Abstract
The purpose of this study was to determine the prevalence of acquired antimicrobial resistance in Streptococcus pneumoniae isolated from nasopharyngeal swabs and blood and cerebrospinal fluid (CSF) specimens of 3,028 children hospitalized with signs or symptoms of pneumonia, sepsis, or meningitis in rural Philippines between 1994 and 2000. Pneumococci were identified using standard methods, serotyped, and their susceptibility to oxacillin, erythromycin, tetracycline, chloramphenicol, and trimethoprim-sulfamethoxazole was determined using the disk diffusion method. Penicillin minimum inhibitory concentrations (MICs) of the oxacillin-resistant isolates were further tested. The clonality of the penicillin-nonsusceptible (PNSP) isolates was analyzed using pulsed-field gel electrophoresis (PFGE) and multi-locus sequence typing (MLST). Altogether 1,048 isolates were analyzed, of which 35 were invasive and 1,013 nasopharyngeal isolates. None was resistant, but 22 (2.1%) were intermediately resistant to penicillin, 4 (0.2%) were resistant to chloramphenicol, 3 (0.2%) to erythromycin, 39 (3.7%) to tetracycline, and 4 (0.2%) to trimethoprim/sulfamethoxazole. Twelve of the 22 PNSP isolates were of serotype 14 and of sequence type 63. These included the two invasive PNSP isolates. PFGE profiling further identified three separate clusters among the sequence of type 63, serotype 14 (ST6314) isolates. Antimicrobial resistance in both invasive and nasopharyngeal pneumococcal pediatric isolates in rural Philippines is rare. In spite of this remote setting, the PNSP isolates of the serotype 14 clusters were of ST63 type, which has been described previously on other continents.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Streptococcus pneumoniae, the pneumococcus, is among the most important human bacterial pathogens as well as a common component of the normal nasopharyngeal microflora. It causes both mild upper respiratory infections like otitis media or sinusitis, and severe diseases like pneumonia, meningitis, and sepsis, especially in children. Pneumonia is the most common cause of death in children under 5 years of age in developing countries, and the pneumococcus its most important causative agent [1]. Resistance of pneumococcus to penicillin and to other antibiotics has been increasing worldwide [2, 3] and threatens the successful management of pneumococcal disease using early antimicrobial therapy, the main preventive strategy recommended by the World Health Organization (WHO) [4].
A generally accepted cause of the increasing frequency of antibiotic resistance is the profuse and inappropriate use of antmicrobial agents [5, 6]. In the Philippines, antibiotics are usually purchased over the counter without prescription and the choices and doses are often inappropriate [7]. The free over-the-counter availability of antibiotics has been shown to be reflected in common pre-admission antibiotic treatment in hospitalized patients [8, 9]. Against this background, the results of our present study in the Philippines are surprising. We studied the antimicrobial susceptibility among a large set of pneumococcal isolates from children under 5 years of age hospitalized for pneumonia, meningitis, and sepsis, and found an unexpectedly low frequency of strains with decreased susceptibility to penicillin, and no fully penicillin-resistant strains at all. However, a slightly increasing trend in the prevalence of penicillin-nonsusceptible pneumococci (PNSP) was observed, most likely due to the spreading of one internationally known pneumococcal clone that seems to express various serotypes and penicillin susceptibility patterns on other continents [10].
Materials and methods
Study site and preparation
Bohol in the Central Philippines is chiefly a rural area with agriculture as its principal source of livelihood. In the coastal areas, fishing is the main source of living. Of the total of one million inhabitants, about 66,000 live in the provincial capital Tagbilaran City, and the rest in small towns or rural villages. The infant mortality rate in the area is 28 per 1,000 live births, and pneumonia is considered the main health problem of children [11]. The Governor Celestino Gallares Memorial Hospital (Gallares Hospital) is a 250-bed tertiary care government hospital located in Tagbilaran City. In February 1994, preparations were started to establish a study site for acute respiratory infections (ARI) in Bohol, as a joint ARIVAC project of the Gallares Hospital, of the Research Institute for Tropical Medicine (RITM), Metro-Manila, the Philippines, the National Public Health Institute (KTL), Helsinki and Oulu, Finland, and the University of Queensland, Brisbane, Australia. This included the establishment of the ARIVAC microbiological laboratory and training of the pediatric staff at the Gallares hospital [12, 13]. The study protocol was approved by the Institutional and Ethical Review Board of the RITM. Parents/guardians of enrolled patients were requested to sign before admission the Gallares Hospital Form for Authorization for Medical and/or Surgical Treatment.
