Abstract
HAND2 antisense RNA 1 (HAND2-AS1) is a newly recognized lncRNA encoded by a gene on 4q34.1. This lncRNA has 10 exons and is predicted to have a positive effect on expression of certain genes. HAND2-AS1 is mainly considered as a tumor suppressive lncRNA in different tissues. Moreover, HAND2-AS1 has been shown to regulate expression of several targets with possible roles in the carcinogenesis through serving as a sponge for miRNAs. This lncRNA can also influence activity of BMP, TGF-β1, JAK/STAT and PI3K/Akt pathways. Down-regulation of HAND2-AS1 in tumor tissues has been associated with larger tumor size, higher tumor grade, higher chance of metastasis and poor clinical outcome. The present study aims at summarization of the impact of HAND2-AS1 in the carcinogenesis and its potential in cancer diagnosis or prediction of cancer prognosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long non-coding RNAs (lncRNAs) are a group of transcripts with established roles in the fundamental process of cell physiology. Abnormal expressions of these transcripts have been linked with the development of several disorders, particularly cancers [1, 2]. This is mainly attributed to the important effects of lncRNAs in the regulation of cell cycle transition, cell proliferation, apoptosis and differentiation as well as stemness [3, 4]. Through interacting with mRNAs, DNA molecules, proteins, and miRNAs, lncRNAs influence expression of genes at almost all possible levels [5]. At epigenetic level, they mediate chromatin modification and DNA methylation. At transcriptional level, they mainly interact with transcription factors and DNA sequences. Moreover, they modulate mRNA processing; thus, they are involved in the regulation of gene expression at the post-transcriptional level. Finally, they have functional interactions with proteins to affect translational and post-translational mechanisms [5].
HAND2 antisense RNA 1 (HAND2-AS1) is a newly recognized lncRNA encoded by a gene on 4q34.1, in an opposite direction to HAND2 gene. Transcription of HAND2AS1 might regulate expression of HAND2 (class A basic helix–loop–helix protein 26), a regulator of heart development which is involved in the pathogenesis of cardiomyopathy [6]. HAND2-AS1 has 10 exons and is predicted to have a positive effect on the expression of certain genes. Moreover, it has been mainly detected in the cytoplasm [6]. Several alternative spliced variants have been identified for this lncRNA with different sets of exons (Fig. 1).
HAND2-AS1 is located on human chromosome 4. It has at least 11 transcripts (based on the NCBI database). HAND2-AS1 has various functions at different levels, such as DNA, mRNA, and protein levels. The early onset and poor prognosis of several tumor types are also related to lower HAND2-AS1 expression
Although the main physiological role of HAND2-AS1 is regulation of cardiac development through influencing expression of HAND2 [6], most of studies conducted on the function of this lncRNA have been performed in the context of cancer. The present study aims at summarization of the impact of HAND2-AS1 in the carcinogenesis and its potential in cancer diagnosis or prediction of cancer prognosis. We have organized the obtained evidence from literature into three distinct subtitles, including cell line studies, studies in xenograft models of cancer and studies in clinical samples.
Search strategy
We searched PubMed and Google Scholar databases with key words "HAND2-AS1" AND "Cancer" OR "Tumor." Then, the relevance of the retrieved articles was assessed through reviewing the abstract and full text of the manuscript. After reviewing 50 articles, a total of 31 articles were selected for inclusion in the manuscript. The inclusion criteria were: 1. type of publication: original study, 2. language: English, 3. study design: in vitro and/or studies in clinical samples.
In vitro studies
Function of HAND2-AS1 has been assessed in a variety of cancer cell lines mainly though knock-in studies. These studies have used some techniques to enhance expression of HAND2-AS1 in a variety of cancer cell lines. Then, they have assessed the functional consequences of over-expression of HAND2-AS1 in these cell lines.
