Abstract
The present study provides new data concerning the morphology of the male genitalia of Aphididae and unifies their nomenclature. The structure of the male genitalia of 31 species from 26 genera of Aphididae was studied with light and scanning electron microscopy. In the studied species, the genitalia of males consist of a phallus composed of the sclerotized basal part with its articulation and a membranous apical part—an aedeagus. Laterally of the phallus, there is a pair of setose parameres. The shape of the aedeagus, the shape and length of the sclerotized basal part and its articulation as well as the variability of parameres in their form and the number of setae are recognized as important systematic signs of the genitalia. These characters are considered in conjunction with the phylogenetic relationships among the studied taxa.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among Hemiptera, Aphidoidea have a special status because of their high polymorphism and distinctive mode of reproduction—cyclical parthenogenesis. In the complete life cycle (holocycle) of most aphids, the parthenogenetic phase ends with the appearance of sexuales—oviparous females and males. In most species, the sexual forms appear in autumn; rarely earlier, at the beginning of the summer. Daylength, temperature and nutrition are the most important factors, which influence the production of sexuales (Kawada 1987).
In comparison with females, aphid males are slender and characterized by numerous rhinaria on the antennal segments III and IV, shorter cauda, visible sclerotization on the abdomen and sclerotized genital structures. Males, winged or wingless, are also less numerous and smaller than females. If they are less than 50% of female size, they are considered to be dwarfs (Vollrath 1998).
The structure of the male internal reproductive system has been studied in about 80 species so far (Vitlaczil 1882; Blochman 1887; Cholodkovsky 1900; Wojciechowski 1977; Klimaszewski et al. 1973; Głowacka et al. 1974a, b; Bochen et al. 1975; Polaszek 1987a, b in lit.; Wieczorek and Wojciechowski 2004; Wieczorek 2006, 2008; Wieczorek and Świątek 2008, 2009), and these studies have made an important contribution to unravelling the phylogeny of Aphididae. However, in contrast to others groups of Hemiptera, the male genitalia of Aphididae have only been used marginally in their taxonomy and classification (Essig and Abernathy 1952; Iglisch and Sobhani 1972; Sobhani and Iglisch 1972) and have never been documented in a comparative study. Only Polaszek (1987b in lit.) reviewed these structures and revealed a number of characters which are of potential use in the phylogeny and higher classification of Aphidoidea.
The male genitalia of aphids are relatively large and discernible under a hand lens or even with the naked eye and are composed of the median intromittent organ (the phallus) and 2 pairs of sclerotized appendices located laterally of the phallus. Nevertheless, the nomenclature and drawings of these appendices are a source of confusion (see Table 1). In general description of the male genitalia of Aphidoidea Ossiannilsson et al. (1970) recognized claspers or opercula lateral to the penis, and the sclerotized basal part of the penis consists of a pair of valves, whereas Matsuda (1976) mentioned the tubular penis and more-or-less reduced parameres. Heie (1980) described only a single pair of structures that he termed parameres or claspers, with the aedeagus having a sclerotized basal part; while according to Miyazaki (1987), both of the pairs of appendices are called valves, claspers or parameres. Yang and Chang (2000) comprehensively investigated the external male genitalia in Hemiptera. According to these authors, the male genitalia of aphids [based on the study of a single species, Periphyllus koelreuteriae (Takahashi, 1919)] consist of the aedeagus, phallobasal plates with distinct cone-shaped projection—a connective directly connected with genital styles. The phallobase and the connective form the upper half-circle-shaped periandrium, and this structure is hypothetically considered to be a primary characteristic among hemipterans. Grimaldi and Engel (2005) mentioned that Aphidomorpha have a reduction in or even a loss of male genitalia. Generally, these structures were believed to be similar across species and not useful taxonomically.
Because numerous terms have been applied to the various components of the external male genitalia of Aphididae, our study firstly standardizes the terminology. We used light (LM) and scanning electron microscopy (SEM) to show the detailed structure of the male genitalia, i.e., the phallus composed of the sclerotized basal part with its articulations (proximal and distal arm), the membranous apical part—aedeagus and parameres (see Table 1).
In the present paper, the general morphologies of these structures are described and compared in those representatives of the Aphididae in which the genitalia are not modified. This paper is part of a larger study into the phylogeny of the Aphididae will provide new data concerning the morphology of the male genitalia in selected species of Aphididae (subfamilies: Aiceoninae, Anoeciinae, Eriosomatinae, Hormaphidinae, Greenideinae, Lachninae, Mindarinae and Thelaxinae), Adelgidae and Phylloxeridae—i.e. representatives of aphids with strongly modified structures of the male genitalia.
Materials and methods
Taxon sampling
The structure of the male genitalia of 31 species belonging to the Drepanosiphinae, Chaitophorinae, Calaphidinae, Phyllaphidinae, Saltusaphidinae, Lizeriinae, Spicaphidinae, Tamalinae, Parachaitophorinae, Phleomyzinae and Aphidinae was studied using light (LM) and scanning electron microscopy (SEM). The adult males of 18 studied species were collected in Poland from 1999 to 2009 (see Table 2). Samples of Drepanosiphum oregonensis Granovsky, 1939; Chaetosiphella stipae subsp. setosa Wieczorek, 2008; Mexicallis spinifer Remaudiere, 1982; Diphyllaphis mordvilkoi (Aizenberg, 1932); Lizerius ocoteae E.E. Blanchard, 1923; Neuquenaphis edwardsi (Laing, 1927); Tamalia sp. (accession number, MNHN (EH) 16392-163930); Parachaitophorus yamashitai Sorin, 1979; Phleomyzus passerini (Signoret, 1875); Cavariella saxifragae Remaudiere, 1959; and Cavariella theobaldi (Gillette & Bragg, 1918), were borrowed from the Muséum national d’Histoire naturelle, Paris, France; samples of Periphyllus singeri (Börner, 1952), were borrowed from the Natural History Museum of Denmark, Copenhagen, Denmark, and samples of Subsaltusaphis flava Hille Ris Lambers, 1939, were borrowed from the Department of Zoology, University of Silesia, Katowice, Poland. Because of the rarity of males, sample sizes consisted of 10 studied individuals for each species, with exception of M. spinifer, L. ocoteae and P. passerini which were studied from 3-4 specimens only. The genitalia of 31 species were studied from aphid material preserved in alcohol and slide-mounted specimens (LM); 9 species were also observed using SEM techniques; nymphs and adult males of Appendiseta robiniae (Gillette, 1907) were prepared for semi-thin sectioning. Collection data and the microscopic techniques used are summarized in Table 2.
