Abstract
Mutations in effectors of the hedgehog signaling pathway are responsible for a wide variety of ocular developmental anomalies. These range from massive malformations of the brain and ocular primordia, not always compatible with postnatal life, to subtle but damaging functional effects on specific eye components. This review will concentrate on the effects and effectors of the major vertebrate hedgehog ligand for eye and brain formation, Sonic hedgehog (SHH), in tissues that constitute the eye directly and also in those tissues that exert indirect influence on eye formation. After a brief overview of human eye development, the many roles of the SHH signaling pathway during both early and later morphogenetic processes in the brain and then eye and periocular primordia will be evoked. Some of the unique molecular biology of this pathway in vertebrates, particularly ciliary signal transduction, will also be broached within this developmental cellular context.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The hedgehog signaling pathway was named after the denticle phenotype of larval Drosophila fruit fly mutants, in which the alternating pattern of abdominal bristles and naked cuticle was replaced by a stubby and uniformly fuzzy appearance. The first description presciently grouped the “hedgehog” (hh) and “cubitus interruptus” (Ci) mutants among those affecting this segmental polarity pattern (Nüsslein-Volhard and Wieschaus 1980). As the corresponding genes were identified and characterized, it turned out that in all vertebrates including humans, hh ligand and Ci transcription factor orthologs, as well as dozens of intermediate effectors, are functionally conserved in many ways and have diversified in others. Hedgehog signaling pathways are now known to regulate many fundamental cellular processes of patterning, cell identity, and environmental responsiveness in all Eumetazoa (Matus et al. 2008; Sigg et al. 2017). They have been doing so over the last 940 million years (Nikoh et al. 1997).
The hedgehog pathway, impinging on similarly conserved/diversified transcriptional gene regulation networks, has been co-opted during the evolution of numerous complex organ systems in the vertebrate clade, particularly the eye (Nilsson 2004; Meulemans and Bronner-Fraser 2005). Non-coding, cis-regulatory elements control when and where not only the ligands, but also the effectors and potential downstream targets may be deployed (Jeong et al. 2006; Cvekl and Duncan 2007); as such, these may also account for some of the complex genetics behind human eye malformations.
The bases of human ocular development
The early eye primordium is an outgrowth of the forebrain
In human embryos, the eye primordia develop as a subdomain of the prospective forebrain (prosencephalon), which can be distinguished morphologically by gestational day (GD)17 (reviewed in Creuzet and Etchevers 2017; Van Cruchten et al. 2017). Work in animal models has shown that the specification of eye fate is accomplished by a network of conserved transcription factors (TFs) that mobilize and coordinate core signaling pathways reiteratively used in organ morphogenesis. These genes are expressed in overlapping domains in the anterior neural primordium and their concerted activity specifies eye fate (reviewed in Beccari et al. 2013; Martinez-Morales 2016). Some of these will also be discussed in more detail further along in this review, as many are either regulated directly by Shh activity or affect its timing and effects during eye formation (Table 1).
As the rostral prosencephalon bulges and initially splays at GD22, bilateral pits emerge on each side midway between the neural fold and floor plate, at an equivalent distance from the edge of the anterior neural fold (Cook et al. 2006; Creuzet and Etchevers 2017). These demarcate the future vesicles that will emerge toward the ectoderm over the next few days. By the time the optic vesicles have evaginated, they are already patterned along the proximal–distal (PD) axis, to give rise to the optic stalk proximally, and neural retina/retinal pigment epithelium (NR/RPE) distally.
By GD28, the optic vesicles have established full contact with the overlying ectoderm and a number of inductive events are triggered in each. This leads to the finer specification of the distal-most portion of the optic vesicle itself as the future neural retina, as well as the induction of lens fate in the overlying ectoderm. These inductive events are accompanied by extensive reshaping of the eye primordium, which then folds over itself to give rise to the optic cup, a bilayered structure in which the external layer will give rise to the RPE and the internal layer to the neural retina (reviewed by Martinez-Morales et al. 2017; Van Cruchten et al. 2017). In parallel to optic cup folding, the lens primordium invaginates and buds off the overlying ectoderm, to give rise to the lens vesicle. During this time, the entire forebrain primordium itself is growing in all dimensions. A flash animation of this dynamic process, to this point highly conserved among vertebrates and under control of homologous genes across species from fish to primates, can be found at the following link (Lamb et al. 2007): https://media.nature.com/full/nature-assets/nrn/journal/v8/n12/extref/nrn2283-s1.swf.
Positional signals are exchanged within and between the ocular neuroepithelium and surrounding mesenchyme
An important population of mesenchymal cells intervenes between the prosencephalon and the overlying ectoderm, later separating the ectoderm from the thinning edge of the optic cup as it becomes the iris, and overlying the lens vesicle. It surrounds and is indispensable for the differentiation of the RPE and subsequently for the entire ocular globe and nerve as they form (Fuhrmann et al. 2000; Creuzet et al. 2005). This mesenchyme is mostly made up of cells known as the neural crest; in older literature it is called “mesectoderm”. Using fate-mapping techniques in model organisms such as chicken or fish embryos, neural crest cells (NCC) have been shown to delaminate from the dorsal neural folds of the caudal forebrain and rostral midbrain to surround the optic vesicles from both above and below to encase the prospective forebrain and eyes (Fuhrmann et al. 2000; Etchevers et al. 2001; Langenberg et al. 2008).
NCC initially outnumber the mesenchymal head mesoderm cells responsible for the formation of the first blood vessels on the dorsal aspect of the brain and eyes, but they rapidly co-mingle to provide a cellular matrix and organize into a primitive vascular network around the entire prosencephalic primordium and within the developing eye itself (Etchevers et al. 2001). NCC later infiltrate the iris to provide its pigmentation, and also differentiate into the corneal stroma and endothelium, the pericytes of the ocular choroid and hyaloid vasculature, as well as the viscoelastic sclera of the eyeball, orbital cartilage and bone, and oculomotor tendons (Johnston et al. 1979; Couly et al. 1993; Kastner et al. 1994; Zieske 2004; Gage et al. 2005; Grenier et al. 2009; Macé et al. 2011).
The NCC mesenchyme constitutes most of the structures of the face and neck around which the sense and secretory organs are organized, particularly the facial skeleton (Le Lièvre and Le Douarin 1975). Shh acts at multiple times on and during cephalic NCC differentiation into these and other structures as well as directly on the ocular primordia (Jeong et al. 2004; Lan and Jiang 2009; Aoto and Trainor 2015). Both posterior cephalic NCC and Shh activity are critical for cardiac outflow tract morphogenesis during heart development (Kirby et al. 1983; Dyer and Kirby 2009). These observations explain clinical associations of facial dysmorphic traits such as hypotelorism, agnathia or cleft lip/palate, or cardiac defects, with ocular developmental anomalies (ODA), some of which had been described many decades before the molecular bases were established (DeMyer et al. 1964; Jeong et al. 2004, 2006; Pasutto et al. 2007; Golzio et al. 2007; Chassaing et al. 2009; Aoto and Trainor 2015).
By the beginning of gestational week 8 (GW8), the optic stalk and the optic cup are fully formed, and the differentiation of the lens as well as the retina is well underway (Van Cruchten et al. 2017). These processes are controlled both directly and indirectly by Shh in the different primordia. Notably, periocular NCC interact with the forming cornea and lens by modulating and transducing Wnt and Tgfb signaling pathways as well as in defining the edge of the optic cup from which the ciliary body, Schlemm’s canal and iris will form (Ittner et al. 2005; Aguiar et al. 2014).
The morphogenetic events leading to optic cup folding also induce the formation of a perpendicular fold, running along the ventral proximo-distal axis of the optic primordium. This opening, called the optic or choroid fissure, runs along the future optic stalk and also comprises the ventral retina. A transient structure as such, it is the conduit to establish vascular irrigation of the differentiating organ, and subsequently guides the exit of retinal ganglion cell axons to innervate the visual centers in the developing brain as the future optic nerve forms (Patel and Sowden 2017). At its most distal aspect, the fissure also attains the iris. Eventually, it will fuse all along its length to generate the fully mature optic nerve, retina and anterior chamber. Defective choroid fissure closure leads to various degrees of coloboma. Again, Shh seems to play a determinant role in this process (Morcillo et al. 2006; Gongal et al. 2011). In humans, the fissure closes around the seventh week of gestation (Barishak 1992).
Further differentiation of RPE cells give rise to a squamous, pigmented epithelium, while differentiation of the neural retina gives rise to the laminated circuit of neurons involved in light reception and processing (reviewed in Creuzet and Etchevers 2017; Van Cruchten et al. 2017). RPE function is critical for the survival and appropriate connectivity of the overlying retinal photoreceptor layer and remains so throughout life—RPE malfunction, as can happen in retinitis pigmentosa, leads to retinal dystrophy (McKechnie et al. 1985; Zhao et al. 2011). These differentiation events continue throughout gestation and throughout the first year after birth, and Hh pathway signaling remains important at multiple points in these processes.
Any given ODA has multiple specific causes
Interrupting any of the steps in ocular development can lead to a range of overlapping pathological outcomes. Brain malformations affecting the future telencephalon and diencephalon are often associated with severe ODA—primary anophthalmia is found with anencephaly and thereby incompatible with postnatal life; severe microphthalmia resulting in secondary anophthalmia (micro/anophthalmia), nanophthalmia and cystic eye are found in syndromes also affecting the structure of the anterior brain (prosencephalon) which will give rise to the cerebral hemispheres of the telencephalon and the hypothalamic–pituitary axis of the diencephalon, and can thus be found associated with combined pituitary hormone deficiencies. These are regularly found with mutations of OTX2, an evolutionarily conserved TF gene highly important for anterior head development (Acampora et al. 1995; reviewed in; Slavotinek 2011). Phenotypic outcomes, even within the same family, of OTX2 mutations range from subclinical to micro/anophthalmia but also include otocephaly and dysgnathia (Ragge et al. 2005a; Chassaing et al. 2012). Otx2 is an active Shh target that is expressed in the ventral optic primordium and subsequently becomes a master regulator of RPE differentiation, necessary for survival and persistence of the optic cup (Martínez-Morales et al. 2003, 2004; Hever et al. 2006; Halilagic et al. 2007; Larsen et al. 2009; Westenskow et al. 2009).
Aphakia results from the interruption of any signals involved in optic cup invagination, lens induction, differentiation or survival—if early enough, the absence of lens will have repercussions for the remaining optic vesicle. Congenital cataract results from affecting later lens differentiation and secondary fiber formation and matrix composition. NCC are indispensable for anterior chamber development, particularly the cornea, iris, trabecular network and ciliary process (reviewed by Williams and Bohnsack 2015; Fuhrmann et al. 2000), associated with myopia, microcornea, iris hypoplasia, corectopia, polycoria and other signs of Axenfeld–Rieger syndrome or congenital glaucoma. NCC are also necessary for the differentiation of the choroid, and they constitute the sclera and attachment tendons as well as contribute to the innervation of the extraocular muscles, by which they may play a part in congenital nystagmus or strabismus (Creuzet et al. 2005; Grenier et al. 2009; reviewed in; Williams and Bohnsack 2015). However, when intervening between prospective lens and the distal point of contact with the ectoderm, ectopic NCC are associated with inhibition of lens formation and differentiation (Sullivan et al. 2004).
Correct optic stalk elongation and induction of the RPE are associated with the differentiation of the lens. In chick embryos where the Pax6 TF was inhibited experimentally, severe colobomas are found with remnants of ectopic RPE, in the absence of lens or an appropriately laminated neural retina (Canto-Soler and Adler 2006). Malformations of the resultant optic nerve, either due to coloboma or problems in retinal ganglion cell axonal growth to the brain, are visible in human pathology as leukocoria.
Hedgehog signaling in early brain and ocular pathogenesis
The holoprosencephaly spectrum impacts eye formation and is the result of SHH signaling deficiency
The Hh signaling pathway is essential for many of the steps leading to the formation of a mature ocular primordium, described above. SHH itself is one of three proteins of the hedgehog protein family in humans (including Sonic, Desert and Indian hedgehog; Pandit and Ogden 2017), and has been the most studied as it appears to play the most varied roles in organogenesis around the body. In humans, SHH is located on chromosome 7q36.3 and the only relevant ligand from this family during embryonic forebrain development (Odent et al. 1999). During these early stages, the role of SHH in controlling eye patterning is intimately linked to its function directing the specification of ventral brain structures. Shh is expressed along the axial mesoderm (the prechordal plate and notochord) and the neural floorplate where it is particularly essential to pattern ventral neural tissues (Krauss et al. 1993; Ekker et al. 1995a; Chiang et al. 1996; Schauerte et al. 1998; Odent et al. 1999). Later, human SHH is transcribed in the superficial lens and posterior retina during neural differentiation while it continues to be produced in the telencephalic floorplate (Bakrania et al. 2008).
