Abstract
There is now robust evidence that the cerebellum—apart from its well-established role in motor control—is crucially involved in a wide spectrum of cognitive and affective functions. Clinical and neuropsychological studies together with evidence from anatomical studies and advanced neuroimaging have yielded significant insights into the specific features and clinical relevance of cerebellar involvement in normal cognition and mood.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The cerebellar cortex shows a complex and remarkably homogeneous cellular organization that is repeated across its entire extension. On the other hand, each part of the cerebellar cortex has specific feedforward and feedback connections to the cerebral cortex that go far beyond the long known connections to motor and premotor cortical areas. Indeed, there are parallel connections to paralimbic and association cortices. Based on these observations, it is thought that the cerebellum performs a universal transform that is applied to different domains, i.e., motor, cognitive and affective. According to current view, the cerebellum—based on its ability to detect errors in performing motor and mental processes—is building internal models, which are crucial for optimizing these processes and the basis for cerebellar learning across contexts [1, 2]. Through automation and adaptation, processes become increasingly stereotyped and efficient, and demand less attention [3]. Motor learning directly exemplifies the close link between the motor and cognitive domain of the cerebellum.
The various cerebellar functions are accordingly based on the specific anatomical connections of parts of the cerebellar cortex rather than on specific neuronal computations of each part. Consequently, a cerebellar functional topography can be defined that distinguishes motor, vestibular, cognitive, and limbic cerebellar regions. Whereas dysfunction of motor and vestibular parts are associated with ataxia, dysfunction of cognitive and limbic parts are associated with the cerebellar cognitive affective syndrome (CCAS) that was first described in the early 90s by Jeremy Schmahmann [4].
Topography of cognitive functions in the cerebellum
The majority of the cerebellar cortex is not primarily involved in motor planning or execution. The motor-related areas of the cerebellum are relatively small and circumscribed in their extent and preserved across subjects [5,6,7]. According to the nomenclature introduced by Larsell (8), 10 cerebellar lobules are distinguished. Lobules I–V correspond to the anterior lobe, lobules VI–IX to the posterior lobe, and lobule X to the flocculonodular lobe. Lobule VII can be further subdivided in lobules Crus I, Crus II and VIIb. Crus I and Crus II together form lobule VIIa. Motor-related areas are located ipsilateral to the moved limb in lobules I–V with some extension into lobule VI and lobule VIII. In recent years, two approaches have largely been chosen to study the functional topography of the human cerebellum beyond sensorimotor processing: resting-state functional MRI (fMRI) for the assessment of cerebellar representation of cerebral networks and task-related functional MRI to directly identify the subsequent task-related areas.
Guell et al. [9] analyzed task fMRI data of the Human Connectome Project. Data from almost 800 human participants were included from a range of motor, working memory, language, social, and emotional tasks in the scanner. Language tasks activated primarily the right posterolateral cerebellar hemisphere, and spatial tasks primarily the left posterolateral cerebellar hemisphere. In the literature, however, lateralization of visuospatial functions is less clear within the cerebellum than language functions. Working memory, executive, social and emotional tasks were accompanied by activation of the posterolateral cerebellar hemispheres bilaterally [9]. In emotional tasks, activation was also found in the vermis. The vermis has known anatomical connections with the limbic system and has been referred to as “limbic cerebellum” [10]. Activations related to cognitive tasks were observed predominantly in lobule VII, with some extension into lobule VI. The most detailed map of motor and non-motor functions within the human cerebellar cortex to date has been published by King et al. in 2019. Their findings confirmed earlier observations that functional representations in the cerebellar cortex do not follow the boundaries of the cerebellar lobules. Furthermore, their findings indicate that the cerebellar cortex is organized in distinct functional subregions. King et al.’s findings are at variance with a recent resting-state fMRI analysis by Guell et al. [12] which suggests that two smooth gradients in the cerebellar cortex exist. According to this analysis, the main gradient extends from motor areas via non-motor task-focused (i.e., attentional/executive network-related) areas to non-motor task-unfocused (i.e., default-mode network related) areas. A second gradient extends from task-focused to task-unfocused non-motor areas [12]. Detailed maps of the functional topography of these non-motor functions based on task-based and resting-state fMRI data are publicly available and are helpful guides in neuroimaging and cerebellar lesion studies [11, 13].
