Abstract
This review summarizes progress in the construction of efficient yeast ethanol producers from glucose/sucrose and lignocellulose. Saccharomyces cerevisiae is the major industrial producer of first-generation ethanol. The different approaches to increase ethanol yield and productivity from glucose in S. cerevisiae are described. Construction of the producers of second-generation ethanol is described for S. cerevisiae, one of the best natural xylose fermenters, Scheffersomyces stipitis and the most thermotolerant yeast known Ogataea polymorpha. Each of these organisms has some advantages and drawbacks. S. cerevisiae is the primary industrial ethanol producer and is the most ethanol tolerant natural yeast known and, however, cannot metabolize xylose. S. stipitis can effectively ferment both glucose and xylose and, however, has low ethanol tolerance and requires oxygen for growth. O. polymorpha grows and ferments at high temperatures and, however, produces very low amounts of ethanol from xylose. Review describes how the mentioned drawbacks could be overcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Global energy demands and environmental problems have stimulated efforts for biofuels’ production from renewable resources, which will be used to supplement gasoline from fossil fuel (oil) [109]. Due to the current political instability in oil-producing nations, and the concerns about global warming (attributed to harmful greenhouse gas emissions), there has been a call for greater reliance on domestic energy sources for the development of economically viable and environmentally friendly biofuels from cellulosic resources.
Fuel ethanol is a renewable liquid transportation fuel widely used in the United States and Brazil that is expected to become a dominant renewable biofuel in the transport sector within the next 20 years [42, 56, 123]. Most ethanol is currently made from maize (US) or sugar cane (Brazil), but future supplies could be derived from cellulosic and hemicellulosic sugars found in herbaceous biomass and agricultural residues. This can be documented by the worldwide exponential growth in ethanol production during the last decade, reaching 120 billion liters in 2018, 100 billion liters of this amount representing fuel ethanol (https://knect365.com/energy/article/c07f7fba-48fa-464f-9f21-12f913fc67f7/world-ethanol-production-to-expand-steadily-in-2019). Ethanol can be blended with gasoline or used as a neat alcohol in dedicated engines, taking advantage of the higher octane number and higher heat of vaporization. Furthermore, it is considered as an excellent fuel for advanced flexi-fuel hybrid vehicles.
Below, we describe different approaches towards construction of more robust and efficient producers of first- and second-generation ethanol in conventional (Saccharomyces cerevisiae) and non-conventional (Scheffersomyces stipitis, Ogataea polymorpha) yeasts.
Saccharomyces cerevisiae
First-generation ethanol
Today, industrial-scale production of fuel ethanol uses primarily conventional feedstocks such as glucose (derived from starch) and sucrose (from sugarcane or sugarbeet), and is known as first-generation (1G) ethanol.
S. cerevisiae is the organism of choice for industrial ethanol production. The ethanol fermentation is the largest industrial biotechnological application of yeast. S. cerevisiae is characterized by several desirable industrial properties which include fast growth, efficient glucose anaerobic metabolism, high ethanol productivity, high yield, and high tolerance to several environmental stress factors, such as ethanol, low pH, and low oxygen. In addition, yeasts in general are insensitive to bacteriophage infection, which is particularly relevant in large industrial processes that use bacteria as the production microorganism. During alcoholic fermentation, S. cerevisiae produces ethanol with a yield close to the theoretical maximum (0.51 g ethanol per g of consumed glucose) [48]. At industrial scale, ethanol is produced with a yield that is higher than 90% of the theoretical maximum [28]. Taking into account the large worldwide ethanol production, an increase of ethanol yield of even 1% can provide additional estimated profits that approach hundreds of millions of dollars annually.
S. cerevisiae catabolizes glucose via the Embden–Meyerhof–Parnas (EMP) pathway yielding anaerobically 2 mol ATP per mole of consumed glucose. The efficiency of this pathway for anabolic processes is low with a maximal biomass yield of around 7% and an ethanol yield in the range of 90–93% from the glucose consumed [68]. In contrast to S. cerevisiae, the bacterium, Zymomonas mobilis, ferments glucose through the Entner–Doudoroff (ED) pathway. This pathway provides only 1 mol of ATP per mole of glucose, and consequently directs only 3% of glucose used to biomass production, while the remaining 97% is converted to ethanol at almost the possible theoretical value [171]. When ATP is used for growth, cell biomass is formed at the expense of glucose that is not converted to ethanol. In other words, cell biomass is a byproduct that is produced during alcoholic fermentation. Therefore, the lowering of ATP yield during alcoholic fermentation increases ethanol yield with reduced substrate conversion to cell mass. To achieve this goal, several strategies can be applied such as: (i) substituting the EMP pathway in yeast by the ED pathway; (ii) activation of energy-consuming plasma membrane sugars symporters; (iii) generation of futile cycles; and (iv) elevating activity of ATP-degrading enzymes [27]. Another possibility is to decrease production of glycerol, which is another major by-product of ethanol production in yeast [48]. This is somewhat more difficult, because glycerol accumulation results from a redox imbalance generated during cell growth.
Notwithstanding the former patent application of Lancashire et al. [104] which describes the functional integration of the ED pathway to bypass glycolysis in S. cerevisiae with positive impact on ethanol yield, more recent work demonstrates the inability to express in this yeast one of the two unique enzymes to the ED pathway, namely, 6-phosphogluconate dehydratase (PGDH) [16]. Several attempts to improve the availability of iron–sulfur clusters in the yeast cells and to attract the iron–sulfur cluster assembly machinery to PGDH protein did not result in improved enzyme activities [16].
The impact of the disaccharide–proton symport on biomass and ethanol yields was studied in S. cerevisiae by comparing anaerobic growth on maltose, which is transported by a maltose–proton symporter and intracellularly hydrolyzed by maltase, to that of glucose [196]. Biomass and ethanol yields in anaerobic maltose-limited cultures were 25% lower and 8% higher, respectively, than that of glucose-limited cultures. Relocation of sucrose hydrolysis from the extracellular space to the cytosol with additional evolutionary engineering resulted in a strain that had elevated sucrose uptake kinetics with a 30% decrease in the biomass yield and an 11% increase in the ethanol yield relative to the reference strain. The evolved strain showed an increased transcript level for AGT1 gene-encoded sucrose–proton symporter [14]. The improved kinetics of sucrose transport with concomitant ATP reduction could be achieved by targeted overexpression of AGT1 or the heterologous genes encoding sucrose–proton symporters from yeasts that naturally hydrolyze sucrose intracellularly [81]. Replacement of S. cerevisiae facilitated diffusion systems by heterologous proton symporters for the other industrially relevant sugars (e.g., glucose) should drop the biomass accumulation and elevate the ethanol yield [27].
Another approach which could lead to increasing the ethanol yield is based on the use of a futile cycle, i.e., a set of at least two biochemical reactions that run simultaneously in opposite directions and one of which splits ATP, thus resulting in energy dissipation. A classical example of the futile cycle is combined expression of phosphofructokinase and fructose-1,6-bisphosphatase. Phosphofructokinase is a glycolytic enzyme that phosphorylates fructose 6-phosphate to fructose-1,6-bisphosphate with ATP expediture. Fructose-1,6-biphosphatase (FBPase) is one of the specific gluconeogenesis enzymes. It hydrolyzes fructose-1,6-bisphosphate to fructose-6-phosphate with no ATP production. The simultaneous action of both enzymes—phosphofructokinase and fructose-1,6-biphosphatase—leads to a futile cycle resulted in ATP dissipation. Phosphofructokinase is primarily regulated at the level of activity and the differences in the amount of the enzyme in glycolytic and gluconeogenic conditions do not exceed twofold [8, 23]. In contrast, the FBPase activity is tightly regulated by catabolic repression, inactivation through ubiquitination, inhibition by AMP and fructose-2,6-biphosphate [126, 127]. Therefore, intracellular FBPase activity level is maintained at basal level in cells grown in media containing fermentable carbon sources. To overcome the tight regulation of the yeast FBPase, the bacterial FBPase from Escherichia coli that is insensitive to fructose-2,6-biphosphate inhibition was constitutively expressed in yeast [127, 164]. The resulting recombinant strain exhibited a threefold increase in FBPase activity, a 30% reduced intracellular ATP level and up to 9% increase of ethanol production relative to that of parental strain [164].
An alternate futile cycle was generated via the simultaneous activation of two enzymes, pyruvate carboxylase and the gluconeogenic enzyme, phosphoenolpyruvate carboxykinase [164]. Pyruvate carboxylase catalyzes the conversion of pyruvate into oxaloacetate in an ATP-dependent reaction, and phosphoenolpyruvate carboxykinase uses ATP energy to convert oxaloacetate into phosphoenolpyruvate. ATP is synthesized when phosphoenolpyruvate is converted into pyruvate-by-pyruvate kinase. Therefore, the resulting total loss in ATP is one molecule for each one turn of the cycle. The specific activity of pyruvate carboxylase was increased 3–5-fold via substitution of target gene promoter with a strong constitutive one. The activity of phosphoenolpyruvate carboxykinase is regulated at the post-translational level [198]. To avoid such regulation, a heterologous gene coding for the corresponding enzyme from E. coli was overexpressed. As a result, the specific activity of phosphoenolpyruvate carboxykinase was elevated 6–7 fold. Ethanol production by the constructed recombinant strains revealed a twofold increase over the parental strain by the end of the first day of fermentation [164]. Potential futile cycle based on the interconversion between glucose and trehalose, which theoretically could lead to ATP dissipation in cells of S. cerevisiae was generated by the activation of trehalose-6-phosphate synthase and the neutral trehalase responsible for the synthesis and degradation of trehalose [164]. Despite the increase in the activities of both enzymes, biomass accumulation remained unchanged.
Lowering the intracellular ATP content in yeast cell with concomitant increase in ethanol yield can also be achieved by the activation of some of the cytosolic ATPases. A decrease in cellular ATP pool and activation of alcoholic fermentation was achieved by the overexpression of the soluble part (F1) of H+-ATPase or a portion of F1 exhibiting ATPase from different origins in S. cerevisiae [75]. Similar results were obtained after the overexpression of PHO5 coding for acid phosphatase which is a non-specific enzyme that also hydrolyzes ATP [158]. Overexpression of the vacuolar alkaline phosphatase Pho8 led to increase in yield and ethanol production from glucose in both a laboratory and an industrial strains of S. cerevisiae (Table 1), whereas the expression of the truncated cytosol localized form is detrimental to cell growth [163]. The galactose induced expression of ATP hydrolysis region of the ribosome-associated molecular chaperon encoded by gene SSB1 of S. cerevisiae and the ATP-diphosphohydrolases also known as apyrases from E. coli, resulted in an increase in ethanol yield of 39 and 29%, respectively, during fermentation of the corresponding recombinant S. cerevisiae strains in galactose containing media [164]. Thus, creation of functional futile cycles or overexpression of ATPases could be the promising approaches for enhancing the ethanol yield in yeast.
Glycerol is the second primary byproduct after cell biomass during the ethanol production. Glycerol is formed at the expense of sugar that is not converted to ethanol [48]. In yeast, the reduction of glycerol formation can most probably result in an increase in ethanol yield. Therefore, significant research efforts have been directed towards reducing glycerol formation during fermentation. This can be accomplished by deleting one or both genes GPD1 and GPD2, coding for glycerol-3-phosphate dehydrogenase [7]. Deletion of both genes affected anaerobic growth. Deletion of GPD2 resulted in an increase in ethanol yield with concomitant decrease in glycerol production; however, this deletion also reduced growth and ethanol productivity [180]. Glycerol formation results from the regeneration of NAD from excess NADH produced during glycolysis under anaerobic conditions. To decrease cytosolic NADH formation, the gene GDH1 encoding NADPH-dependent glutamate dehydrogenase was deleted, while GLN1 and GLT1 coding for glutamine synthetase and NAD-dependent glutamate synthase were overexpressed. During ammonium assimilation that is linked with NADH and ATP consumption, a recombinant strain decreased the glycerol yield by 38% while increasing the ethanol yield by 10% [134]. Another approach for reducing the intracellular pool of NADH and ATP production consisted of replacing the glyceraldehyde-3-phosphate dehydrogenase gene by that of the non-phosphorylating heterologous NADP-dependent analog from Bacillus cereus, or Streptococcus mutans [54, 208]. The combination of this approach with the overexpression of NAD-dependent fumarate reductase or acetaldehyde dehydrogenase increased ethanol yield to 95% of the theoretical maximum [208].
Other work demonstrated that the expression of the NAD-dependent acetaldehyde dehydrogenase from E. coli in an S. cerevisiae gpd1Δ gpd2Δ strain supports the growth in medium with glucose supplemented with acetate under anaerobic conditions [51]. The constructed strains could be used for alcoholic fermentation of acetate containing substrates, as they are able to convert acetate into ethanol. The main drawback of these S. serevisiae strains is that the reduced glycerol production resulted also in reduced osmotolerance and overall viability [63]. This finding imposes certain restrictions on the application of these strains in industrial fermentation processes that normally run at high substrate concentration. Other significant efforts have focused on improving the stress resistance of yeast strains with reduced glycerol production. Osmotolerance, thermotolerance, and tolerance to high concentrations of ethanol of several target strains were elevated by applying methods of metabolic engineering and genomic shuffling [64, 189]. The combination of the deletion of the GPD1 gene with the overexpression of the nadF gene from B. cereus that encodes for an NADP-dependent glyceraldehyde-3-phosphate dehydrogenase with the derepression of homologous genes of trehalose synthesis TPS1 and TPS2 (encoding trehalose-6-phosphate synthase and trehalose-6-phosphate phosphatase), resulted in increased ethanol production, and reduced glycerol formation, but did not exhibit a negative influence on strains viability during alcoholic fermentation [54].
Trehalose protects cells from stress and its intracellular concentration correlates with resistance to high temperatures and high concentrations of ethanol in the medium [176]. Overexpression of the TPS1 gene in S. serevisiae resulted in increased thermotolerance, thereby allowing for the possible reduction in energy costs for cooling of fermentation vessels as well as for savings in energy used for heating due to reduced temperature difference between fermentation and distillation processes [6]. Moreover, high-temperature fermentation has advantages during the process of simultaneous saccharification and fermentation, as high temperature (around 50 °C) is beneficial to hydrolytic enzymes involved in saccharification [165].
The development of improved ethanol-producing strains can be achieved by applying traditional selection and adaptive evolution as a useful alternative to metabolic engineering approaches. Mutants resistant to toxic concentrations of oxythiamine, trehalose, 3-bromopyruvate, glyoxylic acid, and glucosamine have been isolated. Some of these are characterized by 5–8% increase in ethanol yield when compared to the parental industrial ethanol-producing strain (Table 2) [29]. By applying adaptive evolution, useful yeast strains with enhanced maltose utilization and osmotolerance [62] increased ethanol tolerance [174] and yeast with improved ethanol production rate and decreased formation of acetate were selected [21].
Second-generation ethanol
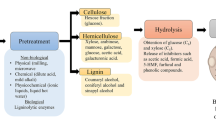
In contrast to 1G, the 2G ethanol is ethanol produced from non-food feedstocks such as dried plant biomass (lignocellulose). The utilization of lignocellulosic biomass for 2G ethanol production would be preferable over sugar and starch-based 1G ethanol production because of significantly lower competition with food and animal feed production and minimal changes to land use [131, 161]. 2G ethanol can utilize a range of different types of lignocellulosic substrates. Currently, a limited amount of 2G ethanol is produced at several pilot and demonstration plants around the world; however, due to higher cost of the large-scale ethanol production from lignocellulosics, it is not yet commercially feasible [40, 109, 135].
The technologies to produce 2G ethanol do exist; however, many improvements are needed. The production of 2G ethanol from lignocellulosics will require the development of robust microbial strains that can grow and produce ethanol from at least glucose and xylose, which are the major fermentable sugars produced from the hydrolysis of lignocellulosic biomass. Other sugars that are produced during hydrolysis include hexoses fructose, mannose, galactose, and the pentose sugar l-arabinose.
The yeast S. cerevisiae efficiently ferments hexoses glucose, fructose, and mannose but to a lesser extent galactose. In this yeast, galactose transporters are subjected to catabolic repression by glucose, thereby limiting co-fermentation of these sugars. In contrast, mannose enters yeast cells using the glucose transport system allowing glucose/mannose co-fermentation to ethanol [118]. The combined deletion of the genes GAL6, GAL80, and MIG1 involved in negative regulation of galactose catabolism in a laboratory S. cerevisiae strain resulted in a partial co-consumption of glucose and galactose in aerobic batch cultures [136]. Normally, galactose metabolism in S. cerevisiae requires respiration. However, a cox9Δ gal80Δ double mutant has been isolated which effectively fermented galactose anaerobically [150]. A natural strain of S. cerevisiae (NRRL Y-1528), which catabolizes galactose more effectively than glucose or mannose and is capable of simultaneously fermenting all hexoses present in hydrolyzed biomass was described. This strain has a proposed deficiency in carbon catabolic repression, which avoids glucose repression of galactose utilization [84].
