Abstract
Central and semiarid north-eastern Asia was subject to twentieth century warming far above the global average. Since forests of this region occur at their drought limit, they are particularly vulnerable to climate change. We studied the regional variations of temperature and precipitation trends and their effects on tree growth and forest regeneration in Mongolia. Tree-ring series from more than 2,300 trees of Siberian larch (Larix sibirica) collected in four regions of Mongolia’s forest zone were analyzed and related to available weather data. Climate trends underlie a remarkable regional variation leading to contrasting responses of tree growth in taiga forests even within the same mountain system. Within a distance of a few hundred kilometers (140–490 km), areas with recently reduced growth and regeneration of larch alternated with regions where these parameters remained constant or even increased. Reduced productivity could be correlated with increasing summer temperatures and decreasing precipitation; improved growth conditions were found at increasing precipitation, but constant summer temperatures. An effect of increasing winter temperatures on tree-ring width or forest regeneration was not detectable. Since declines of productivity and regeneration are more widespread in the Mongolian taiga than the opposite trend, a net loss of forests is likely to occur in the future, as strong increases in temperature and regionally differing changes in precipitation are predicted for the twenty-first century.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The southern limit of the Siberian taiga, the earth’s largest continuous forest area, is located in Mongolia. Southwards, the taiga is displaced by the vast Eurasian steppe belt. Whereas the position of the forest-steppe ecotone has been modified by human activities, including livestock breeding, logging, and arson, the lack of conifers in the steppe is principally due to drought (Gunin et al. 1999; Dulamsuren et al. 2009a, b). This becomes apparent from fluctuations of the forest-steppe borderline along with changes in temperature and precipitation throughout the Holocene (Tsedendash 1995; Gunin et al. 1999; Miehe et al. 2007). Siberian larch (Larix sibirica Ledeb.), Mongolia’s most common forest tree, covering not less than 80% of the forest area (Tsogtbaatar 2004; Dugarjav 2006), migrated northward by 2° latitude during the past 4300 years (Dinesman et al. 1989).
Mongolia’s average temperature has increased by 1.7°C since the 1940s (Batima et al. 2005). During the next 80 years, the temperature is predicted to increase by 2°C in summer and 1°C in winter (Sato and Kimura 2006). The frequency of heat waves increased during the past decades (Nandintsetseg et al. 2007), while approaches of cold fronts from Siberia became rarer, reducing the occurrence of storms especially in spring (Hayasaki et al. 2006; Sato and Kimura 2006). The permafrost area is currently reduced (Bohannon 2008) and likely to vanish from Mongolia during the twenty-first century (Stendel and Christensen 2002; Böhner and Lehmkuhl 2005). Permafrost is not only an essential buffer for the vegetation, alleviating summer droughts (Sugimoto et al. 2002), but also an important carbon pool (Nelson 2003; Zimov et al. 2006). Mongolia’s glaciers have declined by 10–30% in area during the past 60 years (Kadota and Davaa 2005). The response of precipitation to global warming is spatially heterogeneous (Morinaga et al. 2003; Endo et al. 2006), and therefore no significant twentieth century trend is found on a national scale (Batima et al. 2005). However, both regional decreases and increases of annual precipitation by several decimeters have been reported from individual weather stations during the second half of the twentieth century (Batima et al. 2005). For most of northern Mongolia’s taiga forest belt, precipitation and, it is thought, with it, soil moisture will decrease during the twenty-first century (Sato et al. 2007).
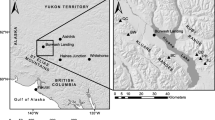
Climate change effects on forest lines have been primarily studied at arctic or alpine limits, where increasing temperatures usually lead to the expansion of forests into tundra or alpine meadows (Dullinger et al. 2004; Mazepa 2004), though this expansion is sometimes hampered by increased fire frequency (Chapin et al. 2004). Remarkably short periods of climate change, lasting only a few decades, can lead to significant shifts of the tree line (Kullman 1998, 2007). In northern Mongolia, a tree-ring study revealed an increased annual increment of Pinus sibirica Du Tour at an alpine forest line (Jacoby et al. 1996; D’Arrigo et al. 2000). Global warming responses at lower forest lines to the steppe are hardly studied, though. Siberian larch growing at such sites was recently shown to be limited by water constraints, both as mature trees and during their reproduction (Dulamsuren et al. 2008, 2009a). These findings and results of tree-ring analyses of a single area in Mongolia (Dulamsuren et al. 2010a) suggest that at the lower forest line taiga forests might generally respond to global warming with decreasing growth and regeneration. In the context of a high spatial variability of precipitation regimes in Mongolia (Morinaga et al. 2003; Batima et al. 2005; Endo et al. 2006, Sato et al. 2007), we selected four study areas in Mongolia’s taiga belt (Fig. 1) for tree-ring analyses in order to assess both the regional variability of climate trends and their potential as a hazard for forest growth and regeneration. In the scope of this analysis, the hypothesis was tested that Siberian larch, growing in the forest-steppe ecotone, shows an overall trend for declining growth and regeneration during the twentieth century along with increasing temperatures.
