Abstract
Key message
Beech and pine respond differently to climate change. June precipitation is of particular influence for beech, and February/March temperature for pine.
Abstract
Climate warming exposes forests to increasing abiotic stress, demanding for difficult silvicultural decisions about the right choice of future timber species. Scots pine (Pinus sylvestris) and European beech (Fagus sylvatica) are major timber species in the North German Lowlands, which have suffered from recent hot droughts, thus raising concern about their suitability for future production forests in the region. We investigated the climate sensitivity of tree growth and long-term growth trends of ten paired beech and pine forests along a precipitation gradient in the North German Lowlands with the aim to compare the species’ climate sensitivity and to search for species-specific climatic thresholds. In the majority of beech stands, basal area increment (BAI) has lost its positive trend since the 1980s or growth declined since then, while the BAI of pine has continually increased. Long-term change in June precipitation is in the study region a more important determinant of beech growth trends than the amount of MAP, while pine growth is largely dependent on the warmth of February/March. Yet, pine growth is also sensitive to dry mid summers, with sensitivity increasing toward low MAP. Climate sensitivity of growth has significantly declined since the 1980s in beech, while the dominant drought signal of June persisted in pine. We conclude that recent climate change is affecting radial growth of beech and pine differently with both species revealing signs of vulnerability to hot droughts, suggesting for the drier part of the study region the preference of more drought-tolerant hardwood timber species over beech and pine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forests play a key role for climate regulation, the water cycle, and as habitats of a rich biodiversity (FAO and UNEP 2020). Moreover, global forests provide humans with an annual harvest of c. 4 billion m3 roundwood, a highly valued resource (FAO 2021). The last four decades have seen rapid climate warming with an increase in the frequency and severity of extreme weather events (IPCC 2021). In parallel, the length of summer heat waves has doubled between 1880 and 2006 in western Europe (Della-Marta et al. 2007; Barriopedro et al. 2011). In many regions on earth, widespread reduction in tree vitality and increased mortality have been reported, which has been linked to the increasing aridification of climate in recent time (van Mantgem et al. 2009; Allen et al. 2010; Carnicer et al. 2011; Braun et al. 2020). Long-term decreases in summer precipitation have locally further increased the drought exposure of forests (Bat-Enerel et al. 2022).
Hot droughts like those of 2003 or 2018/2019 led in Central Europe to widespread dieback of many tree species, notably Norway spruce (Picea abies, Karst.), but also European beech (Fagus sylvatica L.), Scots pine (Pinus sylvestris L.), sessile oak (Quercus petraea (Mat.) Liebl.) and Douglas fir (Pseudotsuga menziesii (Mirbel) Franco) (Bigler et al. 2006; Schuldt et al. 2020; Braun et al. 2020; Obladen et al. 2021). This confronts foresters with the difficult task to establish forest stands with higher drought and heat tolerance, while maintaining timber production to meet a growing wood demand. These efforts have led to the definition of “climate-smart forestry” as a prime goal of future forest policy (Bowditch et al. 2020; Verkerk et al. 2020).
In central Europe, much of the former natural broadleaf forest cover has been replaced by conifer plantations during the last centuries, mostly of Norway spruce and Scots pine (Leuschner and Ellenberg 2017). In Germany, fast-growing conifers stock nowadays on 54% of the current forest area (Thünen-Institut 2022), whereas the natural cover of conifer forests would be less than 1% (Suck et al. 2014). In the North German Lowlands on Pleistocene substrates, where beech forests would naturally cover about 50% of the forested area, planted Scots pine stands represent 53.2% of the current forest area, while contributing with only about 2.3% to the former natural forest cover. Pine plantations occupy between 6% and 75% of the forest cover in the lowlands between the Dutch border in the west and river Oder in the east, while actual beech forest area was reduced to 2–22% of forest cover (Bohn and Neuhäusl 2003; Förster et al. 2021a; Thünen-Institut 2022; Leuschner et al. 2022).
Both beech and pine are important timber species in Central Europe (Müller 2007; Leuschner 2020). Pine is a light-demanding pioneer tree species with marked tolerance of nutrient-poor and dry soils (Schütt and Stimm 2006; Roloff 2008). Distributed through Eurasia from the Atlantic in the West to north-eastern Russia, the species is predominantly a boreal taxon with some of its range extending till the northern Mediterranean region (Carlisle and Brown 1968). Due to its broad range of tolerated climatic and edaphic conditions, Scots pine is the second-most widespread conifer and most widespread pine species of the world (Eckenwalder 2009). Its natural occurrence in Central Europe is on sand dunes (Dicrano-Pinion communities), shallow south-exposed limestone slopes (Erico-Pinion communities) and in swamp forests (e.g. Ledo-Pinetum community) (Leuschner and Ellenberg 2017).
European beech is the most widely distributed species of the genus Fagus (Fang and Lechowicz 2006). Its natural distribution is centered in the temperate zone of Europe and ranges from southern Italy to southern Norway and from northern Spain to eastern Romania (Bohn and Gollub 2007). Beech is a shade-tolerant late-successional species that is susceptible to drought and thus requires sufficient moisture in summer and mild temperatures in winter (Packham et al. 2012; Leuschner 2020).
To meet the challenges of climate change and weather extremes and to reach the declared goal of increasing the conservation status of forest biota (BMUB 2007; EU 2011), the forestry sector of various Central European countries including Germany has undertaken efforts to increase the cover of natural hardwoods at the expense of existing conifer plantations (Bauhus et al. 2021; Bundesministerium für Ernährung und Landwirtschaft (BMEL) 2021). The National Biodiversity Strategy (NBS) of Germany aims at expanding the cover of the natural forest vegetation, which would in large areas be Fagus sylvatica (Bohn and Neuhäusl 2003; Bauhus et al. 2021). Such a shift may also be necessary in the face of climate warming, as the main conifers planted in Central Europe are predominantly boreal species with an assumed adaptation to cooler climates (Hanewinkel et al. 2013; Schueler et al. 2014; Dyderski et al. 2018). Climate projections predict that Scots pine will lose 60% of its present range in Europe in future, depending on the climate scenario (Hanewinkel et al. 2013; van der Maaten et al. 2017; Thurm et al. 2018). However, beech likely will also meet its drought and heat limits in part of its Central European distribution range (Mellert et al. 2016; van der Maaten et al. 2017; Fischer 2019).
In fact, increasing evidence shows that beech is more drought sensitive than many other temperate broadleaf tree species, including the oaks, common ash, common hornbeam and small-leaved linden (Köcher et al. 2009; Rasztovits et al. 2014; Brinkmann et al. 2019; Leuschner 2020). Dendrochronological analyses demonstrate that the radial growth of beech responds negatively to low precipitation and high temperatures in previous year’s or current summer (Scharnweber et al. 2011; Knutzen et al. 2017; Harvey et al. 2020). Long-term growth decline, elevated mortality and even replacement of beech by more drought-tolerant tree species in the course of climate warming were first observed at the species’ southern distribution limit in south-western and south-eastern Europe (Jump et al. 2006; Piovesan et al. 2008; Lakatos and Molnár 2009). However, beech has also been found sensitive to drought and heat in its more northern and north-eastern distribution regions, where it also became increasingly sensitive to winter cold (Farahat and Linderholm 2018; Weigel et al. 2018). In Central Europe, various studies have recently reported negative growth trends from about the 1980s onward, e.g., in northeastern France (Charru et al. 2010), Switzerland (Braun et al. 2017), Belgium (Aertsen et al. 2012) and south-western, central and northern Germany (Scharnweber et al. 2011; Zimmermann et al. 2015; Dulamsuren et al. 2017; Knutzen et al. 2017). Hacket-Pain et al. (2016) suggested that geographical variation in the climate sensitivity of beech may not be strong. However, recent studies indicate higher climate sensitivity and lower resistance to drought at sites in the center and north of the distribution range than in southern-edge populations (Cavin and Jump 2017; Muffler et al. 2020). It is assumed that this is related to local drought adaptation in the southern populations with their higher inter-population genetic diversity (Magri et al. 2006; Cavin and Jump 2017; Muffler et al. 2020). The severe drought episode of 2018/19 has resulted in canopy dieback and tree death in various Central European beech populations, which hints at the vulnerability of the species in the face of climate change (Braun et al. 2020; Schuldt et al. 2020; Obladen et al. 2021).
In contrast to beech, pine shows distinct differences in its climatic sensitivity within its large range. At the southern range edge, summer heat and drought are the principal growth-limiting factors, which are causing increased mortality with advancing warming in certain regions (Giuggiola et al. 2010; Matías et al. 2017; Archambeau et al. 2020). In the north, in contrast, the establishment and growth of pine are primarily limited by low temperatures (Rickebusch et al. 2007; Mathisen and Hofgaard 2011; Matías and Jump 2014). With the recent warming trend, increasing growth has been observed in these northern populations and is predicted to continue in future (Mielikäinen and Sennov 1996; Reich and Oleksyn 2008; Hickler et al. 2012; Socha et al. 2021). Central Europe can be considered as a transition zone (Bauwe et al. 2013, 2015) between the increasing growth trends in the north (Juntunen et al. 2002; Reich and Oleksyn 2008; Kullman 2014; Matías and Jump 2014) and prevailing negative growth trends in the south, where dieback is more frequent (Martínez-Vilalta et al. 2008; Reich and Oleksyn 2008; Giuggiola et al. 2010; Matías and Jump 2014). Evergreen Scots pine is further influenced by the temperature increase in winter, spring and autumn, which is hypothesized to lead to a marked extension of the growing season and growth stimulation (Lebourgeois et al. 2010; Harvey et al. 2020). In the North German Lowlands, Diers et al. (submitted) and (Stolz et al. 2021) suggested that the positive effect of warmer winters currently may offset the negative effect of dry summers, resulting in an overall positive growth trend.