As demonstrated in mice, under the influence of midline Hh activity, transcription of the evolutionarily conserved TF Pax6 normally becomes downregulated in the medial eye field, and these cells in turn transcribe Pax2 (Chiang et al. 1996). As a consequence, two lateral domains of Pax6 expression are generated. The epithelia proliferate and subsequently evaginate from the lateral walls of the forebrain to give rise to the optic pits and eventually the optic vesicles. Loss or downregulation of Hh activity interferes with these early stages of eye as well as forebrain patterning and has been associated both in humans and animal models with a defect called holoprosencephaly (HPE). This malformation has been classically defined as the failure of the forebrain to divide into two cerebral hemispheres. Clinically, this covers a phenotypic spectrum in the brain that always features ocular and related craniofacial malformations, which can range from complete anophthalmia to cyclopia to synophthalmia to microphthalmia, hypotelorism and coloboma (Wallis and Muenke 2000; Hayhurst and McConnell 2003; Gongal et al. 2011). Loss-of-function mutations in SHH can occasionally be associated with isolated coloboma along the spectrum. In one family recently described, a proband presented with unilateral iris coloboma and microcephaly but no HPE, the mother had bilateral iris coloboma, while her brother had multiple malformations compatible with unconfirmed HPE (Bruel et al. 2018).
HPE is relatively common in early embryonic life, but not always compatible with birth, by which time HPE manifests as a rare and sometimes underdiagnosed condition as a result of this phenotypic as well as genetic heterogeneity (Dubourg et al. 2016).
The complexity of Hh signaling transduction is reflected in a continuous range of pathological phenotypes
After the discovery that HPE results from loss-of-function mutations in SHH (Belloni et al. 1996; Roessler et al. 1996), subsequent research showed that HPE is genetically very heterogeneous. The direct pathway effector genes PTCH1 (Ming et al. 2002; Ribeiro et al. 2006), CDON (Bae et al. 2011), DISP (Roessler et al. 2009), GAS1 (Ribeiro et al. 2010; Pineda-Alvarez et al. 2012), and GLI2 (Roessler et al. 2003), as well as other genes also cause various forms of HPE either alone or in compound heterozygous combination (Dubourg et al. 2016; Mouden et al. 2016). Human mutations in LRP2, a non-specific co-receptor of SHH also known as megalin, can lead to Donnai–Barrow syndrome, in which large ocular globes can be associated with high myopia and partial colobomas, in contrast to Lrp2-mutant mice presenting micro/anophthalmia (Kantarci et al. 2007). One study found that two HPE microform cases with large genomic deletions included the LRP2 gene among others (Rosenfeld et al. 2010).
Newer identified components of the HH signaling pathway, such as BOC, seem to be phenotypic modifiers of HPE caused by SHH mutation. Indeed, Boc mutants in mouse do not display HPE, but the allele exacerbates HPE phenotypes when combined with mutations in Cdon or Gas1 (Zhang et al. 2011; Seppala et al. 2014). Moreover, missense BOC variants have recently been found in HPE patients, supporting a potential modifier function for BOC in human HPE (Hong et al. 2017).
Ultimately, mutations in these HPE-causing genes, with their variable effects on the early eye primordium, seem to globally dampen Shh signaling within the forebrain and ocular anlage. However, this overview may be too simplistic (Fig. 1). In addition to a balance of effector transcription factors of the Gli family, with both repressor and activator activities in specific context, there are multiple points at which the Shh pathway interacts, both as target and as modulator, with branches of other signaling pathways. These include bone morphogenetic protein (BMP), fibroblast growth factor (FGF) (Ohkubo et al. 2002) and the vitamin A metabolite, retinoic acid, where signaling to transcriptional targets affect the fates of the same tissues and cause overlapping syndromic malformation phenotypes. Indeed, BMP4 (Bakrania et al. 2008), FGF8 and its receptor FGFR1 (Dubourg et al. 2016), and STRA6, an obligate mediator of RA signal transduction (Pasutto et al. 2007; Golzio et al. 2007; Kawaguchi et al. 2007), have all been identified as responsible for syndromes comprising forebrain malformation and severe micro/anophthalmia. As for Shh itself, each phenotypic spectrum begins at subclinical. These pathways are presented in more depth in other reviews of this issue.
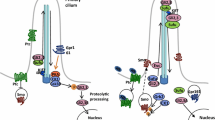
a Before binding by a Hedgehog family ligand (most often Shh, but also Ihh or Dhh), Ptch1 and presumably Ptch2 maintain Smo sequestered, by a currently unknown mechanism. Under these circumstances, both anterograde and retrograde transport in functionally intact primary cilia is necessary to allow PKA-dependent cleavage of the Gli3 transcription factor to its repressor form and its shuttling from the base of the cilium to target DNA, accompanied by Sufu. The same mechanism may govern Gli2 processing. b Hedgehog proteins are post-translationally modified; the secretion and multimeric conformation of Shh is organized by the Disp transmembrane receptor in signaling cells, accompanied by Scube. Binding of Hh to Ptch receptors leads to internalization and degradation, and relocalization of Smo to the primary ciliary membrane, which changes lipid composition. Other receptors include Boc, Cdon and Gas1, which may refine signal integration. Lrp1 is a non-specific but necessary co-receptor. Hh binding leads to ciliary clearance of the G-protein coupled negative regulatory receptor Gpr161, reduction in PKA production, and accompaniment by Sufu of immature Gli2/3 proteins along the ciliary microtubules for their requisite processing to activating forms. Gli1 is transcribed as a Shh target and positively reinforces this cycle, while Ptch1 itself is another target whose increased transcription sharpens thresholds and raises cellular stakes for survival, as continued Hh stimulation becomes necessary for the target cell. The activity of cell cycle-related kinase (Ccrk) is necessary for ciliary integrity and acts between Smo and Gli2 in effecting readout of Shh levels within the optic primordium
Transcriptional targets of Shh signaling mediate ocular morphogenesis: early stages
An important consequence of Hh signaling is the direct or indirect control of a number of TFs that establish positional identity within the ocular primordium. Comprehensive presentation of all potential effectors is beyond the scope of this review, but some important ones are mentioned here. At early stages, midline Hh influences the establishment and maintenance of identity along the proximo-distal axis, which will later translate into the difference between the ocular globe itself and the nerve and chiasm.
Transcriptional repression of the Pax6 TF and concomitant induction of Pax2 in the medial portion of the eye field by early Hh activity at the ventral midline is not only required for the splitting of the eye field, but it also has an essential role in patterning the evaginating optic vesicle along the proximal–distal (PD) and dorsoventral axis in all vertebrates studied. As the optic vesicles grow, they keep this complementary expression, so that the most proximal region keeps expressing Pax2, while the distal-most region expresses Pax6 (Macdonald et al. 1995; Schwarz et al. 2000). The Pax2+ region of the primordium remains always in direct contact with the embryonic ventral midline and maintained by Shh emanating from this region (Ekker et al. 1995b; Macdonald et al. 1995). Pax2 and Pax6, in turn, engage in a cross-repressive interaction that maintains the boundary between proximal and distal domains in the optic primordium (Schwarz et al. 2000). A recent paper shows that Shh also induces the expression of midline-1 (mid1) in the optic stalk. mid1 encodes a E3 ligase involved in the ubiquitination and proteasomal degradation of Pax6 (Pfirrmann et al. 2016). In this way, higher levels of Shh activity in the optic stalk ensure this region is depleted of residual Pax6 protein in a timely fashion. Mutations in human MID1, normally expressed throughout the early developing brain, lead to Opitz G/BBB syndrome, which is characterized by hypertelorism, oesophagolaryngotracheal defects and hypospadias and is also occasionally associated with coloboma (Halal et al. 1981; Pinson et al. 2004).
Shh is chemotactic for chick craniofacial NCC in vitro; an increasing gradient may guide NCC towards the optic stalk as well as toward the ventral prechordal plate, structures each expressing Shh at appropriate stages (Tolosa et al. 2016). This periocular mesenchyme later induces the development of the RPE and anterior chamber, further repressing expression of Pax6 and other neural retina-specific TFs in the intermediate area (Fuhrmann et al. 2000).
Gradients of Shh confer positional coordinates on cells of future retina
At a slightly earlier stage, while the proximo-distal (PD) axis of the evaginating optic vesicle is determined by midline Shh signaling, the primordium also gets patterned along the nasal/temporal (NT) and dorsal/ventral (DV) axes. Zebrafish optic vesicle evagination occurs in a very reduced window of time as compared to this process in mammals. The eye field becomes split in two domains by the same morphogenetic movements driving the lateral evagination of the optic vesicles (Rembold et al. 2006; Ivanovitch et al. 2013). Determination of PD and NT domains occurs simultaneously (Picker and Brand 2005; Picker et al. 2009). As the optic vesicle matures into an optic cup, the ventral connection of the optic vesicle with the neural tube narrows down to a small region located at the most anterior part of the eye primordium to give rise to the optic stalk, and the whole vesicle rotates anteriorly to acquire its final position, in which the NT axis is aligned with the embryonic anterior-posterior (AP) axis (Li et al. 2000). Recent studies in zebrafish have shown that the establishment of nasal and temporal coordinates occurs at the very onset of optic vesicle evagination, as early as or earlier than determination of the PD axis, under the influence of Shh (Hernandez-Bejarano et al. 2015).
Thus, at early stages in zebrafish embryos, the prospective temporal region of the optic vesicle is both continuous with the ventral midline and directly under the influence of Shh. Shh drives the expression of the temporal fate determinant TF foxd1 in this region (Hernandez-Bejarano et al. 2015). In mammals, morphogenesis of the optic cup may not involve as complex spatial rearrangements, since the NT axis seems to be specified already aligned with the AP axis of the developing embryo, in the models commonly studied. Nonetheless, it is likely that the Hh signaling pathway has a similarly determinant role in the formation of the temporal region of the mammalian eye primordium. The temporal region of the eye is specified next to prospective hypothalamic territory, a source of SHH in humans as well as other vertebrates (Odent et al. 1999), and the expression of ocular Foxd1 is first seen at optic vesicle stages in the posterior, future temporal half of the eye primordium in mouse embryos (Hatini et al. 1994; Carreres et al. 2011). Reciprocally, Foxd1 may in turn maintain Shh expression, as suggested by the phenotype in foxd1 mouse mutants, in which ventral Shh is lost, associated with ventral ocular defects (Huh et al. 1999).
PD patterning in vertebrates is also directly linked to DV fate. Like Pax2, the Vax1 and Vax2 TFs are known downstream targets of Shh expressed specifically in the optic stalk (Hallonet et al. 1999; Barbieri et al. 1999; Bertuzzi et al. 1999; Ohsaki et al. 1999; Take-uchi et al. 2003; Lupo et al. 2005; Zhao et al. 2010). Not only is optic nerve differentiation arrested by Shh insufficiency (Macdonald et al. 1997; Bertuzzi et al. 1999; Barbieri et al. 2002; Take-uchi et al. 2003), but ventral optic cup specification and optic fissure fate as well (Hallonet et al. 1999). Ultimately, consistent with its role as a SHH signaling mediator and similar to the mouse mutant phenotype, a human VAX1 mutation has been found in a patient with microphthalmia in association with cleft lip/palate (Slavotinek et al. 2012). VAX2 haploinsufficiency may also be associated with subtler ocular defects related to PD patterning and subsequent retinal dystrophy (Norgett et al. 2015).
In summary, coordination of PD, DV and NT identities is indispensable to later optic vesicle persistence and organization of the various cell types differentiating therein. Defects in any of the patterning processes during the first two months of gestation in humans result in misshaped primordia, leading to microphthalmic and coloboma phenotypes.
Patched (PTCH) receptors are key gatekeepers of Shh signal transduction
The transmembrane protein PTCH1 is both a receptor for SHH and a highly conserved transcriptional target and readout of the hedgehog pathway, from insects to mammals (Goodrich et al. 1996). This receptor works in an unusual way, since SHH binding results in its degradation, and relieves its constitutive repression of Smo, which is the critical transmembrane transducer of SHH signal into the cytoplasm (Fig. 1). Indeed, absence of Patched activity results in widespread transcriptional activation of other targets of Shh signaling.
Ptch receptors are known as “dependence receptors”—when bound, they allow cell survival but once having transduced signal, in the absence of hedgehog proteins, they cause apoptosis (Thibert et al. 2003; Fombonne et al. 2012). In humans, there are two homologues: PTCH1 on 9q22.32, and PTCH2 on 1p34.1. Unbound PTCH1 inhibits signaling through SMO by blocking its associations with intracellular proteins and subsequent phosphorylation. SMO inhibition can also be mimicked by the use of the direct SMO antagonist, cyclopamine (Chen et al. 2002). Both human and mouse cells receptive of Shh signaling, once exposed to Shh, then become dependent on it for their survival. Subsequent lack of Shh induces caspase-9 ubiquitination and subsequent programmed cell death (Fombonne et al. 2012).