Distribution of the Mongolian taiga forests (black areas; black dots represent isolated occurrences) and study areas (A–D; corresponding to areas defined in Table 1): A north-western Khentey, B south-western Khentey, C eastern Khangay, D south-eastern Khentey. Lakes are indicated in gray
Materials and methods
Wood core sampling and processing
Tree-ring series from 2,314 individuals of L. sibirica were analyzed from the north-western, south-western and south-eastern Khentey as well as from the eastern Khangay Mountains (Table 1; Fig. 1). The forest-steppe ecotone in these regions is characterized by a vegetation mosaic of forests on the more humid north-facing slopes and grasslands on the south-facing slopes (Hilbig 1995; Dulamsuren et al. 2005; Bayarsaikhan et al. 2009); consequently only north-facing slopes were included in wood-core sampling. All trees were sampled from 20 m × 20 m plots (five replicate plots per site; three sites in the north-western Khentey, five sites in the other study areas); overall 90 plots were sampled. This means that an average number around 25 trees were sampled on each plot. The average stand density amounted to 535 ± 33 trees/ha (only > 60-year old conifers of the tree layer included) (Fig. 2). In 50% of the plots, the stand density varied between 400 and 700 trees, which is in the lower range of stand densities in boreal forests (Kobak et al. 1996; Luyssaert et al. 2008). With the large number of samples, we intended to analyze the age structure of the forest stands and the dependence of forest regeneration on climate. The high numbers of sampled trees resulted in high values of the expressed population signal (EPS), which clearly exceeded the 0.85-threshold value (Wigley et al. 1984). The samples were collected in October 2007.
The wood cores were sampled with increment borers of 5-mm inner diameter. The borer was driven into the wood parallel to the contour lines of the mountain slopes at 1 m above the ground to avoid compression wood. The wood cores were mounted on wooden strips and cut lengthwise with a scalpel; the contrast between annual tree rings was enhanced with chalk. The tree-ring widths were measured with a precision of 10 μm on a movable object table, the movements of which are electronically transmitted to a computer system equipped with time series analysis and presentation (TSAP)-Win software (Rinntech, Heidelberg).
Evaluation of tree-ring data
Tree-ring data were analyzed following the methods applied by Sarris et al. (2007) and Dulamsuren et al. (2010a) using TSAP-Win software. During cross-dating, the tree-ring series were controlled for missing rings and false rings, which are quite frequent in the semiarid environment of Mongolia in drought-limited forests. Cross-dating was based on the coefficient of agreement [Gleichläufigkeit values (GL); Eckstein and Bauch 1969] and t values (Baillie and Pilcher 1973). Trees of the same site were pooled by calculating mean values of the annual increment. Tree-ring series used for the calculation of means had GL > 65% and t values > 3 (>6 in ca. 90% of samples). Trend lines were calculated using moving 5-year averages.
The interannual (high-frequency) variation of climate was extracted by removing the age-related information from tree-ring width series. This is principally achieved by dividing the observed tree-ring width (r i) by the expected annual increment. The expected annual increment is not constant, but declines throughout the lifetime of a tree for two reasons: the same amount of wood has to be distributed over a larger circumference of the trunk from year to year, trees tend to grow faster during the first decades of their lifetime. Finding the correct function for the age-related growth trend (for estimating the expected growth rate) is easier in semiarid environments, including our study area, than in regions with a good water supply, as the stand density in semiarid woodlands is relatively low and, thus, the individual trees are less influenced by changes in the stand structure than in dense and moist forests (Cook 1985). Therefore, the same type of standardization (i.e., of removing the age-related trend) could be applied for all tree-ring series studied. The annual tree-ring index (z i ) of year i was calculated with the equation z i = 100 × r i /m i , where m i is the 5-year moving average of year i.
In addition to high-frequency variation, the long-term (low-frequency) trend of tree-ring width over time was analyzed to detect long-term changes in climate occurring during the lifespan of the tree. Long-term climate trends can be identified by removing the annual variation of climate from the tree-ring series and conserving the age-related trend (Sarris et al. 2007; Dulamsuren et al. 2010a). If the annual increment is not related to the calendar year, but to tree age, a mean age-related growth curve can be established for a given site, which is largely independent of the annual variation of climate. Such functions are called regional growth curves (RGC; Briffa et al. 1992; Helama et al. 2004; Naurzbaev et al. 2004). Climate trends can be deduced from the RGC by comparing it with tree-ring series from trees of different age. We prefer the comparison of several (partial) RGC for trees of different age classes with one another, because the comparison of an individual growth curve to the RGC calculated for all trees might blur existing trends, as the individual tree-ring series is also included in the RGC. Age is generally specified as the age of the oldest tree ring (cambial age) at the sampling height of 1 m; ca. 10 (to 20) years should be added to deduce tree age from these age specification (Körner et al. 2005; Sankey et al. 2006). Age classes distinguished in the analyses include trees with a cambial age >90 years (old trees), between 50 and 90 years (middle-aged trees) and trees <50 years (young trees). Assuming a difference of 10 years between the cambial age and the year of germination, these groups correspond to tree ages of >100, 60–100, and <60 years, respectively.