Wild-type strains of S. cerevisiae are unable to catabolize and ferment pentoses (d-xylose or l-arabinose, referred to further as xylose and arabinose, respectively) which are major constituents of hydrolysates of plant biomass. This is consequence of the absence of the enzymes that catalyze the initial stages of pentose catabolism. Since the xylose content of plant biomass hydrolysates is significantly higher than that of arabinose, most work has been directed to the construction of S. cerevisiae strains capable of xylose catabolism and fermentation. These efforts have been focused on the functional expression of the heterologous genes for xylose catabolism of prokaryotic and eukaryotic origin. Some efforts have been focused on the expression of the genes encoding xylose isomerase (XI) from different microorganisms. This enzyme does not require cofactors and catalyzes the isomerisation of xylose in xylulose (Fig. 1). Several successful attempts have been made to express XI from the bacteria Thermus thermophilus [188], Clostridium phytofermentans [20], Bacteroides stercoris [55], or from the anaerobic fungus Piromyces sp. E2 [82] or from Orpinomyces sp. [117]. Expression of the codon-optimized XI of C. phytofermentans in S. cerevisiae resulted in a 46% increase in specific growth rate on xylose as compared to the strain expressing a non-optimized version of the gene [20]. XI from the bacterium Propionibacterium acidipropionici became functionally active in S. cerevisiae when co-expressed with GroEL–GroES chaperonin complex from E. coli [178]. More details on functional expression if XI in S. cerevisiae described in a recent reviews [72, 103].
Pathways of xylose, l-arabinose and glucose fermentation to ethanol in yeasts. Gl-6-p is glucose 6-phosphate; Fr-6-p is fructose 6-phosphate; GA-3-p is glyceraldehyde 3-phosphate; SH-7-p is sedoheptulose 7-phosphate; ET-4-p is erythrose 4-phosphate; 1,3-bpG is 1,3-bisphospho-glycerate; DHA-p is dihydroxyacetone phosphate; XR is xylose reductase; XDH is xylitol dehydrogenase; XI is xylose isomerase; XK is xylulokinase; AI is arabinose isomerases; RK is ribulokinase; RPE4 is ribulose-5-phosphate-4-epimerase; AR is aldose reductase; ArDH is arabinitol dehydrogenase; XYR is xylulose reductase; RKI is ribose-5-phosphate ketol-isomerase; RPE is ribulose-5-phosphate-3-epimerase; TKL is transketolase; TAL is translaldolase; PRK is phosphoribulokinase; RCO is ribulose-1,5-bisphosphate carboxylase/oxygenase; G3PDH is glyceraldehyde 3-phosphate dehydrogenase; PDC is pyruvate decarboxylase; ADH is alcohol dehydrogenase
Many studies have been focused on the expression of heterologous yeast genes encoding xylose reductase (XR) and xylitol dehydrogenase (XDH), which catalyze the conversion of xylose into xylitol and subsequently to xylulose (Fig. 1). XR is encoded by XYL1 which can use either NADH or NADPH as cofactors but which generally shows higher affinity to NADPH. XDH is an NAD- and Zn-dependent secondary alcohol dehydrogenase that is encoded by XYL2. The co-expression of both genes resulted in an imbalance of cofactors with a deficiency in NAD and an excess of NADP. It is assumed that this imbalance causes the low efficiency of xylose alcoholic fermentation, primarily due to the xylitol accumulation [77, 94]. A genome-wide scale model of S. cerevisiae was used to predict the maximal achievable growth rate for cofactor balanced xylose utilization pathway. By applying the use of dynamic modeling with experimental results, a balanced cofactor system XR/XDH showed a 24.7% increase in ethanol batch production and simultaneously reduced the predicted substrate utilization time by up to 70% [47]. The combination of computational design [85] and site-specific mutagenesis of domains responsible for binding these cofactors [192], resulted in a modification of the cofactor specificity of XR and XDH and for balanced action. The co-expression of the mutant forms of XR from S. stipitis (K270 M, K270R, K270R/N272D, N272D/P275Q, R276H) with elevated affinity to NADH along with a native NAD-dependent XDH increased productivity and ethanol yield with concomitant reduction of xylitol yield in recombinant strains of S. cerevisiae [15, 108]. A decrease in xylitol and an increase in ethanol yields were also reported as a result of co-expression of the native XR with the modified XDH (D207A/I208R/F209S/N211R) which exhibits affinity to NADP. Xylose consumption was also 32% faster when compared to the strain expressing wild-type alleles of XR and XDH [120]. The ratio between XR and XDH activities is essential to improve xylose alcoholic fermentation. The low level of XYL2 gene expression is the main reason for the accumulation of xylitol in strains expressing XYL1, XYL2, and XYL3 [90].
Comparison of the industrial S. cerevisiae with XI and/or XR/XDH pathways regarding xylose fermentation efficiency showed that XI-expressing strains are advantageous regarding XR/XDH expressing strains; however, the most efficient xylose fermentation was found in the recombinant strain which simultaneously expresses both XI and XR/XDH pathways [25]. Co-expression of both pathways significantly improved ethanol production from non-detoxified hemicellulosic hydrolysates.
Xylulokinase (XK) catalyzes the ATP-dependent phosphorylation of xylulose and is an important enzyme of xylose catabolism (Fig. 1). XK catalyzes the limiting step in xylose alcoholic fermentation as S. serevisiae strains expressing XR/XDH or XI have a significant reduction in xylitol accumulation when XK was overexpressed [139]. The activation of XK was accomplished either by the derepression of the homologous S. cerevisiae gene XKS1 or by heterologous expression of the XYL3 gene from S. stipitis [79]. However, high levels of XK activity have a negative impact on the growth of recombinant strains on xylose, which is probably due to a rapid depletion of ATP caused by xylulose phosphorylation [35].
To further increase ethanol production, xylose-fermenting strains of S. serevisiae expressing the genes for xylose catabolism were subjected to other metabolic modifications. In particular, the heterologous expression of genes for the sugar transporters Sut1 and Sut2 from S. stipitis was carried out [35, 61]. A system for the selection of glucose transporters with high affinity for xylose has been also developed. As a result, a modified version of the Gal2 S. cerevisiae transporter that is able to transport only xylose was constructed [41]. It was also shown that endogenous S. cerevisiae hexose transporters (Hxt) can be engineered into specific xylose transporters [41, 132]. However, xylose transporters synthesized in these strains remain subjected to protein degradation. For example, some Hxt proteins with high xylose transport capacity that are rapidly degraded in the absence of glucose or when glucose is exhausted from the medium [133]. The N-terminal lysine residues of the Hxt proteins were predicted to be the target of ubiquitination. The substitution of N-terminal lysine residues in the hexose transporters Hxt1 and Hxt36, which are subjected to catabolite degradation, resulted in improved retention of these transporters at the cytoplasmic membrane in the absence of glucose and improved xylose fermentation upon the depletion of glucose when cells were grown in xylose alone [133]. An interesting approach proposed to create an artificial hybrid protein consisting of a sugar transporter and xylose isomerase. Such hybrid protein was successfully expressed in S. cerevisiae and the resulting strain showed acceleration of xylose consumption and substantially diminished production of xylitol as an undesired side product, with a concomitant increase in the production of ethanol [179].
To achieve efficient glucose/xylose co-fermentation, a modified xylose-specific, glucose-insensitive transporter Mgt05196 (N360F) from Pichia (Meyerozyma) guilliermondii, was expressed in the background of an xylose-fermenting S. cerevisiae strain overproducing xylose isomerase XI, xylulokinase Xks1 and the enzymes of non-oxidative pentose phosphate pathway (PPP), and in which the aldose reductase Gre3p and the alkaline phosphatase Pho13 had been inactivated. These rationally designed genetic modifications, combined with alternating adaptive evolution in xylose and lignocellulosic hydrolysates, resulted in a final strain, with excellent xylose fermentation that had an enhanced resistance to inhibitors [110]. Glucose/xylose co-fermentation was activated after HXK2 deletion and introduction of a GAL83 G673T allele which provided 2.5-fold higher xylose and glucose co-consumption ratio than its xylose-fermenting parental strain [138].
The co-expression of genes of non-oxidative part of PPP (RKI1, RPE1, TKL1, and TAL1, encoding ribose-5-phosphate isomerase, ribulose-5-phosphate epimerase, transketolase, and transaldolase, respectively) (Fig. 1) on the background of a S. cerevisiae strain expressing XI and XK, resulted in improved growth on xylose [43, 83]. In a recombinant strain expressing XI, xylose consumption and fermentation were also enhanced after homologous or heterologous (from thermotolerant yeast Kluyveromyces marxianus) overexpression of RKI1, TAL1 and TKL1 genes [93]. However, expression of genes of the non-oxidative part of PPP in an S. cerevisiae strain expressing XR, XDH, and XK did not improve its growth on a medium supplemented with xylose [17].
NADPH synthesis mainly occurs in the oxidative part of PPP. A series of studies were conducted to reduce the activity of the enzymes participating in the oxidative branch of PPP. This approach was hypothesized to facilitate the reaction catalyzed by XR using NADH instead of NADPH, with a reduced production of CO2. The deletion of genes ZWF1 (encoding glucose-6-phosphate dehydrogenase) and/or GND1 (decarboxylating 6-phosphogluconate dehydrogenase) with the simultaneous expression of the genes XYL1 and XYL2, increased ethanol yield, but also significantly decrease xylose consumption rate during xylose fermentation [76]. Another successful approach describes the combination of fungal NADP-dependent glyceraldehyde-3-phosphate dehydrogenase overexpression in part with the deletion of the ZWF1 gene, resulting in an increase in ethanol yield and productivity during xylose fermentation [184]. To reduce xylitol production and increase ethanol yield during fermentation of glucose/xylose mixture, a modification of the redox balance of recombinant strains of S. cerevisiae was carried out by the deletion of gene GDH1 (coding for NADPH-dependent glutamate dehydrogenase) and the overexpression of GDH2 gene (NADH-dependent glutamate dehydrogenase) responsible for ammonium assimilation [156]. Xylitol accumulation was reduced by overexpressing the noxE gene from L. lactis, which codes for the water forming NADH-oxidase [206]. Modification of the acetate biosynthetic pathway involving deletion of the ALD6 gene encoding NADP-dependent aldehyde dehydrogenase also increased the efficiency of xylose alcohol fermentation [108].
To increase the production of ethanol during xylose fermentation, recombinant strains of S. cerevisiae were subjected to adaptive evolution and genome shuffling [80, 90, 152]. Sequencing of the genomes of these strains after long-term culturing in xylose containing media revealed new potential target genes for metabolic engineering approach, e.g., XKS1, SOL3 (6-phosphogluconolactonase), GND1 (6-phosphogluconate dehydrogenase), TAL1, TKL1, YCR020C, YBR083 W, and YPR199C [187]. The genes for TAL1 and PHO13 (non-specific alkaline phosphatase) were identified by transposon mutagenesis as new targets. The truncation or deletion of PHO13 particularly increased production of ethanol during xylose fermentation [13, 90, 91, 103, 110, 129].
Carbon dioxide is a major by-product of carbohydrate alcoholic fermentation. An attractive target for manipulation is conserving carbon that is released from the decarboxylation step of pyruvate into acetaldehyde in the last stage of the ethanol-producing pathway. This goal was achieved by the introduction a synthetic reductive PPP into a xylose-fermenting S. cerevisiae strain, which resulted in simultaneous lignocellulosic ethanol production and carbon dioxide recycling. The heterologous enzymes phosphoribulokinase from Spinacia oleracea and ribulose-1,5-bisphosphate carboxylase/oxygenase from Rhodospirillum rubrum, were introduced into the S. cerevisiae strain harboring XR/XDH pathway, up-regulated PPP and knocked out ALD6 and PHO13 genes [90]. The phosphoribulokinase enzyme catalyzes ribulose-5-phosphate conversion to ribulose-1,5-bisphosphate, and the ribulose-1,5-bisphosphate carboxylase/oxygenase converts one ribulose-1,5-bisphosphate and CO2 to two molecules of glycerate-3-phosphate (Fig. 1). The constructed strain with the synthetic reductive PPP revealed a higher yield of ethanol with lower yields of xylitol and glycerol than that of the parental strain. Moreover, a reduced release of CO2 by the engineered strain was observed during xylose fermentation, suggesting that the carbon dioxide generated by pyruvate decarboxylase was partially reassimilated through the synthetic reductive PPP [200]. The strategy of carbon dioxide recycling from the ethanol fermentation pathway in yeast has a great potential in alleviating greenhouse gas emissions during the production of 2G ethanol.
Alcoholic production from l-arabinose appears to be very important, as this pentose is present in hemicellulose and to even higher proportion in pectin [203]. Two different l-arabinose catabolizing pathways were identified in bacteria [199] and fungi [153, 154]. In the bacterial pathway, arabinose is isomerized to ribulose by arabinose isomerase (araA), and then, ribulose is phosphorylated to ribulose-5-phosphate with ribulokinase (araB) which in turn is converted to xylulose-5-phosphate by ribulose-5-phosphate-4-epimerase (araD) [199]. Arabinose fermentation was observed when araA, araB, and araD from Lactobacillus plantarum were co-expressed in S. cerevisiae [199, 203]. In fungi, l-arabinose is reduced to l-arabitol by aldose reductase and then is converted to l-xylulose with arabinitol dehydrogenase. l-xylulose is reduced to xylitol by xylulose reductase, which in turn is oxidized to xylulose by xylitol dehydrogenase (Fig. 1). The expression of the fungal arabinose utilization pathway (aldose reductase Gre3 from S. cerevisiae, arabinitol dehydrogenase, and xylulose reductase from Trichoderma reesi, XDH from S. stipitis and XK from S. cerevisiae) in S. cerevisiae resulted in low ethanol with substantial arabinitol production, which is apparently due to the imbalance caused by the cofactor mismatch among used reductases/dehydrogenases [18, 153, 154, 203]. Based on published results, the expression of genes of the bacterial redox-independent pathway for l-arabinose utilization in S. cerevisiae is a more promising approach [205].
Fermentation of biomass-derived hydrolysates is accompanied by the inhibition of ethanol production due to the presence of toxic compounds produced mainly during lignocellulose pretreatment, e.g., furfural, hydroxymethylfurfural (HMF), weak acids, and phenols [137]. To overcome this limitation, strains of S. cerevisiae that are able to produce ethanol in the presence of these inhibitors have been selected through directed evolution and adaptation [60, 140]. Other alternative molecular approaches were also applied. Furfural and HMF were converted to less toxic furfuryl alcohol and furan dimethanol, respectively, by the overexpression of the endogenous S. cerevisiae oxidoreductases such as alcohol dehydrogenases (ADH1, ADH6, and ADH7) [3, 113, 146] and aldo–keto reductases (GRE2) [124]. It was shown [49] that tolerance to furfural-induced stress is associated with genes of the pentose phosphate pathway ZWF1, GND1, RPE1, and TKL1. The overexpression of the gene PAD1 encoding phenylacrylic-acid decarboxylase in S. cerevisiae resulted in improved growth rate and ethanol productivity in dilute-acid hydrolysates [106]. An original approach to improve tolerance to fermentation inhibitors and ethanol was proposed by modulation of the polyamine (spermidine) content in S. cerevisiae [89]. Intracellular spermidine contents were increased by a double gene deletion OAZ1 (ornithine decarboxylase antizyme) and TPO1 (polyamine transport protein) genes and the overexpression of SPE3 (spermidine synthase) [89].
In summary, 1G ethanol production is a profitable technology that can be further improved by increasing ethanol yield and productivity by applying molecular and classical techniques to industrial ethanol-producing strains of S. cerevisiae. Despite significant efforts, existing technologies of 2G ethanol production are still non-profitable. Although recombinant S. cerevisiae strains capable of hexoses/pentoses co-fermentation were developed, none of the engineered strains are able to ferment glucose/xylose mixture as fast as the rate of glucose fermentation by the parental strain [205]. Another important limitation is the sensitivity of ethanol-producing strains to inhibitors in lignocellulosic hydrolysates. Further studies are needed to develop recombinant S. cerevisiae strains that are capable of rapid fermentation of mixed sugars with improved resistance to fermentation inhibitors.