Patients
From April 1994 to June 2000, 3,028 children under 5 years of age with severe or very severe pneumonia [4], suspected meningitis, or suspected sepsis [12] were enrolled. Blood and nasopharyngeal swab samples were obtained from all the patients and cerebrospinal fluid (CSF) specimens when indicated.
Bacteriological procedures
Blood, CSF, and nasopharyngeal swab samples were cultured in the ARIVAC laboratory as described earlier [12]. In brief, all invasive isolates from blood and CSF were identified using generally accepted methods. The isolates were stored in STGG [14] at −20°C for a maximum of 1 month, after which they were sent in dry ice to the RITM for confirmation, further tests, and long-term storage at −80°C. From nasopharyngeal swab cultures only pneumococci were sought using gentamicin blood agar [12], identified, and sent to the RITM as described for invasive isolates.
Pneumococcal isolates were serotyped using the capsular Quellung test using serum pools and type-/group-specific antisera obtained from the Statens Seruminstitut, Copenhagen, with quality assurance of all invasive isolates and 10% of the nasopharyngeal isolates at the KTL, Oulu [15, 16]. Antimicrobial susceptibility was tested in the ARIVAC laboratory using the disk diffusion method according to the former National Committee for Clinical Laboratory Standards [17]. Mueller–Hinton agar was supplemented with 5% sheep’s blood. To test susceptibility to penicillin, oxacillin disks (1 μg) were used. Other antibiotic disks used were chloramphenicol (15 μg), tetracycline (30 μg), erythromycin (15 μg), and trimethoprim/sulfamethoxazole (1.25/23.75 μg; BBL, Cockeysville, MD, USA; Oxoid, Basingstoke, Hampshire, England; and Difco, Franklin Lakes, NJ, USA).
For all isolates showing decreased susceptibility in the disk diffusion test, the minimum inhibitory concentration (MIC) was determined using the E-test according to the manufacturer’s instructions (AB Biodisk, Solna, Sweden). The susceptibility results were analyzed using the WHONET computer program version 5.3 [18].
All PNSP isolates were analyzed using pulsed-field gel electrophoresis (PFGE) to assess their genetic relatedness [19]. A modification of the method of Matushek et al. was used as a standard procedure for the preparation of bacterial genomic DNA for PFGE analysis [20], which was performed as described by de Lencastre and Tomasz [21]. Restriction endonuclease SmaI (New England BioLabs, Ipswich, MA, USA) was used for digestion and the DNA fragments were separated in a 1.2% PFGE-agarose gel with a CHEF DRR III system (Bio-Rad, Hercules, CA, USA). Gels were stained in 1 mcg/ml ethidium bromide solution and photographed under UV light. Cluster analysis was performed using the BioNumerics software package (Applied Maths, Austin, TX, USA). In addition, all PNSP isolates were analyzed using multi-locus sequence typing (MLST), slightly modified from previously described protocols ([22–24] Bruno Pichon Health Protection Agency, London, UK, personal communication, 4 April 2006). Briefly, bacterial genomic DNA was isolated with the DNeasy Tissue kit (Qiagen, Hilden, Germany) and seven housekeeping genes were partially amplified using PCR. The PCR products were purified using the QiaQuick PCR Purification kit (Qiagen) or Geneclean Turbo kit (Q-BioGene, Aurora, OH, USA) and sequenced bidirectionally. The sequences obtained were compared with the material in the MLST database [10], according to which the sequence types (STs) were assigned.
Results
Among the 3,028 study children, pneumococci were isolated in 1,367 (45%) of the nasopharyngeal samples (carriage isolates) and 40 (1.3%) from blood or CSF samples (invasive isolates). All of the pneumococcal bacteremias were monobacterial. One carriage and one invasive, if existent, isolate per patient were included in the study. Due to accidental defrosting of the storage freezer at the RITM in 2000, some of the isolates were lost before testing was complete. Consequently, serotype and antimicrobial susceptibility results are now available for only 35 invasive and 1,013 nasopharyngeal isolates, and these form the material of the present study.