It is obvious that the climate sensitivity of growth of beech and pine differs not only between the species but also across the distribution ranges. Forestry needs more information on climate change effects on the growth and vitality of these species to define hydrometeorological and thermal thresholds to which beech and pine can be cultivated under the perspective of a warming and drying climate. A few dendrochronological analyses of beech and pine growth exist for the North German Lowlands that address the species’ climate sensitivity (Scharnweber et al. 2011; Bauwe et al. 2013; Knutzen et al. 2017), some of which include a direct species comparison. Mostly, these studied do not explicitly address the pronounced gradient in climate continentality that characterizes the North German Lowlands, but rather focus on north-eastern Germany (Bauwe et al. 2015; Stolz et al. 2021) or the south Baltic coast in the transition between temperate and hemi-boreal climates (Harvey et al. 2020).
We conducted a dendrochronological paired-plot study in nearby beech and Scots pine stands at ten sites that covered the climate continentality gradient from the oceanic North Sea coast to sub-continental eastern Germany near the Polish border (830–560 mm mean annual precipitation, MAP) in full length. The stands also include assumed dry-edge populations in the study region (Kölling 2007). By selecting beech and pine stands on similar soil, we attempted to minimize the influence of specific edaphic conditions on growth patterns and climate sensitivity. Our research was guided by the following three questions:
-
i)
How do beech and pine differ in their climate sensitivity of growth on similar soil?
-
ii)
How has climate sensitivity changed during the last decades in the two species?
-
iii)
How do climate sensitivity and growth trends vary regionally and along the climate continentality gradient?
Materials and methods
Study sites
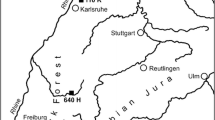
The study took place in ten pairs of European beech and Scots pine forests in the lowlands of northern Germany. The forest stands are located along a climate gradient from a cool-temperate oceanic climate close to Bremen in the west to a cool-temperate sub-continental climate in the east near river Oder at the Polish border (Table 1). Mean annual temperature varied between 9.0 and 9.7 °C and mean annual precipitation (MAP) decreased from 823 mm year− 1 to 564 mm year− 1 along the west–east gradient (Fig. 1). Temperatures have increased in the 1960–2017 observation period in both winter and summer, while precipitation increased only in winter (DWD 2020). All stands stock on acid, relatively nutrient-poor soils developed in glacial sandy to sandy-loamy deposits (except for beech stand no. 10 on Triassic sandstone) (Diers et al. 2021). The selected 20 forests are mature stands with closed canopy (canopy closure > 80%), which had been managed in the past by regular thinning. The beech stands had an age of 95–195 years, the pine stands of 55–74 years, which is about 10–20 years before final harvest in pine, while the beech stands were both younger and older than typical harvest age (about 120 years). In the beech stands, the last thinning operations took place 6–43 years ago, while the pine stands were last thinned approximately 2–14 years ago.
Location of the study sites in the North German Lowlands (inlet shows location of the lowlands within Germany). Area colored by mean annual precipitation (MAP, 1991–2020, DWD Climate Data Center (CDC) 2022). Size and color of dots indicate change in June precipitation (ΔJune precipitation) between the earlier (1960–1988) and later (1989–2017) observation period
Study design and wood core sampling
In each of the ten forest pairs, ten circular plots were established at random positions in the stands (100 circular plots for pine and 100 for beech in total). The plots were 35.68 m in diameter (size 0.1 ha) and their edges were at least 30 m distant from each other. Edge effects were minimized by maintaining a distance of at least 20 m from forest paths and the forest edge.
Each two dominant trees with their crown in the upper canopy layer were selected in every plot and one wood core per tree was extracted at breast height (1.3 m) between December 2017 and April 2018 (i.e., two cores per plot, 20 cores per forest site, and in sum 200 trees sampled per species). Using an increment corer of 5 mm in diameter (Haglöf, Längsele, Sweden), the wood cores were extracted from the western side of a tree except for trees on slopes, which were sampled parallel to the slope inclination to avoid the influence of tension and compression wood. The wood cores were glued on wooden mounts; in case of pine cores, a plain surface was cut on the samples with a microtome, while the beech cores were sanded with sanding paper. Pine samples were colored with safranin, and both beech and pine samples were powdered with chalk in order to increase color contrasts.
Tree ring data
We used a moveable measuring table (Lintab 5, Rinntech, Heidelberg) to measure tree-ring widths of the cores. The samples from each forest were then cross-dated based on the coefficient of agreement (‘Gleichläufigkeit’, GLK; Eckstein and Bauch (1969)), the cross-dating index (CDI; Dobbertin and Grissino-Mayer (2004)), and Student’s t value (TVBP; Baillie and Pilcher (1973)), using TSAP-Win software (Rinntech, Heidelberg, Germany; Rinn 2012)). In most cases, the 20 tree-ring series per forest site had a GLK > 65%, a CDI > 2.0, and a TVBP > 3.5. In a few cases, we excluded 1–2 tree ring series due to low GLK. We used R software version 4.0.4 (R Core Team 2021) for the further analysis of the tree ring data using the packages “dplR” (v.1.7.2, Bunn et al. (2021)) and “treeclim” (v.2.0.5.1, Zang and Biondi (2015)).
The tree-ring series were detrended to the dimensionless ring-width index (RWI) using the function “detrend” (dplR, Bunn et al. (2021)), which applies a smoothing spline function with 20 years (pine) or 30 years (beech) moving windows with frequency cutoff at 50% (Cook and Peters 1981) for eliminating the age trend or long-term effects of forest management. We built stand chronologies from the each (18–) 20 standardized RWI-series per forest stand for all ten forest sites (ten chronologies per species) with Tukey´s bi-weight robust mean (Bunn and Korpela 2021) using the function “chron” (dplR). Chronologies were considered reliable when the expressed population signal (EPS) exceeded 0.85 (Wigley et al. 1984).
Long-term growth trends were analyzed based on time series of annual basal area increment (BAI) which was obtained from the raw tree-ring data with the equation:
where r is the measured stem radius, t the given year and BAIt the corresponding basal area increment. The BAI.out function (dplR, Bunn et al. (2021)) was used to analyze BAI trends for each tree calculated from the DBH data measured in the field and the ring-width series from younger to older rings. From the individual BAI series, we built a BAI chronology separately for each site.
Climate data
Monthly temperature and precipitation data for the period from 1960 to 2017 were extracted from the German Climate Data Center (CDC) of the German Weather Service (DWD, Deutscher Wetterdienst, Offenbach, (DWD Climate Data Center (CDC) 2019a, b)) using the R package “rdwd” v.1.4.0 (Boessenkool 2021). We computed the monthly values of the Standardized Precipitation Evaporation Index (SPEI across a 6 months period) for each site as an indicator of climatic drought intensity using the R package “SPEI” v.1.7 (Beguería and Vicente-Serrano 2017).
Climate–growth analysis and growth trend assessment
To explore the importance of selected climate variables for radial growth, we correlated the annual RWI data with the monthly climate data, considering monthly averages from previous year’s June to current-year September, using the routine “dcc” (“treeclim”, v.2.0.5.1, Zang and Biondi (2015)). Pearson’s r was used as a measure of the strength of the climate–growth relationship. We performed this analysis for each site and species separately. Furthermore, we split the analysis in two periods of equal length of 29 years (earlier period 1960–1988 with only slight temperature increase, later period 1989–2017 with more pronounced warming) to account for assumed non-stationarity of climate–growth relationships (Wilmking et al. 2020) during the observation period. Significance (p < 0.05) of the climate–growth relationships was assessed through a 1000-fold bootstrapping procedure (function “dcc” in “treeclim”) which allowed calculating confidence intervals of Pearson’s r for significance testing (Efron and Tibshirani 1986).
Based on the BAI chronologies, we calculated growth trends by correlating BAI and calendar year for each species, forest site and observation period separately. The significance of trends was assessed with the Mann–Kendall trend test (R-package “Kendall”, McLeod (2015)). Subsequently, we analyzed correlations between the growth trend and long-term averages and changes of climate factors of the periods 1960–2017, 1960–1988 and 1989–2017 that were identified as important limiting factors in the climate–growth analysis (Figs. 2, 3, 4 and 5), notably summer precipitation, June precipitation, the difference in June precipitation between the two periods, summer SPEI, and the climatic water balance (CWB) of the summer.
Correlation coefficients (Pearson's r) for the ten forest sites (ordered by decreasing MAP) and different months for the relation between RWI and precipitation in different months for the two species in the observation period 1960–2017. Significant (p < 0.05) r values according to a 1000-fold bootstrapping procedure are printed in red. Months of the previous year in small letters, months of the current year in capitals
Correlation coefficients (Pearson's r) for the ten forest sites (ordered by decreasing MAP) and different months for the relation between RWI and temperature in different months for the two species in the observation period 1960–2017. Significant (p < 0.05) r values according to a 1000-fold bootstrapping procedure are printed in red. Months of the previous year in small letters, months of the current year in capitals
Correlation coefficients (Pearson's r) for the ten forest sites (ordered by decreasing MAP) and different months for the relation between RWI and the SPEI of different months for the two species in the observation period 1960–2017. Significant (p < 0.05) r values according to a 1000-fold bootstrapping procedure are printed in red. Months of the previous year in small letters, months of the current year in capitals
Spatio-temporal variation in correlation coefficients (Pearson's r) for the relationship of ring width (RWI) of beech and pine with monthly temperature, precipitation or the standardized precipitation evapotranspiration index (SPEI) for the periods 1960–1988 (red boxes) and 1989–2017 (blue boxes), demonstrating non-stationarity of climate–growth relationships since the 1960s. Each monthly box with whiskers represents the spatial variability of climate sensitivity, as it depicts the distribution of the ten climate–growth correlations calculated separately for the ten stands. The threshold for significant correlations is indicated by the red lines (n = 29 years), p < 0.05 corresponds to |r| > 0.38. The black dots mark outlying correlation coefficients of individual stands
Results
Climate sensitivity of growth in beech and pine
Radial growth of beech showed in the observation period 1960–2017 a significant positive impact of June precipitation in the current year at the four sites with lowest MAP, while this effect was visible only at two of the moister sites (no. 4 and 7; Fig. 2). A similar picture emerged also for pine, but this species’ growth was also significantly stimulated by July precipitation especially at the drier sites. In contrast to pine, beech growth also profited from higher precipitation in previous year’s July and June especially at the moister sites (and also at the two driest sites no. 13 and 14). Moreover, it appears that stands with a greater influence of previous year’s precipitation are characterized by a smaller influence of current-year precipitation, as is observed at the moister sites. Carry-over effects of previous’ summer precipitation on growth are lacking in pine.