Furthermore, resting-state data published by Buckner et al. [14] demonstrated a triple representation of cognitive functions in the cerebellar cortex, with two inverted representations in the posterolateral cerebellar hemispheres (attentual/executive network-related areas–default-mode network–default-mode network–attentual/executive network), and a third representation in lobules IX and X. The later overlaps with the well-known cerebellar–vestibular system. Findings were confirmed by Guell et al. [9] using task-related and resting-state fMRI data. The authors describe a triple non-motor representation in lobules VI/Crus I, Crus II/VIIB, and IX/X.
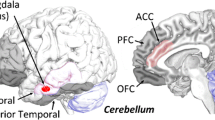
The concept of a non-motor role of the cerebellum is corroborated by cerebellar involvement in several psychiatric disorders, including autism [15, 16], schizophrenia [17, 18] and attention-deficit hyperactivity disorder [16, 19], as well as bipolar, major depressive and anxiety disorders [20]. The variety of psychiatric symptoms may be explained by projections of different cerebellar areas to specific extracerebellar areas.
Cognitive and affective abnormalities in cerebellar patients
The cerebellum optimizes performance according to the context by modulating speed, capacity, appropriateness and consistency of cognitive and affective processes in a similar way, as it modulates force, rate, accuracy and rhythm of movements [21]. CCAS describes the salient features of cognitive and affective abnormalities in cerebellar patients [4, 22, 23]. Typical deficits can be grouped into four domains: deficits of executive functions, visuospatial cognition, linguistic functions and personality changes.
Cerebellar lesions in the posterior lobe of the cerebellum lead to deficits in executive functions that are similar to those seen with prefrontal lesions and include deficits in planning, sequencing, verbal fluency, working memory, abstract reasoning, problem solving strategies, set-shifting and an impaired ability to multi-task and organizing activities [4, 24]. Furthermore, such lesions can impair visuospatial and language functions. The latter may include agrammatism, dysprosodia, anomia, impaired syntax and deficits in verbal fluency [4, 24]. When damage affects the vermal and paravermal regions, patients show inappropriate behavior, mood disturbances including disinhibitions as well as regressive or compulsive behavior. With regard to neurobehavioral profiles, the following domains have been described: disorders of attentional and emotional control, deficits in social behavior as well as autism and psychosis spectrum disorders [21]. In the last few years, all aspects of the CCAS have been replicated in various populations of cerebellar patients in adults and children [24, 25].
In children, the first evidence of cognitive and affective abnormalities was derived from children after surgical resection of a posterior fossa tumors. Posterior fossa syndrome (PFS) is characterized by a reduction of speech or transient mutism, ataxia, hypotonia, executive dysfunction and affective and behavioral abnormalities [26]. Recovery may be prolonged and incomplete, showing residual coordination deficits, dysfluent and slowed speech as well as low social and educational achievements [27,28,29].
Early data about CCAS in adults mainly came from patients with focal injuries such as cerebellar strokes [4]. Typical motor abnormalities were shown in patients with lesions in the anterior lobe. When lesions spared the anterior lobe but affected lobules VI–X of the posterior lobe, patients showed minor or no motor impairment, but cognitive and affective abnormalities [30]. In detail, lesions leading to language deficits (e.g., right Crus I and II extending through lobule IX) could be distinguished from lesions resulting in visuospatial (bilateral Crus I, Crus II, and right lobule VIII) and executive deficits (lobules VII–VIII) [31]. Main predictors of recovery after cerebellar strokes are the localization, the cause and the extent of the lesion. Recovery from cerebellar stroke is better in ischemic stroke than hemorrhage, in strokes affecting the posterior and anterior inferior arteries than the superior cerebellar artery and in strokes leaving the cerebellar nuclei intact [32,33,34]. Additional extracerebellar involvement may complicate the functional recovery. Recovery from lesions of other etiology, e.g., multiple sclerosis seems to be worse [35].