Of the 1,048 pneumococcal isolates, 42 (4.0%) were resistant to oxacillin in the disk diffusion test. These isolates were then tested using the penicillin E-test, and 22 (2.1%) of them were confirmed to be of decreased susceptibility to penicillin (MIC: 0.125–0.25 mg/L). Two of these PNSPs were invasive and 20 were nasopharyngeal isolates (Table 1). In the first 2 years of the study, only 2 PNSPs (0.9%) were found among the 218 isolates tested. In the following 4 years the PNSPs showed a modest increase to 2.4% of the 754 isolates tested, but in the final half-year no PNSPs were found among the 76 isolates.
Among the 1,013 nasopharyngeal isolates, 4 were resistant to chloramphenicol, 3 to erythromycin, 37 (3.6%) to tetracycline, and 4 to trimethoprim/sulfamethoxazole. Two (5.7%) of the 35 invasive isolates were resistant to tetracycline.
Among the 22 PNSP isolates, ten of the nasopharyngeal isolates and both of the invasive isolates were of serotype 14. Five of the remaining 10 PNSP nasopharyngeal isolates were noncapsulated and the serotypes of the remaining 5 were 23F (two isolates), 4, 5, and 28. All of the 12 PNSP serotype 14 isolates were resistant to tetracycline (MIC: 32–64 mg/L). Among the remaining serotype 14 PNSP isolates, this resistance pattern was found in only the one PNSP isolate with serotype 28.
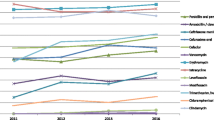
The PFGE profiling of the 12 serotype 14 PNSP isolates revealed three clusters (Fig. 1): cluster A (patterns 994, 1720, and 2118 to 2552b), cluster B (patterns 1969 to 2070), and cluster C (patterns 1867 and 1932). Both of the invasive isolates (2118b and 2552b), as well as the nasopharyngeal isolates from the same patients (2118 and 2552), belonged to the major cluster A. The degree of similarity within the clusters was 100%. The remaining one serotype 14 PNSP isolate (1189) was closely (>85%) related to the major cluster A. In addition to the three PFGE clusters, the ten non-serotype 14 PNSP isolates represented an individual PFGE profile each (data not shown). The MLST results showed that all 12 of the serotype 14 PNSP isolates of the three clusters defined by PFGE profiling shared an identical sequence type, ST63. None of the remaining ten PNSP isolates were alike according to MLST. Among these ten isolates, ST361, ST771, and eight previously undescribed STs were represented.
Discussion
This study is part of prospective surveillance of invasive bacterial infections in children under 5 years of age in the rural province of Bohol in the Central Philippines [12]. This is the first large-scale study of pneumococcal antimicrobial resistance in the Philippines. The patients came from the small city of Tagbilaran and from the surrounding rural municipalities, therefore representing a population different from the urban populations in Metropolitan Manila, where pneumococcal antibiotic resistance has been studied previously [25–27]. In these studies, 65 pediatric blood culture isolates [24], 100 nasopharyngeal isolates from nonhospitalized children under 5 years of age [26], and 22 invasive isolates [27] collected mainly in the 1990s were studied, 3, 2, and 6 of which were reported to be penicillin-nonsusceptible respectively.
The results of the present study show that resistance to penicillin and other clinically important drugs is uncommon in both invasive and nasopharyngeal isolates of pneumococci in the rural Filipino children. This low prevalence of PNSP observed is in contrast to other reports from the South East Asia. In Taiwan, Korea, Sri Lanka, Vietnam, Singapore, and Thailand, 37–92% of pneumococcal nasopharyngeal strains from children under 5 years of age attending daycare centers or outpatient clinics or hospitalized with acute respiratory infection, were penicillin-nonsusceptible [26, 28]. A lower prevalence of PNSP (13%) was reported from China, Malaysia, and India, where the number of pneumococcal isolates varied from 44 to 204 per country [26]. Similar figures from these countries have also been reported by others [29, 30]. A recent report of the ANSORP indicates even higher resistance rates in invasive pneumococci in most of the countries in the area of South East Asia [27].