Light and electron microscopy
Alcohol-preserved specimens and slides were examined and photographed using the light microscope Nikon Eclipse 600. Drawings were made with a camera lucida. A magnified view is provided for each of the photographs and drawings. Additionally, the nymphs and adult specimens of Appendiseta robiniae were fixed for 2 h with 2.5% glutaraldehyde in a 0.1 M phosphate buffer (pH 7.4), then washed 5 times in a phosphate buffer, postfixed for 2 h in 1% OsO4 in the same buffer, dehydrated in a graded series of ethanol replaced by acetone and then embedded in an Epoxy Embedding Medium Kit (Sigma). Semi-thin sections (0.8 μm thick) were cut on a Leica Ultracut UCT ultramicrotome, stained with methylene blue and examined under an Olympus BX60 microscope equipped with a DP12 digital camera and AnalySIS 3.2 (Soft Imaging System) software.
The procedure for preparing samples for SEM was as described earlier (Płachno and Świątek 2009, 2010). Briefly, whole specimens were fixed with 2.5% glutaraldehyde in a 0.1 M phosphate buffer (pH 7.4) for several days or fixed in 70% ethanol. Later, the samples dehydrated in ethanol as well as acetone series were critical point dried in liquid CO2 and coated with gold using a JEOL-JFC 1100E sputter coater. The specimens were viewed in a HITACHI S-4700 microscope (Scanning Microscopy Laboratory of Biological and Geological Sciences, Jagiellonian University, Cracow) at 20 kV.
Results
Morphology
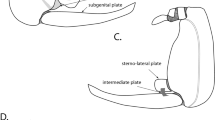
In adult aphids, the abdomen consists of visible segments I-VIII (tergites and sternites) (Fig. 1a). Tergite IX is reduced. The phallus is situated ventrally on the abdominal sternite IX and is composed of a sclerotized basal part (visible in slide-mounted specimens) and a membranous apical part—aedeagus (usually not visible in slide-mounted specimens). The basal part is composed of a pair of various-shaped, partially sclerotized lobes fortified by furcate, strongly sclerotized appendages which consist of a short proximal and a long distal arm. The lobes cover the genital opening, while the aedeagus is withdrawn, during copulation take part in everting the aedeagus and maintaining it in position. Laterally of the phallus, there is a pair of setose parameres (visible in slide-mounted specimens). The cauda (a various-shaped tail-like structure at the end of the abdomen prevents the sticky excrements from flowing into the body) is abdominal tergite X. The anal plate is abdominal sternite X (Fig. 1a–f). In nymphs, the male external genitalia are hidden inside the abdomen and after the last moulting, they are everted (Fig. 1g).
External genitalia of males Cavariella saxifragae laterodorsal view of abdominal segments I–VIII (a), lateroventral view (b). Chaitophorus populeti, basal part of phallus (bp) with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, dorsal view (c), lateral view (d). Appendiseta robiniae, adult, tip of abdomen (e, f). Nymph, tip of abdomen (g). a, b scanning electron microscopy; c, d drawings according to light microscopical preparations; e–g semi-thin sections through abdomen, methylene blue staining. a aedeagus, ag accessory glands, ap anal plate, bp basal part of phallus, c cauda, g gut, p parameres, ph phallus
The general morphologies of these structures of selected species of Aphididae were described, and individual species were then studied systematically (the classification of Aphidoidea after Nieto Nafria et al. 1998 with the exception of Pterocomma populeum (Kaltenbach, 1843) which, according to a molecular study (von Dohlen et al. 2006), now belongs to the tribe Macrosiphini, subfamily Aphidinae).
Drepanosiphinae
Drepanosiphum platanoidis (Schrank, 1801) (male winged)—parameres dusky, large, subtriangular with numerous, rather short setae on the whole surface (Fig. 2a). Basal part of phallus is rather long, finger-like and slightly curved backwards in distal part with few setae. The scanning electron micrographs show a few circular pits distributed mostly in its medial part. Sclerotized arms with distal part rather long and thin, and proximal part shorter and wider. Aedeagus long, inverted question mark-shaped (Fig. 2b).
External genitalia of males. Drepanosiphum platanoidis, ventral view (a), lateroventral view (b). c Chaitophorus populeti, lateral view. Ch. tremulae, dorsal view, membranous area in front of the subgenital plate (asterisk) (d), lateral view membranous area in front of the subgenital plate (asterisk) (e). f Periphyllus coracinus, ventral view, membranous area in front of the subgenital plate (asterisk). g P. singeri, dorsal view. a, c, d, f, g light microscopy; b, e scanning electron microscopy. a aedeagus, bp basal part of phallus with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, c cauda, p parameres
Drepanosiphum oregonensis Granovsky, 1939 (male winged)—similar to those of D. platanoidis with pale, lobate parameres and wider basal part of phallus.
Chaitophorinae
Chaitophorus populeti (Panzer, 1801) (male winged or wingless)—parameres dusky, large, rounded (triangular in lateral view) with numerous, long setae on the whole surface. Basal part of phallus rather long, flattened, oval paddle-shaped with few fine setae. Distal parts of sclerotized arms with rounded processes (Fig. 2c). Aedeagus long, shapeless. In material preserved in alcohol (winged males), membranous area in front of subgenital plate clearly visible.
