, population-based study was to identify risk factors and health measures that predict short-term mortality after elective colon cancer surgery in the aged.
Methods
All patients > 80 years operated electively for stages I–III colon cancer from 2005 to 2016 in four Finnish hospitals were included. The prospectively collected data included comorbidities, functional status, postoperative surgical and medical outcomes as well as mortality data.
Results
A total of 386 patients (mean 84.0 years, range 80–96, 56% female) were included. Male gender (46% vs 35%, p = 0.03), higher BMI (51% vs 37%, p = 0.02), diabetes mellitus (51% vs 37%, p = 0.02), coronary artery disease (52% vs 36%, p = 0.003) and rheumatic diseases (67% vs 39%, p = 0.03) were related to higher risk of complications. The severe complications were more common in patients with increased preoperative hospitalizations (31% vs 15%, p = 0.05) and who lived in nursing homes (30% vs 17%, p = 0.05). The 30-day and 1-year mortality rates were 6.0% and 15% for all the patients compared with 30% and 45% in patients with severe postoperative complications (p < 0.001). Severe postoperative complications were the only significant patient-related variable affecting 1-year mortality (OR 9.60, 95% CI 2.33–39.55, p = 0.002).
Conclusions
The ability to identify preoperatively patients at high risk of decreased survival and thus prevent severe postoperative complications could improve overall outcome of aged colon cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of colorectal cancer has tripled during the past five decades, and the risk of having colorectal cancer increases with age [1]. According to the Finnish Cancer Registry data from 2016, 27% of all colorectal patients were aged 80 years or more [1]. The number of aged people diagnosed with colorectal cancer will increase as the population ages [2].
If possible, colon cancer is managed by radical surgical resection [3, 4]. Previous studies show comparable disease-specific survival rates for all age groups, which advocates tumour resection with curative purpose [5, 6]. In the EUROCARE-5 study, 5-year overall survival rate was 49% for colon cancer patients of 75 years or older while overall survival rate for all ages was 57% in Europe [7]. Corresponding numbers from 2016 in Finland were 60% for patients of 75 years and 66% for all ages [1]. In the oldest patients, however, the survival benefit of operative treatment (compared with other treatments) is only apparent after the first two postoperative years due to the risks of surgery [8,9,10].
Increased age, physical and cognitive disabilities, and previous hospitalizations may predispose to postoperative morbidity and mortality [11]. Colorectal cancer surgery is considered high risk, with reported postoperative major complication and 30-day mortality rates of 16–24% and 1–6%, respectively [12, 13]. Operative treatment of colorectal cancer in the aged is associated with higher morbidity (35–45%) and mortality rates both in the immediate postoperative period and during the first year after surgery (6–16% in 30 days, 14–37% at 1 year) [12,13,14,15,16,17]. Compared with younger people, the 30-day mortality in the group of aged patients underestimates 1-year mortality due to prolonged impact of the perioperative period [8, 18]. However, a recent Dutch study shows better postoperative outcome with almost equal 1-year survival rates for older and younger colorectal patients, supporting tailor-made decisions on surgical treatments for older patients [19].
It is important to have relevant and reliable data concerning older aged colorectal cancer patient’s surgery and postoperative morbidity and survival numbers. Surgeons and oncologists treating patients with colon cancer should become aware of that 43% of their patients are > 75 years and that many of these older patients are frail with comorbidities which demand careful patient assessment [20]. Proper patient selection and careful consideration of appropriate surgical candidates including preoperative optimization of medical comorbidities, nutritional status and physical performance enables colorectal cancer surgery to be performed with reduced morbidity and mortality rates with improved survival [21, 22].
The aim of this study was to identify preoperative risk factors and measures of overall health status affecting postoperative morbidity and mortality and their impact on 30-day and 1-year survival in colon cancer patients aged 80 years and older. In addition, the aim was to investigate time-trends in postoperative short-term mortality and possible factors affecting the outcome.
Material and methods
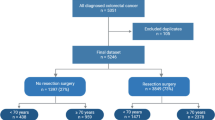
All patients aged 80 years and over who underwent an elective resection for colon cancer during the period of 2005 and 2016 in four Finnish hospitals were retrospectively analyzed. The catchment area of these hospitals ranged from 100,000 to 250,000 inhabitants (in total, approximately 750,000 inhabitants). Exclusion criteria were metastatic or recurrent disease and palliative or emergency surgery. Treatment of malignant diseases is almost exclusively performed by public health care system in Finland. We consider this study population-based because patients are referred to certain hospital based on their place of residence.