Differences in antibiotic policies between countries offer an explanation for the varying frequencies of resistant pneumococcal strains. In Europe in the 1990s, high frequencies of penicillin-intermediate or -resistant strains were observed in Spain (41.2%), France (42.3%), and Hungary (36.2%), but considerably lower in the UK (9%), Germany (4%) [31], Sweden (3–5%) [32], and Finland (6%,) [33]. In Spain and Hungary, antibiotics are freely available, whereas in the UK, Sweden, and Finland a prescription is required. Thus, rational use of antibiotics seems to prevent or at least hinder the appearance and spread of antibiotic resistance [34]. Similar to elsewhere in the Philippines, antibiotics are also available over the counter in Bohol. According to patient histories, antibiotics have often been given to seriously ill children before admission to hospital [12]. Against this background, our finding of a low prevalence of PNSP in Filipino children is surprising. Without a study including a questionnaire for the parents on pre-admission antimicrobial treatment of the children and testing the urine antibacterial activity of the hospitalized children, reasons for this finding can be only speculated upon. In the South East Asian studies referred to above [25–30], the study population consisted of urban children often attending daycare. Our study population represents mostly a rural lifestyle where small children live at home. It stands to reason that transmission of resistant bacteria in a low-density population, such as in our study, is less frequent than in dense urban populations, and this could contribute to the low rate of resistance in our study population.
Epidemiological analysis of PNSP strains in South East Asia [26, 28, 35], as well as in the Americas [36, 37] and in Europe [38], indicate that the major cause of high prevalence of PNSP is due to the introduction and spread of pandemic multi-resistant clones. According to the nomenclature established by the Pneumococcal Molecular Epidemiology Network (PMEN) [39], the Spain23F-1 and Spain9V-3 multi-resistant clones are among the most common and wide-spread, and often the main cause of high prevalence of PNSP. While MLST is nowadays considered the gold standard for typing pneumococci due to its high resolution and ease of comparison, PFGE is more sensitive in detecting subtle changes in the genome [23]. Thus, the results achieved using the two methods reported here are not contradictory. None of the PFGE or MLST patterns of the PNSP isolates in our study resembled any of the patterns of the international clones published by PMEN [39]. However, 15 strains expressing five different serotypes (14, 15A, 19A, 19F, 23F) in the MLST database [10] are identical to the ST6314 PNSP isolates in this study. The antibiotic susceptibility data are not available for all the previously reported sequence type 63 isolates, but the majority of them are intermediately resistant to penicillin (MIC between 0.12–1 μg/ml). The earliest of these findings is a serotype 15A strain isolated in Sweden in 1992 [40]. The ST6314 PNSP isolates were not present in the material of the present study collected in 1994–1995, but were regularly present among the isolates from 1996–1999, consistent with its recent introduction into the population. The question of the further spread of this clone cannot unfortunately be answered because of the discontinuation of the study in June 2000.
A good situation can rapidly turn worse. The introduction of a PNSP clone with the high ability to spread can rapidly increase the proportion of resistant strains [41]. The proportion of penicillin-intermediate or -resistant strains increased in the United States from 6.6% in 1991–1992 to 33.5% in 1996–1997 [42] and to 43.6% in 1997 [43]. During the 1990s, the prevalence of multiply resistant pneumococci increased by approximately 3% in a year in the USA [44]. While resistance in pneumococci is often associated with the pediatric serotypes, i.e., major serotypes causing infections in young children [45] included in the modern pneumococcal vaccines, it has been proposed that vaccination could “clean the table” by eradicating the pediatric PNSP [46]. However, pneumococcus is a highly variable and versatile organism, and it is quite likely that under selection pressure caused by vaccination, for example, anomalous serotypes of common pediatric PNSP clones can become prevalent and the resistance problem will continue. This has already been reported from the US [47]. In our study, the ST6314 PNSP isolates were more common among the invasive isolates, in spite of their low numbers, than among the carriage isolates. One reason for this might be their ability to resist pre-hospitalization antimicrobial treatment, which is likely to prevent growth of susceptible pneumococci in blood cultures taken during admission.
