Abstract
Background
Pioglitazone was used to treat patients of PCOS in many researches, but the treatment has not been recognized by public or recommended by all the guidelines.
Method
We conducted a meta-analysis of the related literatures to objectively evaluate the clinical effectiveness and safety by comparing pioglitazone with metformin administrated by PCOS patients. Searches were performed in Cochrane Library, EMBASE and PubMed (last updated December 2016).
Results
Eleven studies among 486 related articles were identified through searches. Fixed effects and random effects models were used to calculate the overall risk estimates. The results of the meta-analysis suggest that improvement of the menstrual cycle and ovulation in pioglitazone treatment group was better than metformin group [OR = 2.31, 95% CI (1.37, 3.91), P < 0.001, I 2 = 41.8%]. Improvement of the F-G scores in metformin treatment group was better than pioglitazone group [SMD = 0.29, 95% CI (0.0, 0.59), P = 0.048, I 2 = 0.0%]. BMI was more elevated in pioglitazone group than in metformin group [SMD = 0.83, 95% CI (0.24, 1.41), P = 0.006, I 2 = 82.8%]. There were no significant differences of the other data between the two groups.
Conclusions
This meta-analysis indicated that pioglitazone ameliorated menstrual cycle and ovulation better than metformin and metformin ameliorated BMI and F-G scores better than pioglitazone in treating patients with PCOS. Pioglitazone might be a good choice for the patients with PCOS who were intolerant or invalid to metformin for the treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
PCOS (polycystic ovary syndrome) is one of the most common endocrine disorders in women, and it affects about 5–7% women of reproductive age [1] who accounts for 30–60% of anovulatory infertility patients. Its basic characteristics are hyperandrogenism, chronic anovulation and polycystic ovaries. It increases the incidence of endometrial cancer and ovarian cancer. In addition, patients always have the character of insulin resistance, central obesity, impaired glucose, dyslipidemia, cardiovascular risk and subclinical atherosclerosis [2,3,4]. PCOS can increase the risk of type 2 diabetes in 5–10% [5,6,7], and 30–70% of patients with PCOS may be accompanied with obesity [8, 9]. The prevalence of IGT in patients with PCOS in America reached 30–35%, and 5% of these will develop to type 2 diabetes. Adolescent and adult women with PCOS usually need OGTT test of 75 g of glucose, and assessment of waist circumference, BMI, blood lipids, blood pressure and other metabolic factors are also needed. Metformin can ameliorate ovulation and metabolic risk of PCOS and it is currently used as second-line therapy in some PCOS guidelines [10]. Thiazolidinediones (TZDs) are a kind of highly selective synthetic agonists of PPAR-γ (peroxisome proliferation-activated receptor gamma) for the treatment of diabetes. TZDs, the classical insulin sensitizers include troglitazone, rosiglitazone and pioglitazone. The pathogenesis of PCOS may be related to the alteration of PPAR-γ gene, and PPAR-γ seems to play an important role in fertility and metabolism through the effects of its different hypotypes. For example, PPAR-γ1 can specifically regulate ovarian function [11]. Insulin resistance is an important aspect of PCOS and has been observed not only in obese but also in lean women with PCOS and seems to be an intrinsic part of the syndrome [12, 13]; PPAR-γ agonists can decrease androgen synthesis in ovaries by ameliorating peripheral insulin resistance indirectly [14]. Thus, those provide theoretical and practical bases for the treatment of PCOS with TZDs therapy. The treatment of PCOS with TZDs has been investigated in many animals and clinical studies and trials, and most of the trials showed an effective therapeutic result. But it still lacks sufficient evidence-based supports. There are no large-scale clinical trials to verify the efficacy and safety of TZDs drugs for PCOS [15]. So the treatment has not been recognized by public or recommended by all the guidelines. We conducted a meta-analysis of the related literatures to objectively evaluate the clinical effectiveness and safety by comparing pioglitazone with metformin administrated by PCOS patients.
Materials and methods
Literature search
We searched the literatures on Cochrane Library, EMBASE and PubMed with an end date of December 2016. The last searching time was on January 10, 2017. The search terms in full text included the following: “pioglitazone”, “metformin”, “polycystic ovary syndrome” or “PCOS”, and “randomized controlled trial” or “randomized”. Publication time, genre and languages were not limited.
