Abstract
To evaluate the influence of the loss of coronal and radicular tooth structure on the biomechanical behavior and fatigue life of an endodontically treated maxillary premolar with confluent root canals using finite element analysis (FEA). An extracted maxillary second premolar was scanned to produce intact (IT) 3D model. Models were designed with an occlusal conservative access cavity (CAC) with different coronal defects; mesial defect (MO CAC), occlusal, mesial and distal defect (MOD CAC), and 2 different root canal preparations (30/.04, and 40/.04) producing 6 experimental models. FEA was used to study each model. A simulation of cycling loading of 50N was applied occlusally to stimulate the normal masticatory force. Number of cycles till failure (NCF) was used to compare strength of different models and stress distribution patterns via von Mises (vM) and maximum principal stress (MPS). The IT model survived 1.5 × 1010 cycles before failure, the CAC-30.04 had the longest survival of 1.59 × 109, while the MOD CAC-40.04 had the shortest survival of 8.35 × 107 cycles till failure. vM stress analysis showed that stress magnitudes were impacted by the progressive loss of coronal tooth structure rather than the radicular structure. MPS analysis showed that significant loss of coronal tooth structure translates into more tensile stresses. Given the limited size of maxillary premolars, marginal ridges have a critical role in the biomechanical behavior of the tooth. Access cavity preparation has a much bigger impact than radicular preparation on their strength and life span.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The biomechanical behavior of endodontically treated teeth (ETT) is affected by the amount of lost tooth structure before, during, and after treatment [1]. Loss of tooth integrity due to caries and extensive cavity preparation is the main reason for the reduced stiffness of ETT, rather than dehydration or physical changes in dentin [2, 3]. In addition, the lack of vitality severely limits sensory feedback during peak loads and makes non vital teeth more susceptible to fracture [4]. Unfortunately, most fractures are non-restorable [5].
Maxillary premolars have a high incidence [6] and greatest susceptibility [7] to fracture under occlusal loading. Their narrow cervical thickness, presence of a concavity on the mesial aspect of the root, and a radicular groove on the palatal aspect of the buccal root predispose them to cusp fractures, wedging, and splitting [8,9,10].
Previous experimental studies showed that loss of tooth walls, particularly the marginal ridges, causes a significant reduction in tooth fracture resistance more than different access cavity designs [11,12,13,14]. Nonetheless, the current endodontic literature lacks studies examining the interaction between coronal integrity, access designs, and shaping parameters on the biomechanical behavior and fatigue life of ETT.
This study sought to assess the influence of loss of the mesial, or the mesial and distal walls in combination with different access designs and shaping parameters on the biomechanical behavior and fatigue life of an endodontically treated maxillary second premolar with confluent root canals using finite element analysis (FEA) method.
FEA can isolate the effect of each parameter in a scenario that mimics the clinical situation [15,16,17]. Also, it overcomes the limitation of experimental laboratory tests, such as the standardization of teeth because of possible variations in dentin mechanical properties, age, tooth extraction forces, storage time, and storage medium after extraction [18]. The null hypothesis was that there is no difference between the effects of loss of coronal or radicular tooth structure on the biomechanical behavior and fatigue life of ETT.
Materials and methods
Finite element model generation
An intact, single-rooted maxillary second premolar with confluent canals and a mature apex was scanned using high-resolution Cone Beam Computed Tomography machine (Planmeca ProMax 3d MID; Planmeca, Helsinki, Finland) at 90 kV, 12 mA with a voxel dimension of 75 μm. Then, the generated DICOM images were 3D reconstructed using a Materialize interactive medical image control system (MIMICS version 21; Materialise, Leuven, Belgium). The same software was used to identify enamel and dentin and produce the three-dimensional model by forming masks and automatically growing threshold regions. Data were then optimized using the 3-Matic Medical 11.0 software (Materialise, Leuven, Belgium). SolidWorks (Dassault Systemes, Paris, France) to combine enamel and dentin and establish the surrounding periodontal ligaments and the surrounding bone [15,16,17]. Model validation was done according to Nawar et al. [17].
Ethical committee approval
This study has ethical clearance from the research ethics committee Faculty of Dentistry at the British University in Egypt (FD BUE REC 21-003).