References
Bryce J, Boschi-Pinto C, Shibuya K, Black RE, W.C.H.E. Group (2005) WHO estimates of the cause of death in children. Lancet 365:1147–1152
Felmingham D, White AR, Jacobs MR et al (2005) The Alexander Project: the benefits from a decade of surveillance. J Antimicrob Chemother 56 [Suppl S2]:i3–ii21
Bruinsma N, Kristinsson KG, Bronzwaer S et al, on behalf of the EARSS participants (2004) Trends of penicillin and erythromycin resistance among invasive Streptococcus pneumoniae in Europe. J Antimicrob Chemother 54:1045–1050
WHO, Management of the Young Child with an Acute Respiratory Infection (1990) World Health Organization: program for control of acute respiratory infections. World Health Organization, Geneva
Austin DJ, Kristinsson KG, Anderson RM (1999) The relationship between the volume of antimicrobial consumption in human communities and the frequency of resistance. Proc Natl Acad Sci USA 96:1152–1156
Bronzwaer SLAM, Cars O, Buchholz U et al, participants in the European Antimicrobial Resistance Surveillance System (2002) A European study on the relationship of antimicrobial use and antimicrobial resistance. Emerg Infect Dis 8:278–282
Lansang MA, Lucas-Aquino R, Tupasi TE et al (1990) Purchase of antibiotics without prescription in Manila, the Philippines: inappropriate choices and doses. J Clin Epidemiol 43:61–67
Sombrero L, Sunico ME, Quiambao B et al (1999) Reliability of parental history of antibiotic use for Filipino children admitted with acute lower respiratory tract infection. Am J Trop Med Hyg 60:397–399
Liu Y-C, Huang W-K, Huang T-S, Kunin CM (1999) Extent of antibiotic use in Taiwan shown by antimicrobial activity in urine. Lancet 354:1360
Multi Locus Sequence Typing home page, http://www.mlst.net, accessed 15 May 2007
DHE Center (2000) Philippine health statistics. DHE Center, Manila
Lupisan S, Herva E, Sombrero L et al (2000) Invasive bacterial infections in a rural province in Central Philippines. Am J Trop Med Hyg 62:341–346
Herva E, Sombrero L, Lupisan S, Arcay J, Ruutu P (1999) Establishing a laboratory for surveillance of invasive bacterial infections in a tertiary care government hospital in a rural province in the Philippines. Am J Trop Med Hyg 60:1035–1040
Kaijalainen T, Ruokokoski E, Ukkonen P, Herva E (2004) Survival of Streptococcus pneumoniae, Haemophilus influenzae and Moraxella catarrhalis frozen in skim milk-tryptone-glucose-glycerol medium. J Clin Microbiol 42:412–414
Sörensen U (1993) Typing of pneumococci by using 12 pooled antisera. J Clin Microbiol 31:2097–2100
Herva E, Granat S, Mia Z, Ollgren J, Piirainen L, Mäkelä PH (2006) Field evaluation of the chessboard modification for serotyping of Streptococcus pneumoniae in a small laboratory in Bangladesh. Am J Trop Med Hyg 74:863–867
National Committee for Clinical Laboratory Standards (1997) Performance standards for antimicrobial disk susceptibility tests; approved standard, 6th edn. NCCLS Document M2-A6, NCCLS, Villanova
World Health Organization. Drug resistance, WHONET software, http://www.who.int/drugresistance/whonetsoftware/en/
Tenover FC, Arbeit RD, Goering RV et al (1995) Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol 33:2233–2239
Matushek MG, Bonten MJ, Hayden MK (1996) Rapid preparation of bacterial DNA for pulsed-field gel electrophoresis. J Clin Microbiol 34:2598–2600
De Lencastre H, Tomasz A (2002) From ecological reservoir to disease: the nasopharynx, day-care centres and drug-resistant clones of Streptococcus pneumoniae. J Clin Microbiol 50 [Suppl 2]:75–82
Birtles A, McCarthy N, Sheppard CL et al (2005) Multilocus sequence typing directly on DNA from clinical samples and a cultured isolate to investigate linked fatal pneumococcal disease in residents of a shelter for homeless men. J Clin Microbiol 43:2004–2008