Outcome measures
The main outcome was to compare the therapeutic effect between pioglitazone and metformin prescribed for the patients with PCOS. Therapeutic parameters included menstrual cycle, body mass index (BMI), waist hip ratio (WHR), waist circumference (WC), acne, F-G score for hirsutism, fasting blood sugar (FBS), insulin (INS), homeostasis model of assessment for insulin resistance index (HOMA-IR), total cholesterol (TC), triglyceride (TG), free testosterone (T), dehydroepiandrosterone (DHEA), free androgen index (FAI), luteinizing hormone/follicle-stimulating hormone (LH/FSH), sex hormone binding globulin (SHBG), aspartate amino transferase (AST) and glutamic pyruvic transaminase (ALT).
Selection criteria
Identified studies were included in the meta-analysis if [1] the patients were diagnosed with PCOS definitely and the diagnostic criteria conformed to the Rotterdam criteria of the European Society of Human Reproduction and Embryology (ESHRE) and American Society for Reproductive Medicine (ASRM) in 2003. [2] The randomized controlled study (RCT) was conducted in the literature. [3] Groups included at least pioglitazone therapy group and metformin therapy group, and there was comparison between the two groups. [4] Outcome measures included at least one of the above therapeutic parameters in the study. Studies were excluded if [1] the therapeutic group combined using other agents with pioglitazone or metformin. [2] Test design was not reasonable or lacked of effective control. [3] Review, case report, or animal experiments would be excluded.
Data extraction and statistical analysis
No patient consent or ethical approval was required because analyses were based on previously published studies. The first two authors of this article have collected the literatures according to the criteria; the two authors, respectively, evaluated the quality of the literatures independently and negotiated to confirm [1] randomized method; [2] allocation concealment; [3] if patients, executors of the study, and surveyors were blinded; [4] patients withdrawal, loss to follow-up or dropped out. Different opinions of the first two authors of this article would be judged and concluded by the third author. Then the data of the literatures, including the first author’s name, year of publication, study period, average ages of the patients, therapeutic dose, therapeutic period, clinical effect and so on were extracted.
The software of StataSE 12.0 was used to analyze the data by meta-analysis and to draw forest plot. Inter-study heterogeneity among the trials was assessed using Q test. I 2 > 50% indicated significant heterogeneity and random effects model was used for statistical analysis. I 2 < 50% indicated no significant heterogeneity and fixed effects model was used for statistical analysis. The values of OR and 95% CI were used to describe the effect values of categorical data. Mean ± SD was used to measure the effect values of measurement data. Possible heterogeneity among the studies was investigated by meta-regression. Overall quality of the literatures was evaluated by sensitivity analysis. Publication bias was assessed by Begg’s and Egger’s analysis with Begg’s funnel plot. P < 0.05 was considered statistically significant.
Results
Literature search
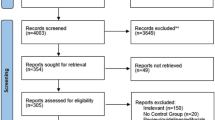
Total 486 related articles were extracted from the three databases and 436 articles were left after removing duplication. Finally, 11 articles meeting the criteria [16,17,18,19,20,21,22,23,24,25,26] were left after the abstract and the full text had been read. Figure 1 shows the search strategy for selection of trials.
Search strategy for selection of trials. From: Lesley et al. [48]
Study characteristics
In total, there were 643 patients, of whom 319 patients were treated with pioglitazone and 324 patients treated with metformin. Table 1 shows the characteristics of the included randomized controlled trials. Detailed data are shown in Tables 2 and 3.
The therapeutic effects of pioglitazone in comparison with metformin
Improvement of the menstrual cycle and ovulation
There were five articles [16, 20, 24,25,26] in which menstrual cycle and ovulation were compared between two groups. Heterogeneity test showed no statistical heterogeneity among the studies. (I 2 = 41.8%, P = 0.143). The fixed effects model was adopted for meta-analysis because of the clinical homogeneity. Results showed that difference of total effective rate was statistically significant between the two groups [OR = 2.31, 95% CI (1.37, 3.91), P < 0.001]. It showed that in pioglitazone treatment group the curative effect of improving menstrual cycle and ovulation was superior to that in metformin treatment group (Fig. 2).
