Abstract
The aim of this study was to investigate the expressions of Toll-like receptor (TLR) 2, TLR4, TLR9, and their correlations with the expression of cytokines that are associated with activation of CD4+ T cells and inflammation including interferon γ (IFNγ), interleukin 4 (IL4), interleukin 17 (IL17), and tumor necrosis factor α (TNFα) in muscle tissues of patients with dermatomyositis (DM) and polymyositis (PM). The expressions of TLR2, TLR4, TLR9, IFNγ, IL4, IL17, and TNFα were measured by real-time reverse transcription–polymerase chain reaction in muscle tissues from 14 patients with DM and PM (nine patients with DM, five patients with PM) and three controls. The expressions of TLR2, TLR4, and TLR9 were also localized with immunohistochemistry. The expression levels of TLR2, TLR4, TLR9, IFNγ, IL4, IL17, and TNFα were significantly high in patients with DM and PM compared with those in the controls, and the expression levels of TLR4 and TLR9 had significant positive correlations with the expressions of IFNγ, IL4, IL17, and TNFα. Immunohistochemistry showed that TLR2, TLR4, and TLR9 were expressed by infiltrating cells of perimysium in DM, whereas they were expressed by infiltrating cells of endomysium in PM. These results suggest that the involvement of TLR4 and TLR9 in immunopathogenesis of DM and PM might be connected with activation of CD4+ T cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dermatomyositis (DM) and polymyositis (PM) are chronic muscle disorders characterized by inflammatory infiltrate in the muscle tissue. DM and PM are clinically characterized by features of symmetric proximal muscle weakness associated with muscle cell destruction [1]. Previous studies have identified different proportions of CD4+, CD8+ T cells and macrophages with various localizations in DM and PM. In PM, cytotoxic CD8+ T cells and macrophages were detected surrounding and invading non-necrotic muscle fibers expressing major histocompatibility complex (MHC) class I [2]. In DM, perivascular infiltrates composed mainly of CD4+ T cells, B cells, and macrophages were observed [3].
The cytokine-driven differentiation of distinct lineages of effector and regulatory T cells (Tregs) from naive CD4+ T cell precursors is a hallmark of the adaptive immune system. T helper (Th) 1 and Th2 are the best understood effector CD4+ T cells generated during immune responses, with each subset characterized by distinct transcription factor activity and cytokine-secreting phenotype. Classically, Th1 cells produce interferon (IFN) γ and mediate immune responses against intracellular bacteria, viruses, and tumor cells through the activation of macrophages and cytotoxic T cells. Th2 cells make mostly interleukin 4 (IL4), which stimulate humoral responses and are thought to have evolved to enhance resistance against extracellular parasites [4, 5]. In recent years, a distinct T-cell subset, termed Th17 cells, has also been identified and seems to play key roles in the activation of neutrophils and immunity to bacteria, particularly at mucosal surfaces. IL17, also termed IL17A, is the signature cytokine of Th17 cells [6].
Recently reported data suggest that CD4+ T cells play roles in immunopathogenesis of DM and PM [7]. Identification of Th1 and Th17 cytokine (IFNγ and IL17) producing cells from muscle tissue of DM and PM contributes to the roles of CD4+ T cells in DM and PM [8].
Cytokines are potent mediators of a number of cell functions and are essential in coordinating inflammatory responses. They can be produced by a large variety of cells and exhibit pro-inflammatory as well as anti-inflammatory effects. Their key role in chronic inflammatory diseases has been well documented by the often strikingly good response to therapies targeting proinflammatory cytokines, one of the best examples being tumor necrosis factor (TNF) α blockade in patients with rheumatoid arthritis and Crohn’s disease. Recent findings suggest cytokines as important key molecules in the pathogenic mechanisms of idiopathic inflammatory myopathies (IIMs), myositis [9].