Scheffersomyces (Pichia) stipitis
Scheffersomyces stipitis (formerly known as Pichia stipitis) belongs to a group of yeasts that naturally ferment xylose while accumulating low amounts of by-products such as xylitol [97]. Other known representatives of the group of native xylose-fermenting yeastsare, Candida shehatae (teleomorph form is known as Scheffersomyces shehatae), Pachysolen tannophilus, Spathaspora passalidarum, and Ogataea polymorpha [38, 128, 160, 162]. As a rule, most natural xylose-fermenting yeasts inhabit the guts of passalid beetles that degrade white-rotted hardwood [125, 130] with the exception of O. polymorpha strains that have been isolated from other natural habitats [46]. S. stipitis is closely related to other yeast endosymbionts of passalid beetles [175]. This yeast has one of the highest native capacities for xylose fermentation among yeast species described so far [39]. In addition to utilize xylose, S. stipitis has the capability to use all of the other major sugars found in wood. It also transforms low-molecular weight lignin moieties, reduces acyclic enones to the corresponding alcohols, and forms various esters and aroma components. It has been recently engineered to produce lactic acid or xylitol in high yield [24, 67, 87, 107, 177]. The published ethanol yield from fermented lignocellulosic sugars by adapted S. stipitis strain approached 80% of theoretical yield [130]. The availability of genetic tools and capability for fermentation of hydrolysates have made S. stipitis an attractive microorganism for bioconversion of lignocellulose to fuels and chemicals. The major drawbacks of S. stipitis are the low fermentation rates, low ethanol tolerance, and the inability to grow anaerobically [39, 50, 167].
S. stipitis is a predominantly haploid, homothallic, hemiascomycetous yeast that forms buds along with pseudomycelia during vegetative growth and two hat-shaped ascospores from each ascus [96]. Genetic manipulation of S. stipitis is much more complicated relative to those of S. cerevisiae, because S. stipitis is resistant to most common antibiotics and the number of strains with convenient auxotrophic markers is limited. This yeast uses an alternative codon system that decodes CUG for serine instead of leucine as common in the classical genetic code [155]. Thus, the expression of foreign proteins, including those used as drug resistance markers, requires codon modification. Unfortunately, random (non-homologous) integration prevails in S. stipitis which makes targeted deletions much more difficult to obtain [74]. Nevertheless, many efficient genetic methods have been developed for S. stipitis that include methods of sexual mating and sporulation [121]. Auxotrophic strains have been created and methods for high-efficiency transformation have been developed for the auxotrophic mutants ura3, leu2, trp5, and his3 of S. stipitis [115, 148, 201]. Genetic tools based on a loxP/Cre recombination system and the dominant marker for zeocine resistance has been developed for S. stipitis [105]. Deletion of the KU80 gene that is responsible for non-homologous end joining significantly increases the fraction of homologous recombinant transformants, albeit at the expense of transformation frequency [116].
The 15.4-Mbp genome of S. stipitis was sequenced [73, 155]. S. stipitis CBS 6054 is known to have eight chromosomes, of which two pairs are very similar in size, accounting for the earlier results that suggested the presence only of six chromosomes [142]. S. stipitis genome annotation predicted that 5841 genes are present in this of which a majority of 72%, have a single exon. The average gene density is 56% and the average gene, transcript, and protein lengths of 1.6 kb, 1.5 kb and 493 amino acids, respectively. Expressed sequence tags (ESTs) confirmed the identity of 40% of the predicted genes with 84% showing strong similarity to proteins in other fungi [73, 155]. So far, protein function can be tentatively assigned to about 70% of the genes. Synteny analysis of S. stipitis with its nearest completely sequenced yeast genome neighbor, Debaryomyces hansenii, shows extensive recombination and shuffling of the chromosomes, which appear to be a common feature. S. stipitis and D. hansenii share 151 gene families that are not found in the other genomes. The S. stipitis gene set is missing 81 gene families (442 proteins) relative to the other yeast genomes in the analysis.
The most frequent domains characterized so far include protein kinases, helicases, transporters (sugar and MFS), and domains involved in transcriptional regulation (fungal-specific transcription factors, RNA recognition motifs, and WD40 domains). A majority of these are shared with other hemiascomycota. These range from 1534 domains in common with Schizosaccharomyces pombe and 1639 domains with D. hansenii. One of the few S. stipitis-specific domains belong to one of the glycosyl hydrolase families, a subgroup of cellulases and xylanases. All of the genes for xylose assimilation, including the oxidative PPP, glycolytic cycle, the tricarboxylic acid cycle (TCA), and ethanol production, were present in isoforms similar to those found in other yeasts [73, 155]. Genes of the first three enzymes of xylose metabolism, XYL1, XYL2, and XYL3 and that of PPP (ZWF1, GND1, TKL1, TAL1, and RPI1), are also induced by xylose and the expression of XYL2 yields one of the most abundant transcripts in xylose-grown cells [74]. Another interesting pattern of the regulation of gene expression was found through oxygen limitation. Such limitation led to strong derepression of some glycolytic genes that include two genes of glyceraldehyde-3-phosphate dehydrogenase TDH1 and TDH2, pyruvate decarboxylase PDC1 and PDC2, and of the secondary alcohol dehydrogenase SAD2 (the function of the last gene/enzyme in metabolism is not known) [74].
S. stipitis has genes for sensing and regulatory proteins (< 200 putative Zn-finger regulatory proteins) which in many cases code for proteins similar to those in S. cerevisiae.
The genome S. stipitis also revealed many gene clusters representing either pairs/clusters of non-homologous genes in which each cluster has a single function such as galactose metabolism, or tandem repeats of paralogous genes. Gene clusters seem to be particularly abundant in S. stipitis, as there are at least 35 clusters of functionally related genes [155]. The studies of structure, function, and regulation in S. stipitis genome has an important impact on understanding its physiology and could be used for metabolic engineering of this organism.
The study of S. stipitis has attracted scientists and technologists primarily due to its natural ability to produce large amounts of ethanol during xylose fermentation with small or no production of xylitol. It was found that fed-batch cultures of S. stipitis produce around 47 g/L of ethanol with yields of 0.36 g/g xylose at 30 °C [182]. However, S. stipitis fermentation rate on xylose is low relative to that of S. cerevisiae on glucose, so increasing the rate of fermentation of xylose by S. stipitis could greatly improve its usefulness in commercial applications [74]. Another drawback to using S. stipitis is that it is much more susceptible to ethanol inhibition relative to S. cerevisiae [167].
There are many detailed publications on the physiology of S. stipitis. Oxygen plays an important role in cell growth, redox balance, functioning of the mitochondria and generation of energy for xylose transport in S. stipitis [169]. Fermentation in S. stipitis is activated by oxygen limitation [92, 141, 144]. It is interesting to note that S. stipitis can metabolize xylose anaerobically, even though it does not grow under anaerobic conditions [197]. The optimal temperature for S. stipitis fermentation is between 25 and 33 °C and the optimal pH is in the range of 4.5–5.5 [36]. The nutrients in the fermentation media play an important part in the growth and ethanol production in S. stipitis. Ethanol production increased with the addition of amino acids and nitrogen was required for non-growth associated ethanol production [170]. Ammonium salts increased the ethanol productivity and the ethanol to biomass yield in S. stipitis [2, 52]. Magnesium has also been shown to play an important role in redox balance and, therefore, has an effect on xylitol production [119]. Low levels of Mg2+ resulted in xylitol accumulation and a high intracellular NADH content. Corn steep liquor is a viable nutrient source for S. stipitis fermentation when used as a sole nitrogen source compared to amino acids, vitamins, and other nutrients [5]. The initial xylose concentration has an effect on the fermentation parameters of S. stipitis with maximum ethanol productivities occurring at a xylose concentration of 50 g/L [37].
The conversion of xylose to ethanol in S. stipitis consists of three stages: (i) xylose transport and the initial reaction to enter the PPP; (ii) non-oxidative reactions of PPP; and (iii) glycolysis (Fig. 1). Little is known about xylose transport in S. stipitis. The low-affinity transport system is shared between glucose and xylose for sugar transport. Glucose inhibits xylose transport by noncompetitive inhibition in the low-affinity proton symport system [86]. The low-affinity transport is used when sugar concentrations are high and the high-affinity systems are used when sugar concentrations are low. Repression of xylose uptake occurs in fermentation media containing glucose and xylose. Therefore, glucose is the preferred sugar by S. stipitis in ethanol production. The rate of glucose consumption is higher than xylose under similar growth conditions [1]. The transport of sugars into the cells is the rate-limiting step in the utilization of sugars for ethanol production in S. stipitis [111]. A high-affinity xylose-transporting system has been described that is specific for xylose in this yeast [57]. Three genes, SUT1, SUT2, and SUT3, have been characterized that encode glucose transporters in S. stipitis [195]. Sut2 and Sut3 are highly similar to the S. cerevisiae glucose transporter family and the Sut2 and Sut3 transporters have a higher affinity for glucose than for xylose. Transcription of SUT1 is induced in S. stipitis independently of oxygen supply, whereas SUT2 and SUT3 are expressed only under aerobic conditions, but independently of the carbon source. Disruption of SUT1 eliminates the low-affinity xylose transport system in S. stipitis [195].
Initial metabolism of xylose in S. stipitis is similar to other natural xylose-fermenting yeasts (Fig. 1). Xylose first is reduced by xylose reductase (aldose reductase, gene XYL1) to xylitol [183]. This enzyme has affinity to both NADH and NADPH, but shows much higher affinity towards NADPH. By comparison, S. stipitis xylitol dehydrogenase (gene XYL2), which converts xylitol to xylulose, has affinity only for NAD [122]. The third reaction of xylose metabolism is catalyzed by xylulokinase (gene XYL3), which converts xylulose to the PPP intermediate xylulose-5-phosphate [78]. The cofactor imbalance, resulting from the first two reactions involving xylose reductase and xylitol dehydrogenase, leads to xylitol accumulation in most natural xylose-fermenting yeasts, but not in S. stipitis [169]. In S. stipitis, there are efficient systems for NADH reoxidation to NAD and NADPH regeneration [12]. One particular pathway that has been observed for xylose metabolism that is induced under oxygen-limited conditions is effective in tackling the cofactor imbalance caused by the first two steps in xylose utilization [73, 74]. This pathway involves the four enzymes; NAD-dependent glutamate dehydrogenase (GDH2), which converts 2-oxoglutarate to l-glutamate consuming NADH; glutamate decarboxylase (GAD2), which decarboxylates l-glutamate to 4-aminobutyrate; 4-aminobutyrate aminotransferase (UGA1), which transaminates 4-aminobutyrate to succinate semialdehyde; and succinate semialdehyde dehydrogenase (UGA2), which oxidizes succinate semialdehyde to succinate using NADP. The net result of the four reactions is the conversion of NADH to NADPH.
The PPP plays an important role in xylose fermentation (Fig. 1) and the corresponding genes (ZWF1, GND1, TKL1, TAL1, RPI1) are known to be induced by xylose [74]. Alcohol dehydrogenase is also an important enzyme in ethanol production with the deletion of either ADH1 or ADH2 gene significantly reducing ethanol formation, and the deletion of both entirely abolishes ethanol production [22, 143]. Relatively little is known about the rate-limiting steps in ethanol production from xylose. It has been shown that XR and XDH are repressed by glucose and are induced during growth on xylose. Xylose is generally not consumed in the presence of glucose; hence, under glucose repression, these activities, along with xylose transport, are rate limiting [19]. The XK does not, however, appear to be rate limiting in S. stipitis once its activity is induced by xylose [78].
In addition to the ultimate goal of using S. stipitis for ethanol production from lignocellulosic feedstock, this yeast can also be successfully used for xylitol production from xylose using a mutant defective in XYL2 that codes for xylitol dehydrogenase [87, 157]. Efficient producers of lactic acid have also been constructed on S. stipitis after expression of the lactate dehydrogenase LDH gene from Lactobacillus helveticus under control of the yeast ADH1 promoter. It is interesting to note that xylose was more efficient substrate for lactate synthesis than glucose [67]. A strain of S. stipitis hyperaccumulating S-adenosylmethionine has been isolated [95]. Other S. stipitis recombinant strains have been constructed that produce fumaric acid from xylose after acquiring a heterologous reductive pathway from Rhizopus oryzae [194].
In spite of the fact that S. stipitis is one of the best natural xylosefermenting yeasts with no xylitol accumulation, it have several drawbacks which include a low rate of fermentation, low tolerance to ethanol and requirement of oxygen for growth. When grown on lignocellulosic hydrolysates, other limitations to the use of this yeast are observed. Among these, there is lack of simultaneous fermentation of glucose and xylose and the poor tolerance to inhibitors that are present in hydrolysates. In addition, as oxygen is required, and S. stipitis has a tendency to utilize the ethanol produced when there is still a considerable amount of xylose remaining in the medium [58]. Comparison of the advanced recombinant S. cerevisiae strain with the wild-type S. stipitis strain regarding xylose fermentation showed that S. cerevisiae showed a higher maximum ethanol titer and xylose consumption rate when cultured with a high concentration of xylose, mixed sugars, and under anaerobic conditions than S. stipitis. However, its ethanol productivity was less on 40 g/L xylose as the sole carbon source, mainly due to the formation of xylitol and glycerol [168].
One of the most popular methods used to increase S. stipitis tolerance towards inhibitors is adaptation by repeated sub-culturing or recycled of yeast cells while increasing concentrations of the inhibitor(s) in a stepwise fashion by adding more concentrated lignocellulosic hydrolysate solutions [4, 65, 193, 202]. Strains adapted to inhibitors present in a specific hydrolysate may exhibit cross tolerance to other hydrolysates. Adaptation has been done to individual inhibitor(s) as well as to mixtures of inhibitors, with the latter approach being most common. Ability to tolerate inhibitory compounds in lignocellulose hydrolysates could reduce the need for detoxification procedures, and this can decrease the overall production cost of ethanol. Adaptation is frequently substituted by random mutagenesis [11] or using genome shuffling via protoplast fusion of strains with different genotypes [9, 10]. To improve ethanol tolerance and production, similar approaches of random mutagenesis and genome shuffling were combined and used [58]. Evolution engineering has been used for selection of S. stipitis strains adapted to undetoxified hardwood spend liquor [145]. Selection of higher ethanol tolerance S. stipitis strains has been accomplished using UV mutagenesis has also led to improved ethanol production [193]. Protoplast fusion of S. stipitis with S. cerevisiae has allowed the isolation of a hybrid strain with higher ethanol productivity from xylose relative to the S. stipitis parental strain; however, such hybrid was unstable [204]. A popular method for genetic improvement that consists of genome shuffling uses transformation of a yeast strain with total DNA isolated from another strain of the same species or even from different yeast species. In one series of experiments, total DNA of S. stipitis was introduced in S. cerevisiae by electroporation and the best xylose-fermenting strain isolated was used as the source of DNA for the next round of transformation. This strain showed improvement in ethanol production from xylose and higher ethanol tolerance [209]. To isolate S. stipitis strains with improved growth and fermentation characteristics on the xylose/glucose mixture, several fast growing mutants were isolated on a xylose medium with respiration inhibitors antimicyn A and salicyl hydroxamate. Several other mutants have also been isolated which produced more ethanol on xylose/glucose mixture [172]. In another approach, mutants of S. stipitis which grow anaerobically on xylose plates were isolated. In contrast to the wild-type strain, these isolated mutants grew and fermented xylose and glucose anaerobically though very slowly [66].
Glucose prevents xylose utilization, as it competes with xylose for transport and its use is subject to glucose catabolite repression. To obtain strains of S. stipitis that can ferment simultaneously glucose and xylose, 2-deoxyglucose-resistant mutants were isolated [173]. This was also accomplished by deletion of the gene HXK1 coding for hexokinase I [26]. The last experiment could be considered as one of very few, in which a metabolic engineering approach was applied for this organism [26]. As a rule, experiments in metabolic engineering of S. stipitis are hampered by our limited knowledge of the limiting steps of the fermentation process. In another approach, a mutant of S. stipitis with disruption of cytochrome c was isolated. Due to defects in respiration, this mutant appeared to be superior when compared to the wild-type strain in xylose alcoholic fermentation, as it accumulated elevated amounts of ethanol [166]. No strains with glucose-insensitive xylose transport have been reported for S. stipitis.
To determine the gene(s) affecting S. stipitis fermentative capabilities, insertional mutants with altered ethanol production from glucose and xylose have been isolated (M. Semkiv, K. Berezka, K. Dmytruk, V. Passoth, A. Sibirny, unpublished). Mutants obtained by random insertional mutagenesis were screened for their growth abilities on solid media with different sugars and for resistance to the glycolysis inhibitor, 3-bromopyruvate. Fermentations in shake flask agitated cultures were carried out to measure sugar consumption and ethanol formation rates. Subsequently, the most interesting strains were analyzed to determine the genetic background of the observed alterations. Of more than 1300 screened mutants, 17 were identified that have significantly changed ethanol yields during the fermentation. In one of the best fermenting strains, a single insertion event resulted in the enhancement of ethanol formation in the media with both glucose and xylose. This strain had to have within the ORF a gene homologous to S. cerevisiae gene YDL119C that encoded for a not yet described mitochondrial transporter and was designated TMI1 (Transport to MItochondria). Mutant exhibited defects in glucose and xylose respiration. Wild-type phenotype was restored via complementation of the insertion mutation of the wild allele of TMI1 gene. In addition, the mentioned gene has been deleted and the fragment of TMI1 gene which is expressed in insertion mutant has been introduced into the deletion tmi1Δ strain. It could be suggested that the gene TMI1 is apparently involved in the control of hexose and pentose alcoholic fermentation in S. stipitis.