By this mechanism, Shh is particularly vital for the maintenance of multipotent periocular and craniofacial NCC viability (Ahlgren and Bronner-Fraser 1999; Jeong et al. 2004; Calloni et al. 2007). In the upper jaw, epithelial Shh signals through Smo to the mesenchyme, maintaining cyclin D2 and palate cell proliferation during shelf outgrowth (Lan and Jiang 2009). This sensitivity of facial NCC to Shh availability underlies at least in part, the frequent association of cleft palate and ODA and/or HPE. The NCC mesenchyme of the first pharyngeal arch in all vertebrate embryos gives rise to both a rostral/dorsal maxillary component, precursor to the palate, and a caudal/ventral mandibular component, precursor to the lower jaw. Indirectly, SHH may also mediate the local effects of OTX2 mutations on both eye and jaw in human micro/anophthalmia with or without otocephaly and micro/agnathia (Chassaing et al. 2012).
Both homozygous knockout and overexpression of Ptch in mice lead to embryonic lethality (Goodrich et al. 1996). PTCH1 mutations and deletions have been identified in patients with a dominant form of syndromic and variable expression of HPE, comprising infrequent cleft lip/palate (Ming et al. 2002; Richieri-Costa et al. 2016). Other inactivating variants may contribute to nonsyndromic CLP (Cobourne et al. 2009). However, relative to ocular development, mutations in PTCH1 have been identified to be a major cause of human micro/anophthalmia or anterior segment dysgenesis (Chassaing et al. 2016; Richieri-Costa et al. 2016). Ptch1 continues to be expressed in the adult mouse retina (Fig. 2). Families with one type of ocular malformation can present individuals with the other and/or both, reflecting great clinical heterogeneity within this class of ocular developmental anomalies as well as relative to other craniofacial effects of such mutations.
Ptch1 mRNA expression in the adult mouse retina (as reported in Chassaing et al. 2016). Transcripts in bluish-purple are present within all cell bodies of the retina. From external to internal: scl sclera, ch choroid plexus, heavily invested with melanocytes, RPE retinal pigmented epithelium, ES external segment of photoreceptors—both rods and cones in contact with the RPE, elm the external limiting “membrane” (aligned tight junctions of photoreceptors and Müller cells), EGL external granular layer, composed of photoreceptor soma, EPL external plexiform layer, which contains the dendrites of bipolar and horizontal interneurons that integrate photoreceptor signals, IGL internal granular layer, with the cell bodies of the bipolar, horizontal and amacrine cells, IPL internal plexiform layer, with the axons of bipolar cells connected to amacrine cells and retinal ganglion cell (RGC) dendrites, RGC cell body layer, NL nerve fiber layer of RGC axons converging on the optic nerve, ILM inner limiting membrane, vitr vitreous
To make the clinical picture even more complex, somatic loss-of-function mutations in either PTCH1 or PTCH2 are among the multiple genetic causes of basal cell nevus (BCN) syndrome (Bresler et al. 2016). The result of such mutations is overactivation of Shh pathway transduction. Homozygous loss-of-function mouse embryos show dramatic ventralization of the CNS, with total repression of Pax6 expression and absent optic evaginations (Zhang et al. 2017). Ectopic and sustained Shh expression in mouse facial epithelia leads to cleft lip/palate, hypodontia and hypertelorism (Cobourne et al. 2009). PTCH1/2 mutations that have been identified in both BCN syndrome and HPE are usually missense variants and dominant, implying that additional haploinsufficiency also would be embryonic lethal in humans. A truncating, dominant germline mutation in PTCH1 was identified in one BCN syndrome patient with myelinated fibers and optic nerve anomalies in one eye and epiretinal membranes in both (Romano et al. 2011). Another patient with a truncating PTCH1 mutation was reported with both BCN syndrome and unilateral microphthalmia with cyst (Ragge et al. 2005b). For the moment, no PTCH2 mutations have been associated with ODA.
These varied effects are likely the result of the complex coordination of eye morphogenesis and the differentiation of its critical but more superficial tissues. Non-autonomous cell–cell communication between the epithelial (retinal, lens) and mesenchymal (neural crest) eye progenitors is essential for the correct induction and patterning of each, as has been examined in numerous animal models over the years. Such studies have been a source for candidate genes and pathways in human ODA pathogenesis.
Indeed, ODA resulting from interference with growth and differentiation of the optic vesicle, stalk and disc are genetically heterogeneous, and the major causative genes, PTCH1 and SOX2, account together for at most one in five micro/anophthalmia cases (Chassaing et al. 2016). SOX2 is a positive upstream regulator of SHH signaling (Zhao et al. 2012). Interestingly, nearly all the other genes that had been identified to date in severe ODA and anterior segment dysgenesis are also transcription factors, often found to work in gene regulatory networks. Another syndromic ODA gene, STRA6, is an apparent exception to this observation (Pasutto et al. 2007; Golzio et al. 2007). STRA6 encodes a ubiquitous membrane protein enabling the intracellular passage of vitamin A, the metabolic precursor to retinoic acid (RA) (Kawaguchi et al. 2007). RA, like SHH, is another extremely important developmental signaling molecule but, unlike SHH, acts directly in concert with nuclear receptors to control transcriptional regulation, including that of SHH itself (Rhinn and Dollé 2012). Teratogenic or experimentally induced changes in RA levels at early stages of embryogenesis have dramatic effects for the establishment of the AP axis of the whole organism. In the context of the murine eye, prevention of RA signaling in NCC alone, by conditional knockout of all three heterodimeric nuclear receptors necessary for its transduction, leads to frontonasal truncation, eyelid agenesis and severe microphthalmia, with loss of the ventral retina and optic nerve (Matt et al. 2008; Dupé and Pellerin 2009). Double-germline mutants in RA receptor subunits also show malformations of the anterior chamber, aphakia, congenital cataract and retinal colobomas, attributed to ectopic NCC (Mark et al. 1998). The effects of RA deficiency in the eye may be partly mediated by disrupting the integration of Shh signaling at the transcriptional level, perhaps potentiating it.
Hedgehog signaling in malformations comprising later ocular derivatives
SHH genotype–phenotype correlations in ocular developmental anomalies
At later stages of optic cup maturation, Shh is required for RPE differentiation (Zhang and Yang 2001; Perron et al. 2003; Dakubo et al. 2008), choroid fissure formation and closure, and optic nerve maturation (Dakubo et al. 2003; Morcillo et al. 2006). Continued Shh signaling from the hypothalamus is necessary for optic disc maturation, the site through which retinal ganglion cell axons project into the optic nerve. Reduced signaling from this specific source, as engineered by conditionally driving Shh inactivation with a Cre recombinase under control of a hypothalamic-specific Shh regulatory element, leads to hypoplastic optic nerve in mouse models (Zhao et al. 2012).
Most mutations in SHH associated with HPE cases are not in the C-terminal region. However, these are also phenotypically heterogeneous or show incomplete penetrance within families. HPE-causative mutations in SHH, including in the C-terminal region, as well as mutations in other pathway effectors can also lead to more subtle defects not always diagnosed in addition to uveal and chorioretinal coloboma and microphthalmia, including refraction errors induced by myopia, retinal thinning, malposition of the optic nerve, and borderline microcornea (Pineda-Alvarez et al. 2011).
Occasionally, mutations in SHH have been reported in milder, isolated ODA cases presenting microphthalmia and/or coloboma (Schimmenti et al. 2003; Bakrania et al. 2010). During post-translational processing, the hedgehog protein precursor undergoes autocatalytic internal cleavage, yielding an active N-terminal domain covalently attached to cholesterol and palmitate, and a C-terminal domain necessary for that attachment (Porter et al. 1996). The mutations found in these isolated ODA patients lead to the deletion of eight amino acids in the C-terminal autocatalytic domain of SHH and may interfere with lipid attachment. The effect is incompletely penetrant: of family members in both these reports who were carrying such mutations, some were entirely affected, others had unilateral or partial colobomas, while one proband had bilateral microphthalmia in addition to severe uveoretinal coloboma. Defective lipid attachment is likely to have dramatic consequences for SHH signaling. Cholesterol annexation is essential for a fully functional Shh molecule and establishing tissue-appropriate titers (e.g., Huang et al. 2007; Dessaud et al. 2008). Recent reports suggest that cholesterol may be able to also modulate the activity of the pathway by directly binding to a hydrophobic groove in the extracellular portion of Smo (Luchetti et al. 2016; Byrne et al. 2018). Smith–Lemli–Opitz syndrome (SLOS), in which cataracts and blepharoptosis are frequent among a wide constellation of congenital anomalies attributed to reduced SHH signaling, is caused by mutations in DHCR7, which encodes a rate-limiting enzyme in the cholesterol biosynthesis pathway (Wassif et al. 1998; Fitzky et al. 1998). The subsequent cholesterol deficit in SLOS impairs Smo translocation within the lipid membrane to the primary cilium (Blassberg et al. 2016), as discussed further below.
It seems likely that pathway effector mutations in humans will increasingly be found to be pathogenic concerning later outcomes of maturation of specific tissue components of the eye. Indeed, many ciliopathies discussed briefly below, may be imputed in part to such roles for Shh signaling.
DHH and IHH may affect later corneal and retinal maintenance
Other Hedgehog family members in humans include Desert hedgehog (DHH, on 12q13.12) and Indian hedgehog (IHH, on 2q35). Although Dhh has been reported to be the major ligand present and involved with corneal homeostasis in the adult mouse, it has not to date been implicated in human ODA or later ocular phenotypes (Kucerova et al. 2012). Mouse null mutants in Ihh have no reported ocular defects, although they die shortly after birth with numerous skeletal anomalies, mildly foreshortened snout and mandible and a rounded skull (St-Jacques et al. 1999). Pathogenic IHH mutations in humans, consistent with these important roles demonstrated in endochondral ossification, tend to be missense mutations causing acrocapitofemoral dysplasia or brachydactyly type A1, but do not affect the eye (Byrnes et al. 2009). However, in addition to Shh, both Dhh and Ihh are expressed in the postnatal rat retina. Ihh secretion by choroidal endothelial cells impacts both the formation of the RPE inside, and the sclera outside, of this meningeal-like layer (Dakubo et al. 2008). We predict that future functional as well as malformative roles of other hedgehog signaling ligands will be discovered in human ocular pathology.
DISP1 mutations indirectly affect the eyes
DISP1 (dispatched Resistance Nodulation Division transporter family member 1), in contrast to most other molecules reviewed here that are present in target tissues, encodes a protein needed for the secretion of the mature SHH peptide (Tian et al. 2008; Tukachinsky et al. 2012, Fig. 1). Mouse Disp mutations lead to HPE and misplaced early eye primordia (Tian et al. 2008). However, human mutations in DISP1 are rarely diagnosed in HPE and have been reported in microforms with incomplete penetrance, at the level of the eyes showing hypotelorism and upslanting palpebral fissures (Roessler et al. 2009); other patients with compound heterozygous DISP1 mutations sometimes in combination with other HPE gene mutations, tend to only have hypotelorism in conjunction with lobar or microform HPE (Dubourg et al. 2016; Dupé et al. 2017).
It is interesting that patients carrying deletions of the 1q41q42 region including DISP1, have rather opposite symptoms, again affecting tissues around the eyes—hypertelorism, deep-set eyes, some effects on the palpebral fissures, and frontal bossing, but no clinical HPE (Shaffer et al. 2007). Such signs, evocative of a subset of BCN syndrome symptoms, are the Shh hyperactivation counterpart to the cranial midline hypoplasias seen in HPEs. They may reflect the fact that genomic regulatory elements are lost as well as DISP1 haploinsufficiency or paradoxical effects on the repressor/activator balance of effector transcription factors of the GLI family. Ultimately, although implicated in human ocular pathology, DISP1 mutations and truncations affect SHH signal transduction in an incomplete manner and do not phenocopy even partial SHH loss-of-function.
GLI proteins are context-dependent transcriptional activators and repressors
The three members of the GLI family of proteins (glioma-associated oncogene family zinc finger; homologous to the single Drosophila segment polarity protein Ci, for “cubitus interruptus”) are the direct transcriptional effectors of Shh signaling. The three homologues in humans, GLI1, GLI2 and GLI3, are on chromosomes 12q13.3, 2q14.2, and 7p14.1, respectively. These TFs act in both repressor and activator forms, post-transcriptionally dependent on Hh signaling activity. This dual role was first established in Drosophila (Méthot and Basler 1999). As an activator (in response to Hh), Ci directly controls ptc transcription, and this role is evolutionarily conserved through to mammals—Ptch1 is often used as a readout of Shh activity (Goodrich et al. 1996).