Effect of sex hormones and the clinical manifestations
Only one article compared LH/FSH, hirsutism, acne and hair loss before and after the therapy between the two groups, so the above-mentioned factors were not analyzed.
Measure of free testosterone: Free testosterone of the two groups was measured before and after the therapy in four articles [17, 21, 23, 25]. Heterogeneity test showed no statistical heterogeneity among the studies (I 2 = 0.0%, P = 0.694). The fixed effects model was adopted for meta-analysis because of the clinical homogeneity. Results showed that difference of free testosterone was not significant between the two groups [SMD = 0.04, 95% CI (−0.22, 0.31), P > 0.05]. It showed that there was no difference of free testosterone induced by pioglitazone or metformin (Fig. 3).
Measure of DHEA: DHEA of the two groups was measured before and after the therapy in four articles [17, 23, 25, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 60.6%, P = 0.054). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of DHEA was not significant between the two groups [SMD = −0.12, 95% CI (−0.61, 0.36), P > 0.05]. It showed that there was no difference of DHEA induced by pioglitazone or metformin (Fig. 4).
Measure of FAI: FAI of the two groups was measured before and after the therapy in three articles [18, 19, 21]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 82.3%, P = 0.004). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of FAI was not significant between the two groups [SMD = −0.23, 95% CI (−1.16, 0.7), P > 0.05]. It showed that there was no difference of FAI induced by pioglitazone or metformin (Fig. 5).
Measure of SHB: SHBG of the two groups was measured before and after the therapy in three articles [18, 19, 21]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 96.7%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of SHBG was not significant between the two groups [SMD = 2.28, 95% CI (−0.5, 5.06), P > 0.05]. It showed that there was no difference of SHBG induced by pioglitazone or metformin (Fig. 6).
Measure of F-G score: F-G score of the two groups was measured before and after the therapy in four articles [16, 17, 19, 21]. Heterogeneity test showed no statistical heterogeneity among the studies (I 2 = 0.0%, P = 0.543). The fixed effects model was adopted for meta-analysis because of the clinical homogeneity. Results showed that difference of F-G score was significant between the two groups [SMD = 0.29, 95% CI (0.0, 0.59), P = 0.048]. It showed that in metformin treatment group the improvement of F-G score was superior to that in pioglitazone treatment group (Fig. 7).
Effects on glucose metabolism
Measure of FBS: FBS of the two groups was measured before and after the therapy in seven articles [16, 17, 19, 22, 23, 25, 26]. Heterogeneity test showed no statistical heterogeneity among the studies (I 2 = 47.9%, P = 0.074). The fixed effects model was adopted for meta-analysis because of the clinical homogeneity. Results showed that the difference of FBS was not significant between the two groups [SMD = 0.14, 95% CI (−0.09, 0.38), P > 0.05]. It showed that there was no difference of FBS induced by pioglitazone or metformin (Fig. 8).
Measure of INS: INS of the two groups was measured before and after the therapy in nine articles [16,17,18,19, 21,22,23, 25, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 90.4%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of INS was not significant between the two groups [SMD = −0.69, 95% CI (−1.39, 0.01), P = 0.054]. It showed that there was no difference of INS induced by pioglitazone or metformin (Fig. 9).
Measure of HOMA-IR: HOMA-IR of the two groups was measured before and after the therapy in seven articles [16,17,18, 21,22,23, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 88.2%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of HOMA-IR was not significant between the two groups [SMD = −0.57, 95% CI (−1.28, 0.14), P > 0.05]. It showed that there was no difference of HOMA-IR induced by pioglitazone or metformin (Fig. 10).
Effects on other metabolic indexes
Only one article compared WC, AST and ALT before the therapy with those after the therapy, so the above-mentioned factors were not analyzed.
Measure of BMI: BMI of the two groups was measured before and after the therapy in eight articles [16,17,18,19, 22, 23, 25, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 82.8%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of BMI was significant between the two groups [SMD = 0.83, 95% CI (0.24, 1.41), P = 0.006]. It showed that in pioglitazone treatment group BMI added more than that in metformin treatment group (Fig. 11).