All the patients had histologically confirmed primary adenocarcinoma of the colon, and preoperatively had colonoscopy and computed tomography. They underwent radical surgery for the primary tumour. Patients were evaluated for their fitness for elective surgery with general anaesthesia but were not preselected any other way. Decision to proceed with surgery was based on patient’s general condition and comorbidities. Prior geriatric evaluation was not routinely used.
For this study, cancer- and surgery-related data was collected from prospectively maintained, institutionally approved and password-protected electronic colorectal databases in the study hospitals and was supplemented by review of medical records. Patients with colorectal cancer were identified using ICD codes C18-C19 [23]. The collected clinical data included patients´ characteristics, living status, hospitalizations in the 6 months before surgery, comorbidities, modified age-adjusted Charlson Comorbidity Index [24] score (4–15; solid tumour excluded with two points, all patients > 80 years which contributes four points for all patients), other malignancies, symptoms, operative procedures and postoperative recovery. Postoperative complications were defined and determined using the Clavien-Dindo classification [25], and severe complications were graded into classes III–V. Tumours were staged according to the Union for International Cancer Control (UICC) TNM classification [26] and graded according to the World Health Organization histological classification system [27]. The number of lymph nodes was recorded in every case. Patients with macroscopic or residual tumour were excluded.
Postoperative surveillance was performed in out-patient clinics according to the follow-up programs of the four hospitals. Local recurrences and distant metastasis were identified radiologically (ultrasound, computed tomography) or histologically. The information was collected from the hospital medical records and Statistics Finland for all patients with or without arranged follow-up program.
The primary outcome measures were postoperative morbidity (surgical and non-surgical complications) and mortality 30 days and 1 year after primary treatment. Mortality data including causes of death were obtained from the population-based statistics of Statistics Finland. Mortality was assessed at 30 days and 1 year following surgery. The follow-up duration data was calculated from the date of surgery to the date of death or date of active follow-up in the clinics (1 year after surgery).
The associations between categorical variables were tested with the chi-square test or the Fisher exact test, when appropriate, and continuous variables with the Student’s t test. Survival rates were calculated from the time of primary surgery using the Kaplan-Meier method and were compared by the log-rank test. A uni- and multivariable analysis of the factors which influenced 30-day and 1-year mortality was carried out using binary logistic regression. All variables (Table 4) that were statistically significant in the univariate model were included in the multivariable model. Statistical analyses were performed using SPSS version 23.
The study was approved by the ethics committee of Pirkanmaa Hospital District, Tampere, Finland (R18188).
Results
A total of 386 patients were identified from the prospective records of the four hospitals. The mean age of the study population was 84.1 years (range 80–96 years). Most of the patients were female (56%), had an ASA III classification (69%) and CCI score 4–5 (62%). Most of the patients lived at home (90%), were not dependent on others for daily help (56%) and had no hospital admission in the previous 6 months (73%). Of the patients, 106 (28%) were operated in 2005–2008, 143 (37%) in 2009–2012 and 137 (35%) in 2013–2016. Table 1 shows the patients` baseline characteristics.
Most of the operative procedures were performed for right-sided colon cancer (68%). An intended laparoscopic resection was performed in 252 patients (65%) and in 35 cases (14%), the operation was converted to an open surgery due to anatomical or technical reasons. One hundred eighty-seven patients (50%) were discharged to other hospitals or wards of healthcare centres. Readmission within 30 days of discharge occurred for 6.5% of the patients. Postoperative adjuvant chemotherapy was given to 46% (67/146) of stage III patients. Table 2 shows surgical characteristics and outcomes.
Morbidity
Overall postoperative morbidity was 40% (154/386), with 24% (92/386) of patients having surgical complications. The most common surgical complications were ileus (7.0%), intra-abdominal or anastomotic bleeding (6.2%) and anastomotic leakage (5.7%). The most common non-surgical complications were cardiovascular (6.0%) and pulmonary (4.7%). Both surgical and non-surgical complications were recorded only for 1.8% (7/386) of the patients. According to the Clavien-Dindo classification, 18% (69/386) of patients had severe complications (grades III–V), accounting for 45% (69/154) of all complications. (Table 3).