Toll-like receptors (TLRs) are primarily involved in the innate immune response to microbial pathogens through the recognition of conserved pathogen-associated molecular patterns. However, they also contribute to sterile inflammation by sensing “danger signals”, the endogenous molecules that are generated during tissue damage or inflammation [10–13]. The activation of TLRs is an important bridge between innate and adaptive immunity by regulating the expression of co-stimulatory molecules on antigen-presenting cells that drive T-cell activation and by creating a cytokine milieu in which the differentiation of T cells into the desired subsets occurs [14]. It was known that activation of TLR4 and TLR9 is generally to induce a Th1 response in dendritic cells (DCs), and TLR2 might induce Th2-based immune response in experimental asthma, and activation of TLR4 also induces Th17 response through IL17 production [14–21].
The aim of this study was to investigate the expression of TLR2, TLR4, TLR9, and cytokines that are related to activation of CD4+ T cells (Th1, Th2, and Th17 cells) and inflammations including IFNγ, IL4, IL17, and TNFα, and to examine the connection of TLRs to these cytokines in muscle tissues of patients with DM and PM.
Here, we demonstrate that the TLR4 and TLR9 expressions are significantly increased, and they are connected with the expressions of those cytokines in muscle tissues of patients with DM and PM.
Patients and methods
Patients
Muscle biopsy samples from patients newly diagnosed with active DM (n = 9) or PM (n = 5), according to the classification system of Bohan and Peter [22, 23], were evaluated in this study. Seven patients were women and seven were men. The time from symptom onset to diagnosis was 1–12 months, and they had not been treated. The mean age at diagnosis was 41 years (41 ± 14 years). Patient data are presented in Table 1. Muscle tissue sections from three patients with non-specific muscle manifestations but normal histological findings were used as the controls. All patients and controls gave their informed consent, and the local ethics committee at Pusan National University Hospital approved the study.
Laboratory assessments
Serum levels of C-reactive protein (CRP), the erythrocyte sedimentation rate (ESR), and serum aldolase and creatinine kinase (CK) levels were analyzed at the Department of Clinical Chemistry, Pusan National University Hospital.
Muscle biopsies
Biopsy samples were obtained from the vastus lateralis or deltoid muscle with a “semi-open” muscle biopsy technique, with the patient under local anesthesia. The biopsy specimens were immediately frozen in dry ice and isopentane and stored at −70°C until analysis.
Real-time RT–PCR
After biopsy of the muscle tissues from patients with PM or DM, these were immersed immediately in liquid nitrogen. Total RNA was extracted from the frozen muscle biopsies with TRIzol Reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). Each solution containing 1 μg of RNA was heated at 65°C for 15 min, and a mixture containing reverse transcriptase was added to the solution. cDNA was transcribed with reaction cycles of 25°C for 10 min, 42°C for 60 min, 99°C for 5 min, and 4°C for 5 min. A First Strand cDNA Synthesis Kit for reverse transcription–polymerase chain reaction (RT–PCR; AMV, Roche Applied Science, Indianapolis, IN, USA) was used for the reactions described above, and real-time PCR was performed in a LightCycler System Instrument (Roche Applied Science). LightCycler-DNA Master SYBR Green I (Roche Applied Science), the cDNA template, each primer, and 25 mM of MgCl2 were added to microcapillary tubes to a final volume of 20 μl. The PCR cycling parameters were 50 cycles of denaturation at 95°C for 10 s, annealing at 60°C for 5 s, and primer extension at 72°C for 20 s.
The primers 5′-GCCACCATTTCCACGGACT-3′ (sense) and 5′-GGCTTCCTCTTGGCCTGG-3′ (antisense) were used to amplify TLR2, 5′-CTCTGCCTTCACTACA GAGACTTT-3′ (sense) and 5′-TGTGGAAGC CTTCCTGGATG-3′ (antisense) for TLR4, 5′-ACTGAGCACCCCTGCTTCTA-3′ (sense) and 5′-AGATTAGTCACGG CAGGAA-3′ (antisense) for TLR9, 5′-TCCCATGGGTTGTGTGTTTA-3′ (sense) and 5′-AAGCA CCAGGCATGAAATCT-3′ (antisense) for IFNγ, 5′-ACCCAAACTAGGCCTCACCT-3′ (sense) and 5′-ACAGGTGGCATCTTGGAAAC-3′ (antisense) for IL4, 5′-TCTCAT CCAGCAAGAGATCC-3′ (sense) and 5′-AGTTTGGGACCCCTTTACAC-3′ (antisense) for IL17, and 5′-CAAACCACCAAGTGGAGGAG-3′ (sense) and 5′-AGATAGCAAATC GGCTGACG-3′ (antisense) for TNFα, all purchased from Bioneer (Daejun, Korea).