Chaitophorus tremulae Koch, 1854 (male winged)—very similar to those of Ch. populeti, with narrower basal part of phallus. In material preserved in alcohol (Fig. 2d) and in the scanning electron micrographs (Fig. 2e), membranous area in front of subgenital plate clearly visible.
Periphyllus coracinus (Koch, 1854) (male winged)—parameres darker on the apices, large, triangular, with numerous, very long setae on the whole surface. Basal part of phallus rather short, flattened, with numerous setae. Distal parts of sclerotized arms with rounded processes. Aedeagus long, shapeless. In material preserved in alcohol, membranous area in front of the subgenital plate clearly visible (Fig. 2f).
Periphyllus singeri (Börner, 1952) (male winged)—very similar to those of P. coracinus with paler parameres and wide, subquadrangular basal part of phallus (Fig. 2g).
Chaetosiphella stipae Hille Ris Lambers, 1947 (male wingless)—parameres uniformly dark, rather small, lobe-shaped with numerous, rather long setae on the whole surface (Fig. 3a). Basal part of phallus rather short, flattened, with few fine setae (Fig. 3b). Sclerotized arms much wider than in above-studied species (Fig. 3c). Aedeagus short, shapeless (Fig. 3b).
External genitalia of males. Chaetosiphella stipae, ventral view (a), laterodorsal view (b), dorsal view (c). d Ch. stipae subsp. setosa, dorsal view. e Sipha (Rungsia) maydis, laterodorsal view. f Laingia psammae, ventral view. a, b, e scanning electron microscopy; c, d, f light microscopy. a aedeagus, ap anal plate, bp basal part of phallus with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, c cauda, p parameres
Chaetosophella stipae subsp. setosa Wieczorek, 2008 (male wingless)—very similar to those of Ch. stipae with darker and wider parameres (Fig. 3d).
Sipha (Rungsia) maydis Passerini, 1860 (male wingless)—similar to those of Ch. stipae with darker parameres, and basal part of phallus sharply pointed distally. Aedeagus slightly curved (Fig. 3e).
Laingia psammae Theobald, 1922 (male wingless)—parameres pale, small, triangular with numerous, rather long setae. Basal part of phallus rather short, flattened, with few fine setae; distal and proximal part of arms short (Fig. 3f). Aedeagus rather short, shapeless.
Calaphidinae
Clethrobius comes (Walker, 1848) (male winged)—parameres dark, large, broadly rounded with numerous (more numerous than in the above-studied species) long setae on the whole surface. Basal part of phallus is long, with few, short setae; sclerotized arms similar to those of Drepanosiphum platanoidis (Fig. 4a). Aedeagus long, shapeless.
External genitalia of males. a Clethrobius comes, lateral view. b Appendiseta robiniae, ventral view. Mexicallis spinifer, lateral view (c), lateroventral view (d), aedeagus (a) with spermatozoa (s). e Myzocallis (Lineomyzocallis) walshii, ventral view. f Panaphis juglandis, ventral view. a–c, e, f light microscopy; d scanning electron microscopy. a aedeagus, bp basal part of phallus with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, c cauda, p parameres
Appendiseta robiniae (Gillette, 1907) (male winged)—parameres dark, large, lobate, with numerous long setae on the whole surface. Basal part of phallus long, flattened with few setae; sclerotized arms are of a similar length to the proximal and distal parts. Aedeagus long, with distal part about two times wider than its basal part (Fig. 4b).
Mexicallis spinifer Remaudiere, 1982 (male winged)—parameres dark, large, triangular, with numerous long setae on the whole surface (Fig. 4c). Basal part of phallus rather long, flattened, spatulate, with few, long setae laying in a row in outer margin. Sclerotized arms have a rather long and thin distal part; proximal part is shorter and wider. Aedeagus rather short, shapeless (Fig. 4d).
Myzocallis (Lineomyzocallis) walshii Monell, 1879 (male winged)—parameres dusky, large, subtriangular with numerous, rather short setae on the inner margin. Basal part of phallus similar to those of the above-studied species, with the exception of few, short setae. Sclerotized arms have a very long and thin distal part; the proximal part is very short. Aedeagus long, with distal part about two times wider than its basal part (Fig. 4e).
Panaphis juglandis (Goeze, 1778) (male winged)—parameres darker on the inner margin, large, rounded, with numerous short setae on the whole surface. Basal part of phallus short, oval-shaped, with few, short setae. Distal and proximal parts of sclerotized arms are of a similar length (Fig. 4f). Aedeagus long, shapeless.
Pterocallis alni (De Geer, 1773) (male winged)—similar to those of P. juglandis with wider basal part of phallus.
Phyllaphidinae
Phyllaphis fagi (Linne, 1767) (male winged)—parameres darker on the inner margin, large, half-rounded, with numerous, long setae on the whole surface (Fig. 5a). Basal part of phallus rather short, widely rounded, with few setae (Fig. 5b). Distal and proximal parts of sclerotized arms are of a similar length. Aedeagus long, inverted question mark-shaped.
External genitalia of males. Phyllaphis fagi, ventral view (a), dorsal view (b). c Diphyllaphis mordvilkoi, ventral view. d Subsaltusaphis flava, ventral view. e Lizerius ocoteae, ventral view. f Neuquenaphis edwardsi, lateral view. a, c–f light microscopy; b scanning electron microscopy. a aedeagus, bp basal part of phallus, c cauda, p parameres, short proximal (solid arrows) and long distal (dotted arrows) part of sclerotized arms
Diphyllaphis mordvilkoi (Aizenberg, 1932) (male wingless)—parameres dusky, large, subtriangular with sharply pointed edges and rather long, numerous setae. Basal part of phallus is similar to those of P. fagi. Sclerotized arms have a long and thin distal part; proximal part is short and wide (Fig. 5c).