Male gender (46% vs 35%, p = 0.03), diabetes mellitus (51% vs 37%, p = 0.02), coronary artery disease (52% vs 36%, p = 0.003) and rheumatic diseases (67% vs 39%, p = 0.03) were related to higher number of complications. Patients with complications had higher BMI (26 vs 25, p = 0.04). Operative approach (open or laparoscopic) and operating time were not associated with increased rate of complications.
More severe postoperative complications (Clavien-Dindo III–V) were experienced by patients living in a nursing home compared with those living in their own home (30% vs 17%, p = 0.05), and by those who had hospital admissions during the 6 months prior surgery compared with those who had none (31% vs 15%, p = 0.05). Patients with severe complications also had more intraoperative blood loss (median 50 ml vs 100 ml, p = 0.006).
Mortality
The overall 30-day mortality was 6.0% (23/386). Six patients died due to anastomotic leakage and three due to other surgical complications (ileus, intraoperative haemorrhage and small intestine perforation). Seven patients died due to cardiac complications, three due to pulmonary problems and three due to cerebral haemorrhage. One patient died of a ruptured abdominal aneurysm 6 days after surgery.
The overall 1-year mortality was 15% (59/386). The most important causes of death were cardiopulmonary (43%) and surgery-related postoperative complications (17%). Cancer-related mortality was 4.4%, accounting for 29% (17/59) of deaths within 1 year. Over half (55%, 17/31) of the patients who had severe, surgery-related complications (anastomotic leakage, intra-abdominal haemorrhage, ileus) died within 1 year after surgery whereas 1-year mortality was 8.6% (20/232) for the patients without postoperative complications. In patients with mild complications (Clavien-Dindo I–II), mortality was 9.4%. Patients with severe postoperative complications (Clavien-Dindo III–V) had mortality 45% (31/69). (Fig. 1).
Patients with stage III disease (n = 146) and treated with postoperative chemotherapy had lower 1-year mortality rate compared with patients not receiving adjuvant treatment (10.4% vs 27.8%, p = 0.007).
Univariate logistic regression analysis showed that living in a nursing home, hospital admissions 6 months before surgery, CCI score > 6, longer operation time, greater intraoperative blood loss and severe postoperative complications were significant patient-related variables affecting 30-day mortality. Higher age, hospital admissions 6 months before surgery, open compared with laparoscopic operation, longer operative time, greater intraoperative blood loss and severe postoperative complications were associated with greater 1-year mortality after surgery (Table 4). In multivariate logistic regression analysis, hospital admissions 6 months before surgery and severe postoperative complications were the only significant patient-related variables affecting 30-day mortality (OR 3.05, 95% CI 1.25–7.43, p = 0.014 and OR 85.61, 95% CI 10.68–686.1, p < 0.001). Severe postoperative complications were the only significant patient-related variable affecting 1-year mortality (OR 9.60, 95% CI 2.33–39.55, p = 0.002).
Mortality rates in 30 days seemed to decrease between operation years 2005–2008 (7.5%), 2009–2012 (5.6%) and 2013–2016 (5.1%). The corresponding numbers in 1 year were 21% (2005–2008), 14% (2009–2012) and 12% (2013–2016). For severe complications (CD III–V), the 30-day mortality rates were 2005–2008 (30.8%), 2009–2012 (35%) and 2013–2016 (26.1%). The corresponding numbers in 1 year were 50% (2005–2008), 45% (2009–2012) and 39% (2013–2016). There was no statistically significant difference between time-trends and mortality.
Table 4 shows predictors for 30-day and 1-year mortality in univariate analysis (binary logistic regression).
Discussion
For this retrospective population-based, cohort study, short-term morbidity and mortality differences were analyzed in the electively operative non-metastatic colon cancer patients aged 80 years and over in a large, regionally representable materials. To our knowledge, our material consisting of 386 patients is the largest dataset regarding elective colon cancer surgery with curative intent and focusing on both postoperative complications and short-term mortality in this age group. Most of the similar studies featured surgical patients with colorectal cancers [6, 8, 14, 28]. Patients undergoing resection for rectal cancer have more personalized, multimodal treatment options and a greater number of severe complications such as anastomotic leakage compared with colon cancer surgery [29]. Thus, the patient data in our study was more homogenous focusing only on colon surgery. Furthermore, in Finland, treatment of colorectal cancer is based on national EBM guidelines [30]. The study hospitals also had standardized protocols for colon cancer treatments, and the patients’ baselines as well as operative outcomes and tumour characteristics were similar.