The threshold cycle (C T) was determined by monitoring the fluorescent signal for each cycle, and the amounts of mRNA in the experimental groups were determined relative to those of the control group.
Immunohistochemistry for TLR2, TLR4, and TLR9
The cryotissue was fixed in 4% paraformaldehyde, and endogenous peroxidase activity was quenched with 3% H2O2 in methanol. Immunohistochemistry was performed using the Vectastain ABC kit (Vector Laboratories, Burlingame, CA, USA). The tissues were incubated with the primary antibody directed against TLR2 (sc-8689), TLR4 (sc-10741), or TLR9 (sc16247) overnight at 4°C. All the primary antibodies were purchased from Santa Cruz Biotechnology, Inc. The slides were incubated with biotinylated secondary antibody solution and then treated with the avidin/biotinylated enzyme complex. The final color product was developed using diaminobenzidine chromogen (Dako, Carpinteria, CA, USA).
Statistical analysis
Differences between experimental groups were tested using the Mann–Whitney U test and Student’s t test, depending on the parameter. The data are expressed as means ± standard deviations (SD).
Results
The expression levels of TLR2, TLR4, TLR9, IFNγ, IL4, IL17, and TNFα were significantly high in patients with DM and PM compared to controls
The mRNA expression levels of TLR2 (32.3 ± 18.1), TLR4 (170.0 ± 104.8), TLR9 (18.6 ± 13.8), IFNγ (1121.3 ± 978.9), IL4 (18.1 ± 19.5), IL17 (66.4 ± 60.6), and TNFα (22.7 ± 21.5) were significantly high in patients compared to those of the controls, and the levels of TLR4 and IFNγ were prominently high (Table 1). These expression levels were not significantly different between PM and DM, and any of these levels did not significantly correlate with disease duration, age, serum level of ESR, CRP, CK, and aldolase.
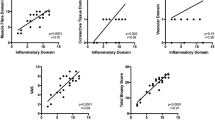
The expression levels of TLR4 and TLR9 had significant positive correlations with those of IFNγ, IL4, IL17, and TNFα
The expression levels of TLR2 had no significant correlations with those of IFNγ, IL4, IL17, and TNFα, whereas the levels of TLR4 and TLR9 had positive correlations with those of IFNγ (r = 0.76 in TLR4 and 0.73 in TLR9, p < 0.01), IL4 (r = 0.8 in TLR4 and 0.76 in TLR9, p < 0.01), IL17 (r = 0.92 in TLR4 and 0.9 in TLR9, p < 0.01), and TNFα (r = 0.71 in TLR4, p < 0.01; 0.66 in TLR9, p = 0.01; Fig. 1).
Correlations of TLR2, TLR4, TLR9, IFNγ, IL4, L17, and TNFα expressions in patients with DM and PM. The expression levels of TLR4 and TLR9 had significant positive correlations with those of IFNγ, IL4, IL17, and TNFα. The mRNA expressions were measured by real-time RT–PCR from muscle tissues of patients with DM and PM
Immunohistochemistry of TLR2, TLR4, and TLR9
TLR2, TLR4, and TLR9 were expressed by perimysial infiltrating cells in DM, whereas they were expressed by endomysial infiltrating cells in PM (Figs. 2, 3, and 4).