Influences other than climate and tree age, including internal (e.g., the natural death of a neighboring tree) and external disturbances (e.g., insect infestations, fire) of stands (Dulamsuren et al. 2010b), tree-specific characters caused by genetic variations or the small-scale variation of site parameters (Wilmking et al. 2004) were minimized due to the large sample size and the collection of wood cores on several mountain slopes per study area.
Forest regeneration
The temporal development of the establishment of larch trees was analyzed in all 90 plots. Past forest regeneration was deduced from the starting points of the individual tree-ring series. The years of establishment (i.e., germination) of the individual trees were inferred from the wood cores taken at 1 m above the ground by adding 10 years to the year of the oldest tree ring. There is some uncertainty inherent in this assumption, as the exact number of tree rings, which are not detectable at 1-m height, is not known and underlies some tree-to-tree variation. This has to be considered when interpreting the abscissas of Figs. 4a, 7 and 9a. However, the expected shifts along the abscissa would amount only to 1 year or a few years and would thus not affect the principle information which can be extracted from these analyses. It is improbable that less than 10 years would have to be added (Sankey et al. 2006). Therefore, any error inherent in this method would lead to a small overestimation of recent regeneration. In addition to wood core sampling, all sample plots were thoroughly searched for seedlings and saplings below 1-m height to collect stem cross-sections. These samples taken from seedlings and saplings at <1-m height were used for the determination of the age structure, but were not included in the tree-ring chronologies.
δ13C signatures of tree-ring wood
Tree-ring wood from 1988 to 1997 and from 1998 to 2007 was dried at 105°C for 24 h, ground to a fine powder, and bulk samples for the 10-year periods (ca. 1 mg) were weighed in tin capsules for the determination of δ13C signatures. The analyses were conducted with a Delta V Advantage isotope ratio mass spectrometer (Thermo Fisher Scientific, Waltham, Mass.), which was connected to an NA 1500 C/N Elementar Analyzer (Carlo Erba Strumentazione, Milan) via a Conflo III interface (Thermo Fisher Scientific). Acetanilide was used as an internal standard. Using this internal standard, the δ13C signature was related to the Pee Dee belemnite limestone standard using the equation δ13C (‰) = [(R sample/R standard) − 1] × 1,000, with R = 12C/13C. Enrichment of 13C indicating drought stress results in high (less negative) values of δ13C.
Climate data
The positions of the weather stations (Tables 2, 3) are: 49°48′N, 106°42′E, 900 m a.s.l., station Eroo (study area A); 47°90′N, 107°0′E, 1,360 m, Ulan Bator (study area B); 47°40′N, 101°50′E, 1,700 m, Tsenkher (study area C); 47°30′N, 110°70′E, 1,030 m, Underkhan (study area D). The positions of the weather stations remained unchanged during the sampling periods, which started in: 1961 (A), 1950 (B), 1937 (C), and 1942 (D).
Mongolia’s climate is characterized by the Asiatic anticyclone in winter, which typically has its center southwest of Lake Baikal and causes dry and cold winters. In summer, warm air masses from the south flow into northern Mongolia resulting in the formation of cyclones when they meet the cold air from Siberia. Therefore, most precipitation is received during summer. Mongolia’s average temperature shows a long-term trend of increase throughout the period since 1940 when sufficient instrumental data are available for the calculation of a regional average (Batima et al. 2005). Consistent with the global development of temperatures, the period from 1940 to 1960s was generally cooler than the subsequent decades.
Results
In the twentieth century, the growth trends strongly varied between the four study areas, even though the distance between neighboring areas varied only between 140 and 490 km (Fig. 1). In three out of four study areas, both the annual increment and the potential for forest regeneration strongly declined from the 1940s. This decline was most severe in the north-western Khentey Mountains (study area A), an area where summer and winter temperatures are increasing (Fig. 3a; Table 2), concomitant to a significant decrease in annual precipitation (Fig. 3b; Table 3). The increase in the annual mean temperature amounted to 2.5°C within 45 years (Table 2), whereas the annual precipitation decreased by 100 mm from 330 to 230 mm (Table 3). A characteristic of Mongolia’s climate is a pronounced peak of precipitation in July and August, which is essential for the water supply of the vegetation. It was precisely then that precipitation decreased most strongly in the north-western Khentey, from 185 to 105 mm within 45 years.
Our tree-ring chronologies of the north-western Khentey date back to the late nineteenth century. An outstanding character of these chronologies is the abrupt decline of the annual increment in the 1940s irrespective of the age of the trees (Fig. 4a, b). Growth declines in drought years and recoveries in subsequent wet years were the rule for centuries, but the larch trees never recovered from 2 subsequent drought years in 1948 and 1954 (Fig. 4a). This can be attributed to the constant increase in aridity in the north-western Khentey since that time (Fig. 3). This conclusion is supported by recently increasing δ13C values of the tree-ring wood. In more than 100-year-old trees, the δ13C values were significantly higher (P ≤ 0.001, t test, n = 10) during the recent decade (−24.6 ± 0.2‰; means ± SE) than in the decade before (−25.6 ± 0.1‰). In the age class from 50 to 90 cambial years, δ13C values increased from −25.3 ± 0.2‰ to −24.0 ± 0.3‰ (P ≤ 0.001, n = 15), indicating an increasing stomatal limitation of photosynthesis. Cumulative growth curves, where the yearly increment is related to tree age (or more exactly to the age of the cambium at the sampling height) instead of the calendar year, show that old trees grew much faster during the first decades of their lifespan than trees that are currently young (Fig. 4c).