Measure of WHR: WHR of the two groups was measured before and after the therapy in eight articles [16, 17, 20, 23, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 91.6%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of WHR was not significant between the two groups [SMD = 0.58, 95% CI (−0.53, 1.7), P > 0.05]. It showed that there was no difference of WHR induced by pioglitazone or metformin (Fig. 12).
Measure of TC: TC of the two groups was measured before and after the therapy in seven articles [17, 19, 21,22,23, 25, 26]. Heterogeneity test showed significant statistical heterogeneity among the studies (I 2 = 91.7%, P < 0.001). The random effects model was adopted for meta-analysis because of the clinical heterogeneity. Results showed that difference of TC was not significant between the two groups [SMD = −0.49, 95% CI (−1.29, 0.31), P > 0.05]. It showed that there was no difference of TC induced by pioglitazone or metformin (Fig. 13).
Measure of TG: TG of the two groups was measured before and after the therapy in six articles [17, 19, 22, 23, 25, 26]. Heterogeneity test showed no statistical heterogeneity among the studies (I 2 = 43.9%, P = 0.113). The fixed effects model was adopted for meta-analysis because of the clinical homogeneity. Results showed that difference of TG was not significant between the two groups [SMD = −0.01, 95% CI (−0.26, 0.24), P > 0.05]. It showed that there was no difference of TG induced by pioglitazone or metformin (Fig. 14).
Analysis of publication bias
The possible publication bias was analyzed by Begg’s and Egger’s test (Table 4, P > 0.05). In Begg’s funnel plot (Fig. 15), the researches included were distributed relatively symmetrically. The results showed that there was less likely publication bias.
Analysis of meta-regression
Publication year, number of cases, therapeutic doses of pioglitazone and duration of therapy were taken as covariant for meta-regression analysis. It showed that the above factors did not result in the significant heterogeneity of the study (Table 5, P > 0.05).
Sensitivity analysis
To investigate how much a single research affecting overall effect size by sensitivity analysis. Results showed that each of the researches included had no significant influence on the overall effect size, i.e., there might be certain heterogeneity among the researches included, but the heterogeneity did not affect the results significantly. So the results of meta-analysis were quite steady (Fig. 16).
Discussion
Metformin can significantly reduce body mass and fasting insulin levels in patients with PCOS. It can obviously improve insulin sensitivity, hyperandrogenism, menstrual cycle and ovulation. Metformin is relatively safe for the fetus during pregnancy (FDA Pregnancy Category B) [27,28,29]. Thiazolidinediones (TZDs) are the PPAR-γ (peroxisome proliferator-activated receptor gamma) agonists and act by activating PPARs (nuclear receptors) with greatest specificity. Free fatty acids (FFAs) and eicosanoids are the endogenous ligands for the receptors. When activated, the receptor binds to DNA in complex with the retinoid X receptor (RXR), another nuclear receptor, increasing transcription of a number of specific genes and decreasing transcription of others. That regulated a series of specific gene expression including adiponectin, resistin, leptin, and TNF-α. And that would speed up the differentiation of preadipocytes. Mature adipose cells would be more sensitive to insulin and have the effects of anti-inflammatory. TZDs can decrease the insulin resistance, modify the adipocyte differentiation [30], inhibit the VEGF-induced angiogenesis [31], decrease leptin levels (perhaps leading to an increased appetite) and have the effects of anti-inflammatory. TZDs include rosiglitazone, pioglitazone and troglitazone. Troglitazone has been eliminated due to its serious liver toxicity. In 2010, the FDA carried on the strict restrictions of rosiglitazone for possible increased risk of cardiovascular events. Its restriction was relieved in December 2015 because related researches had not confirmed the increased risk. Animal experiments and epidemiological survey found that pioglitazone might lead to increased risk of bladder cancer. In recent years, many clinical studies have shown that pioglitazone treatment is not associated with increased bladder cancer risk [32,33,34,35,36,37,38,39]. In 2017, FDA determined that [40] the public should be informed about the uncertainty in the literature, while retaining the current warning against the use of pioglitazone in patients with active bladder cancer and for careful considerations of risks and benefits in patients with a prior history of bladder cancer because the results of literatures were inconsistent [32, 41, 42]. A possible association with bladder cancer has also largely been refuted in the consensus statement by the American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) on the comprehensive type 2 diabetes management algorithm-2017 executive summary [43]. Pioglitazone was not recommended for PCOS in the clinical practice guideline of AES in 2013 [10]. But TZDs can be used as the second-line choice in the statement of European Society of Endocrinology in 2014 [44]. Our analysis results show that pioglitazone could improve the menstrual cycle and ovulation better than metformin, but significantly increase BMI compared with metformin. Improvement of hirsutism by metformin was superior to that by pioglitazone. Pioglitazone had the similar effects on FBS, INS, HOMA-IR, TG, TC, T, DHEA, FAI, SHBG and WHR as metformin.