Saltusaphidinae
Subsaltusaphis flava Hille Ris Lambers, 1939 (male wingless)—parameres dusky, large, elongated, lying almost parallel with numerous long setae on the inner margin (Fig. 5d). Basal part of phallus short, flattened. Distal and proximal parts of sclerotized arms are of a similar length.
Lizeriinae
Lizerius ocoteae E.E. Blanchard, 1923 (male wingless)—parameres dusky, large, lobate with numerous setae. Basal part of phallus wide with sharply pointed edges. Sclerotized arms have a long and thin distal part; proximal part is short and wide (Fig. 5e).
Spicaphidinae
Neuquenaphis edwardsi (Laing, 1927) (male winged)—parameres darker on the outer margin, large, triangular, with numerous long setae on the whole surface. Basal part of phallus long, pointed. Sclerotized arms have a rather long and thin distal part; proximal part is shorter and wider (Fig. 5f).
Tamalinae
Tamalia sp. (male winged)—parameres darker on outer margin, large, triangular, with numerous long setae on outer margin and shorter ones on the whole surface. Basal part of phallus long, pointed. Proximal part of sclerotized arms short and wider than in the above-studied species; distal part long and thin (Fig. 6a). Aedeagus long, shapeless (Fig. 6b).
External genitalia of males. Tamalia sp., ventral view (a). lateral view (b). c Parachaitophorus yamashitai, dorsal view. d Phleomyzus passerini, dorsal view. e Aphis pomi, dorsal view. f Brachycaudus divaricatae, ventral view. a, c–e light microscopy; b, f scanning electron microscopy. a aedeagus, ap anal plate, bp basal part of phallus with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, c cauda, p parameres
Parachaitophorinae
Parachaitophorus yamashitai Sorin, 1979 (male winged)—parameres pale with darker margin, large, lobate with numerous, long setae on the margin. Basal part of phallus rather long, flattened, with few setae. Distal parts of sclerotized arms have distinct, rounded processes (Fig. 6c).
Phleomyzinae
Phleomyzus passerini (Signoret, 1875) (male winged)—parameres pale, large, almost circular with numerous, long setae on the whole surface. Basal part of phallus paddle-shaped, with few setae. Both proximal and distal parts of sclerotized arms are rather long and thin (Fig. 6d).
Aphidinae
Aphis pomi De Geer, 1773 (male winged)—parameres dark, large, lobate with long setae on the outer margin. Basal part of phallus short, hooked-shaped, with few setae. Sclerotized arms have a long and thin distal part; proximal part is much shorter (Fig. 6e). Aedeagus long, S-shaped.
Brachycaudus divaricatae Shaposhnikov, 1956 (male winged)—parameres dark, large, subtriangular with visible sculpturation and numerous, very long setae on the whole surface (Fig. 6f). Basal part of phallus rather long, hooked-shaped, without setae. Sclerotized arms have a long and thin distal part; proximal part is much shorter. Aedeagus rather short, oval-shaped (Fig. 6f).
Cavariella saxifragae Remaudiere, 1959 (male wingless)—parameres smaller than in above-studied species, subtriangular with numerous, long setae on the whole surface. Basal part of phallus short, hooked-shaped, with distinctly stronger sclerotized articulations than in the above-studied species (Fig. 7a, b). Aedeagus long, inverted question mark-shaped (Fig. 1b).
External genitalia of males. Cavariella saxifragae, dorsal view (a), dorsal view (b). C. theobaldi, lateroventral view (c), lateral view (d). e Myzus cerasi, ventral view. f Pterocomma populeum, ventral view. a, e, f light microscopy; b–d scanning electron microscopy. a aedeagus, ap anal plate, bp basal part of phallus with sclerotized arms consists of short proximal (solid arrows) and long distal (dotted arrows) part, c cauda, p parameres
Cavariella theobaldi (Gillette & Bragg, 1918) (male winged)—parameres large, lobate with numerous, long setae on the whole surface. Basal part of phallus short, hooked-shaped but thinner than in C. saxifragae. The scanning electron micrographs show a few circular pits distributed in two rows on the inner margin of basal part of phallus (Fig. 7c). Distal parts of sclerotized arms are long and thin with distinct capitate processes; proximal part is much shorter. Aedeagus long, with distal part about two times wider than its basal part (Fig. 7d).
Hyperomyzus pallidus Hille Ris Lambers, 1935 (male winged)—parameres dusky on the apices, large, subtriangular with numerous long setae on the inner margin. Basal part of phallus and its sclerotizations are similar to those of B. divaricatae.
Myzus cerasi Fabricius, 1775 (male winged)—parameres dusky on the inner margin, large, subtriangular with numerous, very long setae on the whole surface. Basal part of phallus large, oval-shaped, with few, short setae. Aedeagus long, shapeless (Fig. 7e).
Pterocomma populeum (Kaltenbach, 1843) (male winged)—parameres uniformly dark, large, inverted triangle-shaped, with numerous, very long setae on the whole surface. Basal part of phallus rather long and thin, club-shaped, with few, short setae. Distal parts of sclerotized arms are wide and robust (Fig. 7f). Aedeagus long, inverted question mark-shaped.
Discussion
The external genitalia of males in many species of Hemiptera are extremely characteristic and used for species determination (e.g. Gullan 1978; Gnezdilov 2007; Chłond and Junkiert 2010; Redei and Tsai 2010). However, these structures have not been used in Aphididae taxonomy to date—mostly because of the rarity of males. The present study shows that in all examined species (representatives of 11 taxa traditionally classified as subfamilies within 24 of the Aphididae), the external genitalia of males are well developed and typically consist of the phallus composed of the sclerotized basal part with its articulation and a membranous apical part—aedeagus. Laterally of the phallus, there is a pair of setose parameres.