The temperature influence on growth differs markedly between the two species (Fig. 3). In beech, growth was negatively influenced by higher temperatures in previous year’s July (and partly also August and September), or alternatively by a warmer current-year June. Winter temperature was of no relevance in our beech stands (except for a positive effect of December temperature in stand 11). Pine shows a very distinct growth stimulation by higher temperatures in February and March of the current year at most sites. Higher temperatures in current-year May and June (and July) had a negative effect on pine growth at some of the drier sites, while three pine stands were also negatively affected by higher temperatures in previous year’s July, August and September (Fig. 3).
A lower climatic drought exposition (higher SPEI) in previous year’s late summer and autumn (July–December) had a significant positive influence on beech growth at the four moistest and two driest sites (Fig. 4). Elevated current-year SPEI values in spring and summer stimulated beech growth in five of the sites, mostly with lower MAP. Two sites (no. 11 and 16) showed no significant SPEI influence on beech growth at all. Most pine stands showed a positive influence of reduced drought exposition in current-year June to September on growth. The driest stand (no. 14) was also stimulated by a higher SPEI in previous year’s June–December. Only one of the pine stands (no. 8) was also stimulated by a lower drought exposition of previous year’s June. Pine stand no. 9 revealed no significant SPEI effect on growth in any month.
Temporal change in the climate sensitivity of growth
Beech shows a distinct shift in climate sensitivity between the 1960–1988 and the 1989–2017 period. The significant climate–growth relationships depicted in Figs. 2, 3 and 4 for the 1960–2017 period are in case of SPEI, temperature and precipitation more pronounced in the earlier period and diminish toward the second period (Fig. 5). The significant negative correlations of beech growth with June temperature (current year), and July and August temperature (previous year) as well as positive correlations with current-year June precipitation in the 1960–1988 period, have disappeared in the 1989–2017 period. The same is true for the significant positive correlation with previous year’s SPEI from June–December, which has disappeared in the second period. In fact, beech growth shows no significant sensitivity to any of the three climate variables in the recent observation period any more (Fig. 5). The overall climate sensitivity of beech clearly has decreased compared to the earlier period. A moving-window analysis shows that especially at the sites, where June precipitation has decreased (or remained stable) since the 1960–1988 period, growth sensitivity has decreased (decreasing r value), while it remained constant at sites with stable or increasing June precipitation (Fig. 8, Appendix). In pine, no general long-term trend toward decreasing climate sensitivity is visible. Yet, the positive effect of June precipitation and of lower drought exposition (SPEI) in August and September on growth has disappeared from 1960–1988 to 1989–2017, while the sensitivity to July precipitation and SPEI has remained (Fig. 5). The stimulating effect of higher February temperature has become significant only after 1988.
a Mean annual precipitation (1989–2017), b difference in June-precipitation (Δ-precipitation June) between the 1960–1988 and 1989–2017 period (negative values: decrease), and c growth trends in the period 1989–2017 of beech and pine in the ten forests obtained by regressing the BAI chronology against year. Forest sites are ordered according to the Δ-precipitation June value (decreasing from left to right). Asterisks show significant BAI trends
Long-term growth trends
The ten studied sites differ in their long-term trend of June precipitation, which has increased at three sites (no. 11, 13 and 14), but decreased at seven sites from the 1960–1988 to the 1989–2017 period (Fig. 6b). The long-term trend in June precipitation was not related to the long-term MAP of the sites (Fig. 6a). Interestingly, the two driest sites (13, 14) are characterized by an increase in June precipitation. The long-term BAI trend of beech was positive at the three sites with a June precipitation increase, but was negative at all sites with a June precipitation decrease (except for stand no. 7 with a slight growth increase despite decreasing June precipitation; Figs. 6c and 7 in the Appendix).
A correlation analysis between the slope of the growth trend (Pearson’s r) and the size of the difference in June precipitation between earlier and later period shows a significant positive relation for beech (Table 2). Since the extent of June precipitation decline is unrelated to MAP (r = − 0.44, p > 0.05, Fig. 6a & b), no relationship between site MAP and the extent of beech growth decline does exist (r = − 0.46, n.s.).
For pine, this relation is not significant, as positive long-term growth trends appeared not only with increasing June precipitation, but also at several sites with a decreasing June precipitation (Fig. 6c). In fact, a negative growth trend was only recorded at pine site no. 8.
As the time since management had been abandoned varied in the beech stands between 6 and 43 years, we additionally examined the influence of this factor on growth trends. Accordingly, beech BAI was not influenced by the time since management abandonment (Table 3, Appendix).
Discussion
Differential climate sensitivity of beech and pine growth
Our comparison of beech and pine growth patterns at nearby sites in the North German Lowlands reveals marked differences in the climate sensitivity of the two species, which are partly explained by the different tree functional types (deciduous broadleaf vs. evergreen conifer). Pine growth is in the studied ten stands primarily influenced by the weather conditions of the current year, both in late winter/early spring and in summer, while beech growth is to a larger degree dependent on the weather of the previous summer. This reflects the different carbohydrate allocation patterns of the two life strategies. According to Čufar et al. (2008), xylem growth in deciduous beech starts in Central Europe at the same time as leaf unfolding and usually peaks in June with about 35% of the current ring width being formed. Beech uses a large part of the stored starch reserves of previous year’s carbon gain to enfold its foliage in April/May and advance cambial growth in early summer. After cessation of ring formation, the beech foliage continues to photosynthesize for about 2–3 months, with the carbohydrates mainly transferred to storage organs, which explains the carry-over effect in terms of past weather influences on beech growth (Čufar et al. 2008). If the summer is hot and dry, the reserves cannot be built up sufficiently and less growth is possible in the following year. The development of cumulative soil water deficits in consecutive dry years as in 2018/19 may amplify the depletion of carbohydrate stores, thereby increasing the precipitation and SPEI effects on beech growth (Scharnweber et al. 2020). In addition, high temperatures and radiation intensities during summer droughts trigger masting in beech in the following year (Müller-Haubold et al. 2015; van der Maaten-Theunissen et al. 2016; Hacket-Pain et al. 2018), causing reduced radial growth and thus amplifying the effect of previous summer precipitation and SPEI on beech growth.
In contrast, evergreen pine produces new needles only from mid-summer onwards and thus is able to invest the C assimilated from late winter to early summer primarily into radial growth. This also explains the strong February/March temperature signal visible in pine radial growth. Higher fruit production in pine in certain years usually does not lead to reduced radial growth, and no change in the frequency of Scots pine fructification has been observed with climate warming in Europe so far (Martínez-Alonso et al. 2007; Nussbaumer et al. 2016). While low summer precipitation in the current year reduces the growth of both species, beech is sensitive mainly to low June precipitation, while pine is negatively impacted by low June and especially July rainfall. We speculate that cambial activity is impaired by water deficits in early/mid summer in both species, but pine frequently responds to dry spells later in summer by pre-senescent shedding of the oldest (2 years) needle generation, which may reduce C assimilation, while pre-senescent leaf browning and shedding occurs in beech only during exceptional droughts (Braun et al. 2020; Leuschner 2020). Pine showed a distinct influence of previous year’s drought exposition on growth only at the driest study site (no. 14) with a MAP of only around 560 mm year− 1. We speculate that, at this site, needle loss or fine root dieback in dry summers might have weakened the vitality of the trees with the consequence of reduced growth in the following year.
In addition to differing weather effects, pine and beech reveal contrasting recent growth trends in the study region. In pine, four of the stands showed a significant positive growth trend in the 1989–2017 period, and another five an insignificant growth increase or no change, while only one stand (site no. 8) revealed a significant recent growth decline. This indicates that increasing climatic aridity (and locally decreasing summer precipitation) in the course of climate warming has not yet negatively affected radial growth of pine at most of the studied sites. In beech, in contrast, decreasing growth trends were clearly related to climate aridification in summer at the sites, indicating a higher drought vulnerability than in pine.
Whether trees withstand drought at drier sites may partly depend on the species’ capacity to acclimate to low site water availability. A fine root study at our sites found along the precipitation gradient increases in fine root biomass and total root surface area toward the drier pine stands, which points at a plastic root system response to water deficits in Pinus (Förster et al. 2021b). A similar response lacked in the beech stands (Förster et al. 2021b). Yet, an earlier study has reported increases in fine root biomass and productivity with decreasing MAP in other North German beech forests (Hertel et al. 2013). Contrary to expectation, evidence for deeper rooting at drier sites is scarce for mature beech stands (Meier et al. 2018). Moreover, beech has been found to increase average leaf size and stand leaf area from moister to drier sites along precipitation transects in northern and central Germany (Meier and Leuschner 2008; Weithmann et al. 2022), which we interpret as an additional weak point in the drought response strategy of beech that could explain the apparently higher drought sensitivity.