Immunohistochemistry of TLR2 in muscle tissues of patients with DM, PM, and controls. In the muscle tissues of DM, TLR2 was expressed by infiltrating cells in the perimysial area (A3), whereas in PM, TLR2 was expressed by infiltrating cells in the endomysium (A4). Control muscle tissues were obtained from patients with non-specific muscle manifestations but with normal histological findings (A1, 2). Magnification was ×400
Immunohistochemistry of TLR4 in muscle tissues of patients with DM, PM, and controls. In the muscle tissues of DM, TLR4 was expressed by infiltrating cells in the perimysial area (B3), whereas in PM, TLR4 was expressed by infiltrating cells in the endomysium (B4). Control muscle tissues were obtained from patients with non-specific muscle manifestations but with normal histological findings (B1, 2). Magnification was ×400
Immunohistochemistry of TLR9 in muscle tissues of patients with DM, PM, and controls. In the muscle tissues of DM, TLR9 was weakly expressed by infiltrating cells in the perimysial area (C3), whereas in PM, TLR9 was expressed by infiltrating cells in the endomysium (C4). Control muscle tissues were obtained from patients with non-specific muscle manifestations but with normal histological findings (C1, 2). Magnification was ×400
Discussion
Data originating predominantly from animal models of autoimmune disease and circumstantial data from human patients suggest that the inappropriate activation of the TLR pathway by endogenous or exogenous ligands may lead to the initiation and/or perpetuation of autoimmune responses and tissue injury [24]. The expression of TLRs by both immune cells and the resident cells in the involved tissues supports their important roles in tissue injury, destruction, and repair. We showed that the expression levels of TLR2, TLR4, and TLR9 were elevated in the muscle tissues of patients with DM and PM compared to controls, and as far as we know, this is the first report of this phenomenon in DM and PM.
We reported that inflammatory cytokines such as IL17, TNFα, IL1, and IL6 increase the production of TLR2, TLR4, and TLR9 from mice synoviocytes in autoimmune arthritis [25]. The possible mechanisms of TLR overexpressions are as follows: (1) various microbial pathogens considered as etiologic agents of IIMs contribute to overexpressions; (2) endogenous molecules and cytokines that are generated during muscle damage or immune response induce the overexpressions.
DM is a microangiopathy affecting the skin and muscle, in which the early activation and deposition of complement causes the lysis of endomysial capillaries and muscle ischemia. In PM, clonally expanded CD8+ cytotoxic T cells invade muscle fibers that express MHC class I antigens, leading to fiber necrosis via the perforin pathway. Perimysial and perivascular inflammation with CD4+ T cell, B cells, and macrophage infiltrations are histological features of DM, whereas endomysial inflammation with CD8+ T-cell infiltration is a histological feature of PM [1]. We have demonstrated that TLR2, TLR4, and TLR9 are expressed by perimysial infiltrating cells in DM and by endomysial infiltrating cells in PM. These expression patterns of TLR2, TLR4, and TLR9 were consistent with the histopathological features of DM and PM.
CD4+ T cells play roles in immunopathogenesis of DM and PM [7]. Detection of IFNγ and IL17 producing cells in muscle tissues of DM and PM suggests the involvement of activated CD4+ T cells in the pathophysiology of both DM and PM. These data showed that non-specific amplification of muscle inflammation by T lymphocytes may result from the local production of cytokines and chemokines [8]. We have demonstrated that the expressions of cytokines that are associated with CD4+ T-cell activation including IFNγ (Th1 cytokine), IL4 (Th2 cytokine), and IL17 (Th17 cytokine) were increased in patients with DM and PM compared to controls. These results imply that activation of CD4+ T cells is involved in immunopathogenesis of DM and PM.
TLR signaling may promote autoimmunity by several mechanisms. TLR signaling of B lymphocytes enhance their antigen-presenting capacity towards CD4+ T cells through up-regulation of HLA-DR. TLR signaling in plasmacytoid DCs results in maturation of DCs, and mature DCs promote induced activation of CD4+ T cells [24]. CD4+ T cells are categorized as Th1, Th2, or Th17 cells, and the function of these cells is to produce reciprocal sets of cytokines: IFNγ by Th1 cells, IL4 by Th2 cells, and IL17 by Th17 cells [4–6]. Specific TLRs might be associated with CD4+ T-cell activation. Activation of TLR2 might induce Th2 immune response, TLR4 induced Th1 and Th17 immune response, and TLR9 induced Th1 immune response [14–21]. We showed that the expression levels of TLR4 and TLR9 were increased compared to controls and had significant positive correlations with those of IFNγ, IL4, and IL17. These results suggest that the overexpression of TLR4 and TLR9 might be involved in the immunopathogenesis of DM and PM through activation of Th1, Th2, and Th17 cells. And the data showed by our study that the expression levels of TLR4 and IFNγ were prominently high and had significantly positive correlations with each other suggest that TLR4-induced Th1 response might play a more important role in immunopathogenesis of DM and PM.