The external genitalia of males—phallus
The phallus is a median intromittent organ as a whole, arising embryologically from the interno-median parts of larval phallic lobes, the externo-lateral parts of which give rise to parameres (Dupuis 1970). The phallus is often used synonymously with aedeagus or penis; however, in aphids, it is distinctly composed of a membranous part—aedeagus and the sclerotized basal part. The aedeagus is the largest component of the male genitalia in most of the species studied (Figs. 2b, 3e). This copulatory organ, continuating ductus ejaculatorius, is withdrawn within the body and everted during copulation. When the aedeagus is everted, the ejaculatory duct and outlets of vasa deferentia and accessory glands are pulled into it as has been shown in the present paper in the case of Appendiseta robiniae (Fig. 1f). The aedeagus is characterized by a large amount of variation in the shape—an inverted question mark-shaped (Drepanosiphum platanoidis Fig. 2b) or an S-shaped (Sipha (Rungsia) maydis Fig. 3e), sometimes oval-shaped but most often shapeless (Tamalia sp. Fig. 6b). However, in slide preparation, the aedeagus is not usually visible, and its shape is estimated mostly on the basis of the material preserved in alcohol or the scanning electron micrographs. The shape of the aedeagus is variable between different genera and species, but varies very little within each species (Polaszek 1987b in lit.). This characteristic is useful for the distinction of males belonging to different species; however, for diagnosing species, larger sample sizes would be necessary. Polaszek (1987b in lit.), after Bonhag and Wick (1953) terminology, distinguished the basal part of the aedeagus—a conjuctiva–– and its distal part—a vesica in some species. The present study does not support this division; however, in two species (Myzocallis (Lineomyzocallis) walshii Fig. 4e and Cavariella theobaldi Fig. 7c, d), the distal part of aedeagus was bulbous—about two times wider than its proximal part.
Laterally to the aedeagus, there is a pair of partially sclerotized lobes, which are called valves by some authors (Balbiani 1869; Ossiannilsson et al. 1970; Polaszek 1987b in lit.; Blackman et al. 2001). According to Snodgrass (1957), the so-called penis valves are merely a lateral sclerotization of the aedeagal wall. The scanning electron micrographs clearly show the lateral sclerotization of the otherwise membranous aedeagus which enfolds it and is characterized by various shapes (finger-like Fig. 2b, hooked-shaped Fig. 7f, paddle-shaped Fig. 2c and flattened Fig. 6d) and sizes (from elongated Fig. 6c to rather short Fig. 3f). Iglisch and Sobhani (1972), Sobhani and Iglisch (1972) and Heie (1980) in descriptions of the male genitalia of aphids also termed these structures the sclerotic basal part, as in the present paper. This paired dark pigmented and setose structure is distinctly visible in slide-mounted specimens (Fig. 6c–e). In all of the species studied, it is additionally fortified by the heavily sclerotized arms. They are probably apophyses (spiniform projections of the endoskeleton giving support for muscle attachment). In descriptions of the male genitalia of Aphididae, they are usually ignored; only Iglisch and Sobhani (1972) and Sobhani and Iglisch (1972) called them a “sclerotized fastener” and figured in Macrosiphoniella ptarmicae and Macrosiphoniella usquertensis. They also were figured by Balbiani (1869) in his description of the male genitalia of Macrosiphoniella millefolli (De Geer, 1773) and mentioned as the coriaceous ring by Essig and Abernathy (1952) in males of Periphyllus species. The present study shows that these structures are not uniform and vary in form and length. They are usually furcate; however, the proportion of the proximal and distal part of the arm as well as their width is different in particular species (e.g. rather long and thin in Drepanosiphum platanoidis Fig. 2a, long and wide in Pterocomma populeum Fig. 7f or short and wide in Laingia psammae Fig. 3f). The distal parts of the arms have additional distinct, rounded processes as in the studied representatives of Chaitophorus, Periphyllus (Fig. 2f) and Parachaitophorus yamashitai (Fig. 6c). When the aedeagus is withdrawn, the lobes of the basal part cover the genital opening, remaining in contact with each other. During copulation, supported by sclerotized arms, they take part in everting the aedeagus and maintaining it in position. Moreover, in some species (e.g. Drepanosiphum platanoidis Fig. 2b and Cavariella theobaldi Fig. 7c), the SEM micrographs show a few small circular pits distributed in rows on the inner margin of the basal part of the phallus. They may have a sensory function; however, no comparative study concerning these structures has been investigated so far.