Colorectal cancer
Forced overexpression of HAND2-AS1 in colorectal cancer cells has reduced the proliferation ability and invasive properties of these cells. Mechanistically, HAND2-AS1 can serve as a molecular sponge for miR-1275. This miRNA can target KLF14; therefore, HAND2-AS1 suppresses progression of colorectal cancer via enhancing KLF14 expression [7]. This study has confirmed the tumor suppressor role of HAND2-AS1 in colorectal cancer.
HAND2-AS1 has also been found to be down-regulated in 5-fluororacil-resistant colorectal cancer cells, parallel with down-regulation of PDCD4 and up-regulation of miR-20a. HAND2-AS1 could suppress 5-fluororacil resistance, cell proliferation, migratory potential and invasive properties and promote cells apoptosis in 5-fluororacil-resistant colorectal cancer cells. Mechanistically, HAND2-AS1 acts as a sponge for miR-20a to modulate levels of PDCD4 [8]. Thus, HAND2-AS1 is a target for modulating resistance of colorectal cancer cells to 5-fluororacil.
Esophageal cancer
Up-regulation of HAND2-AS1 in squamous cell carcinoma cells of esophagus has resulted in downregulation of miR-21 and inhibition of cell proliferation, migration, and invasive aptitude of these cells. miR-21 up-regulation could not influence expression level of HAND2-AS1, yet it could attenuate the suppressive impact of HAND2-AS1 up-regulation on these cells [9]. Cumulatively, tumor suppressive roles of HAND2-AS1 in esophageal cancer are exerted through regulation of miR-21 expression.
Lung cancer
An experiment in lung cancer cells has shown that HAND2-AS1 over-expression blocks migration and invasive aptitude and stemness features of cells, while TGF-β1 has the opposite effects. Up-regulation of HAND2-AS1 in these cells has led to down-regulation of TGF-β1, while TGF-β1 has failed to affect expression of HAND2-AS1 (Fig. 2). However, TGF-β1 could attenuate the inhibitory effect of HAND2-AS1 up-regulation on invasive properties of lung cancer cells. Therefore, HAND2-AS1 has a role in the regulation of migratory potential, invasion, and stemness of lung cancer cells via interacting with TGF-β1 [10].
Hepatocellular carcinoma
In hepatocellular carcinoma, HAND2-AS1 reduces viability of cancer cells through modulation of miR-300/SOCS5 axis [11]. Moreover, it can suppress cancer cell proliferation through down-regulation of RUNX2 [12] and ROCK2 [13]. Besides, HAND2-AS1 suppresses proliferation of liver cancer cells and their migration through enhancing expression of SOCS5 and inactivating the JAK-STAT signaling [14]. Thus, malignant feature of hepatocellular carcinoma cells is modulated by HAND2-AS1 via different mechanisms.
In spite of the observed down-regulation of HAND2-AS1 in most of cancer cells, this lncRNA has been found to be over-expressed in liver cancer stem cells (CSCs). Most notably, HAND2-AS1 has a fundamental role in the maintenance of self-renewal of these CSCs and is required for development of hepatocellular carcinoma. From a mechanistical point of view, HAND2-AS1 can recruit the INO80 chromatin-remodeling complex to the BMPR1A promoter to induce expression of this gene and subsequently activate BMP signaling [15]. Table 1 shows the role of HAND2-AS1 in different cancer cell lines.
Animal studies
Up-regulation of HAND2-AS1 has been shown to inhibit propagation of colorectal cancer [7] and its in vivo growth [8]. Similar effects have been reported for HAND2-As1 up-regulation in cervical, breast, ovarian, bladder, and gastric cancers (Table 2).
However, experiments in the humanized models of hepatocellular carcinoma have shown that antisense oligonucleotide-mediated suppression of HAND2-AS1 exerts anti-tumor activity in a synergic manner with siRNA-mediated inhibition of BMPR1A. Besides, silencing of lncHand2 or Bmpr1a in mice hepatocytes has been shown to impair BMP signaling and suppress development of liver cancer [15]. Therefore, there is a discrepancy between the bulk of evidence from most types of cancers versus hepatocellular carcinoma.