In summary, S. stipitis remains as one of the most efficient organisms for xylose and, in general, lignocellulose fermentation. Access to genome sequence and the development of methods of molecular genetics have been used to alleviate some of the shortcomings of this yeast (low tolerance to ethanol, glucose inhibition of xylose metabolism, and inability to grow anaerobically). This can be accomplished using the combination of rational design methods of metabolic engineering and random selection. Once the before mentioned shortcomings have been addressed, this organism yeast has the potential for use in industrial fermentation.
Ogataea polymorpha
O. polymorpha (earlier was designated as Hansenula polymorph or Pichia angusta) is one of the most thermotolerant yeasts known. It has the ability to grow up to 50 °C. Wild-type strains of O. polymorpha are able to ferment glucose, xylose, mannose, maltose, and cellobiose into ethanol, but are not able to utilize and ferment galactose and l-arabinose [160]. Sugar fermentation in this yeast is most efficient under conditions of limited aeration and even at 45–48 °C. At the industrial scale, high-temperature tolerance can reduce cooling costs and the risk of contamination with more energy-efficient removal of ethanol due to lower difference between fermentation and distillation temperatures. Several successful attempts have been made to improve thermotolerance of this yeast even further [70]. It is known that, similar to other fungi, O. polymorpha accumulates trehalose and expresses heat shock proteins (Hsps) under heat shock conditions [53]. The increase in the intracellular level of trehalose in O. polymorpha following the knock out of acid trehalase gene ATH1 resulted in a sixfold higher ethanol production of xylose fermentation at 50 °C. The overexpression of the heat shock proteins Hsp16 and Hsp104 has also led to three to six times improved ethanol production at 50 °C [70].
High ethanol tolerance is another important feature of ethanol producers for industrial applications. O. polymorpha appears to be more resistant to ethanol than S. stipitis; but it is more sensitive than S. cerevisiae [32]. Overexpression of the endogenous ETT1 gene (a homolog of S. cerevisiae MPE1 gene) significantly increased the resistance of O. polymorpha to ethanol, resulting in 10- and 3-fold improvements in the growth on agar and in liquid media with ethanol, respectively. The resistance of O. polymorpha to ethyl alcohol was also enhanced by heterologous overexpression of S. cerevisiae MPR1, which codes for acetyltransferase [69].
Direct microbial conversion of polysaccharides into ethanol is a promising technology for the production of alcohols from lignocellulosic raw material. The optimal temperature for the activity of hydrolytic enzymes used during microbial conversion of polymers into ethanol is about 50 °C. Recombinant O. polymorpha strains that ferment starch were constructed by the expression of heterologous secretory α-amylase and glucoamylase coding for the SWA2 and GAM1 genes of Schwanniomyces occidentalis [190]. Heterologous expression of XYN2 of Trichoderma reesei and xlnD of Aspergillus niger that code for secretory endoxylanase and secretory β-xylosidase resulted in strains of O. polymorpha with an ability for direct xylan fermentation at high-temperature [185].
O. polymorpha has the potential to be used for efficient simultaneous saccharification and fermentation (SSF) due to its temperature tolerance and ability to ferment xylose to ethanol [151]. However, in spite of the robust growth on xylose, the ethanol yield and productivity from this sugar in wild-type strains of O. polymorpha are very low (0.03 g/g and 0.02 g/L/h, respectively). Identification of rate-limiting enzymes for xylose conversion to ethanol is necessary for rational strain modification to improve the fermenting efficiency.
The molecular tools for this yeast species are well-developed [45] and a complete genome sequence of the strain NCYC495 of O. polymorpha is publically available (http://genome.jgi-psf.org/Hanpo2/Hanpo2.home.html). The genome of O. polymorpha has been annotated [155]. As a consequence, a combination of metabolic engineering and classical selection approaches was successfully used to improve the efficiency of xylose alcoholic fermentation in O. polymorpha [100].
The difference in cofactor specificity at the first steps of xylose metabolism results in cofactor imbalance causing a substantially reduced ethanol production with accumulation of xylitol as a by-product in yeasts, including that of O. polymorpha. The problem arises during consecutive action of NADPH-dependent XR and NAD-dependent XDH that catalyze the reduction of xylose to xylitol and the oxidation of xylitol to xylulose (Fig. 1). To avoid the cofactor imbalance, XR and XDH were replaced with bacterial XI, which directly converts xylose into xylulose with no cofactors required. The bacterial genes xylA from E. coli or Streptomyces coelicolor coding for XI was successfully expressed in a xylose negative O. polymorpha strain in which the XYL1 gene coding for XR and two paralogs XYL2A and XYL2B of XDH had been deleted. The recombinant strains were able to grow on xylose as carbon source; however, the amount of accumulated ethanol remained at the level of the parental wild-type strain CBS4732 (0.15 g/L) [186]. The overexpression of E. coli xylA together with O. polymorpha XYL3 that codes for XK has led to fourfold increase of ethanol production, but still, the maximal ethanol accumulation did not exceed 0.6 g/L at 48 °C [34].
The XR of O. polymorpha can use both NADPH and NADH as cofactors. However, the affinity of XR to NADH is significantly lower than that to NADPH. Therefore, another approach to eliminate the imbalance of these two cofactors was based on engineering of the O. polymorpha XR with reduced affinity towards NADPH. Using site-specific mutagenesis, a modified XR was constructed by the substitution of lysine and asparagine for arginine and aspartic acid at amino acid positions 341 and 343 [34, 147]. As a result of the modification of the primary structure of the protein, the affinity of XR to NADPH decreased 17-fold as compared to the native enzyme, while the affinity of modified XR to NADH remained unchanged. A recombinant strain of O. polymorpha with enhanced expression of modified XR (engineered gene was designated as XYL1 m) and a native XDH and XK was characterized by a fivefold decrease of xylitol accumulation as compared to the wild-type strain and twofold higher ethanol production reaching 1.3 g of ethanol/L [33].
During high-temperature xylose alcoholic fermentation, the wild-type O. polymorpha strain NCYC495 can accumulate up to 0.5 g of ethanol/L as compared to the 0.15 g/L seen with strain CBS4732. Pyruvate decarboxylase (PDC) is one of the key enzymes of the final steps of alcoholic fermentation, catalyzing the conversion of pyruvate into acetaldehyde and CO2. Subsequently, acetaldehyde is reduced to ethanol by alcohol dehydrogenase (ADH) that is encoded by the ADH1 gene (Fig. 1). Under conditions of limited aeration, a sufficient activity of PDC is important to redirect pyruvate towards ethanol formation instead of respiration. Overexpression of endogeneous PDC1 under control of the strong constitutive GAPp from glyceraldehyde-3-phosphate dehydrogenase using a plasmid with multicopy integration doubled ethanol production from xylose in the wild-type strain NCYC495 [71]. Overexpression of both PDC1 and ADH1 genes in O. polymorpha has resulted in an additional twofold activation of xylose alcoholic fermentation when compared to the strain expressing solely PDC1 [102].
The O. polymorpha mutant 2EthOH− unable to utilize ethanol as a sole carbon source was isolated from strain NCYC495 by UV mutagenesis and characterized by a threefold increase in ethanol production from xylose. Subsequently, overexpression of the gene PDC1 in this mutant− further improved ethanol accumulation, reaching 2.5 g of ethanol/L at 48 °C [71]. To achieve higher ethanol production from xylose, several successful metabolic engineering approaches were combined to modify the genome of the 2EthOH− mutant. The overexpression of the genes XYL1 m, XYL2, and XYL3, that code for the modified XR and native XDH and XK, on the background of non-identified mutation in the strain 2EthOH− led to a substantial increase in ethanol accumulation during xylose fermentation (7.4 g/L at 45 °C relative to 0.6 g/L in the wild-type strain NCYC495) [100]. The additional activation of PDC did not lead to any further improvement of xylose conversion to ethanol though the overexpression of PDC1 on the background of XYL1 m and XYL2 overexpressed strain, increased ethanol production. The impact of XK on ethanol production during xylose alcoholic fermentation is more pronounced relative to PDC, suggesting that PDC does not limit xylose conversion in strain with higher activities of XR, XDH, and XK.
Mutants selected on a medium supplemented with toxic concentrations of 3-bromopyruvate (BrPA) were characterized by additional increase in ethanol production from xylose (to 10 g/L at 45 °C) [30, 100]. BrPA is known as a compound causing ATP depletion by the inhibition of the glycolytic enzymes, hexokinase II, glyceraldehyde-3-phosphate dehydrogenase and 3-phosphoglycerate kinase [44]. While mutation(s) causing resistance to BrPA in the ethanol overproducing strain remain to be identified, a corresponding mutation was mapped in the strain with the wild-type background. Insertional mutagenesis was used for NCYC495 strain of O. polymorpha with subsequent selection of transformants using a mineral medium supplemented with 25 mM of BrPA. Sequencing of the flanking regions revealed that the insertional cassette disrupted the ORF of a gene homologous to the S. cerevisiae autophagy-related gene ATG13 [31]. This gene encodes a regulatory subunit of the Atg1-signaling complex, stimulating Atg1 kinase activity, which is required for vesicle formation during autophagy and the cytoplasm-to-vacuole targeting pathway. The ATG13 gene mutation led to a 40% increase in ethanol production from xylose as compared to the parental strain. However, the mechanism of such regulation remains unknown [99].
Further possible increase in ethanol yield and productivity from xylose in O. polymorpha is hampered by the lack of the knowledge of the regulation of xylose metabolism and fermentation. Therefore, it is important to identify the corresponding genes and, depending on their functions, activate or repress them. Xylose is a unique carbon source that can be fermented to ethanol, similar to glucose, and simultaneously able to be converted to glucose and other hexoses. It is possible mostly in PPP, though partial contribution of gluconeogenesis in hexose synthesis from xylose cannot be neglected. The CAT8 gene that codes for a zinc-finger cluster protein regulates at least 30 genes involved in gluconeogenesis, ethanol utilization, glyoxylate cycle, and diauxic shift from fermentation to respiration [59]. The roles of CAT8 gene in the regulation of cell metabolism are well understood in S. cerevisiae. It was shown that the deletion of this gene in S. cerevisiae and P. guilliermondii activated glucose alcoholic fermentation, though maximally achieved level of ethanol in the latter species was still very low [149, 191]. The role of CAT8 in regulation of xylose metabolism was poorly understood. Transcriptome analysis of the natural xylose-metabolizing yeast O. polymorpha did not find changes in CAT8 expression between xylose- and glucose-containing media [88].
To define the role of Cat8 transcriptional factor in xylose fermentation, O. polymorpha cat8Δ knock out mutants were constructed and analyzed from either wild type or ethanol overproducing strain (from xylose) [159]. In O. polymorpha, CAT8 deletion did not lead to any significant changes in ethanol production from glucose, while a considerable increase in xylose alcoholic fermentation was observed (Fig. 2). The cell respiration of cat8Δ mutants on xylose was impaired to a higher extent relative to that on glucose as a substrate. Moreover, the impaired ethanol and glycerol utilization in cat8Δ mutants were observed, suggesting the involvement of CAT8 in the regulation of gluconeogenesis in O. polymorpha, similar to that described for S. cerevisiae. Remarkably, growth on xylose of the cat8Δ mutant strain was also slightly worsened, suggesting that xylose can be considered, at least partially, as a gluconeogenic substrate. Only slight decrease in the specific activity of fructose-1,6-bisphosphatase in cat8Δ mutants was observed, suggesting differences in Cat8 action between S. cerevisiae and O. polymorpha. It could be suggested that the drop in other enzymes of gluconeogenesis, e.g., phosphoenolpyruvate carboxykinase, explains growth impairment of O. polymorpha cat8Δ mutants on gluconeogenic substrates. Overexpression of CAT8 had the opposite effect on xylose alcoholic fermentation as compared to that in cat8Δ mutants, as transformants overexpressing CAT8 gene were characterized by a decrease in ethanol production from xylose (Fig. 2) [159].
Ethanol production during xylose fermentation of O. polymorpha WT, cat8∆ and WT/CAT8 strains. Alcoholic fermentation of yeast strains was fulfilled by cultivation in liquid mineral medium at oxygen-limited conditions at 45 °C. The conditions were provided by agitation at 140 rpm. 9% xylose was added into the medium used for the fermentation. The cells were pregrown in 100 ml of liquid YPX medium (1% yeast extract, 2% peptone and 4% xylose) in 300 ml Erlenmeyer flasks at 220 rpm till the mid-exponential growth phase. Then the cells were precipitated by centrifugation, washed by water and inoculated into 40 ml of the fermentation medium in 100 mL Erlenmeyer flasks covered with cotton plugs. The initial biomass concentration for fermentation experiments was 2 g (dry weight)/l
Thus, the CAT8 gene is one of the first identified genes that are involved in the regulation of xylose alcoholic fermentation in natural xylose-fermenting yeasts. Mutant O. polymorpha cat8Δ isolated from the advanced ethanol producer accumulated 30% more ethanol relative to the parental strain, reaching 12.5 g ethanol/L at 45 °C, which is the highest ethanol titer for high-temperature xylose fermentation [159]. The ethanol yield of the constructed O. polymorpha recombinant strain (0.34 g/g xylose) is close to that described for S. stipitis (0.35–0.44 g/g xylose) [73] and S. passalidarum (0.42 g/g xylose) [114]. However, this was achieved for O. polymorpha at 45 °C, whereas the compared organisms are mesophilic and, therefore, unable to grow and ferment at such a high temperature. Among the thermotolerant ethanol-producing strains, the most promising one is an engineered K. marxianus strain with ethanol yield 0.38 g/g xylose at 42 °C, but lower yield at 45 °C (0.27 g/g xylose). The additional advantage for O. polymorpha recombinant strain in contrast to recombinant K. marxianus was that no xylitol accumulation can be observed [207].
O. polymorpha belongs to the methylotrophic yeasts that are capable for growth on methanol as sole source of carbon and energy. To enable methanol utilization, these organisms have evolved highly specialized metabolic pathways that are partly compartmentalized in peroxisomes. The first enzyme of methanol catabolism, alcohol oxidase, catalyzes oxidation of methanol into the two reactive compounds, formaldehyde, and hydrogen peroxide. Alcohol oxidase is localized in peroxisomes together with catalase, which decomposes hydrogen peroxide into water and oxygen. A third peroxisomal enzyme of methanol metabolism is dihydroxyacetone synthase (DHAS). DHAS is a component of the xylulose-5-phosphate cycle and catalyzes the transfer of two-carbon moieties from xylulose-5-phosphate to formaldehyde with the production of glyceraldehyde-3-phosphate (an intermediate of glycolysis) and dihydroxyacetone, which after phosphorylation is converted to a glycolytic intermediate [181]. DHAS can also display classical transketolase activity using aldose phosphates (such as ribose 5-phosphate) as the acceptors for the glycolyl group from the donor substrate xylulose 5-phosphate, therefore, playing role in xylose utilization through PPP [112]. Screening for other enzymes that are putatively involved in xylose utilization in O. polymorpha has revealed a peroxisomal transaldolase coded for the gene TAL2. Fluorescent labeling proved the peroxisomal localization of Tal2 protein [101]. The functional roles of peroxisome-localized transaldolase and the specific peroxisomal transketolase in xylose utilization and fermentation in O. polymorpha remain unclear. To investigate the role of these enzymes in ethanol production during xylose fermentation, the corresponding genes DAS1 and TAL2 were overexpressed in O. polymorpha NCYC495 strain under control of the strong constitutive promoter of GAP1 gene (encodes glycerol-3-phosphate dehydrogenase) using a plasmid for multicopy integration pGLG61 [181]. The recombinant strains overexpressing DAS1 and TAL2 revealed 4.6- and 1.5-fold increase in the specific activity of the corresponding enzymes. The overexpression of TAL2 gene resulted in a 1.5-fold increase in ethanol production at the 4th day of xylose fermentation as compared to the wild-type strain (Fig. 3). The effect of the overexpression of DAS1 gene was more pronounced. A strain overexpressing DAS1 gene synthesized 2.3-fold higher amount of ethanol than that of the parental strain after 4 days of xylose fermentation (Fig. 3) [98, 101]. Both das1Δ and tal2Δ mutants did not show any growth retardation on xylose as carbon source, but were impaired in xylose alcoholic fermentation as compared to the wild-type strain (Fig. 3). Overexpression of DAS1 and TAL2 genes in O. polymorpha in an advanced ethanol producer increased ethanol production by 40% up to 16 g ethanol/L during xylose alcoholic fermentation at 45 °C [101]. As a consequence, it was shown for the first time that peroxisomal enzymes Das1 and Tal2 are involved in the xylose alcoholic fermentation in O. polymorpha; however, the functions of peroxisomes during alcoholic fermentation of xylose require further investigation. It could be concluded that peroxisomal transketolase and transaldolase are important for xylose fermentation, but not for utilization of this sugar. O. polymorpha mutants with knock out and overexpression of genes TKL1 and TAL1 coding for cytosolic transketolase and transaldolase, respectively, also were constructed. The mutants tkl1Δ and tal1Δ with knock out of these genes did not grow on xylose as sole carbon source, whereas fermented this pentose to ethanol [101]. These data show different roles of cytosolic and peroxisomal transketolases and transaldolases in xylose utilization and alcoholic fermentation. Overexpression of TKL1 and TAL1 genes activated xylose fermentation (O. Kurylenko, K. Dmytruk, A. Sibirny, unpublished). It is also worth to mention that xylose but not glucose fermentation in O. polymorpha was blocked in the mutants defective in peroxisome biogenesis pex3Δ and pex6Δ, whereas pex3Δ mutants of the non-methylotrophic yeast S. stipitis did not differ from the wild-type strain regarding xylose fermentation [101].