The same pattern of deficits has been replicated in degenerative ataxias. Even before detailed genetic testing was available, impaired executive function was shown to be present in patients with dominantly inherited spinocerebellar ataxias. In a subgroup additional mild generalized cognitive impairment was present [36]. Since most subtypes of SCAs and other degenerative ataxias have significant extracerebellar pathology, studies of SCA6, are of great interest in furthering our understanding of cerebellar contribution to cognition because in this disorder the patients have an almost exclusive cerebellar pathology. Indeed, executive deficits involving inhibition of response and verbal reasoning, cognitive flexibility and abstraction have all been found in SCA6 [37, 38], whereas deficits seem to be more diverse and pronounced in multisystemic SCAs, such as SCA2 and SCA3 [37, 39]. Cognitive decline in SCA patients especially memory and learning abilities have been shown to deteriorate over time independently of motor functions or depressive symptoms [40].
Clinical relevance and perspectives
Cerebellar involvement in cognition has long been a research topic but is gaining increasing clinical attention. Consideration of the CCAS in clinical routine is greatly facilitated by the recent availability of a validated bedside test that assesses the presence and severity of CCAS [23]. Thus, conventional bedside tests that were originally designed to measure cognitive and affective deficits in patients with dementia and depression are now dispensable. The CCAS scale allows screening of patients with cerebellar disease in a short time. Moreover, longitudinal assessment of cognitive and affective deficits in these patients is possible. One needs to be aware, however, that reference values which take age and education into account, are currently missing. The reference values published [23] may not apply for elderly patients [41].
Detailed knowledge of the presence, severity, and individual course of CCAS in patients with cerebellar disease is crucial because the presence of a CCAS may have an impact on the daily life of patients and their families due to cognitive and neuropsychiatric abnormalities, including inappropriate behavior or impulsive actions. The awareness of these symptoms provides the opportunity not only for better counselling, but also for active therapeutic intervention by cognitive rehabilitation, psychological treatment and in some cases pharmacological therapy of neuropsychiatric symptoms. In addition, the specific cognitive problems of cerebellar patients need to be taken into account by rehabilitation specialists. Rehabilitation programs for cerebellar patients that currently focus on coordination training and speech therapy need to be complemented by training programs that address the accompanying cognitive and affective symptoms. Such extended programs need to focus not only on executive and visuospatial functions and language, but also on psychological well-being and social behavior.
The cerebellum is increasingly considered as target for invasive and non-invasive neurostimulation, such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS). Current efforts are mainly focused on improvement of motor coordination in patients with cerebellar disease. However, it is conceivable that non-invasive stimulation could also be effective for improving cognitive function in these patients [42, 43]. However, due the highly convoluted nature of the cerebellar cortex, effects of non-invasive cerebellar brain stimulation are hard to predict, and robustness and replicability of previous findings will need to be seen before any recommendations on these types of therapy can be made [44].
Conversely, the understanding of the role of the cerebellum in cognition and affect could provide clues for the treatment of mental illnesses like autism-spectrum disorders, affective disorders or psychosis spectrum disorders as well as Alzheimer’s disease and frontotemporal dementia.
References
Schmahmann JD (1998) Dysmetria of thought: clinical consequences of cerebellar dysfunction on cognition and affect. Trends Cogn Sci 2:362–371