The marked positive effect of higher February/March temperatures on pine radial growth suggests that the growth-depressing effect of summer drought is still compensated by the positive effect of late-winter and early-spring warming (Stolz et al. 2021). In addition, Scots pine has been found to clearly profit from the extended growing season, which may cancel out assumed carbon gain reductions due to water deficits and high temperatures in summer (Martinez del Castillo et al. 2018). Beech, in contrast, has profited from the recent warming through growing season extension less than other broadleaf species including the oaks (Menzel et al. 2003; Vitasse et al. 2009). Despite a so far less consistent drought signal in pine growth in comparison to beech, the significant current-year precipitation and SPEI sensitivity of pine growth and its increase toward dry sites indicate that advancing climate warming and aridification will have an increasingly negative influence on this species in the future as well.
Recent decline in the climate sensitivity of growth in beech
We found a marked change in the climate sensitivity of growth between the 1960–1988 and the 1989–2017 observation periods in both species. While pine exhibited an increase in the winter temperature-sensitivity of growth, as it was also observed by Stolz et al. (2021) in our study region, the growth sensitivity of beech to summer precipitation, temperature and drought (SPEI) declined over the last three decades with the consequence that none of the tested climate variables exerted a significant influence on beech growth in the recent observation period any more. The latter finding was unexpected, as it contrasts with results reported from dendrochronological studies on beech and other tree species in central to northern Europe (Scharnweber et al. 2011; Kint et al. 2012; Harvey et al. 2020; Stolz et al. 2021), even though declining sensitivity of beech growth to summer drought has been noticed previously at some sites in Europe (Hacket-Pain and Friend 2017; Muffler et al. 2020; Weigel et al. 2022).
Our results also do not fit to the findings of van der Maaten-Theunissen et al. (2016) who reported for beech a shift in drought sensitivity from current-year to previous-year weather influences, as climate sensitivity of beech growth decreased in our data in the recent year as well. These authors explained the observed shift in drought sensitivity toward the previous year by the marked temperature increase in the second half of the 20th century, which increases the atmospheric evaporative demand and would render the water status of the previous year and soil water recharge in winter more important (van der Maaten-Theunissen et al. 2016; Scharnweber et al. 2020).
A decrease in climate sensitivity of beech growth since about 1990 was also observed by Hacket-Pain and Friend (2017) at the southern distribution range of the species, where growth rate is increasing despite a relatively warm and dry climate and a warming trend. The authors explain the unexpected drought resistance of beech with successful adaptation to the local climatic conditions. Comparable observations were made by Fuchs et al. (2021) who report a decreasing growth sensitivity in recent decades in several temperate broadleaf tree species in a dry region in Central Germany, which was accompanied by a constant or positive growth trend. The authors also assume successful acclimation or adaption to increasing drought in these species in recent time (Fuchs et al. 2021).
Our results deviate from these findings in that the sensitivity decrease was accompanied by stable or declining growth rates in beech. A possible explanation may be provided by the “sensitivity loss” phenomenon, i.e., non-stationarity in the climate–growth relationship, which was first observed in northern forests since the mid 20th century and has now been reported from various regions around the globe (Wilmking et al. 2020). We speculate that the beech trees of our study coped fairly well with the drought regime prevailing in the 1960s, 70s and 80s, even though being sensitive to summer drought and heat, which resulted in positive growth trends in that time. With increasing warming and climate aridification especially since the 1980s, the BAI of beech has ceased to increase or even declined since then in part of the study region, resulting in smaller tree rings which weakens the climate signal. Particularly influential is a decrease in June precipitation due to its large influence on radial growth in beech (Weigel et al. 2022). In fact, regions with a decadal decrease in June precipitation show the greatest decrease in beech radial growth and also in growth sensitivity to climate since the 1960–1988 observation period. This result differs from the findings of Stolz et al. (2021) who report an increasing growth sensitivity to drought in beech in the last decades in part of the North German Lowlands. We assume that this discrepancy to our results is partly explained by the considerable regional variability in climate and also climate trends in the lowlands (Bat-Enerel et al. 2022), with the north-western coastal region experiencing a recent humidification of climate and positive growth trends in beech, while the climate became more arid further inland. Moreover, June precipitation has decreased in parts of the central and south-eastern lowlands with negative effects on beech growth (see Fig. 1), while precipitation increased elsewhere. This could explain increases and decreases in climate sensitivity of growth in neighboring regions.
Possible influences of tree age and size, and forest management
An alternative explanation of the apparently greater summer drought sensitivity of beech than pine growth and the weakening of the drought signal in beech growth over time could be the ageing of trees between the earlier and later observation period. Since the studied pine trees were on average about 78 years younger than the beech trees, tree size and age differences between beech and pine might have enhanced the species differences in climate-sensitivity of growth. As regular thinning is usually promoting the radial growth of the remaining trees, the more recent occurrence of stand thinning in the pine forests as compared to the beech forests could be one explanation for the prevalence of positive recent growth trends in this species, compared to beech. Moreover, the beeches were on average also larger than the pines in our study. Taller trees have frequently been found to be more sensitive to climatic signals than smaller ones due to greater canopy exposure to the atmosphere (Mérian and Lebourgeois 2011; Zang et al. 2014; Hacket-Pain et al. 2016). Yet, a recent leveling-off or decline in radial growth has been observed in parts of the study region also in beech stands with more recent thinning (Knutzen et al. 2017). In addition, canopy exposure was not different between the cored beeches and pines, as all trees were dominant in the upper canopy layer. This makes it likely that the growth trends revealed in our study are caused by a greater climate change impact on beech than on pine growth.
While tree ontogeny can indeed modulate climate sensitivity (Carrer and Urbinati 2004; Gallardo et al. 2022), the tree age effect on climate sensitivity seems to vary with species and environment, being greater either in older or younger trees (Pompa-García and Hadad 2016; Jiao et al. 2017; Peng et al. 2018; Sanchez-Salguero et al. 2018). Instead of age, Trouillier et al. (2019) suggested that climate sensitivity is rather affected by the tree size change throughout a tree’s lifespan, which is increasingly acknowledged in tree-ring studies that address the effects of tree ontogeny (Bernal-Escobar et al. 2022; Martinez del Castillo et al. 2022). As xylem vessels usually widen in the stem xylem with increasing stem diameter in order to compensate for increasing flow resistances in the xylem path with height growth, higher drought sensitivity in older, taller trees could result from larger vessels and increased xylem vulnerability to cavitation (Anfodillo et al. 2013). An increase in climate sensitivity with age was indeed demonstrated for beech growth by Latte et al. (2015). Mérian and Lebourgeois (2011) thus concluded that shade-tolerant tree species, such as beech, are more sensitive to summer drought at larger than smaller size when age is similar, likely due to larger vessel diameters in young annual rings and the exposure of a larger crown to the atmosphere. This seems not to be valid for Scots pine as a shade-intolerant tree species, where no clear differences in drought sensitivity were found between size (diameter) classes (Mérian and Lebourgeois 2011). Yet, the temperature sensitivity of pine radial growth has been found to decrease with cambial age (Konter et al. 2016). These species-specific patterns offer no explanation for the observed sensitivity decline over time in beech, but it would suggest that the higher climate sensitivity of beech growth compared to pine is not caused by the higher age of the studied beeches.
Recent changes in forest management might also be considered as explanation for the recently decreasing sensitivity of beech growth, as growth often decreases when thinning is abandoned in managed stands. Indeed, Mausolf et al. (2018) observed a concomitant decrease in growth sensitivity to climate fluctuation in beech forests with management cessation. In correspondence, Bosela et al. (2016) found only managed beech stands in Slovakia to be negatively affected by increasing drought frequency in recent time, but not unmanaged stands. In fact, heavy thinning in beech forests may contribute to increased tree mortality even 60 years later, probably due to an increase in drought sensitivity after stand opening (Dulamsuren et al. 2022). This contrasts with the results of van der Maaten (2013), who found a decrease in drought vulnerability in German beech forests upon thinning. In our study, in only three of six beech stands with decreasing growth sensitivity, management had been abandoned since the earlier observation period; in the others it continued. In correspondence, the time since management abandonment had no influence in our model built to explain recent growth trends. Thus, it is not yet clear how forest management has affected the climate sensitivity of beech growth in our study. Further research is needed to convincingly explain the observed decline in beech growth sensitivity in our stands.
Regional variation in growth trends and their drivers
While the recent growth trend of the pines is positive at nearly all studied sites despite increasing climatic aridity, irrespective of MAP or recent trends in June precipitation, beech growth trends vary across the study region. We identified the direction of June precipitation change between the 1960–1988 and 1989–2017 period as the most important influential factor for this variation in beech. Regions with largest declines in June precipitation as southern Lower Saxony, Saxony-Anhalt and the south of Brandenburg with reductions by up to 10 mm and significant negative growth trends contrast with coastal regions where June precipitation has increased, and beech growth trends were mostly positive. However, negative growth trends can also be detected in regions with June precipitation increase due to the influence of other, non-climate drivers (Weigel et al. 2022). While the negative correlation between June precipitation trend and beech growth trend was highly significant (r = 0.86, p < 0.001), our results do not allow defining a MAP threshold, which separates positive and negative growth trends in beech. Possible reasons are the relatively low number of study sites in our study (N = 10) or too short environmental gradients. For example, Stolz et al. (2021) did not find any relationship between radial increment and MAP across a short precipitation gradient in northern Germany. In contrast, a more comprehensive study with 30 beech stands in northern Germany detected a tipping point at about 360 mm growing-season precipitation (MGSP, April – September), which separated positive and negative growth trends (Weigel et al. 2022). This matches the dendrochronological results of Knutzen et al. (2017) in the western part of our study region which suggest a similar threshold of about 350 mm MGSP or of ca. 660 mm MAP for the beech growth trend to turn negative.