The external genitalia of males—parameres
At the anterior end of the genital area and above basal part of phallus, there is a pair of cone-shaped ventral processes, the parameres. These structures are described as synonymous with claspers by most authors (Sorin 1965; Polaszek 1987b in lit.), or opercula (Ossiannilsson et al. 1970). Parameres are independent of the phallus but arise embryologically from primary phallic lobes (Dupuis 1970). As is shown in Fig. 1g in nymphs of Appendiseta robiniae, parameres are positioned ventrally at the base of the canal where the phallus is withdrawn. In adult males of all of the species studied, parameres are rather large, united and vary in form: subtriangular in Drepanosiphum oregonensis, D. platanoidis (Fig. 2a), Myzocallis (Lineomyzocallis) walshii (Fig. 4e), Brachycaudus divaricatae (Fig. 6f), Hyperomyzus pallidus, Myzus cerasi (Fig. 7e) and Pterocomma populeum (Fig. 7f) or rounded in Clethrobius comes (Fig. 4a), Panaphis juglandis (Fig. 4f) or Pterocallis alni. The most variable shape of parameres is observed in Chaitophorinae (triangular in Periphyllus coracinus, subquadrangular in Periphyllus singeri Fig. 2g, rounded in Chaitophorus populeti and lobe-shaped in Chaetosiphella stipae Fig. 3c), whereas the most variable length is in Calaphidinae (e.g. elongated in Myzocallis (Lineomyzocallis) walshii (Fig. 4e) and shortened in Clethrobius comes Fig. 4a). The largest parameres are present in Pterocomma populeum (Aphidinae) (Fig. 7f), whereas in representatives of the tribe Siphini (Chaitophorinae), the parameres are small, lobe-shaped (Wieczorek 2010), and especially in Laingia psammae weakly visible (Fig. 3f). Among the species studied, males of Siphini are the smallest (but not dwarfish), and the small size of their parameres results from their smaller body size. In most of the species studied, parameres are dark pigmented, covered with numerous setae and with visible sculpturation (visible in SEM micrographs—e.g. Brachycaudus divaricatae Fig. 6f). In male insects, these structures are used during copulation to hold the female—the grasping apparatus; however, data concerning the mating behaviour of aphids are rather scarce (Kozłowski 1991; Dagg and Scheurers 1997; Dagg 2003; Hales 2005). Further research, i.e., to describe the role of parameres in the copulation of aphids is also necessary. Similarly, the role of the membranous area in front of the subgenital plate, visible in some species of the Chaitophorinae, is unknown. This membranous area was observed only in winged males of Chaitophorus and Periphyllus species (Fig. 2d–f) and was not present in wingless males (own unpublished data) or in closely related wingless males of Siphini species (Wieczorek 2010). Similar structures were described in aphids only in viviparous females of the Neophyllaphis species and Paoliella (Lizerocallis) (Quednau 2010). These adhesive vesicles were everted when insects were disturbed and may prevent the aphid from being removed from the host plant (White and Carver 1971). Nevertheless, in males, this membranous area may be involved in mounting and copulation, but observation of these species in copula is necessary.
The external genitalia of males—phylogenetic implications
Males of the species studied, which represent the Drepanosiphinae, Chaitophorinae, Calaphidinae, Phyllaphidinae, Saltusaphidinae, Lizeriinae, Spicaphidinae, Tamalinae, Parachaitophorinae, Phleomyzinae and Aphidinae, taxa that traditionally are classified as subfamilies, have normal size (i.e. not dwarfish) and are mostly winged. Males with modified genitalia are usually dwarfish (Anoeciinae, Eriosomatinae, Hormaphidinae, Mindarinae and Thelaxinae with the exception of Aiceoninae, Greenideinae and Lachninae) and wingless, so these modifications may result from their smaller body size (own unpublished data). On the other hand, molecular studies on the phylogeny of Aphididae (Ortiz-Rivas et al. 2004; Ortiz-Rivas and Martinez-Torres 2010) have indicated the existence of three main lineages of aphids that have been named A+D (Aphidinae, Calaphidinae, Chaitophorinae and Drepanosiphinae), E+T (Anoeciinae, Eriosomatinae, Hormaphidinae, Mindarinae and Thelaxinae) and L (Lachninae) (Ortiz-Rivas and Martinez-Torres 2010). The results of the present study largely agree with this proposal. The male genitalia studied in species representing lineage A+D are well developed and typically consist of the phallus (composed of the basal part and aedeagus) and parameres. Our own preliminary data show that the lineage E+T comprises taxa of dwarfish males with strongly modified genitalia, whereas lineage L (Lachninae) has normal-sized males with the most varied genitalia among Aphididae. According to Quednau’s (2010) morphological study, three major groups of aphids can also be distinguished: 1. Eriosomatinae, Hormaphidinae and Anoeciinae; 2. Calaphidinae, Chaitophorinae, Drepanosiphinae, Israelaphidinae, Phyllaphidinae, Pterastheniinae, Saltusaphidinae, Lizeriinae, Macropodaphidinae, Mindarinae, Neophyllaphidinae, Parachaitophorinae, Spicaphidinae, Taiwanaphidinae and Tamalinae (the drepanosiphine aphids); and 3. Aphidinae, Greenideinae and Lachninae. Our results support grouping subfamilies in lineages 1 and 2. Lineage 1 comprises taxa of dwarfish males with strongly modified genitalia. The closely related drepanosiphine aphids (lineage 2) have normal-sized males with not modified genitalia. The lineage 3 comprises taxa with normal-sized males; however, Aphidinae have not modified genitalia, whereas Greenideinae and Lachninae have strongly modified genitalia (own unpublished data). The present study shows that Aphidinae are characterized by similar male genitalia to those in the drepanosiphine aphids (lineage 2); nevertheless, Aphidinae was usually thought to be a sister to subfamily Lachninae (Heie 1987; Wojciechowski 1992).
Summary
To unify the nomenclature of the external male genitalia elements of Aphididae, we propose using the following nomenclature: the phallus, which consists membranous apical part—the aedeagus and basal part with its sclerotization (proximal and distal part of sclerotized arms), parameres (Table 1). Representatives of the Drepanosiphinae, Chaitophorinae, Calaphidinae, Phyllaphidinae, Saltusaphidinae, Lizeriinae, Spicaphidinae, Tamalinae, Parachaitophorinae, Phleomyzinae and Aphidinae have normal-sized males without modified external genitalia. The paper supports the phylogenetic analysis of aphid evolution based on molecular studies of Ortiz-Rivas and Martinez-Torres (2010); our results are in agreement with the Quednau’s division of aphids into three major groups (especially relationships within drepanosiphine aphids), with the exception of the position of Aphidinae and its relation to Lachninae.