For non-metastatic colorectal cancer patients, surgical resection is the best option for curative treatment [3, 4]. A recent multinational study showed that patients over 80 years have 30-day and 1-year mortality rates of 5.5–11.4% and 17.1–23.6%, respectively [16]. In the present study, 1-year survival rate was 91.4% for the patients without postoperative complications, and the overall 1-year mortality was 15.3%. Most of the older patients do as well as younger patients after elective surgery and have acceptable survival numbers. For example, recent data from Netherlands showed similar 1-year overall survival rates for patients under and over 75 years [18]. However, 30-day mortality rate remains high among the oldest, and it continues to increase well beyond the initial postoperative month causing excess 1-year mortality. Previous studies show that major postoperative complications are related to higher mortality rates and are among the strongest risk factors for reduced survival in the aged [28, 31]. Our data showed similar postoperative morbidity and survival numbers reflecting the results of previous studies considering excess mortality beyond initial postoperative month [8, 13]. In our study, 1-year mortality rate of patients with major postoperative complications was 44.9%, this being the most important factor for diminished short-term survival after colon surgery.
Our study indicated high morbidity rates in early postoperative period. Almost 40% (153/386) of the patients developed postoperative complications, and 45% (69/153) of these were major complications. These figures are comparable with other studies of colorectal cancer surgery reporting total complication rates 35–45% and major complications 20–25%, respectively [14,15,16,17, 31]. Our data included all patients who were willing and fit enough for surgery according to ASA classification and other comorbidities. Risk factors for complications were male gender, diabetes, coronary artery and rheumatic diseases. Severe complications were more common in patients who had increased preoperative hospitalizations and who lived in nursing homes. These findings support recent studies from Norway and Japan [32, 33] emphasizing the importance of preoperative evaluation of patient’s physical and cognitive performance, to prevent complications in colon cancer surgery. In Finland, aged people move to nursing homes when they cannot survive with maximal help at home and most of them have some degree of cognitive impairment. Therefore, residing in a nursing home is a kind of proxy for cognitive impairment and disability in activities of daily living. Unfortunately, the diagnoses for hospitalizations before surgery were not documented accurately in our study data, and the reasons for hospitalizations were heterogenous.
Postoperative complications are associated with a disproportionately high risk of 30-day and 1-year mortality in elderly patients [28, 31]. In our study, the overall 30-day mortality rate was 6.0% but increased to 30.4% in patients with severe postoperative complications. Other factors associated with increased 30-day mortality included living in a nursing home, hospital admissions 6 months before surgery, CCI score > six, longer operation time, greater intraoperative blood loss and severe postoperative complications. Patients with diminished physical and mental resources had more severe postoperative complications leading to higher mortality rates indicating that limited performance state of patients plays an important role in postoperative outcome [33]. Cardiopulmonary problems and surgery-related issues were the most important factors contributing to 1-year mortality whereas cancer-related mortality was significantly low. This finding emphasizes the importance of preoperative evaluation on patient’s physical and cognitive performance with specific focus on preoperative cardiopulmonary status and medical optimization. Non-surgical, symptomatic treatments are worth considering when individualized treatment decisions are made for patients with limited performance status. On the contrary, patients with stage III disease treated with postoperative chemotherapy had significantly lower 1-year mortality rate compared with those who were not giving postoperative adjuvant treatment. That underlines the importance of identify patients who can benefit from the radical cancer treatments.
30-day and 1-year mortality rates decreased progressively between operation years 2005–2008, 2009–2012 and 2013–2016, but this was not statistically significant. During the study period, there were no preoperative arrangements for nutrition and physical performance status assessment, or geriatric assessment. Conversely, all the participating hospitals in our study had high proportion (56%) of operations done by laparoscopy, and enhanced recovery after surgery (ERAS) program was gradually adopted to clinical work during the study period. A recent meta-analysis demonstrated that laparoscopic colorectal surgery has positive impact in terms of significant decrease in postoperative morbidity and mortality among the aged compared with open colorectal surgery [34]. ERAS is a multimodal approach that aims to optimize perioperative management [35]. Old patients adhered to and benefited from an ERAS program have similar postoperative outcome advantages to their younger counterparts [36]. These facts can partly explain the similar time-trends in morbidity and mortality between the study years, together with developments in perioperative anaesthesia care, surgical performance (laparoscopy, technical standardization) and oncological treatments.