The high importance of June precipitation for beech growth in Central Europe is supported by other studies (Scharnweber et al. 2011; Harvey et al. 2020; Stolz et al. 2021), while MAP seems to be of secondary importance. In fact, beech thrives in the driest region of Central Europe (near Halle/S. in central Germany) at < 500 mm MAP on deep loess soils and shows a positive recent growth trend (Weigel et al. 2022). Similarly, beech growth reveals a strong positive trend in low MAP regions in northern Mecklenburg-Western Pomerania (sites no. 13 and 14), where June precipitation has recently increased (see Fig. 6). In regions where June precipitation has decreased, in contrast, beech shows a negative growth trend (except for site no. 7), even when MAP exceeds 770 mm. We explain the fact that the June precipitation trend is more important for the direction of recent growth trends than absolute amounts of GSP or MAP, with local acclimation or adaptation to the prevailing drought regime. Long-term change in water availability, especially in physiologically important June, has the potential to reduce beech vitality, probably because foliage, hydraulic architecture and fine root system are not well adjusted to the altered conditions. This might trigger further acclimation or, if not possible, lead to vitality decline and eventually death (Obladen et al. 2021; Walthert et al. 2021).
Our comparison of sites revealed that beech growth is favored by higher summer precipitation and reduced drought exposure in the previous year at both the driest and the moistest sites, but the former stands were additionally dependent on rainfall and drought in the current summer, a factor not relevant at the moister sites. This suggests a higher sensitivity to low precipitation and climatic drought at the sites with low MAP and may hint at limited adaptation in drier climates. The published work on regional differences in the drought adaptation of beech is partly controversial. Martinez del Castillo et al. (2018) compared beech trees at dry sites and of marginal populations with beeches at wetter sites in the distribution center and found no better adaption of the former, which is in line with the findings of Scharnweber et al. (2011) and Stolz et al. (2021) observed in northern Germany. In contrast, Hacket-Pain et al. (2016), Cavin and Jump (2017) and Muffler et al. (2020) found in continent-wide studies a reduced drought sensitivity at drier sites and for dry-edge populations, which point at successful adaptation.
We cannot explain the performance of beech trees in stand no. 7, which show a positive growth trend despite decreasing June precipitation. Forest management can be largely excluded as a driver, as the last thinning took place in this stand in 1996 and no sudden increase in the growth curves can be detected thereafter (Fig. 7, Appendix). It is possible that natural disturbance events have altered the competitive balance between dominant and subdominant trees in this stand in the recent past.
Pine has in common with beech, that signs of its sensitivity to low summer precipitation and drought (and less pronounced to high summer temperatures) increased with decreasing MAP and MGSP; nevertheless, positive growth trends prevailed at most sites (except for site no. 8). Stolz et al. (2021) similarly found increasing drought sensitivity with decreasing MAP for the two species in northeastern Germany, but for pine only during the 1964–1993 interval, and not thereafter. In our comparison, pine, in contrast to beech, shows stable sensitivity to measures of summer water deficits of the current year, and this sensitivity apparently has shifted from June to July from the 1960–1988 to the 1989–2017 period (Diers et al., submitted). The negative effect of increasingly dry summers is currently offset by a positive influence of warm winters, so that pine continues to show a positive growth trend along the entire studied climate gradient, in contrast to beech. Certainly, we cannot fully exclude an effect of stand age on this difference, because the different rotation time for the two species caused different stand ages of the sampled trees. Nevertheless, even old beech trees > 300 to 400 years have the potential to sustain high growth rates (Piovesan et al. 2003, 2008), which suggests that climate change, and not natural senescence, is the main driver of the observed growth decline in beech in our stands, where age varied between 95 and 195 years.
Precipitation and water deficits are modulated by soil physical factors in their effect on tree water status and growth. While all 20 stands were selected on sandy to sandy-loamy soils on glacial deposits, it is likely that the capacity for plant-available water (AWC) differs between the stands and thus may explain part of the variation in growth trends and climate sensitivity of growth (Fuchs et al. 2020; Scharnweber et al. 2020). Since soil physical data are lacking for our sites, the soil influence remains elusive. However, another study in 30 beech forests in the North German Lowlands on similar soil revealed a significantly higher xylem embolism resistance of beech sun-canopy branches with a decrease in AWC from 300 mm to < 100 mm on sandy soil, indicating successful acclimation of the hydraulic system of beech to reduced soil water storage capacity (Weithmann et al. 2022). Variation in soil properties could also influence the drought tolerance of trees through altered nutrient availability (Lévesque et al. 2016; Braun et al. 2021). Future research on tree growth patterns has to consider regional variation in both climatic and edaphic water availability to reach at a more complete picture.
Conclusions
The necessary shift to climate-smart forests requires comprehensive understanding of the drought and heat tolerance of tree species as well as of their effects on the biogeochemical cycles of forest landscapes. Comparative dendroecological studies of several co-occurring tree species across climate gradients, such as our study on paired pine and beech stands on similar soil along a steep precipitation gradient, can provide valuable information to achieve this goal. A higher carbon sequestration potential and positive effects on the water cycle, soil fertility and biodiversity suggest to replace pine plantations by beech forests in parts of the North German Lowlands. However, the sensitivity of beech to increasing drought exposure and declining growth rates, as demonstrated here, suggest selecting more drought-tolerant broadleaf species in all regions with declining June precipitation, notably in Saxony-Anhalt (and adjacent regions of western Brandenburg and eastern Lower Saxony). Promising silvicultural alternatives can be Quercus species (notably Q. petraea and robur, but also Q. frainetto (Kasper et al. 2022), Acer platanoides, Carpinus betulus, Sorbus torminalis and Tilia cordata (Fuchs et al. 2021), among others.
Beech will be a relatively safe option close to the North Sea (Schleswig-Holstein, north-western Lower Saxony) and Baltic Sea coasts (parts of northern Mecklenburg-Western Pomerania) and in northern Brandenburg, where the summer climatic water balance has turned more favorable in the recent past. Although our pine stands revealed until recently mostly positive growth trends, it is likely that the next pine generation will also suffer vitality and growth declines, should the current thermal and hydrometeorological trends continue in future.
Author contribution statement
CL conceived the study idea. CL and MD designed the study. MD performed the research and analyzed the data together with RW. MD wrote the manuscript together with CL and RW. All authors read and approved the final manuscript.
Appendix
See Table 3.
Temporal course of basal area increment (BAI) of the individual trees (grey lines) and of the stand chronologies (bold black line) in the period 1960–2017 in the each ten beech (B) and pine (P) stands. Linear trends for the earlier period are depicted in green, for the later period in red. Given are Pearson’s r values for the trend lines. Significance was calculated with a Mann-Kendall trend test. The spline curve (thin blue line) is calculated using local polynomial regression fitting (loess) with a smoothing parameter (span) of 0.5
Moving-window analysis of the correlation (Pearson’s r) between ring width (RWI) with long-term June precipitation change since the 1960–1988 interval in the ten beech stands. The sites are ordered by the amount of change in June precipitation between the 1960–1988 and the 1989–2017 period (sites no. 11–14: increase in June precipitation, no. 4–10: increasingly larger decrease in June precipitation). The x-axis shows the central year of the 30-years moving window. The grey background marks the range with |r| > 0.37 at p < 0.05 for n = 30 years
Data availability
The data set generated during the current study is available from the corresponding author on reasonable request. All climate data from the German Weather Service (DWD) is openly available at www.opendata.dwd.de.
References
Aertsen W, Kint V, De Vos B et al (2012) Predicting forest site productivity in temperate lowland from forest floor, soil and litterfall characteristics using boosted regression trees. Plant Soil 354:157–172. https://doi.org/10.1007/s11104-011-1052-z
Allen CD, Macalady AK, Chenchouni H et al (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259:660–684. https://doi.org/10.1016/j.foreco.2009.09.001
Anfodillo T, Petit G, Crivellaro A (2013) Axial conduit widening in woody species: a still neglected anatomical pattern. IAWA J 34:352–364. https://doi.org/10.1163/22941932-00000030
Archambeau J, Ruiz-Benito P, Ratcliffe S et al (2020) Similar patterns of background mortality across Europe are mostly driven by drought in European beech and a combination of drought and competition in Scots pine. Agric For Meteorol 280:107772. https://doi.org/10.1016/j.agrformet.2019.107772
Baillie MGL, Pilcher JR (1973) A simple crossdating program for tree-ring research. Tree-Ring Bull 33:7–14
Barriopedro D, Fischer EM, Luterbacher J et al (2011) The hot summer of 2010: redrawing the temperature record map of Europe. Science 332:220–224. https://doi.org/10.1126/science.1201224
Bat-Enerel B, Weigel R, Leuschner C (2022) Changes in the thermal and hydrometeorological forest growth climate during 1948–2017 in northern Germany. Front For Glob Change 5:830977. https://doi.org/10.3389/ffgc.2022.830977
Bauhus J, Seeling U, Dieter M et al (2021) Die Anpassung von Wäldern und Waldwirtschaft an den Klimawandel. Berichte über Landwirtsch - Z für Agrarpolit Landwirtsch, Sonderheft 233, November 2021. https://doi.org/10.12767/BUEL.VI233.386
Bauwe A, Jurasinski G, Scharnweber T et al (2015) Impact of climate change on tree-ring growth of Scots pine, common beech and pedunculate oak in northeastern Germany. IForest Biogeosci For 9:1–11. https://doi.org/10.3832/ifor1421-008.