The menstrual cycle and ovulation was ameliorated by pioglitazone or metformin in four literatures, and pioglitazone was better than metformin. In nine literatures, pioglitazone obviously elevated BMI but metformin decreased BMI, and this was in accordance with the guideline or consensus. In five literatures, pioglitazone or metformin had no effects on WHR; there was no difference between the two agents and this indirectly showed that pioglitazone elevated BMI not for increasing central obesity. Studies in diabetics suggest that pioglitazone can increase peripheral fat and may reduce visceral fat stores [45]. Pioglitazone can reduce ectopic fat including the liver and muscle, and thus enhance tissue sensitivity to insulin [46, 47]. In some patients, TZDs may increase body weight due to fluid retention [44]. In four literatures, F-G scores of hirsutism were decreased by pioglitazone or metformin, and metformin was a little better than pioglitazone. In seven literatures, FBS, insulin and HOMA-IR were deceased by pioglitazone or metformin, and there were no differences between the two agents. The effects on androgen and lipid were different in the literatures. Many literatures showed that pioglitazone ameliorated androgen and lipid better than metformin, but there was no difference between the two agents from meta-analysis. Based on different dosages and time of treatment, it shows that the pioglitazone therapy for 3 or 6 months seems better than that for 1.5 months, and either 30 or 45 mg of pioglitazone therapy is effective. But 45 mg of pioglitazone therapy seems more BMI gained. Side effects may be mitigated using a moderate dose (e.g., ≤30 mg) of pioglitazone [43]. So 30 mg daily of pioglitazone therapy for 3–6 months may be a better choice for PCOS. There was only descriptive information and few statistical data of side effects in most literatures. Adverse reactions such as headache, rash and myalgia were minor. Side effects were recorded and compared in detail only in one literature of Kashani. Gastrointestinal reaction was more significant in metformin group than in pioglitazone group. Pioglitazone increased appetite more than metformin. The two agents both had no effects on liver function such as AST and ALT.
There are some deficiencies in this study because apparent heterogeneity exists in some data of different literatures. We analyzed the possible causes of the heterogeneity: (1) there are not enough literatures conformed to the diagnostic criteria and inclusion criteria entering our study. The number of patients in each research is from 20 to 50, so there are not very enough cases in the literatures. Multi-center large RCT studies were insufficient and there might be some sampling error. (2) There were no detailed instructions for the race of patients in the researches. Only one study gave clear indication for the race of the Caucasus ethnic groups, and others did not; (3) not all the research methods of the literatures are perfect. Blind method was conducted only in two articles. Concealment and analysis of intention-to-treat (ITT) was conducted only in one article. There were no specific statistics on the data of the patients retreated or loss to follow-up. There was no analysis if the patients retreated for pregnancy might benefit from the medicine which could lead to conception or if that might decrease the efficiency of the agents. All of the above might increase the error of the results. (4) The dosages of both pioglitazone and metformin used in the individual studies are very heterogeneous. Dosages of pioglitazone were 30 and 45 mg per day, and dosages of metformin were 1000, 1500, 1700, and 2550 mg per day. This fact might strongly affect the outcomes of the studies. (5) The duration of the individual studies is likewise very heterogeneous—some were 3 and others 6 months long and one just 1.5 months. This might also affect the outcomes of the studies. The course of treatment was short which was from 6 weeks to 6 months. This might lead to inconsistent results which were dependent on time. (6) No exact statistics were for side effects. Most only described as symptoms such as rash, nausea, and vomiting. Only in one article were there detailed data such as liver function before and after treatment in both groups.