GLI3 has been particularly well-studied. In mice, Gli3 is normally expressed in the optic stalk. Gli3 knockout mutants develop microphthalmia, with reduction in the Pax6-expressing distal domain of the optic vesicle, and concomitant upregulation of Pax2 and Vax1/2 TFs in the stalk, which thickens. Otx2 expression in the ventral optic cup is also reduced secondarily (Aoto et al. 2002). Interestingly, these effects phenocopy the effects of augmenting Shh signaling in the optic primordium and translate the loss of the Gli3 repressor form in particular (Gli3-R).
However, the activator form of GLI3 is also necessary for human health, though not implicated specifically in eye formation. Pallister–Hall syndrome (PHS) is a rare, often lethal condition comprising massive benign hamartomic tumors of the hypothalamus, effects on the pituitary gland and subsequent adrenal function, and effects on digit numbers (polydactyly or syndactyly). Such effects on digits had been anticipated in experimental embryology by directly or indirectly manipulating Shh levels in the outgrowing limb (Niswander et al. 1994), many years before the other signal transducers in the cascade had been identified, much less in human pathology. The dominant truncating mutations in GLI3 that cause PHS result in constitutive availability of the GLI3-R form, and a consequent imbalance relative to its activator form (Démurger et al. 2015, Fig. 1). However, other mutations, including large deletions, leading to GLI3 haploinsufficiency are associated with the distinct Grieg syndrome, also including poly/syndactyly, but characterized by different craniofacial abnormalities, such as macrocephaly (occasionally associated with macrosomia) and hypertelorism. Further abrogation of GLI3 may be embryonic lethal even earlier in humans, such that its potential ocular phenotype would not have been reported.
A major gene of Axenfeld–Rieger syndrome responsible for anterior segment dysgenesis and congenital glaucoma when mutated, the FOXC1 transcription factor (Nishimura et al. 1998), is a known SHH signaling target in the periocular mesenchyme. Recently, it has been found that FOXC1 directly activates the transcription of GLI2-mediated SHH targets in human basal-like breast cancer cells by binding and acting as a co-factor to GLI2, bypassing SMO (Han et al. 2015). This is but one example that the contextual interplay between the many effector TFs directly and indirectly integrating SHH signaling effects on eye morphogenesis merits further work.
Mutations in SMO, the critical effector of Shh signal transduction, can have repercussions for the anterior chamber
Smoothened (Smo) is a G protein-coupled receptor that partners intracellularly with scaffolding proteins such as Suppressor of Fused (Sufu, discussed below) to relieve the constitutive processing of Gli TFs into their N-terminal truncated repressor forms, and allow their transcriptional activator forms to predominate. Cyclopamine is a historic and well-known Hh signaling antagonist acting on SMO, which is initially bound and repressed by PTCH1 at the cell membrane (Taipale et al. 2000). The identification of cyclopamine as the teratogenic mediator of cyclopia and severe alobar HPE in pregnant animals grazing on Veratrum californicum (false hellebore or corn lily) dates to the mid-twentieth century (James 1999).
A few patients with presumably heterozygous deletions comprising the SMO locus at 7q32.3 have been reported. These mutations did not lead to even minor forms of HPE, but their clinical spectrum was not further delineated (Rosenfeld et al. 2010). Of the nine patients reported with heterozygous deletions including the SMO locus in the DECIPHER database (https://decipher.sanger.ac.uk), seven have no ocular symptoms aside from strabismus, while patient 258,033 with a nearly 7-Mb deletion comprising 74 genes has an epibulbar dermoid tumor, proptosis, and an eyelid coloboma, and patient 261,321, with a larger deletion yet, is reported simply with abnormal scleral morphology. Thus, SMO haploinsufficiency does not clearly affect the human eye or brain, reflecting possible rescue by the remaining allele.
Murine Smo null (homozygous) mutants also do not demonstrate HPE, but they do have ocular defects that correlate with mispatterning of the DV axis, reminiscent of loss of the Gli2 or Otx2 effectors of Shh signaling. In these mutants, Bmp4, usually expressed in the dorsal portion of the developing eye, expands ventrally, accompanied by reduction in transcription of Vax2 and Pax2 transcription factors and subsequent impairment of ventral eye formation (Zhao et al. 2010). This antagonistic relationship between Bmp4 and Shh in patterning the DV axis of the ocular primordium has also been described in Xenopus (Sasagawa et al. 2002) and chick (Ohkubo et al. 2002; Kobayashi et al. 2010).
Shh-mediated signaling, by binding Ptch, frees up Smo for actual transduction. In vertebrates, this involves Smo modification by cholesterol and relocalization to the sensory organelle, the primary cilium (Fig. 1; reviewed by Briscoe and Thérond 2013; Byrne et al. 2018). This process remains incompletely understood to date. In Drosophila, activated Smo liberates the heterotrimeric G protein subunit, Gαi, decreasing levels of adenylate cyclase-mediated PKA and attenuating Ci cleavage to the repressor form (Ogden et al. 2008). However, Smo may also affect Gli-independent, Shh-induced cellular responses, in the light of conflicting data concerning the inhibitory effects of Gαi activation on PKA (Pandit and Ogden 2017). This may in part explain the chemotaxis displayed by some cranial NCC in the presence of Shh (Tolosa et al. 2016) as well as the very heterogeneous phenotypes in the presence of mosaic, constitutively activating mutations in SMO. Depending on the lineages targeted, these can cause Curry–Jones syndrome, which includes iris colobomas and microphthalmia (Twigg et al. 2016), while within the epidermis in postnatal somatic mutation, the same hotspot mutations are found in basal cell carcinomas (Xie et al. 1998).
CDON, GAS1 and BOC
CDON, GAS1 and BOC are transmembrane proteins that act as co-receptors for SHH and are involved in the modulation of cellular response to SHH activity. Cdon complexes with many proteins, including Ptch1 but also Boc (“brother of CDON”), through its fibronectin domains. Missense substitutions in CDON induce HPE by continuing to bind SHH but not PTCH1, thereby sequestering and diminishing the perceived levels of SHH in target cells (Bae et al. 2011). These studies indicate a role for CDON in mammals as a positive modulator of the Hh pathway. However, cdon may also have a role as a negative modulator of the pathway. In zebrafish and chick embryos, unmutated Cdon has been shown to work as a decoy by sequestering, and thereby attenuating, Shh activity. Consistently, downregulation of cdon leads to an expansion of optic stalk fate at the expense of distal optic vesicle fates (Cardozo et al. 2014).
BOC has only recently been identified as a true HPE modifier gene in concert with SHH mutations (Hong et al. 2017). GAS1 is a GPI-linked co-receptor that also interacts directly with SHH in ways that remain biochemically unclear; missense heterozygous mutations are an uncommon cause of HPE along the entire phenotypic spectrum (Ribeiro et al. 2010; Pineda-Alvarez et al. 2012).
In mice homozygous null for Gas1, presenting a mild HPE midline phenotype, it has been shown that additional loss of Boc alleles plays a modifier role that demonstrates that many of the pleiotropic effects of Shh signaling are mediated by combinatorial and sometimes unique transduction through these receptors (Seppala et al. 2014). These may also be modulated by the non-specific Lrp2/megalin co-receptor. A recent review addresses this newly uncovered complexity in Shh signaling modulation (Xavier et al. 2016), although to date, no clear phenotype–genotype correlations have been drawn in animal models or human genetics between a particular receptor mutation subtype or combination, and the outcome with respect to HPE or ODA.
Ciliopathies and Shh signal transduction
Cilia are specialized organelles present on nearly all cell types and which play a particularly important role in the organization and function of ocular tissues such as the RPE throughout life. They are also necessary for signal transduction by Shh in vertebrates (Briscoe and Thérond 2013). As such, many syndromic ciliopathies, such as Joubert syndrome and associated disorders, show disruption of processes in ocular formation that depend on intact signaling mediated by cilia (Khan et al. 2008; Valente et al. 2008). The many complex roles of cilia in ocular physiology and homeostasis are the subject of a complete and recent review in itself (May-Simera et al. 2017).
In short, Ptch binding of Shh leads to its replacement in the ciliary membrane by Smo (Rohatgi et al. 2007) and subsequent action on Gli subcellular localization (cf. Fig. 1). This mobilization is carried out specifically by the Sufu protein, in concert with kinesins and intraflagellar transport (IFT) proteins that are more generally critical for ciliary function. Although ancestral Hh signaling may not have required a primary cilium, transduction through ciliary Smo is also conserved in Drosophila olfactory neurons (Kuzhandaivel et al. 2014).
Suppressor of fused (SUFU) is a key molecular chaperone of Gli effector TFs
SUFU is a molecular chaperone protein critical for the ciliary trafficking and processing of Gli proteins. To regulate its localization, it can be phosphorylated by protein kinase A (PKA) or glycogen synthase kinase 3 (GSK3). Mutations in SUFU are a rarer cause of BCN syndrome, along with mutations in PTCH2 or the principal gene PTCH1 (Johnson et al. 1996), compatible with a normal role in modulating and restraining the effects of SHH signaling. High-throughput sequencing in human patients suggests that variants in additional SHH pathway genes may also contribute to BCN syndrome phenotypes (Onodera et al. 2017). Ocular anomalies, a minor diagnostic criterion, may be under-reported but include congenital cataract, coloboma and glaucoma—while further emerging reports of somatic mosaicism in any of the BCN causative genes, may yet prove to be the cause of some apparently de novo ODA.
In the absence of SHH, SUFU constitutively sequesters GLI2/3 proteins at the base of ciliary microtubules. Translocation of this complex to the tips of cilia then promotes Gli TF cleavage into repressor forms, and Sufu has recently been shown to accompany these to the nucleus as far as the chromatin itself. In the presence of SHH, SUFU instead traffics GLI1/2/3 activator forms into the nucleus and the GLI2/3 repressor forms out (Zhang 2017).
Examples of kinesins crucial for SHH signal transduction
The kinesins are a superfamily of molecular motors that hydrolyse ATP to translocate cargoes along the microtubules of primary cilia, towards the tip. Mutations affecting intraflagellar transport and kinesin proteins can cause ocular ciliopathies like retinitis pigmentosa (RP) by preventing the transport of proteins necessary for photoreceptor development, function and survival. They also can cause misregulation of Shh signal transduction by playing havoc with the trafficking of Gli TFs (reviewed in Bisgrove and Yost 2006).
Structural organization of the tip of cilia by the kinesin KIF7 (He et al. 2014) is critical for association with the small GTPase ARL3 and the GTPase-activating protein, Retinitis Pigmentosa 2 (RP2), mutations of which, as its name implies, cause an X-linked form of RP (Schwarz et al. 2017). Certain missense mutations in KIF7 (Dafinger et al. 2011), and more infrequently, ARL3 (Strom et al. 2016) and other proteins in this complex, as well as SUFU itself (De Mori et al. 2017), are all causes of Joubert syndrome. Early reports of this syndrome and the related but more severe Meckel syndrome, including fetal phenotyping, have shown multiple associations with coloboma and micro/anophthalmia (Delous et al. 2007; reviewed by; Valente et al. 2008; May-Simera et al. 2017).
However, even fetuses affected with the severe hydrolethalus syndrome, due to KIF7 mutation, are much more frequently affected by brain malformations to the point of anencephaly, and by hypertelorism, than ODA, although one case was described with bilateral optic atrophy (Putoux et al. 2011). Indeed, the very KIF7 mutations identified in these severe cases lead to upregulation of dozens of direct and indirect SHH signaling targets, through augmenting the ratio of activator to repressor forms of GLI3 protein (Putoux 2011). Similarly, Kif3a mutations in mouse NCC lead to complete loss of their cilia, increased proliferation and Gli1 expression, and thereby phenocopy the effects of excessive Shh signaling on the forming face (Brugmann et al. 2010). This leads to hypertelorism and frontonasal dysplasia but have no major effects on eye formation before E12.5, by which time retinal ganglion cells are extending axons into the optic nerve, the corneal mesenchyme is in place, the lens is differentiating and the RPE beginning to pigment. Many other mutations in genes for ciliary proteins have a similar effect on craniofacial mesenchyme and are associated with hypertelorism but not with ODA as such.
Ciliary shape and integrity determines Shh signaling efficacy
A number of recent studies in mice show a requirement for IFT proteins in controlling eye patterning through the modulation of Hh activity. For example, Ift172 is required for anterograde ciliary transport (toward the tip) while Ift122 is required for retrograde transport (toward the base). Mouse mutants in Ift172 do not produce cilia, while loss of Ift122 leads to bulbous cilia, where cargo accumulates at the tip of these structures (Burnett et al. 2017; Fig. 1). These mutants also show defective patterning of the optic primordium along the proximo-distal axis, suggesting perturbed Shh signaling.