Bauwe A, Koch M, Kallweit R et al (2013) Tree-ring growth response of Scots pine (pinus sylvestris l.) to climate and soil water availability in the lowlands of north-eastern Germany. Balt For 19:14
Beguería S, Vicente-Serrano SM (2017) Calculation of the Standardised Precipitation-Evapotranspiration Index. R Foundation for Statistical Computing, Vienna, Austria
Bernal-Escobar M, Zuleta D, Feeley KJ (2022) Changes in the climate suitability and growth rates of trees in eastern North America. Ecography. https://doi.org/10.1111/ecog.06298
Bigler C, Bräker OU, Bugmann H et al (2006) Drought as an inciting mortality factor in Scots pine stands of the Valais, Switzerland. Ecosystems 9:330–343. https://doi.org/10.1007/s10021-005-0126-2
BMUB (2007) Nationale Strategie zur biologischen Vielfalt. Federal Ministry for the Environment, Berlin, Germany
Boessenkool B (2021) Select and download climate data from “DWD” (German Weather Service). https://cran.r-project.org/web/packages/rdwd/index.html
Bohn U, Gollub G (2007) Buchenwälder als natürliche vegetation in Europa. Nat Landsch 82:391–397
Bohn U, Neuhäusl R (2003) Map of the natural vegetation of Europe. Landwirtschaftsverlag, Münster/W, Germany
Bosela M, Štefančík I, Petráš R, Vacek S (2016) The effects of climate warming on the growth of European beech forests depend critically on thinning strategy and site productivity. Agric For Meteorol 222:21–31. https://doi.org/10.1016/j.agrformet.2016.03.005
Bowditch E, Santopuoli G, Binder F et al (2020) What is Climate-Smart Forestry? A definition from a multinational collaborative process focused on mountain regions of Europe. Ecosyst Serv 43:101113. https://doi.org/10.1016/j.ecoser.2020.101113
Braun S, Schindler C, Rihm B (2017) Growth trends of beech and Norway spruce in Switzerland: the role of nitrogen deposition, ozone, mineral nutrition and climate. Sci Total Environ 599–600:637–646. https://doi.org/10.1016/j.scitotenv.2017.04.230
Braun S, de Witte LC, Hopf SE (2020) Auswirkungen des Trockensommers 2018 auf Flächen der Interkantonalen Walddauerbeobachtung. Schweiz Z Forstwes 171:270–280. https://doi.org/10.3188/szf.2020.0270
Braun S, Hopf S-E, Tresch S et al (2021) 37 years of forest monitoring in Switzerland: Drought effects on Fagus sylvatica. Front For Glob Change 4:765782. https://doi.org/10.3389/ffgc.2021.765782
Brinkmann N, Eugster W, Buchmann N, Kahmen A (2019) Species-specific differences in water uptake depth of mature temperate trees vary with water availability in the soil. Plant Biol 21:71–81. https://doi.org/10.1111/plb.12907
Bundesministerium für Ernährung und Landwirtschaft (BMEL) (ed) (2021) Waldstrategie 2050 - Herausforderungen und Chancen für Mensch, Natur und Klima. Berlin
Bunn A, Korpela M (2021) Chronology building in dplr. R Foundation for Statistical Computing, Vienna, Austria
Bunn A, Korpela M, Biondi F et al (2021) dplR: Dendrochronology Program Library in R. R package version 1.7.2. R Foundation for Statistical Computing, Vienna, Austria
Carlisle A, Brown AHF (1968) Pinus sylvestris L. J Ecol 56:269. https://doi.org/10.2307/2258078
Carnicer J, Coll M, Ninyerola M et al (2011) Widespread crown condition decline, food web disruption, and amplified tree mortality with increased climate change-type drought. Proc Natl Acad Sci 108:1474–1478. https://doi.org/10.1073/pnas.1010070108
Carrer M, Urbinati C (2004) Age-dependent tree‐ring growth responses to climate in Larix decidua and Pinus cembra. Ecology 85:730–740
Cavin L, Jump AS (2017) Highest drought sensitivity and lowest resistance to growth suppression are found in the range core of the tree Fagus sylvatica L. not the equatorial range edge. Glob Change Biol 23:362–379. https://doi.org/10.1111/gcb.13366
Charru M, Seynave I, Morneau F, Bontemps J-D (2010) Recent changes in forest productivity: an analysis of national forest inventory data for common beech (Fagus sylvatica L.) in north-eastern France. For Ecol Manag 260:864–874. https://doi.org/10.1016/j.foreco.2010.06.005
Cook ER, Peters K (1981) The smoothing spline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree-Ring Bull 41:45–53
Čufar K, Prislan P, de Luis M, Gričar J (2008) Tree-ring variation, wood formation and phenology of beech (Fagus sylvatica) from a representative site in Slovenia, SE Central Europe. Trees 22:749–758. https://doi.org/10.1007/s00468-008-0235-6
Della-Marta PM, Haylock MR, Luterbacher J, Wanner H (2007) Doubled length of western European summer heat waves since 1880. J Geophys Res 112:D15103. https://doi.org/10.1029/2007JD008510
Diers M, Weigel R, Culmsee H, Leuschner C (2021) Soil carbon and nutrient stocks under Scots pine plantations in comparison to European beech forests: a paired-plot study across forests with different management history and precipitation regimes. For Ecosyst 8:47. https://doi.org/10.1186/s40663-021-00330-y
Dobbertin MK, Grissino-Mayer HD (2004) The online bibliography of dendrochronology. Dendrochronologia 21:85–90. https://doi.org/10.1078/1125-7865-00042
Dulamsuren C, Hauck M, Kopp G et al (2017) European beech responds to climate change with growth decline at lower, and growth increase at higher elevations in the center of its distribution range (SW Germany). Trees 31:673–686. https://doi.org/10.1007/s00468-016-1499-x
Dulamsuren C, Bat-Enerel B, Meyer P, Leuschner C (2022) Did stand opening 60 years ago predispose a European beech population to death? Trees For People 8:100265. https://doi.org/10.1016/j.tfp.2022.100265
DWD (2020) Nationaler Klimareport, 4th corr. ed. Deutscher Wetterdienst, Potsdam, Germany
DWD Climate Data Center (CDC) (2019a) Raster der Monatsmittel der Lufttemperatur (2m) für Deutschland, v1.0. https://opendata.dwd.de/climate_environment/CDC/grids_germany/monthly/air_temperature_mean/. Accessed 15 Dec 2019
DWD Climate Data Center (CDC) (2019b) Raster der Monatssumme der Niederschlagshöhe für Deutschland, Version v1.0. https://opendata.dwd.de/climate_environment/CDC/grids_germany/monthly/precipitation/. Accessed 17 Dec 2019
DWD Climate Data Center (CDC) (2022) Multi-annual grids of precipitation height over Germany 1991–2020, version v1.0. https://opendata.dwd.de/climate_environment/CDC/grids_germany/multi_annual/precipitation/. Accessed 3 Feb 2022
Dyderski MK, Paź S, Frelich LE, Jagodziński AM (2018) How much does climate change threaten European forest tree species distributions? Glob Change Biol 24:1150–1163. https://doi.org/10.1111/gcb.13925
Eckenwalder JE (2009) Conifers of the World: The Complete Reference. Timber Press, Portland, USA
Eckstein D, Bauch J (1969) Beitrag zur Rationalisierung eines dendrochronologischen Verfahrens und zur Analyse seiner Aussagesicherheit. Forstwiss Centralbl 88:230–250. https://doi.org/10.1007/BF02741777
Efron B, Tibshirani R (1986) Bootstrap methods for standard errors, confidence intervals, and other measures of statistical accuracy. Stat Sci 1:54–77. https://doi.org/10.1214/ss/1177013815
EU (2011) Our life insurance, our natural capital: an EU biodiversity strategy to 2020. European Commission, Brussels, published 3.5.2011.