Ethanol yield and the productivity of the best O. polymorpha strain with overexpressed genes DAS1 and TAL2 at 45 °C are like that described for another thermotolerant yeast K. marxianus at 42 °C; however, at 45 °C, K. marxianus showed a drop in the mentioned parameters. Moreover, the strain of O. polymorpha did not accumulate xylitol during xylose fermentation, whereas K. marxianus accumulated large amounts of this by-product [207]. Apparently, the main drawback of the constructed O. polymorpha strain is incomplete xylose consumption under fermentation condition. The ethanol yield in the best obtained O. polymorpha ethanol producer also is not high enough for economic feasibility. Further improvement of the parameters of alcoholic fermentation of xylose in O. polymorpha could be obtained by optimization of the transport of this pentose into cells, amplification of the limiting genes of glycolysis and the PPP, as well as that of the genes determining the resistance to toxic and inhibitory compounds derived after pretreatment of lignocellulosic biomass. At present, the resistance of O. polymorpha to toxic products (aldehydes, phenols, acetic and formic acids) accumulated in lignocellulose hydrolysates under the conditions of acidic hydrolysis has not been precisely studied.
Concluding remarks
Commercial strains of S. cerevisiae which are characterized by increased ethanol yield from glucose and sucrose (1G ethanol) have been deployed extensively for industrial production. However, apparently, all of these strains have some drawbacks. Strains of S. cerevisaie which accumulate more ethanol by cost of biomass typically show lower robustness and cannot compete with wild-type contaminants during non-sterile production process. Strains that accumulate more ethanol due to lower glycerol production display worse performance being more susceptible to osmotic stress relative to the wild-type strains. Mentioned shortcomings should be overcome by additional metabolic changes.
S. cerevisiae strains constructed for the production of 2G ethanol efficiently ferment, in addition to glucose, abundant pentose sugars of lignocellulosic hydrolyzates, xylose, and l-arabinose. Strains fermenting galactose have also been constructed. The strains are also known which could ferment different sugars of hydrolyzates simultaneously due to elaboration of specific xylose transporters and expression of genes responsible for xylose catabolism under control of strong constitutive promoters. Strains resistant to inhibitors of lignocellulosic hydrolyzates are also described. Pilot-plant production of 2G ethanol using engineered S. cerevisiae strains has been started.
S. stipitis strains with further improvements of xylose fermentation on lignocellulosic hydrolyzates have been constructed. Such strains have defects in glucose catabolite repression and are more resistant to inhibitors present in hydrolyzates. Still, low ethanol tolerance and the need in oxygen for growth are major drawbacks of S. stipitis that need to still be addressed. Plans to start pilot plants for 2G ethanol production based on S. stipitis are known (T.W. Jeffries, personal communication). If so, such plant could be good platform for further development of S. stipitis strains.
O. polymorpha also looks as another promising organism for 2G ethanol production especially as it could ferment at temperatures of 45 °C and higher which is compatible with SSF process. However, the level of ethanol production from xylose is still low, glucose inhibits xylose utilization, and fermentation characteristics of glucose and xylose from the real lignocellulosic hydrolyzates are not known. l-arabinose and galactose are not metabolized by O. polymorpha. These questions and problems need to be addressed prior to commercial deployment of this yeast.
References
Agbogbo FK, Coward-Kelly G, Torry-Smith M, Wenger KS (2006) Fermentation of glucose/xylose mixtures using P. stipitis. Process Biochem 41:2333–2336. https://doi.org/10.1016/j.procbio.2006.05.004
Agbogbo FK, Wenger KS (2006) Effect of pretreatment chemicals on xylose fermentation by P. stipitis. Biotechnol Lett 28:2065–2069. https://doi.org/10.1007/s10529-006-9192-6
Almeida JR, Roder A, Modig T, Laadan B, Liden G, Gorwa-Grauslund MF (2008) NADH- vs NADPH-coupled reduction of 5-hydroxymethyl furfural (HMF) and its implications on product distribution in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 78:939–945. https://doi.org/10.1007/s00253-008-1364-y
Amartey S, Jeffries T (1996) An improvement in Pichia stipitis fermentation of acid-hydrolysed hemicellulose achieved by overliming (calcium hydroxide treatment) and strain adaptation. World J Microbiol Biotechnol 12:281–283. https://doi.org/10.1007/bf00360928
Amartey SA, Jeffries TW (1994) Comparison of corn steep liquor with other nutrients in the fermentation of D-xylose by Pichia stipitis CBS6054. Biotechnol Lett 16:211–214. https://doi.org/10.1007/bf01021673
An MZ, Tang YQ, Mitsumasu K, Liu ZS, Shigeru M, Kenji K (2011) Enhanced thermotolerance for ethanol fermentation of Saccharomyces cerevisiae strain by overexpression of the gene coding for trehalose-6-phosphate synthase. Biotechnol Lett 33:1367–1374. https://doi.org/10.1007/s10529-011-0576-x
Ansell R, Granath K, Hohmann S, Thevelein JM, Adler L (1997) The two isoenzymes for yeast NAD+-dependent glycerol 3-phosphate dehydrogenase encoded by GPD1 and GPD2 have distinct roles in osmoadaptation and redox regulation. EMBO J 16:2179–2187. https://doi.org/10.1093/emboj/16.9.2179
Bafuelos M, Gancedo C (1978) In situ study of the glycolytic pathway in Saccharomyces cerevisiae. Arch Microbiol 117:197–201. https://doi.org/10.1007/bf00402308
Bajwa PK, Phaenark C, Grant N, Zhang X, Paice M, Martin VJ, Trevors JT, Lee H (2011) Ethanol production from selected lignocellulosic hydrolysates by genome shuffled strains of Scheffersomyces stipitis. Bioresour Technol 102:9965–9969. https://doi.org/10.1016/j.biortech.2011.08.027
Bajwa PK, Pinel D, Martin VJ, Trevors JT, Lee H (2010) Strain improvement of the pentose-fermenting yeast Pichia stipitis by genome shuffling. J Microbiol Methods 81:179–186. https://doi.org/10.1016/j.mimet.2010.03.009
Bajwa PK, Shireen T, D’Aoust F, Pinel D, Martin VJ, Trevors JT, Lee H (2009) Mutants of the pentose-fermenting yeast Pichia stipitis with improved tolerance to inhibitors in hardwood spent sulfite liquor. Biotechnol Bioeng 104:892–900. https://doi.org/10.1002/bit.22449
Balagurunathan B, Jonnalagadda S, Tan L, Srinivasan R (2012) Reconstruction and analysis of a genome-scale metabolic model for Scheffersomyces stipitis. Microb Cell Fact 11:27. https://doi.org/10.1186/1475-2859-11-27
Bamba T, Hasunuma T, Kondo A (2016) Disruption of PHO13 improves ethanol production via the xylose isomerase pathway. AMB Express 6:4. https://doi.org/10.1186/s13568-015-0175-7
Basso TO, de Kok S, Dario M, do Espirito-Santo JC, Müller G, Schlölg PS, Silva CP, Tonso A, Daran JM, Gombert AK, van Maris AJ, Pronk JT, Stambuk BU (2011) Engineering topology and kinetics of sucrose metabolism in Saccharomyces cerevisiae for improved ethanol yield. Metab Eng 13:694–703. https://doi.org/10.1016/j.ymben.2011.09.005
Bengtsson O, Hahn-Hägerdal B, Gorwa-Grauslund MF, Bengtsson O (2009) Xylose reductase from Pichia stipitis with altered coenzyme preference improves ethanolic xylose fermentation by recombinant Saccharomyces cerevisiae. Biotechnol Biofuels 2:9. https://doi.org/10.1186/1754-6834-2-9
Benisch F, Boles E (2014) The bacterial Entner-Doudoroff pathway does not replace glycolysis in Saccharomyces cerevisiae due to the lack of activity of iron-sulfur cluster enzyme 6-phosphogluconate dehydratase. J Biotechnol 171:45–55. https://doi.org/10.1016/j.jbiotec.2013.11.025
Bera AK, Ho NW, Khan A, Sedlak M (2011) A genetic overhaul of Saccharomyces cerevisiae 424A(LNH–ST) to improve xylose fermentation. J Ind Microbiol Biotechnol 38:617–626. https://doi.org/10.1007/s10295-010-0806-6
Bettiga M, Bengtsson O, Hahn-Hagerdal B, Gorwa-Grauslund MF (2009) Arabinose and xylose fermentation by recombinant Saccharomyces cerevisiae expressing a fungal pentose utilization pathway. Microb Cell Fact 8:40. https://doi.org/10.1186/1475-2859-8-40
Bicho PA, Runnals PL, Cunningham JD, Lee H (1988) Induction of xylose reductase and xylitol dehydrogenase activities in Pachysolen tannophilus and Pichia stipitis on mixed sugars. Appl Environ Microb 54:50–54
Brat D, Boles E, Wiedemann B (2009) Functional expression of a bacterial xylose isomerase in Saccharomyces cerevisiae. Appl Environ Microbiol 75:2304–2311. https://doi.org/10.1128/aem.02522-08
Cadiere A, Ortiz-Julien A, Camarasa C, Dequin S (2011) Evolutionary engineered Saccharomyces cerevisiae wine yeast strains with increased in vivo flux through the pentose phosphate pathway. Metab Eng 13:263–271. https://doi.org/10.1016/j.ymben.2011.01.008
Cho JY, Jeffries TW (1998) Pichia stipitis genes for alcohol dehydrogenase with fermentative and respiratory functions. Appl Environ Microb 64:1350–1358
Clifton D, Fraenkel DG (1981) The gcr (glycolysis regulation) mutation of Saccharomyces cerevisiae. J Biol Chem 256:13074–13078
Conceicao GJA, Moran PJS, Rodrigues JAR (2003) Highly efficient extractive biocatalysis in the asymmetric reduction of an acyclic enone by the yeast Pichia stipitis. Tetrahedron-Asymmetr 14:43–45. https://doi.org/10.1016/s0957-4166(02)00798-x
Cunha JT, Soares PO, Romaní A, Thevelein JM, Domingues L (2019) Xylose fermentation efficiency of industrial Saccharomyces cerevisiae yeast with separate or combined xylose reductase/xylitol dehydrogenase and xylose isomerase pathways. Biotechnol Biofuels 12:20. https://doi.org/10.1186/s13068-019-1360-8
Dashtban M, Wen X, Bajwa PK, Ho CY, Lee H (2015) Deletion of hxk1 gene results in derepression of xylose utilization in Scheffersomyces stipitis. J Ind Microbiol Biotechnol 42:889–896. https://doi.org/10.1007/s10295-015-1614-9
De Kok S, Kozak B, Pronk J, Van Maris A (2012) Energy coupling in Saccharomyces cerevisiae: selected opportunities for metabolic engineering. FEMS Yeast Res 12:387–397. https://doi.org/10.1111/j.1567-1364.2012.00799.x
Della-Bianca BE, Basso TO, Stambuk BU, Basso LC, Gombert AK (2013) What do we know about the yeast strains from the Brazilian fuel ethanol industry? Appl Micro Biotechnol 97:979–991. https://doi.org/10.1007/s00253-012-4631-x
Dmytruk K, Kshanovska B, Abbas C, Sibirny A (2016) New methods for positive selection of yeast ethanol overproducing mutants. Bioethanol 2:24–31. https://doi.org/10.1515/bioeth-2015-0003
Dmytruk K, Kshanovska B, Sibirny A (2015) New methods for selection of Saccharomyces cerevisiae ethanol overproducing mutants resistant to oxythiamine, trehalose, bromopyruvate, glyoxylic acid and glucosamine. In: Sibirny A, Fedorovyvh D, Gonchar M, Grabek-Lejko D (eds) Living organisms and bioanalytical approaches for detoxification and monitoring of toxic compounds. University of Rzeszow, Rzeszow, pp 237–246
Dmytruk KV, Ruchala J, Grabek-Lejko D, Puchalski C, Bulbotka NV, Sibirny AA (2018) Autophagy-related gene ATG13 is involved in control of xylose alcoholic fermentation in the thermotolerant methylotrophic yeast Ogataea polymorpha. FEMS Yeast Res 18:foy010. https://doi.org/10.1093/femsyr/foy010
Dmytruk KV, Sibirny AA (2013) Metabolic engineering of yeast Hansenula polymorpha for construction of efficient ethanol producers. Tsitol Genet 47:329–342. https://doi.org/10.3103/s0095452713060029
Dmytruk OV, Dmytruk KV, Abbas CA, Voronovsky AY, Sibirny AA (2008) Engineering of xylose reductase and overexpression of xylitol dehydrogenase and xylulokinase improves xylose alcoholic fermentation in the thermotolerant yeast Hansenula polymorpha. Microb Cell Fact 7:21. https://doi.org/10.1186/1475-2859-7-21
Dmytruk OV, Voronovsky AY, Abbas CA, Dmytruk KV, Ishchuk OP, Sibirny AA (2008) Overexpression of bacterial xylose isomerase and yeast host xylulokinase improves xylose alcoholic fermentation in the thermotolerant yeast Hansenula polymorpha. FEMS Yeast Res 8:165–173. https://doi.org/10.1111/j.1567-1364.2007.00289.x
Du J, Li S, Zhao H (2010) Discovery and characterization of novel D-xylose-specific transporters from Neurospora crassa and Pichia stipitis. Mol BioSyst 6:2150–2156. https://doi.org/10.1039/c0mb00007h
du Preez JC, Bosch M, Prior B (1986) The fermentation of hexose and pentose sugars by Candida shehatae and Pichia stipitis. Appl Microbiol Biotechnol 23:228–233. https://doi.org/10.1007/bf00261920
du Preez JC, Bosch M, Prior BA (1985) Xylose fermentation by Candida shehatae and Pichia stipitis: effects of pH, temperature and substrate concentration. Enzyme Microb Technol 8:360–364. https://doi.org/10.1016/0141-0229(86)90136-5
du Preez JC, van der Walt JP (1983) Fermentation of D-xylose to ethanol by a strain of Candida shehatae. Biotechnol Lett 5:357–362. https://doi.org/10.1007/bf01141138
du Preez JC, van Driessel B, Prior BA (1989) Ethanol tolerance of Pichia stipitis and Candida shehatae strains in fed-batch cultures at controlled low dissolved-oxygen levels. Appl Microbiol Biot 30:53–58. https://doi.org/10.1007/bf00255996
Duwe A, Tippkötter N, Ulber R (2019) Lignocellulose-biorefinery: ethanol-focused. Adv Biochem Eng Biotechnol 166:177–215. https://doi.org/10.1007/10_2016_72
Farwick A, Bruder S, Schadeweg V, Oreb M, Boles E (2014) Engineering of yeast hexose transporters to transport D-xylose without inhibition by D-glucose. Proc Natl Acad Sci 111:5159–5164. https://doi.org/10.1073/pnas.1323464111
Favaro L, Jansen T, van Zyl WH (2019) Exploring industrial and natural Saccharomyces cerevisiae strains for the bio-based economy from biomass: the case of bioethanol. Crit Rev Biotechnol 39:800–816. https://doi.org/10.1080/07388551.2019.1619157
Feng Q, Liu ZL, Weber SA, Li S (2018) Signature pathway expression of xylose utilization in the genetically engineered industrial yeast Saccharomyces cerevisiae. PLoS One 13:e0195633. https://doi.org/10.1371/journal.pone.0195633