Cavity designs
After producing the intact (IT) model, 6 experimental models (3 coronal variations with 2 parameters for root canal shaping for each) were generated (Figs. 1 and 2);
-
A)
Conservative Endodontic Access (CAC): designed by drawing lines from the center of each of the buccal and palatal root canal orifices at the furcation level, then extending them to the occlusal surface, resulting in 2 cross-points that were connected to produce the access outline [19, 20].
-
B)
Conservative Endodontic Access along with loss of the mesial wall (MO CAC): To create the proximal part, the middle 1/3 of the buccopalatal width of the tooth in the mesial one third was removed including the marginal ridge with an occlusogingival depth of 1.5–2 mm on the outer surface [21, 22].
-
C)
Conservative Endodontic Access with loss of both proximal walls (MOD CAC). Two proximal parts are created the same way done with the MO CAC.
The junctions between the occlusal and the proximal cavities were made to be smooth and curvy to avoid establishing stress concentration areas. Access cavities and missing proximal wall(s) were filled with simulated composite resin material (Fig. 1), and the volume of simulated composite representing the amount of lost tooth structure is listed in Table 1.
Root canal preparation
Root canal preparations were simulated as either (#30/0.04 taper) or (#40/0.04 taper) as shaping parameters [16]. This was done by drawing a line in the central axis of the root canal, then creating a conical shape around it with the target dimensions. The prepared root canals were filled with simulated gutta-percha filling materials, 0.5 mm short from the apex of the root canal up to 2 mm from the canal orifices (Fig. 2).
Meshing and set material properties
All models were imported into the Cosmos software package (Solid works software package; Dassault Systems) for meshing. Teeth, as well as all materials used, were considered homogeneous, linear, and isotropic [23]. The elastic modulus and Poisson’s ratio of structures used to set up FEA models are listed in Table 2, whereas the plot of stress/Number of cycles to failure (SN curve) for both enamel and dentin was set according to Gao et al. [24] and Kinney et al. [25]. The numbers of nodes and tetrahedral elements ranged from 38,411 and 63,715, respectively (solid model), to 40,639 and 69,287, respectively (MOD CAC model). Considering the bounding conditions, the cancellous bone block was fixed mesially and distally and all components were simulated to have bonded contacts (Fig. 3).
Finite element analysis
The models were subjected to cyclic loading with a magnitude of 50 N [26] to simulate the clinical masticatory loading. Loading areas used followed the pattern of Lim et al. [27]. First, the solid model loading simulation was done and the number of cycles until failure (NCF) was registered as well as the failure location. Other models’ simulations were then performed, and their life span was calculated as the percentage of the NCF of each model compared with the solid model. After load application of all models, mathematical analysis of the stress distribution patterns, von Mises (VM) stresses, and Maximum Principal Stress (MPS) were assessed using Cosmos software package (Solid works software package; Dassault Systems).
Results
The increase (%) in stress within each model and the decrease (%) in the NCF compared with the IT model are presented in Table 3. Also, stress distribution patterns, maximum vM and MPS are shown in (Figs. 4 and 5).
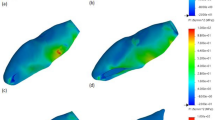
Composite figure showing the stress distribution of the VMS from different views (occlusal, isometric “mesio palatal line angle”, coronal cut at 12.5 mm from the apex, middle cut at 7 mm from the apex, and apical cut at 1.20 mm from the apex) of all models with different access cavities, and different root canal preparation
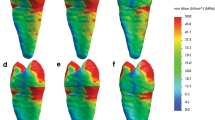
Composite figure showing the stress distribution of the MPS from different views (occlusal, isometric “mesio palatal line angle”, coronal cut at 12.5 mm from the apex, middle cut at 7 mm from the apex, and apical cut at 1.20 mm from the apex) of all models with different access cavities, and different root canal preparation
The IT model had the least maximum vM stress with a value of 6.14 MPa and survived 1.5 × 1010 cycles before failure. CAC-30.04 had the longest survival of 1.59 × 109 NCF and the least maximum vM of 7.7 MPa, while the MOD CAC-40.04 had the shortest survival of 8.35 × 107 NCF and the highest maximum vM of 10.37 MPa.