Experiments in clinical samples
HAND2-AS1 has been demonstrated to be under-expressed in colorectal cancer tissues. Moreover, expression levels of HAND2-AS1 have been negatively correlated with metastasis and advanced stages. Besides, HAND2-AS1 down-regulation has been identified as a marker of poor prognosis [7]. Another study in colorectal cancer tissues has demonstrated down-regulation of this lncRNA in 5-fluorurcil-resistant tissues [8].
In addition, HAND2-AS1 has been reported to be downregulated in esophagus squamous cell carcinoma tissues, while miR-21 has been upregulated in these tissues compared with paired adjacent healthy tissues. Transcript levels of HAND2-AS1 and miR-21 have been inversely correlated in tumor samples but not in non-tumoral samples of these patients. Notably, authors have reported down-regulation of plasma levels of HAND2-AS1 in esophagus squamous cell carcinoma patients compared with normal persons. Besides, downregulation of plasma levels of HAND2-AS1 could distinguish early stage of this cancer from healthy status [9]. Thus, HAND2-AS1 can be regarded as a marker for early detection of cancer.
HAND2-AS1 expression has also been lower in lung cancer tissues compared with adjacent healthy tissues. Moreover, plasma levels of HAND2-AS1 have been lower in patients with lung cancer compared with controls, while TGF-β levels have been higher in these patients. Notably, plasma levels of HAND2-AS1 and TGF-β1 have been negatively correlated in patients but not in control subjects [10]. This study further supports the potential of HAND2-AS1 in cancer diagnosis.
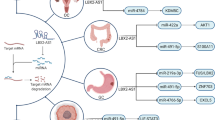
In chronic myeloid leukemia (CML), down-regulation of HAND2-AS1 has been much prominent in accelerated and blast phases compared with chronic phase [18], indicating a putative prognostic role for this transcript. Totally, down-regulation of HAND2-AS1 has been associated with metastasis in colorectal cancer [7] and hepatocellular carcinoma [11]. Moreover, its down-regulation has been associated with advanced stage in colorectal cancer [7] and with high pathological grade in hepatocellular carcinoma [11], endometrial cancer [24], bladder cancer [27], and glioma [37]. Finally, levels of this lncRNA are associated with tumor size in non-small cell lung cancer [16], osteosarcoma [20], and melanoma [36]. Table 3 shows the role of HAND2-AS1 as described by studies in clinical samples.
Discussion
This review provides a comprehensive summary of the expression level, function, and prognostic value of HAND2-AS1 in different cancers. Based on the above-mentioned data, HAND2-AS1 is mainly regarded as a tumor suppressive lncRNA in different tissues. The most possible explanation for its down-regulation in cancer tissues is hypermethylation of its promoter as demonstrated in ovarian cancer tissues [39]. Moreover, HAND2-AS1 has been shown to regulate expression of several targets with possible roles in the carcinogenesis through serving as a sponge for miRNAs. miR-1275, miR-20a, miR‐21, miR-300, miR-590-3p, miR-184, miR-769–5p, miR‐340‐5p, miR‑106a, miR‑146, miR‑330‑5p, miR-106a-5p, and miR-3118 are the main miRNAs that are influenced by this lncRNA. The interaction between HAND2-AS1 and miR-1275 has been verified in different contexts, including colorectal cancer, CML, and breast cancer. Most notably, the rs2276941 polymorphism within HAND2-AS1 has been shown to affect its binding with hsa-miR-1275 and influence the susceptibility to colorectal cancer [40].
BMP, TGF-β1, JAK/STAT, and PI3K/Akt pathways are the main cancer-related pathways being influenced by HAND2-AS1. In addition to regulation of cell proliferation and apoptosis, it can affect cancer metabolism and angiogenesis through modulation of a number of targets including GLUT1, VEGFA, and HIF1α. Most importantly, HAND2-AS1 can modulate response of cancer cells to 5-fluouracil and cisplatin.
The impact of HAND2-AS1 on the maintenance of CSCs should be further investigated, since data regarding this function are contradictory [10, 15]. Based on the importance of this population of cells in tumor metastasis and recurrence, this issue has practical significance.