In addition to declining growth of already established trees, failure of forest regeneration (Fig. 4a) is a threat for the persistence of the larch forests covering the mountain slopes of the north-western Khentey. After a period of rich regeneration in the 1930s and 1940s, the rejuvenation of larch strongly decreased concomitant to the decline of the annual increment of mature trees (Fig. 4a). Since the 1970s, larch seedlings have been virtually absent in the mountain forests (Fig. 4a), while they still occur in flood plains.
In the south-western Khentey (B) and eastern Khangay Mountains (C; Fig. 1), only temperature increased (Table 2), whereas precipitation remained constant (Table 3). However, the increase in temperature in the south-western Khentey since 1950 by 4.4°C is remarkably high and exceeds the levels in the other study areas (Table 2). This increase is especially due to an enormous increase in winter temperatures, whereas summer temperatures increased by the same magnitude as in the north-western Khentey and the eastern Khangay (Table 2). The highest increase in temperature was in February at not less than 8.9°C since 1950 (Fig. 5). The eastern Khangay differs from the other study areas by its higher amount of annual precipitation (Table 3) and warmer winters (Table 2). In the eastern Khangay and south-western Khentey, the RGC for trees of different age classes reveal that, as in the north-western Khentey, young trees today grow more slowly than did the now old trees at the same age (Fig. 6). However, the decline in the annual increment during the twentieth century in the south-western Khentey and eastern Khangay is less pronounced than in the north-western Khentey, which is consistent with the constant precipitation in the south-western Khentey and eastern Khangay, but decreasing summer rainfall in the north-western Khentey. The rejuvenation of larch in the south-western Khentey strongly decreased in the 1940s, but in contrast to the north-western Khentey, sparse regeneration has occurred until the present time (Fig. 7). In the more humid eastern Khangay, strong regeneration of larch occurred until the early 1960s; afterwards, trees could establish only occasionally (Fig. 7).
In the eastern Khangay, the oldest tree-ring series in our sample collection reaches back to the late sixteenth century (Fig. 8). In this individual tree-ring series, the nineteenth century is peculiar because of narrow tree rings and a low annual variability of the tree-ring width. During the wet phase starting in the early twentieth century, both tree-ring width and its variability increased, before it recently decreased along with increasing temperatures.
The south-eastern Khentey (D; Fig. 1) strongly differs from the other three study areas. Here, the increase in the annual mean temperature (Table 2) is limited to an increase in winter temperature in contrast to all other regions. In addition, the annual precipitation increased from 210 to 260 mm in the past 65 years (Table 3). These conditions apparently promoted tree growth. Even old larch trees increased in annual stem increment in the late twentieth century (Fig. 9a) and were capable of overcompensating the age-dependent increment decline occurring in any tree primarily due to the merely geometric fact that wood production is distributed over a larger circumference with increasing age. In contrast to our other study areas, young trees grew faster during the recent decades than the trees formerly did at the same age in past decades (Fig. 9b). Moreover, larch forests in the south-eastern Khentey are regenerating well (Fig. 9a).
Discussion
Spatio-temporal fluctuations of L. sibirica forest lines in Mongolia’s forest-steppe ecotone have often been discussed in the context of a potential encroachment of trees into the steppe, because L. sibirica was thought to be excluded from many steppe areas only by livestock grazing and not for climatic reasons (Korotkov and Dorjsuren 1988; Hilbig 1995; Sankey et al. 2006). Recent studies in the north-western Khentey suggested that natural site factors alone are sufficient to prevent L. sibirica from establishing in grasslands along the lower forest line at least in that particular area. Relevant natural site factors include water shortages and high soil temperatures as well as herbivory by insects and small mammals (Dulamsuren et al. 2008, 2009a, 2010a, b; Hauck et al. 2008). The present results indicate that the potential of L. sibirica to invade the steppe has continuously decreased during the late twentieth century. Declines in the annual stem increment and, even more importantly, in regeneration suggest that in parts of the Mongolian forest-steppe ecotone the conversion of forests into steppe is much more probable than the succession of grassland to Siberian larch forests.
The comparative study of four regions within the Mongolian forest zone showed that twentieth century climate trends and responses of L. sibirica to changes in temperature and precipitation underlie a striking spatial variation. The differences between the three areas in the Khentey Mountains are especially remarkable, because they are only separated by 140–200 km. Within this small area, locations both with increasing and decreasing aridity are found and result in diverging responses of L. sibirica. The increase by 4.4°C within 55 years in the south-western Khentey is notable for its scale, but agrees with the general high increase in temperature in central Asia during the second half of the twentieth century, which exceeded the global trend by a factor of 3–4 (IPCC 2007).