Some limitations exist in our study. First, although we were able to categorize specific comorbidities, we found little relationship between multiple morbidities and overall survival. This may reflect the fact that there was no information available regarding the severity of the comorbidities. Additionally, we were unable to fully assess disability and functional as well as cognitive performance state of patients because of imprecise patient files. Information about daily physical and cognitive functional activity and general performance was not routinely recorded. It is likely that the conditions that lead to a patient being considered frail are only documented in those patients whose condition had significant negative impact on their overall well-being. The same applies to comorbidities. However, undervaluation of such conditions would lead to underrate of the risks and false negative results, and hence, the reported risks are rather underestimates than overestimates. In addition, and result of the previous reasons, healthier patients were likely selected for surgical treatment. However, our findings suggest that surgical treatment in aged patients diagnosed with colon cancer can lead to acceptable and similar results compared with younger patients.
Our data indicate that postoperative morbidity following elective colon cancer surgery is common in aged patients and results in a significant increase in mortality, which lasts beyond the first postoperative month. Age, comorbidities, disability and the occurrence of severe postoperative complications were factors most strongly associated with reduced 30-day and 1-year survival after surgery. Although we have demonstrated an increase in survival rates, the impact of surgical treatment on postoperative functional outcome is not clear. Regardless of the new achievements, there seems to be a tendency to offer fewer surgical resections to older compared with younger patients [37, 38]. Therefore, the information on present-day postoperative morbidity and mortality rates of older patients is important for shared decision-making regarding surgical treatment. If aged colon cancer patients survive the first year after surgery, they have a reasonably good long-term survival. Older patients often give higher priority to functional outcomes than to survival [39]. Further research should therefore focus not only on further increasing postoperative survival rates in older patients but also on quality of life and improvement of postoperative physical functioning. Prehabilitation programs could play a role in achieving this goal [40, 41].
In conclusion, mortality in the present study is related to postoperative complications. Identifying effective strategies for both prevention and tailor-made treatments of postoperative complications in colon cancer surgery in the aged could potentially improve patient overall outcomes.
References
Finnish Cancer Registry. www.cancerregistry.fi. Accessed 6 Oct 2019
Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F (2017) Global patterns and trends in colorectal cancer incidence and mortality. Gut 66(4):683–691
Schmoll HJ, Van Cutsem E, Stein A, Valentini V, Glimelius B, Haustermans K et al (2012) ESMO consensus guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol 23(10):2479–2516
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S (2009) Standardized surgery for colonic cancer: complete mesocolic excision and central ligation--technical notes and outcome. Color Dis 11(4):354–364
Mothes H, Bauschke A, Schuele S, Eigendorff E, Altendorf-Hofmann A, Settmacher U (2017) Surgery for colorectal cancer in elderly patients: how can we improve outcome? J Cancer Res Clin Oncol 143(9):1879–1889
Mäkelä JT, Klintrup KH, Rautio TT (2017) Mortality and survival after surgical treatment of colorectal cancer in patients aged over 80 years. Gastrointest Tumors 4(1–2):36–44
Holleczek B, Rossi S, Domenic A, Innos K, Minicozzi P, Francisci S et al (2015) Ongoing improvement and persistent differences in the survival for patients with colon and rectum cancer across Europe 1999-2007. Results from the EUROCARE-5 study. Eur J Cancer 51(15):2158–2168