Experiments in clinical samples have shown association between dysregulation of HAND2-AS1 and a number of tumor characteristics such as level of differentiation, tumor size, lymph node metastasis, and most importantly overall survival of patients. Thus, evidence regarding the prognostic role of HAND2-AS1 is ample. On the other hand, diagnostic value of this lncRNA has not been investigated thoroughly.
Although the clinical application of HAND2-AS1 as a therapeutic target has not been assessed, based on the observed down-regulation of this lncRNA in a vast variety of tumors with different origins, up-regulation of HAND2-AS1 can be regarded as a therapeutic target for several types of cancer. This can be achieved by specific epigenetic regulators that affect epigenetic marks in the promoter of this lncRNA. This therapeutic modality can also affect response of patients to conventional chemotherapeutic agents.
Finally, the impact of polymorphisms within HAND2-AS1 gene on risk of cancer has not been investigated. These polymorphisms can affect binding of this lncRNA to certain miRNAs, thus influencing the functional roles of HAND2-AS1.
Future perspectives
Forced up-regulation of HAND2-AS1 in cancer cells is a possible route for reduction in malignant features. Thus, the applicability and safety of this method should be assessed in clinical settings to find a novel treatment option for cancer.
Data availability
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
References
Ghafouri-Fard S, Hussen BM, Gharebaghi A, Eghtedarian R, Taheri M. LncRNA signature in colorectal cancer. Pathol Res Practice. 2021;222:153432.
Hussen BM, Hidayat HJ, Ghafouri-Fard S. Identification of expression of CCND1-related lncRNAs in breast cancer. Pathol Res Practice. 2022;236:154009.
Ghafouri-Fard S, Hajiesmaeili M, Shoorei H, Bahroudi Z, Taheri M, Sharifi G. The impact of lncRNAs and miRNAs in regulation of function of cancer stem cells and progression of cancer. Front Cell Develop Biol. 2021;9:696820.
Hussen BM, Kheder RK, Abdullah ST, et al. Functional interplay between long non-coding RNAs and Breast CSCs. Cancer Cell Int. 2022;22(1):233.
Zhang X, Wang W, Zhu W, et al. Mechanisms and functions of long non-coding RNAs at multiple regulatory levels. Int J Mole Sci. 2019;20(22):5573.
Anderson KM, Anderson DM, McAnally JR, Shelton JM, Bassel-Duby R, Olson EN. Transcription of the non-coding RNA upperhand controls Hand2 expression and heart development. Nature. 2016;539(7629):433–6.
Zhou J, Lin J, Zhang H, Zhu F, Xie R. LncRNA HAND2-AS1 sponging miR-1275 suppresses colorectal cancer progression by upregulating KLF14. Biochem Biophys Res Commun. 2018;503(3):1848–53.
Jiang Z, Li L, Hou Z, et al. LncRNA HAND2-AS1 inhibits 5-fluorouracil resistance by modulating miR-20a/PDCD4 axis in colorectal cancer. Cell Signal. 2020;66:109483.
Yan Y, Li S, Wang S, et al. Long noncoding RNA HAND2-AS1 inhibits cancer cell proliferation, migration, and invasion in esophagus squamous cell carcinoma by regulating microRNA-21. J Cell Biochem. 2019;120(6):9564–71.
Miao F, Chen J, Shi M, Song Y, Chen Z, Pang L. LncRNA HAND2-AS1 inhibits non-small cell lung cancer migration, invasion and maintains cell stemness through the interactions with TGF-beta1. 2019. Biosci Rep. https://doi.org/10.1042/BSR20181525.
Bi HQ, Li ZH, Zhang H. Long noncoding RNA HAND2-AS1 reduced the viability of hepatocellular carcinoma via targeting microRNA-300/SOCS5 axis. Hepatobiliary Pancreat Dis Int HBPD INT. 2020;19(6):567–74.