Increased drought during the growing season due to the simultaneous increase in summer temperature and decrease in precipitation, as in the north-western Khentey, is obviously most detrimental for L. sibirica, as its growth is strongly reduced and reproduction fails to occur. This conclusion agrees with the δ13C signatures as well as the results of climate response analyses of tree-ring widths in Larix sibirica from the north-western Khentey (Dulamsuren et al. 2010c) and Larix gmelinii in central Siberia (Sidorova et al. 2009). The present lack of larch seedlings on the mountain slopes of the north-western Khentey is not only attributable to an inhibition of germination by high soil temperatures and low soil moisture (Dulamsuren et al. 2008), but also to the conspicuous lack of cones of larch trees in this area. This contrasts with other forest regions of Mongolia, where Siberian larch is often richly fertile. Increased temperatures during the growing season at constant precipitation, as in the south-western Khentey or the eastern Khangay, lead to less rigorously reduced growth and regeneration of L. sibirica. This is consistent with the fact that the tree line to the steppe is drought limited (Dulamsuren et al. 2009a), in contrast to alpine tree lines (Jacoby et al. 1996; D’Arrigo et al. 2000). Increased annual precipitation at constant summer temperature, as found in the south-eastern Khentey, results in constant or even improved growth and regeneration. Even >300-year-old larch trees are capable of resuming increased growth rates as a response to improving climatic conditions. This is inferred from Fig. 8, where an increasing annual increment is observed after 80 years of depressed growth at the end of the Little Ice Age in the late nineteenth century (Pederson et al. 2001). This behavior is characteristic of most old trees established in the eighteenth century or earlier. Crucial for the increased growth and regeneration of L. sibirica in the south-eastern Khentey is the increase in annual precipitation, and not the simultaneously increasing winter temperatures, as can be inferred from the summer drought-induced reduced tree-ring widths and regeneration in the south-western Khentey, despite the remarkable increase in winter temperatures since 1950 by 7.1°C (December–February), which is far above that in the south-eastern Khentey or the Mongolian average (Jacoby et al. 1999; Batima et al. 2005).
The present results for the Khentey and Khangay Mountains indicate that the improvement of growth conditions for L. sibirica by climate change may occur much more rarely in Mongolia than their deterioration. Considering the dominance of L. sibirica in the Mongolian taiga, this suggests a future loss of forest area, though Siberian larch might locally encroach into the steppe in the south-eastern Khentey and potential areas with similar climate trend. In addition to the direct effect of climate, more frequent forest fires and insect calamities in areas with increased aridity (Oberhuber 2001; Chapin et al. 2004; Hauck et al. 2008) may deteriorate conditions both for mature trees and seedlings of L. sibirica. Insect herbivory and fire, however, are unlikely to be the cause of the observed variation in tree-ring widths and regeneration between the four study areas, as three to five mountain slopes per area were studied. Insects or fire probably cause considerable variation between slopes of the same study area and the individual replicate plots on each slope, which was not observed.
In the case of the north-western Khentey, livestock grazing can be ruled out as the cause of the present lack of larch regeneration, in contrast to many other places in Mongolia. The present and historic land use of the specific study area in the north-western Khentey is known in detail (Schlütz et al. 2008). Pastoral nomads traditionally avoid the region with their livestock because of the poor accessibility and the small size of good pastures as well as the abundance of carnivores, including wolves and bears (Schlütz et al. 2008). The population densities of large herbivores, including deer, are low due to widespread poaching. In the south-western Khentey, increased livestock grazing can also be ruled out as the cause of reduced regeneration in the late twentieth century, since the plots studied there are located in one of the world’s oldest nature reserves at Mt. Bogd Uul (Hilbig et al. 2004). Therefore, increased aridity is the most plausible cause of the recently reduced or lack of reproduction in L. sibirica on mountain slopes. Recent larch regeneration in floodplains, even in the north-western Khentey, supports this conclusion. The dependence of seedling emergence on climate is also evident from parallels of peaks in the annual increment of mature trees with peaks in seedling establishment in all study areas (Figs. 4a, 7, 9a). Rich regeneration of larch in the 1930s and 1940s coincides with above-average precipitation throughout north-eastern Asia (Quian and Zhu 2001; Zhang et al. 2003). Relatively low documented numbers of seedlings in years of high annual stem increment before 1920 (Fig. 4a) are probably due to timber logging on the plots of the north-western Khentey in the late 1970s and 1980s (Schlütz et al. 2008), whereas the relative significance of past logging activities or natural mortality are not documented for the other study areas.