Fang J, Lechowicz MJ (2006) Climatic limits for the present distribution of beech (Fagus L.) species in the world. J Biogeogr 33:1804–1819. https://doi.org/10.1111/j.1365-2699.2006.01533.x
FAO (2021) FAO Yearbook of Forest Products 2019. FAO, Rome
FAO and UNEP (2020) The State of the World’s Forests 2020. Forests, biodiversity and people. FAO, Rome
Farahat E, Linderholm HW (2018) Growth–climate relationship of European beech at its northern distribution limit. Eur J For Res 137:619–629. https://doi.org/10.1007/s10342-018-1129-9
Fischer AP (2019) Adapting and coping with climate change in temperate forests. Glob Environ Change 54:160–171. https://doi.org/10.1016/j.gloenvcha.2018.10.011
Förster A, Culmsee H, Leuschner C (2021a) Thinned northern German Scots pine forests have a low carbon storage and uptake potential in comparison to naturally developing beech forests. For Ecol Manag 479:118575. https://doi.org/10.1016/j.foreco.2020.118575
Förster A, Hertel D, Werner R, Leuschner C (2021b) Belowground consequences of converting broadleaf to conifer forest: comparing the fine root systems of European beech and Scots pine. For Ecol Manag 496:119457. https://doi.org/10.1016/j.foreco.2021.119457
Fuchs S, Hertel D, Schuldt B, Leuschner C (2020) Effects of summer drought on the fine root system of five broadleaf tree species along a precipitation gradient. Forests 11:289. https://doi.org/10.3390/f11030289
Fuchs S, Schuldt B, Leuschner C (2021) Identification of drought-tolerant tree species through climate sensitivity analysis of radial growth in central European mixed broadleaf forests. For Ecol Manag 494:119287. https://doi.org/10.1016/j.foreco.2021.119287
Gallardo VB, Hadad MA, Ribas-Fernández YA et al (2022) Age-related tree-ring sensitivity at the dry forest-steppe boundary in northwestern Patagonia. Trees 36:1607–1620. https://doi.org/10.1007/s00468-022-02312-1
Giuggiola A, Kuster T, Saha S (2010) Drought-induced mortality of Scots pines at the southern limits of its distribution in Europe: causes and consequences. IForest - Biogeosciences For 3:95–97. https://doi.org/10.3832/ifor0542-003
Hacket-Pain AJ, Friend AD (2017) Increased growth and reduced summer drought limitation at the southern limit of Fagus sylvatica L., despite regionally warmer and drier conditions. Dendrochronologia 44:22–30. https://doi.org/10.1016/j.dendro.2017.02.005
Hacket-Pain AJ, Cavin L, Friend AD, Jump AS (2016) Consistent limitation of growth by high temperature and low precipitation from range core to southern edge of European beech indicates widespread vulnerability to changing climate. Eur J For Res 135:897–909. https://doi.org/10.1007/s10342-016-0982-7
Hacket-Pain AJ, Ascoli D, Vacchiano G et al (2018) Climatically controlled reproduction drives interannual growth variability in a temperate tree species. Ecol Lett 21:1833–1844. https://doi.org/10.1111/ele.13158
Hanewinkel M, Cullmann DA, Schelhaas M-J et al (2013) Climate change may cause severe loss in the economic value of European forest land. Nat Clim Change 3:203–207. https://doi.org/10.1038/nclimate1687
Harvey JE, Smiljanić M, Scharnweber T et al (2020) Tree growth influenced by warming winter climate and summer moisture availability in northern temperate forests. Glob Change Biol 26:2505–2518. https://doi.org/10.1111/gcb.14966
Hertel D, Strecker T, Müller-Haubold H, Leuschner C (2013) Fine root biomass and dynamics in beech forests across a precipitation gradient - is optimal resource partitioning theory applicable to water-limited mature trees? J Ecol 101:1183–1200. https://doi.org/10.1111/1365-2745.12124
Hickler T, Vohland K, Feehan J et al (2012) Projecting the future distribution of European potential natural vegetation zones with a generalized, tree species-based dynamic vegetation model. Glob Ecol Biogeogr 21:50–63
IPCC (2021) Summary for Policymakers. In: Masson-Delmotte V, Zhai P, Pirani A (eds) Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press
Jiao L, Jiang Y, Wang M et al (2017) Age-effect radial growth responses of Picea schrenkiana to climate change in the Eastern Tianshan Mountains, Northwest China. Forests 8:294. https://doi.org/10.3390/f8090294
Jump AS, Hunt JM, Peñuelas J (2006) Rapid climate change-related growth decline at the southern range edge of Fagus sylvatica. Glob Change Biol 12:2163–2174. https://doi.org/10.1111/j.1365-2486.2006.01250.x
Juntunen V, Neuvonen S, Norokorpi Y, Tasanen T (2002) Potential for timberline advance in northern Finland, as revealed by monitoring during 1983-99. Arctic 55:348–361. https://doi.org/10.14430/arctic719
Kasper J, Leuschner C, Walentowski H et al (2022) Winners and losers of climate warming: declining growth in Fagus and Tilia vs. stable growth in three Quercus species in the natural beech–oak forest ecotone (western Romania). For Ecol Manag 506:119892. https://doi.org/10.1016/j.foreco.2021.119892
Kint V, Aertsen W, Campioli M et al (2012) Radial growth change of temperate tree species in response to altered regional climate and air quality in the period 1901–2008. Clim Change 115:343–363. https://doi.org/10.1007/s10584-012-0465-x
Knutzen F, Dulamsuren C, Meier IC, Leuschner C (2017) Recent climate warming-related growth decline impairs European beech in the center of its distribution range. Ecosystems 20:1494–1511. https://doi.org/10.1007/s10021-017-0128-x
Köcher P, Gebauer T, Horna V, Leuschner C (2009) Leaf water status and stem xylem flux in relation to soil drought in five temperate broad-leaved tree species with contrasting water use strategies. Ann For Sci 66:101–101. https://doi.org/10.1051/forest/2008076
Kölling VC (2007) Klimahüllen für 27 Waldbaumarten. AFZ-Der Wald 23:1242–1245
Konter O, Büntgen U, Carrer M et al (2016) Climate signal age effects in boreal tree-rings: Lessons to be learned for paleoclimatic reconstructions. Quat Sci Rev 142:164–172. https://doi.org/10.1016/j.quascirev.2016.04.020
Kullman L (2014) Treeline (Pinus sylvestris) landscape evolution in the Swedish Scandes—a 40-year demographic effort viewed in a broader temporal context. Nord Geogr Tidsskr - Nord J Geogr 68:155–167. https://doi.org/10.1080/00291951.2014.904402
Lakatos F, Molnár M (2009) Mass mortality of beech (Fagus sylvatica L.) in South-West Hungary. Acta Silv Lignar Hungarica 5:75–82
Latte N, Lebourgeois F, Claessens H (2015) Increased tree-growth synchronization of beech (Fagus sylvatica L.) in response to climate change in northwestern Europe. Dendrochronologia 33:69–77. https://doi.org/10.1016/j.dendro.2015.01.002
Lebourgeois F, Rathgeber CBK, Ulrich E (2010) Sensitivity of French temperate coniferous forests to climate variability and extreme events (Abies alba, Picea abies and Pinus sylvestris). J Veg Sci 21:364–376. https://doi.org/10.1111/j.1654-1103.2009.01148.x
Leuschner C (2020) Drought response of European beech (Fagus sylvatica L.)—a review. Perspect Plant Ecol Evol Syst 47:125576. https://doi.org/10.1016/j.ppees.2020.125576
Leuschner C, Ellenberg H (2017) Ecology of Central European Forests: Vegetation Ecology of Central Europe, volume I. Springer, Cham
Leuschner C, Förster A, Diers M, Culmsee H (2022) Are northern German Scots pine plantations climate-smart? The impact of large-scale conifer planting on climate, soil and the water cycle. For Ecol Manag 507:120013. https://doi.org/10.1016/j.foreco.2022.120013
Lévesque M, Walthert L, Weber P (2016) Soil nutrients influence growth response of temperate tree species to drought. J Ecol 104:377–387. https://doi.org/10.1111/1365-2745.12519
Magri D, Vendramin GG, Comps B et al (2006) A new scenario for the quaternary history of European beech populations: palaeobotanical evidence and genetic consequences. New Phytol 171:199–221. https://doi.org/10.1111/j.1469-8137.2006.01740.x
Martinez del Castillo E, Prislan P, Gričar J et al (2018) Challenges for growth of beech and co-occurring conifers in a changing climate context. Dendrochronologia 52:1–10. https://doi.org/10.1016/j.dendro.2018.09.001
Martinez del Castillo E, Zang CS, Buras A et al (2022) Climate-change-driven growth decline of European beech forests. Commun Biol 5:163. https://doi.org/10.1038/s42003-022-03107-3
Martínez-Alonso C, Valladares F, Camarero JJ et al (2007) The uncoupling of secondary growth, cone and litter production by intradecadal climatic variability in a mediterranean Scots pine forest. For Ecol Manag 253:19–29. https://doi.org/10.1016/j.foreco.2007.06.043
Martínez-Vilalta J, López BC, Adell N et al (2008) Twentieth century increase of Scots pine radial growth in NE Spain shows strong climate interactions. Glob Change Biol 14:2868–2881. https://doi.org/10.1111/j.1365-2486.2008.01685.x
Mathisen IE, Hofgaard A (2011) Recent height and diameter growth variation in Scots pine (Pinus sylvestris L.) along the Arctic margin: the importance of growing season versus non-growing season climate factors. Plant Ecol Divers 4:1–11. https://doi.org/10.1080/17550874.2011.570797
Matías L, Jump AS (2014) Impacts of predicted climate change on recruitment at the geographical limits of Scots pine. J Exp Bot 65:299–310. https://doi.org/10.1093/jxb/ert376
Matías L, Linares JC, Sánchez-Miranda Á, Jump AS (2017) Contrasting growth forecasts across the geographical range of Scots pine due to altitudinal and latitudinal differences in climatic sensitivity. Glob Change Biol 23:4106–4116. https://doi.org/10.1111/gcb.13627
Mausolf K, Wilm P, Härdtle W et al (2018) Higher drought sensitivity of radial growth of European beech in managed than in unmanaged forests. Sci Total Environ 642:1201–1208. https://doi.org/10.1016/j.scitotenv.2018.06.065
McLeod AI (2015) Package Kendall. https://www.stats.uwo.ca/faculty/aim
Meier IC, Leuschner C (2008) Leaf size and leaf area index in Fagus sylvatica forests: competing effects of precipitation, temperature, and nitrogen availability. Ecosystems 11:655–669. https://doi.org/10.1007/s10021-008-9135-2
Meier IC, Knutzen F, Eder LM et al (2018) The deep root system of Fagus sylvatica on sandy soil: structure and variation across a precipitation gradient. Ecosystems 21:280–296. https://doi.org/10.1007/s10021-017-0148-6
Mellert KH, Ewald J, Hornstein D et al (2016) Climatic marginality: a new metric for the susceptibility of tree species to warming exemplified by Fagus sylvatica (L.) and Ellenberg’s quotient. Eur J For Res 135:137–152. https://doi.org/10.1007/s10342-015-0924-9
Menzel A, Jakobi G, Ahas R et al (2003) Variations of the climatological growing season (1951–2000) in Germany compared with other countries. Int J Climatol 23:793–812. https://doi.org/10.1002/joc.915
Mérian P, Lebourgeois F (2011) Size-mediated climate–growth relationships in temperate forests: a multi-species analysis. For Ecol Manag 261:1382–1391. https://doi.org/10.1016/j.foreco.2011.01.019
Mielikäinen K, Sennov SN (1996) Growth trends of forests in Finland and North-Western Russia. In: Spiecker H, Mielikäinen K, Köhl M, Skovsgaard JP (eds) Growth Trends in European Forests. Springer, Berlin Heidelberg, pp 19–27
Muffler L, Weigel R, Hacket-Pain AJ et al (2020) Lowest drought sensitivity and decreasing growth synchrony towards the dry distribution margin of European beech. J Biogeogr 47:1910–1921. https://doi.org/10.1111/jbi.13884
Müller K (2007) Die aktuelle Verbreitung der Kiefer in Brandenburg. Ministerium für Ländliche Entwicklung, Umwelt und Verbraucherschutz (MLUV) (ed) Die Kiefer im Nordostdeutschen Tiefland - Ökologie und Bewirtschaftung, 1st edn. Brandenburgische Universitätsdruckerei und Verlagsgesellschaft Potsdam mbH, Potsdam, Germany
Müller-Haubold H, Hertel D, Leuschner C (2015) Climatic drivers of mast fruiting in European beech and resulting C and N allocation shifts. Ecosystems 18:1083–1100. https://doi.org/10.1007/s10021-015-9885-6
Nussbaumer A, Waldner P, Etzold S et al (2016) Patterns of mast fruiting of common beech, sessile and common oak, Norway spruce and Scots pine in Central and Northern Europe. For Ecol Manag 363:237–251. https://doi.org/10.1016/j.foreco.2015.12.033
Obladen N, Dechering P, Skiadaresis G et al (2021) Tree mortality of European beech and Norway spruce induced by 2018–2019 hot droughts in Central Germany. Agric For Meteorol 307:108482. https://doi.org/10.1016/j.agrformet.2021.108482
Packham JR, Thomas PA, Atkinson MD, Degen T (2012) Biological flora of the British Isles: Fagus sylvatica. J Ecol 100:1557–1608. https://doi.org/10.1111/j.1365-2745.2012.02017.x
Peng J, Peng K, Li J (2018) Climate-growth response of Chinese white pine (Pinus armandii) at different age groups in the Baiyunshan National Nature Reserve, central China. Dendrochronologia 49:102–109. https://doi.org/10.1016/j.dendro.2018.02.004
Piovesan G, Bernabei M, Di Filippo A et al (2003) A long-term tree ring beech chronology from a high-elevation old-growth forest of Central Italy. Dendrochronologia 21:13–22. https://doi.org/10.1078/1125-7865-00036
Piovesan G, Biondi F, Filippo AD et al (2008) Drought-driven growth reduction in old beech (Fagus sylvatica L.) forests of the central Apennines, Italy: drought-driven growth decline in beech forests. Glob Change Biol 14:1265–1281. https://doi.org/10.1111/j.1365-2486.2008.01570.x
Pompa-García M, Hadad MA (2016) Sensitivity of pines in Mexico to temperature varies with age. Atmosfera 29:209–219. https://doi.org/10.20937/ATM.2016.29.03.03.