Conclusions
Our study shows that pioglitazone improved menstrual cycle and ovulation of PCOS patients better than metformin. In contrast with pioglitazone, metformin could reduce weight and ameliorate symptoms of hirsutism better. Two agents had similar effects on other metabolic targets. Pioglitazone may be an alternative treatment in insulin-resistant or obese PCOS women who do not tolerate or do not respond to metformin therapy and 30 mg daily of pioglitazone therapy for 3–6 months may be a better choice. In the future, high quality and multi-center study of RCT with sufficient cases are required to confirm the curative effects and side effects of pioglitazone.
References
Carmina E, Lobo RA (1999) Polycystic ovary syndrome (PCOS): arguably the most common endocrinopathy is associated with significant morbidity in women. J Clin Endocrinol Metab 84:1897–1899
Pepene CE, Ilie IR, Marian I, Duncea I (2011) Circulating osteoprotegerin and soluble receptor activator of nuclear factor κB ligand in polycystic ovary syndrome: relationships to insulin resistance and endothelial dysfunction. Eur J Endocrinol 164(1):61–68
Glueck CJ, Morrison JA, Goldenberg N, Wang P (2009) Coronary heart disease risk factors in adult premenopausal white women with polycystic ovary syndrome compared with a healthy female population. Metab Clin Exp 58(5):714–721
Ilie IR, Marian I, Mocan T, Ilie R, Mocan L, Duncea I et al (2012) Ethinylestradiol30μg-drospirenone and metformin: could this combination improve endothelial dysfunction in polycystic ovary syndrome? BMC Endocr Disord 12:9
Ehrmann DA, Barnes RB, Rosenfield RL, Cavaghan MK, Imperial J (1999) Prevalence of impaired glucose tolerance and diabetes in women with polycystic ovary syndrome. Diabetes Care 22(1):141–146
Legro RS, Kunselman AR, Dodson WC, Dunaif A (1999) Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: a prospective, controlled study in 254 affected women. J Clin Endocrinol Metab 84(1):165–169
Palmert MR, Gordon CM, Kartashov AI, Legro RS, Emans SJ, Dunaif A (2002) Screening for abnormal glucose tolerance in adolescents with polycystic ovary syndrome. J Clin Endocrinol Metab 87(3):1017–1023
Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO (2004) The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab 89(6):2745–2749
Ehrmann DA (2005) Polycystic ovary syndrome. N Engl J Med 352(12):1223–1236
Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R, Welt CK (2013) Diagnosis and treatment of polycystic ovary syndrome: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 98:4565–4592 (Online 1945-7197:1-29)
Froment P, Gizard F, Staels B, Dupont J, Monget P (2005) A role of PPARgamma in reproduction? Med Sci 21(5):507–511
Anastasiou OE, Canbay A, Fuhrer D, Reger-Tan S (2017) Metabolic and androgen profile in underweight women with polycystic ovary syndrome. Arch Gynecol Obstet 296:363–371
Doganay M, Ozyer SS, Var T, Tonguc E, Gun Eryilmaz O, Ozer I et al (2015) Associations between adipocyte fatty acid-binding protein and clinical parameters in polycystic ovary syndrome. Arch Gynecol Obstet 291(2):447–450
Schoppee PD, Garmey JC, Veldhuis JD (2002) Putative activation of the peroxisome proliferator-activated receptor gamma impairs androgen and enhances progesterone biosynthesis in primary cultures of porcine theca cells. Biol Reprod 66(1):190–198
Tang T, Lord JM, Norman RJ, Yasmin E, Balen AH (2010) Insulin-sensitising drugs (metformin, rosiglitazone, pioglitazone, d-chiro-inositol) for women with polycystic ovary syndrome, oligo amenorrhoea and subfertility. Cochrane Database Syst Rev 1:CD003053
Ortega-González C, Cardoza L, Coutiño B, Hidalgo R, Arteaga-Troncoso G, Parra A (2005) Insulin sensitizing drugs increase the endogenous dopaminergic tone in obese insulin-resistant women with polycystic ovary syndrome. J Endocrinol 184(1):233–239