Ganapathy-Kanniappan S, Vali M, Kunjithapatham R, Buijs M, Syed LH, Rao PP, Ota S, Kwak BK, Loffroy R, Geschwind JF (2010) 3-bromopyruvate: a new targeted antiglycolytic agent and a promise for cancer therapy. Curr Pharm Biotechnol 11:510–517. https://doi.org/10.2174/1389210204205652010
Gellisen G (2000) Heterologous protein production in methylotrophic yeasts. Appl Microbiol Biotechnol 54:741–750. https://doi.org/10.1007/s002530000464
Gellisen G (2002) Hansenula polymorpha—biology and applications. Wiley VCH, Weinheim
Ghosh A, Zhao H, Price ND (2011) Genome-scale consequences of cofactor balancing in engineered pentose utilization pathways in Saccharomyces cerevisiae. PLoS One 6:e27316. https://doi.org/10.1371/journal.pone.0027316
Gombert AK, van Maris AJ (2015) Improving conversion yield of fermentable sugars into fuel ethanol in 1st generation yeast-based production processes. Curr Opin Biotechnol 33:81–86. https://doi.org/10.1016/j.copbio.2014.12.012
Gorsich SW, Dien BS, Nichols NN, Slininger PJ, Liu ZL, Skory CD (2006) Tolerance to furfural-induced stress is associated with pentose phosphate pathway genes ZWF1, GND1, RPE1, and TKL1 in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 71:339–349. https://doi.org/10.1007/s00253-005-0142-3
Grootjen DRJ, van der Lans RGJM, Luyben KChA (1990) Effects of the aeration rate on the fermentation of glucose and xylose by Pichia stipitis CBS 5773. Enzyme Microb Technol 12:20–23. https://doi.org/10.1016/0141-0229(90)90174-o
Guadalupe Medina V, Almering MJH, van Maris AJA, Pronk JT (2010) Elimination of glycerol production in anaerobic cultures of a Saccharomyces cerevisiae strain engineered to use acetic acid as an electron acceptor. Appl Environ Microbiol 76:190–195. https://doi.org/10.1128/aem.01772-09
Guebel DV, Cordenons A, Cascone O, Giulietti AM, Nudel C (1992) Influence of the nitrogen source on growth and ethanol production by Pichia stipitis NRRL Y-7124. Biotechnol Lett 14:1193–1198. https://doi.org/10.1007/bf01027027
Guerra E, Chye PP, Berardi E, Piper PW (2005) Hypoxia abolishes transience of the heat-shock response in the methylotrophic yeast Hansenula polymorpha. Microbiology 151:805–811. https://doi.org/10.1099/mic.0.27272-0
Guo ZP, Zhang L, Ding ZY, Shi GY (2011) Minimization of glycerol synthesis in industrial ethanol yeast without influencing its fermentation performance. Metab Eng 13:49–59. https://doi.org/10.1016/j.ymben.2010.11.003
Ha SJ, Wei Q, Kim SR, Galazka JM, Cate JH, Jin YS (2011) Cofermentation of cellobiose and galactose by an engineered Saccharomyces cerevisiae strain. Appl Environ Microbiol 77:5822–5825. https://doi.org/10.1128/aem.06776-11
Hahn-Hägerdal B, Galbe M, Gorwa-Grauslund MF, Lidén G, Zacchi G (2006) Bio-ethanol—the fuel of tomorrow from the residues of today. Trends Biotechnol 24:549–556. https://doi.org/10.1016/j.tibtech.2006.10.004
Hahn-Hägerdal B, Wahlbom CF, Gárdonyi M, van Zyl WH, Cordero Otero RR, Jönsson LJ (2001) Metabolic engineering of Saccharomyces cerevisiae for xylose utilization. Adv Biochem Eng. Biotechnol 73:53–84. https://doi.org/10.1007/3-540-45300-8_4
Harner NK, Wen X, Bajwa PK, Austin GD, Ho CY, Habash MB, Trevors JT, Lee H (2015) Genetic improvement of native xylose-fermenting yeasts for ethanol production. J Ind Microbiol Biotechnol 42:1–20. https://doi.org/10.1007/s10295-014-1535-z
Haurie V, Perrot M, Mini T, Jenö P, Sagliocco F, Boucherie H (2001) The transcriptional activator Cat8p provides a major contribution to the reprogramming of carbon metabolism during the diauxic shift in Saccharomyces cerevisiae. J Biol Chem 276:76–85. https://doi.org/10.1074/jbc.m008752200
Hawkins GM, Ghose D, Russel J, Peterson J (2013) Production of ethanol from high dry matter of pretreated loblolly pine by an evolved strain of Saccharomyces cerevisiae. J Bioremediation Biodegrad 4:195. https://doi.org/10.4172/2155-6199.1000195
Hector RE, Qureshi N, Hughes SR, Cotta MA (2008) Expression of a heterologous xylose transporter in a Saccharomyces cerevisiae strain engineered to utilize xylose improves aerobic xylose consumption. Appl Microbiol Biotechnol 80:675–684. https://doi.org/10.1007/s00253-008-1583-2
Higgins VJ, Bell PJL, Dawes IW, Attfield PV (2001) Generation of a novel Saccharomyces cerevisiae strain that exhibits strong maltose utilization and hyperosmotic resistance using nonrecombinant techniques. Appl Environ Microbiol 67:4346–4348. https://doi.org/10.1128/aem.67.9.4346-4348.2001
Hohmann S (2002) Osmotic stress signaling and osmoadaptation in yeasts. Microbiol Mol Biol Rev 66:300–372. https://doi.org/10.1128/mmbr.66.2.300-372.2002
Hou L (2010) Improved production of ethanol by novel genome shuffling in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 160:1084–1093. https://doi.org/10.1007/s12010-009-8552-9
Huang CF, Lin TH, Guo GL, Hwang WS (2009) Enhanced ethanol production by fermentation of rice straw hydrolysate without detoxification using a newly adapted strain of Pichia stipitis. Bioresour Technol 100:3914–3920. https://doi.org/10.1016/j.biortech.2009.02.064
Hughes SR, Gibbons WR, Bang SS, Pinkelman R, Bischoff KM, Slininger PJ, Qureshi N, Kurtzman CP, Liu S, Saha BC, Jackson JS, Cotta MA, Rich JO, Javers JE (2012) Random UV-C mutagenesis of Scheffersomyces (formerly Pichia) stipitis NRRL Y-7124 to improve anaerobic growth on lignocellulosic sugars. J Ind Microbiol Biotechnol 39:163–173. https://doi.org/10.1007/s10295-011-1012-x
Ilmen M, Koivuranta K, Ruohonen L, Suominen P, Penttilä M (2007) Efficient production of L-lactic acid from xylose by Pichia stipitis. Appl Environ Microb 73:117–123. https://doi.org/10.1128/aem.01311-06
Ingledew WM (1999) Alcohol production by Saccharomyces cerevisiae: a yeast primer. In: Lyons TP, Kelsall DR (eds) The Alcohol Textbook, 3rd edn. Nottingham University Press, Nottingham, pp 49–87. https://doi.org/10.3390/beverages2040030
Ishchuk OP, Abbas CA, Sibirny AA (2010) Heterologous expression of Saccharomyces cerevisiae MPR1 gene confers tolerance to ethanol and L-azetidine-2-carboxylic acid in Hansenula polymorpha. J Ind Microbiol Biotechnol 37:213–218. https://doi.org/10.1007/s10295-009-0674-0
Ishchuk OP, Voronovsky AY, Abbas CA, Sibirny AA (2009) Construction of Hansenula polymorpha strains with improved thermotolerance. Biotechnol Bioeng 104:911–919. https://doi.org/10.1002/bit.22457
Ishchuk OP, Voronovsky AY, Stasyk OV, Gayda GZ, Gonchar MV, Abbas CA, Sibirny AA (2008) Overexpression of pyruvate decarboxylase in the yeast Hansenula polymorpha results in increased ethanol yield in high-temperature fermentation of xylose. FEMS Yeast Res 8:1164–1174. https://doi.org/10.1111/j.1567-1364.2008.00429.x
Jansen MLA, Bracher JM, Papapetridis I, Verhoeven MD, de Bruijn H, de Waal PP, van Maris AJA, Klaassen P, Pronk JT (2017) Saccharomyces cerevisiae strains for second-generation ethanol production: from academic exploration to industrial implementation. FEMS Yeast Res 17:fox044. https://doi.org/10.1093/femsyr/fox044
Jeffries TW, Grigoriev IV, Grimwood J, Laplaza JM, Aerts A, Salamov A, Schmutz J, Lindquist E, Dehal P, Shapiro H, Jin YS, Passoth V, Richardson PM (2007) Genome sequence of the lignocellulose-bioconverting and xylose-fermenting yeast Pichia stipitis. Nat Biotechnol 25:319–326. https://doi.org/10.1038/nbt1290
Jeffries TW, Van Vleet JR (2009) Pichia stipitis genomics, transcriptomics, and gene clusters. FEMS Yeast Res 9:793–807. https://doi.org/10.1111/j.1567-1364.2009.00525.x
Jensen P, Snoep J, Westerhoff H (2006) Method of improving the production of biomass or a desired product from a cell. US Patent 20060094078: A1
Jeppsson M, Johansson B, Hahn-Hägerdal B, Gorwa-Grauslund MF (2002) Reduced oxidative pentose phosphate pathway flux in recombinant xylose-utilizing Saccharomyces cerevisiae strains improves the ethanol yield from xylose. Appl Environ Microbiol 68:1604–1609. https://doi.org/10.1128/aem.68.4.1604-1609.2002
Jin YS, Jeffries TW (2003) Changing flux of xylose metabolites by altering expression of xylose reductase and xylitol dehydrogenase in recombinant Saccharomyces cerevisiae. Appl Biochem Biotechnol 105–108:277–286
Jin YS, Jones S, Shi NQ, Jeffries TW (2002) Molecular cloning of XYL3 (D-xylulokinase) from Pichia stipitis and characterization of its physiological function. Appl Environ Microb 68:1232–1239. https://doi.org/10.1128/aem.68.3.1232-1239.2002
Jin YS, Ni H, Laplaza JM, Jeffries TW (2003) Optimal growth and ethanol production from xylose by recombinant Saccharomyces cerevisiae require moderate D-xylulokinase activity. Appl Environ Microbiol 69:495–503. https://doi.org/10.1128/aem.69.1.495-503.2003
Jingping G, Hongbing S, Gang S, Hongzhi L, Wenxiang P (2012) A genome shuffling-generated Saccharomyces cerevisiae isolate that ferments xylose and glucose to produce high levels of ethanol. J Ind Microbiol Biotechnol 39:777–787. https://doi.org/10.1007/s10295-011-1076-7
Kaliterna J, Weusthuis RA, Castrillo JI, van Dijken JP, Pronk JT (1995) Coordination of sucrose uptake and respiration in the yeast Debaryomyces yamadae. Microbiology 141:1567–1574. https://doi.org/10.1099/13500872-141-7-1567
Karhumaa K, Garcia Sanchez R, Hahn-Hägerdal B, Gorwa-Grauslund MF (2007) Comparison of the xylose reductase-xylitol dehydrogenase and the xylose isomerase pathways for xylose fermentation by recombinant Saccharomyces cerevisiae. Microb Cell Fact 6:5. https://doi.org/10.1186/1475-2859-6-5
Karhumaa K, Hahn-Hägerdal B, Gorwa-Grauslund MF (2005) Investigation of limiting metabolic steps in the utilization of xylose by recombinant Saccharomyces cerevisiae using metabolic engineering. Yeast 22:359–368. https://doi.org/10.1002/yea.1216
Keating JD, Robinson J, Cotta MA, Saddler JN, Mansfield SD (2004) An ethanologenic yeast exhibiting unusual metabolism in the fermentation of lignocellulosic hexose sugars. J Ind Microbiol Biotechnol 31:235–244. https://doi.org/10.1007/s10295-004-0145-6
Khoury GA, Fazelinia H, Chin JW, Pantazes RJ, Cirino PC, Maranas CD (2009) Computational design of Candida boidinii xylose reductase for altered cofactor. Protein Sci 18:2125–2138. https://doi.org/10.1002/pro.227
Kilian SG, van Uden N (1988) Transport of xylose and glucose in the xylose fermenting yeast Pichia stipitis. Appl Microb Biotechnol 27:545–548. https://doi.org/10.1007/bf00451629
Kim MS, Chung YS, Seo JH, Jo DH, Park YH, Ryu YW (2001) High-yield production of xylitol from xylose by a xylitol dehydrogenase defective mutant of Pichia stipitis. J Microbiol Biotechn 11:564–569
Kim OC, Suwannarangsee S, Oh DB, Kim S, Seo JW, Kim CH, Kang HA, Kim JY, Kwon O (2013) Transcriptome analysis of xylose metabolism in the thermotolerant methylotrophic yeast Hansenula polymorpha. Bioprocess Biosyst Eng 36:1509–1518. https://doi.org/10.1007/s00449-013-0909-3
Kim SK, Jin YS, Choi IG, Park YC, Seo JH (2015) Enhanced tolerance of Saccharomyces cerevisiae to multiple lignocellulose-derived inhibitors through modulation of spermidine contents. Metab Eng 29:46–55. https://doi.org/10.1016/j.ymben.2015.02.004
Kim SR, Skerker JM, Kang W, Lesmana A, Wei N, Arkin AP, Jin YS (2013) Rational and evolutionary engineering approaches uncover a small set of genetic changes efficient for rapid xylose fermentation in Saccharomyces cerevisiae. PLoS One 8:e57048. https://doi.org/10.1371/journal.pone.0057048
Kim SR, Xu H, Lesmana A, Kuzmanovic U, Au M, Florencia C, Oh EJ, Zhang G, Kim KH, Jin YS (2015) Deletion of PHO13, encoding haloacid dehalogenase type IIa phosphatase, results in upregulation of the pentose phosphate pathway in Saccharomyces cerevisiae. Appl Environ Microbiol 81:1601–1609. https://doi.org/10.1128/aem.03474-14
Klinner U, Fluthgraf S, Freese S, Passoth V (2005) Aerobic induction of respiro-fermentative growth by decreasing oxygen tensions in the respiratory yeast Pichia stipitis. Appl Microbiol Biot 67:247–253. https://doi.org/10.1007/s00253-004-1746-8
Kobayashi Y, Sahara T, Ohgiya S, Kamagata Y, Fujimori KE (2018) Systematic optimization of gene expression of pentose phosphate pathway enhances ethanolproduction from a glucose/xylose mixed medium in a recombinant Saccharomyces cerevisiae. AMB Express 8:139. https://doi.org/10.1186/s13568-018-0670-8
Krahulec S, Klimacek M, Nidetzky B (2012) Analysis and prediction of the physiological effects of altered coenzyme specificity in xylose reductase and xylitol dehydrogenase during xylose fermentation by Saccharomyces cerevisiae. J Biotechnol 158:192–202. https://doi.org/10.1016/j.jbiotec.2011.08.026
Križanović S, Butorac A, Mrvčić J, Krpan M, Cindrić M, Bačun-Družina V, Stanzer D (2015) Characterization of a S-adenosyl-l-methionine (SAM)-accumulating strain of Scheffersomyces stipitis. Int Microbiol 18:117–125. https://doi.org/10.2436/20.1501.01.241
Kurtzman CP (1990) Candida shehatae—genetic diversity and phylogenetic relationships with other xylose-fermenting yeasts. Antonie Van Leeuwenhoek 57:215–222. https://doi.org/10.1007/bf00400153
Kurtzman CP, Suzuki M (2010) Phylogenetic analysis of ascomycete yeasts that form coenzyme Q-9 and the proposal of the new genera Babjeviella, Meyerozyma, Millerozyma, Priceomyces, and Scheffersomyce. Mycoscience 51:2–14. https://doi.org/10.1007/s10267-009-0011-5
Kurylenko O, Ruchala J, Dmytruk K, Sibirny A (2015) New targets for improvement of xylose alcoholic fermentation in the methylotrophic yeast Hansenula polymorpha. In: Sibirny A, Fedorovyvh D, Gonchar M, Grabek-Lejko D (eds) Living organisms and bioanalytical approaches for detoxification and monitoring of toxic compounds. University of Rzeszow, Rzeszow, pp 247–257
Kurylenko O, Semkiv M, Ruchala J, Kshanovska B, Abbas C, Dmytruk K, Sibirny A (2016) New approaches for improving the production of the 1st and 2nd generation ethanol by yeast. Acta Biochim Polon 63:1156. https://doi.org/10.18388/abp.2015_1156
Kurylenko OO, Ruchala J, Hryniv OB, Abbas CA, Dmytruk KV, Sibirny AA (2014) Metabolic engineering and classical selection of the methylotrophic thermotolerant yeast Hansenula polymorpha for improvement of high-temperature xylose alcoholic fermentation. Microb Cell Fact 13:122. https://doi.org/10.1186/s12934-014-0122-3