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Rasztovits E, Berki I, Mátyás C et al (2014) The incorporation of extreme drought events improves models for beech persistence at its distribution limit. Ann For Sci 71:201–210. https://doi.org/10.1007/s13595-013-0346-0
Reich PB, Oleksyn J (2008) Climate warming will reduce growth and survival of Scots pine except in the far north: Scots pine growth and survival following climate transfer. Ecol Lett 11:588–597. https://doi.org/10.1111/j.1461-0248.2008.01172.x
Rickebusch S, Lischke H, Bugmann H et al (2007) Understanding the low-temperature limitations to forest growth through calibration of a forest dynamics model with tree-ring data. For Ecol Manag 246:251–263. https://doi.org/10.1016/j.foreco.2007.04.030
Rinn F (2012) TSAP-Win Professional - Benutzerhandbuch. Rinntech, Heidelberg
Roloff A (2008) Baum des Jahres 2007: die Wald-Kiefer (Pinus sylvestris L.)—Biologie, Ökologie, Verwendung, Schäden. In: Nordwestdeutschen Forstlichen Versuchsanstalt (ed) Die Waldkiefer – Fachtagung zum Baum des Jahres 2007. Göttingen, Germany
Sanchez-Salguero R, Camarero J, Gutiérrez E et al (2018) Climate warming alters age-dependent growth sensitivity to temperature in Eurasian alpine treelines. Forests 9:688. https://doi.org/10.3390/f9110688
Scharnweber T, Manthey M, Criegee C et al (2011) Drought matters – declining precipitation influences growth of Fagus sylvatica L. and Quercus robur L. in north-eastern Germany. For Ecol Manag 262:947–961. https://doi.org/10.1016/j.foreco.2011.05.026
Scharnweber T, Smiljanic M, Cruz-García R et al (2020) Tree growth at the end of the 21st century - the extreme years 2018/19 as template for future growth conditions. Environ Res Lett 15:074022. https://doi.org/10.1088/1748-9326/ab865d
Schueler S, Falk W, Koskela J et al (2014) Vulnerability of dynamic genetic conservation units of forest trees in Europe to climate change. Glob Change Biol 20:1498–1511. https://doi.org/10.1111/gcb.12476
Schuldt B, Buras A, Arend M et al (2020) A first assessment of the impact of the extreme 2018 summer drought on central european forests. Basic Appl Ecol 45:86–103. https://doi.org/10.1016/j.baae.2020.04.003
Schütt P, Stimm B (2006) Pinus sylvestris. In: Schütt P (ed) Enzyklopädie der Holzgewächse. Ecomed, Landsberg am Lech
Socha J, Solberg S, Tymińska-Czabańska L et al (2021) Height growth rate of Scots pine in Central Europe increased by 29% between 1900 and 2000 due to changes in site productivity. For Ecol Manag 490:119102. https://doi.org/10.1016/j.foreco.2021.119102
Stolz J, van der Maaten E, Kalanke H et al (2021) Increasing climate sensitivity of beech and pine is not mediated by adaptation and soil characteristics along a precipitation gradient in northeastern Germany. Dendrochronologia 67:125834. https://doi.org/10.1016/j.dendro.2021.125834
Suck R, Bushart M, Hofmann G, Schröder L (2014) Karte der Potentiellen Natürlichen Vegetation Deutschlands. Band 1 - Grundeinheiten. BfN-Skripten 348:1–449
Thünen-Institut (2022) Dritte Bundeswaldinventur - Ergebnisdatenbank, Waldfläche (gemäß Standflächenanteil) [ha] nach Land und Baumartengruppe (rechnerischer Reinbestand). https://bwi.info/inhalt1.3.aspx?Text=1.04%20Baumartengruppe%20(rechnerischer%20Reinbestand)&prRolle=public&prInv=BWI2012&prKapitel=1.04. Accessed 25 Oct 2021
Thurm EA, Hernandez L, Baltensweiler A et al (2018) Alternative tree species under climate warming in managed European forests. For Ecol Manag 430:485–497. https://doi.org/10.1016/j.foreco.2018.08.028
Trouillier M, van der Maaten-Theunissen M, Scharnweber T et al (2019) Size matters—a comparison of three methods to assess age- and size-dependent climate sensitivity of trees. Trees 33:183–192. https://doi.org/10.1007/s00468-018-1767-z
van der Maaten E (2013) Thinning prolongs growth duration of European beech (Fagus sylvatica L.) across a valley in southwestern Germany. For Ecol Manag 306:135–141. https://doi.org/10.1016/j.foreco.2013.06.030
van der Maaten E, Hamann A, van der Maaten-Theunissen M et al (2017) Species distribution models predict temporal but not spatial variation in forest growth. Ecol Evol 7:2585–2594. https://doi.org/10.1002/ece3.2696
van der Maaten-Theunissen M, Bümmerstede H, Iwanowski J et al (2016) Drought sensitivity of beech on a shallow chalk soil in northeastern Germany – a comparative study. For Ecosyst 3:24. https://doi.org/10.1186/s40663-016-0083-6
van Mantgem PJ, Stephenson NL, Byrne JC et al (2009) Widespread increase of tree mortality rates in the western United States. Science 323:521–524. https://doi.org/10.1126/science.1165000
Verkerk PJ, Costanza R, Hetemäki L et al (2020) Climate-Smart Forestry: the missing link. For Policy Econ 115:102164. https://doi.org/10.1016/j.forpol.2020.102164
Vitasse Y, Delzon S, Bresson CC et al (2009) Altitudinal differentiation in growth and phenology among populations of temperate-zone tree species growing in a common garden. Can J For Res 39:1259–1269. https://doi.org/10.1139/X09-054
Walthert L, Ganthaler A, Mayr S et al (2021) From the comfort zone to crown dieback: sequence of physiological stress thresholds in mature European beech trees across progressive drought. Sci Total Environ 753:141792. https://doi.org/10.1016/j.scitotenv.2020.141792
Weigel R, Muffler L, Klisz M et al (2018) Winter matters: sensitivity to winter climate and cold events increases towards the cold distribution margin of European beech (Fagus sylvatica L.). J Biogeogr 45:2779–2790. https://doi.org/10.1111/jbi.13444
Weigel R, Banzragch B-E, Dulamsuren C et al (2022) Summer drought exposure, stand structure, and soil properties jointly control the growth of European beech along a steep precipitation gradient in northern Germany. Glob Chang Biol. https://doi.org/10.1111/gcb.16506
Weithmann G, Link RM, Banzragch B-E et al (2022) Soil water availability and branch age explain variability in xylem safety in European beech in Central Europe. Oecologia 198:629–644
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23:201-213
Wilmking M, Maaten-Theunissen M, Maaten E et al (2020) Global assessment of relationships between climate and tree growth. Glob Change Biol 26:3212–3220. https://doi.org/10.1111/gcb.15057
Zang C, Biondi F (2015) treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38:431–436. https://doi.org/10.1111/ecog.01335
Zang C, Hartl-Meier C, Dittmar C et al (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Change Biol 20:3767–3779. https://doi.org/10.1111/gcb.12637
Zimmermann J, Hauck M, Dulamsuren C, Leuschner C (2015) Climate warming-related growth decline affects Fagus sylvatica, but not other broad-leaved tree species in central european mixed forests. Ecosystems 18:560–572. https://doi.org/10.1007/s10021-015-9849-x
Acknowledgements
We would like to thank Johanna Steen and Thore Christian Schulz for help during the field work, and Banzragch Bat-Enerel for his support during the work in the dendro lab.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study is part of the WiNat-project, funded by the Federal Ministry for Education and Research (BMBF) (project number: 01LC1314B).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Camarero.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Diers, M., Weigel, R. & Leuschner, C. Both climate sensitivity and growth trend of European beech decrease in the North German Lowlands, while Scots pine still thrives, despite growing sensitivity. Trees 37, 523–543 (2023). https://doi.org/10.1007/s00468-022-02369-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-022-02369-y