Increasing the radicular shaping parameters from 30.04 to 40.04 had a minimal effect on vM stresses and NCF of all coronal configurations accounting for a lifespan reduction of 0.11% within the CAC models, 0.13% within the MO CAC models, and 0.39% within the MOD CAC models.
When the MO CAC and the MOD CAC models were compared together, the NCF ranged from 8.35 × 107 for the MOD CAC-40.04 to 4.49 × 108 for the MO CEC-30.04. Maximum vM stress ranged from 8.75 MPA with the MO CAC 30.04 model to 10.37 MPa for the MOD CAC 40.04.
When MPS was analyzed, stress distribution patterns did not vary among experimental models with the maximum tensile stress located at the buccal restoration/tooth interface, however, stresses character leans more toward being tensile with the loss of more coronal structures. MPS values are displayed in Fig. 6.
Discussion
Minimally invasive endodontics has been suggested to increase the life span of ETT through minimal dentin removal and preservation of the pericervical dentin [28]. This study investigated the impact of each of the loss of coronal and radicular tooth structure alone and combined on the life span of a tooth with a high fracture susceptibility.
FEA was used for stress analysis in the present study, because it has the merit of standardization through the evaluation of one tested variable while virtually fixating all other contributors, thus providing reliable results and numerically controlled testing [29]. Also, fracture methodology used for in vitro analyses of ETT does not accurately reflect intraoral conditions in which failures occur primarily because of fatigue [30].
The conservative access design was used in this study as previous reports demonstrated its superiority over the traditional access from the biomechanical perspective, whereas the ultraconservative access cavity designs such as the ninja access or orifice directed access were not included in this study, because the literature does not support the notion that they provide any additional mechanical advantage beyond what is provided by conservative designs [20]. On the other hand, evidence suggests that they compromise cleaning and predispose to procedural errors [31].
In this study, simulated cyclic loading was applied, given the fact that clinically most of the failures of ETT are caused by cyclic fatigue with a subcritical load or fluctuating stresses which are much lower than the load capacity required to cause a catastrophic failure [25]. Such repeated masticatory loading cycles cause fatigue failure due to the cumulative effect of crack initiation and propagation over time [32].
This study presented that the loss of a proximal wall had a considerable impact on the life span of ETT with a reduction of approximately 6% of the tooth’s expected life. Analyzing the maximum vM stress values demonstrated that the recorded stress magnitude is directly proportional to the amount of dental structure lost. When the CAC model is compared to the intact model, stresses are raised by approximately 25.4%. This stress increase reached 42.5% with the loss of the mesial marginal ridge in the MO CAC model, and 67.7% with the additional loss of the distal marginal ridge in the MOD CAC model. This agrees with Reeh et al. [2] and Corsentino et al. [12] who recommended the preservation of marginal ridge(s) integrity to maintain tooth strength.
Analysis of stresses whether in Maximum Principal stresses or von Mises stresses were reflected in the fatigue, the solid model was used as a control reference, and the CAC showed the highest life log percentage of 90.42% followed by the MC CAC with 85.03% and the least lifelog percentage was recorded for MOD CAC model by 78.24%. This comes in agreement with previous studies [15,16,17]. It is worth mentioning that the value of tensile stresses in the tooth with a MOD cavity (≈ 4.5 MPa) was approximately 50% more than that found in the intact tooth (3.1 MPa). This may be a concern given that dental tissues are more vulnerable to stresses with tensile character [33, 34] and justify adopting full coverage restorations in cases where both proximal sides of a maxillary premolar are compromised.
Finally, our study showed that varying the apical preparation of the root canals did not appear to make a substantial mechanical difference regardless of the amount of coronal structures lost. This agrees with previous studies [15,16,17] and emphasizes that most of the functional loads are absorbed and housed by the coronal structures. This finding contrasts with Smoljan et al. [35] who found that wider progressive taper preparations have less fracture resistance than narrow progressive taper canal preparations. This can be attributed to the difference in loading conditions as they utilized 200N in static loading and the numerical values of total deformation as an indicator of fracture [33].