TNFα gene expression can be detected in the muscle tissues of most patients with IIMs [26–28]. TNFα acts via several possible mechanisms, including the positive feedback loop in which increased numbers of TNFα-producing cells in infiltrating cell clusters enhance the inflammatory reaction by releasing more TNFα. This mediates the upregulation of adhesion molecules on endothelial cells and hence increases transendothelial cell trafficking as well as enhancing the T-cell response and thereby also T-cell-mediated muscle injury [29, 30]. We have showed that the expression level of TNFα was elevated and had significant positive correlations with those of TLR4 and TLR9 in the muscle tissues of patients with DM and PM.
According to recent studies, including our previous reported study and our present data, the possible immunopathogenic involvement of TLRs and CD4+ T cells are as follows: (1) overexpression of TLR4 and TLR9 induced by microbial pathogen, endogenous molecules, and inflammatory cytokines contributed to activation of CD4+ T cells resulting in increased expression of IFNγ, IL4, and IL17; (2) overexpressed CD4+ T-cell cytokines induce the inflammatory response, resulting in an increase of various inflammatory cytokines and endogenous molecules; (3) overexpressed CD4+ T-cell and inflammatory cytokines, including IFNγ, IL4, IL17, and TNFα, and endogenous molecules induce the overexpression of TLRs, causing the vicious cycle of amplification of chronic inflammation in muscle tissues of DM and PM.
In conclusion, we have demonstrated that the expressions of TLR2, TLR4, and TLR9 were elevated in muscle tissue of patients with DM and PM, and their expression patterns were consistent with pathophysiology of DM and PM. We also showed that the expression levels of TLR4 and TLR9 had significant positive correlations with those of Th1, Th2, and Th17 cell cytokines. These results suggest that the involvement of TLR4 and 9 in the immunopathogenesis of DM and PM may be connected with activation of CD4+ T cells.
References
Dalakas M, Hohlfeld R (2003) Polymyositis and dermatomyositis. Lancet 362:971–982
Nyberg P, Wikman AL, Nennesmo I, Lundberg I (2000) Increased expression of interleukin 1 alpha and MHC class I in muscle tissue of patients with chronic inactive polymyositis and dermatomyositis. J Rheumatol 27:940–948
Arahata K, Engel AG (1984) Monoclonal antibody analysis of mononuclear cells in myopathies. I: Quantitation of subsets according to diagnosis and sites of accumulation and demonstration and counts of muscle fibers invaded by T cells. Ann Neurol 16:193–208
Moss RB, Moll T, El-Kalay M et al (2004) Th1/Th2 cells in inflammatory disease states: therapeutic implications. Expert Opin Biol Ther 4:1887–1896
Coffman RL (2006) Origins of the T(H)1-T(H)2 model: a personal perspective. Nat Immunol 7:539–541
Stockinger B, Veldhoen M (2007) Differentiation and function of Th17 T cells. Curr Opin Immunol 19:281–286
Grundtman C, Malmström V, Lundberg I (2007) Immune mechanisms in the pathogenesis of idiopathic inflammatory myopathies. Arthritis Res Ther 9:208–220
Page G, Chevrel G, Miossec P (2004) Anatomic localization of immature and mature dendritic cell subsets in dermatomyositis and polymyositis: interaction with chemokines and Th1 cytokine-producing cells. Arthritis Rheum 50:199–208
Salomonsson S, Lundberg I (2006) Cytokines in idiopathic inflammatory myopathies. Autoimmunity 39:177–190
Akira S, Uematsu S, Takeuchi O (2006) Pathogen recognition and innate immunity. Cell 124:783–801
Johnson GB, Brunn GJ, Kodaira Y, Platt JL (2002) Receptor-mediated monitoring of tissue well-being via detection of soluble heparin sulfate by Toll-like receptor 4. J Immunol 168:5233–5239
Okamura Y, Watari M, Jerud ES et al (2001) The extra domain A of fibronection activates Toll-like receptor 4. J Biol Chem 276:10229–10233