Enright MC, Spratt BG (1998) A multilocus sequence typing scheme for Streptococcus pneumoniae: identification of clones associated with serious invasive disease. Microbiology 144:3049–3060
Enright MC, Knox K, Griffiths D, Crook DWM, Spratt BG (2000) Molecular typing of bacteria directly from cerebrospinal fluid. Eur J Clin Microbiol Infect Dis 19:627–630
Capeding MR, Sombrero LT, Lucero MG, Saniel MC (1994) Serotype distribution and antimicrobial resistance of invasive Streptococcus pneumoniae isolates in Filipino children. J Infect Dis 169:479–480
Lee NY, Song JH, Kim S et al (2001) Carriage of antibiotic-resistant pneumococci among Asian children: a multinational surveillance by the Asian Network for Surveillance of Resistant Pathogens (ANSORP). Clin Infect Dis 32:1463–1469
Song JH, Jung SI, Ko KS et al (2004) High prevalence of antimicrobial resistance among clinical Streptococcus pneumoniae isolates in Asia (an ANSORP study). Antimicrob Agents Chemother 48:2101–2107
Dejsirilert S, Overweg K, Sluijter M et al (1999) Nasopharyngeal carriage of penicillin-resistant Streptococcus pneumoniae among children with acute respiratory tract infections in Thailand: a molecular epidemiological survey. J Clin Microbiol 37:1832–1838
Kim S-N, Kim S-W, Choi I-H, Pjo S-N, Rhee D-K (1996) High incidence of multi-drug-resistant Streptococcus pneumoniae in South Korea. Microb Drug Resist 2:401–406
Chion C-CC, Liu Y-C, Huang T-S et al (1998) Extremely high prevalence of nasopharyngeal penicillin-resistant Streptococcus pneumoniae in children in Kaohsiung, Taiwan. J Clin Microbiol 36:1933–1937
Felmingham D, Washington J, the Alexander Project Group (1999) Trends in antimicrobial susceptibiity of bacterial respiratory tract pathogens—findings of the Alexander project 1992–1996. J Chemother 11:5–21
Kihlström E, Normann B (1995) Occurrence of pneumococci with resistance or decreased susceptibility to penicillin in southeast Sweden. Scand J Infect Dis 27:489–494
Pihlajamaki M, Jalava J, Huovinen P, Kotilainen P, Finnish Study Group for Antimicrobial Resistance (2003) Antimicrobial resistance of invasive pneumococci in Finland in 1999–2000. Antimicrob Agents Chemother 47:1832–1835
Melander E, Ekdahl K, Jonsson G, Molstad S (2000) Frequency of penicillin-resistant pneumococci in children is correlated to community utilization of antibiotics. Pediatr Infect Dis J 19:1172–1177
Parry CM, Duong NM, Zhou J et al (2002) Emergence in Vietnam of Streptococcus pneumoniae resistant to multiple antimicrobial agents as a result of dissemination of the multiresistant Spain(23F)-1 clone. Antimicrob Agents Chemother 46:3512–3517
Richter SS, Heilmann KP, Coffman SL et al (2002) The molecular epidemiology of penicillin-resistant Streptococcus pneumoniae in the United States, 1994–2000. Clin Infect Dis 34:330–339
Wolf B, Rey LC, Brisse S et al (2000) Molecular epidemiology of penicillin-resistant Streptococcus pneumoniae colonizing children with community-acquired pneumonia and children attending day-care centres in Fortaleza, Brazil. J Antimicrob Chemother 46:757–765
Bogaert D, Syrogiannopoulos GA, Grivea JN, de Groot R, Beratis NG, Hermans PWM (2000) Molecular epidemiology of penicillin-nonsusceptible Streptococcus pneumoniae among children in Greece. J Clin Microbiol 38:4361–4366
McGee L, McDougal L, Zhou J et al (2001) Nomenclature of major antimicrobial-resistant clones of Streptococcus pneumoniae defined by the pneumococcal molecular epidemiology network. J Clin Microbiol 39:2565–2571
Sa-Leao R, Tomasz A, de Lencastre H (2001) Multilocus sequence typing of Streptococcus pneumoniae clones with unusual drug resistance patterns: genetic backgrounds and relatedness to other epidemic clones. J Infect Dis 184:1206–1210
Soares S, Kristinsson KG, Musser JM, Tomasz A (1993) Evidence for the introduction of a multiresistant clone of serotype 6B Streptococcus pneumoniae from Spain to Iceland in the late 1980s. J Infect Dis 168:158–163