Though Matveev and Usoltzev (1996) found regeneration peaks in Siberian larch after fire due to the degradation of litter and herbs as well as reduced competition for water and nutrients, Danilin (1995) emphasized that fire only enhances the regeneration of Siberian larch, but is not mandatory. This agrees with our long-standing field experiences from the north-western Khentey, where larch seedlings in the river valleys definitely establish without preceding fire. Furthermore, sowing experiments in the western Khentey showed the germination of Siberian larch seeds in the field without any influence of fire (Dulamsuren and Hauck 2008). The slower growth of young trees than of old trees at a given age in three out of four study areas is not attributable to the assumption that the old trees represent the most quickly growing trees of their generation. Such an explanation would not match with the faster growth of young than old trees at a given age in the south-eastern Khentey. If effects of stand density play a key role in the tree-ring width of young trees, the stand density in the south-eastern Khentey should be lower than in the other study areas. However, Fig. 2 shows that the plots of the south-eastern Khentey are evenly distributed along the range of stand densities observed in the present study. This supports the hypothesis that the rich regeneration in the south-eastern Khentey and the high growth rates of the young trees result from the increased precipitation and not from the forest structure.
Conclusion
The high regional variability of global warming responses on a small geographical scale highlights that Mongolia’s natural forest resources have to cope with a highly diverse spatial pattern of climate trends. Regionally contrasting perspectives for the Mongolian taiga should be incorporated into the country’s development policy. Since declines of productivity and regeneration are more widespread than the opposite trend, a net loss of forests is likely to occur in the future, as strong changes in temperature and precipitation are predicted for the twenty-first century. Taiga forests harbor an important part of Mongolia’s biodiversity and are already under increasing pressure by the fast-growing Mongolian population, which heavily depends on the country’s natural resources. Any change in Mongolia’s vegetation pattern is not only a threat for Mongolia’s biodiversity and weak economy, but is also likely to interfere with the traditional nomadic lifestyle in the country. Tree increment is correlated with pasture biomass (Liang et al. 2003; Batima 2006). Over decades reduced stem increments may, thus, indicate an ongoing reduction of livestock carrying capacities in important rangeland areas of Mongolia and forewarn of a future disaster, as the Mongolian economy and cultural identity still heavily rely on nomadic pastoralism. Most of the 40% of the Mongolian population working in the agricultural sector pursues a nomadic lifestyle, which includes not only the utilization of pastures, but also of timber and other forest products (Neupert 1999; Havstad et al. 2008).
References
Baillie MGL, Pilcher JR (1973) A simple cross-dating program for tree-ring research. Tree Ring Bull 33:7–14
Batima P (2006) Climate change vulnerability and adaptation in the livestock sector of Mongolia. International START Secretariat, Washington, DC
Batima P, Natsagdorj L, Gombluudev P, Erdenetsetseg B (2005) Observed climate change in Mongolia. Assess Imp Adapt Clim Change Work Pap 12:1–26
Bayarsaikhan U, Boldgiv B, Kim K-R, Park K-A, Lee D (2009) Change detection and classification of land cover at Hustai National Park in Mongolia. Int J Appl Earth Obs Geoinf 11:273–280
Bohannon J (2008) The big thaw reaches Mongolia’s pristine north. Science 319:567–568
Böhner J, Lehmkuhl F (2005) Environmental change modelling for Central and High Asia: Pleistocene, present and future scenarios. Boreas 34:220–231
Briffa KR, Jones PD, Batholin TS, Eckstein D, Schweingruber FH, Karlén W, Zetterberg P, Eronen M (1992) Fennoscandian summers from AD 500: temperature changes on short and long timescales. Clim Dynamics 7:111–119
Chapin FS, Callaghan TV, Bergeron Y, Fukuda M, Johnstone JF, Juday G, Zimov SA (2004) Global change and the boreal forest: thresholds, shifting states or gradual change? Ambio 33:361–365
Cook ER (1985) A time series analysis approach to tree ring standardization. Dissertation, University of Arizona, Tucson
D’Arrigo R, Jacoby G, Pederson N, Frank D, Buckley B, Baatarbileg N, Mijiddorj R, C Dugarjav (2000) Mongolian tree-rings, temperature sensitivity and reconstructions of northern hemisphere temperature. Holocene 10:669–672
Danilin I (1995) Structure and biomass of larch stands regenerating naturally after clearcut logging. Water Air Soil Pollut 82:125–131
Dinesman LG, Knyazev AV, Kiseleva NK (1989) History of the steppe ecosystems of the Mongolian People’s Republic. Nauka, Moscow