Indeed, Ift172−/− optic cups show expansion of Pax2 and Sox1 expression at the expense of Pax6, which is reduced to a small patch at the distal-most region of the primordium, indicating relative reduction in optic cup- and expansion of optic stalk-fated territories. Likewise, expression of the TFs Mitf and Otx2, markers of the RPE, abnormally extends into the inner layer of the optic cup, with a concomitant reduction in the NR-specific TF Chx10, indicating that loss of Ift-mediated Shh signaling leads to expansion of proximal RPE fate at the expense of the more distal NR fate. Curiously, Ift122−/− optic cups show a more severe phenotype, with no invagination of the optic vesicle at all. Pax2 and Sox1 are dramatically expanded throughout the mutant optic vesicle, while Pax6 expression is virtually absent, and neither RPE nor NR is specified at all. Thus, Ift122-negative optic primordia adopt a full optic stalk fate. The phenotypes observed in Ift177 and Ift122 mutant embryos suggest overactivation of the HH pathway under conditions in which cilia formation is compromised, correlated in phenotypes with the extent of stimulation. Astonishingly, the concomitant knockout of Ift122 and Gli2 can partially rescue optic cup patterning and morphogenesis (Burnett et al. 2017).
We can hypothesize that overexpressing PHS-causing truncated forms of Gli3-R would make for a fascinating demonstration that some ciliary production of Gli3-R is necessary to counter baseline Gli2 activity, perhaps even in the absence of Shh as facilitated by Foxc1 and perhaps other co-factors. Altogether, defective cilia formation in these IFT mutants seems to lead to an increased proportion of activating to repressor forms of Gli TFs, mimicking constitutive signaling from the Hh pathway, and thereby affecting proximo-distal cell identity in the optic vesicle and its derivatives.
Activating intracellular cAMP (as mimicked by phorbol esters) and thereby, protein kinase A (PKA) early in development to further the proteolytic processing of Gli repressor forms may also counter these effects (Lauth et al. 2007). Application to human malformations can only be speculative concerning the use of autologous induced pluripotent cells for tissue reconstruction. Accordingly, since the Ptch-expressing RPE depends on Shh signaling via Otx2 and Mitf induction, augmenting cAMP prevents RPE cell proliferation (Hecquet et al. 2002; Fig. 2).
The mutation of a lipid-modifying enzyme encoded by the INPP5E gene induces a form of Joubert syndrome (Bielas et al. 2009). INPP5E is an evolutionarily divergent paralog of the X-linked OCRL gene, which encodes another inositol polyphosphate phosphatase whose mutation can cause either the oculocerebrorenal syndrome of Lowe or a more restricted renal tubulopathy (Zhang et al. 1995). Although OCRL haploinsufficiency is frequently associated with congenital cataracts and glaucoma, these do not overlap with the retinitis pigmentosa or other ocular signs sometimes found in Joubert patients. However, INPP5E has been shown to compartmentalize certain lipids in the ciliary membrane, thereby regulating the concentration of an inhibitory transmembrane receptor known as GPR161 that directly augments cAMP content in the cilia and, thereby, prevents Shh-dependent GLI1 activity (Chávez et al. 2015). INPP5E-mutated primary cilia are shorter (Luo et al. 2012), indirectly accumulate too much GPR161 and constitutively repress at least some aspects of SHH signaling. In contrast, OCRL-mutated primary cilia can vary in length, depending on the model system studied, and are deficient in trafficking from endosomes to the ciliary membrane, thereby potentially affecting the levels of the non-specific SHH co-receptor, LRP2, or otherwise interfering with other signaling processes mediated by the primary cilium (Rbaibi et al. 2012).
Recently published work demonstrates that Ccrk (cell cycle-related kinase)-null mouse mutants show abnormal, shorter cilia, and display a number of corresponding developmental defects, amongst which microphthalmia due to fully penetrant coloboma and frequent loss of the lens primordium (Lupu et al. 2018). A result of disrupting Shh-dependent proximo-distal patterning at optic cup stages, malformations and effects on target gene transcription induced by mutant Ccrk confirm the hypothesis that different Shh activity readout levels lead to different ocular fates. Ccrk is required in regions of high levels of HH activity (proximal) to promote pathway activation, but it is simultaneously required in regions of low HH activity (distal) to maintain the pathway in a repressed state (Fig. 1). In Ccrk knockouts, the Gli1 expression domain of the distal optic vesicle is expanded and Mitf is ectopically expressed within the optic cup, promoting a RPE fate. Furthermore, Ccrk mutation rescues the effect of Smo mutants, in which Pax6 is expressed throughout the entire optic vesicle following loss of Shh signaling, demonstrating that Ccrk acts downstream of Smo. The analysis of both Ccrk−/−;Gli2−/− double mutants showed compensation for the excess distal Shh signaling activity mediated by the loss of Ccrk in terms of ocular vesicle morphology, although at the expense of ventral and midline tissues with normally highest exposure to Shh. Likewise, Ccrk−/−;Gli3−/− double mutants show exacerbation of the distal RPE phenotype at the expense of neural retina fate, and total loss of lens specification (Lupu et al. 2018), supporting the idea that such effects result from loss of the Gli3-R TF form and expansion of midline fates at the expense of distal tissues.
Conclusions
The vertebrate eye is a highly complex structure whose assembly is coordinated over time from four distinct sources of embryological cells. Hedgehog signaling plays a role from the very first stages of specification of the neural epithelium, to determining the eye fields, to the maintenance of healthy functional ocular tissues in the adult organism. Over the last 25 years, the number of molecular participants in this pathway has grown exponentially, as have demonstrations that changes in their activity or availability cause human ocular disease. The primary cilium has been shown to be the principal organelle necessary for hedgehog signal reception and integration. The presence of the cilium is cell cycle-dependent, implying that Hh activity may also be cell cycle-dependent, adding one more level of complexity to the modulation of cellular functions by this signaling pathway. A “rheostat” model, first used to describe the gradation in signaling levels of the TF Mitf and in opposition to a binary on–off switch model (Carreira et al. 2006), best describes the continuous transcriptional integration of Gli and other TF at the level of Shh target gene chromatin. Perhaps we should consider the “trimpot” (trimmer potentiometer) as a more apt metaphor for the variable transcriptional effects of Shh signaling, as the adjustment periods for reactivity to thresholds are limited during development. These fascinating mechanisms will continue to surprise and inform our understanding of human pathology, as additional biological circuitry is explored and brought to light in the future.
References
Acampora D, Mazan S, Lallemand Y et al (1995) Forebrain and midbrain regions are deleted in Otx2−/− mutants due to a defective anterior neuroectoderm specification during gastrulation. Development 121:3279–3290
Aguiar DP, Sghari S, Creuzet S (2014) The facial neural crest controls fore- and midbrain patterning by regulating Foxg1 expression through Smad1 activity. Development 141:2494–2505. https://doi.org/10.1242/dev.101790
Ahlgren SC, Bronner-Fraser M (1999) Inhibition of sonic hedgehog signaling in vivo results in craniofacial neural crest cell death. Curr Biol 9:1304–1314
Alsaif HS, Khan AO, Patel N et al (2018) Congenital glaucoma and CYP1B1: an old story revisited. Hum Genet. https://doi.org/10.1007/s00439-018-1878-z
Aoto K, Trainor PA (2015) Co-ordinated brain and craniofacial development depend upon Patched1/XIAP regulation of cell survival. Hum Mol Genet 24:698–713. https://doi.org/10.1093/hmg/ddu489
Aoto K, Nishimura T, Eto K, Motoyama J (2002) Mouse GLI3 regulates Fgf8 expression and apoptosis in the developing neural tube, face, and limb bud. Dev Biol 251:320–332. https://doi.org/10.1006/dbio.2002.0811
Bae GU, Domene S, Roessler E et al (2011) Mutations in CDON, encoding a hedgehog receptor, result in holoprosencephaly and defective interactions with other hedgehog receptors. Am J Hum Genet 89:231–240. https://doi.org/10.1016/j.ajhg.2011.07.001
Bakrania P, Efthymiou M, Klein JC et al (2008) Mutations in BMP4 cause eye, brain, and digit developmental anomalies: overlap between the BMP4 and Hedgehog signaling pathways. Am J Hum Genet 82:304–319. https://doi.org/10.1016/j.ajhg.2007.09.023
Bakrania P, Ugur Iseri SA, Wyatt AW et al (2010) Sonic hedgehog mutations are an uncommon cause of developmental eye anomalies. Am J Med Genet A 152A:1310–1313. https://doi.org/10.1002/ajmg.a.33239
Barbieri AM, Lupo G, Bulfone A et al (1999) A homeobox gene, vax2, controls the patterning of the eye dorsoventral axis. Proc Natl Acad Sci USA 96:10729–10734
Barbieri AM, Broccoli V, Bovolenta P et al (2002) Vax2 inactivation in mouse determines alteration of the eye dorsal-ventral axis, misrouting of the optic fibres and eye coloboma. Development 129:805–813
Barishak YR (1992) Embryology of the eye and its adnexae. Dev Ophthalmol 24:1–142
Beccari L, Marco-Ferreres R, Bovolenta P (2013) The logic of gene regulatory networks in early vertebrate forebrain patterning. Mech Dev 130:95–111. https://doi.org/10.1016/j.mod.2012.10.004
Belloni E, Muenke M, Roessler E et al (1996) Identification of Sonic hedgehog as a candidate gene responsible for holoprosencephaly. Nat Genet 14:353–356. https://doi.org/10.1038/ng1196-353
Bertuzzi S, Hindges R, Mui SH et al (1999) The homeodomain protein vax1 is required for axon guidance and major tract formation in the developing forebrain. Genes Dev 13:3092–3105
Bielas SL, Silhavy JL, Brancati F et al (2009) Mutations in INPP5E, encoding inositol polyphosphate-5-phosphatase E, link phosphatidyl inositol signaling to the ciliopathies. Nat Genet 41:1032–1036. https://doi.org/10.1038/ng.423
Bisgrove BW, Yost HJ (2006) The roles of cilia in developmental disorders and disease. Development 133:4131–4143. https://doi.org/10.1242/dev.02595
Blassberg R, Macrae JI, Briscoe J, Jacob J (2016) Reduced cholesterol levels impair smoothened activation in Smith–Lemli–Opitz syndrome. Hum Mol Genet 25:693–705. https://doi.org/10.1093/hmg/ddv507
Bresler SC, Padwa BL, Granter SR (2016) Nevoid basal cell carcinoma syndrome (Gorlin syndrome). Head Neck Pathol 10:119–124. https://doi.org/10.1007/s12105-016-0706-9
Briscoe J, Thérond PP (2013) The mechanisms of Hedgehog signalling and its roles in development and disease. Nat Rev Mol Cell Biol 14:416–429. https://doi.org/10.1038/nrm3598
Bruel AL, Thevenon J, Huet F et al (2018) Unexpected diagnosis of a SHH nonsense variant causing a variable phenotype ranging from familial coloboma and intellectual disability to isolated microcephaly. Clin Genet. https://doi.org/10.1111/cge.13211
Brugmann SA, Allen NC, James AW et al (2010) A primary cilia-dependent etiology for midline facial disorders. Hum Mol Genet 19:1577–1592. https://doi.org/10.1093/hmg/ddq030
Burnett JB, Lupu FI, Eggenschwiler JT (2017) Proper ciliary assembly is critical for restricting Hedgehog signaling during early eye development in mice. Dev Biol 430:32–40. https://doi.org/10.1016/j.ydbio.2017.07.012