Dekker JW, van den Broek CB, Bastiaannet E, van de Geest LG, Tollenaar RA, Liefers GJ (2011) Importance of the first postoperative year in the prognosis of elderly colorectal cancer patients. Ann Surg Oncol 18(6):1533–1539
Ng O, Watts E, Bull CA, Morris R, Acheson A, Banerjea A (2016) Colorectal cancer outcomes in patients aged over 85 years. Ann R Coll Surg Engl 98(3):216–221
Abdel-Halim M, Wu H, Poustie M, Beveridge A, Scott N, Mitchell PJ (2019) Survival after non-resection of colorectal cancer: the argument for including non-operatives in consultant outcome reporting in UK. Ann R Coll Surg Engl 101(2):126–132
Neuman HB, Weiss JM, Leverson G, O'Connor ES, Greenblatt DY, Loconte NK et al (2013) Predictors of short-term postoperative survival after elective colectomy in colon cancer patients ≥ 80 years of age. Ann Surg Oncol 20(5):1427–1435
Morris EJ, Taylor EF, Thomas JD, Quirke P, Finan PJ, Coleman MP, Rachet B, Forman D (2011) Thirty-day postoperative mortality after colorectal cancer surgery in England. Gut 60(6):806–813
Al-Refaie WB, Parsons HM, Habermann EB, Kwaan M, Spencer MP, Henderson WG, Rothenberger DA (2011) Operative outcomes beyond 30-day mortality: colorectal cancer surgery in oldest old. Ann Surg 253(5):947–952
Ihedioha U, Gravante G, Lloyd G, Sangal S, Sorge R, Singh B, Chaudhri S (2013) Curative colorectal resections in patients aged 80 years and older: clinical characteristics, morbidity, mortality and risk factors. Int J Color Dis 28(7):941–947
Aquina CT, Mohile SG, Tejani MA, Becerra AZ, Xu Z, Hensley BJ et al (2017) The impact of age on complications, survival, and cause of death following colon cancer surgery. Br J Cancer 116(3):389–397
Claassen YHM, Bastiaannet E, van Eycken E, Van Damme N, Martling A, Johansson R et al (2019) Time trends of short-term mortality for octogenarians undergoing a colorectal resection in North Europe. Eur J Surg Oncol 45(8):1396–1402
Duraes LC, Stocchi L, Dietz D, Kalady MF, Kessler H, Schroeder D, Remzi FH (2016) The disproportionate effect of perioperative complications on mortality within 1 year after colorectal cancer resection in octogenarians. Ann Surg Oncol 23(13):4293–4301
Bos ACRK, Kortbeek D, van Erning FN, Zimmerman DDE, Lemmens VEPP, Dekker JWT et al (2019) Postoperative mortality in elderly patients with colorectal cancer: the impact of age, time-trends and competing risks of dying. Eur J Surg Oncol 45(9):1575–1583
Brouwer NPM, Heil TC, Olde Rikkert MGM, Lemmens VEPP, Rutten HJT, de Wilt JHW, van Erning F (2019) The gap in postoperative outcome between older and younger patients with stage I-III colorectal cancer has been bridged; results from the Netherlands cancer registry. Eur J Cancer 116:1–9
Papamichael D, Audisio RA, Glimelius B, de Gramont A, Glynne-Jones R, Haller D et al (2013) Treatment of colorectal cancer in older patients: International Society of Geriatric Oncology (SIOG) consensus recommendations 2013. Ann Oncol 26(3):463–476
Örum M, Gregersen M, Jensen K, Meldgaard P, Damsgaard EMS (2018) Frailty status but not age predict complications in elderly cancer patients: a follow-up study. Acta Oncol 57(11):1458–1466
Kunitake H, Zingmond DS, Ryoo J, Ko CY (2010) Caring for octogenarian and nonagenarian patients with colorectal cancer: what should our standards and expectations be? Dis Colon Rectum 53(5):735–743
WHO (2010) International statistical classification of diseases and related health problems. 10th revision
Chang CM, Yin WY, Wei CK, Wu CC, Su YC, Yu CH, Lee CC (2016 Feb 5) Adjusted age-adjusted Charlson comorbidity index score as a risk measure of perioperative mortality before cancer surgery. PLoS One 11(2):e0148076. https://doi.org/10.1371/journal.pone.0148076
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Sobin LH, Gospodarowicz MK, Wittekind CH (2009) TNM classification of malignant tumours, 7th edn. Wiley-Blackwell, Hoboken
Loughrey MB, Quirke P, Shepherd NA (2017) Dataset for histopathological reporting of colorectal cancer, 4th edn. The Royal College of Pathologists, London
Weerink LBM, Gant CM, van Leeuwen BL, de Bock GH, Kouwenhoven EA, Faneyte IF (2018) Long-term survival in octogenarians after surgical treatment for colorectal cancer: prevention of postoperative complications is key. Ann Surg Oncol 25(13):3874–3882