References
Baker AC (1915) The woolly apple aphis. Rep US Dep Agric 101:1–55
Balbiani EG (1869) Memoire sur la generation des aphides. Ann Sci Nat Zool Paleontol 11:5–89
Blackman RL, De Bois E, Czylok A (2001) Occurrences of sexual morphs in Trama troglodytes von Heyden, 1837 (Hemiptera, Aphididae). J Nat Hist 35:779–785
Blochman F (1887) Uber den Geschlechtsgeneration von Chermes abietis L. Biol Centralbl 7:417–420
Bochen K, Klimaszewski SM, Wojciechowski W (1975) Budowa męskiego układu rozrodczego Macrosiphoniella artemisiae (B.De Fonsc.) i M. millefolli (De Geer) (Homoptera, Aphididae). Acta Biol Univ Śl Katowice 90:73–81
Bonhag PF, Wick JR (1953) The functional anatomy of the male and female reproductive system of the milkweed bug Oncopeltus fasciatus (Dallas) (Heteroptera: Lygeidae). J Morphol 93:177–284
Chłond D, Junkiert Ł (2010) Peyrierocoris a new genus of Harpactorinae (Hemiptera: Heteroptera: Reduviidae) from Madagascar. Zootaxa 2487:33–42
Cholodkovsky N (1900) Uber den mannlichen Geschlechtsapparat von Chermes. Biol Chl 20:1–619
Dagg JL (2003) Copula duration and sperm economy in the large thistle aphid, Uroleucon cirsii (Hemiptera: Aphididae). Eur J Entomol 100:201–203
Dagg JL, Scheurers S (1997) Observations on some patterns of the males’ sexual behavior of certain aphid species indicate the existence of the male marking pheromones. In: Nieto Nafría JM, Dixon AFG (eds) Aphids in natural and managed ecosystems. Universidad de León, León, pp 167–171
Dupuis C (1970) Heteroptera. In: Tuxen SL (ed) Taxonomist’s glossary of genitalia of insects, 2nd edn. Ejnar Munksgaard, Copenhagen, pp 1–359
Essig EO, Abernathy F (1952) The Aphid genus Periphyllus. A systematic biological and ecological study. University of California Press, Berkeley, pp 1–166
Favret C, Tooker JF, Hanks LM (2004) Iowana frisoni Hottes (Hemiptera: Aphididae) redescribed with notes on its biology. Proc Entomol Soc Wash 106:26–34
Głowacka E, Klimaszewski SM, Szelegiewicz H, Wojciechowski W (1974a) Uber den Bau des mannlichen Fortpflanzungssystems der Lachniden (Homoptera, Aphidoidea). Ann Zool Warszawa 32:39–49
Głowacka E, Klimaszewski SM, Szelegiewicz H, Wojciechowski W (1974b) Uber den Bau des mannlichen Fortpflanzungssystems der Aphiden (Homoptera, Aphidoidea). Ann UMCS Lublin Sec C 29:133–138
Gnezdilov VM (2007) On the systematic positions of the Bladinini Kirkaldy, Tonginae Kirkaldy, and Trienopinae Fennah (Homoptera, Fulgoroidea). Zoosystematica Rossica 15(2):293–297
Grimaldi DA, Engel MS (2005) Evolution of insects. Cambridge University Press, Cambridge, pp 1–755
Gullan PJ (1978) Male insects and galls of the genus Cylindrococcus Maskell (Homoptera: Coccoidea). J Aust Entomol Soc 17:53–61
Hales DF (2005) Physiological determinants of male mating performance in aphids (Hemiptera: Aphididae). Eur J Entomol 102:641–646
Heie OE (1980) The Aphidoidea (Hemiptera) of Fennoscandia and Denmark. I. General Part. The families Mindaridae, Hormaphididae, Thelaxidae, Anoeciidae, and Pemphigidae. Klampenborg, Fauna Ent. Scand., Scandinavian Science Press LTD, Klampenborg 9:1–235
Heie OE (1987) Paleontology and phylogeny. In: Minks AK, Harrewijn P (eds) Aphids, their biology, natural enemies and control, vol 2A. Elsevier, Amsterdam, pp 367–391
Iglisch I, Sobhani P (1972) Ein Beitrag zur Morphologie der Genitalien von Blattläusen (Homoptera: Aphidoidea). Teil 1: Zur Morphologie des männlichen Geschlechtstieres von Macrosiphoniella ptarmicae H.R.L. Z Angew Zoologie 59:5–15
Kawada K (1987) Polymorphism and morph determination. In: Minks AK, Harrewijn P (eds) Aphids, their biology, natural enemies and control, vol 2A. Elsevier, Amsterdam, pp 315–320
Klimaszewski SM, Szelegiewicz H, Wojciechowski W (1973) Uber den Bau des mannlichen Fortpflanzungssystems von Drepanosiphum platanoidis (Schr.) (Homoptera, Aphidoidea). Bull Acad Pol Sci Warszawa 10:671–674
Kozłowski MW (1991) Mating behavior and high-level polygamy in the aphids Periphyllus acericola and Drepanosiphum platanoidis (Homoptera, Drepanosiphidae). Ethol Ecol Evol 3:285–294
Matsuda R (1976) Morphology and evolution of the insect abdomen. Pergamon Press Ltd, Oxford, pp 1–534
Miyazaki K (1987) Forms and morphs of aphids. In: Minks AK, Harrewijn P (eds) Aphids, their biology, natural enemies and control, vol 2A. Elsevier, Amsterdam, pp 27–50
Nieto Nafria JM, Mier Durante MP, Remaudiere G (1998) Les noms des taxa du group-famille chez les Aphididae (Hemiptera). Rev Fr Entomol (NS) 19:77–92
Ortiz-Rivas B, Martinez-Torres D (2010) Combination of molecular data support the existence of three main lineages in the phylogeny of aphids (Hemiptera: Aphididae) and the basal position of the subfamily Lachninae. Mol Phylogenet Evol 55(1):305–317
Ortiz-Rivas B, Moya A, Martinez-Torres D (2004) Molecular systematics of aphids (Homoptera: Aphididae): new insights from the long-wavelength opsin gene. Mol Phylogenet Evol 30:24–37
Ossiannilsson BF, Russel LM, Weber H (1970) Homoptera. In: Tuxen SL (ed) Taxonomist’s glossary of genitalia of insects, 2nd edn. Ejnar Munksgaard, Copenhagen, pp 1–359