Byrne EF, Luchetti G, Rohatgi R, Siebold C (2018) Multiple ligand binding sites regulate the Hedgehog signal transducer smoothened in vertebrates. Curr Opin Cell Biol 51:81–88. https://doi.org/10.1016/j.ceb.2017.10.004
Byrnes AM, Racacho L, Grimsey A et al (2009) Brachydactyly A-1 mutations restricted to the central region of the N-terminal active fragment of Indian Hedgehog. Eur J Hum Genet 17:1112–1120. https://doi.org/10.1038/ejhg.2009.18
Calloni GW, Glavieux-Pardanaud C, Le Douarin NM, Dupin E (2007) Sonic Hedgehog promotes the development of multipotent neural crest progenitors endowed with both mesenchymal and neural potentials. Proc Natl Acad Sci 104:19879–19884. https://doi.org/10.1073/pnas.0708806104
Canto-Soler MV, Adler R (2006) Optic cup and lens development requires Pax6 expression in the early optic vesicle during a narrow time window. Dev Biol 294:119–132. https://doi.org/10.1016/j.ydbio.2006.02.033
Cardozo MJ, Sanchez-Arrones L, Sandonis A et al (2014) Cdon acts as a Hedgehog decoy receptor during proximal-distal patterning of the optic vesicle. Nat Commun 5:4272. https://doi.org/10.1038/ncomms5272
Carreira S, Goodall J, Denat L et al (2006) Mitf regulation of Dia1 controls melanoma proliferation and invasiveness. Genes Dev 20:3426–3439. https://doi.org/10.1101/gad.406406
Carreres MI, Escalante A, Murillo B et al (2011) Transcription factor Foxd1 is required for the specification of the temporal retina in mammals. J Neurosci 31:5673–5681. https://doi.org/10.1523/JNEUROSCI.0394-11.2011
Chassaing N, Golzio C, Odent S et al (2009) Phenotypic spectrum of STRA6 mutations: from Matthew–Wood syndrome to non-lethal anophthalmia. Hum Mutat 30:E673–E681. https://doi.org/10.1002/humu.21023
Chassaing N, Sorrentino S, Davis EE et al (2012) OTX2 mutations contribute to the otocephaly–dysgnathia complex. J Med Genet 49:373–379. https://doi.org/10.1136/jmedgenet-2012-100892
Chassaing N, Davis EE, McKnight KL et al (2016) Targeted resequencing identifies PTCH1 as a major contributor to ocular developmental anomalies and extends the SOX2 regulatory network. Genome Res 26:474–485. https://doi.org/10.1101/gr.196048.115
Chávez M, Ena S, Van Sande J et al (2015) Modulation of ciliary phosphoinositide content regulates trafficking and sonic Hedgehog signaling output. Dev Cell 34:338–350. https://doi.org/10.1016/j.devcel.2015.06.016
Chen JK, Taipale J, Cooper MK, Beachy PA (2002) Inhibition of Hedgehog signaling by direct binding of cyclopamine to smoothened. Genes Dev 16:2743–2748. https://doi.org/10.1101/gad.1025302
Chiang C, Litingtung Y, Lee E et al (1996) Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature 383:407–413. https://doi.org/10.1038/383407a0
Cobourne MT, Xavier GM, Depew M et al (2009) Sonic hedgehog signalling inhibits palatogenesis and arrests tooth development in a mouse model of the nevoid basal cell carcinoma syndrome. Dev Biol 331:38–49. https://doi.org/10.1016/j.ydbio.2009.04.021
Cook CS, Sulik KK, Wright KW (2006) Embryology. In: Wright KW, Spiegel PH, Thompson LS (eds) Handbook of pediatric eye and systemic disease. Springer, New York, pp 1–61
Couly GF, Coltey PM, Le Douarin NM (1993) The triple origin of skull in higher vertebrates: a study in quail-chick chimeras. Development 117:409–429
Creuzet SE, Etchevers HC (2017) Embryologie de l’œil. In: Denis D, Qui Buoc E, Aziz Alessi A (eds) Ophtalmologie pédiatrique: Rapport SFO 2017, 1st edn. Elsevier-Masson, Paris, pp 739–756. https://doi.org/10.6084/m9.figshare.4983362.v1
Creuzet S, Vincent C, Couly G (2005) Neural crest derivatives in ocular and periocular structures. Int J Dev Biol 49:161–171. https://doi.org/10.1387/ijdb.041937sc
Cvekl A, Duncan MK (2007) Genetic and epigenetic mechanisms of gene regulation during lens development. Prog Retin Eye Res 26:555–597. https://doi.org/10.1016/j.preteyeres.2007.07.002
Dafinger C, Liebau MC, Elsayed SM et al (2011) Mutations in KIF7 link Joubert syndrome with Sonic Hedgehog signaling and microtubule dynamics. J Clin Investig 121:2662–2667. https://doi.org/10.1172/JCI43639
Dakubo GD, Wang YP, Mazerolle C et al (2003) Retinal ganglion cell-derived sonic hedgehog signaling is required for optic disc and stalk neuroepithelial cell development. Development 130:2967–2980
Dakubo GD, Mazerolle C, Furimsky M et al (2008) Indian hedgehog signaling from endothelial cells is required for sclera and retinal pigment epithelium development in the mouse eye. Dev Biol 320:242–255. https://doi.org/10.1016/j.ydbio.2008.05.528
De Mori R, Romani M, D’Arrigo S et al (2017) Hypomorphic recessive variants in SUFU impair the Sonic Hedgehog pathway and cause Joubert syndrome with cranio-facial and skeletal defects. Am J Hum Genet 101:552–563. https://doi.org/10.1016/j.ajhg.2017.08.017
Delous M, Baala L, Salomon R et al (2007) The ciliary gene RPGRIP1L is mutated in cerebello-oculo-renal syndrome (Joubert syndrome type B) and Meckel syndrome. Nat Genet 39:875–881. https://doi.org/10.1038/ng2039
Démurger F, Ichkou A, Mougou-Zerelli S et al (2015) New insights into genotype–phenotype correlation for GLI3 mutations. Eur J Hum Genet 23:92–102. https://doi.org/10.1038/ejhg.2014.62
DeMyer W, Zeman W, Palmer C (1964) The face predicts the brain: Diagnostic significance of median facial anomalies for holoprosencephaly (arhinencephaly). Pediatrics 34:256
Dessaud E, McMahon AP, Briscoe J (2008) Pattern formation in the vertebrate neural tube: a sonic hedgehog morphogen-regulated transcriptional network. Development 135:2489–2503. https://doi.org/10.1242/dev.009324
Dubourg C, Carré W, Hamdi-Rozé H et al (2016) Mutational spectrum in holoprosencephaly shows that FGF is a new major signaling pathway. Hum Mutat 37:1329–1339. https://doi.org/10.1002/humu.23038
Dupé V, Pellerin I (2009) Retinoic acid receptors exhibit cell-autonomous functions in cranial neural crest cells. Dev Dyn 238:2701–2711. https://doi.org/10.1002/dvdy.22087
Dupé V, Dubourg C, Tayrac M, De David V (2017) Un nouveau regard sur la génétique de l’holoprosencéphalie. Médecine/sciences 33:924–926
Dyer LA, Kirby ML (2009) Sonic hedgehog maintains proliferation in secondary heart field progenitors and is required for normal arterial pole formation. Dev Biol 330:305–317. https://doi.org/10.1016/j.ydbio.2009.03.028
Ekker SC, McGrew LL, Lai CJ et al (1995a) Distinct expression and shared activities of members of the hedgehog gene family of Xenopus laevis. Development 121:2337–2347
Ekker SC, Ungar AR, Greenstein P et al (1995b) Patterning activities of vertebrate hedgehog proteins in the developing eye and brain. Curr Biol 5:944–995
Etchevers HC, Vincent C, Le Douarin NM, Couly GF (2001) The cephalic neural crest provides pericytes and smooth muscle cells to all blood vessels of the face and forebrain. Development 128:1059–1068
Fitzky BU, Witsch-Baumgartner M, Erdel M et al (1998) Mutations in the Delta7-sterol reductase gene in patients with the Smith–Lemli–Opitz syndrome. Proc Natl Acad Sci USA 95:8181–8186
Fombonne J, Bissey P-A, Guix C et al (2012) Patched dependence receptor triggers apoptosis through ubiquitination of caspase-9. Proc Natl Acad Sci 109:10510–10515. https://doi.org/10.1073/pnas.1200094109
Fuhrmann S, Levine EM, Reh TA (2000) Extraocular mesenchyme patterns the optic vesicle during early eye development in the embryonic chick. Development 127:4599–4609
Gage PJ, Rhoades W, Prucka SK, Hjalt T (2005) Fate maps of neural crest and mesoderm in the mammalian eye. Invest Ophthalmol Vis Sci 46:4200–4208. https://doi.org/10.1167/iovs.05-0691
Golzio C, Martinovic-Bouriel J, Thomas S et al (2007) Matthew–Wood syndrome is caused by truncating mutations in the retinol-binding protein receptor gene STRA6. Am J Hum Genet 80:1179–1187. https://doi.org/10.1086/518177
Gongal PA, French CR, Waskiewicz AJ (2011) Aberrant forebrain signaling during early development underlies the generation of holoprosencephaly and coloboma. Biochim Biophys Acta 1812:390–401. https://doi.org/10.1016/j.bbadis.2010.09.005
Goodrich LV, Johnson RL, Milenkovic L et al (1996) Conservation of the hedgehog/patched signaling pathway from flies to mice: induction of a mouse patched gene by Hedgehog. Genes Dev 10:301–312. https://doi.org/10.1101/gad.10.3.301
Grenier J, Teillet M-A, Grifone R et al (2009) Relationship between neural crest cells and cranial mesoderm during head muscle development. PLoS One 4:e4381. https://doi.org/10.1371/journal.pone.0004381
Halal F, Farsky K, Opitz JM (1981) Brief clinical report: coloboma hypospadias. Am J Med Genet 8:53–58
Halilagic A, Ribes V, Ghyselinck NB et al (2007) Retinoids control anterior and dorsal properties in the developing forebrain. Dev Biol 303:362–375. https://doi.org/10.1016/j.ydbio.2006.11.021
Hallonet M, Hollemann T, Pieler T, Gruss P (1999) Vax1, a novel homeobox-containing gene, directs development of the basal forebrain and visual system. Genes Dev 13:3106–3114
Han B, Qu Y, Jin Y et al (2015) FOXC1 activates smoothened-independent hedgehog signaling in basal-like breast cancer. Cell Rep 13:1046–1058. https://doi.org/10.1016/J.CELREP.2015.09.063
Hatini V, Tao W, Lai E (1994) Expression of winged helix genes, BF-1 and BF-2, define adjacent domains within the developing forebrain and retina. J Neurobiol 25:1293–1309. https://doi.org/10.1002/neu.480251010
Hayhurst M, McConnell SK (2003) Mouse models of holoprosencephaly. Curr Opin Neurol 16:135–141. https://doi.org/10.1097/01.wco.0000063761.15877.40
He M, Subramanian R, Bangs F et al (2014) The kinesin-4 protein Kif7 regulates mammalian Hedgehog signalling by organizing the cilium tip compartment. Nat Cell Biol 16:663–672. https://doi.org/10.1038/ncb2988
Hecquet C, Lefevre G, Valtink M et al (2002) cAMP inhibits the proliferation of retinal pigmented epithelial cells through the inhibition of ERK1/2 in a PKA-independent manner. Oncogene 21:6101–6112. https://doi.org/10.1038/sj.onc.1205765
Hernandez-Bejarano M, Gestri G, Spawls L et al (2015) Opposing Shh and Fgf signals initiate nasotemporal patterning of the zebrafish retina. Development 142:3933–3942. https://doi.org/10.1242/dev.125120
Hever AM, Williamson KA, van Heyningen V (2006) Developmental malformations of the eye: the role of PAX6, SOX2 and OTX2. Clin Genet 69:459–470. https://doi.org/10.1111/j.1399-0004.2006.00619.x
Hong M, Srivastava K, Kim S et al (2017) BOC is a modifier gene in holoprosencephaly. Hum Mutat 38:1464–1470. https://doi.org/10.1002/humu.23286
Huang X, Litingtung Y, Chiang C (2007) Region-specific requirement for cholesterol modification of sonic hedgehog in patterning the telencephalon and spinal cord. Development 134:2095–2105. https://doi.org/10.1242/dev.000729
Huh S, Hatini V, Marcus RC et al (1999) Dorsal-ventral patterning defects in the eye of BF-1-deficient mice associated with a restricted loss of shh expression. Dev Biol 211:53–63
Ittner LM, Wurdak H, Schwerdtfeger K et al (2005) Compound developmental eye disorders following inactivation of TGFbeta signaling in neural-crest stem cells. J Biol 4:11. https://doi.org/10.1186/jbiol29
Ivanovitch K, Cavodeassi F, Wilson SW (2013) Precocious acquisition of neuroepithelial character in the eye field underlies the onset of eye morphogenesis. Dev Cell 27:293–305. https://doi.org/10.1016/j.devcel.2013.09.023
James LF (1999) Teratological research at the USDA-ARS poisonous plant research laboratory. J Nat Toxins 8:63–80
Jeong J, Mao J, Tenzen T et al (2004) Hedgehog signaling in the neural crest cells regulates the patterning and growth of facial primordia. Genes Dev 18:937–951. https://doi.org/10.1101/gad.1190304