Ortega-González C, Luna S, Hernández L, Crespo G, Aguayo P, Arteaga-Troncoso G et al (2005) Responses of serum androgen and insulin resistance to metformin and pioglitazone in obese, insulin-resistant women with polycystic ovary syndrome. J Clin Endocrinol Metab 90(3):1360–1365
Cho LW, Kilpatrick ES, Keevil BG, Coady AM, Atkin SL (2009) Effect of metformin, orlistat and pioglitazone treatment on mean insulin resistance and its biological variability in polycystic ovary syndrome. Clin Endocrinol 70(2):233–237
Naka KK, Kalantaridou SN, Kravariti M, Bechlioulis A, Kazakos N, Calis KA et al (2011) Effect of the insulin sensitizers metformin and pioglitazone on endothelial function in young women with polycystic ovary syndrome: a prospective randomized study. Fertil Steril 95(1):203–209
Navali N, Shokoufe LA, Mallah F, Bastani P, Mashrabi O (2012) Comparing therapeutic effects of metformin and pioglitazone in polycystic ovary syndrome (PCOS). Pak J Med Sci 28(3):390–394
Sangeeta S (2012) Metformin and pioglitazone in polycystic ovarian syndrome: a comparative study. J Obstet Gynecol India 62(5):551–556
Ziaee A, Oveisi S, Abedini A, Hashemipour S, Karimzadeh T, Ghorbani A (2012) Effect of metformin and pioglitazone treatment on cardiovascular risk profile in polycystic ovary syndrome. Acta Med Indones 44(1):16–22
Kashani L, Omidvar T, Farazmand B, Modabbernia A, Ramzanzadeh F, Tehraninejad ES et al (2013) Does pioglitazone improve depression through insulin-sensitization? Results of a randomized double-blind metformin-controlled trial in patients with polycystic ovarian syndrome and comorbid depression. Psychoneuroendocrinology 38(6):767–776
Chaudhry I, Sadaf Un N, Shams Un N (2016) Comparison between pioglitazone and metformin in terms of efficacy in patients with polycystic ovarian syndrome. Pak J Med Health Sci 10(2):574–577
Shahebrahimi K, Jalilian N, Bazgir N, Rezaei M (2016) Comparison clinical and metabolic effects of metformin and pioglitazone in polycystic ovary syndrome. Indian J Endocrinol Metab 20(6):805–809
Sohrevardi SM, Nosouhi F, Khalilzade SH, Kafaie P, Karimi-Zarchi M, Halvaei I et al (2016) Evaluating the effect of insulin sensitizers metformin and pioglitazone alone and in combination on women with polycystic ovary syndrome: an RCT. Int J Reprod BioMed 14(12):743–754
Bridger T, MacDonald S, Baltzer F, Rodd C (2006) Randomized placebo-controlled trial of metformin for adolescents with polycystic ovary syndrome. Arch Pediatr Adolesc Med 160(3):241–246
De Leo V, Musacchio MC, Morgante G, Piomboni P, Petraglia F (2006) Metformin treatment is effective in obese teenage girls with PCOS. Hum Reprod 21(9):2252–2256
Ghazeeri GS, Nassar AH, Younes Z, Awwad JT (2012) Pregnancy outcomes and the effect of metformin treatment in women with polycystic ovary syndrome: an overview. Acta Obstet Gynecol Scand 91(6):658–678
Waki HY, Yamauchi T, Kadowaki T (2010) Regulation of differentiation and hypertrophy of adipocytes and adipokine network by PPARgamma. Nihon Rinsho 68(2):210–216
Panigrahy D, Singer S, Shen LQ et al (2002) PPARγ ligands inhibit primary tumor growth and metastasis by inhibiting angiogenesis. J Clin Invest 110(7):923–932
Lewis JD, Habel LA, Quesenberry CP, Strom BL, Peng T, Hedderson MM et al (2015) Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 314(3):265–277
Levin D, Bell S, Sund R, Hartikainen SA, Tuomilehto J, Pukkala E et al (2015) Pioglitazone and bladder cancer risk: a multipopulation pooled, cumulative exposure analysis. Diabetologia 58(3):493–504
Lee MY, Hsiao PJ, Yang YH, Lin KD, Shin SJ (2014) The association of pioglitazone and urinary tract disease in type 2 diabetic Taiwanese: bladder cancer and chronic kidney disease. PLoS One 9(1):e85479