Kurylenko OO, Ruchala J, Vasylyshyn RV, Stasyk OV, Dmytruk OV, Dmytruk KV, Sibirny AA (2018) Peroxisomes and peroxisomal transketolase and transaldolase enzymes are essential for xylose alcoholic fermentation by the methylotrophic thermotolerant yeast, Ogataea (Hansenula) polymorpha. Biotechnol Biofuels 11:197. https://doi.org/10.1186/s13068-018-1203-z
Kurylenko O, Vasylyshyn R, Kata I, Ruchala J, Semkiv M, Dmytruk K, Sibirny A (2016) Improvement of high-temperature xylose and glucose alcoholic fermentation in methylotrophic yeast Ogataea (Hansenula) polymorpha by overexpression of PDC1 and ADH1 genes. In Visnyk Lviv Univ 73:147–153
Kwak S, Jin YS (2017) Production of fuels and chemicals from xylose by engineered Saccharomyces cerevisiae: a review and perspective. Microb Cell Fact 16:82. https://doi.org/10.1186/s12934-017-0694-9
Lancashire WE, Dickinson JR, Malloch RA (1998) DNA encoding enzymes of the glycolytic pathway for use in alcohol producing yeast. US Patent 5786186 A
Laplaza JM, Torres BR, Jin YS, Jeffries TW (2006) Sh ble and Cre adapted for functional genomics and metabolic engineering of Pichia stipitis. Enzyme Microb Tech 38:741–747. https://doi.org/10.1016/j.enzmictec.2005.07.024
Larsson S, Nilvebrant NO, Jonsson LJ (2001) Effect of overexpression of Saccharomyces cerevisiae Pad1p on the resistance to phenylacrylic acids and lignocellulose hydrolysates under aerobic and oxygen limited conditions. Appl Microbiol Biotechnol 57:167–174. https://doi.org/10.1007/s002530100742
Lee H, James AP, Zahab DM, Mahmourides G, Maleszka R, Schneider H (1986) Mutants of Pachysolen tannophilus with improved production of ethanol d-xylose. Appl Environ Microbiol 51:1252–1258
Lee SH, Kodaki T, Park YC, Seo JH (2012) Effects of NADH-preferring xylose reductase expression on ethanol production from xylose in xylose-metabolizing recombinant Saccharomyces cerevisiae. J Biotechnol 158:184–191. https://doi.org/10.1016/j.jbiotec.2011.06.005
Lennartsson PR, Erlandsson P, Taherzadeh MJ (2014) Integration of the first and second generation bioethanol processes and the importance of by-products. Bioresour Technol 165:3–8. https://doi.org/10.1016/j.biortech.2014.01.127
Li H, Shen Y, Wu M, Hou J, Jiao C, Li Z, Liu X, Bao X (2016) Engineering a wild-type diploid Saccharomyces cerevisiae strain for second-generation bioethanol production. Bioresour Bioprocess 3:51. https://doi.org/10.1186/s40643-016-0126-4
Ligthelm ME, Prior JC, du Preez JC, Brandt V (1988) An investigation of D-xylose metabolism in Pichia stipitis under aerobic and anaerobic conditions. Appl Microb Biotechnol 28:293–296. https://doi.org/10.1007/bf00250458
Lindley ND, Waites MJ, Quayle JR (1981) Simultaneous assay of dihydroxyacetone synthase and transketolase in a methylotrophic yeast grown in continuous culture. A cautionary note. J Gen Microbiol 126:253–259. https://doi.org/10.1099/00221287-126-2-253
Liu ZL, Moon J, Andersh BJ, Slininger PJ, Weber S (2008) Multiple gene-mediated NAD(P)H-dependent aldehyde reduction is a mechanism of in situ detoxification of furfural and 5-hydroxymethylfurfural by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 81:743–753. https://doi.org/10.1007/s00253-008-1702-0
Long TM, Su YK, Headman J, Higbee A, Willis LB, Jeffries TW (2012) Cofermentation of glucose, xylose and cellobiose by the beetle-associated yeast Spathaspora passalidarum. Appl Environ Microbiol 78:5492–5500. https://doi.org/10.1128/aem.00374-12
Lu P, Davis BP, Hendrick J, Jeffries TW (1998) Cloning and disruption of the beta-isopropylmalate dehydrogenase gene (LEU2) of Pichia stipitis with URA3 and recovery of the double auxotroph. Appl Microbiol Biot 49:141–146. https://doi.org/10.1007/s002530051150
Maassen N, Freese S, Schruff B, Passoth V, Klinner U (2008) Nonhomologous end joining and homologous recombination DNA repair pathways in integration mutagenesis in the xylose-fermenting yeast Pichia stipitis. FEMS Yeast Res 8:735–743. https://doi.org/10.1111/j.1567-1364.2008.00383.x
Madhavan A, Tamalampudi S, Ushida K, Kanai D, Katahira S, Srivastava A, Fukuda H, Bisaria VS, Kondo A (2009) Xylose isomerase from polycentric fungus Orpinomyces: gene sequencing, cloning, and expression in Saccharomyces cerevisiae for bioconversion of xylose to ethanol. Appl Microbiol Biotechnol 82:1067–1078. https://doi.org/10.1007/s00253-008-1794-6
Madhavan A, Srivastava A, Kondo A, Bisaria VS (2012) Bioconversion of lignocellulose-derived sugars to ethanol by engineered Saccharomyces cerevisiae. Crit Rev Biotechnol 32:22–48. https://doi.org/10.3109/07388551.2010.539551
Mahler G, Nudel C (2000) Effect of magnesium ions on the fermentative and respirative functions in Pichia stipitis under oxygen-restricted growth. Microbiol Res 155:31–35. https://doi.org/10.1016/s0944-5013(00)80019-1
Matsushika A, Sawayama S (2008) Efficient bioethanol production from xylose by recombinant Saccharomyces cerevisiae requires high activity of xylose reductase and moderate xylulokinase activity. J Biosci Bioeng 106:306–309. https://doi.org/10.1263/jbb.106.306
Melake T, Passoth V, Klinner U (1996) Characterization of the genetic system of the xylose-fermenting yeast Pichia stipitis. Curr Microbiol 33:237–242. https://doi.org/10.1007/s002849900106
Metzger MH, Hollenberg CP (1995) Amino acid substitutions in the yeast Pichia stipitis xylitol dehydrogenase coenzyme-binding domain affect the coenzyme specificity. Eur J Biochem 228:50–54. https://doi.org/10.1111/j.1432-1033.1995.0050o.x
Mohd Azhar S, Abdulla R, Jambo SA, Marbawi H, Gansau JA, Mohd Faik AA, Rodrigues KF (2017) Yeasts in sustainable bioethanol production. A review. Biochem Biophys Rep 6:52–61. https://doi.org/10.1016/j.bbrep.2017.03.003
Moon J, Liu ZL (2012) Engineered NADH-dependent GRE2 from Saccharomyces cerevisiae by directed enzyme evolution enhances HMF reduction using additional cofactor NADPH. Enzyme Microb Technol 50:115–120. https://doi.org/10.1016/j.enzmictec.2011.10.007
Nardi JB, Bee CM, Miller LA, Nguyen NH, Suh SO, Blackwell M (2006) Communities of microbes that inhabit the changing hindgut landscape of a subsocial beetle. Arthropod Struct Dev 35:57–68. https://doi.org/10.1016/j.asd.2005.06.003
Navas MA, Cerdan S, Gancedo JM (1993) Futile cycles in Saccharomyces cerevisiae strains expressing the gluconeogenic enzymes during growth on glucose. Prot Natl Acad Sci USA 90:1290–1294. https://doi.org/10.1073/pnas.90.4.1290
Navas MA, Gancedo JM (1996) The regulatory characteristics of yeast fructose-1,6-bisphosphatase confer only a small selective advantage. J Bacteriol 178:1809–1812. https://doi.org/10.1128/jb.178.7.1809-1812.1996
Nguyen NH, Suh SO, Marshall CJ, Blackwell M (2006) Morphological and ecological similarities: wood-boring beetles associated with novel xylose-fermenting yeasts, Spathaspora passalidarum gen. sp. nov. and Candida jeffriesii sp. nov. Mycol Res 110:1232–1241. https://doi.org/10.1016/j.mycres.2006.07.002
Ni H, Laplaza JM, Jeffries TW (2007) Transposon mutagenesis to improve the growth of recombinant Saccharomyces cerevisiae on D-xylose. Appl Environ Microbiol 73:2061–2066. https://doi.org/10.1128/aem.02564-06
Nigam JN (2001) Development of xylose-fermenting yeast Pichia stipitis for ethanol production through adaptation on hardwood hemicellulose acid prehydrolysate. J Appl Microbiol 90:208–215. https://doi.org/10.1046/j.1365-2672.2001.01234.x
Nigam PS, Singh A (2011) Production of liquid biofuels from renewable resources. Prog Energy Combust Sci 37:52–68. https://doi.org/10.1016/j.pecs.2010.01.003
Nijland JG, Shin HY, de Jong RM, de Waal PP, Klaassen P, Driessen AJ (2014) Engineering of an endogenous hexose transporter into a specific d-xylose transporter facilitates glucose-xylose co-consumption in Saccharomyces cerevisiae. Biotechnol Biofuels 7:168. https://doi.org/10.1186/s13068-014-0168-9
Nijland JG, Vos E, Shin HY, de Waal PP, Klaassen P, Driessen AJ (2016) Improving pentose fermentation by preventing ubiquitination of hexose transporters in Saccharomyces cerevisiae. Biotechnol Biofuels 9:158. https://doi.org/10.1186/s13068-016-0573-3
Nissen TL, Kielland-Brandt MC, Nielsen J, Villadsen J (2000) Optimization of ethanol production in Saccharomyces cerevisiae by metabolic engineering of the ammonium assimilation. Metab Eng 2:69–77. https://doi.org/10.1006/mben.1999.0140
Oh YK, Hwang KR, Kim C, Kim JR, Lee JS (2018) Recent developments and key barriers to advanced biofuels: a short review. Bioresour Technol 257:320–333. https://doi.org/10.1016/j.biortech.2018.02.089
Ostergaard S, Walloe KO, Gomes SG, Olsson L, Nielsen J (2001) The impact of GAL6, GAL80, and MIG1 on glucose control of the GAL system in Saccharomyces cerevisiae. FEMS Yeast Res 1:47–55. https://doi.org/10.1111/j.1567-1364.2001.tb00012.x
Palmqvist E, Hahn-Hägerdal B (2001) Fermentation of lignocellulosic hydrolysates. I: inhibition and detoxification. Bioresour Technol 74:17–24. https://doi.org/10.1016/s0960-8524(99)00160-1
Papapetridis I, Verhoeven MD, Wiersma SJ, Goudriaan M, van Maris AJA, Pronk JT (2018) Laboratory evolution for forced glucose-xylose co-consumption enables identification of mutations that improve mixed-sugar fermentation by xylose-fermenting Saccharomyces cerevisiae. FEMS Yeast Res 1:18. https://doi.org/10.1093/femsyr/foy056
Parachin NS, Bergdahl B, van Niel EWJ, Gorwa-Grauslund MF (2011) Kinetic modelling reveals current limitations in the production of ethanol from xylose by recombinant Saccharomyces cerevisiae. Metab Eng 13:508–517. https://doi.org/10.1016/j.ymben.2011.05.005
Parreiras LS, Breuer RJ, Avanasi Narasimhan R, Higbee AJ, La Reau A, Tremaine M, Qin L, Willis LB, Bice BD, Bonfert BL, Pinhancos RC, Balloon AJ, Uppugundla N, Liu T, Li C, Tanjore D, Ong IM, Li H, Pohlmann EL, Serate J, Withers ST, Simmons BA, Hodge DB, Westphall MS, Coon JJ, Dale BE, Balan V, Keating DH, Zhang Y, Landick R, Gasch AP, Sato TK (2014) Engineering and two-stage evolution of a lignocellulosic hydrolysate-tolerant Saccharomyces cerevisiae strain for anaerobic fermentation of xylose from AFEX pretreated corn stover. PLoS One 9:e107499. https://doi.org/10.1371/journal.pone.0107499
Passoth V, Cohn M, Schafer B, Hahn-Hägerdal B, Klinner U (2003) Analysis of the hypoxia-induced ADH2 promoter of the respiratory yeast Pichia stipitis reveals a new mechanism for sensing of oxygen limitation in yeast. Yeast 20:39–51. https://doi.org/10.1002/yea.933
Passoth V, Hansen M, Klinner U, Emeis CC (1992) The electrophoretic banding patterns of the chromosomes of Pichia stipitis and Candida shehatae. Curr Genet 22:429–431. https://doi.org/10.1007/bf00352445
Passoth V, Schafer B, Liebel B, Weierstall T, Klinner U (1998) Molecular cloning of alcohol dehydrogenase genes of the yeast Pichia stipitis and identification of the fermentative ADH. Yeast 14:1311–1325. https://doi.org/10.1002/(sici)1097-0061(1998100)14:14%3c1311:aid-yea315%3e3.0.co;2-t
Passoth V, Zimmermann M, Klinner U (1996) Peculiarities of the regulation of fermentation and respiration in the crabtree-negative, xylose-fermenting yeast Pichia stipitis. Appl Biochem Biotechnol 57(58):201–212. https://doi.org/10.1007/978-1-4612-0223-3_18
Pereira SR, Nogué SI, Frazão CJ, Serafim LS, Gorwa-Grauslund MF, Xavier AM (2015) Adaptation of Scheffersomyces stipitis to hardwood spent sulfite liquor by evolutionary engineering. Biotechnol Biofuels 8:50. https://doi.org/10.1186/s13068-015-0234-y
Petersson A, Almeida JR, Modig T, Karhumaa K, Hahn-Hägerdal B, Gorwa-Grauslund MF, Lidén G (2006) A 5-hydroxymethyl furfural reducing enzyme encoded by the Saccharomyces cerevisiae ADH6 gene conveys HMF tolerance. Yeast 23:455–464. https://doi.org/10.1002/yea.1370
Petschacher B, Nidetzky B (2008) Altering the coenzyme preference of xylose reductase to favor utilization of NADH enhances ethanol yield from xylose in a metabolically engineered strain of Saccharomyces cerevisiae. Microb Cell Fact 7:9. https://doi.org/10.1186/1475-2859-7-9
Piontek M, Hagedorn J, Hollenberg CP, Gellissen G, Strasser AW (1998) Two novel gene expression systems based on the yeasts Schwanniomyces occidentalis and Pichia stipitis. Appl Microbiol Biot 50:331–338. https://doi.org/10.1007/s002530051300
Qi K, Zhong JJ, Xia XX (2014) Triggering respirofermentative metabolism in the Crabtree-negative yeast Pichia guilliermondii by disrupting the CAT8 gene. Appl Environ Microbiol 80:3879–3887. https://doi.org/10.1128/aem.00854-14
Quarterman J, Skerker JM, Feng X, Liu IY, Zhao H, Arkin AP, Jin YS (2016) Rapid and efficient galactose fermentation by engineered Saccharomyces cerevisiae. J Biotechnol 229:13–21. https://doi.org/10.1016/j.jbiotec.2016.04.041
Radecka D, Mukherjee V, Mateo RQ, Stojiljkovic M, Foulquié-Moreno MR, Thevelein JM (2015) Looking beyond Saccharomyces: the potential of non-conventional yeast species for desirable traits in bioethanol fermentation. FEMS Yeast Res 15:fov053. https://doi.org/10.1093/femsyr/fov053
Ren X, Wang J, Yu H, Peng C, Hu J, Ruan Z, Zhao S, Liang Y, Peng N (2016) Anaerobic and sequential aerobic production of high-titer ethanol and single cell protein from NaOH-pretreated corn stover by a genome shuffling-modified Saccharomyces cerevisiae strain. Bioresour Technol 218:623–630. https://doi.org/10.1016/j.biortech.2016.06.118
Richard P, Londesborough J, Putkonen M, Kalkkinen N, Penttila M (2001) Cloning and expression of a fungal L-arabinitol-4-dehydrogenase gene. J Biol Chem 276(44):40631–40637. https://doi.org/10.1074/jbc.m104022200
Richard P, Putkonen M, Vaananen R, Londesborough J, Penttila M (2002) The missing link in the fungal L-arabinose catabolic pathway, identification of the L-xylulose reductase gene. Biochemistry 41:6432–6437. https://doi.org/10.1021/bi025529i