Jing GY, Zheng XZ, Ji XX. lncRNA HAND2-AS1 overexpression inhibits cancer cell proliferation in hepatocellular carcinoma by downregulating RUNX2 expression. J Clin Lab Anal. 2021;35(4):e23717.
Jiang L, He Y, Shen G, et al. lncRNA HAND2-AS1 mediates the downregulation of ROCK2 in hepatocellular carcinoma and inhibits cancer cell proliferation, migration and invasion. Mole Med Rep. 2020. https://doi.org/10.3892/mmr.2020.10928.
Yan D, Jin F, Lin Y. lncRNA HAND2-AS1 inhibits liver cancer cell proliferation and migration by upregulating SOCS5 to inactivate the JAK-STAT pathway. Cancer Biother Radiopharm. 2020;35(2):143–52.
Wang Y, Zhu P, Luo J, et al. LncRNA HAND2-AS1 promotes liver cancer stem cell self-renewal via BMP signaling. EMBO J. 2019;38(17):e101110.
Gao T, Dai X, Jiang Y, He X, Yuan S, Zhao P. LncRNA HAND2-AS1 inhibits proliferation and promotes apoptosis of non-small cell lung cancer cells by inactivating PI3K/Akt pathway. 2020. Biosci Rep. https://doi.org/10.1042/BSR20201870.
Zhang L, Jin C, Yang G, Wang B, Hua P, Zhang Y. LncRNA WTAPP1 promotes cancer cell invasion and migration in NSCLC by downregulating lncRNA HAND2-AS1. BMC Pulm Med. 2020;20(1):153.
Yang JR, Shi MX, Zeng Y. LncRNA HAND2-AS1 inhibits proliferation and promotes apoptosis of chronic myeloid leukemia cells by sponging with micRNA-1275. Eur Rev Med Pharmacol Sci. 2019;23(5):2103–11.
Kang Y, Zhu X, Xu Y, et al. Energy stress-induced lncRNA HAND2-AS1 represses HIF1α-mediated energy metabolism and inhibits osteosarcoma progression. Am J Cancer Res. 2018;8(3):526–37.
Chen S, Xu X, Lu S, Hu B. Long non-coding RNA HAND2-AS1 targets glucose metabolism and inhibits cancer cell proliferation in osteosarcoma. Oncol Lett. 2019;18(2):1323–9.
Yu L, Li H, Li Z, et al. Long non-coding RNA HAND2-AS1 inhibits growth and migration of gastric cancer cells through regulating the miR-590-3p/KCNT2 Axis. Onco Targets Ther. 2020;13:3187–96.
Xu Z, Lv H, Wang Y, et al. HAND2-AS1 inhibits gastric adenocarcinoma cells proliferation and aerobic glycolysis via miRNAs sponge. Cancer Manag Res. 2020;12:3053–68.
Yu L, Luan W, Feng Z, et al. Long non-coding RNA HAND2-AS1 inhibits gastric cancer progression by suppressing TCEAL7 expression via targeting miR-769-5p. Dig Liver Dis. 2021;53(2):238–44.
Yang X, Wang CC, Lee WYW, Trovik J, Chung TKH, Kwong J. Long non-coding RNA HAND2-AS1 inhibits invasion and metastasis in endometrioid endometrial carcinoma through inactivating neuromedin U. Cancer Lett. 2018;413:23–34.
Chen J, Lin Y, Jia Y, Xu T, Wu F, Jin Y. LncRNA HAND2-AS1 exerts anti-oncogenic effects on ovarian cancer via restoration of BCL2L11 as a sponge of microRNA-340-5p. J Cell Physiol. 2019;234(12):23421–36.
Li L, Li L, Hu L, Li T, Xie D, Liu X. Long noncoding RNA HAND2AS1/miR106a/PTEN axis resensitizes cisplatin-resistant ovarian cells to cisplatin treatment. Mol Med Rep. 2021. https://doi.org/10.3892/mmr.2021.12402.