Schaefer L, Babelova A, Kiss E et al (2005) The matrix component bigylcan is proinflammatory and signals through Toll-like receptors 4 and 2 in macrophages. J Clin Invest 115:2223–2233
Re F, Strominger JL (2001) Toll-like receptor 2 (TLR2) and TLR4 differentially activated human dendritic cells. J Biol Chem 276:37692–37699
Krieg AM (2002) CpG motifs in bacterial DNA and their immune effects. Annu Rev Immunol 20:709–760
Reis e Sousa C, Hieny S, Scharton-Kersten T et al (1997) In vivo microbial stimulation induces rapid CD40 ligand-independent production of interleukin 12 by dendritic cells and their redistribution to T cell areas. J Exp Med 186:1819–1829
Trinchieri G (2003) Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat Rev Immunol 3:133–146
Agrawal S, Agrawal A, Doughty B et al (2003) Cutting edge: different Toll-like receptor agonists instruct dendritic cells to induce distinct Th responses via differential modulation of extracellular signal-regulated kinase-mitogen-activated protein kinase and c-Fos. J Immunol 171:4984–4989
Dillon S, Agrawal A, Van Dyke T et al (2004) A Toll-like receptor 2 ligand stimulates Th2 responses in vivo, via induction of extracellular signal-regulated kinase mitogen-activated protein kinase and c-Fos in dendritic cells. J Immunol 172:4733–4743
Redecke V, Häcker H, Datta SK et al (2004) Cutting edge: activation of Toll-like receptor 2 induces a Th2 immune response and promotes experimental asthma. J Immunol 172:2739–2743
Abdollahi-Roodsaz S, Joosten LA, Koenders MI et al (2008) Stimulation of TLR2 and TLR4 differentially skews the balance of T cells in a mouse model of arthritis. J Clin Invest 118:205–216
Bohan A, Peter JB (1975) Polymyositis and dermatomyositis (first part). N Engl J Med 292:344–347
Bohan A, Peter JB (1975) Polymyositis and dermatomyositis (second part). N Engl J Med 292:403–407
Papadimitraki ED, Bertsias GK, Boumpas DT (2007) Toll-like receptors and autoimmunity: a critical appraisal. J Autoimmun 29:310–318
Lee JH, Cho ML, Kim JI et al (2009) IL17 increases the expression of Toll-like receptor-2, 4, and 9 by increasing IL-1 and IL-6 production in autoimmune arthritis. J Rheumatol 36:684–692
Lundberg I, Brengman JM, Engel AG (1995) Analysis of cytokine expression in muscle in inflammatory myopathies, Duchenne dystrophy, and non-weak controls. J Neuroimmunol 63:9–16
Lindberg C, Oldfors A, Tarkowski A (1995) Local T-cell proliferation and differentiation in inflammatory myopathies. Scand J Immunol 41:421–426
Tateyama M, Nagano I, Yoshioka M et al (1997) Expression of tumor necrosis factor-α in muscles of polymyositis. J Neurol Sci 146:45–51
Seko Y, Matsuda H, Kato K et al (1993) Expression of intercellular adhesion molecule-1 in murine hearts with acute myocarditis caused by coxsackievirus B3. J Clin Invest 91:1327–1336
Scheurich P, Thoma B, Ucer U, Pfizenmaier K (1987) Immunoregulatory activity of recombinant human tumor necrosis factor (TNF)-alpha: induction of TNF receptors on human T cells and TNF-alpha-mediated enhancement of T cell responses. J Immunol 138:1786–1790
Acknowledgments
This work was supported by a grant (R11-2002-098-05001-0) from the Korea Science and Engineering Foundation through the Rheumatism Research Center (RhRC) at the Catholic University of Korea and by a project grant from Pusan National University Hospital Institute.
Disclosures
None
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Geun-Tae Kim and Mi-La Cho are co-first authors.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Kim, GT., Cho, ML., Park, YE. et al. Expression of TLR2, TLR4, and TLR9 in dermatomyositis and polymyositis. Clin Rheumatol 29, 273–279 (2010). https://doi.org/10.1007/s10067-009-1316-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-009-1316-7