Montroni I, Ugolini G, Saur NM, Spinelli A, Rostoft S, Millan M, Wolthuis A, Daniels IR, Hompes R, Penna M, Fürst A, Papamichael D, Desai AM, Cascinu S, Gèrard JP, Myint AS, Lemmens VEPP, Berho M, Lawler M, de Liguori Carino N, Potenti F, Nanni O, Altini M, Beets G, Rutten H, Winchester D, Wexner SD, Audisio RA (2018) Personalized management of elderly patients with rectal cancer: expert recommendations of the European Society of Surgical Oncology, European Society of Coloproctology, International Society of Geriatric Oncology, and American College of Surgeons Commission on Cancer. Eur J Surg Oncol 44(11):1685–1702
https://www.terveysportti.fi/dtk/ltk/koti?p_artikkeli=hsu00007. Accessed 6. Oct 2019
Gooiker GA, Dekker JW, Bastiaannet E, van der Geest LG, Merkus JW, van de Velde CJ, Tollenaar RA, Liefers GJ (2012) Risk factors for excess mortality in the first year after curative surgery for colorectal cancer. Ann Surg Oncol 19(8):2428–2434
Furnes B, Storli KE, Forsmo HM, Karliczek A, Eide GE, Pfeffer F (2019) Risk factors for complications following introduction of radical surgery for colon cancer: a consecutive patient series. Scand J Surg 108(2):144–151
Okabe H, Ohsaki T, Ogawa K, Ozaki N, Hayashi H, Akahoshi S et al (2019) Frailty predicts severe postoperative complications after elective colorectal surgery. Am J Surg 217(4):677–681
Seishima R, Okabayashi K, Hasegawa H, Tsuruta M, Shigeta K, Matsui S, Yamada T, Kitagawa Y (2015) Is laparoscopic colorectal surgery beneficial for elderly patients? A systematic review and meta-analysis. J Gastrointest Surg 19(4):756–765
Fearon K, Ljungqvist O, Von Meyenfeldt M, Revhaug A, Dejong CH, Lassen K et al (2005) Enhanced recovery after surgery: a consensus review of clinical care for patients undergoing colonic resection. Clin Nutr 24(3):466–477
Forsmo HM, Erichsen C, Rasdal A, Körner H, Pfeffer F (2017) Enhanced recovery after colorectal surgery (ERAS) in elderly patients is feasible and achieves similar results as in younger patients. Geront Geriatr Med 3:2333721417706299
Egenvall M, Schubert Samuelsson K, Klarin I, Lökk J, Sjövall A, Martling A et al (2014) Management of colon cancer in the elderly: a population-based study. Color Dis 16(6):433–441
Doat S, Thiébaut A, Samson S, Ricordeau P, Guillemot D, Mitry E (2014) Elderly patients with colorectal cancer: treatment modalities and survival in France. National data from the ThInDiT cohort study. Eur J Cancer 50(7):1276–1283
Banks E, Byles JE, Gibson RE, Rodgers B, Latz IK, Robinson IA et al (2010) Is psychological distress in people living with cancer related to the fact of diagnosis, current treatment or level of disability? Findings from a large Australian study. Med J Aust 193(5 Suppl):S62–S67
Ommundsen N, Wyller TB, Nesbakken A, Bakka AO, Jordhøy MS, Skovlund E et al (2018) Preoperative geriatric assessment and tailored interventions in frail older patients with colorectal cancer: a randomized controlled trial. Color Dis 20(1):16–25
Van Rooijen S, Carli F, Dalton S, Thomas G, Bojesen R, Le Guen M et al (2019) Multimodal prehabilitation in colorectal cancer patients to improve functional capacity and reduce postoperative complications: the first international randomized controlled trial for multimodal prehabilitation. BMC Cancer 19(1):98. https://doi.org/10.1186/s12885-018-5232-6
Acknowledgments
The authors thank warmly M.D. Ville Väyrynen Central Hospital of Central Finland and M.D. Anu Välikoski Tays Hatanpää Tampere University Hospital for their dedicated data collection.
Funding
This study was financially partly supported by the Competitive State Research Financing of the Expert Responsibility area of Tampere University Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the ethics committee of Pirkanmaa Hospital District, Tampere, Finland (R18188).
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niemeläinen, S., Huhtala, H., Ehrlich, A. et al. Risk factors of short-term survival in the aged in elective colon cancer surgery: a population-based study. Int J Colorectal Dis 35, 307–315 (2020). https://doi.org/10.1007/s00384-019-03488-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-019-03488-8