Dugarjav Ch (2006) Larch forests of Mongolia (in Mongolian). Bembi San, Ulan Bator
Dulamsuren Ch, Hauck M (2008) Spatial and seasonal variation of climate on steppe slopes of the northern Mongolian mountain taiga. Grassland Sci 54:217–230
Dulamsuren Ch, Hauck M, Mühlenberg M (2005) Ground vegetation in the Mongolian taiga forest-steppe ecotone does not offer evidence for the human origin of grasslands. Appl Veg Sci 8:149–154
Dulamsuren Ch, Hauck M, Mühlenberg M (2008) Insect and small mammal herbivores limit tree establishment in northern Mongolian steppe. Plant Ecol 195:143–156
Dulamsuren Ch, Hauck M, Bader M, Osokhjargal D, Oyungerel Sh, Nyambayar S, Runge M, Leuschner C (2009a) Water relations and photosynthetic performance in Larix sibirica growing in the forest-steppe ecotone of northern Mongolia. Tree Physiol 29:99–110
Dulamsuren Ch, Hauck M, Bader M, Oyungerel Sh, Osokhjargal D, Nyambayar S, Leuschner C (2009b) The different strategies of Pinus sylvestris and Larix sibirica to deal with summer drought in a northern Mongolian forest-steppe ecotone suggest a future superiority of pine in a warming climate. Can J For Res 39:2520–2528
Dulamsuren Ch, Hauck M, Leuschner C (2010a) Recent drought stress leads to growth reductions in Larix sibirica in the western Khentey, Mongolia. Global Change Biol (in press)
Dulamsuren Ch, Hauck M, Leuschner HH, Leuschner C (2010b) Gypsy moth-induced growth decline of Larix sibirica in a forest-steppe ecotone. Dendrochronologia (in press)
Dulamsuren Ch, Hauck M, Leuschner (2010c) Climate response of tree-ring width in Larix sibirica growing in the drought-stressed forest-steppe ecotone of northern Mongolia. Ann For Sci (in press)
Dullinger S, Dirnböck T, Grabherr G (2004) Modelling climate change-driven treeline shifts: relative effects of temperature increase, dispersal and invasibility. J Ecol 92:241–252
Eckstein D, Bauch J (1969) Beitrag zur Rationalisierung eines dendrochronologischen Verfahrens und zur Analyse seiner Aussagesicherheit. Forstwiss Centralbl 88:230–250
Endo N, Kadota T, Matsumoto J, Ailikun B, Yasunari T (2006) Climatology and trends in summer precipitation characteristics in Mongolia for the period 1960–1998. J Meteorol Soc Jpn 84:543–551
Gunin PD, Vostokova EA, Dorofeyuk NI, Tarasov PI, Black CC (1999) Vegetation dynamics of Mongolia. Kluwer, Dordrecht
Hauck M, Dulamsuren Ch, Heimes C (2008) Effects of insect herbivory on the performance of Larix sibirica in a forest-steppe ecotone. Environ Exp Bot 62:351–356
Havstad KM, Herrick J, Tseelei E-A (2008) Mongolia’s rangelands: is livestock production the key to the future? Frontiers Ecol 8:386–391
Hayasaki M, Sugata S, Tanaka HL (2006) Interannual variation of cold frontal activity in spring in Mongolia. J Meteorol Soc Jpn 84:463–475
Helama S, Lindholm M, Timonen M, Eronen M (2004) Detection of climate signal in dendrochronological data analysis: a comparison of tree-ring standardization methods. Theor Appl Clim 79:239–254
Hilbig W (1995) The vegetation of Mongolia. SFB, Amsterdam
Hilbig W, Jäger EJ, Knapp HD (2004) Vegetation of Mt. Bogd-uul near Ulan Bator (Mongolia): site preferences and phytogeographical characters. Feddes Repert 115:265–342
IPCC (2007) Climate change 2007: the physical science basis. Intergovernmental Panel on Climate Change, Geneva
Jacoby GC, D’Arrigo RD, Davaajamts T (1996) Mongolian tree rings and 20th-century warming. Science 273:771–773
Jacoby GC, D’Arrigo RD, Pederson N, Buckley B, C Dugarjav, Mijiddorj R (1999) Temperature and precipitation in Mongolia based on dendroclimatic investigations. IAWA J 20:339–350
Kadota T, Davaa G (2005) Recent glacier variations in Mongolia. Proceedings of the Third International Workshop Terrestrial Change Mongolia, pp 20–22
Kobak KI, Turchinovich IYe, Kondrasheva NYu, Schulze E-D, Schulze W, Koch H, Vygodskaya NN (1996) Vulnerability and adaptation of the larch forest in eastern Siberia to climate change. Water Air Soil Pollut 92:119–127
Körner C, Sarris D, Christodoukalis D (2005) Long-term increase in climatic dryness in the East-Mediterranean evidenced for the island of Samos. Reg Environ Change 5:27–36
Korotkov IA, Dorjsuren Ch (1988) Dynamics of the phytocoenoses in pseudotaiga larch forests under the influence of logging and fire. Biologicheskie Resursi Prirodnikh Uslovii MNR 30:50–61
Kullman L (1998) Tree-limits and montane forests in the Swedish Scandes: sensitive biomonitors of climate change and variability. Ambio 27:312–321
Kullman L (2007) Tree line population monitoring of Pinus sylvestris in the Swedish Scandes, 1973–2005: implications for tree line theory and climate change ecology. J Ecol 95:41–52
Liang E, Vennetier M, Lin J, Shao X (2003) Relationships between tree increment, climate and above-ground biomass of grass: a case study in the typical steppe, north China. Acta Oecol 24:87–94
Luyssaert S, Schulze E-D, Börner A, Knohl A, Hessenmöller D, Law BE, Ciais P, Grace J (2008) Old-growth forests as global carbon sinks. Nature 455:213–215