Ito M (2008) Control of mental activities by internal models in the cerebellum. Nat Rev Neurosc 9:304–313
Koziol LF, Budding D, Andreasen N, D’Arrigo S, Bulgheroni S, Imamizu H, Ito M, Manto M, Marvel C, Parker K, Pezzulo G, Ramnani N, Riva D, Schmahmann J, Vandervert L, Yamazaki T (2014) Consensus paper: the cerebellum’s role in movement and cognition. Cerebellum 13(1):151–177
Schmahmann JD, Sherman JC (1998) The cerebellar cognitive affective syndrome. Brain 121:561–579
Diedrichsen J, Zotow E (2015) Surface-based display of volume-averaged cerebellar imaging data. PLoS ONE 10(7):e0133402
Marek S, Siegel JS, Gordon EM, Raut RV, Gratton C, Newbold DJ et al (2018) Spatial and temporal organization of the individual human cerebellum. Neuron 100(4):977-93e7
Stoodley CJ, Schmahmann JD (2018) Functional topography of the human cerebellum. Handb Clin Neurol 154:59–70
Larsell O (1958) Lobules of the mammalian and human cerebellum. Anat Rec 130:329–330
Guell X, Gabrieli JDE, Schmahmann JD (2018) Triple representation of language, working memory, social and emotion processing in the cerebellum: convergent evidence from task and seed-based resting-state fMRI analyses in a single large cohort. Neuroimage 15(172):437–449
Blatt G.J., Oblak A.L., Schmahmann J.D. (2013) Cerebellar Connections with Limbic Circuits: Anatomy and Functional Implications. In: Manto M., Schmahmann J.D., Rossi F., Gruol D.L., Koibuchi N. (eds) Handbook of the Cerebellum and Cerebellar Disorders
King M, Hernandez-Castillo CR, Poldrack RA, Ivry RB, Diedrichsen J (2019) Functional boundaries in the human cerebellum revealed by a multi-domain task battery. Nat Neurosci 22(8):1371–1378
Guell X, Schmahmann JD, Gabrieli J, Ghosh SS (2018) Functional gradients of the cerebellum. Elife 14(7):e36652
Guell X, Schmahmann J (2020) Cerebellar functional anatomy: a didactic summary based on human fMRI evidence. Cerebellum 19(1):1–5
Buckner RL, Krienen FM, Castellanos A, Diaz JC, Yeo BT (2011) The organization of the human cerebellum estimated by intrinsic functional connectivity. J Neurophysiol 106(5):2322–2345
D’Mello AM, Crocetti D, Mostofsky SH, Stoodley CJ (2015) Cerebellar gray matter and lobular volumes correlate with core autism symptoms. Neuroimage Clin 7:631–639
Marko MK, Crocetti D, Hulst T, Donchin O, Shadmehr R, Mostofsky SH (2015) Behavioural and neural basis of anomalous motor learning in children with autism. Brain 138(Pt 3):784–797
Nopoulos PC, Ceilley JW, Gailis EA, Andreasen NC (1999) An MRI study of cerebellar vermis morphology in patients with schizophrenia: evidence in support of the cognitive dysmetria concept. Biol Psychiatry 46(5):703–711
Chen YL, Tu PC, Lee YC, Chen YS, Li CT, Su TP (2013) Resting-state fMRI mapping of cerebellar functional dysconnections involving multiple large-scale networks in patients with schizophrenia. Schizophr Res 149(1–3):26–34
Tomasi D, Volkow ND (2012) Abnormal functional connectivity in children with attention-deficit/hyperactivity disorder. Biol Psychiatry 71(5):443–450
Phillips JR, Hewedi DH, Eissa AM, Moustafa AA (2015) The cerebellum and psychiatric disorders. Front Public Health 5(3):66
Schmahmann JD, Weilburg JB, Sherman JC (2007) The neuropsychiatry of the cerebellum—insights from the clinic. Cerebellum 6:254–267
Ahmadian N, van Baarsen K, van Zandvoort M, Robe PA (2019) The Cerebellar Cognitive Affective Syndrome—a Meta-analysis. Cerebellum 18(5):941–950
Hoche F, Xavier G, Mark GV, Janet CS, Jeremy DS (2018) The cerebellar cognitive affective/Schmahmann syndrome scale. Brain 141(1):248–270
Argyropoulos GPD, van Dun K, Adamaszek M, Leggio M, Manto M, Masciullo M, Molinari M, Stoodley CJ, Van Overwalle F, Ivry RB, Schmahmann JD (2020) The cerebellar cognitive affective/schmahmann syndrome: a task force paper. Cerebellum 19(1):102–125
Levisohn L, Cronin-Golomb A, Schmahmann JD (2000) Neuropsychological consequences of cerebellar tumor resection in children: cerebellar cognitive affective syndrome in a pediatric population. Brain 123:1041–1050
Daly DD, Love JG (1958) Akinetic mutism. Neurology 8(3):238–242