Mufson MA (1998) Editorial response: penicillin-resistant Streptococcus pneumoniae increasingly threatens the patient and challenges the physician. Clin Infect Dis 27:771–773
Doern GV, Pfaller MA, Kugler K, Freeman J, Jones RN (1998) Prevalence of antimicrobial resistance among respiratory tract isolates of Streptococcus pneumoniae in North America: 1997 results from the SENTRY antimicrobial surveillance program. Clin Infect Dis 27:764–770
Mera RM, Miller LA, Daniels JJ, Weil JG, White AR (2005) Increasing prevalence of multidrug-resistant Streptococcus pneumoniae in the United States over a 10-year period: Alexander Project. Diagn Microbiol Infect Dis 51:195–200
Tomasz A (1997) Antibiotic resistance in Streptococcus pneumoniae. Clin Infect Dis 24 [Suppl 1]:585–588
Talbot TR, Poehling KA, Hartert TV et al (2004) Reduction in high rates of antibiotic-nonsusceptible invasive pneumococcal disease in Tennessee after introduction of the pneumococcal conjugate vaccine. Clin Infect Dis 39:641–648
Farrell DJ, Klugman KP, Pichichero M (2007) Increased antimicrobial resistance among nonvaccine serotypes of Streptococcus pneumoniae in the pediatric population after the introduction of 7-valent pneumococcal vaccine in the United States. Pediatr Infect Dis J 26:123–128
Acknowledgements
Members of the ARIVAC consortium who contributed to this work are: Dr Socorro Lupisan, RITM, Manila, Philippines, for directing the laboratory; Professor Helen Mäkelä, KTL, Helsinki, Finland, for consulting ARIVAC laboratory work; Dr Erma Abucejo-Ladesma, Gallares Hospital, Tagbilaran, Philippines, for being in charge of clinical activities; medical technologists Gene Baluran and Maria Felnore Girasol, Gallares Hospital, Tagbilaran, Philippines, for laboratory work; medical technologist Tarja Kaijalainen, KTL, Oulu, Finland, for quality assurance of serotyping of pneumococci.
We thank the parents of the children enrolled in this study without whose support this study would not have been possible and other members of the ARIVAC consortium for their contribution to the project. In particular we express thanks to the ARIVAC laboratory and RITM microbiology laboratory staff for laboratory work, the study nurses for patient enrollment and follow-up and the staff of Gallares Hospital, Bohol, for their contribution (the pediatric and emergency room staff for patient enrollment and management, Dr. Juanita Arcay for support in establishing the ARIVAC laboratory and acting as liaison between the hospital and the ARIVAC project).
This publication made use of the Multi Locus Sequence Typing website (http://www.mlst.net) at Imperial College London, developed by David Aanensen and Man-Suen Chan and funded by the Wellcome Trust.
This work was supported by grants from The Academy of Finland (contract Nos. 2041057 and 5569), Directorate General Research of the European Union (contract Nos. 18CT950025, IC18-CT97–0219, and ICA4-CT1999–10008), the Public Health Research and Development Committee (PHRDS) of the National Health & Medical Research Council (MHMRC), Australia (Project No. 964161) and the Finnish International Development Aid to Physicians for Social Responsibility, Finland.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
The members of the ARIVAC consortium contributing to this study are listed in the Acknowledgements
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Sombrero, L., Nissinen, A., Esparar, G. et al. Low incidence of antibiotic resistance among invasive and nasopharyngeal isolates of Streptococcus pneumoniae from children in rural Philippines between 1994 and 2000. Eur J Clin Microbiol Infect Dis 27, 929–935 (2008). https://doi.org/10.1007/s10096-008-0524-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-008-0524-4