Riley R, Haridas S, Wolfe KH, Lopes MR, Hittinger CT, Göker M, Salamov AA, Wisecaver JH, Long TM, Calvey CH, Aerts AL, Barry KW, Choi C, Clum A, Coughlan AY, Deshpande S, Douglass AP, Hanson SJ, Klenk HP, LaButti KM, Lapidus A, Lindquist EA, Lipzen AM, Meier-Kolthoff JP, Ohm RA, Otillar RP, Pangilinan JL, Peng Y, Rokas A, Rosa CA, Scheuner C, Sibirny AA, Slot JC, Stielow JB, Sun H, Kurtzman CP, Blackwell M, Grigoriev IV, Jeffries TW (2016) Comparative genomics of biotechnologically important yeasts. Proc Natl Acad Sci USA 113:9882–9887. https://doi.org/10.1073/pnas.1603941113
Roca C, Nielsen J, Olsson L (2003) Metabolic engineering of ammonium assimilation in xylose-fermenting Saccharomyces cerevisiae improves ethanol production. Appl Environ Microbiol 69(8):4732–4736. https://doi.org/10.1128/aem.69.8.4732-4736.2003
Rodrigues R, Lu CF, Lin B, Jeffries TW (2008) Fermentation kinetics for xylitol production by a Pichia stipitis D-xylulokinase mutant previously grown in spent sulfite liquor. Appl Biochem Biotech. 148:199–209. https://doi.org/10.1007/s12010-007-8080-4
Rogers DT, Szostak JW (1993) Strains of yeast with increased rates of glycolysis. US Patent 5268285:A1
Ruchala J, Kurylenko OO, Soontorngun N, Dmytruk KV, Sibirny AA (2017) Transcriptional activator Cat8 is involved in regulation of xylose alcoholic fermentation in the thermotolerant yeast Ogataea (Hansenula) polymorpha. Microb Cell Fact 16:36. https://doi.org/10.1186/s12934-017-0652-6
Ryabova OB, Chmil OM, Sibirny AA (2003) Xylose and cellobiose fermentation to ethanol by the thermotolerant methylotrophic yeast Hansenula polymorpha. FEMS Yeast Res 4:157–164. https://doi.org/10.1016/s1567-1356(03)00146-6
Sánchez ÓJ, Cardona CA (2008) Trends in biotechnological production of fuel ethanol from different feedstocks. Biores Technol 99:5270–5295. https://doi.org/10.1016/j.biortech.2007.11.013
Schneider H, Wang PY, Chan YK, Kurtzman CP (1981) Conversion of d-xylose into ethanol by the yeast Pachysolen tannophilus. Biotechnol Lett 3:89–92. https://doi.org/10.1007/bf00145116
Semkiv MV, Dmytruk KV, Abbas CA, Sibirny AA (2014) Increased ethanol accumulation from glucose via reduction of ATP level in a recombinant strain of Saccharomyces cerevisiae overexpressing alkaline phosphatase. BMC Biotechnol 14:42. https://doi.org/10.1186/1472-6750-14-42
Semkiv MV, Dmytruk KV, Abbas CA, Sibirny AA (2016) Activation of futile cycles as an approach to increase ethanol yield during glucose fermentation in Saccharomyces cerevisiae. Bioengineered 7:106–111. https://doi.org/10.1080/21655979.2016.1148223
Shahsavarani H, Sugiyama M, Kaneko Y, Chuenchit B, Harashima S (2012) Superior thermotolerance of Saccharomyces cerevisiae for efficient bioethanol fermentation can be achieved by overexpression of RSP5 ubiquitin ligase. Biotechnol Adv 30:1289–1300. https://doi.org/10.1016/j.biotechadv.2011.09.002
Shi NQ, Davis B, Sherman F, Cruz J, Jeffries TW (1999) Disruption of the cytochrome c gene in xylose-utilizing yeast Pichia stipitis leads to higher ethanol production. Yeast 15:1021–1030. https://doi.org/10.1002/(sici)1097-0061(199908)15:11%3c1021:aid-yea429%3e3.0.co;2-v
Shi NQ, Jeffries TW (1998) Anaerobic growth and improved fermentation of Pichia stipitis bearing a URA1 gene from Saccharomyces cerevisiae. Appl Microbiol Biotechnol 50:339–345. https://doi.org/10.1007/s002530051301
Shin M, Kim JW, Ye S, Kim S, Jeong D, Lee DY, Kim JN, Jin YS, Kim KH, Kim SR (2019) Comparative global metabolite profiling of xylose-fermenting Saccharomyces cerevisiae SR8 and Scheffersomyces stipitis. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-019-09829-5
Skoog K, Hahn-Hagerdahl B (1990) Effect of oxygenation on xylose fermentation by Pichia stipitis. Appl Environ Microbiol 56:3389–3394
Slininger PJ, Dien BS, Gorsick SW, Liu ZL (2006) Nitrogen source and mineral optimization enhances D-xylose conversion to ethanol by the yeast Pichia stipitis NRRL Y-7124. Appl Microbial Cell Physiol 72:1285–1296. https://doi.org/10.1007/s00253-006-0435-1
Sprenger GA (1996) Carbohydrate metabolism in Zymomonas mobilis: a catabolic highway with some scenic routes. FEMS Microbiol Lett 145:301–307. https://doi.org/10.1111/j.1574-6968.1996.tb08593.x
Sreenath HK, Jeffries TW (1997) Diminished respirative growth and enhanced assimilative sugar uptake result in higher specific fermentation rates by the mutant Pichia stipitis FPL-061. Appl Biochem Biotechnol 63–65:109–116. https://doi.org/10.1007/bf02920417
Sreenath HK, Jeffries TW (1999) 2-deoxyglucose as a selective agent for derepressed mutants of Pichia stipitis. Appl Biochem Biotechnol 77:211–222. https://doi.org/10.1385/abab:77:1-3:211
Stanley D, Fraser S, Chambers PJ, Rogers P, Stanley GA (2010) Generation and characterisation of stable ethanol-tolerant mutants of Saccharomyces cerevisiae. J Ind Microbiol Biot 37:139–149. https://doi.org/10.1007/s10295-009-0655-3
Suh SO, Marshall CJ, McHugh JV, Blackwell M (2003) Wood ingestion by passalid beetles in the presence of xylose-fermenting gut yeasts. Mol Ecol 12(11):3137–3145. https://doi.org/10.1046/j.1365-294x.2003.01973.x
Tao X, Zheng D, Liu T, Wang P, Zhao W, Zhu M, Jiang X, Zhao Y, Wu X (2012) A novel strategy to construct yeast Saccharomyces cerevisiae for very high gravity fermentation. PLoS One 7:e31235. https://doi.org/10.1371/journal.pone.0031235
Targonski Z (1992) Biotransformation of lignin-related aromatic-compounds by Pichia stipitis Pignal. Zentralbl Mikrobiol 147:244–249. https://doi.org/10.1111/j.1567-1364.2009.00525.x
Temer B, Dos Santos LV, Negri VA, Galhardo JP, Magalhães PHM, José J, Marschalk C, Corrêa TLR, Carazzolle MF, Pereira GAG (2017) Conversion of an inactive xylose isomerase into a functional enzyme by co-expression of GroEL-GroES chaperonins in Saccharomyces cerevisiae. BMC Biotechnol 17:71. https://doi.org/10.1186/s12896-017-0389-7
Thomik T, Wittig I, Choe JY, Boles E, Oreb M (2017) An artificial transport metabolon facilitates improved substrate utilization in yeast. Nat Chem Biol 13:1158–1163. https://doi.org/10.1038/nchembio.2457
Valadi H, Larsson C, Gustafsson L (1998) Improved ethanol production by glycerol-3-phosphate dehydrogenase mutants of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 50(4):434–439. https://doi.org/10.1007/s002530051317
van der Klei IJ, Yurimoto H, Sakai Y, Veenhuis M (2006) The significance of peroxisomes in methanol metabolism in methylotrophic yeast. Biochim Biophys Acta 1763:1453–1462. https://doi.org/10.1016/j.bbamcr.2006.07.016
van Dijken JP, van den Bosch E, Hermans JJ, de Miranda LR, Scheffers WA (1986) Alcoholic fermentation by ‘nonfermentative’ yeasts. Yeast 2:123–127. https://doi.org/10.1002/yea.320020208
Verduyn C, Van Kleef R, Frank J, Schreuder H, van Dijken JP, Scheffers WA (1985) Properties of the NAD(P)H-dependent xylose reductase from the xylose-fermenting yeast Pichia stipitis. Biochem J 226:669–677. https://doi.org/10.1042/bj2260669
Verho R, Londesborough J, Penttilä M, Richard P (2003) Engineering redox cofactor regeneration for improved pentose fermentation in Saccharomyces cerevisiae. Appl Environ Microbiol 69:5892–5897. https://doi.org/10.1128/aem.69.10.5892-5897.2003
Voronovsky AY, Rohulya OV, Abbas CA, Sibirny AA (2009) Development of strains of the thermotolerant yeast Hansenula polymorpha capable of alcoholic fermentation of starch and xylan. Metab Eng 11:234–242. https://doi.org/10.1016/j.ymben.2009.04.001
Voronovsky AY, Ryabova OB, Verba OV, Ishchuk OP, Dmytruk KV, Sibirny AA (2005) Expression of xylA genes encoding xylose isomerases from Escherichia coli and Streptomyces coelicolor in the methylotrophic yeast Hansenula polymorpha. FEMS Yeast Res 5:1055–1062. https://doi.org/10.1016/j.femsyr.2005.09.001
Wahlbom CF, Cordero Otero RR, van Zyl WH, Hahn-Hägerdal B, Jönsson LJ (2003) Molecular analysis of a Saccharomyces cerevisiae mutant with improved ability to utilize xylose shows enhanced expression of proteins involved in transport, initial xylose metabolism, and the pentose phosphate pathway. Appl Environ Microbiol 69:740–746. https://doi.org/10.1128/aem.69.2.740-746.2003
Walfridsson M, Bao X, Anderlund M, Lilius G, Bülow L, Hahn-Hägerdal B (1996) Ethanolic fermentation of xylose with Saccharomyces cerevisiae harboring the Thermus thermophilus xylA gene, which expresses an active xylose (glucose) isomerase. Appl Environ Microbiol 62:4648–4651
Wang PM, Zheng DQ, Liu TZ, Tao XL, Feng MG, Min H, Jiang XH, Wu XC (2012) The combination of glycerol metabolic engineering and drug resistance marker-aided genome shuffling to improve very-high-gravity fermentation performances of industrial Saccharomyces cerevisiae. Bioresour Technol 108:203–210. https://doi.org/10.1016/j.biortech.2011.12.147
Wang TT, Lin LL, Hsu WH (1989) Cloning and expression of a Schwanniomyces occidentalis α-amylase gene in Saccharomyces cerevisiae. Appl Environ Microbiol 55:3167–3172. https://doi.org/10.1007/bf02882897
Watanabe D, Hashimoto N, Mizuno M, Zhou Y, Akao T, Shimoi H (2013) Accelerated alcoholic fermentation caused by defective gene expression related to glucose derepression in Saccharomyces cerevisiae. Biosci Biotechnol Biochem 77:2255–2262. https://doi.org/10.1271/bbb.130519
Watanabe S, Kodaki T, Makino K (2005) Complete reversal of coenzyme specificity of xylitol dehydrogenase and increase of thermostability by the introduction of structural zinc. J Biol Chem 280:10340–10349. https://doi.org/10.1074/jbc.m409443200
Watanabe T, Watanabe I, Yamamoto M, Ando A, Nakamura T (2011) A UV-induced mutant of Pichia stipitis with increased ethanol production from xylose and selection of a spontaneous mutant with increased ethanol tolerance. Bioresour Technol 102:1844–1848. https://doi.org/10.1016/j.biortech.2010.09.087
Wei L, Liu J, Qi H, Wen J (2015) Engineering Scheffersomyces stipitis for fumaric acid production from xylose. Bioresour Technol 187:246–254. https://doi.org/10.1016/j.biortech.2015.03.122
Weierstall T, Hollenberg CP, Boles E (1999) Cloning and characterization of three genes (SUT1-3) encoding glucose transporters of the yeast Pichia stipitis. Mol Microbiol 31:871–883. https://doi.org/10.1046/j.1365-2958.1999.01224.x
Weusthuis RA, Adams H, Scheffers WA, van Dijken JP (1993) Energetics and kinetics of maltose transport in Saccharomyces cerevisiae: a continuous culture study. Appl Environ Microbiol 59:3102–3109
Wijsman MR, Bruinenberg PM, van Diken JP, Scheffers WA (1985) Incapacity for anaerobic growth in xylose-fermenting yeasts. Antonie Van Leeuwenhoek 51:563–564. https://doi.org/10.1007/bf00404547
Wilson AJ, Bhattacharjee JK (1986) Regulation of phosphoenolpyruvate carboxykinase and pyruvate kinase in Saccharomyces cerevisiae grown in the presence of glycolytic and gluconeogenic carbon sources and the role of mitochondrial function on gluconeogenesis. Can J Microbiol 32:969–972. https://doi.org/10.1128/mcb.01055-07
Wisselink HW, Toirkens MJ, del Rosario Franco Berriel M, Winkler AA, van Dijken JP, Pronk JT, van Maris AJ (2007) Engineering of Saccharomyces cerevisiae for efficient anaerobic alcoholic fermentation of L-arabinose. Appl Environ Microbiol 73:4881–4891. https://doi.org/10.1111/j.1567-1364.2012.00808.x
Xia PF, Zhang GC, Walker B, Seo SO, Kwak S, Liu JJ, Kim H, Ort DR, Wang SG, Jin YS (2016) Recycling carbon dioxide during xylose fermentation by engineered Saccharomyces cerevisiae. ACS Synth Biol. https://doi.org/10.1021/acssynbio.6b00167
Yang VW, Marks JA, Davis BP, Jeffries TW (1994) High efficiency transformation of Pichia stipitis based on its URA3 gene and a homologous autonomous replication sequence, ARS2. Appl Environ Microb 60:4245–4254
Yang XS, Zhang SJ, Zuo Z, Men X, Tian S (2011) Ethanol production from the enzymatic hydrolysis of non-detoxified steamexploded corn stalk. Bioresour Technol 102:7840–7844. https://doi.org/10.1016/j.biortech.2011.05.048
Ye S, Kim JW, Kim SR (2019) Metabolic engineering for improved fermentation of L-arabinose. J Microbiol Biotechnol 29:339–346. https://doi.org/10.4014/jmb.1812.12015
Yoon GS, Lee TS, Kim C, Seo JH, Ryu YW (1996) Characterization of alcohol fermentation and segregation of protoplast fusant of Saccharomyces cerevisiae and Pichia stipitis. J Microbiol Biotechnol 6:286–291
Zhang GC, Liu JJ, Kong II, Kwak S, Jin YS (2015) Combining C6 and C5 sugar metabolism for enhancing microbial bioconversion. Curr Opin Chem Biol 29:49–57. https://doi.org/10.1016/j.cbpa.2015.09.008
Zhang GC, Liu JJ, Ding WT (2012) Decreased xylitol formation during xylose fermentation in Saccharomyces cerevisiae due to overexpression of water-forming NADH oxidase. Appl Environ Microbiol 78:1081–1086. https://doi.org/10.1128/aem.06635-11
Zhang J, Zhang B, Wang D, Gao X, Sun L, Hong J (2015) Rapid ethanol production at elevated temperatures by engineered thermotolerant Kluyveromyces marxianus via the NADP(H)-preferring xylose reductase-xylitol dehydrogenase pathway. Metab Eng 31:140–152. https://doi.org/10.1016/j.ymben.2015.07.008
Zhang L, Tang Y, Guo ZP, Ding ZY, Shi GY (2011) Improving the ethanol yield by reducing glycerol formation using cofactor regulation in Saccharomyces cerevisiae. Biotechnol Lett 33:1375–1380. https://doi.org/10.1007/s10529-011-0588-6
Zhang W, Geng AL (2012) Improved ethanol production by a xylose-fermenting recombinant yeast strain constructed through a modified genome shuffling method. Biotechnol Biofuels 5:46–57. https://doi.org/10.1186/1754-6834-5-46
Acknowledgements
This work was supported by grant of National Science Centre of Poland (NCN) Opus UMO-2016/21/B/NZ1/00280 awarded to A. Sibirny, National Academy of Sciences of Ukraine (Grants 2-19, 6-17 and 35-19). Authors are grateful to Prof. T. Jeffries (Madison, WI, USA) for careful reading the manuscript and useful ideas and suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ruchala, J., Kurylenko, O.O., Dmytruk, K.V. et al. Construction of advanced producers of first- and second-generation ethanol in Saccharomyces cerevisiae and selected species of non-conventional yeasts (Scheffersomyces stipitis, Ogataea polymorpha). J Ind Microbiol Biotechnol 47, 109–132 (2020). https://doi.org/10.1007/s10295-019-02242-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-019-02242-x