Balaji V, Seshiah V, Ashtalakshmi G, Ramanan SG, Janarthinakani M (2014) A retrospective study on finding correlation of pioglitazone and incidences of bladder cancer in the Indian population. Indian J Endocrinol Metab 18(3):425–427
Wei L, MacDonald TM, Mackenzie IS (2013) Pioglitazone and bladder cancer: a propensity score matched cohort study. Br J Clin Pharmacol 75(1):254–259
Vallarino C, Perez A, Fusco G, Liang H, Bron M, Manne S et al (2013) Comparing pioglitazone to insulin with respect to cancer, cardiovascular and bone fracture endpoints, using propensity score weights. Clin Drug Investig 33(9):621–631
Tseng CH (2012) Pioglitazone and bladder cancer: a population-based study of Taiwanese. Diabetes Care 35(2):278–280
Song SO, Kim KJ, Lee BW (2012) Response: the risk of bladder cancer in korean diabetic subjects treated with pioglitazone (diabetes metab j 2012;36:371–8). Diabetes Metab J 36(6):462–463
Hampp C, Pippins J (2017) Pioglitazone and bladder cancer: FDA’s assessment. Pharmacoepidemiol Drug Saf 26:117–118
Erdmann E, Harding S, Lam H, Perez A (2016) Ten-year observational follow-up of PROactive: a randomized cardiovascular outcomes trial evaluating pioglitazone in type 2 diabetes. Diabetes Obes Metab 18(3):266–273
Tuccori M, Filion KB, Yin H, Yu OH, Platt RW, Azoulay L (2016) Pioglitazone use and risk of bladder cancer: population based cohort study. BMJ 352:i1541
Garber AJ, Abrahamson MJ, Barzilay JI, Blonde L, Bloomgarden ZT, Bush MA et al (2017) Consensus statement by the American Association Of Clinical Endocrinologists and American College of endocrinology on the comprehensive type 2 diabetes management algorithm—2017 executive summary. Endocr Pract 23(2):207–238
Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R et al (2013) Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 98(12):4565–4592
Rasouli N, Raue U, Miles LM, Lu T, Di Gregorio GB, Elbein SC et al (2005) Pioglitazone improves insulin sensitivity through reduction in muscle lipid and redistribution of lipid into adipose tissue. Am J Physiol Endocrinol Metab 288(5):E930–E934
Gupta AK, Bray GA, Greenway FL, Martin CK, Johnson WD, Smith SR (2010) Pioglitazone, but not metformin, reduces liver fat in Type-2 diabetes mellitus independent of weight changes. J Diabetes Complicat 24(5):289–296
Bajaj M, Baig R, Suraamornkul S, Hardies LJ, Coletta DK, Cline GW et al (2010) Effects of pioglitazone on intramyocellular fat metabolism in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab 95(4):1916–1923
Stewart LA, Clarke M, Rovers M et al (2015) Preferred Reporting Items for a systematic review and meta-analysis of individual participant data: The PRISMA-IPD Statement. JAMA 313(16):1657–1665. doi:10.1001/jama.2015.3656
Acknowledgements
The authors would like to thank Wang Chaoqun and Guo Qinjun for the advice on the statistics of StataSE.
Author information
Authors and Affiliations
Contributions
XY, the first author: project development, data collection, data analysis, and manuscript writing. WY, co-first author: data collection. HQ, corresponding author: manuscript editing
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest.
Ethical approval and informed consent
No patient consent or ethical approval was required because analyses were based on previous published studies.
Additional information
Yifeng Xu and Yanxiang Wu are co-first authors.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Xu, Y., Wu, Y. & Huang, Q. Comparison of the effect between pioglitazone and metformin in treating patients with PCOS:a meta-analysis. Arch Gynecol Obstet 296, 661–677 (2017). https://doi.org/10.1007/s00404-017-4480-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-017-4480-z