Huber JF, Bradley K, Spiegler BJ, Dennis M (2006) Long-term effects of transient cerebellar mutism after cerebellar astrocytoma or medulloblastoma tumor resection in childhood. Childs Nerv Syst 22(2):132–138
Huber JF, Bradley K, Spiegler B, Dennis M (2007) Long-term neuromotor speech deficits in survivors of childhood posterior fossa tumors: effects of tumor type, radiation, age at diagnosis, and survival years. J Child Neurol 22(7):848–854
Kieffer V, Chevignard MP, Dellatolas G, Puget S, Dhermain F, Grill J, Valteau-Couanet D, Dufour C (2019) Intellectual, educational, and situation-based social outcome in adult survivors of childhood medulloblastoma. Dev Neurorehabil 22(1):19–26
Schmahmann JD, Macmore J, Vangel M (2009) Cerebellar stroke without motor deficit: clinical evidence for motor and non-motor domains within the human cerebellum. Neuroscience 162(3):852–861
Stoodley CJ, MacMore JP, Makris N, Sherman JC, Schmahmann JD (2016) Location of lesion determines motor vs. cognitive consequences in patients with cerebellar stroke. Neuroimage Clin. 12:765–775
Kelly PJ, Stein J, Shafqat S (2001) Functional recovery after rehabilitation for cerebellar stroke. Stroke 32:530–534
Tohgi H, Takahashi S, Chiba K, Hirata Y (1993) Cerebellar infarction. Clinical and neuroimaging analysis in 293 patients. The Tohoku Cerebellar Infarction Study Group. Stroke 24:1697–1701
Schoch B, Dimitrova A, Gizewski ER, Timmann D (2006) Functional localization in the human cerebellum based on voxelwise statistical analysis: a study of 90 patients. Neuroimage 30(1):36–51
Mills RJ, Yap L, Young CA (2007) Treatment for ataxia in multiple sclerosis. Cochrane Database Syst Rev 24(1):CD005029
Kish SJ, el-Awar M, Stuss D, Nobrega J, Currier R, Aita JF, Schut L, Zoghbi HY, Freedman M (1994) Neuropsychological test performance in patients with dominantly inherited spinocerebellar ataxia: relationship to ataxia severity. Neurology 44(9):1738–1746
Klinke I, Minnerop M, Schmitz-Hübsch T, Hendriks M, Klockgether T, Wüllner U, Helmstaedter C (2010) Neuropsychological features of patients with spinocerebellar ataxia (SCA) types 1, 2, 3, and 6. Cerebellum 9(3):433–442
Cooper FE, Grube M, Elsegood KJ, Welch JL, Kelly TP, Chinnery PF, Griffiths TD (2010) The contribution of the cerebellum to cognition in Spinocerebellar Ataxia Type 6. Behav Neurol 23(1–2):3–15
Bürk K, Globas C, Bösch S, Klockgether T, Zühlke C, Daum I, Dichgans J (2003) Cognitive deficits in spinocerebellar ataxia type 1, 2, and 3. J Neurol 250(2):207–211
Roeske S, Filla I, Heim S, Amunts K, Helmstaedter C, Wüllner U, Wagner M, Klockgether T, Minnerop M (2013) Progressive cognitive dysfunction in spinocerebellar ataxia type 3. Mov Disord 28(10):1435–1438
Thieme A, Röske S, Faber J, Sulzer P, Minnerop M, Elben S, Reetz K, Dogan I, Barkhoff M, Konczak J, Wondzinski E, Siebler M, Hetze S, Müller O, Sure U, Klockgether T, Synofzik M, Timmann D (2020) Reference values for the Cerebellar Cognitive Affective Syndrome Scale: age and education matter. Brain 27:417
Farzan F, Wu Y, Manor B, Anastasio EM, Lough M, Novak V, Greenstein PE, Pascual-Leone A (2013) Cerebellar TMS in treatment of a patient with cerebellar ataxia: evidence from clinical, biomechanics and neurophysiological assessments. Cerebellum 12(5):707–712
Block HJ, Celnik P (2012) Can cerebellar transcranial direct current stimulation become a valuable neurorehabilitation intervention? Expert Rev Neurother 12(11):1275–1277
Miterko LN, Baker KB, Beckinghausen J, Bradnam LV, Cheng MY, Cooperrider J, DeLong MR, Gornati SV, Hallett M, Heck DH, Hoebeek FE, Kouzani AZ, Kuo SH, Louis ED, Machado A, Manto M, McCambridge AB, Nitsche MA, Taib NOB, Popa T, Tanaka M, Timmann D, Steinberg GK, Wang EH, Wichmann T, Xie T, Sillitoe RV (2019) Consensus paper: experimental neurostimulation of the cerebellum. Cerebellum 18(6):1064–1097
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jacobi, H., Faber, J., Timmann, D. et al. Update cerebellum and cognition. J Neurol 268, 3921–3925 (2021). https://doi.org/10.1007/s00415-021-10486-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10486-w