Most of the limitations of this study are linked to FEA, which is a computerized virtual method that differs from the clinical scenario that cannot be easily replicated. The mechanical properties of materials are set as uniform isotropic materials, when in fact it is well established that dental structures including the tubular structure of dentin and dentin-enamel junction are functionally graded materials with varying elastic models and creep-related behavior [17].
Conclusion
-
1.
The biomechanical behavior and the life span of maxillary premolars were more influenced by the loss of coronal tooth structure.
-
2.
The marginal ridges are structurally very important and should be preserved whenever possible.
-
3.
As long as the extent of the radicular preparation is kept within the widely accepted preparation sizes, it has almost no effect on the fatigue life of endodontically treated single-rooted maxillary premolars.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Haridy MF, Ahmed HS, Kataia MM, Saber SM, Schafer E. Fracture resistance of root canal-treated molars restored with ceramic overlays with/without different resin composite base materials: an in vitro study. Odontology. 2022;110:497–507.
Reeh ES, Messer HH, Douglas WH. Reduction in tooth stiffness as a result of endodontic and restorative procedures. J Endod. 1989;15:512–6.
Huang TJG, Schilder H, Nathanson D. Effects of moisture content and endodontic treatment on some mechanical properties of human dentin. J Endod. 1992;18:209–15.
Chang CY, Kuo JS, Lin YS, Chang YH. Fracture resistance and failure modes of CEREC endo-crowns and conventional post and core-supported CEREC crowns. J Dent Sci. 2009;4:110–7.
McDaniel RJ, Davis RD, Murchison DF, Cohen RB. Causes of failure among cuspal-coverage: amalgam restorations: a clinical survey. J Am Dent Assoc. 2000;131:173–7.
Abe Y, Nogami K, Mizumachi W, Tsuka H, Hiasa K. Occlusal-supporting ability of individual maxillary and mandibular teeth. J Oral Rehabil. 2012;39:923–30.
Cohen S, Berman LH, Blanco L, Bakland L, Kim JS. A demographic analysis of vertical root fractures. J Endod. 2006;32:1160–3.
Lagouvardos P, Sourai P, Douvitsas G. Coronal fractures in posterior teeth. Oper Dent. 1989;14:28–32.
Zelic K, Vukicevic A, Jovicic G, Aleksandrovic S, Filipovic N, Djuric M. Mechanical weakening of devitalized teeth: three-dimensional finite element analysis and prediction of tooth fracture. Int Endod J. 2015;48:850–63.
Yanık D, Nalbantoğlu AM. Radicular groove of maxillary premolar: is a “danger zone.” Cumhur Dent J. 2022;25:7–12.
Ibrahim AMBR, Richards LC, Berekally TL. Effect of remaining tooth structure on the fracture resistance of endodontically-treated maxillary premolars: an in vitro study. J Prosthet Dent. 2016;115:290–5.
Corsentino G, Pedullà E, Castelli L, Liguori M, Spicciarelli V, Martignoni M, Grandini S. Influence of access cavity preparation and remaining tooth substance on fracture strength of endodontically treated teeth. J Endod. 2018;44:1416–21.
Özyürek T, Ülker Ö, Demiryürek EÖ, Yılmaz F. The effects of endodontic access cavity preparation design on the fracture strength of endodontically treated teeth: traditional versus conservative preparation. J Endod. 2018;44:800–5.
Soares PV, Santos-Filho PCF, Gomide HA, Araujo CA, Martins LRM, Soares CJ. Influence of restorative technique on the biomechanical behavior of endodontically treated maxillary premolars. Part II: strain measurement and stress distribution. J Prosthet Dent. 2008;99:114–22.
Elkholy MMA, Nawar NN, Ha WN, Saber SM, Kim HC. Impact of canal taper and access cavity design on the life span of an endodontically treated mandibular molar: a finite element analysis. J Endod. 2021;47:1472–80.
Saber SM, Hayaty DM, Nawar NN, Kim HC. The effect of access cavity designs and sizes of root canal preparations on the biomechanical behavior of an endodontically treated mandibular first molar: a finite element analysis. J Endod. 2020;46:1675–81.
Nawar NN, Kataia M, Omar N, Kataia EM, Kim HC. Biomechanical behavior and life span of maxillary molar according to the access preparation and pericervical dentin preservation: finite element analysis. J Endod. 2022;48:902–8.