Jeong Y, El-Jaick K, Roessler E et al (2006) A functional screen for sonic hedgehog regulatory elements across a 1 Mb interval identifies long-range ventral forebrain enhancers. Development 133:761–772. https://doi.org/10.1242/dev.02239
Johnson RL, Rothman AL, Xie J et al (1996) Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 272:1668–1671
Johnston MC, Noden DM, Hazelton RD et al (1979) Origins of avian ocular and periocular tissues. Exp Eye Res 29:27–43. https://doi.org/10.1016/0014-4835(79)90164-7
Jordan T, Hanson I, Zaletayev D et al (1992) The human PAX6 gene is mutated in two patients with aniridia. Nat Genet 1:328–332. https://doi.org/10.1038/ng0892-328
Kantarci S, Al-Gazali L, Hill RS et al (2007) Mutations in LRP2, which encodes the multiligand receptor megalin, cause Donnai–Barrow and facio-oculo-acoustico-renal syndromes. Nat Genet 39:957–959. https://doi.org/10.1038/ng2063
Kastner P, Grondona JM, Mark M et al (1994) Genetic analysis of RXRa developmental convergence of RXR and RAR signaling in heart and eye morphogenesis function. Pathways 78:987–1003
Kawaguchi R, Yu J, Honda J et al (2007) A membrane receptor for retinol binding protein mediates cellular uptake of vitamin A. Science 315:820–825. https://doi.org/10.1126/science.1136244
Khan AO, Oystreck DT, Seidahmed MZ et al (2008) Ophthalmic features of Joubert syndrome. Ophthalmology 115:2286–2289. https://doi.org/10.1016/j.ophtha.2008.08.005
Kirby ML, Gale TF, Stewart DE (1983) Neural crest cells contribute to normal aorticopulmonary septation. Science 220:1059–1061
Kobayashi T, Yasuda K, Araki M (2010) Coordinated regulation of dorsal bone morphogenetic protein 4 and ventral Sonic hedgehog signaling specifies the dorso-ventral polarity in the optic vesicle and governs ocular morphogenesis through fibroblast growth factor 8 upregulation. Dev Growth Differ 52:351–363. https://doi.org/10.1111/j.1440-169X.2010.01170.x
Krauss S, Concordet JP, Ingham PW (1993) A functionally conserved homolog of the Drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75:1431–1444
Kucerova R, Dorà N, Mort RL et al (2012) Interaction between hedgehog signalling and PAX6 dosage mediates maintenance and regeneration of the corneal epithelium. Mol Vis 18:139–150
Kuzhandaivel A, Schultz SW, Alkhori L, Alenius M (2014) Cilia-mediated hedgehog signaling in Drosophila. Cell Rep 7:672–680. https://doi.org/10.1016/j.celrep.2014.03.052
Lamb TD, Collin SP, Pugh EN (2007) Evolution of the vertebrate eye: opsins, photoreceptors, retina and eye cup. Nat Rev Neurosci 8:960–976. https://doi.org/10.1038/nrn2283
Lan Y, Jiang R (2009) Sonic hedgehog signaling regulates reciprocal epithelial-mesenchymal interactions controlling palatal outgrowth. Development 136:1387–1396. https://doi.org/10.1242/dev.028167
Langenberg T, Kahana A, Wszalek JA, Halloran MC (2008) The eye organizes neural crest cell migration. Dev Dyn 237:1645–1652. https://doi.org/10.1002/dvdy.21577
Larsen KB, Lutterodt M, Rath MF, Møller M (2009) Expression of the homeobox genes PAX6, OTX2, and OTX1 in the early human fetal retina. Int J Dev Neurosci 27:485–492. https://doi.org/10.1016/j.ijdevneu.2009.04.004
Lauth M, Bergström Å, Toftgård R (2007) Phorbol esters inhibit the Hedgehog signalling pathway downstream of Suppressor of Fused, but upstream of Gli. Oncogene 26:5163–5168. https://doi.org/10.1038/sj.onc.1210321
Le Lièvre CS, Le Douarin NM (1975) Mesenchymal derivatives of the neural crest: analysis of chimaeric quail and chick embryos. J Embryol Exp Morphol 34:125–154
Li Z, Joseph NM, Easter SS Jr (2000) The morphogenesis of the zebrafish eye, including a fate map of the optic vesicle. Dev Dyn 218:175–188
Luchetti G, Sircar R, Kong JH et al (2016) Cholesterol activates the G-protein coupled receptor smoothened to promote Hedgehog signaling. Elife. https://doi.org/10.7554/eLife.20304
Luo N, Lu J, Sun Y (2012) Evidence of a role of inositol polyphosphate 5-phosphatase INPP5E in cilia formation in zebrafish. Vis Res 75:98–107. https://doi.org/10.1016/j.visres.2012.09.011
Lupo G, Liu Y, Qiu R et al (2005) Dorsoventral patterning of the Xenopus eye: a collaboration of retinoid, Hedgehog and FGF receptor signaling. Development 132:1737–1748. https://doi.org/10.1242/dev.01726
Lupu FI, Burnett JB, Eggenschwiler JT (2018) Cell cycle-related kinase regulates mammalian eye development through positive and negative regulation of the hedgehog pathway. Dev Biol 434:24–35. https://doi.org/10.1016/j.ydbio.2017.10.022
Macdonald R, Barth KA, Xu Q et al (1995) Midline signalling is required for Pax gene regulation and patterning of the eyes. Development 121:3267–3278
Macdonald R, Scholes J, Strahle U et al (1997) The Pax protein Noi is required for commissural axon pathway formation in the rostral forebrain. Development 124:2397–2408
Macé M, Galiacy SD, Erraud A et al (2011) Comparative transcriptome and network biology analyses demonstrate antiproliferative and hyperapoptotic phenotypes in human keratoconus corneas. Investig Ophthalmol Vis Sci 52:6181–6191. https://doi.org/10.1167/iovs.10-70981
Mark M, Ghyselinck NB, Kastner P et al (1998) Mesectoderm is a major target of retinoic acid action. Eur J Oral Sci 106:24–31. https://doi.org/10.1111/j.1600-0722.1998.tb02149.x
Martinez-Morales JR (2016) Vertebrate eye gene regulatory networks. In: Castelli-Gair Hombría J, Bovolenta P (eds) Organogenetic gene networks. Springer International Publishing, Cham, pp 259–274
Martinez-Morales JR, Cavodeassi F, Bovolenta P (2017) Coordinated morphogenetic mechanisms shape the vertebrate eye. Front Neurosci 11:721. https://doi.org/10.3389/fnins.2017.00721
Martínez-Morales JR, Dolez V, Rodrigo I et al (2003) OTX2 activates the molecular network underlying retina pigment epithelium differentiation. J Biol Chem 278:21721–21731. https://doi.org/10.1074/jbc.M301708200
Martínez-Morales JR, Rodrigo I, Bovolenta P (2004) Eye development: a view from the retina pigmented epithelium. Bioessays 26:766–777. https://doi.org/10.1002/bies.20064
Matt N, Ghyselinck NB, Pellerin I, Dupé V (2008) Impairing retinoic acid signalling in the neural crest cells is sufficient to alter entire eye morphogenesis. Dev Biol 320:140–148. https://doi.org/10.1016/j.ydbio.2008.04.039
Matus DQ, Magie CR, Pang K et al (2008) The Hedgehog gene family of the cnidarian, Nematostella vectensis, and implications for understanding metazoan Hedgehog pathway evolution. Dev Biol 313:501–518. https://doi.org/10.1016/j.ydbio.2007.09.032
May-Simera H, Nagel-Wolfrum K, Wolfrum U (2017) Cilia—the sensory antennae in the eye. Prog Retin Eye Res 60:144–180. https://doi.org/10.1016/j.preteyeres.2017.05.001
McKechnie NM, King M, Lee WR (1985) Retinal pathology in the Kearns–Sayre syndrome. Br J Ophthalmol 69:63–75. https://doi.org/10.1136/BJO.69.1.63
Méthot N, Basler K (1999) Hedgehog controls limb development by regulating the activities of distinct transcriptional activator and repressor forms of Cubitus interruptus. Cell 96:819–831. https://doi.org/10.1016/S0092-8674(00)80592-9
Meulemans D, Bronner-Fraser M (2005) Central role of gene cooption in neural crest evolution. J Exp Zool B Mol Dev Evol 304B:298–303
Ming JE, Kaupas ME, Roessler E et al (2002) Mutations in PATCHED-1, the receptor for SONIC HEDGEHOG, are associated with holoprosencephaly. Hum Genet 110:297–301. https://doi.org/10.1007/s00439-002-0695-5
Morcillo J, Martinez-Morales JR, Trousse F et al (2006) Proper patterning of the optic fissure requires the sequential activity of BMP7 and SHH. Development 133:3179–3190. https://doi.org/10.1242/dev.02493
Mouden C, Dubourg C, Carré W et al (2016) Complex mode of inheritance in holoprosencephaly revealed by whole exome sequencing. Clin Genet 89:659–668. https://doi.org/10.1111/cge.12722
Nikoh N, Iwabe N, Kuma K et al (1997) An estimate of divergence time of Parazoa and Eumetazoa and that of Cephalochordata and Vertebrata by aldolase and triose phosphate isomerase clocks. J Mol Evol 45:97–106
Nilsson D-E (2004) Eye evolution: a question of genetic promiscuity. Curr Opin Neurobiol 14:407–414
Nishimura DY, Swiderski RE, Alward WLM et al (1998) The forkhead transcription factor gene FKHL7 is responsible for glaucoma phenotypes which map to 6p25. Nat Genet 19:140–147. https://doi.org/10.1038/493
Niswander L, Jeffrey S, Martin GR, Tickle C (1994) A positive feedback loop coordinates growth and patterning in the vertebrate limb. Nature 371:609–612. https://doi.org/10.1038/371609a0
Norgett EE, Yii A, Blake-Palmer KG et al (2015) A role for VAX2 in correct retinal function revealed by a novel genomic deletion at 2p13.3 causing distal renal tubular acidosis: case report. BMC Med Genet 16:38. https://doi.org/10.1186/s12881-015-0182-1
Nüsslein-Volhard C, Wieschaus E (1980) Mutations affecting segment number and polarity in drosophila. Nature 287:795–801. https://doi.org/10.1038/287795a0
Odent S, Atti-Bitach T, Blayau M et al (1999) Expression of the Sonic hedgehog (SHH) gene during early human development and phenotypic expression of new mutations causing holoprosencephaly. Hum Mol Genet 8:1683–1689
Ogden SK, Fei DL, Schilling NS et al (2008) G protein Galphai functions immediately downstream of Smoothened in Hedgehog signalling. Nature 456:967–970
Ohkubo Y, Chiang C, Rubenstein JL (2002) Coordinate regulation and synergistic actions of BMP4, SHH and FGF8 in the rostral prosencephalon regulate morphogenesis of the telencephalic and optic vesicles. Neuroscience 111:1–17
Ohsaki K, Morimitsu T, Ishida Y et al (1999) Expression of the Vax family homeobox genes suggests multiple roles in eye development. Genes Cells 4:267–276
Onodera S, Saito A, Hasegawa D et al (2017) Multi-layered mutation in hedgehog-related genes in Gorlin syndrome may affect the phenotype. PLoS One 12:e0184702. https://doi.org/10.1371/journal.pone.0184702
Pandit T, Ogden S (2017) Contributions of noncanonical smoothened signaling during embryonic development. J Dev Biol 5:11. https://doi.org/10.3390/jdb5040011
Pasutto F, Sticht H, Hammersen G et al (2007) Mutations in STRA6 cause a broad spectrum of malformations including anophthalmia, congenital heart defects, diaphragmatic hernia, alveolar capillary dysplasia, lung hypoplasia, and mental retardation. Am J Hum Genet 80:550–560
Patel A, Sowden JC (2017) Genes and pathways in optic fissure closure. Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2017.10.010
Perron M, Boy S, Amato MA et al (2003) A novel function for Hedgehog signalling in retinal pigment epithelium differentiation. Development 130:1565–1577
Pfirrmann T, Jandt E, Ranft S et al (2016) Hedgehog-dependent E3-ligase Midline1 regulates ubiquitin-mediated proteasomal degradation of Pax6 during visual system development. Proc Natl Acad Sci USA 113:10103–10108
Picker A, Brand M (2005) Fgf signals from a novel signaling center determine axial patterning of the prospective neural retina. Development 132:4951–4962. https://doi.org/10.1242/dev.02071
Picker A, Cavodeassi F, Machate A et al (2009) Dynamic coupling of pattern formation and morphogenesis in the developing vertebrate retina. PLoS Biol 7:e1000214. https://doi.org/10.1371/journal.pbio.1000214
Pineda-Alvarez DE, Solomon BD, Roessler E et al (2011) A broad range of ophthalmologic anomalies is part of the holoprosencephaly spectrum. Am J Med Genet Part A 155:2713–2720. https://doi.org/10.1002/ajmg.a.34261
Pineda-Alvarez DE, Roessler E, Hu P et al (2012) Missense substitutions in the GAS1 protein present in holoprosencephaly patients reduce the affinity for its ligand, SHH. Hum Genet 131:301–310. https://doi.org/10.1007/s00439-011-1078-6