Płachno BJ, Świątek P (2009) Functional anatomy of the ovule in Genlisea with remarks on ovule evolution in Lentibulariaceae. Protoplasma 236:39–48
Płachno BJ, Świątek P (2010) Unusual embryo structure in viviparous Utricularia nelumbifolia with remarks on embryo evolution in genus Utricularia. Protoplasma 239:69–80
Polaszek A (1987a) Studies on the comparative anatomy of aphid reproductive systems. In: Population, structure, genetics and taxonomy of Aphids and Thysanoptera. Proceedings of the International Symposium, Smolenice, Czechoslovakia, 1985. SPB Acad Publ, The Hague, pp 261–266
Polaszek A (1987b) Comparative anatomy of the male aphid reproductive system. Unpublished Ph.D. Thesis
Quednau FW (2010) Atlas of the Drepanosiphine aphids of the world. Part III: Mindarinae Tullgren 1909 to Saltusaphidinae Baker 1920 (Hemiptera: Sternorrhyncha, Aphididae). Mem Am ent Inst 83:1–361
Redei D, Tsai JF (2010) A survey of the saicine assassin bugs of Taiwan (Hemiptera: Heteroptera: Reduviidae). Acta Entomol Mus Nat Pragae 50:15–32
Snodgrass RE (1957) A revised interpretation of the external reproductive organs of male insects. Smithson Miscell Coll 135:1–60
Sobhani P, Iglisch I (1972) Ein Beitrag zur Morphologie der Genitalien von Blattläusen (Homoptera: Aphidoidea). Teil 2: Zur Morphologie des männlichen Geschlechtstieres von Macrosiphoniella usqertensis H.R.L. Z Angew Zoologie 59:447–461
Sorin M (1965) Three new species of the genus Stomaphis in Japan with a redescription of S. yanonis Takahashi (Aphididae, Homoptera). Bull Univ Osaka Prefect 16:81–88
Takahashi R (1960) Kurisakia and Aiceona of Japan (Homoptera, Aphididae). Insecta Matsumurana 23(1):1–10
Vitlaczil E (1882) Zur Anatomie der Aphiden. Arb Zool Inst Univ Vien 4:397–441
Vollrath F (1998) Dwarf males. Tree 13:159–163
von Dohlen CD, Rowe CA, Heie OE (2006) A test for morphological hypotheses for tribal and subtribal relationships of Aphidinae (Insecta: Hemiptera: Aphididae) using DNA sequences. Mol Phylogenet Evol 38:316–329
White WS, Carver M (1971) Adhesive vesicles in some species of Neophyllaphis Takahashi, 1920 (Homoptera: Aphididae). J Aust Entomol Soc 10:281–284
Wieczorek K (2006) Anatomical investigations of the male reproductive system of five species of Calaphidinae (Hemiptera, Aphidoidea). Insect Syst Evol 37:457–465
Wieczorek K (2008) Structure of the male reproductive system of Anoecia (Anoecia) corni Fabricius, 1775 (Hemiptera, Aphidoidea) a representative of the family Anoeciidae. Acta Zool 89:163–167
Wieczorek K (2010) A monograph of Siphini Mordvilko, 1928 (Hemiptera, Aphidoidea: Chaitophorinae). Wydawnictwo Uniwersytetu Śląskiego ISSN 0208-6336 ISBN 978-83-226-1966-7 Katowice 1–297
Wieczorek K, Świątek P (2008) Morphology and ultrastructure of the male reproductive system of the woolly beech aphid Phyllaphis fagi (Hemiptera, Aphidoidea: Phyllaphidinae). Eur J Entomol 105:707–712
Wieczorek K, Świątek P (2009) Comparative study of the structure of the male reproductive system of monoecius and heteroecius aphid species Glyphina betulae and Anoecia (Anoecia) corni (Hemiptera, Aphididae). Zool Anz 248:153–159
Wieczorek K, Wojciechowski W (2004) The systematic position of Chaitophorinae (Hemiptera, Aphidoidea) in the light of anatomy research. Insect Syst Evol 35(3):317–327
Wojciechowski W (1977) Procesy oligomeryzacji w budowie męskiego układu rozrodczego miodownic (Homoptera, Lachnidae). Acta Biol Univ Śl Katowice 3:140–164
Wojciechowski W (1992) Studies on the systematic system of Aphids (Homoptera, Aphidinea). Prac Nauk. Univ Śl, Katowice, 1–75
Yang CT, Chang TY (2000) The external male genitalia of Hemiptera (Homoptera-Heteroptera). Shih Way Publishers, Taiwan, pp 1–745
Acknowledgments
We are sincerely grateful to Andrev Polaszek for access to his unpublished Ph. D. Thesis concerning the comparative anatomy of the male aphid reproductive system. We thank Roger Blackman, the Natural History Museum, London and David Redei, the Natural History Museum, Budapest for a critical reading of the manuscript and valuable comments. We would also like to express our thanks to Barbara Wilkaniec and Beata Borowiak-Sobkowiak Agricultural University, Poznań for their help in collecting the material of B. divaricatae and A. robiniae, to Georges Remaudière, Daniele Matile-Ferrero and Thierry Bourgoin, Museum national d’Histoire naturelle, Paris and to Niels P. Kristensen and Lars Bjørn Vilhelmsen, the Natural History Museum of Denmark, Copenhagen for the loan of M. spinifer, D. mordvilkoi, I. lambersi, L. ocoteae, Tamalia sp., P. yamashitai, P. passerini, C. saxifragae, C. theobaldi and P. singeri. Special thanks go to Łukasz Junkiert for preparing the drawings. The authors are grateful the Reviewers and the Editor for all suggestions and comments that improved the manuscript.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Bartolomaeus.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wieczorek, K., Płachno, B.J. & Świątek, P. Comparative morphology of the male genitalia of Aphididae (Insecta, Hemiptera): part 1. Zoomorphology 130, 289–303 (2011). https://doi.org/10.1007/s00435-011-0134-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-011-0134-z