Shan L, Liu W, Zhan Y. LncRNA HAND2-AS1 exerts anti-oncogenic effects on bladder cancer via restoration of RARB as a sponge of microRNA-146. Cancer Cell Int. 2021;21(1):361.
Jin L, Ji J, Shi L, Jin S, Pei L. lncRNA HAND2-AS1 inhibits cancer cell proliferation, migration and invasion by downregulating ROCK1 in HPV-positive and negative cervical squamous cell carcinoma. Exp Ther Med. 2019;18(4):2512–8.
Gong J, Fan H, Deng J, Zhang Q. LncRNA HAND2-AS1 represses cervical cancer progression by interaction with transcription factor E2F4 at the promoter of C16orf74. J Cell Mol Med. 2020;24(11):6015–27.
Gao Y, Zou T, Liang W, Zhang Z, Qie M. Long non-coding RNA HAND2-AS1 delays cervical cancer progression via its regulation on the microRNA-21-5p/TIMP3/VEGFA axis. Cancer Gene Ther. 2021;28(6):619–33.
Chen S, Wang J. HAND2-AS1 inhibits invasion and metastasis of cervical cancer cells via microRNA-330-5p-mediated LDOC1. Cancer Cell Int. 2019;19:353.
Xing L, Tang X, Wu K, Huang X, Yi Y, Huan J. LncRNA HAND2-AS1 suppressed the growth of triple negative breast cancer via reducing secretion of MSCs derived exosomal miR-106a-5p. Aging (Albany NY). 2020;13(1):424–36.
Dong G, Wang X, Jia Y, et al. HAND2-AS1 works as a ceRNA of miR-3118 to suppress proliferation and migration in breast cancer by upregulating PHLPP2. Biomed Res Int. 2020;2020:8124570.
Wang Y, Cai X. Long noncoding RNA HAND2-AS1 restrains proliferation and metastasis of breast cancer cells through sponging miR-1275 and promoting SOX7. Cancer Biomark. 2020;27(1):85–94.
Wei M, Liu L, Wang Z. Long non-coding RNA heart and neural crest derivatives expressed 2-antisense RNA 1 overexpression inhibits the proliferation of cancer cells by reducing RUNX2 expression in triple-negative breast cancer. Oncol Lett. 2019;18(6):6775–80.
Liu P, Du R, Yu X. LncRNA HAND2-AS1 overexpression inhibits cancer cell proliferation in melanoma by downregulating ROCK1. Oncol Lett. 2019;18(2):1005–10.
Liu S, Li Y. LncRNA HAND2-AS1 attenuates glioma cell proliferation, invasion and migration by targeting CDK6. Neurol Res. 2022;44(8):677–83.
Jiang L, He Y, Shen G, et al. lncRNA HAND2-AS1 mediates the downregulation of ROCK2 in hepatocellular carcinoma and inhibits cancer cell proliferation, migration and invasion. Mol Med Rep. 2020;21(3):1304–9.
Gokulnath P, de Cristofaro T, Manipur I, Di Palma T, Soriano AA, Guarracino MR, et al. Long non-coding RNA HAND2-AS1 acts as a tumor suppressor in high-grade serous ovarian carcinoma. Int J Mol Sci. 2020;21(11):4059.
Gao X, Zhang S, Wang X. HAND2-AS1 rs2276941 polymorphism affecting the binding of hsa-miR-1275 Is associated with the risk of colorectal cancer. DNA Cell Biology. 2022;41:600–5.
Acknowledgements
The authors would like to thank the clinical Research Development Unit (CRDU) of Loghman Hakim Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran, for their support, cooperation, and assistance throughout the period of study.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
SGF wrote the draft and revised it. MT designed and supervised the study. MD, BMH, SRA, and AK collected the data and designed the figures and tables. All the authors read the submitted version and approved it.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participant
Not applicable.
Consent of publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghafouri-Fard, S., Hussen, B.M., Abdullah, S.R. et al. A review on the role of HAND2-AS1 in cancer. Clin Exp Med 23, 3179–3188 (2023). https://doi.org/10.1007/s10238-023-01092-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01092-3