Matveev PM, Usoltzev VA (1996) Post-fire mortality and regeneration of Larxi sibirica and Larix dahurica in conditions of long-term permafrost. In: Goldammer JG, Furyaev VV (eds) Fire in ecosystems of boreal Eurasia. Kluwer, Dordrecht, pp 366–371
Mazepa VS (2004) Stand density in the last millenium at the upper tree-line ecotone in the Polar Ural Mountains. Can J For Res 35:2082–2091
Miehe G, Schlütz F, Miehe S, Opgenoorth L, Cermak J, Samiya R, Jäger EJ, Wesche K (2007) Mountain forest islands and Holocene environmental changes in Central Asia: a case study from the southern Gobi Altay, Mongolia. Palaeogeogr Palaeoclim Palaeoecol 250:150–166
Morinaga Y, Tian S-F, Shinoda M (2003) Winter snow anomaly and atmospheric circulation in Mongolia. Int J Clim 23:1627–1636
Nandintsetseg B, Greene JS, Goulden CE (2007) Trends in extreme daily precipitation and temperature near Lake Hövsgöl, Mongolia. Int J Clim 27:341–347
Naurzbaev MM, Hughes MK, Vaganov EA (2004) Tree-ring growth curves as sources of climatic information. Quat Res 62:126–133
Nelson FE (2003) (Un)frozen in time. Science 299:1673–1675
Neupert R (1999) Population, nomadic pastoralism and the environment in the Mongolian Plateau. Pop Environ 20:413–441
Oberhuber W (2001) The role of climate in the mortality of Scots pine (Pinus sylvestris L.) exposed to soil dryness. Dendrochronologia 19:45–55
Pederson N, Jacoby GC, D’Arrigo RD, Cook ER, Buckley BM (2001) Hydrometeorological reconstructions for northeastern Mongolia derived from tree rings: 1651–1995. J Clim 14:872–881
Quian W, Zhu Y (2001) Climate change in China from 1880 to 1998 and its impact on the environmental condition. Clim Change 50:419–444
Sankey TT, Montagne C, Graumlich L, Lawrence R, Nielsen J (2006) Lower forest-grassland ecotones and 20th century livestock herbivory effects in northern Mongolia. For Ecol Manage 233:36–44
Sarris D, Christodoukalis D, Körner C (2007) Recent decline in precipitation and tree growth in the eastern Mediterranean. Global Change Biol 13:1187–1200
Sato T, Kimura F (2006) Regional climate simulations to diagnose environmental changes in Mongolia. Bull Terr Environ Res Cent Univ Tsukuba 7:59–69
Sato T, Kimura F, Kitoh A (2007) Projection of global warming onto regional precipitation over Mongolia using a regional climate model. J Hydrol 333:144–154
Schlütz F, Dulamsuren Ch, Wieckowska M, Mühlenberg M, Hauck M (2008) Late Holocene vegetation history suggests natural origin of steppes in the northern Mongolian mountain taiga. Palaeogeogr Palaeoclim Palaeoecol 261:203–217
Sidorova OV, Siegwolf RTW, Saurer M, Shaskin AV, Knorre AA, Prokushkin AS, Vaganov EA, Kirdyanov AV (2009) Do centennial tree-ring and stable isotope trends of Larix gmelinii (Rupr.) Rupr. indicate increasing water shortage in the Siberian north? Oecologia 161:825–835
Stendel M, Christensen JH (2002) Impact of global warming on permafrost conditions in a coupled GCM. Geophys Res 29. doi:10.1029/2001GL014345
Sugimoto A, Yanagisawa N, Naito D, Fujita N, Maximov TC (2002) Importance of permafrost as a source of water for plants in east Siberian taiga. Ecol Res 17:493–503
Tsedendash G (1995) The forest vegetation of the Khentey Mountains (in Mongolian). Dissertation, National University of Mongolia, Ulan Bator
Tsogtbaatar J (2004) Deforestation and reforestation needs in Mongolia. For Ecol Manage 201:57–63
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23:201–213
Wilmking M, Juday GP, Barber VA, Zald HS (2004) Recent climate warming forces contrasting growth responses of white spruce at treeline in Alaska through temperature thresholds. Global Change Biol 10:1–13
Zhang T, Serreze M, Barry RG, Gilichinsky D, Etringer A (2003) Climate change: evidence from Russian historical soil temperature measurements. Geophys Res Abstr 5:01485
Zimov SA, Schuur EAG, Chapin FS (2006) Permafrost and the global carbon budget. Science 312:1612–1613
Acknowledgments
The study was supported by a grant of the German Science Foundation (Deutsche Forschungsgemeinschaft) to Ch. Dulamsuren (Du 1145/1-1, 1-2). We are thankful to M. Runge (Göttingen) for constant advice. M. Mühlenberg (Göttingen) made available the facilities of Khonin Nuga Research Station in the north-western Khentey Mountains to us. S. Nyambayar (Ulan Bator) is thanked for his assistance in field work and with tree ring measurements in Göttingen. Prof. Dr D. Suran, L. Bazarragchaa, L. Jadambaa, and D. Osokhjargal (Ulan Bator) helped during field work.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Detlef Schulze.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Dulamsuren, C., Hauck, M., Khishigjargal, M. et al. Diverging climate trends in Mongolian taiga forests influence growth and regeneration of Larix sibirica . Oecologia 163, 1091–1102 (2010). https://doi.org/10.1007/s00442-010-1689-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-010-1689-y