Versiani MA, Cavalcante DM, Belladonna FG, Silva EJNL, Souza EM, De-Deus G. A critical analysis of research methods and experimental models to study dentinal microcracks. Int Endod J. 2022;55:178–226.
Yuan K, Niu C, Xie Q, Jiang W, Gao L, Huang Z, Ma R. Comparative evaluation of the impact of minimally invasive preparation vs. conventional straight-line preparation on tooth biomechanics: a finite element analysis. Eur J Oral Sci. 2016;124:591–6.
Silva EJNL, De-Deus G, Souza EM, Belladonna FG, Cavalcante DM, Simões-Carvalho M, Versiani MA. Present status and future directions – Minimal endodontic access cavities. Int Endod J. 2022;55:531–87.
Kantardžić I, Vasiljević D, Lužanin O, Maravić T, Blažić L. Influence of the restorative procedure factors on stress values in premolar with MOD cavity: a finite element study. Med Bio Engin Comput. 2018;56:1875–86.
Maravić T, Comba A, Mazzitelli C, et al. Finite element and in vitro study on biomechanical behavior of endodontically treated premolars restored with direct or indirect composite restorations. Sci Rep. 2022;12:12671.
Wang Q, Liu Y, Wang Z, Yang T, Liang Y, Gao Z, Zhang Y. Effect of access cavities and canal enlargement on biomechanics of endodontically treated teeth: a finite element analysis. J Endod. 2020;46:1501–7.
Gao SS, An BB, Yahyazadehfar M, Zhang D, Arola DD. Contact fatigue of human enamel: experiments, mechanisms, and modeling. J Mech Behav Biomed Mater. 2016;60:438–50.
Kinney JH, Marshall SJ, Marshall GW. The mechanical properties of human dentin: a critical review and re-evaluation of the dental literature. Crit Rev Oral Biol Med. 2003;14:13–29.
Dorado S, Arias A, Jimenez-Octavio JR. Biomechanical modelling for tooth survival studies: mechanical properties, loads and boundary conditions—a narrative review. Materials. 2022;15:7852.
Lim DY, Kim HC, Hur B, Kim KH, Son K, Park JK. Stress distribution of endodontically treated maxillary second premolars restored with different methods: three-dimensional finite element analysis. J Korean Acad Conserv Dent. 2009;34:69.
Clark D, Khademi J. Modern molar endodontic access and directed dentin conservation. Dent Clin North Am. 2010;54:249–73.
Askerbeyli Örs S, Aksel H, Küçükkaya Eren S, Serper A. Effect of perforation size and furcal lesion on stress distribution in mandibular molars: a finite element analysis. Int Endod J. 2019;52:377–84.
Moore B, Verdelis K, Kishen A, Dao T, Friedman S. Impacts of contracted endodontic cavities on instrumentation efficacy and biomechanical responses in maxillary molars. J Endod. 2016;42:1779–83.
Neelakantan P, Khan K, Hei Ng GP, Yip CY, Zhang CF, Pan Cheung GS. Does the orifice-directed dentin conservation access design debride pulp chamber and mesial root canal systems of mandibular molars similar to a traditional access design? J Endod. 2018;44:274–9.
Kishen A. Mechanisms and risk factors for fracture predilection in endodontically treated teeth. Endod Topics. 2006;13:57–83.
Wan B, Chung BH, Zhang MR, Kim SA, Swain M, Peters OA, et al. The effect of varying occlusal loading conditions on stress distribution in roots of sound and instrumented molar teeth: a finite element analysis. J Endod. 2022;48:893–901.
Sakaguchi RL, Powers JM. Craig’s restorative dental materials. 13th ed. St. Louis: Elsevier Health Sciences; 2012.
Smoljan M, Hussein MO, Guentsch A, Ibrahim M. Influence of progressive versus minimal canal preparations on the fracture resistance of mandibular molars: a 3-dimensional finite element analysis. J Endod. 2021;47:932–8.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdelfattah, R.A., Nawar, N.N., Kataia, E.M. et al. How loss of tooth structure impacts the biomechanical behavior of a single-rooted maxillary premolar: FEA. Odontology 112, 279–286 (2024). https://doi.org/10.1007/s10266-023-00829-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-023-00829-6