Pinson L, Augé J, Audollent S et al (2004) Embryonic expression of the human MID1 gene and its mutations in Opitz syndrome. J Med Genet 41:381–386
Porter JA, Young KE, Beachy PA (1996) Cholesterol modification of hedgehog signaling proteins in animal development. Science 274:255–259
Putoux A, Thomas S, Coene KLM et al (2011) KIF7 mutations cause fetal hydrolethalus and acrocallosal syndromes. Nat Genet 43:601–606. https://doi.org/10.1038/ng.826
Quaderi NA, Schweiger S, Gaudenz K et al (1997) Opitz G/BBB syndrome, a defect of midline development, is due to mutations in a new RING finger gene on Xp22. Nat Genet 17:285–291. https://doi.org/10.1038/ng1197-285
Ragge NK, Brown AG, Poloschek CM et al (2005a) Heterozygous mutations of OTX2 cause severe ocular malformations. Am J Hum Genet 76:1008–1022
Ragge NK, Salt A, Collin JRO et al (2005b) Gorlin syndrome: the PTCH gene links ocular developmental defects and tumour formation. Br J Ophthalmol 89:988–991. https://doi.org/10.1136/bjo.2004.061390
Rbaibi Y, Cui S, Mo D et al (2012) OCRL1 modulates cilia length in renal epithelial cells. Traffic 13:1295–1305. https://doi.org/10.1111/j.1600-0854.2012.01387.x
Rembold M, Loosli F, Adams RJ, Wittbrodt J (2006) Individual cell migration serves as the driving force for optic vesicle evagination. Science 313:1130–1134. https://doi.org/10.1126/science.1127144
Rhinn M, Dollé P (2012) Retinoic acid signalling during development. Development 139:843–858. https://doi.org/10.1242/dev.065938
Ribeiro LA, Murray JC, Richieri-Costa A (2006) PTCH mutations in four Brazilian patients with holoprosencephaly and in one with holoprosencephaly-like features and normal MRI. Am J Med Genet A 140:2584–2586. https://doi.org/10.1002/ajmg.a.31369
Ribeiro LA, Quiezi RG, Nascimento A et al (2010) Holoprosencephaly and holoprosencephaly-like phenotype and GAS1 DNA sequence changes: report of four Brazilian patients. Am J Med Genet A 152A:1688–1694. https://doi.org/10.1002/ajmg.a.33466
Richieri-Costa A, Vendramini-Pittoli S, Kokitsu-Nakata N et al (2016) Multisystem involvement in a patient with a PTCH1 mutation: clinical and imaging findings. J Pediatr Genet 06:103–106
Roessler E, Belloni E, Gaudenz K et al (1996) Mutations in the human Sonic Hedgehog gene cause holoprosencephaly. Nat Genet 14:357–360. https://doi.org/10.1038/ng1196-357
Roessler E, Du YZ, Mullor JL et al (2003) Loss-of-function mutations in the human GLI2 gene are associated with pituitary anomalies and holoprosencephaly-like features. Proc Natl Acad Sci USA 100:13424–13429. https://doi.org/10.1073/pnas.22357341002235734100
Roessler E, Ma Y, Ouspenskaia MV et al (2009) Truncating loss-of-function mutations of DISP1 contribute to holoprosencephaly-like microform features in humans. Hum Genet 125:393–400. https://doi.org/10.1007/s00439-009-0628-7
Rohatgi R, Milenkovic L, Scott MP (2007) Patched1 regulates hedgehog signaling at the primary cilium. Science 317:372–376. https://doi.org/10.1126/science.1139740
Romano M, Iacovello D, Cascone NC, Contestabile MT (2011) Identification of a novel mutation in the PTCH gene in a patient with Gorlin–Goltz syndrome with unusual ocular disorders. Eur J Ophthalmol 21:516–519. https://doi.org/10.5301/EJO.2010.6186
Rosenfeld JA, Ballif BC, Martin DM et al (2010) Clinical characterization of individuals with deletions of genes in holoprosencephaly pathways by aCGH refines the phenotypic spectrum of HPE. Hum Genet 127:421–440. https://doi.org/10.1007/s00439-009-0778-7
Sasagawa S, Takabatake T, Takabatake Y et al (2002) Axes establishment during eye morphogenesis in Xenopus by coordinate and antagonistic actions of BMP4, Shh, and RA. Genesis 33:86–96. https://doi.org/10.1002/gene.10095
Schauerte HE, van Eeden FJ, Fricke C et al (1998) Sonic hedgehog is not required for the induction of medial floor plate cells in the zebrafish. Development 125:2983–2993
Schimmenti LA, de la Cruz J, Lewis RA et al (2003) Novel mutation in sonic hedgehog in non-syndromic colobomatous microphthalmia. Am J Med Genet A 116A:215–221. https://doi.org/10.1002/ajmg.a.10884
Schwarz M, Cecconi F, Bernier G et al (2000) Spatial specification of mammalian eye territories by reciprocal transcriptional repression of Pax2 and Pax6. Development 127:4325–4334
Schwarz N, Lane A, Jovanovic K et al (2017) Arl3 and RP2 regulate the trafficking of ciliary tip kinesins. Hum Mol Genet 26:2480–2492. https://doi.org/10.1093/hmg/ddx143
Semina EV, Reiter R, Leysens NJ et al (1996) Cloning and characterization of a novel bicoid-related homeobox transcription factor gene, RIEG, involved in Rieger syndrome. Nat Genet 14:392–399. https://doi.org/10.1038/ng1296-392
Seppala M, Xavier GM, Fan CM, Cobourne MT (2014) Boc modifies the spectrum of holoprosencephaly in the absence of Gas1 function. Biol Open 3:728–740. https://doi.org/10.1242/bio.20147989
Shaffer LG, Theisen A, Bejjani BA et al (2007) The discovery of microdeletion syndromes in the post-genomic era: Review of the methodology and characterization of a new 1q41q42 microdeletion syndrome. Genet Med 9:607–616. https://doi.org/10.1097/GIM.0b013e3181484b49
Sigg MA, Menchen T, Lee C et al (2017) Evolutionary Proteomics Uncovers Ancient Associations of Cilia with Signaling Pathways. Dev Cell 43:744–762.e11. https://doi.org/10.1016/j.devcel.2017.11.014
Slavotinek AM (2011) Eye development genes and known syndromes. Mol Genet Metab 104:448–456. https://doi.org/10.1016/j.ymgme.2011.09.029
Slavotinek AM, Chao R, Vacik T et al (2012) VAX1 mutation associated with microphthalmia, corpus callosum agenesis, and orofacial clefting: the first description of a VAX1 phenotype in humans. Hum Mutat 33:364–368. https://doi.org/10.1002/humu.21658
St-Jacques B, Hammerschmidt M, McMahon AP (1999) Indian hedgehog signaling regulates proliferation and differentiation of chondrocytes and is essential for bone formation. Genes Dev 13:2072–2086. https://doi.org/10.1101/gad.13.16.2072
Strom SP, Clark MJ, Martinez A et al (2016) De novo occurrence of a variant in ARL3 and apparent autosomal dominant transmission of retinitis pigmentosa. PLoS One 11:e0150944. https://doi.org/10.1371/journal.pone.0150944
Sullivan CH, Braunstein L, Hazard-Leonards RM et al (2004) A re-examination of lens induction in chicken embryos: In vitro studies of early tissue interactions. Int J Dev Biol 48:771–782. https://doi.org/10.1387/ijdb.041894cs
Taipale J, Chen JK, Cooper MK et al (2000) Effects of oncogenic mutations in smoothened and patched can be reversed by cyclopamine. Nature 406:1005–1009. https://doi.org/10.1038/35023008
Take-uchi M, Clarke JD, Wilson SW (2003) Hedgehog signalling maintains the optic stalk-retinal interface through the regulation of Vax gene activity. Development 130:955–968
Thibert C, Teillet M-A, Lapointe F et al (2003) Inhibition of neuroepithelial patched-induced apoptosis by sonic hedgehog. Science 301:843–846. https://doi.org/10.1126/science.1085405
Tian H, Jeong J, Harfe BD et al (2008) Mouse Disp1 is required in sonic hedgehog-expressing cells for paracrine activity of the cholesterol-modified ligand. Development 135:3471–3472. https://doi.org/10.1242/dev.029678
Tolosa EJ, Fernández-Zapico ME, Battiato NL, Rovasio RA (2016) Sonic hedgehog is a chemotactic neural crest cell guide that is perturbed by ethanol exposure. Eur J Cell Biol 95:136–152. https://doi.org/10.1016/j.ejcb.2016.02.003
Tukachinsky H, Kuzmickas RP, Jao CY et al (2012) Dispatched and scube mediate the efficient secretion of the cholesterol-modified hedgehog ligand. Cell Rep 2:308–320. https://doi.org/10.1016/j.celrep.2012.07.010
Twigg SRF, Hufnagel RB, Miller KA et al (2016) A recurrent mosaic mutation in SMO, encoding the Hedgehog signal transducer smoothened, is the major cause of Curry–Jones syndrome. Am J Hum Genet 98:1256–1265. https://doi.org/10.1016/j.ajhg.2016.04.007
Valente EM, Brancati F, Dallapiccola B (2008) Genotypes and phenotypes of Joubert syndrome and related disorders. Eur J Med Genet 51:1–23. https://doi.org/10.1016/J.EJMG.2007.11.003
Van Cruchten S, Vrolyk V, Perron Lepage M-F et al (2017) Pre- and postnatal development of the eye: a species comparison. Birth Defects Res 109:1540–1567. https://doi.org/10.1002/bdr2.1100
Wallis D, Muenke M (2000) Mutations in holoprosencephaly. Hum Mutat 16:99–108. https://doi.org/10.1002/1098-1004(200008)16:2%3C99::AID-HUMU2%3E3.0.CO;2-0
Wassif CA, Maslen C, Kachilele-Linjewile S et al (1998) Mutations in the human sterol delta7-reductase gene at 11q12–13 cause Smith–Lemli–Opitz syndrome. Am J Hum Genet 63:55–62. https://doi.org/10.1086/301936
Westenskow P, Piccolo S, Fuhrmann S (2009) Beta-catenin controls differentiation of the retinal pigment epithelium in the mouse optic cup by regulating Mitf and Otx2 expression. Development 136:2505–2510. https://doi.org/10.1242/dev.032136
Wild A, Kalff-Suske M, Vortkamp A et al (1997) Point mutations in human GLI3 cause Greig syndrome. Hum Mol Genet 6:1979–1984
Williams AL, Bohnsack BL (2015) Neural crest derivatives in ocular development: discerning the eye of the storm. Birth Defects Res Part C Embryo Today Rev 105:87–95. https://doi.org/10.1002/bdrc.21095
Williams AL, Eason J, Chawla B, Bohnsack BL (2017) Cyp1b1 regulates ocular fissure closure through a retinoic acid—independent pathway. Investig Opthalmol Vis Sci 58:1084. https://doi.org/10.1167/iovs.16-20235
Xavier GM, Seppala M, Barrell W et al (2016) Hedgehog receptor function during craniofacial development. Dev Biol 415:198–215. https://doi.org/10.1016/j.ydbio.2016.02.009
Xie J, Murone M, Luoh SM et al (1998) Activating Smoothened mutations in sporadic basal-cell carcinoma. Nature 391:90–92. https://doi.org/10.1038/34201
Zhang XM, Yang XJ (2001) Temporal and spatial effects of Sonic hedgehog signaling in chick eye morphogenesis. Dev Biol 233:271–290. https://doi.org/10.1006/dbio.2000.0195S0012-1606(00)90195-7
Zhang X, Jefferson AB, Auethavekiat V, Majerus PW (1995) The protein deficient in Lowe syndrome is a phosphatidylinositol-4,5-bisphosphate 5-phosphatase. Proc Natl Acad Sci USA 92:4853–4856
Zhang W, Hong M, Bae GU et al (2011) Boc modifies the holoprosencephaly spectrum of Cdo mutant mice. Dis Model Mech 4:368–380. https://doi.org/10.1242/dmm.005744
Zhang Z, Shen L, Law K et al (2017) Suppressor of fused chaperones gli proteins to generate transcriptional responses to Sonic Hedgehog signaling. Mol Cell Biol 37:e00421–e00416. https://doi.org/10.1128/MCB.00421-16
Zhao L, Saitsu H, Sun X et al (2010) Sonic hedgehog is involved in formation of the ventral optic cup by limiting Bmp4 expression to the dorsal domain. Mech Dev 127:62–72. https://doi.org/10.1016/j.mod.2009.10.006
Zhao C, Yasumura D, Li X et al (2011) mTOR-mediated dedifferentiation of the retinal pigment epithelium initiates photoreceptor degeneration in mice. J Clin Invest 121:369–383. https://doi.org/10.1172/JCI44303
Zhao L, Zevallos SE, Rizzoti K et al (2012) Disruption of SoxB1-dependent sonic hedgehog expression in the hypothalamus causes septo-optic dysplasia. Dev Cell 22:585–596. https://doi.org/10.1016/j.devcel.2011.12.023
Zieske JD (2004) Corneal development associated with eyelid opening. Int J Dev Biol 48:903–911. https://doi.org/10.1387/ijdb.041860jz
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Cavodeassi, F., Creuzet, S. & Etchevers, H.C. The hedgehog pathway and ocular developmental anomalies. Hum Genet 138, 917–936 (2019). https://doi.org/10.1007/s00439-018-1918-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-018-1918-8